Plant Products for Pharmacology: Application of Enzymes in Their Transformations
Abstract
1. Introduction
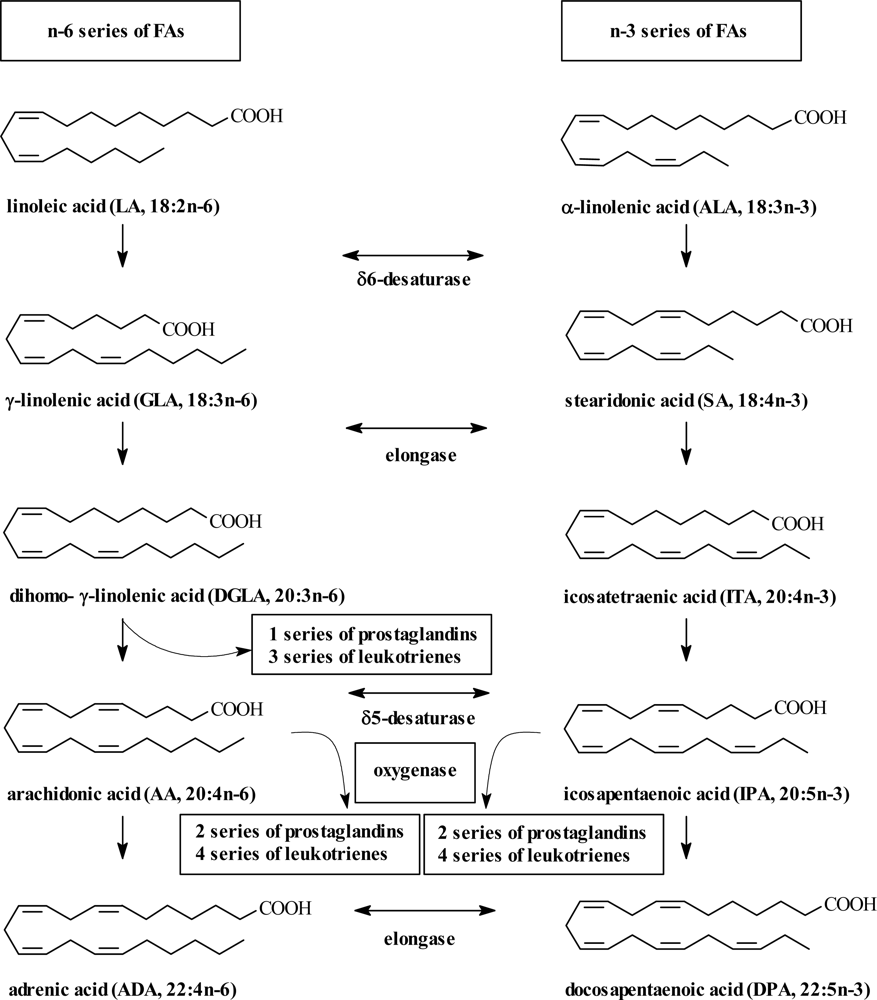
2. PUFA Sources
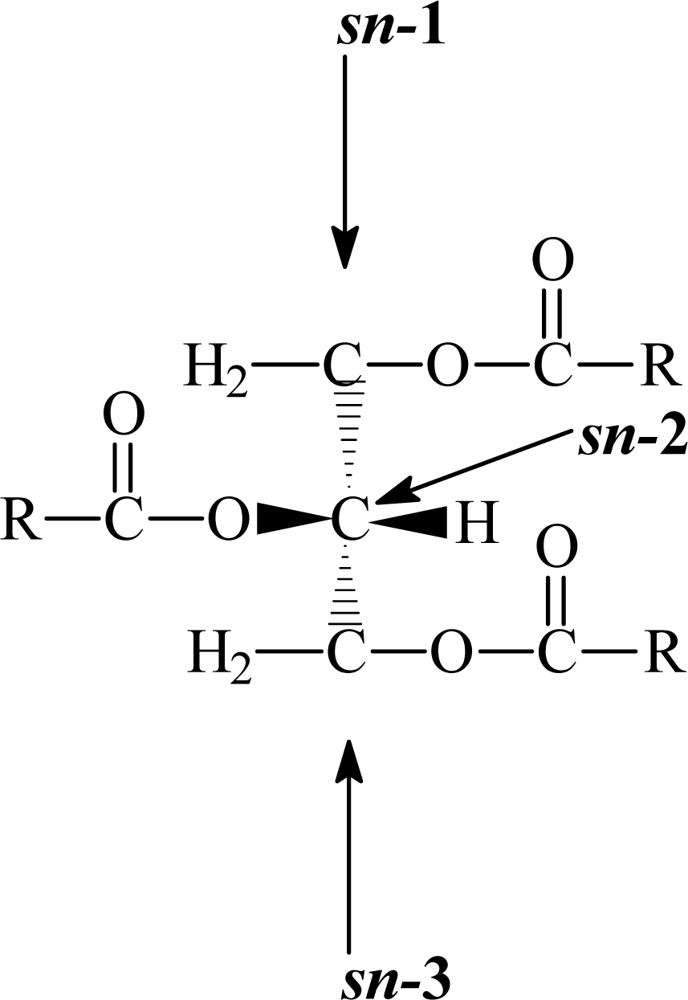
3. Triacylglycerol structure
4. Enzymes acting in lipid modifications
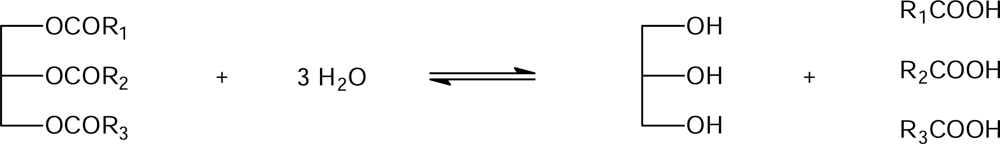
4.1. Lipases
4.2. Lipase structure and catalytic ability
4.3. Lipase-catalyzed hydrolysis under conventional conditions
4.4. Lipase-catalyzed reactions under non-conventional conditions
4.4.1. Properties of supercritical carbon dioxide
4.4.2. Properties of ionic liquids
4.4.3. Lipase-catalyzed hydrolysis in supercritical carbon dioxide
4.5. Lipase-catalyzed esterification in organic solvents
4.6. Lipase-catalyzed transesterification and acidolysis under conventional conditions
4.7. Structured lipids
4.7.1. Lipase-catalyzed esterification in supercritical carbon dioxide
4.7.2. Other types of lipase-catalyzed reactions in supercritical carbon dioxide
4.9. Lipase-catalyzed reactions in ionic liquid / supercritical carbon dioxide biphasic systems
5. Conclusions: Importance of the reaction media for sustainability
Acknowledgments
References
- Emken, EA; Adlof, RO; Gulley, RM. Dietary linoleic acid influences desaturation and acylation of deuterium-labeled linoleic and linolenic acids in young adult males. Biochim. Biophys. Acta 1994, 1213, 277–288. [Google Scholar]
- Holub, DJ; Holub, BJ. Omega-3 fatty acids from fish oils and cardiovascular disease. Mol. Cell Biochem 2004, 263, 217–225. [Google Scholar]
- Peet, M; Brind, J; Ramchand, CN; Shah, S; Vankar, GK. Two double blind placebo controlled pilot studies of eicosapentaenoic acid in the treatment of schizophrenia. Schizophr. Res 2001, 49, 243–251. [Google Scholar]
- Simopoulos, AP. Omega-3 fatty acids in health and disease and in growth and development. Am. J. Clin. Nutr 1991, 54, 438–463. [Google Scholar]
- von Schacky, C. Omega-3 fatty acids and cardiovascular disease. Curr. Opin. Clin. Nutr. Metab.Care 2004, 7, 31–136. [Google Scholar]
- Stahl, SM. Essential psychopharmacology. Neuroscientific basis and practical applications, 2nd Ed ed; Cambridge University Press: Cambridge, 2000. [Google Scholar]
- Glen, AIM; Ross, BM. Eicosanoids in the central nervous system. In The Eicosanoids; Curtis-Prior, P, Ed.; Wiley: London, UK, 2004; pp. 210–220. [Google Scholar]
- Carlezon, WA, Jr; Mague, SD; Parow, AM; Stoll, AL; Cohen, BM; Renshaw, PF. Antidepressant-like effects of uridine and omega-3 fatty acids are potentiated by combined treatment in rats. Biol. Psychiatry 2005, 57, 343–350. [Google Scholar]
- Ross, BM; Seguin, J; Sieswerda, LE. Omega-3 fatty acids as treatments for mental illness: Which disorder and which fatty acid? Lipids Health Dis 2007, 6, 21–30. [Google Scholar]
- Xiao, YF; Sigg, DC; Leaf, A. The antiarrhythmic effect of n-3 polyunsaturated fatty acids: Modulation of cardiac ion channels as a potential mechanism. J. Membr. Biol 2005, 206, 141–154. [Google Scholar]
- Wigmore, SJ; Ross, JA; Falconer, JS; Plester, CE; Tisdade, MJ; Carter, DC; Faron, KCH. Effect of PUFA on the progress of cachexia in patiens with pancreatic cancer. Nutrition 1996, 12, 27–30. [Google Scholar]
- Beaubaire, NA; Simon, JE. Production potential of Borago oficinalis L. Acta Horticult 1987, 208, 101–103. [Google Scholar]
- Senanayake, SPJN; Shahidi, F. Enzyme-assisted acidolysis of borage (Borago officinalis L.) and evening primrose (Oenothera biennis L.) oils: Incorporation of omega-3 polyunsaturated fatty acids. J. Agric. Food Chem 1999, 47, 3105–3112. [Google Scholar]
- Stránský, K; Zarevúcka, M; Wimmer, Z. Gas chromatography analysis of blackcurrant oil in relation to its stability. Food Chem 2005, 92, 569–573. [Google Scholar]
- Traitler, H; Wille, HJ; Studer, A. Fractionation of Blackcurrant seed oil. J. Am. Oil Chem. Soc 1988, 65, 755–760. [Google Scholar]
