Dietary Polyphenols and Their Biological Significance
Abstract
:1. Introduction
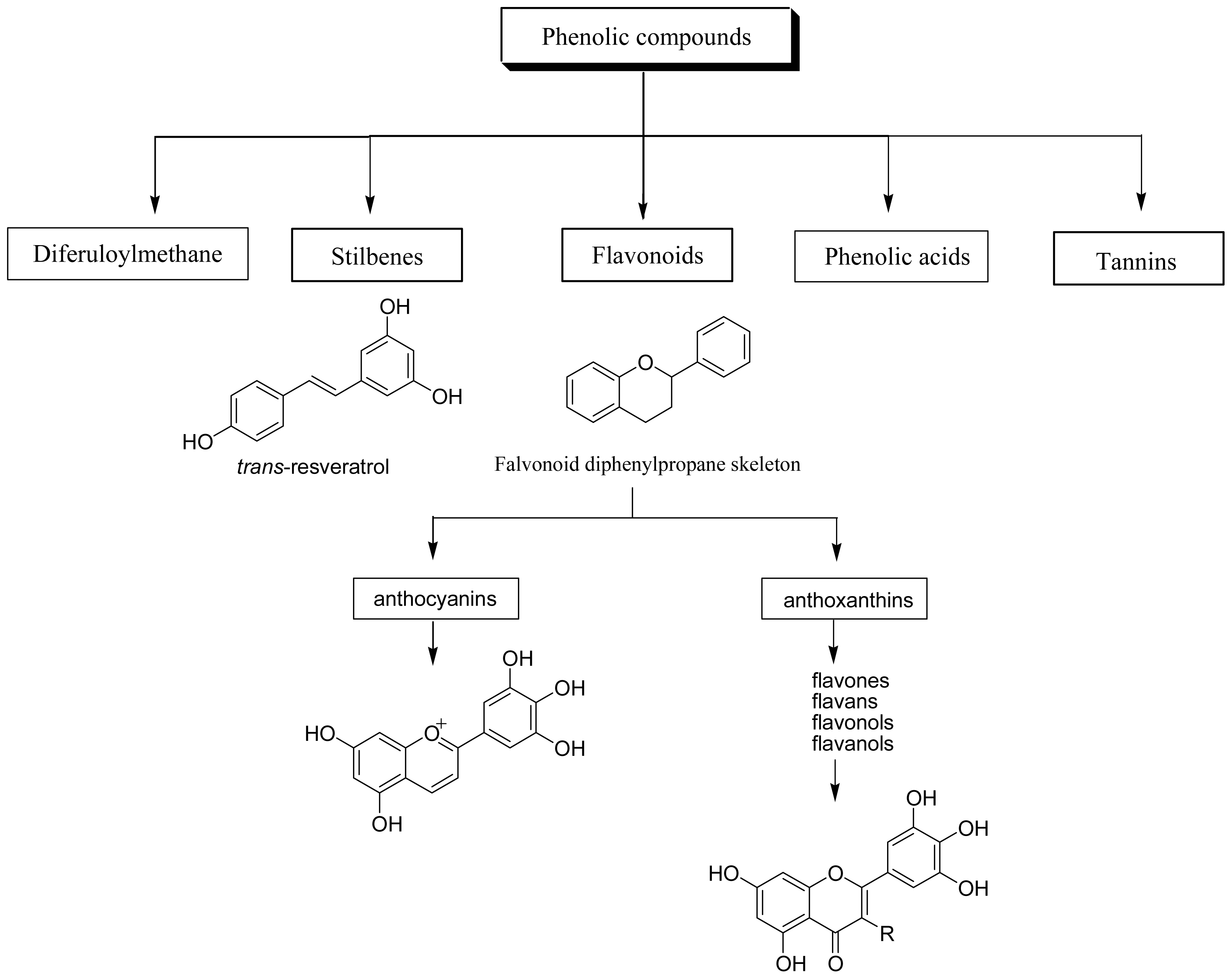
2. Classification and occurrence of dietary polyphenols
2.1 Phenolic acids
2.2 Flavonoids
2.3 Stilbenes
2.4 Tannins
2.5 Diferuloylmethanes
3. Bioactivities of dietary polyphenols
3.1 Antioxidant and free radical scavenging properties
3.2. Anti-atherosclerosis and cardioprotection
3.3 Neuroprotective effects on anti-aging and neurodegenerative diseases
3.4 Anti-inflammatory properties
3.5 Antimutagenic/anticarcinogenic properties
3.6 Maintenance of gastrointestinal health and effects on digestive enzymes
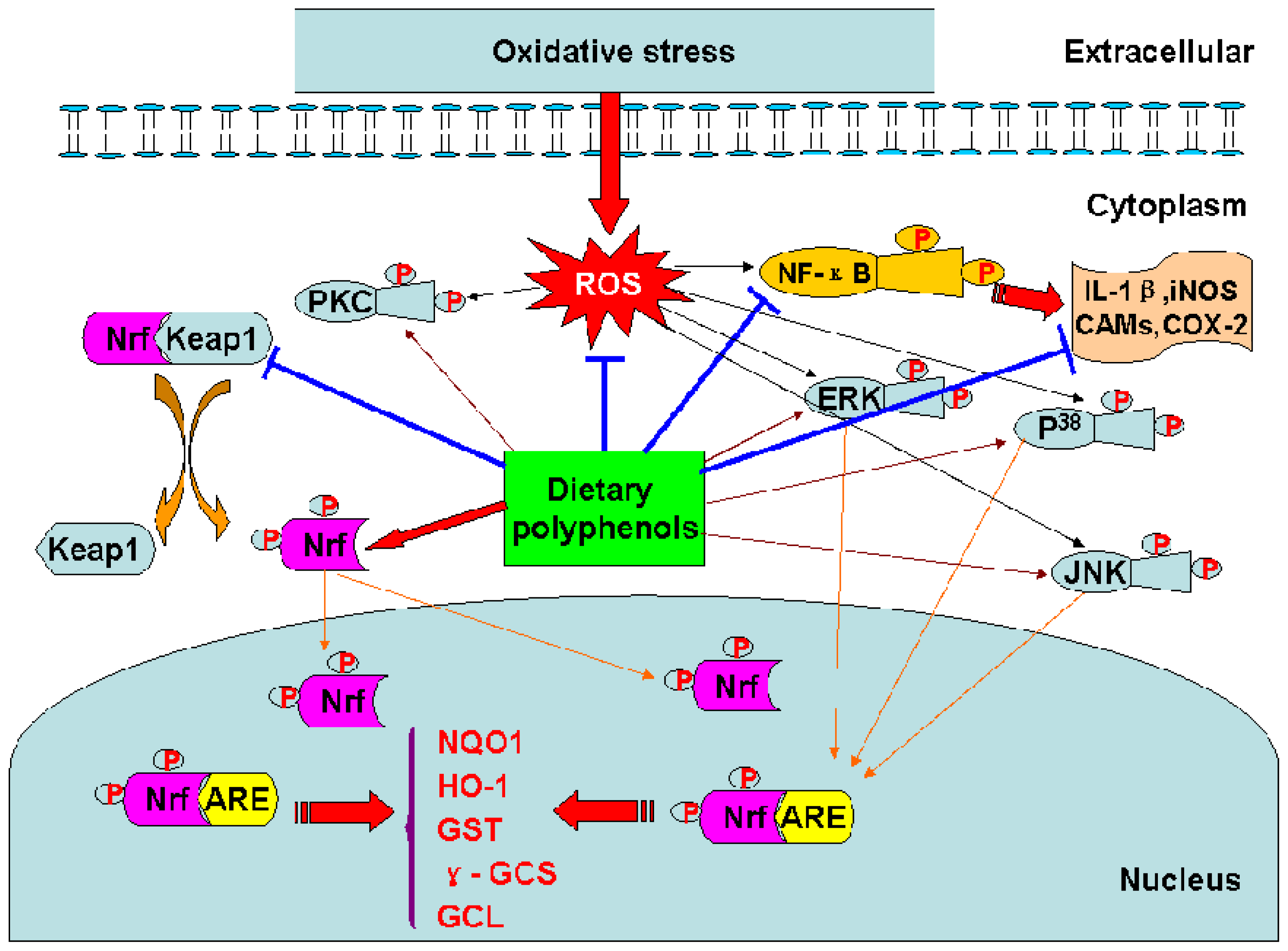
3.7 Modulation of signal transduction pathways
3.8 Improvement of endothelium functions
3.9 Protective effect on immune cell functions
3.10 Antiallergic activity
3.11 Antidiabetic effects
3.12 Regulation of cell cycle progression
3.13 Modulation of hormonal effects and contraceptive activity
3.14 Effect in the treatment of chronic obstructive pulmonary disease (COPD)
3.15 Other bioactive effects
4. Prooxidant activity and cellular effects of the phenoxyl radicals of dietary polyphenols
5. Bioavailability of dietary polyphenols
6. Conclusions

| Class and subclass | Dietary polyphenol | Foods or beverages | Ref |
|---|---|---|---|
| Flavonoids | |||
| Anthocyanidins | Cyanidin 3-galactoside Cyanidin 3-glucoside Cyanidin 3-arabinoside Cyanidin 3-xyloside Malvidin Delphinidin Pelargonidin | Fruits: blackberries,black currant,blueberries, black grape, elderberries,strawberries, cherries, plums, cranberry, pomegranate juice, raspberry Others: red wine | 6 14 15 16 |
| Anthoxanthins | |||
| Flavonols | Myricetin Fisetin Quercetin Kaempferol Isorhamnetin | Vegetables: capers, celery, chives, onions, red onions, dock leaves, fennel, hot peppers,cherry tomatoes, spinach, sweet potato leaves, lettuce, celery, broccoli, Hartwort leaves, kale Cereal: buckwheat, beans(green/yellow) Fruits: apples, apricots, grapes, plums, bilberries, blackberries, blueberries, cranberries, olive elderberries, currants, cherries, black currant juice, apple juice, ginkgo biloba Spices and herbs: dill weed Others: red wine, tea (green, black), tea (black beverage), cocoa powder, turnip (green), endive, leek | 7 17 14 |
| Flavanones | Naringenin Eriodictyol Hesperetin | Citrus fruits and juices: lemon, lemon juice, lime juice, orange, orange juice, grapefruit, tangerine juice Spices and herbs: peppermint | 18 19 20 |
| Flavones | Apigenin Luteolin | Fruits: celery, olives Vegetables: hot peppers, celery hearts, fresh parsley Spices and herbs: oregano, rosemary, dry parsley, thyme | 14 21 22 |
| Flavanols (Flavan-3-ols) | (+)-Catechin (−)-Epicatechin (−)-Epicatechin 3-gallate Morin (−)-Epigallocatechin (−)-Epigallocatechin-3-gallate (+)-Gallocatechin Procyanidins Prodelphinidins | Fruits: apples, apricots, grapes, peaches, nectarines, pears,plums, raisins, raspberries, cherries, blackberries, blueberries, cranberries Others: red wine, tea (green, black), chocolate (dark, milk), white wine, cocoa | 23 24 |
| Isoflavones (Flavans) | Genistein Daidzein Equol | Fruits: grape seed/skin Others: soybean, soy nuts, soy flour/bread, tofu, miso, soy milk, tofu yogurt, soy cheese/sauce/hot dog | 25 |
| Flavonoid glycoside | Rutin Hesperidin Naringin | Fruits: lemon, orange, orange juice, grapefruit, tangerine juice | 26 |
| Phenolic acids | 27 | ||
| Hydroxycinnamic acids | Caffeic acid Chlorogenic acid Ferulic acid Neochlorogenic acid P-coumaric acid Sinapic acid Caftaric acids | Fruits: bluberry, cranberry, pear, cherry(sweet), apple, orange, grapefruit, cherry juice,apple juice, lemon, peach, Vegetables: potato, lettuce, spinach Others: coffee beans, tea, coffee, cider | |
| Hydroxybenzoic acids | Ellagic acid Gallic acid Corilagin | Fruits: strawberry, raspberry grape juice( black/green), longan seed, pomegranate juice | 28 29 |
| Trihydroxy-stilbenes | Resveratrol Trans-resveratrol | Fruits: grapes, peanuts, Others: red wine | 30 31 |
| Tannins | Catechin polymers Epicatechin polymers Ellagitannins Proanthocyanidins Casuarictin Sanguin H6 Tannic acids | Fruits: grape (dark/light) seed/skin, apple juice, strawberries, longan, raspberries, pomegranate, walnuts, muscadine grape, muscadine grape, peach, blackberry (juices/jams/jellies), olive, plum, Vegetables: chick pea, black-eyed peas, lentils, Cereal: haricot bean, Others: red wine, white wine, cocoa, chocolate, oak-aged red wine, tea, cider, tea, coffee, immature fruits | 14 29 32 |
| Diferuloylmethane | Curcumin | herbal remedy, dietary spice turmeric | 33 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Epigallocatechin, EGCG, ECG | Inhibiting lipoxygenase and cyclooxygenase | In human colon mucosa and colon tumor tissues | In vitro | 34 |
| EGCG ECG | Inducing ARE-mediated gene expression through the activation of MAPK proteins (ERK, JNK and p38) | In HepG2-ARE-C8 cell | In vitro | 10 |
| Hydroxytyrosol | Increasing CAT and SOD activities | In rats fed a cholesterol-rich diet | In vivo | 35 |
| Inhibiting the activities of 12-lipoxygenase and 5-lipoxygenase Reducing leukotriene B4 production | In rat platelets and rat polymorphonuclear leukocytes (PMNL) | In vitro | 36 | |
