Phosphorescent Organic Light-Emitting Devices: Working Principle and Iridium Based Emitter Materials
Abstract
1. Introduction
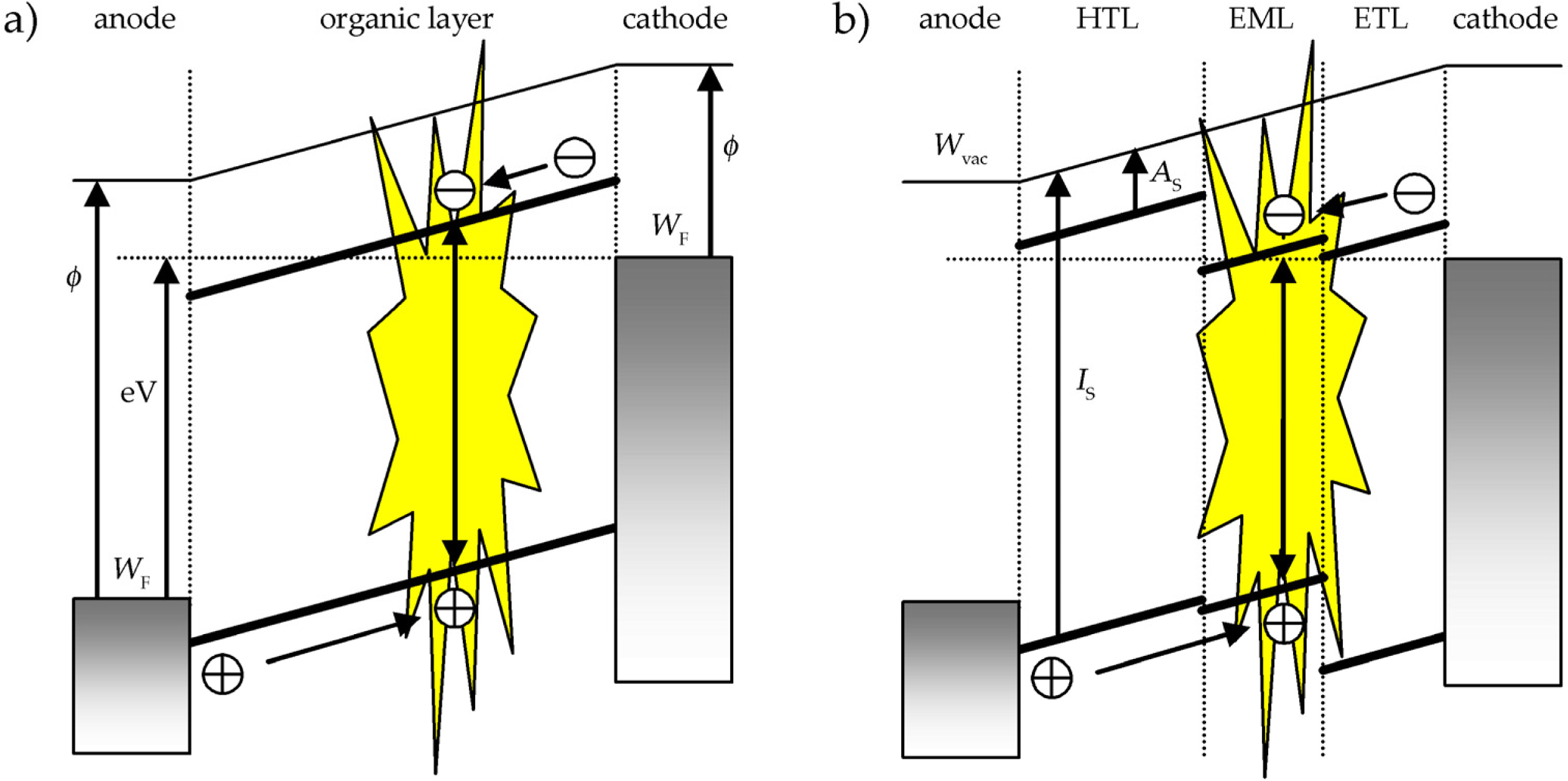
2. The working principle of OLEDs
2.1. Small molecule OLEDs (smOLEDs) and polymeric light-emitting devices (PLEDs)
2.2. Single and multilayer OLEDs
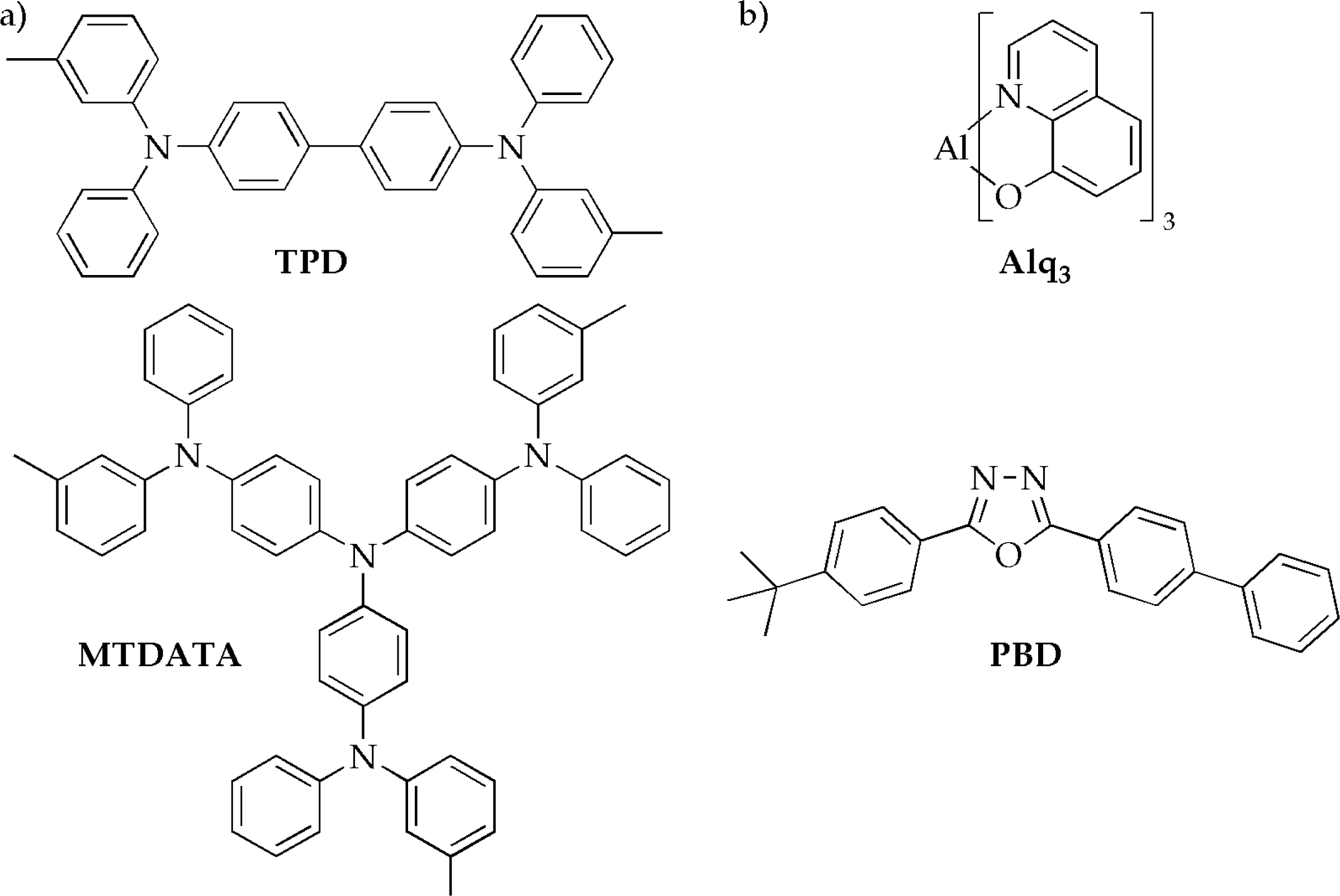
2.3. Fluorescent and phosphorescent materials in OLEDs
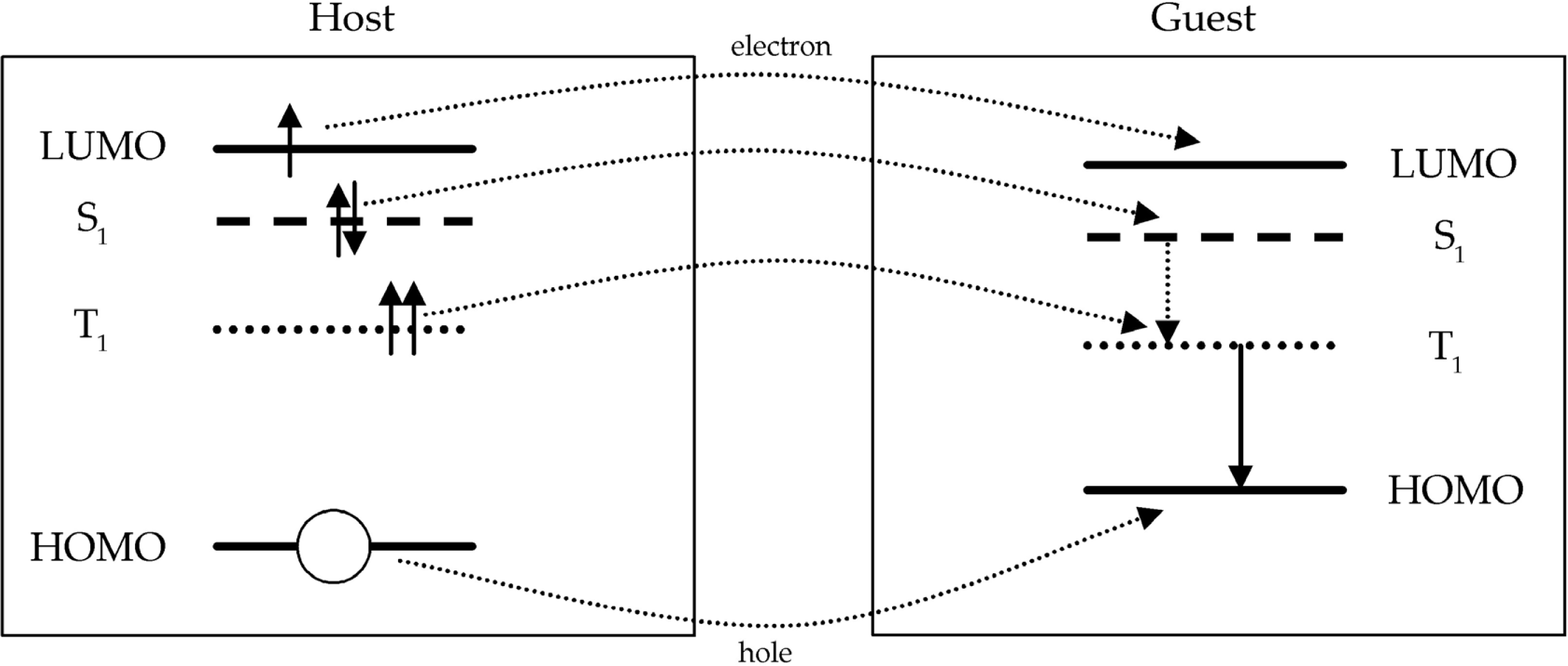
2.4. Transfer processes in host materials doped with phosphorescent dyes
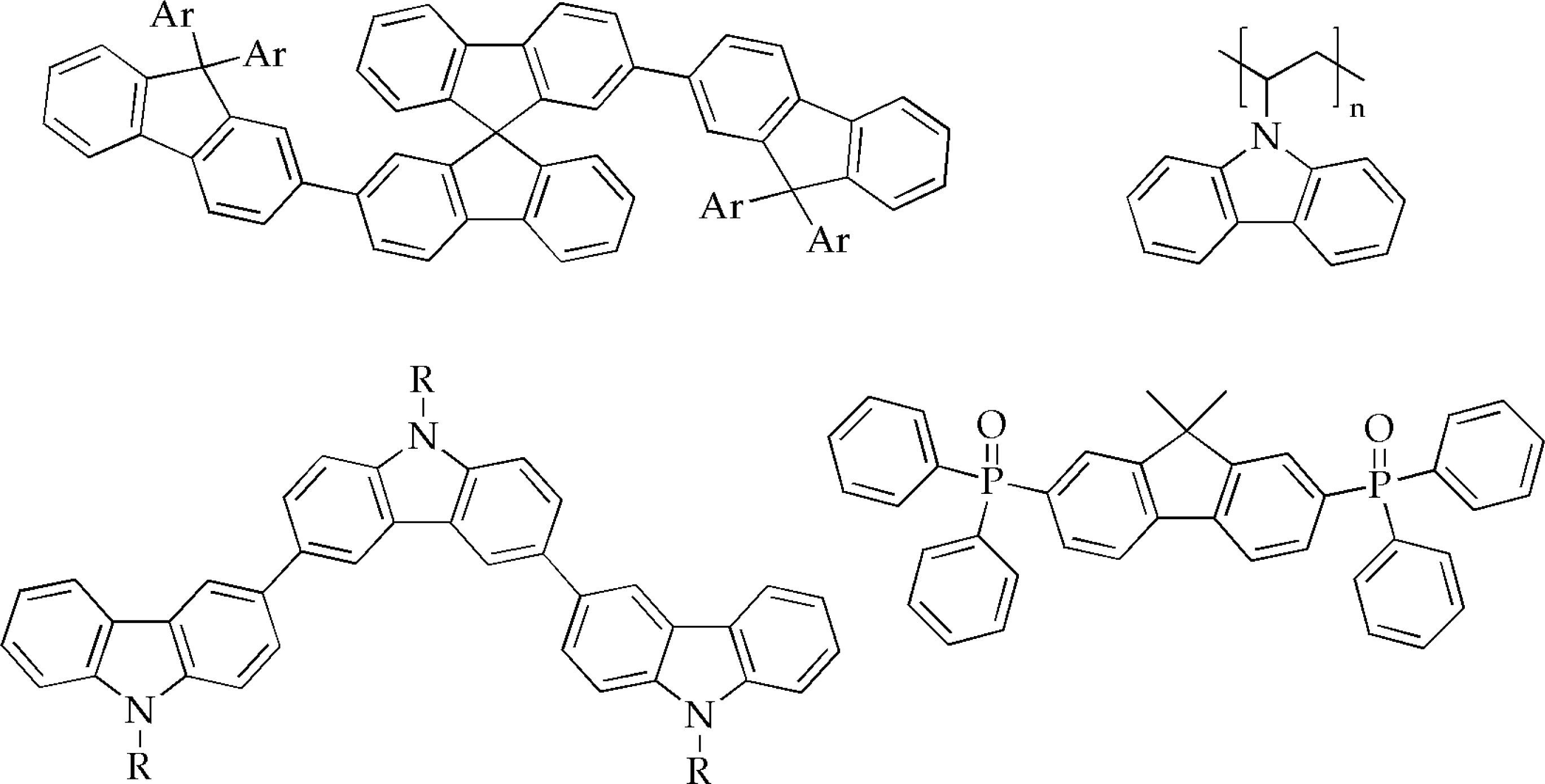
2.5. Host materials for phosphorescent OLEDs
2.6. Advantages of OLEDs in display technology
3. Phosphorescent iridium(III) complexes
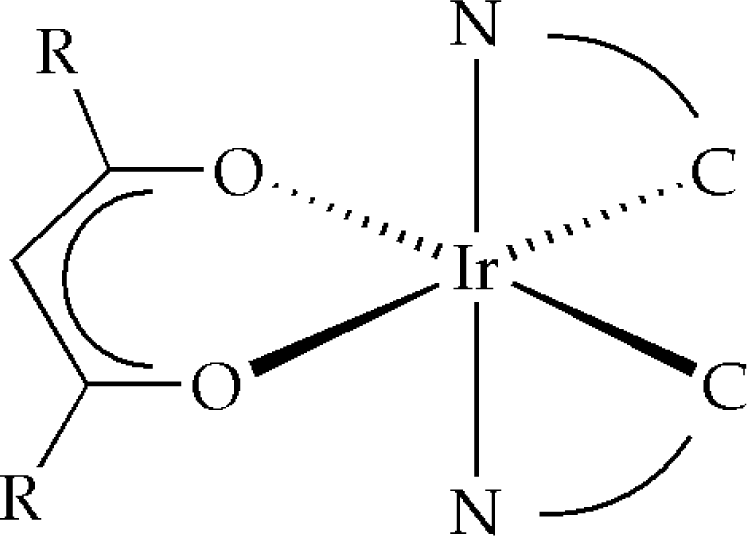
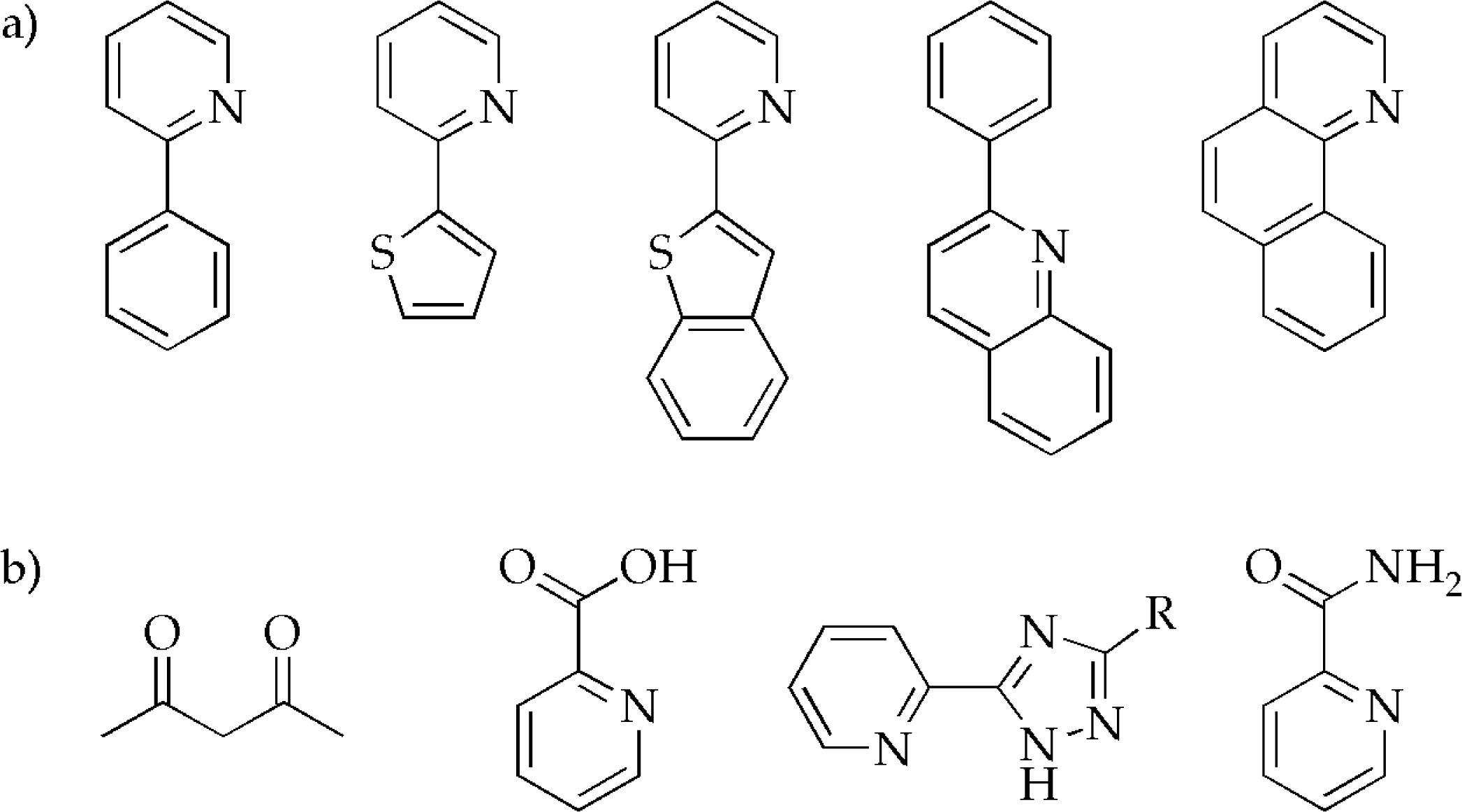
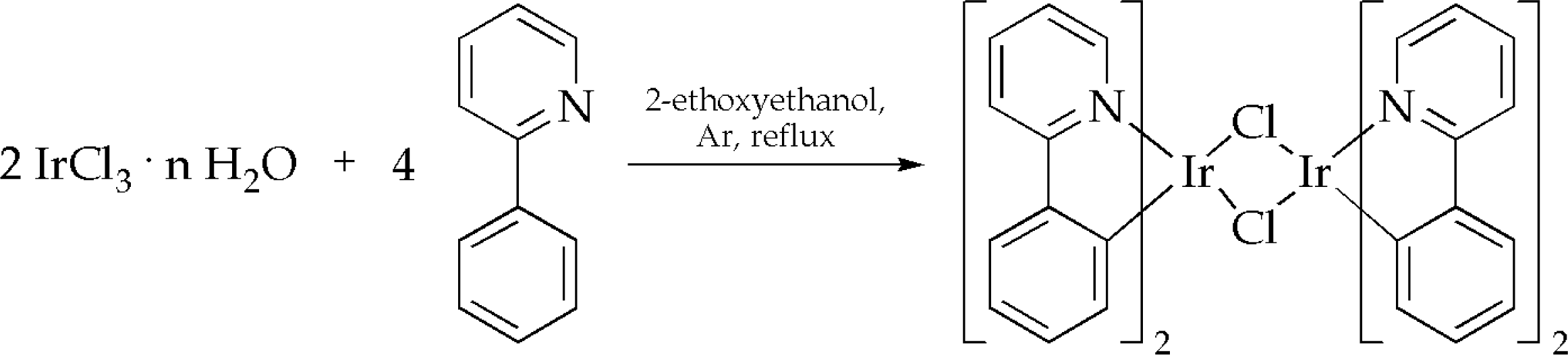
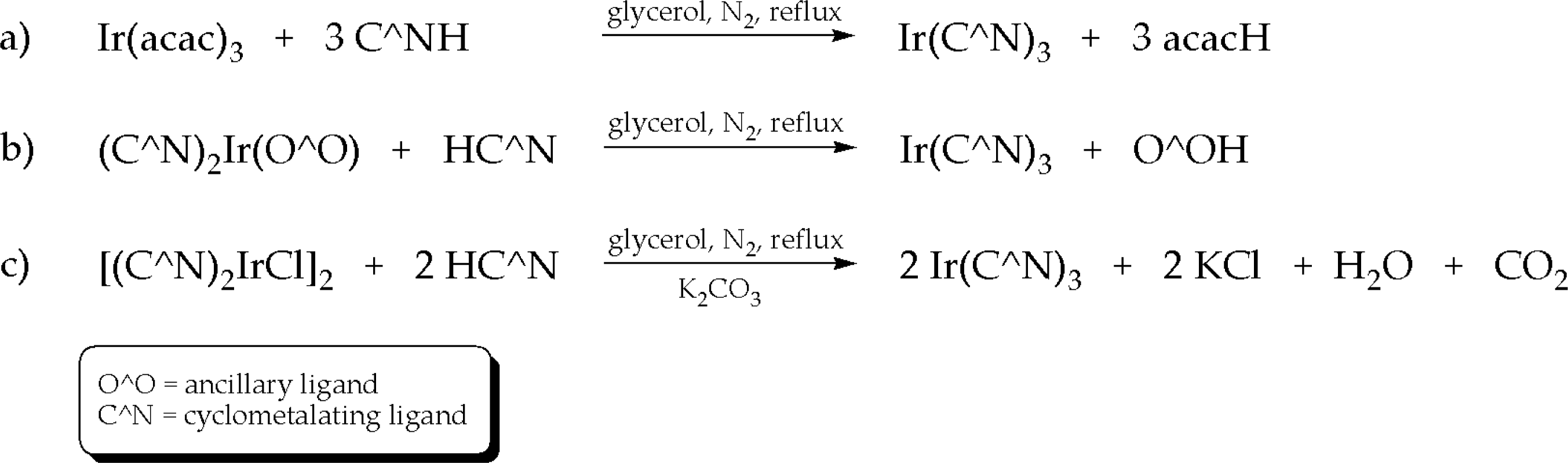
3.1. Synthesis of iridium(III) complexes
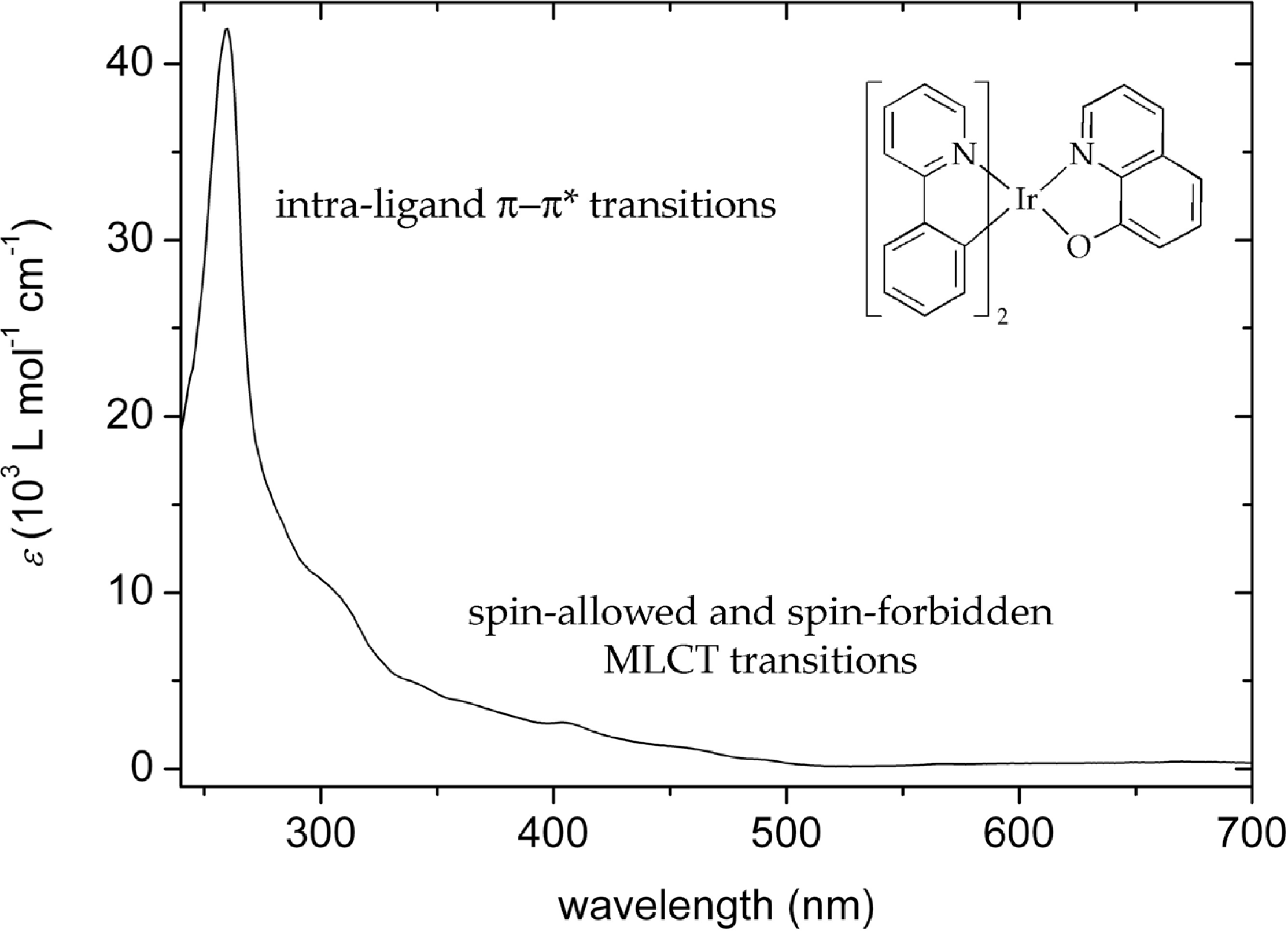
3.2. Photophysical properties of iridium(III) complexes
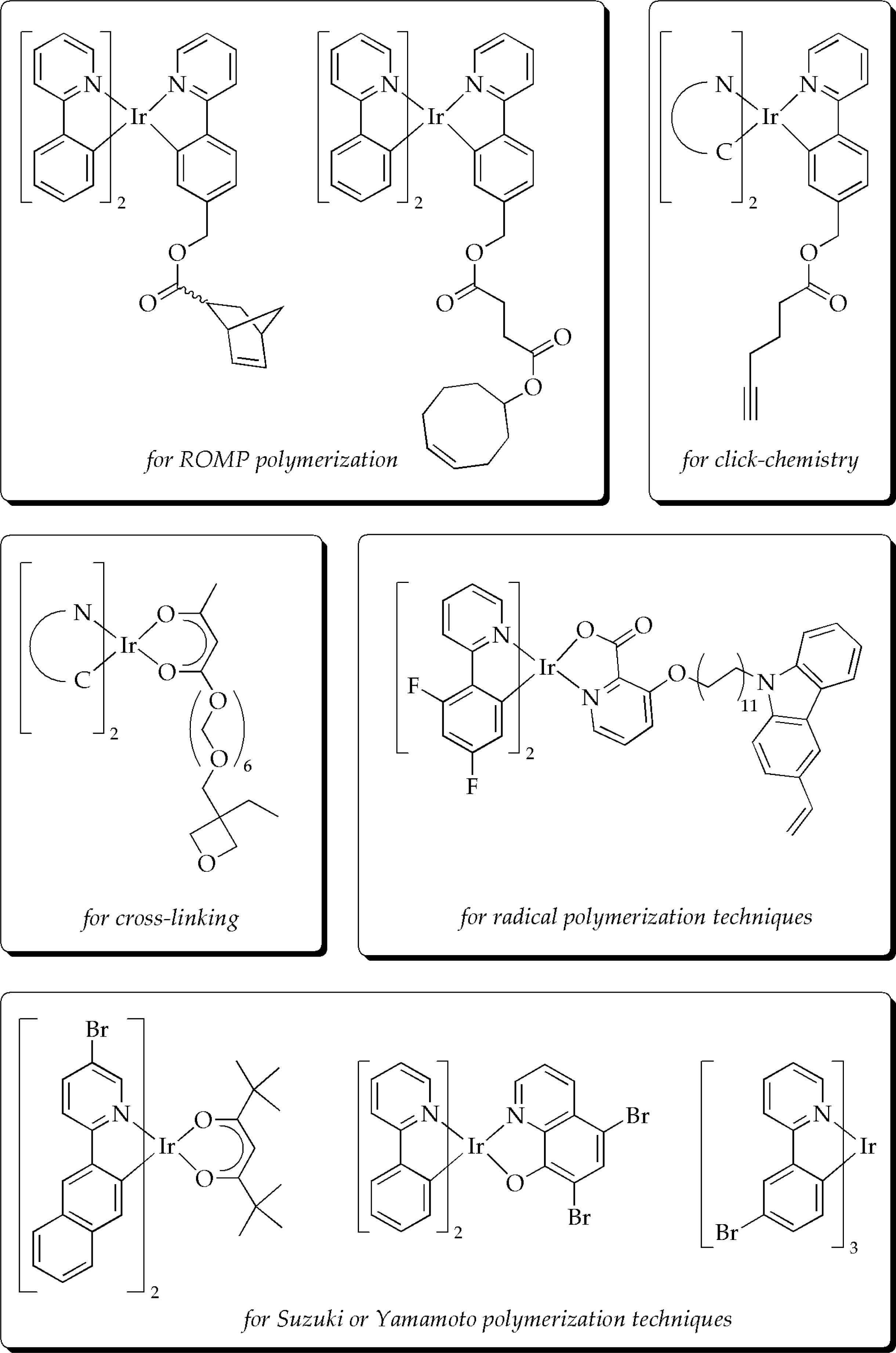
3.3. Polymeric phosphorescent materials
3. Conclusions and Outlook
Acknowledgments
References
- Pope, M; Kallmann, H; Magnante, P. Electroluminescence in Organic Crystals. J. Chem. Phys 1963, 38, 2042–2043. [Google Scholar]
- Vardeny, ZV; Heeger, AJ; Dodabalapur, A. Fundamental Research Needs in Organic Electronic Materials. Synth. Met 2005, 148, 1–3. [Google Scholar]
- Tang, CW; VanSlyke, SA. Organic Electroluminescent Diodes. Appl. Phys. Lett 1987, 51, 913–915. [Google Scholar]
- Haskal, EI; Büchel, M; Duineveld, PC; Sempel, A; van de Weijer, P. Passive-Matrix Polymer Light-Emitting Displays. MRS Bull 2002, 864–869. [Google Scholar]
- Nuyken, O; Jungermann, S; Wiederhirn, V; Bacher, E; Meerholz, K. Modern Trends in Organic Light-Emitting Devices (OLEDs). Monatsh. Chem 2006, 137, 811–824. [Google Scholar]
- Kido, J; Matsumoto, T. Bright Organic Electroluminescent Devices Having a Metal-doped Electron-injecting Layer. Appl. Phys. Lett 1998, 73, 2866–2868. [Google Scholar]
- Walzer, K; Maennig, B; Pfeiffer, M; Leo, K. Highly Efficient Organic Devices Based on Electrically Doped Transport Layers. Chem. Rev 2007, 107, 1233–1271. [Google Scholar]
- Sović, T; Kappaun, S; Koppitz, A; Zojer, E; Saf, R; Bartl, K; Fodor-Csorba, K; Vajda, A; Diele, S; Pelzl, G; Slugovc, C; Stelzer, F. Main-Chain Liquid Crystalline Polymers Based on Bis-Etherified 9,9-Dihexyl-2,7-bis(40-hydroxy-1,10-biphen-4-yl)fluorenes. Macromol. Chem. Phys 2007, 208, 1458–1468. [Google Scholar]
- Waser, R (Ed.) Nanoelectronics and Information Technology-Advanced Electronic Materials and Novel Devices; Wiley-VCH: Weinheim, 2003; p. 917.
- Blom, PWM; Tanase, C; de Leeuw, DM; Coehoorn, R. Thickness Scaling of the Space-charge-limited Current in poly(p-phenylene vinylene). Appl. Phys. Lett 2005, 86, 092105–092105-3. [Google Scholar]
- Wohlgenannt, M; Tandon, K; Mazumdar, S; Ramasesha, S; Vardeny, ZV. Formation Cross-sections of Singlet and Triplet Excitons in π-conjugated Polymers. Nature 2001, 409, 494–497. [Google Scholar]