- Wille, HJ; Traitler, H; Lagarde, M. Preparation of stearidonic acid concentrates. Fat Sci. Technol 1991, 93, 362–368. [Google Scholar]
- Traitler, H; Wille, HJ. Isolation of pure fatty acids from fats and oils. Fat Sci. Technol 1992, 94, 506–511. [Google Scholar]
- Syed Rahmatullah, MSK; Shukla, VKS; Mukherjee, KD. Enrichment of γ-linolenic acid from evening primrose oil and borage oil via lipase-catalyzed hydrolysis. J. Am. Oil Chem. Soc 1994, 71, 560–573. [Google Scholar]
- Syed Rahmatullah, MSK; Shukla, VKS; Mukherjee, KD. γ-Linolenic acid concentrates from borage and evening priprose oil via lipase-catalyzed esterification. J. Am. Oil Chem. Soc 1994, 71, 563–577. [Google Scholar]
- Kawashima, H; Sakuradani, E; Kamada, N; Akimoto, K; Konishi, K; Ogawa, J; Shimizu, S. Production of 8,11,14,17-cis-eicosatetraenoic acid (20 : 4 omega-3) by a Delta 5 and Delta 12 desaturase-defective mutant of an arachidonic acid-producing fungus Mortierella alpina 1S-4. J. Am. Oil Chem. Soc 1998, 75, 1495–1500. [Google Scholar]
- Shinmen, Y; Shimizu, S; Akimoto, D; Kawashima, H; Yamada, H. Production of arachidonic acid by Mortierella fungi. Selection of a potent producer and optimization of culture conditions for large-scale production. Appl. Microbiol. Biotechnol 1989, 31, 11–16. [Google Scholar]
- Yongmanitchai, W; Ward, OP. Screening of algae for potential alternative sources of eicosapentaenoic acid. Phytochemistry 1991, 30, 2963–2967. [Google Scholar]
- Nichols, BW; Appleby, RS. The distribution and biosynthesis of arachidonic acid in algae. Phytochemistry 1969, 8, 1907–1915. [Google Scholar]
- Monroig, O; Navarro, JC; Amat, F; Gonzáles, P; Bermejo, A; Hontoria, F. Enrichment of Artemia nauplii in essential fatty acids with different types of liposomes and their use in the rearing of gilthead sea bream (Sparus aurata) larvae. Aquaculture 2006, 251, 491–508. [Google Scholar]
- Zhu, J-J; Shi, J-H; Qian, W-B; Cai, Z-Z; Li, D. Effects of krill oil on serum lipids of hyperlipidemic rats and human SW480 cells. Lipids Health Dis 2008, 7, 30. [Google Scholar]
- Shimada, Y; Sugihara, A; Nakano, H; Kuramoto, T; Nagao, T; Gemba, M; Tominaga, Y. Purification of docosahexaenoic acid by selective esterification of fatty acids from tuna oil with Rhizopus delemar lipase. J. Am. Oil Chem. Soc 1997, 74, 97–101. [Google Scholar]
- Salunkhe, DK; Chavan, JK; Adsule, RN; Kadam, SS. World Oilseeds: Chemistry, Technology, and Utilization; Salunkhe, DK, Van Nostrand, R, Eds.; Springer: New York, USA, 1992. [Google Scholar]
- Gunstone, FD; Ilyas-Qureshi, M. Glyceride studies. Part IV. The component glycerides of ten seed oils containing linoleic acid. J. Am. Oil. Chem. Soc 1965, 42, 961–965. [Google Scholar]
- Gunstone, FD; Hamilton, RJ; Padley, FB; Ilyas-Qureshi, M. Glyceride studies. V. The distribution of unsaturated acyl groups in vegetable triglycerides. J. Am. Oil. Chem. Soc 1965, 42, 965–970. [Google Scholar]
- Biermann, U; Friedt, W; Lang, S; Lühs, W; Machmüller, G; Metzger, JO; Rüsch gen. Klaas, M; Schäfer, J; Schneider, MP. New syntheses with oils and fats as renewable raw materials for the chemical industry. Angew. Chem. Int. Ed. Engl 2000, 39, 2206–2224. [Google Scholar]
- Hayes, DG. Enzyme-catalyzed modification of oilseed materials to produce eco-friendly products. J. Am. Oil Chem. Soc 2004, 81, 1077–1103. [Google Scholar]
- Gandhi, NN. Applications of lipase. J. Am. Oil Chem. Soc 1997, 74, 621–634. [Google Scholar]
- Albasi, C; Bertrand, N; Riba, JP. Enzymatic hydrolysis of sunflower oil in a standardized agitated tank reactor. Bioprocess Eng 1999, 20, 77–81. [Google Scholar]
- Martinez, O; Wilhelm, AM; Riba, JP. Kinetic-study of an enzymatic liquid liquid reaction – the hydrolysis of tributyrin by Candida cylindracea lipase. J. Chem. Tech. Biotechnol 1992, 53, 373–378. [Google Scholar]