| Catechin Proanthocyanidin B4 | Increasing CAT, glutathione S-transferase (GST) and SOD activities Elevating cellular GSH content | In cardiac H9C2 cells | In vitro | 37 |
| Curcumin | Inhibiting CYP1A2, CYP3A4, CYP2B6, CYP2D6, and CYP2C9 | The plasmids with human cytochrome P450 NADPH reductase | In vitro | 38 |
| Inhibiting mitochondrial proton F0F1-ATPase/ATP synthase | Rat brain F0F1-ATPase | In vitro | 39 | |
| Increasing the expression of GSTP1 by activing ARE and Nrf2 | In HepG2 cells | In vivo | 40 | |
| Increasing CAT, SOD activity and heat shock proteins 70 expression Decreasing the activity of iNOS Decreasing malondialdehyde (MDA), NO(2)(−) + NO(3)(−) and myeloperoxidase (MPO) level and serum transaminase concentration | In rat model | In vivo | 8 | |
| Kaempferol-3-Ogalactoside | Inhibiting human recombinant synovial phospholipase A2 (PLA2) | In mice | In vivo | 41 |
| EGCG, Quercetin, Kaempferol Morin, Apigenin, Daidzein, ECG | Inhibiting mitochondrial proton F0F1-ATPase/ATP synthase | Rat brain F0F1-ATPase | In vitro | 39 |
| Ellagic acid Gallic acid Corilagin | Inhibiting tyrosinase, xanthine oxidase, and the formation of superoxide radical | In substrate of L-tyrosine | In vitro | 29 |
| Dihydrocaffeic acid | Enhancing eNOS activity and protein expression Scavenging intracellular ROS | In human EA.hy926 endothelial cells | In vitro | 42 |
| Caffeic acid (+)-catechin | Inhibiting peroxynitrite-mediated oxidation of dopamine | In dopamine | In vitro | 43 |
| Quercetin | Preventing lactate dehydrogenase (LDH ) leakage Increasing SOD, CAT, GSH, GPx, and GR activity | In mouse liver | In vivo | 9 |
| Decreasing MDA and lipoperoxidation Increasing Cu/Zn SOD and GPx mRNA | In HepG2 cells | In vitro | 44 | |
| Increasing the expression and activity of NADPH:quinone oxidoreductase-1( NQO1) | In the MCF-7 human breast carcinoma cellse | In vitro | 45 | |
| Enhancing γ-glutamylcysteine synthetase (γ-GCS) | In HepG2 cells | In vitro | 47 | |
| Enhancing the ARE binding activity and Nrf2-mediated transcription activity Upregulating and stabilizing Nrf2 Reducing the level of Keap1 protein | In HepG2 cells | In vitro | 48 | |
| Resveratrol | Inhibiting O-acetyltransferase and sulfotransferase activities Preventing the oxidative DNA damage | In male Wistar rats treated with potassium bromate | In vivo | 49 |
| Inhibiting the production of H2O2 and MPO activity Increasing GSH levels and SOD activities Decreasing the levels of MPO and oxidized GR | In mouse skin | Ex vivo | 50 | |
| Reducing PhIP-DNA-adduct formation by O-acetyltransferase and sulfotransferase catalysis | In primary cultures of human mammary epithelial cells | In vitro | 51 | |
| Inhibiting the expression and activity of CYP 1A1/1A2 | In microsomes and intact HepG2 cells | In vitro | 52 | |
| Inhibiting mitochondrial proton F0F1-ATPase/ATP synthase | Rat brain and liver F0F1-ATPase | In vitro | 39 | |
| Suppressing CYP1A1 and IL-1 β transcription by blocking aryl hydrocarbon receptor | Ex vivo In vivo | 53 | ||
| (−)-Epicatechin Procyanidin EGCG, ECG | Inhibiting recombinant human platelet 12-lipoxygenase and 15-lipoxygenase | In rabbit smooth muscle cells and in J774A.1 cells | In vitro | 54 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Resveratrol | Suppresing the expression and activity of COX-2 Suppresing activation of AP-1 | In human mammary epithelial cells | In vitro | 55 |
| Inhibiting the activity and expression of MMP-9 | In U937 cells | In vitro | 56 | |
| Enhancing myocardial angiogenesis by induction of VEGF, thioredoxin-1 (Trx-1), and HO-1 | In male Sprague Dawley rats | In vivo | 57 | |
| Inhibiting the expression and binding activity of the monocyte chemotactic protein-1 (MCP-1) receptor, CC-chemokine receptor-2 (CCR2) | on THP-1 monocytes | In vitro | 58 | |
| Increasing NO and NOS levels Increasing intracellular cyclc GMP (cGMP) level and decreasing atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP) levels | In cultured rat cardiac fibroblasts | In vitro | 59 | |
| (−)-Epicatechin | Inhibiting 7β-OH-cholesterol formation | In endothelial cells | In vitro | 60 |
| Hydroxytyrosol | Preventing platelet aggregation and eicosanoid formation Inhibiting thromboxane B2 production | In platelet rich plasma | In vitro | 61 |
| Inhibit thromboxane B2 production | In patients with uncomplicated type I diabetes | In vivo | 62 | |
| Hydroxytyrosol Oleuropein Caffeic acid | Inhibiting leukotriene B4 generation Inhibiting 5-lipoxygenase | In rat peritoneal leukocytes | In vitro | 63 |
| Oleuropein Hydroxytyrosol Resveratrol | Reducing monocytoid cell adhesion to stimulated endothelium Decreasing VCAM-1 mRNA and protein | In human umbilical vein endothelial cells (HUVECs) | In vitro | 64 |
| Oleuropein | Decreasing creatine kinase and GSH release | In the isolated rat heart | Ex vivo | 65 |
| Quercetin Kaempferol Apigenin | Upregulating the expression of serum HDL-associated PON-1 | In the HuH7 human hepatoma cell line | In vitro | 66 |
| Inducing interferon-gamma (IFN-γ) gene expression Downregulating IL-4 gene expression | In peripheral blood mononuclear cells | In vitro | 67 | |