- Köhler, A; Wilson, JS; Friend, RH. Fluorescence and Phosphorescence in Organic Materials. Adv. Mater 2002, 14, 701–707. [Google Scholar]
- Shuai, Z; Beljonne, D; Silbey, RJ; Brédas, JL. Singlet and Triplet Exciton Formation Rates in Conjugated Polymer Light-Emitting Diodes. Phys. Rev. Lett 2000, 84, 131–134. [Google Scholar]
- Yang, X; Neher, D. Polymer Electrophosphorescence Devices. In Organic Light-Emitting Devices; Müllen, K, Scherf, U, Eds.; Wiley-VCH: Weinheim, 2006; p. 333. [Google Scholar]
- Kido, J; Kimura, M; Nagai, K. Multilayer White Light-Emitting Organic Electroluminescent Device. Science 1995, 267, 1332–1334. [Google Scholar]
- Campbell, IH; Crone, BK; Smith, DL. Physics of Organic Light-Emitting Diodes. In Semiconducting Polymers; Hadziioannou, G, Malliaras, GG, Eds.; Wiley-VCH: Weinheim, 2007; p. 421. [Google Scholar]
- Sheats, JR. Manufacturing and Commercialization Issues in Organic Electronics. J. Mater. Res 2004, 19, 1974–1989. [Google Scholar]
- Gong, X; Moses, D; Heeger, AJ. Polymer-Based Light-Emitting Diodes (PLEDs) and Displays-Fabrication from Arrays of PLEDs. In Semiconducting Polymers; Hadziioannou, G, Malliaras, GG, Eds.; Wiley-VCH: Weinheim, 2007; p. 151. [Google Scholar]
- Fung, M-K; Lee, C-S; Lee, S-T. Metal/Polymer Interface Studies for Organic Light-Emitting Devices. In Organic Light-Emitting Devices; Müllen, K, Scherf, U, Eds.; Wiley-VCH: Weinheim, 2006; p. 181. [Google Scholar]
- Burroughes, JH; Bradley, DDC; Brown, AR; Marks, RN; Mackay, K; Friend, RH; Burns, PL; Holmes, AB. Light-emitting Diodes Based on Conjugated Polymers. Nature 1990, 347, 539–541. [Google Scholar]
- Lu, J; Jin, Y; Ding, J; Tao, Y; Day, M. High-efficiency Multilayer Polymeric Blue Light-emitting Diodes Using Boronate Esters as Cross-linking Linkages. J. Mater. Chem 2006, 16, 593–601. [Google Scholar]
- Lim, SC; Kim, SH; Chu, HY; Lee, JH; Lee, J-I; Oh, JY; Kim, D; Zyung, T. New Method of Driving an OLED with an OTFT. Synth. Met 2005, 151, 197–201. [Google Scholar]
- Shao, Y; Yang, Y. Organic Solid Solutions: Formation and Applications in Organic Light-Emitting Diodes. Adv. Funct. Mater 2005, 15, 1781–1786. [Google Scholar]
- Smith, LH; Wasey, JAE; Samuel, IDW; Barnes, WL. Light Out-Coupling Efficiencies of Organic Light-Emitting Diode Structures and the Effect of Photoluminescence Quantum Yield. Adv. Funct. Mater 2005, 15, 1839–1844. [Google Scholar]
- Yersin, H. Triplet Emitters for OLED Applications. Mechanisms of Exciton Trapping and Control of Emission Properties. Top. Curr. Chem 2004, 241, 1–26. [Google Scholar]
- Meerholz, K; Müller, C-D; Nuyken, O. Crosslinkable Organic Semiconductors for Use in Organic Light-Emitting Diodes (OLEDs). In Organic Light-Emitting Devices; Müllen, K, Scherf, U, Eds.; Wiley-VCH: Weinheim, 2006; p. 293. [Google Scholar]
- Baldo, MA; O’Brien, DF; You, Y; Shoustikov, A; Sibley, S; Thompson, ME; Forrest, SR. Highly Efficient Phosphorescent Emission from Organic Electroluminescent Devices. Nature 1998, 395, 151–154. [Google Scholar]
