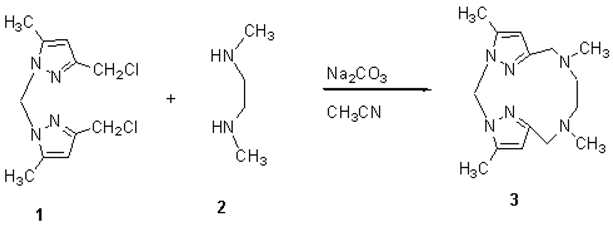
4,8,11,15-Tetramethyl-1,3,8,11,16,17-hexaazatricyclo[11.2.1.13,6]heptadeca-4,6(17), 13(16), 14-tetraene
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References
- El Kadiri, S. Ph. D. Thesis, U. S. T. L. Montpellier, 1988.
- Malek, F.; Persin, M.; Ramdani, A.; Sarrasin, J.; Zidane, I. New. J. Chem. 2002, 2, 26.
© 2003 MDPI. All rights reserved.
Share and Cite
Berhili, F.; Touzani, R.; Ramdani, A.; El Kadiri, S. 4,8,11,15-Tetramethyl-1,3,8,11,16,17-hexaazatricyclo[11.2.1.13,6]heptadeca-4,6(17), 13(16), 14-tetraene. Molbank 2003, 2003, M308. https://doi.org/10.3390/M308
Berhili F, Touzani R, Ramdani A, El Kadiri S. 4,8,11,15-Tetramethyl-1,3,8,11,16,17-hexaazatricyclo[11.2.1.13,6]heptadeca-4,6(17), 13(16), 14-tetraene. Molbank. 2003; 2003(1):M308. https://doi.org/10.3390/M308
Chicago/Turabian StyleBerhili, Farid, Rachid Touzani, Abdelkrim Ramdani, and Sghir El Kadiri. 2003. "4,8,11,15-Tetramethyl-1,3,8,11,16,17-hexaazatricyclo[11.2.1.13,6]heptadeca-4,6(17), 13(16), 14-tetraene" Molbank 2003, no. 1: M308. https://doi.org/10.3390/M308
APA StyleBerhili, F., Touzani, R., Ramdani, A., & El Kadiri, S. (2003). 4,8,11,15-Tetramethyl-1,3,8,11,16,17-hexaazatricyclo[11.2.1.13,6]heptadeca-4,6(17), 13(16), 14-tetraene. Molbank, 2003(1), M308. https://doi.org/10.3390/M308