| Increasing the intracellular GSH and activating γ-GCS heavy subunit (GCS(h)) promoter | In COS-1 cells | In vitro | 68 | |
| EGCG and ECG | Inhibiting rat VSMCs adhesion on collagen and laminin Interference with VSMC’s integrin β1 receptor and binding to extracellular matrix (ECM) proteins | In rat VSMCs | In vitro | 69 |
| Genistein | Decreasing hydroxyproline concentrations Suppressing the progression of myocardial fibrosis | In Long-Evans Tokushima Otsuka non-diabetic rats | In vivo | 70 |
| Genistein Daidzein | Incorporating into LDLs, increasing their oxidation resistance and antiproliferative efficacy | In cultured U937 cells | Ex vivo | 71 |
| Procyanidins | Decreasing leukotriene-prostacyclin ratio in plasma | In humans and human aortic endothelial cells | In vivo In vitro | 72 |
| Proanthocyanidin | Inhibiting CD36 mRNA expression | In peripheral blood mononuclear cell | In vitro | 73 |
| Proanthocyanidin | Reducing cardiomyocyte apoptosis by inhibiting ischemia–reperfusion-induced activation of JNK-1 and c-Jun | In Male Sprague Dawley rats | In vivo | 74 |
| Hydroxytyrosol | Lowering serum TC and LDL-C Slowing the lipid peroxidation process | In rats fed a cholesterol-rich diet | In vivo | 35 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Hydroxytyrosol | Attenuating Fe2+-and NO-induced cytotoxicity Increasing cellular ATP Reducing lipid peroxidation Hyperpolarizing basal mitochondrial membrane potential | In murine-dissociated brain cells and mice | In vitro Ex vivo | 12 |
| Resveratrol | Stimulating AMP kinase activity | In Neuro2a cells and primary neurons | In vitro | 75 |
| Preventing fibrosis, NF-κB activation and TGF-β increases induced by chronic CCl(4) treatment | In rats | In vivo | 76 | |
| Activating the phosphorylation of PKC Secreting transthyretin to prevent Aβ aggregation | In cultured rat hippocampal cells | In vitro | 77 | |
| Protecting dopaminergic neurons Activating sirtuin family of NAD-dependent histone deacetylases | In organotypic midbrain slice culture | In vitro | 78 | |
| EGCG ECG Myricetin | Inhibiting IL-6, IL-8, VEGF and prostaglandin E2 (PGE2) production Attenuating the expression of COX-2 and activation of NF-κB Inducing the expression of MAPK phosphatase-1 Suppressing the phosphorylation of MAPK (p38 and JNK) | In human astrocytoma U373MG cells | In vitro | 79 |
| Attenuating disruption of mitochondrial membrane potential and release of cytochrome c Decreasing the activities of caspase-9 and caspase-3 and increase in the Bax to Bcl-2 ratio | In rat PC12 cells | In vitro | 115 | |
| Stimulating the deacetylase activity of recombinant SIRT1 protein | In human HT29 cells | In vitro | 80 | |
| Increasing the activities of PKC and ERK1/2 Decreasing the expression of Bax, Bad, and Mdm2 Increasing the expression of Bcl-2, Bcl-w, and Bcl-xL | In human neuroblastoma SH-SY5Y cell | In vitro | 81 | |
| Catechin Quercetin Genestein Naringenin | Attenuating the apoptotic injury induced N-methyl-4-phenyl-1,2,3,6-tetrahydropyridinium hydrochloride (MPP+) | In mesencephalic dopamine neurones | In vivo | 82 |
| Epicatechin Kaempferol | Protecting neurons from oxLDL-induced apoptosis by inhibiting the activation of JNK, c-Jun and caspase-3 | In cultured primary neurons | In vitro | 83 |
| Curcumin | Disrupting existing plaques and restoring distorted neurites Crossing the blood-brain barrier and labels senile plaques and cerebrovascular amyloid angiopathy | In an Alzheimer mouse model In APPswe/PS1dE9 mice | In vivo | 84 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Procyanidins | Inhibiting transcription and secretion of IL-1β | In peripheral blood mononuclear cells | In vitro | 85 |
| EGCG ECG | Inducing apoptosis by activating caspases 3, 8, and 9 | In Isolated peripheral blood monocytes | In vitro | 86 |
| Downregulating CD11b expression Attenuating adhesion and migration of peripheral blood CD8+T cells | In peripheral blood CD8+ T cells | In vitro | 87 | |
| Resveratrol | Inhibiting stimulation of caspase-3 and cleavage of PARP induced by IL-1β | In human articular chondrocytes | In vitro | 88 |
| Suppressing the expression of iNOS mRNA and protein by inhibiting the activation of NF-κB Inhibiting NO generation | In RAW 264.7 cells | In vitro | 89 | |
| Upregulating MAP kinase phosphatase-5 | In prostate cells | In vitro | 90 | |
| Apigenin Luteolin Quercetin | Blocking the expression of intercellular adhesion molecule-1 (ICAM-1), VCAM-1, and E-selectin Inhibiting prostaglandin synthesis and IL-6, 8 production | In human endothelial cells | In vitro | 91 |
| Inhibiting the upregulation of THP-1 adhesion and VCAM-1 expression Inhibiting the activity of the NF-κB | In HUVECs | In vitro | 92 | |
| Inhibiting NO production and iNOS protein expression | In NR8383 macrophages | In vitro | 93 | |
| Anthocyanins Hydroxy-cinnamic acids | Localizing into endothelial cells Reducing the upregulation of IL-8, MCP-1, and ICAM-1 | In human microvascular endothelial cells | In vitro | 94 |
| Curcumin | Decreasing MPO activity and TNF-α on chronic colitis Reducing nitrites levels and the activation of p38 MAPK Downregulating COX-2 and iNOS expression | In rats | In vivo | 95 |
| Upregulating MAP kinase phosphatase-5 | In prostate cells | In vitro | 90 | |