- Evans, RC; Douglas, P; Winscom, CJ. Coordination Complexes Exhibiting Room-temperature Phosphorescence: Evaluation of Their Suitability as Triplet Emitters in Organic Light Emitting Diodes. Coord. Chem. Rev 2006, 250, 2093–2126. [Google Scholar]
- Tsuboi, T; Aljaroudi, N. Electronic Relaxation Processes in the T1 State of Phosphorescent factris(2-phenylpyridine) Iridium Molecule in 4,4’-N,N’-dicarbazole-biphenyl Host. Jpn. J. Appl. Phys 2008, 47, 1266–1270. [Google Scholar]
- Hertel, D; Müller, CD; Meerholz, K. Organische Leuchtdioden: Bilderzeugung. Chem. Unserer Zeit 2005, 39, 336–347. [Google Scholar]
- Wang, Y; Herron, N; Grushin, VV; LeCloux, D; Petrov, V. Highly Efficient Electroluminescent Materials Based on Fluorinated Organometallic Iridium Compounds. Appl. Phys. Lett 2001, 79, 449–451. [Google Scholar]
- Wong, K-T; Chen, Y-M; Lin, Y-T; Su, H-C; Wu, C-C. Nonconjugated Hybrid of Carbazole and Fluorene: A Novel Host Material for Highly Efficient Green and Red Phosphorescent OLEDs. Org. Lett 2005, 7, 5361–5364. [Google Scholar]
- van Dijken, A; Bastiaansen, JJAM; Kiggen, NMM; Langeveld, BMW; Rothe, C; Monkman, A; Bach, I; Stössel, P; Brunner, K. Carbazole Compounds as Host Materials for Triplet Emitters in Organic Light-Emitting Diodes: Polymer Hosts for High-Efficiency Light-Emitting Diodes. J. Am. Chem. Soc 2004, 126, 7718–7727. [Google Scholar]
- Zhu, W; Mo, Y; Yuan, M; Yang, W; Cao, Y. Highly Efficient Electrophosphorescent Devices Based on Conjugated Polymers Doped with Iridium Complexes. Appl. Phys. Lett 2002, 80, 2045–2047. [Google Scholar]
- Brunner, K; van Dijken, A; Börner, H; Bastiaansen, JJAM; Kiggen, NMM; Langeveld, BMW. Carbazole Compounds as Host Materials for Triplet Emitters in Organic Light-Emitting Diodes: Tuning the HOMO Level without Influencing the Triplet Energy in Small Molecules. J. Am. Chem. Soc 2004, 126, 6035–6042. [Google Scholar]
- Chen, Y-C; Huang, G-S; Hsiao, C-C; Chen, S-A. High Triplet Energy Polymer as Host for Electrophosphorescence with High Efficiency. J. Am. Chem. Soc 2006, 128, 8549–8558. [Google Scholar]
- King, SM; Al-Attar, HA; Evans, RJ; Congreve, A; Beeby, A; Monkman, AP. The Use of Substituted Iridium Complexes in Doped Polymer Electrophosphorescent Devices: The Influence of Triplet Transfer and Other Factors on Enhancing Device Performance. Adv. Funct. Mater 2006, 16, 1043–1050. [Google Scholar]
- Vecchi, PA; Padmaperuma, AB; Qiao, H; Sapochak, LS; Burrows, PEA. Dibenzofuran-Based Host Material for Blue Electrophosphorescence. Org. Lett 2006, 8, 4211–4214. [Google Scholar]
- Lin, C-H; Chang, Y-T; Li, C-S; Liu, C-H; Duan, J-P. Bis(arylquinoxalinyl)carbazole Derivatives as Saturated Blue Emitters for Electroluminescent Devices. Synth. Met 2006, 156, 671–676. [Google Scholar]
- Tsai, M-H; Lin, H-W; Su, H-C; Ke, T-H; Wu, C-c; Fang, F-C; Liao, Y-L; Wong, K-T; Wu, C-I. Highly Efficient Organic Blue Electrophosphorescent Devices Based on 3,6-Bis(triphenylsilyl)carbazole as the Host Material. Adv. Mater 2006, 18, 1216–1220. [Google Scholar]