- Habulin, M; Knez, Ž. High-pressure enzymatic hydrolysis of oil. Eur. J. Lipid Sci. Technol 2002, 104, 381–386. [Google Scholar]
- Malcata, FX; Reyes, HR; Garcia, HS; Hill, CG, Jr; Amundson, CH. Immobilized lipase reactions for modification of fats and oils – A review. J. Am. Oil Chem. Soc 1990, 67, 890–910. [Google Scholar]
- Bogel-Lukasik, R. Sustainable processes employing ionic liquids for secondary alcohols separation. Monatsh. Chem 2007, 138, 1137–1144. [Google Scholar]
- Dordick, JS. Nonaqueous enzymology. Curr. Opin. Biotechnol 1991, 2, 401–407. [Google Scholar]
- Reverchon, E; De Marco, I. Supercritical fluid extraction and fractionation of natural matters. J. Supercrit Fluids 2006, 38, 146–166. [Google Scholar]
- Klibanov, AM. Improving enzymes by using them in organic solvents. Nature 2001, 409, 241–246. [Google Scholar]
- Schoffers, E; Golebioeski, A; Johnson, CR. Enantioselective synthesis through enzymatic asymmetrization. Tetrahedron 1996, 52, 3769–3826. [Google Scholar]
- Klibanov, AM. Why are enzymes less active in organic solvents than in water? Trends Biotechnol 1997, 15, 97–101. [Google Scholar]
- Schmid, RD; Verger, R. Lipases: Interfacial enzymes with attractive applications. Angew. Chem. Int. Ed. Engl 1998, 37, 1608–1633. [Google Scholar]
- Beisson, F; Tiss, A; Riviere, C; Verger, R. Methods for lipase detection and assay: A critical review. Eur. J. Lipid Sci. Technol 2000, 133–153. [Google Scholar]
- Krishna, HS; Karanth, NG. Lipases and lipase-catalyzed esterification reactions in nonaqueous media. Catal. Rev 2002, 44, 499–591. [Google Scholar]
- Patel, RN. Microbial/enzymatic synthesis of chiral intermediates for pharmaceuticals. Enzyme Microb. Technol 2002, 31, 804–826. [Google Scholar]
- Patel, RN. Biocatalytic synthesis of chiral pharmaceutical intermediate. Food Technol. Biotechnol 2004, 42, 305–325. [Google Scholar]
- Wong, C-H; Whitesides, GM. Enzymes in Synthetic Organic Chemistry; Pergamon: Oxford, UK, 1994. [Google Scholar]
- Dias, SF; Vilas-Boas, L; Cabral, JMS; Fonseca, MMR. Production of ethyl butyrate by Candida rugosa lipase immobilized in polyurethane. Biocatalysis 1991, 5, 21–34. [Google Scholar]
- Macrae, AR. Biocatalysts in Organic Synthesis; Tramper, J, van der Plas, HC, Linko, P, Eds.; Elsevier: Amsterdam, The Netherlands, 1985. [Google Scholar]
- Muderhwa, JM; Pina, M; Graille, J. Interesterification catalyzed by 1–3 regioselective lipases without solvent-valorization of palm oil and its solid fraction. Rev. Fr. Corps Gras 1989, 36, 11–19. [Google Scholar]
- Kyotani, S; Nakashima, T; Izumoto, E; Fukuda, H. Continuous interesterification of oils and fats using dried fungus immobilized in biomass support particles. J. Ferment. Bioeng 1991, 71, 286–288. [Google Scholar]
- Mojovič, L; Siler-Marinkovič, S; Kukič, G; Vunjak-Novakovič, G. Rhizopus arrhizus lipase-catalyzed interesterification of the midfraction of palm oil to a cocoa butter equivalent fat. Enzyme Microb. Technol 1993, 15, 438–443. [Google Scholar]
- Forssell, P; Paruvuori, P; Linko, P; Poutanen, K. Enzymatic transesterification of rapeseed oil and lauric acid in a continuous reactor. J. Am. Oil Chem. Soc 1993, 70, 1105–1109. [Google Scholar]
- Ghazali, HM; Hamidah, S; Che Man, YB. Enzymatic transesterification of palm olein with nonspecific and 1,3-specific lipases. J. Am. Oil Chem. Soc 1995, 72, 633–639. [Google Scholar]
- Soumanou, MM; Bornscheuer, UT; Menge, U; Schmid, RD. Synthesis of structured triglycerides from peanut oil with immobilized lipase. J. Am. Oil Chem. Soc 1997, 74, 427–433. [Google Scholar]
- Ghosh, S; Bhattacharyya, DK. Utilization of high-melting palm stearin in lipase-catalyzed interesterification with liquid oils. J. Am. Oil Chem. Soc 1997, 74, 589–592. [Google Scholar]