| Suppressing the induction of COX-2 and iNOS Inhibiting the expression of ICAM-1 and MCP-1 Suppressing the Janus kinase (JAK)-STAT via activation of Src homology 2 domain-containing protein tyrosine phosphatases (SHP-2 ) | In both rat primary microglia and murine BV2 microglial cells | In vitro | 97 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Hydroxytyrosol | Inhibiting cell proliferation Inducing apoptosis by arresting the cells in the G0/G1 phase with a concomitant decrease in the cell percentage in the S and G2/M phases | In human promyelocytic leukaemia cells HL60 | In vitro | 98 |
| Resveratrol | Inhibiting cell proliferation and downregulating telomerase activity | In human colon tumor cells | In vitro | 99 |
| Inducing apoptosis mediated by p53-dependent pathway | In HepG2 cells | In vitro | 100 | |
| Inhibiting cell proliferation by interfering with an estrogen receptor-α (ERα)-associated PI3K pathway | In estrogen-responsive MCF-7 human breast cancer cells | In vitro | 101 | |
| Suppressing COX-2 expression by blocking the activation of MAPKs and AP-1 | In dorsal skin of female ICR mice | In vitro | 102 | |
| Decreasing the expression of COX-1, COX-2, c-myc, c-fos, c-jun, transforming growth factor-beta1 (TGF-β1) and TNF-α | In mouse skin | Ex vivo | 50 | |
| Inhibiting oncogenic disease through the inhibition of protein kinase CKII activity | In HeLa cell lysates | In vitro | 103 | |
| Inhibiting the Ca(2+)-dependent activities of PKCα and PKCβI | On the activities of PKC isozymes | In vitro | 104 | |
| Inhibiting nitrobenzene(NB)-DNA adducts and NB–Hb adducts | In male Kunming mice | In vivo | 105 | |
| Chlorogenic acid | Inhibiting the formation of DNA single strand breaks | In supercoiled pBR322 DNA | In vitro | 106 |
| Quercetin Luteolin | Blocking EGFR tyrosine kinase activity | In MiaPaCa-2 cancer cells | In vitro | 107 |
| Myricetin Apigenin Quercetin Kaempferol | Inhibiting human CYP1A1 activities Inhibiting the formation of diolepoxide 2(DE2) and B[a]P activation | On 7-ethoxyresorufin O-deethylation | In vitro | 26 |
| Silymarin Hesperetin Quercetin Daidzein | Interacting with P-glycoprotein and modulating the activity of ATP-binding cassette transporter, breast cancer resistance protein (BCRP/ABCG2) | In two separate BCRP-overexpressing cell lines | In vitro | 108 |
| EGCG | Inhibiting telomerase | In human cancer cells In nude mice models | In vitro In vivo | 114 |
| Curcumin | Suppressing proliferation and angiogenesis Inhibiting NF-κB-regulated gene products (cyclin D1, c-myc, Bcl-2, Bcl-xL, cellular inhibitor of apoptosis protein-1, COX-2, MMP, and VEGF) | In various pancreatic cancer cell lines and nude mice | In vitro In vivo | 109 |
| Inducing apoptosis by sustained phosphorylation of JNK and p38 MAPK Inhibitiing NF-κB transcriptional activity Inducing phosphorylation of c-jun and stimulation of AP-1 transcriptional activity | In HCT116 cells | In vitro | 110 | |
| Inducing apoptosis through activation of caspase-8, BID cleavage and cytochrome c release Suppressing ectopic expression of Bcl-2 and Bcl-xl | In human acute myelogenous leukemia HL-60 cells | In vitro | 111 | |
| Inhibiting the Akt/mTOR/p70S6K pathway and activating the ERK1/2 pathway Inhibiting tumor growth and inducing autophagy | In U87-MG and U373-MG malignant glioma cells In the subcutaneous xenograft model of U87-MG cells | In vitro In vivo | 112 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Curcumin | Inhibiting both myeloid differential factor 88 (MyD88)-and TIR domain-containing adapter inducing IFN-β (TRIF)-dependent pathways Inhibiting homodimerization of Toll-like receptor 4(TLR4) Suppressing the activation of NF-κB by inhibiting IκB kinase β activity in MyD88-dependent pathway Inhibiting IFN-regulatory factor 3 (IRF3) activation | In 293T cells | In vitro | 124 |
| Inhibiting the level of NOS mRNA and protein Suppressing NF-κB activation through inhibitory of IκB kinase activity | In macrophages | In vitro | 125 | |
| Suppressing COX-2 expression by inhibiting AP-1 and NF-κB | In BV2 microglial cells | In vitro | 126 | |
| Inhibiting IL-6-inducible STAT3 phosphorylation and nuclear translocation | In human multiple myeloma cells | In vitro | 118 | |
| Upregulating CYP3A4 via pregnane X receptor (PXR) activation Activating the electrophile responsive element (EpRE) of HO-1 and enhancing the gastrointestinal (GI)-GPx activity | In HepG2 cells | In vitro | 127 | |
| Suppressing JAK-STAT inflammatory signaling through activation of SHP-2 | In both rat primary microglia and murine BV2 microglial cells | In vitro | 97 | |
| Proanthocyanidins | Promoting apoptosis through alterations in Cdki-Cdk-cyclin cascade, and caspase-3 activation via loss of mitochondrial membrane potential | In human epidermoid carcinoma A431 cells | In vitro | 128 |
| Proanthocyanidins | Inhibiting the phosphorylation of ERK1/2, JNK and p38 Inhibiting the activation of NF-κB/p65 through inhibition of degradation of IκBα and activation of IκB kinase α | In SKH-1 hairless mice | In vivo | 129 |
| Caffeic acid | Modulating ceramide-induced signal transduction pathway and NF-κB activation Inhibiting protein tyrosine kinase activity | In U937 cells | In vitro | 113 |