- Padmaperuma, AB; Sapochak, LS; Burrows, PE. New Charge Transporting Host Material for Short Wavelength Organic Electrophosphorescence: 2,7-Bis(diphenylphosphineoxide)-9,9-dimethylfluorene. Chem. Mater 2006, 18, 2389–2396. [Google Scholar]
- Gong, X; Ostrowski, JC; Moses, D; Bazan, GC; Heeger, AJ. High-performance Polymer-based Electrophosphorescent Light-emitting Diodes. J. Polym. Sci., Part B: Polym. Phys 2003, 41, 2691–2705. [Google Scholar]
- Li, Y; Ding, J; Day, M; Tao, Y; Lu, J; D’iorio, M. Synthesis and Properties of Random and Alternating Fluorene/Carbazole Copolymers for Use in Blue Light-Emitting Devices. Chem. Mater 2004, 16, 2165–2173. [Google Scholar]
- Diaz, JL; Dobarro, A; Villacampa, B; Velasco, D. Structure and Optical Properties of 2,3,7,9-Polysubstituted Carbazole Derivatives. Experimental and Theoretical Studies. Chem. Mater 2001, 13, 2528–2536. [Google Scholar]
- Grigalevicius, S. 3,6(2,7),9-Substituted carbazoles as Electroactive Amorphous Materials for Optoelectronics. Synth. Met 2006, 156, 1–12. [Google Scholar]
- Wong, K-T; Liao, Y-L; Lin, Y-T; Su, H-C; Wu, C-c. Nonconjugated Hybrid of Carbazole and Fluorene: A Novel Host Material for Highly Efficient Green and Red Phosphorescent OLEDs. Org. Lett 2005, 7, 5131–5364. [Google Scholar]
- Wu, F-I; Shih, P-I; Yuan, M-C; Dixit, AK; Shu, C-F; Chung, Z-M; Diau, EW-G. Novel Distyrylcarbazole Derivatives as Hole-transporting Blue Emitters for Electroluminescent Devices. J. Mater. Chem 2005, 15, 4753–4760. [Google Scholar]
- Cha, SW; Joo, S-H; Jeong, M-Y; Jin, J-I. Balancing Charge Carrier Mobility by Constructing Chemical Structures to Contain both Hole- and Electron-transporting Moieties in Electroluminescent Organic Compounds. Synth. Met 2005, 150, 309–316. [Google Scholar]
- Kremser, G; Hofmann, OT; Sax, S; Kappaun, S; List, EJW; Zojer, E; Slugovc, C. Synthesis and Photophysical Properties of 3,6-Diphenyl-9-hexyl-9H-carbazole Derivatives Bearing Electron Withdrawing Groups. Monatsh. Chem 2008, 139, 223–231. [Google Scholar]
- Niedermair, F; Waich, K; Kappaun, S; Mayr, T; Trimmel, G; Mereiter, K; Slugovc, C. Heteroleptic κ2(N,C2)-2-phenylpyridine Platinum Complexes: The Use of bis(pyrazolyl)borates as Ancillary Ligands. Inorg. Chim. Acta 2007, 360, 2767–2777. [Google Scholar]
- Niedermair, F; Kwon, O; Zojer, K; Kappaun, S; Trimmel, G; Mereiter, K; Slugovc, C. Heteroleptic Platinum(II) Complexes of 8-Quinolinolates Bearing Electron Withdrawing Groups in 5 Position. Dalton Trans 2008, 4006–4014. [Google Scholar]
- Dixon, IM; Collin, JP; Sauvage, JP; Flamigni, L; Encinas, S; Barigelletti, F. A Family of Luminescent Coordination Compounds: Iridium(III) Polyimine Complexes. Chem. Soc. Rev 2000, 29, 385–391. [Google Scholar]
- Lowry, MS; Bernhard, S. Synthetically Tailored Excited States: Phosphorescent, Cyclometalated Iridium(III) Complexes and Their Applications. Chem. Eur. J 2006, 12, 7970–7977. [Google Scholar]
- Goushi, K; Kwong, R; Brown, JJ; Sasabe, H; Adachi, C. Triplet Exciton Confinement and Unconfinement by Adjacent Hole-transport Layers. J. Appl. Phys 2004, 95, 7798–7802. [Google Scholar]