- Mukesh, D; Banerji, AA; Newadkar, R; Bevinakatti, HS. Lipase catalysed transesterification of vegetable oils - A comparative study in batch and tubular reactors. Biotechnol. Lett 1993, 15, 77–82. [Google Scholar]
- Xu, X; Balchen, S; Høy, C-E; Adler-Nissen, J. Pilot batch production of specific-structured lipids by lipase-catalyzed interesterification: Preliminary study on incorporation and acyl migration. J. Am. Oil Chem. Soc 1998, 75, 301–308. [Google Scholar]
- Seriburi, V; Akoh, CC. Enzymatic transesterification of triolein and stearic acid and solid fat content of their products. J. Am. Oil Chem. Soc 1998, 75, 511–516. [Google Scholar]
- Mu, H; Xu, X; Høy, C-E. Production of specific-structured triacylglycerols by lipase-catalyzed interesterification in a laboratory-scale continuous reactor. J. Am. Oil Chem. Soc 1998, 75, 1187–1193. [Google Scholar]
- Ferreira-Dias, S; Duarte, CS; Falaschi, V; Marques, SR; Gusmao, JH; da Fonseca, MMR. Stability and Stabilization of Biocatalysts; Ballesteros, A, Plou, FJ, Iborra, JL, Halling, P, Eds.; Elsevier: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Fomuso, LB; Akoh, CC. Enzymatic modification of triolein: Incorporation of caproic and butyric acids to produce reduced-calorie structured lipids. J. Am. Oil Chem. Soc 1997, 74, 269–272. [Google Scholar]
- Xu, X; Skands, ARH; Høy, C-E; Mu, H; Balchen, S; Adler-Nissen, J. Production of specific-structured lipids by enzymatic interesterification: Elucidation of acyl migration by response surface design. J. Am. Oil Chem. Soc 1998, 75, 1179–1186. [Google Scholar]
- Miura, S; Ogawa, A; Konishi, H. A rapid method for enzymatic synthesis and purification of the structured triacylglycerol, 1,3-dilauroyl-2-oleoyl-glycerol. J. Am. Oil Chem. Soc 1999, 76, 927–931. [Google Scholar]
- Adachi, S; Okumura, K; Ota, Y; Mankura, M. Acidolysis of sardine oil by lipase to concentrate eicosapentaenoic and docosahexanoic acids in glycerides. J. Ferment. Bioeng 1993, 75, 259–264. [Google Scholar]
- Yamane, T; Suzuki, T; Hoshino, T. Increasing n-3 polyunsaturated fatty acid content of fish oil by temperature control of lipase-catalyzed acidolysis. J. Am. Oil Chem. Soc 1993, 70, 1285–1287. [Google Scholar]
- Li, Z; Ward, OP. Enzyme catalysed production of vegetable oils containing omega-3 polyunsaturated fatty acid. Biotechnol. Lett 1993, 15, 185–188. [Google Scholar]
- Huang, K-H; Akoh, CC. Lipase-catalyzed incorporation of n-3 polyunsaturated fatty acids into vegetable oils. J. Am. Oil Chem. Soc 1994, 71, 1277–1288. [Google Scholar]
- Lee, K-T; Akoh, CC. Immobilized lipase-catalyzed production of structured lipids with eicosapentaenoic acid at specific positions. J. Am. Oil Chem. Soc 1996, 73, 611–615. [Google Scholar]
- Senanayake, SPJN; Shahidi, F. Enzymatic incorporation of docosahexaenoic acid into borage oil. J. Am. Oil Chem. Soc 1999, 76, 1009–1015. [Google Scholar]
- Jennings, BH; Akoh, C. Enzymatic modification of triacylglycerols of high eicosapentaenoic and docosahexaenoic acids content to produce structured lipids. J. Am. Oil Chem. Soc 1999, 76, 1133–1137. [Google Scholar]
- Haraldsson, G; Thorarensen, A. Preparation of phospholipids highly enriched with n-3 polyunsaturated fatty acids by lipase. J. Am. Oil Chem. Soc 1999, 76, 1143–1149. [Google Scholar]
- Muderhwa, JM; Pina, M; Montet, D; Feuillard, P; Graille, J. 1–3 Regioselective enzymatic interesterification in a melted medium and a continuous reactor – valorization of palm oil. Oleagineux 1989, 44, 36–43. [Google Scholar]
- Zainal, Z; Yusoff, MSA. Enzymatic interesterification of palm stearin and palm kernel olein. J. Am. Oil Chem. Soc 1999, 76, 1003–1008. [Google Scholar]
- Bergstrom, B; Brochmann, HL. Lipases, 1st Ed, Elsevier: Amsterdam, The Netherlands, 1984.