| Quercetin | Inhibiting phosphorylation of JNK and p38 MAPK on ROS-mediated signaling | In HUVECs | In vitro | 117 |
| Modulating Akt/PKB and ERK1/2 signalling cascades on neuronal viability | In primary cortical neurons | In vitro In vivo | 130 | |
| Equol | Mediating rapid vascular relaxation by Ca2+-independent activation of eNOS/Hsp90 involving ERK1/2 and Akt phosphorylation | In human endothelial cells | In vitro | 131 |
| Resveratrol | Inhibiting monocyte CCR2 binding activity in an NO-, MAPK- and PI3K-dependent manner Inhibiting CCR2 mRNA in an NO- and MAPK-independent, PI3K-dependent manner | on THP-1 monocytes | In vitro | 58 |
| Inhibiting proliferation of cardiac fibroblasts by NO-cGMP signaling pathway | In cultured rat cardiac fibroblasts | In vitro | 59 | |
| Inducing phase II genes by regulating ARE/EpRE activation Modifying the capability of Keap1 in sequestering Nrf2 | In PC12 cells | In vitro | 132 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| EGCG Quercetin | Inhibiting apoptosis through modulation of Bcl-2 and Bax Inhibiting nuclear transactivation of p53 Decreasing the activity of caspase-3 Blocking JNK- and p38 MAPK-related signaling | In HUVECs | In vitro | 117 |
| RWPCs | Inhibiting the expression of VEGF mRNA and protein Preventing the activation of the p38 MAPK pathway | In VSMCs | In vitro | 133 |
| Inhibiting the invasion and migration of VSMCs Inhibiting pro-MMP-2 expression and its activation via inhibition of membrane type 1-MMP (MT1-MMP) activity | In VSMCs | In vitro | 134 | |
| Inhibiting VSMCs migration through inhibiting the PI3K activity and p38 MAPK phosphorylation Inhibiting the phosphorylation of MKK3/6 | In cultured VSMCs | In vitro | 135 | |
| Inducing EDHF-mediated relaxations through activation of the PI3-kinase/Akt pathway | In porcine coronary arteries | In vivo | 136 | |
| Increasing intracellular Ca2+ and activate tyrosine kinases Increasing NO production | In bovine aortic endothelial cells | In vitro | 137 | |
| Inhibiting NADPH oxidase activity and/or reducing endothelin-1(ET-1) release | In Twelve-week-old male Wistar rats | In vivo | 138 | |
| Inhibiting the synthesis of ET-1 | In cultured bovine aortic endothelial cells | In vitro | 139 | |
| Elevating NO and prostacyclin (PGI2) | In rats | In vivo | 140 | |
| Ehancing PGI2 release | In endothelial cell | In vitro | 141 | |
| Cy3G | Enhancing eNOS activity and expression Inducing NO production Regulating phosphorylation of eNOS and Akt Increasing cGMP production | In bovine vascular endothelial cells | In vitro | 142 |
| EGCG | Having endothelial-dependent vasodilator actions Activatiing phosphatidylinositol 3-kinase, Akt, and eNOS | In bovine aortic endothelial cells | In vitro | 143 |
| Increasing eNOS activity Inducing a sustained activation of Akt, ERK1/2, and eNOS Ser1179 phosphorylation | In bovine aortic endothelial cells | In vitro | 144 | |
| Catechins | Reducing the vascularization induced by the angiogenin-like protein on chicken CAM | In chichen | In vitro | 145 |
| Activin | Reducing ICAM-1, VCAM-1 and E-selectin | In systemic sclerosis | In vivo | 146 |
| Proanthocyanidin | Downregulating VCAM-1 expression; Decreasing TNFα-induced adherence of T-cells to HUVECs | In primary HUVECs | In vitro | 147 |
| Procyanidins Flavan-3-ols | Inhibiting angiotensin I converting enzyme (ACE) activity | In two substrates | In vitro | 148 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Curcumin | Inhibiting diabetes-induced elevation in the levels of IL-1β, VEGF, and NF-κB Decreasing oxidatively modified DNA and nitrotyrosine | In streptozotocin-induced diabetic rats | In vivo | 153 |
| EGCG, ECG, (−)-epigallochatechin | Inhibiting SGLT1 and sodium-free GLUT | In polarised Caco-2 intestinal cells | In vitro | 154 |
| Inhibiting SGLT1 and glucose uptake | In the rabbit small intestine | In vivo | 155 | |
| Quercetin | Reducing blood glucose levels Inhibiting sodium-dependent vitamin C transporter 1 (SVCT1) and GLUT2 | In Chinese hamster ovary cells | In vitro | 156 |
| Mangiferin | Inhibiting sucrase, isomaltase, and aldose reductase | In rats | In vivo | 157 |
| Tannins Anthocyanin | Inhibiting α-amylase and α-glucosidase | In the substrate of 2-chloro-4-nitro-phenyl-4-O-b-D-galactopyranosyl-maltoside | In vitro | 158 |
| Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|
| Resveratrol | Upregulating p21 expression and cause G1 phase arrest | In HepG2 cells | In vitro | 163 |
| Inhibiting cyclin D1/D2-cdk6, cyclin D1/D2-cdk4, and cyclin E-cdk2 complexes | In human epidermoid carcinoma A431 cells | In vitro | 164 | |
| Downregulatiing cyclin D1/Cdk4 complex and upregulating cyclin E and A expression | In the human colonic adenocarcinoma cell line Caco-2 | In vitro | 165 | |
| Decreasing in the hyperphosphorylated form of pRb and increasing in hypophosphorylated pRb Downregulating the protein expression of E2F (1–5) family members of transcription factors and their heterodimeric partners DP1 and DP2 Leading to a G0/G1 arrest | In human epidermoid carcinoma A431 cells | In vitro | 166 | |
| Inhibiting the expression of cyclin B1, D1, A1 and β-catenin | In six human cancer cell lines (MCF7, SW480, HCE7, Seg-1, Bic-1, and HL60) | In vitro | 167 | |