- Holder, E; Langeveld, BMW; Schubert, US. New Trends in the Use of Transition Metal-Ligand Complexes for Applications in Electroluminescent Devices. Adv. Mater 2005, 17, 1109–1121. [Google Scholar]
- Slinker, JD; Gorodetsky, AA; Lowry, MS; Wang, J; Parker, S; Rohl, R; Bernhard, S; Malliaras, GG. Efficient Yellow Electroluminescence from a Single Layer of a Cyclometalated Iridium Complex. J. Am. Chem. Soc 2004, 126, 2763–2767. [Google Scholar]
- Nonoyama, M. Benzo[h]quinolin-10-yl-N Iridium(III) Complexes. Bull. Chem. Soc. Jpn 1974, 47, 767–768. [Google Scholar]
- Ionkin, AS; Marshall, WJ; Fish, BM. Synthesis and Structural Characterization of a Series of Novel Polyaromatic Ligands Containing Pyrene and Related Biscyclometalated Iridium(III) Complexes. Organometallics 2006, 25, 1461–1471. [Google Scholar]
- King, KA; Spellane, PJ; Watts, RJ. Excited-state Properties of a Triply Ortho-metalated Iridium(III) Complex. J. Am. Chem. Soc 1985, 107, 1431–1432. [Google Scholar]
- Ionkin, AS; Marshall, WJ; Roe, DC; Wang, Y. Synthesis, Structural Characterization and the First Electroluminescent Properties of Tris- and Bis-cycloiridiated Complexes of Sterically Hindered Electron-poor 2-(3,5-bis(trifluoromethyl)phenyl)-4-trifluoromethylpyridine. Dalton Trans 2006, 2468–2478. [Google Scholar]
- Tamayo, AB; Alleyne, BD; Djurovich, PI; Lamansky, S; Tsyba, I; Ho, NN; Bau, R; Thompson, ME. Synthesis and Characterization of Facial and Meridional Tris-cyclometalated Iridium(III) Complexes. J. Am. Chem. Soc 2003, 125, 7377–7387. [Google Scholar]
- Huo, S; Deaton, JC; Rajeswaran, M; Lenhart, WC. Highly Efficient, Selective, and General Method for the Preparation of Meridional Homo- and Heteroleptic Tris-cyclometalated Iridium Complexes. Inorg. Chem 2006, 45, 3155–3157. [Google Scholar]
- Dedeian, K; Shi, J; Shepard, N; Forsythe, E; Morton, DC. Photophysical and Electrochemical Properties of Heteroleptic Tris-Cyclometalated Iridium(III) Complexes. Inorg. Chem 2005, 44, 4445–4447. [Google Scholar]
- Kappaun, S; Rentenberger, S; Pogantsch, A; Zojer, E; Mereiter, K; Trimmel, G; Saf, R; Möller, KC; Stelzer, F; Slugovc, C. Organoboron Quinolinolates with Extended Conjugated Chromophores: Synthesis, Structure, and Electronic and Electroluminescent Properties. Chem. Mater 2006, 18, 3539–3547. [Google Scholar]
- You, Y; Park, SY. Inter-Ligand Energy Transfer and Related Emission Change in the Cyclometalated Heteroleptic Iridium Complex: Facile and Efficient Color Tuning over the Whole Visible Range by the Ancillary Ligand Structure. J. Am. Chem. Soc 2005, 127, 12438–12439. [Google Scholar]
- Kappaun, S; Sax, S; Eder, S; Möller, KC; Waich, K; Niedermair, F; Saf, R; Mereiter, K; Jacob, J; Müllen, K; List, EJW; Slugovc, C. 8-Quinolinolates as Ligands for Luminescent Cyclometalated Iridium Complexes. Chem. Mater 2007, 19, 1209–1211. [Google Scholar]
- Kappaun, S; Eder, S; Sax, S; Mereiter, K; List, EJW; Slugovc, C. Organoiridium Quinolinolate Complexes: Synthesis, Structures, Thermal Stabilities and Photophysical Properties. Eur. J. Inorg. Chem 2007, 4207–4215. [Google Scholar]
