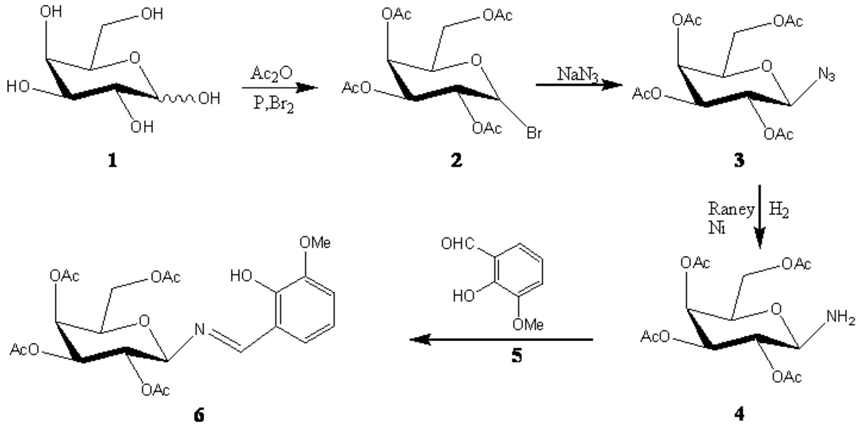
Synthesis of N-(2-hydroxy-3-methoxybenzylidene) - 2, 3, 4, 6-tetra-O-acetyl -β-D galactopyranosylamine as a new chiral Schiff base for asymmetric [2+2] cycloadditions
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgment
References
- Kunz, H.; Pfrengle, W. J. Am. Chem. Soc. 1988, 110, 651. [CrossRef]
- Kunz, H.; Pfrengle, W. Tetrahedron 1988, 44, 5487. [CrossRef]
- Sharma, G V M; Reddy, G.; Radha Krishna, P. Tetrahedron Lett. 1999, 40, 1783.
- Totani, K.; Takao, K.; Tadano, K. Synlett 2004, 12, 2066.
- Kunz, H. Modern Amination Methods; Ricci, A., Ed.; WILEY-VCH: einheim, 2000; p. 103. [Google Scholar]
- Danishefsky, S. J.; Aِِِِِِِllen, J. R. Angew. Chem. Int. Ed. Engl. 2000, 39, 836.
- Pili, R.; Chang, J.; Patris, R.A.; Mueller, R. A.; Chrest, F. J.; Passaniti, A. Cancer Res. 1995, 55, 2920.
- Wonge, C. H.; Sears, P. Angew. Chem. Int. Ed. Engl. 1999, 38, 2300.
- Dotz, K.; Jakel, C.; Hasse, W. J. Organomet. Chem. 2001, 617-618, 119.
- Babiano, R.; Fuentes Mota, J. Carbohydr. Res. 1986, 154, 280.
- Cusack, N. J.; Hildick, B. J.; Robinson, D. H.; Rugg, P. W.; Shaw, G. J. Chem.Soc. 1973, 1720.
- Cusack, N. J.; Robinson, D. H.; Rugg, P. W.; Shaw, G.; Lofthouse, R. J. Chem. Soc. 1974, 73.
- Barton, D. H. R.; Getau-Olesker, A.; Anaya-Mateos, J.; Cleophax, J.; Gero, S. D.; Chiaroni, A.; Riche, C. J. Chem. Soc., Perkin Trans.1 1990, 3211.
- Jarrahpour, A. A.; Shekarriz, M.; Taslimi, A. Molecules 2004, 9, 939. [CrossRef] [PubMed]
- Jarrahpour, A. A.; Shekarriz, M.; Taslimi, A. Molecules 2004, 9, 29. [CrossRef] [PubMed]
- Krank, B.; Hebrault, D.; Schultz, M.; Kunz, H. Synlett 2003, 4, 671.
- Kunz, H.; Sager, W.; Schanzenbach, D.; Decker, M. liebigs Ann. Chem. 1991, 7, 649.
- Furniss, B. S.; Hannaford, A. J.; Smith, P. W. G.; Tartchel, A.R. Textbook of Practical Organic Chemistry (Vogel’s), 5th ed.; John Wiley & Sons, Inc.: New York, 1991; p. 648. [Google Scholar]
- Sample Availability: Available from MDPI.
© 2005 MDPI. All rights reserved.
Share and Cite
Jarrahpour, A.A.; Alvand, P.; Arab, R.; Beheshti, A. Synthesis of N-(2-hydroxy-3-methoxybenzylidene) - 2, 3, 4, 6-tetra-O-acetyl -β-D galactopyranosylamine as a new chiral Schiff base for asymmetric [2+2] cycloadditions. Molbank 2005, 2005, M436. https://doi.org/10.3390/M436
Jarrahpour AA, Alvand P, Arab R, Beheshti A. Synthesis of N-(2-hydroxy-3-methoxybenzylidene) - 2, 3, 4, 6-tetra-O-acetyl -β-D galactopyranosylamine as a new chiral Schiff base for asymmetric [2+2] cycloadditions. Molbank. 2005; 2005(4):M436. https://doi.org/10.3390/M436
Chicago/Turabian StyleJarrahpour, A. A., P. Alvand, R. Arab, and A. Beheshti. 2005. "Synthesis of N-(2-hydroxy-3-methoxybenzylidene) - 2, 3, 4, 6-tetra-O-acetyl -β-D galactopyranosylamine as a new chiral Schiff base for asymmetric [2+2] cycloadditions" Molbank 2005, no. 4: M436. https://doi.org/10.3390/M436
APA StyleJarrahpour, A. A., Alvand, P., Arab, R., & Beheshti, A. (2005). Synthesis of N-(2-hydroxy-3-methoxybenzylidene) - 2, 3, 4, 6-tetra-O-acetyl -β-D galactopyranosylamine as a new chiral Schiff base for asymmetric [2+2] cycloadditions. Molbank, 2005(4), M436. https://doi.org/10.3390/M436



