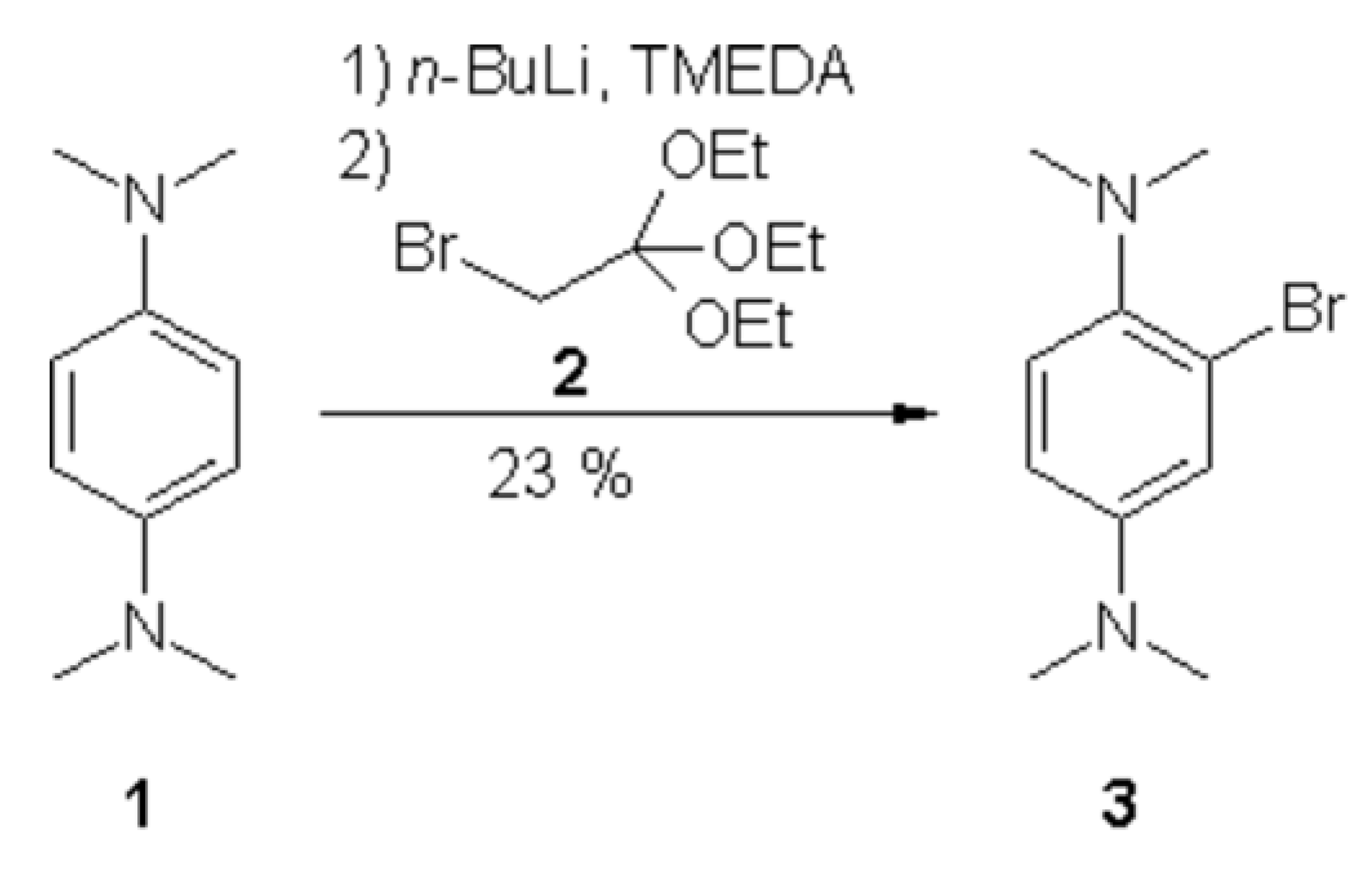
2-Bromo-N1, N1, N4, N4-tetramethyl-benzene-1,4-diamine
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References and Notes
- O’Toole, T. R. US Patent 1997. Cont.-in-part of U.S. Ser. No. 362,384.
- Sample availability: available form the authors.
© 2003 MDPI. All rights reserved.
Share and Cite
Zieg, H.; Pitsch, W.; Koenig, B. 2-Bromo-N1, N1, N4, N4-tetramethyl-benzene-1,4-diamine. Molbank 2002, 2002, M288. https://doi.org/10.3390/M288
Zieg H, Pitsch W, Koenig B. 2-Bromo-N1, N1, N4, N4-tetramethyl-benzene-1,4-diamine. Molbank. 2002; 2002(1):M288. https://doi.org/10.3390/M288
Chicago/Turabian StyleZieg, Harald, Wolfgang Pitsch, and Burkhard Koenig. 2002. "2-Bromo-N1, N1, N4, N4-tetramethyl-benzene-1,4-diamine" Molbank 2002, no. 1: M288. https://doi.org/10.3390/M288
APA StyleZieg, H., Pitsch, W., & Koenig, B. (2002). 2-Bromo-N1, N1, N4, N4-tetramethyl-benzene-1,4-diamine. Molbank, 2002(1), M288. https://doi.org/10.3390/M288