- Lamansky, S; Djurovic, P; Murphy, D; Abdel-Razzaq, F; Kwong, R; Tsyba, I; Bortz, M; Mui, B; Bau, R; Thompson, ME. Synthesis and Characterization of Phosphorescent Cyclometalated Iridium Complexes. Inorg. Chem 2001, 40, 1704–1711. [Google Scholar]
- Lamansky, S; Djurovich, P; Murphy, D; Abdel-Razzaq, F; Lee, H-E; Adachi, C; Burrows, PE; Forrest, SR; Thompson, ME. Highly Phosphorescent Bis-Cyclometalated Iridium Complexes: Synthesis, Photophysical Characterization, and Use in Organic Light Emitting Diodes. J. Am. Chem. Soc 2001, 123, 4304–4312. [Google Scholar]
- Hsu, N-M; Li, W-R. Accelerated Discovery of Red-Phosphorescent Emitters through Combinatorial Organometallic Synthesis and Screening. Angew. Chem 2006, 118, 4244–4248. [Google Scholar]
- Yang, C-H; Li, S-W; Chi, Y; Cheng, Y-M; Yeh, Y-S; Chou, P-T; Lee, G-H; Wang, C-H; Shu, C-F. Heteroleptic Cyclometalated Iridium(III) Complexes Displaying Blue Phosphorescence in Solution and Solid State at Room Temperature. Inorg. Chem 2005, 44, 7770–7780. [Google Scholar]
- Sajoto, T; Djurovich, PI; Tamayo, A; Yousufuddin, M; Bau, R; Thompson, ME. Blue and Near-UV Phosphorescence from Iridium Complexes with Cyclometalated Pyrazolyl or N-Heterocyclic Carbene Ligands. Inorg. Chem 2005, 44, 7992–8003. [Google Scholar]
- Mak, CSK; Hayer, A; Pascu, SI; Watkins, SE; Holmes, AB; Köhler, A; Friend, RH. Blue-to-green Electrophosphorescence of Iridium-based Cyclometalated Materials. Chem. Commun 2005, 4708–4710. [Google Scholar]
- Jang, H; Shin, CH; Kim, NG; Hwang, KY; Do, Y. New Phosphorescent ppy-based Iridium Complexes Containing Electron-withdrawing Groups. Synth. Met 2005, 154, 157–160. [Google Scholar]
- Ragni, R; Plummer, EA; Brunner, K; Hofstraat, JW; Babudri, F; Farinola, GM; Naso, F; De Cola, L. Blue Emitting Iridium Complexes: Synthesis, Photophysics and Phosphorescent Devices. J. Mater. Chem 2006, 16, 1161–1170. [Google Scholar]
- Chin, CS; Eum, M-S; Kim, Sy; Kim, C; Kang, SK. Blue-Light-Emitting Complexes: Cationic (2-Phenylpyridinato)iridium(III) Complexes with Strong-Field Ancillary Ligands. Eur. J. Inorg. Chem 2007, 372–375. [Google Scholar]
- Lo, S-C; Shipley, CP; Bera, RN; Harding, RE; Cowley, AR; Burn, PL; Samuel, IDW. Blue Phosphorescence from Iridium(III) Complexes at Room Temperature. Chem. Mater 2006, 18, 5119–5129. [Google Scholar]
- Chou, P-T; Chi, Y. Phosphorescent Dyes for Organic Light-Emitting Diodes. Chem. Eur. J 2007, 13, 380–395. [Google Scholar]
- Li, J; Djurovich, PI; Alleyne, BD; Yousufuddin, M; Ho, NN; Thomas, JC; Peters, JC; Bau, R; Thompson, ME. Synthetic Control of Excited-State Properties in Cyclometalated Ir(III) Complexes Using Ancillary Ligands. Inorg. Chem 2005, 44, 1713–1727. [Google Scholar]
- Liu, T; Xia, B-H; Zhou, X; Zhang, H-X; Pan, Q-J; Gao, J-S. Theoretical Studies on Structures and Spectroscopic Properties of Bis-Cyclometalated Iridium Complexes. Organometallics 2007, 26, 143–149. [Google Scholar]