- Quinlan, P; Moore, S. Modification of triglycerides by lipases: Process technology and its application to the production of nutritionally improved fats. INFORM 1993, 4, 580–585. [Google Scholar]
- Brady, L; Brzozowski, AM; Derewenda, ZS; Dodson, E; Dodson, G; Tolley, S; Turkenburg, JP; Christiansen, L; Huge-Jensen, B; Norskov, L; Thim, L; Menge, U. A serine protease triad forms the catalytic centre of a triacylglycerol lipase. Nature 1990, 343, 767–770. [Google Scholar]
- Klibanov, AM. Why are enzymes less active in organic solvents than in water? Trends Biotechnol 1997, 15, 97–101. [Google Scholar]
- Pleiss, J; Fischer, M; Peiker, M; Thiele, C; Schmid, RD. Lipase engineering database. Understanding and exploiting sequence–structure–function relationships. J. Mol. Catal. B: Enzym 2000, 10, 491–508. [Google Scholar]
- Balcao, VM; Paiva, AL; Malcata, FX. Review. Bioreactors with immobilized lipases: State of the art. Enzyme Microb. Technol 1996, 18, 392–416. [Google Scholar]
- Deng, HT; Xu, ZK; Huang, XJ; Wu, J; Seta, P. Adsorption and activity of Candida rugosa lipase on polypropylene hollow fiber membrane modified with phospholipid analogous polymers. Langmuir 2004, 20, 10168–10173. [Google Scholar]
- Homquist, M; Norin, M; Hult, K. The role of arginines in stabilizing the active open-lid conformation of Rhizomucor meihei lipase. Lipids 1993, 28, 721–726. [Google Scholar]
- Villeneuve, P; Muderwha, J; Graille, J; Haas, M. Customizing lipases for biocatalysis: A survey of chemical, physical and molecular biological approaches. J. Mol. Catal. B: Enzym 2000, 9, 113–148. [Google Scholar]
- Holmquist, M; Martinelle, M; Berglund, P; Clausen, IG; Patkar, S; Svendsen, A; Hult, K. Lipases from Rhizomucor miehei and Thermomyces lanuginosa: modification of the lid covering the active site alters enantioselectivity. J. Protein Chem 1993, 12, 749–757. [Google Scholar]
- Gutierrez-Ayesta, C; Carelli, AA; Ferreira, ML. Relation between lipase structures and their catalytic ability to hydrolyse triglycerides and phospholipids. Enzyme Microb. Technol 2007, 41, 35–43. [Google Scholar]
- Pleiss, J; Fisher, M; Schmid, RD. Anatomy of lipase binding sites: the scissile fatty acid binding site. Chem. Phys. Lipids 1998, 93, 67–80. [Google Scholar]
- Bottino, NR; Vandenburg, GA; Reiser, R. Resistance of certain long chain polyunsaturated fatty acids of marine oils to pancreatic lipase hydrolysis. Lipids 1967, 2, 489–493. [Google Scholar]
- Jensen, RG; Gordon, DT. Specificity of Geotrichum candidum lipase with respect to double bond position in triglycerides containing cis-octadecenoic acids. Lipids 1972, 7, 738–741. [Google Scholar]
- Lopez-Martinez, JC; Campra-Madrid, P; Ramirez-Fajardo, A; Esteban-Cerdan, L; Guil-Guerrero, JL. Screening of lipases for enzymatic concentration of gamma-linolenic acid (GLA) from seed oils. J. Food Lipids 2006, 13, 362–374. [Google Scholar]
- Hoshino, T; Yamane, T; Shimizu, S. Selective hydrolysis of fish oil by lipase to concentrate n-3 polyunsaturated fatty acid. Agric. Biol. Chem 1990, 54, 1459–1467. [Google Scholar]
- Tanaka, Y; Hirano, J; Funada, T. Concentration of docosahexaenoic acid glyceride by hydrolysis of fish oil with Candida cylindracea lipase. J. Am. Oil Chem. Soc 1992, 69, 1210–1214. [Google Scholar]
- Basheer, S; Plat, D. Enzymatic modification of sterols using sterol-specific lipase. WO patent 2001-075083, 2001. [Google Scholar]
- Weber, N; Weikamp, P; Mukherjee, KD. Cholesterol-lowering food additives: Lipase-catalyzed preparation of phytosterol and phytostanol esters. Food Res. Intl 2002, 35, 177–181. [Google Scholar]
- Bertinotti, A; Carrea, G; Ottolina, G; Riva, S. Regioselective esterification of polyhydroxylated steroids by Candida antarctica lipase B. Tetrahedron 1994, 50, 13165–13172. [Google Scholar]
- Norinobu, S; Senoo, N; Kaneko, S; Sato, F; Mankura, M. Supercritical preparation of sterol fatty esters with enzyme. Japanese patent 2002-233396, 2002. [Google Scholar]
- Stamatis, H; Sereti, V; Kolisis, FN. Studies on the enzymatic synthesis of lipophilic derivatives of natural antioxidants. J. Am. Oil Chem. Soc 1999, 76, 1505–1510. [Google Scholar]
- Jessop, PG; Ikariya, T; Noyori, R. Homogenous catalysis in supercritical fluids. Chem. Rev 1999, 99, 475–494. [Google Scholar]
- Brennecke, JF; Chateauneuf, JE. Homogenous organic reactions as mechanistic probes in supercritical fluids. Chem. Rev 1999, 99, 433–452. [Google Scholar]
- Baiker, A. Supercritical fluids in heterogenous catalysis. Chem. Rev 1999, 99, 453–474. [Google Scholar]
- Darr, JA; Poliakoff, M. New directions in inorganic and metal-organic coordination chemistry in supercritical fluids. Chem. Rev 1999, 99, 495–542. [Google Scholar]
- Kiran, E; Debenedetti, PG; Peters, CJ. Supercritical Fluids Fundamentals and Applications; Kluwer Academic: Dordrecht, The Netherlands, 2000. [Google Scholar]
- Leitner, W. Supercritical carbon dioxide as a green reaction medium for catalysis. Acc. Chem. Res 2002, 35, 746–756. [Google Scholar]
- Matsuda, T; Watanabe, K; Harada, T; Nakamura, K. Enzymatic reactions in supercritical CO2: carboxylation, asymmetric reduction and esterification. Catal. Today 2004, 96, 103–111. [Google Scholar]
- Kragl, U; Eckstein, M; Kraftzik, N. Biocatalytic reactions in ionic liquids. In Ionic Liquids in Synthesis; Wasserscheid, P, Welton, T, Eds.; Wiley-VCH: Weinheim, Germany, 2003; pp. 336–347. [Google Scholar]