| Arresting cell cycle in the G1-S phase | In VSMCs | In vitro | 168 | |
| Upregulating the expression of cyclins A, E, and B1 | In human SK-Mel-28 melanoma cells | In vitro | 169 | |
| Proanthocyanidins | Increasing G1-phase arrest Inhibiting cyclin-dependent kinases (Cdk) Cdk2, Cdk4, Cdk6 and cyclins D1, D2 and E Increasing the protein expression of cyclin-dependent kinase inhibitors (Cdki), Cip1/p21 and Kip1/p27 Enhancing the binding of Cdki-Cdk | In human epidermoid carcinoma A431 cells | In vitro | 170 |
| Type of Activity | Dietary polyphenols | Protective effects and mechanisms | Conditions | Levels | Ref |
|---|---|---|---|---|---|
| Antibacterial activity | Gnemonol B and gnetin E | Exhibiting strong antibacterial activities against vancomycin-resistant Enterococci (VRE) and methicillin-resistant Staphylococcus aureus (MRSA) | In Enterococci and Staphylococcus aureus | In vitro | 174 |
| Hydroxytyrosol | Antimycoplasmal activity against M. pneumoniae, M. hominis, and M. fermentans | In Mycoplasma | In vitro | 175 | |
| Anti-HIV effects | Proanthocyanidins | Downregulating the expression of the HIV-1 entry co-receptors, CCR2b, CCR3 and CCR5 | In normal peripheral blood mononuclear cells | In vitro | 176 |
| Angiogenesis effect | Proanthocyanidins Resveratrol | Upregulating VEGF expression | In cultured keratinocytes | In vitro | 177 |
| Hepato-protective ability | A novel Proanthocyanidins IH636 | Increasing the expression of Bcl-xL Attenuating acetaminophen-induced hepatic DNA damage, apoptotic and necrotic cell death of liver cells | In male ICR mice | In vivo | 178 |
| Daidzein | Ameliorating the d-galactosamine-induced increase in malondialdehyde-protein adducts and cytosolic SOD activities | In the rat liver | In vivo | 179 | |
| Genistein | Reducing experimental liver damage caused by CCl(4) by preventing lipid peroxidation and strengthening antioxidant systems | In rats | In vitro | 180 |
Acknowledgements
References
- Gutteridge, J.M. Free radicals in diseases processes: a compilation of cause and consequence. Free Radic Res. Commun 1993, 19, 141–158. [Google Scholar]
- Kehrer, J.P. Free radicals as mediators of tissue injury and disease. Crit. Rev. Toxicol 1993, 23, 21–48. [Google Scholar]
- Becker, L.B. New concepts in reactive oxygen species and cardiovascular reperfusion physiology. Cardiovasc. Res 2004, 61, 461–470. [Google Scholar]
- Hayes, J.D.; McLellan, L.I. Glutathione and glutathione-dependent enzymes represent a co-ordinately regulated defences against oxidative stress. Free Radic. Res 1999, 31, 273–300. [Google Scholar]
- Masella, R.; Di Benedetto, R.; Vari, R.; Filesi, C.; Giovannini, C. Novel mechanisms of natural antioxidant compounds in biological systems: involvement of glutathione and glutathione-related enzymes. J. Nutr. Biochem 2005, 16, 577–586. [Google Scholar]
- Hartman, R.E.; Shah, A.; Fagan, A.M.; Schwetye, K.E.; Parsadanian, M.; Schulman, R. N.; Beth Finn, M.; Holtzman, D.M. Pomegranate juice decreases amyloid load and improves behavior in a mouse model of Alzheimer’s disease. Neurobiol. Dis 2006, 24, 506–515. [Google Scholar]
- Hollman, P.C.; van Trijp, J.M.; Buysman, M.N.; van der Gaag, M.S.; Mengelers, M.J.; de Vries, J.H.; Katan, M.B. Relative bioavailability of the antioxidant flavonoid quercetin from various foods in man. FEBS Lett 1997, 418, 152–156. [Google Scholar]
- Shen, S.Q; Zhang, Y.; Xiang, J.J.; Xiong, C.L. Protective effect of curcumin against liver warm ischemia/reperfusion injury in rat model is associated with regulation of heat shock protein and antioxidant enzymes. World J. Gastroenterol 2007, 13, 1953–1961. [Google Scholar]
- Molina, M.F.; Sanchez-Reus, I.; Iglesias, I.; Benedi, J. Quercetin, a flavonoid antioxidant, prevents and protects against ethanol-induced oxidative stress in mouse liver. Biol. Pharm. Bull 2003, 26, 1398–1402. [Google Scholar]
- Chen, C.; Yu, R.; Owuor, E.D.; Kong, A.N. Activation of antioxidant response element (ARE), mitogen-activated protein kinases (MAPKs) and caspases by major green tea polyphenol components during cell survival and death. Arch. Pharm. Res 2000, 23, 605–612. [Google Scholar]
- Butterfield, D.A.; Castegna, A.; Pocernich, C. B.; Drake, J.; Scapagninib, G.; Calabresec, V. Nutritional approaches to combat oxidative stress in Alzheimer’s disease. J. Nutr. Biochem 2002, 13, 444–461. [Google Scholar]
- Schaffer, S.; Podstawa, M.; Visioli, F.; Bogani, P.; Müller, W.E.; Eckert, G.P. Hydroxytyrosol-rich olive mill wastewater extract protects brain cells in vitro and ex vivo. J. Agric. Food Chem 2007, 55, 5043–5049. [Google Scholar]
- Tuck, K.L.; Freeman, M.P.; Hayball, P.J.; Stretch, G.L.; Stupans, I. The in vivo fate of hydroxytyrosol and tyrosol, antioxidant phenolic constituents of olive oil, after intravenous and oral dosing of labeled compounds to rats. J. Nutr 2001, 131, 1993–1996. [Google Scholar]
- Rice-Evans, C.A.; Mmiller, N.J.; Paganga, G. Antioxidant properties of phenolic compounas. Trends Plant Sci 1997, 2, 152–159. [Google Scholar]