- Kwon, T-H; Cho, HS; Kim, MK; Kim, J-W; Kim, J-J; Lee, KH; Park, SJ; Shin, I-S; Kim, H; Shin, DM; Chung, YK; Hong, J-I. Color Tuning of Cyclometalated Iridium Complexes through Modification of Phenylpyrazole Derivatives and Ancillary Ligand Based on ab Initio Calculations. Organometallics 2005, 24, 1578–1585. [Google Scholar]
- Bansal, AK; Holzer, W; Penzkofer, A; Tsuboi, T. Absorption and Emission Spectroscopic Characterization of Platinum-octaethyl-porphyrin (PtOEP). Chem. Phys 2006, 330, 118–129. [Google Scholar]
- Holzer, W; Penzkofer, A; Tsuboi, T. Absorption and Emission Spectroscopic Characterization of Ir(ppy)3. Chem. Phys 2005, 308, 93–102. [Google Scholar]
- Koga, Y; Ueno, K; Matsubara, K. New Preparative Procedure for Photoluminescent Metallopolymers Having a Biphenyl-2,2’-diyl Iridium(III) Unit Bound to a Phosphine Copolymer Ligand. J. Polym. Sci., Part A: Polym. Chem 2006, 44, 4204–4213. [Google Scholar]
- You, Y; Kim, SH; Jung, HK; Park, SY. Blue Electrophosphorescence from Iridium Complex Covalently Bonded to the Poly(9-dodecyl-3-vinylcarbazole): Suppressed Phase Segregation and Enhanced Energy Transfer. Macromolecules 2006, 39, 349–356. [Google Scholar]
- Jiang, J; Jiang, C; Yang, W; Zhen, H; Huang, F; Cao, Y. High-Efficiency Electrophosphorescent Fluorene-alt-carbazole Copolymers N-Grafted with Cyclometalated Ir Complexes. Macromolecules 2005, 38, 4072–4080. [Google Scholar]
- Carlise, JR; Wang, X-Y; Weck, M. Phosphorescent Side-Chain Functionalized Poly(norbornene)s Containing Iridium Complexes. Macromolecules 2005, 38, 9000–9008. [Google Scholar]
- Wang, X-Y; Prabhu, RN; Schmehl, RH; Weck, M. Polymer-Based Tris(2-phenylpyridine)iridium Complexes. Macromolecules 2006, 39, 3140–3146. [Google Scholar]
- Chen, X; Liao, J-L; Liang, Y; Ahmed, MO; Tseng, H-E; Chen, S-A. High-Efficiency Red-Light Emission from Polyfluorenes Grafted with Cyclometalated Iridium Complexes and Charge Transport Moiety. J. Am. Chem. Soc 2003, 125, 636–637. [Google Scholar]
- Deng, L; Furuta, PT; Garon, S; Li, J; Kavulak, D; Thompson, ME; Frechet, JMJ. Living Radical Polymerization of Bipolar Transport Materials for Highly Efficient Light Emitting Diodes. Chem. Mater 2006, 18, 386–395. [Google Scholar]
- Wang, X-Y; Kimyonok, A; Weck, M. Functionalization of Polymers with Phosphorescent Iridium Complexes via Click Chemistry. Chem. Commun 2006, 3933–3925. [Google Scholar]
- Kimyonok, A; Weck, M. Poly(cyclooctene)s with Pendant Fluorescent and Phosphorescent Metal Complexes. Macromol. Rapid Commun 2007, 28, 152–157. [Google Scholar]
- Zhang, K; Chen, Z; Yang, C; Gong, S; Qin, J; Cao, Y. Saturated Red-Emitting Electrophosphorescent Polymers with Iridium Coordinating to β-Diketonate Units in the Main Chain. Macromol. Rapid Commun 2006, 27, 1926–1931. [Google Scholar]
- Evans, NR; Devi, LS; Mak, CSK; Watkins, SE; Pascu, SI; Köhler, A; Friend, RH; Williams, CK; Holmes, AB. Triplet Energy Back Transfer in Conjugated Polymers with Pendant Phosphorescent Iridium Complexes. J. Am. Chem. Soc 2006, 128, 6647–6656. [Google Scholar]