- Seddon, KR. Ionic liquids for clean technology. J. Chem. Technol. Biotechnol 1997, 68, 351–356. [Google Scholar]
- Welton, T. Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem. Rev 1999, 99, 2071–2084. [Google Scholar]
- Yang, Z; Pan, W. Ionic liquids: Green solvents for nonaqueous biocatalysis. Enzyme Microb. Technol 2005, 37, 19–28. [Google Scholar]
- Chowdhury, S; Mohan, RS; Scott, JL. Reactivity in ionic liquids. Tetrahedron 2007, 63, 2363–2389. [Google Scholar]
- Freemantle, M. Ionic liquids in organic synthesis. Chem. Eng. News 2004, 82, 44–49. [Google Scholar]
- Roberts, NJ; Seago, A; Carey, JS; Freer, R; Preston, C; Lye, GJ. Lipase catalysed resolution of the Lotrafiban intermediate 2,3,4,5-tetrahydro-4-methyl-3-oxo-1H-1,4-benzodiazepine-2-acetic acid methyl ester in ionic liquids: Comparison to the industrial t-butanol process. Green Chem 2004, 6, 475–482. [Google Scholar]
- Lau, RM; Sorgedrager, MJ; Carrea, G; van Rantwijk, F; Secundo, F; Sheldon, RA. Dissolution of Candida antarctica lipase B in ionic liquids: effects on structure and activity. Green Chem 2004, 6, 483–487. [Google Scholar]
- Oliveira, JV; Oliveira, D. Kinetics of the enzymatic alcoholysis of palm kernel oil in supercritical CO2. Ind. Eng. Chem. Res 2000, 39, 4450–4454. [Google Scholar]
- Kamat, SV; Beckman, EJ; Russell, AJ. Enzyme-activity in supercritical fluids. Crit. Rev. Biotechnol 1995, 15, 41–71. [Google Scholar]
- Connstein, W; Hoyer, E; Wartenberg, H. The enzymic decomposition of fat. Chem. Ber 1902, 34, 3988–4006. [Google Scholar]
- Baumann, H; Bühler, M; Fochem, H; Hirsinger, F; Zoebelein, H; Falbe, J. Natural fats and oils - renewable raw materials for the chemical industry. Angew. Chem. Int. Ed. Engl 1988, 27, 41–62. [Google Scholar]
- Moquin, PHL; Temelli, F. Production of monoolein from oleic acid and glycerol in supercritical carbon dioxide media: a kinetic approach. J. Supercrit. Fluids 2008, 44, 40–47. [Google Scholar]
- Promožič, M; Habulin, M; Knez, Ž. Parameter optimization for enzymic hydrolysis of sunflower oil in high-pressure reactors. J. Am. Oil Chem. Soc 2003, 80, 643–646. [Google Scholar]
- Promožič, M; Habulin, M; Knez, Ž. Thermodynamic properties of the enzymatic hydrolysis of sunflower oil in high-pressure reactors. J. Am. Oil Chem. Soc 2003, 80, 785–788. [Google Scholar]
- Promožič, M; Habulin, M; Knez, Ž. Modeling of kinetics for the enzymatic hydrolysis of sunflower oil in a high-pressure reactors. J. Am. Oil Chem. Soc 2005, 82, 543–547. [Google Scholar]
- Zarevúcka, M; Vacek, M; Wimmer, Z; Stránský, K; Koutek, B; Demnerová, K. Enzymic transformations of blackcurrant oil: enrichment with γ-linolenic acid and α-linolenic acid. Chem. Listy 2003, 97, 206–213. [Google Scholar]
- Sovová, H; Zarevúcka, M. Lipase-catalysed hydrolysis of blackcurrant oil in supercritical carbon dioxide. Chem. Eng. Sci 2003, 58, 2339–2350. [Google Scholar]
- Bártlová, M; Bernášek, P; Sýkora, J; Sovová, H. HPLC in reversed phase mode: Tool for investigation of kinetics of blackcurrant seed oil lipolysis in supercritical carbon dioxide. J. Chromatogr. B 2006, 839, 80–84. [Google Scholar]
- Sýkora, J; Bernášek, P; Zarevúcka, M; Kurfürst, M; Sovová, H; Schraml, J. High-performance liquid chromatography with nuclear magnetic resonance detection - A method for quantification of α- and γ-linolenic acids in their mixtures with free fatty acids. J. Chromatogr. A 2007, 1139, 152–155. [Google Scholar]
- Kosugi, Y; Azuma, N. Synthesis of triacylglycerol from polyunsaturated fatty acid by immobilized lipase. J. Am. Oil Chem. Soc 1994, 71, 1397–1403. [Google Scholar]
- Cerdán, LE; Medina, AR; Giménez Giménez, A; Ibánez González, MJ; Molina Grima, E. Synthesis of PUFA enriched triglycerides by lipase catalyzed esterification. J. Am. Oil Chem. Soc 1998, 75, 1329–1337. [Google Scholar]
- Medina, AR; Cerdán, LE; Giménez Giménez, A; Camacho Páez, B; Ibánez González, MJ; Molina Grima, E. Lipase-catalyzed esterification of glycerol and polyunsaturated fatty acids from fish and microalgae oils. J. Biotechnol 1999, 70, 379–391. [Google Scholar]
- Akoh, CC; Moussata, CO. Lipase-catalyzed modification of borage oil: Incorporation of capric and eicosapentaenoic acids to form structured lipids. J. Am. Oil Chem. Soc 1998, 75, 697–701. [Google Scholar]
- McKenna, MC; Hubbard, VS; Aeri, JG. Linoleic acid absorption for lipid supplements in patients with cystic fibrosis with pancreatic insufficiency and in control subjects. J. Pediatr. Gastroenterol. Nutr 1985, 4, 45–48. [Google Scholar]
- Kawashima, A; Shimada, Y; Nagao, T; Ohara, A; Matsuhisa, T; Sugihara, A; Tominaga, Y. Production of structured TAG rich in 1,3- dicapryloyl-2-c-linolenoyl glycerol from borage oil. J. Am. Oil Chem. Soc 2002, 79, 871–877. [Google Scholar]
- Kawashima, A; Shimada, Y; Yamamoto, M; Sugihara, A; Nagao, T; Komemushi, S; Tominaga, Y. Enzymatic synthesis of high-purity structured lipids with caprylic acid at 1,3-positions and polyunsaturated fatty acid at 2-position. J. Am. Oil Chem. Soc 2001, 78, 611–616. [Google Scholar]
- Senanayake, SPJN; Shahidi, F. Chemical and stability of structured lipids from borage (Borago officinalis L.) and evening primrose (Oenothera biennis L.) oils. J. Food Sci 2002, 67, 2038–2045. [Google Scholar]
- Shimada, Y; Suenaga, M; Sugihara, A; Seiichi, S; Tominaga, Y. Continuous production of structured lipids containing clinolenic and caprylic acids by immobilizedRhizopus delemar lipase. J. Am. Oil Chem. Soc 1996, 76, 189–193. [Google Scholar]