- Nielsen, I.L.; Dragsted, L.O.; Ravn-Haren, G.; Freese, R.; Rasmussen, S.E. Absorption and excretion of black currant anthocyanins in humans and watanabe heritable hyperlipidemic rabbits. J. Agric. Food Chem 2003, 51, 2813–2820. [Google Scholar]
- Bub, A.; Watzl, B.; Heeb, D.; Rechkemmer, G.; Briviba, K. Malvidin-3-glucoside bioavailability in humans after ingestion of red wine, dealcoholized red wine and red grape juice. Eur. J. Nutr 2001, 40, 113–120. [Google Scholar]
- McAnlis, G.T.; McEneny, J.; Pearce, J.; Young, I.S. Absorption and antioxidant effects of quercetin from onions, in man. Eur. J. Clin. Nutr 1999, 53, 92–96. [Google Scholar]
- Manach, C.; Morand, C.; Gil-Izquierdo, A.; Bouteloup-Demange, C.; Remesy, C. Bioavailability in humans of the flavanones hesperidin and narirutin after the ingestion of two doses of orange juice. Eur. J. Clin. Nutr 2003, 57, 235–242. [Google Scholar]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radic. Biol. Med 2006, 41, 1727–1746. [Google Scholar]
- Erlund, I.; Meririnne, E.; Alfthan, G.; Aro, A. Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice. J. Nutr 2001, 131, 235–241. [Google Scholar]
- Henning, S.M.; Niu, Y.; Liu, Y.; Lee, N.H.; Hara, Y.; Thames, G.D.; Minutti, R.R.; Carpenter, C.L.; Wang, H.; Heber, D. Bioavailability and antioxidant effect of epigallocatechin gallate administered in purified form versus as green tea extract in healthy individuals. J. Nutr. Biochem 2005, 16, 610–616. [Google Scholar]
- Widlansky, M.E.; Duffy, S.J.; Hamburg, N.M.; Gokce, N.; Warden, B.A.; Wiseman, S.; Keaney, J.F., Jr.; Frei, B.; Vita, J.A. Effects of black tea consumption on plasma catechins and markers of oxidative stress and inflammation in patients with coronary artery disease. Free Radic. Biol. Med 2005, 38, 499–506. [Google Scholar]
- Bell, J.R.; Donovan, J.L.; Wong, R.; Waterhouse, A.L.; German, J.B.; Walzem, R. L.; Kasim-Karakas, S.E. (+)-Catechin in human plasma after ingestion of a single serving of reconstituted red wine. Am. J. Clin. Nutr 2000, 71, 103–108. [Google Scholar]
- Holt, R.R.; Lazarus, S.A.; Sullards, M.C.; Zhu, Q.Y.; Schramm, D.D.; Hammerstone, J.F.; Fraga, C.G.; Schmitz, H.H.; Keen, C.L. Procyanidin dimer B2 [epicatechin-(4beta-8)-epicatechin] in human plasma after the consumption of a flavanol-rich cocoa. Am. J. Clin. Nutr 2002, 76, 798–804. [Google Scholar]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radic. Biol. Med 2006, 41, 1727–1746. [Google Scholar]
- Schwarz, D.; Roots, I. In vitro assessment of inhibition by natural polyphenols of metabolic activation of procarcinogens by human CYP1A1. Biochem. Biophys. Res. Commun 2003, 303, 902–907. [Google Scholar]
- Gonthier, M.P.; Remesy, C.; Scalbert, A.; Cheynier, V.; Souquet, J.M.; Poutanen, K.; Aura, A.M. Microbial metabolism of caffeic acid and its esters chlorogenic and caftaric acids by human faecal microbiota in vitro. Biomed. Pharmacother 2006, 60, 536–540. [Google Scholar]
- Seeram, N. P.; Lee, R.; Heber, D. Bioavailability of ellagic acid in human plasma after consumption of ellagitannins from pomegranate (Punica granatum L.) juice. Clin. Chim. Acta 2004, 348, 63–68. [Google Scholar]
- Rangkadilok, N.; Sitthimonchai, S.; Worasuttayangkurn, L.; Mahidol, C.; Ruchirawat, M.; Satayavivad, J. Evaluation of free radical scavenging and antityrosinase activities of standardized longan fruit extract. Food Chem. Toxicol 2007, 45, 328–336. [Google Scholar]
- Ray, P.S.; Maulik, G.; Cordis, G.A.; Bertelli, A.A.; Bertelli, A.; Das, D.K. The red wine antioxidant resveratrol protects isolated rat hearts from ischemia reperfusion injury. Free Radic. Biol. Med 1999, 27, 160–169. [Google Scholar]
- Zhang, Y.; Liu, Y.; Wang, T.; Li, B.; Li, H.; Wang, Z.; Yang, B. Resveratrol, a natural ingredient of grape skin:Antiarrhythmic efficacy and ionic mechanisms. Biochem. Biophys. Res. Commun 2006, 340, 1192–1199. [Google Scholar]
- Chung, KT.; Wong, TY.; Wei, CI.; Huang, YW.; Lin, Y. Tannins and human health: a review. Crit. Rev. Food Sci. Nutr 1998, 38, 421–464. [Google Scholar]
- Sharma, R.A.; Gescher, A.J.; Steward, W.P. Curcumin: The story so far. Eur. J. Cancer 2005, 41, 1955–1968. [Google Scholar]
- Hong, J.; Smith, T.J.; Ho, C.T.; August, D.A.; Yang, C.S. Effects of purified green and black tea polyphenols on cyclooxygenase-and lipoxygenase-dependent metabolism of arachidonic acid in human colon mucosa and colon tumor tissues. Biochem. Pharmacol 2001, 62, 1175–1183. [Google Scholar]
- Fki, I.; Sahnoun, Z.; Sayadi, S. Hypocholesterolemic effects of phenolic extracts and purified hydroxytyrosol recovered from olive mill wastewater in rats fed a cholesterol-rich diet. J. Agric. Food Chem 2007, 55, 624–631. [Google Scholar]
- Kohyama, N.; Nagata, T.; Fujimoto, S.; Sekiya, K. Inhibition of arachidonate lipoxygenase activities by 2-(3, 4-dihydroxyphenyl) ethanol, a phenolic compound from olives. Biosci. Biotechnol. Biochem 1997, 61, 347–350. [Google Scholar]
- Du, Y.; Guo, H.; Lou, H. Grape seed polyphenols protect cardiac cells from apoptosis via induction of endogenous antioxidant enzymes. J. Agric. Food Chem 2007, 55, 1695–1701. [Google Scholar]
- Appiah-Opong, R.; Commandeur, J.N.; van Vugt-Lussenburg, B.; Vermeulen, N.P. Inhibition of human recombinant cytochrome P450s by curcumin and curcumin decomposition products. Toxicology 2007, 235, 83–91. [Google Scholar]