- Kimyonok, A; Domercq, B; Haldi, A; Cho, J-Y; Carlise, JR; Wang, X-Y; Hayden, LE; Jones, SC; Barlow, S; Marder, SR; Kippelen, B; Weck, M. Norbornene-Based Copolymers with Iridium Complexes and Bis(carbazolyl)fluorene Groups in Their Side-Chains and Their Use in Light-Emitting Diodes. Chem. Mater 2007, 19, 5602–5608. [Google Scholar]
- Rehmann, N; Ulbricht, C; Köhnen, A; Zacharias, P; Gather, MC; Hertel, D; Holder, E; Meerholz, K; Schubert, U. Advanced Device Architecture for Highly Efficient Organic Light-Emitting Diodes with an Orange-Emitting Crosslinkable Iridium(III) Complex. Adv. Mater 2008, 20, 129–133. [Google Scholar]
- Yang, W; Zhen, HY; Jiang, CY; Su, LJ; Jiang, JX; Shi, HH; Cao, Y. Synthesis of Electrophosphorescent Polymers Based on Para-phenylenes with Iridium Complexes. Synth. Met 2005, 153, 189–192. [Google Scholar]
- Zhen, H; Jiang, C; Yang, W; Jiang, J; Huang, F; Cao, Y. Synthesis and Properties of Electrophosphorescent Chelating Polymers with Iridium Complexes in the Conjugated Backbone. Chem. Eur. J 2005, 11, 5007–5016. [Google Scholar]
- Zhen, H; Luo, C; Yang, W; Song, W; Du, B; Jiang, J; Jiang, C; Zhang, Y; Cao, Y. Electrophosphorescent Chelating Copolymers Based on Linkage Isomers of Naphthylpyridine-Iridium Complexes with Fluorene. Macromolecules 2006, 39, 1693–1700. [Google Scholar]
- Liu, S-J; Zhao, Q; Chen, R-F; Deng, Y; Fan, Q-L; Li, F-Y; Wang, L-H; Huang, C-H; Huang, W. π-Conjugated Chelating Polymers with Charged Iridium Complexes in the Backbones: Synthesis, Characterization, Energy Transfer, and Electrochemical Properties. Chem. Eur. J 2006, 12, 4351–4361. [Google Scholar]
- Zhuang, W; Zhang, Y; Hou, Q; Wang, L; Cao, Y. High-Efficiency, Electrophosphorescent Polymers with Porphyrin–Platinum Complexes in the Conjugated Backbone: Synthesis and Device Performance. J. Polym. Sci., Part A: Polym. Chem 2006, 44, 4174–4186. [Google Scholar]
- Kappaun, S; Eder, S; Sax, S; Saf, R; Mereiter, K; List, EJW; Slugovc, C. WPLEDs Prepared from Main-chain Fluorene–iridium(III) Polymers. J. Mater. Chem 2006, 16, 4389–4392. [Google Scholar]

© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/). This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kappaun, S.; Slugovc, C.; List, E.J.W. Phosphorescent Organic Light-Emitting Devices: Working Principle and Iridium Based Emitter Materials. Int. J. Mol. Sci. 2008, 9, 1527-1547. https://doi.org/10.3390/ijms9081527
Kappaun S, Slugovc C, List EJW. Phosphorescent Organic Light-Emitting Devices: Working Principle and Iridium Based Emitter Materials. International Journal of Molecular Sciences. 2008; 9(8):1527-1547. https://doi.org/10.3390/ijms9081527
Chicago/Turabian StyleKappaun, Stefan, Christian Slugovc, and Emil J. W. List. 2008. "Phosphorescent Organic Light-Emitting Devices: Working Principle and Iridium Based Emitter Materials" International Journal of Molecular Sciences 9, no. 8: 1527-1547. https://doi.org/10.3390/ijms9081527
APA StyleKappaun, S., Slugovc, C., & List, E. J. W. (2008). Phosphorescent Organic Light-Emitting Devices: Working Principle and Iridium Based Emitter Materials. International Journal of Molecular Sciences, 9(8), 1527-1547. https://doi.org/10.3390/ijms9081527