- Hamam, F; Shahidi, F. Synthesis of structured lipids via acidolysis of docosahexaenoic acid single cell oil (DHASCO) with capric acid. J. Agric. Food Chem 2004, 52, 2900–2906. [Google Scholar]
- Hamam, F; Shahidi, F. Enzymatic acidolysis of arachidonic acid single cell oil (ARASCO) with capric acid. J. Am. Oil Chem. Soc 2004, 81, 887–892. [Google Scholar]
- Hamam, F; Shahidi, F. Enzymatic incorporation of capric acid into a single cell oil rich in docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA). Food Chem 2005, 91, 583–591. [Google Scholar]
- Iwasaki, Y; Han, JJ; Narita, M; Rosu, R; Yamane, T. Enzymatic synthesis of structured lipids from single cell oil of high docosahexaenoic acid content. J. Am. Oil Chem. Soc 1999, 76, 563–569. [Google Scholar]
- Yang, T; Xu, X; Li, L. Comparison of linoleic and conjugated linoleic acids in enzymatic acidolysis of tristearin. J. Food Lipids 2001, 8, 149–161. [Google Scholar]
- Ronne, TH; Pederson, LS; Xu, X. Triglyceride selectivity of immobilized Thermomyces lanuginose lipase in interesterification. J. Am. Oil Chem. Soc 2005, 82, 737–743. [Google Scholar]
- Tsuzuki, W. Acidolysis between triolein and short-chain fatty acids by lipase in organic solvents. Biosci. Biotechnol. Biochem 2005, 69, 1256–1261. [Google Scholar]
- Paez, BC; Medina, AR; Rubio, FC; Cerdan, LE; Grima, EM. Kinetics of lipase-catalyzed interesterification of triolein and caprylic acid to produce structured lipids. J. Chem. Technol. Biotechnol 2003, 78, 461–470. [Google Scholar]
- Sellappan, S; Akoh, CC. Synthesis of structured lipids by transesterification of trilinolein catalyzed by Lipozyme IM60. J. Agric. Food Chem 2001, 49, 1–9. [Google Scholar]
- Fomus, LB; Akoh, CC. Structured lipids: Lipase-catalyzed interesterification of tricaproin and trilinolein. J. Am. Oil Chem. Soc 1998, 75, 405–410. [Google Scholar]
- Hamam, F; Shahidi, F. Incorporation of selected long-chain fatty acids into trilinolein and trilinolenin. Food Chem 2008, 106, 33–39. [Google Scholar]
- Hamam, F; Shahidi, F. Enzymatic incorporation of selected long-chain fatty acids into triolein. J. Am. Oil Chem. Soc 2007, 84, 533–541. [Google Scholar]
- Hamam, F; Shahidi, F. Acidolysis of tristearin with selected long chain fatty acids. J. Agric. Food Chem 2007, 55, 1955–1960. [Google Scholar]
- Lee, K; Akoh, C. Characterization of enzymatically synthesized structured lipids containing eicosapentaenoic, docosahexaenoic, and caprylic acids. J. Am. Oil Chem. Soc 1998, 75, 495–499. [Google Scholar]
- McNeill, GP; Ackman, RG; Moore, SR. Lipase-catalyzed enrichment of long chain polyunsaturated fatty acids. J. Am. Oil Chem. Soc 1996, 73, 1403–1414. [Google Scholar]
- Schmid, U; Bornscheuer, UT; Soumanou, MM; Mc- Neill, GP; Schmid, RD. Optimization of the reaction conditions in the lipase-catalyzed synthesis of structured triglycerides. J. Am. Oil Chem. Soc 1998, 75, 1527–1531. [Google Scholar]
- Soumanou, MM; Bornscheuer, UT; Schmid, RD. Two step enzymatic reaction for synthesis of pure structured triacylglycerides. J. Am. Oil Chem. Soc 1998, 75, 703–710. [Google Scholar]
- Tsujisaka, Y; Okumura, S; Iwai, M. Glyceride synthesis by four kinds of microbial lipase. Biochim. Biophys. Acta 1977, 489, 415–422. [Google Scholar]
- Okumura, S; Iwai, M; Tsujisaka, Y. Synthesis of various kinds of esters by four microbial lipases. Biochim. Biophys. Acta 1979, 575, 156–165. [Google Scholar]
- Liu, K-J; Chang, H-M; Liu, K-M. Enzymatic synthesis of cocoa butter analog through interesterification of lard and tristearin in supercritical carbon dioxide by lipase. Food Chem 2007, 100, 1303–1311. [Google Scholar]
- Sakurai, H; Pokorný, J. The development and application of novel vegetable oils tailor-made for specific human dietary needs. Eur. J. Lipid Sci. Technol 2003, 105, 769–778. [Google Scholar]
- Jessop, P; Leitner, W. Chemical synthesis using supercritical fluids. Wiley-VCH: Weinheim, 1999. [Google Scholar]
- King, JW; Snyder, JM; Frykman, H; Neese, A. Sterol ester production using lipase-catalyzed reactions in supercritical carbon dioxide. Eur. Food Res. Technol 2001, 212, 566–569. [Google Scholar]
- Gunnlaugsdottir, H; Järemo, M; Sivik, B. Process parameters influencing ethanolysis of cod liver oil in supercritical carbon dioxide. J. Supercrit. Fluids 1998, 12, 85–93. [Google Scholar]
- Overmeyer, A; Schrader-Lippelt, S; Kasche, V; Brunner, G. Lipase-catalysed kinetic resolution of racemates at temperatures from 40 °C to 160 °C in supercritical CO2. Biotechnol. Lett 1999, 21, 65–69. [Google Scholar]
- Habulin, M; Krmelj, V; Knez, Ž. Synthesis of oleic acid esters catalyzed by immobilized lipase. J. Agric. Food Chem 1996, 44, 338–342. [Google Scholar]
- Knez, Ž; Habulin, M. Compressed gases as alternative enzymatic-reaction solvents: A short review. J. Supercrit. Fluids 2002, 23, 29–42. [Google Scholar]
- Leitner, W. Die “bessere Lösung”? Chemische Synthese in überkritischem Kohlendioxid. Chem. Unserer Zeit 2003, 37, 32–38. [Google Scholar]
- King, J; Jackson, M; Temelli, F. Development of industrially-useful synthetic processes in supercritical carbon dioxide. In I Fluidi Supercritici e le Lorno Applicazioni; Kikic, I, Alessi, P, Eds.; Trieste: Italy, 1995; pp. 19–26. [Google Scholar]
- Bornscheuer, U; Kazlauskas, R. Hydrolases in Organic Synthesis: Regio- and Stereoselective Biotransformations; Wiley-VCH: Weinheim, Germany, 1999. [Google Scholar]
- Snyder, J; King, J; Jackson, M. Fat content for nutritional labeling by supercritical fluid extraction and an on-line lipase catalyzed reaction. J. Chromatogr. A 1996, 750, 201–207. [Google Scholar]
- Anderson, M; King, J; Blomberg, L. Synthesis of fatty alcohol mixtures from oleochemicals in supercritical fluids. Green Chem 2000, 2, 230–234. [Google Scholar]
- Nakaya, H; Nakamura, K; Miyawaki, O. Lipase-catalyzed esterification of stearic acid with ethanol, and hydrolysis of ethyl stearate, near the critical point in supercritical carbon dioxide. J. Am. Oil Chem. Soc 2002, 79, 23–27. [Google Scholar]
- Ikushima, Y; Saito, N; Arai, M; Blanch, HW. Activation of lipase triggered by interactions with supercritical carbon dioxide in the near-critical region. J. Phys. Chem 1995, 99, 8941–8944. [Google Scholar]
- Srivastava, S; Madras, G; Modak, J. Esterification of myristic acid in supercritical carbon dioxide. J. Supercrit. Fluids 2003, 27, 55–64. [Google Scholar]
- Gunnlaugsdottir, H; Wannerberger, K; Sivik, B. Alcoholysis and glyceride synthesis with immobilized lipase on controlled-pore glass of varying hydrophobicity in supercritical carbon dioxide. Enzyme Microb. Technol 1998, 22, 360–367. [Google Scholar]
- Laudani, CG; Habulin, M; Primožič, M; Knez, Ž; Della Porta, G; Reverchon, E. Optimisation of n-octyl oleate enzymatic synthesis over Rhizomucor miehei lipase. Bioprocess Biosyst. Eng 2006, 29, 119–127. [Google Scholar]
- Laudani, CG; Habulin, M; Knez, Ž; Della Porta, G; Reverchon, E. Immobilized lipase-mediated long-chain fatty acid esterification in dense carbon dioxide: bench-scale packed-bed reactor study. J. Supercrit. Fluids 2007, 41, 74–81. [Google Scholar]
- Pomier, E; Galy, J; Paolucci-Jeanjean, D; Pina, M; Sarrade, S; Rios, GM. A new reactor design combining enzyme, membrane and SC CO2: Application to castor oil modification. J. Membrane Sci 2005, 249, 127–132. [Google Scholar]
- Liu, K-J; Liu, K-M; Chang, H-M. Biocatalytic synthesis of palmitoyl vanillylamide in supercritical carbon dioxide through amidation of vanillylamide hydrochloride and palmitic anhydride by lipase. Food Chem 2007, 102, 1020–1026. [Google Scholar]
- Hakoda, M; Shiragami, N; Enomoto, A; Nakamura, K. Effect of moisture on enzymatic reaction in supercritical carbon dioxide. Bioprocess Biosyst. Eng 2002, 24, 355–361. [Google Scholar]
- Hlavsová, K; Wimmer, Z; Xanthakis, E; Bernášek, P; Sovová, H; Zarevúcka, M. Lipase activity enhancement by SC-CO2 treatment. Z. Naturforsch 2008, 63B, 779–784. [Google Scholar]
- Eisenmenger, M; Dunford, NT; Eller, F; Tailor, S; Martinez, J. Pilot-scale supercritical carbon dioxide extraction and fractionation of wheat germ oil. J. Am. Oil Chem. Soc 2006, 83, 863–868. [Google Scholar]
- Lau, HLN; Choo, YM; Ma, AN; Chuah, CH. Quality of residual oil from palm-pressed mesocarp fiber (Elaeis guineensis) using supercritical CO2 with and without ethanol. J. Am. Oil Chem. Soc 2006, 83, 893–898. [Google Scholar]
- Üstündag, ÖG; Temelli, F. Column fractionation of canola oil deodorizer distillate using supercritical carbon dioxide. J. Am. Oil Chem. Soc 2007, 84, 953–961. [Google Scholar]
- Nagesha, GK; Manohar, B; Sankar, KU. Enzymatic esterification of free fatty acids of hydrolyzed soy deodorizer distillate in supercritical carbon dioxide. J. Supercrit. Fluids 2004, 32, 137–145. [Google Scholar]
- Frykman, HB; Snyder, JM; King, JW. Screening catalytic lipase activities with an analytical supercritical fluid extractor. J. Am. Oil Chem. Soc 1998, 75, 517–520. [Google Scholar]
- Hernández, FJ; de los Ríos, AP; Gómez, D; Rubio, M; Víllora, G. A new recirculating enzymatic membrane reactor for ester synthesis in ionic liquid/supercritical carbon dioxide biphasic systems. Appl. Catal. B: Environ 2006, 67, 121–126. [Google Scholar]
- Gumí, T; Paolucci-Jeanjean, M; Belleville, M-P; Rios, GM. Enzymatic membrane reactor involving a hybrid membrane in supercritical carbon dioxide. J. Membrane Sci 2007, 297, 98–103. [Google Scholar]
- Wang, L; Weller, CL. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Technol 2006, 17, 300–312. [Google Scholar]
- Ghanem, A. Trends in lipase-catalyzed asymmetric access to enantiomerically pure/enriched compounds. Tetrahedron 2007, 63, 1721–1754. [Google Scholar]
- Jurček, O; Wimmerová, M; Wimmer, Z. Selected chiral alcohols: Enzymic resolution and reduction of convenient substrates. Coord. Chem. Rev 2008, 252, 767–781. [Google Scholar]


© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/). This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zarevúcka, M.; Wimmer, Z. Plant Products for Pharmacology: Application of Enzymes in Their Transformations. Int. J. Mol. Sci. 2008, 9, 2447-2473. https://doi.org/10.3390/ijms9122447
Zarevúcka M, Wimmer Z. Plant Products for Pharmacology: Application of Enzymes in Their Transformations. International Journal of Molecular Sciences. 2008; 9(12):2447-2473. https://doi.org/10.3390/ijms9122447
Chicago/Turabian StyleZarevúcka, Marie, and Zdeněk Wimmer. 2008. "Plant Products for Pharmacology: Application of Enzymes in Their Transformations" International Journal of Molecular Sciences 9, no. 12: 2447-2473. https://doi.org/10.3390/ijms9122447
APA StyleZarevúcka, M., & Wimmer, Z. (2008). Plant Products for Pharmacology: Application of Enzymes in Their Transformations. International Journal of Molecular Sciences, 9(12), 2447-2473. https://doi.org/10.3390/ijms9122447