- Zheng, J.; Ramirez, V.D. Inhibition of mitochondrial proton F0F1-ATPase/ATP synthase by polyphenolic phytochemicals. Br. J. Pharmacol 2000, 130, 1115–1123. [Google Scholar]
- Nishinaka, T.; Ichijo, Y.; Ito, M.; Kimura, M.; Katsuyama, M.; Iwata, K.; Miura, T.; Terada, T.; Yabe-Nishimura, C. Curcumin activates human glutathione S-transferase P1 expression through antioxidant response element. Toxicol Lett 2007, 170, 238–247. [Google Scholar]
- Gil, B.; Sanz, M.J.; Terencio, M.C.; Ferrandiz, M.L.; Bustos, G.; Paya, M.; Gunasegaran, R.; Alcaraz, M.J. Effects of flavonoids on Naja naja and human recombinant synovial phospholipase A2 and inflammatory responses in mice. Life Sci 1994, 54, 333–338. [Google Scholar]
- Huang, J.; de Paulis, T.; May, J.M. Antioxidant effects of dihydrocaffeic acid in human EA.hy926 endothelial cells. J. Nutr. Biochem 2004, 15, 722–729. [Google Scholar]
- Kerry, N.; Rice-Evans, C. Inhibition of peroxynitrite-mediated oxidation of dopamine by flavonoid and phenolic antioxidants and their structural relationship. J. Neurochem 1999, 73, 247–253. [Google Scholar]
- Alía, M.; Ramos, S.; Mateos, R.; Granado-Serrano, A.B.; Bravo, L.; Goya, L. Quercetin protects human hepatoma HepG2 against oxidative stress induced by tert-butyl hydroperoxide. Toxicol. Appl. Pharmacol 2006, 212, 110–118. [Google Scholar]
- Valerio, L.G., Jr; Kepa, J.K.; Pickwell, G.V.; Quattrochi, L.C. Induction of human NAD(P)H:quinone oxidoreductase (NQO1) gene expression by the flavonol quercetin. Toxicol. Lett 2001, 119, 49–57. [Google Scholar]
- Motohashi, H.; Yamamoto, M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol. Med 2004, 10, 549–557. [Google Scholar]
- Scharf, G; Prustomersky, S.; Knasmuller, S.; Schulte-Hermann, R.; Huber, W.W. Enhancement of glutathione and g-glutamylcysteine synthetase, the rate limiting enzyme of glutathione synthesis, by chemoprotective plant-derived food and beverage components in the human hepatoma cell line HepG2. Nutr. Cancer 2003, 45, 74–83. [Google Scholar]
- Tanigawa, S.; Fujii, M.; Hou, D.X. Action of Nrf2 and Keap1 in ARE-mediated NQO1 expression by quercetin. Free Radic. Biol. Med 2007, 42, 1690–1703. [Google Scholar]
- Cadenas, S.; Barja, G. Resveratrol, melatonin, vitamin E, and PBN protect against renal oxidative DNA damage induced by the kidney carcinogen KBrO3. Free Radic. Biol. Med 1999, 26, 1531–1537. [Google Scholar]
- Jang, M.; Pezzuto, J.M. Effects of resveratrol on 12-O-tetradecanoylphorbol -13-acetate -induced oxidative events and gene expression in mouse skin. Cancer Lett 1998, 134, 81–89. [Google Scholar]
- Dubuisson, J.G.; Dyess, D.L.; Gaubatz, J.W. Resveratrol modulates human mammary epithelial cell O-acetyltransferase, sulfotransferase, and kinase activation of the heterocyclic amine carcinogen N-hydroxy-PhIP. Cancer Lett 2002, 182, 27–32. [Google Scholar]
- Ciolino, H.P.; Yeh, G.C. Inhibition of aryl hydrocarbon induced cytochrome P-4501A1 enzyme activity and CYP1A1 expression by resveratrol. Mol. Pharmacol 1999, 56, 760–767. [Google Scholar]
- Casper, R.F.; Quesne, M.; Rogers, I.M.; Shirota, T.; Jolivet, A.; Milgrom, E.; Savouret, J.F. Resveratrol has antagonist activity on the aryl hydrocarbon receptor: implications for prevention of dioxin toxicity. Mol. Pharmacol 1999, 56, 784–790. [Google Scholar]
- Schewe, T.; Sadik, C.; Klotz, L.O.; Yoshimoto, T.; Kuhn, H.; Sies, H. Polyphenols of cocoa: inhibition of mammalian 15-lipoxygenase. Biol. Chem 2001, 382, 1687–1696. [Google Scholar]
- Subbaramaiah, K.; Chung, W.J.; Michaluart, P.; Telang, N.; Tanabe, T.; Inoue, H.; Jang, M.; Pezzuto, J.M.; Dannenberg, A.J. Resveratrol inhibits cyclooxygenase-2 transcription and activity in phorbol ester-treated human mammary epithelial cells. J. Biol. Chem 1998, 273, 21875–21882. [Google Scholar]
- Li, Y.T.; Shen, F.; Liu, B.H.; Cheng, G.F. Resveratrol inhibits matrix metalloproteinase-9 transcription in U937 cells. Acta Pharmacol. Sin 2003, 24, 1167–1171. [Google Scholar]
- Kaga, S.; Zhan, L.; Matsumoto, M.; Maulik, N. Resveratrol enhances neovascularization in the infarcted rat myocardium through the induction of thioredoxin-1, heme oxygenase-1 and vascular endothelial growth factor. J. Mol. Cell. Cardiol 2005, 39, 813–822. [Google Scholar]
- Cullen, J.P.; Morrow, D.; Jin, Y.; von Offenberg Sweeney, N.; Sitzmann, J.V.; Cahill, P.A.; Redmond, E.M. Resveratrol inhibits expression and binding activity of the monocyte chemotactic protein-1 receptor, CCR2, on THP-1 monocytes. Atherosclerosis 2007, in press. [Google Scholar]
- Wang, S.; Wang, X.; Yan, J.; Xie, X.; Fan, F.; Zhou, X.; Han, L.; Chen, J. Resveratrol inhibits proliferation of cultured rat cardiac fibroblasts: Correlated with NO-cGMP signaling pathway. Eur. J. Pharmacol 2007, 567, 26–35. [Google Scholar]
- Steffen, Y.; Wiswedel, I.; Peter, D.; Schewe, T.; Sies, H. Cytotoxicity of myeloperoxidase/nitrite-oxidized low-density lipoprotein toward endothelial cells is due to a high 7β-hydroxycholesterol to 7-ketocholesterol ratio. Free Radic. Biol. Med 2006, 41, 1139–1150. [Google Scholar]
- Petroni, A.; Blasevich, M.; Salami, M.; Papini, N.; Montedoro, G.F.; Galli, C. Inhibition of platelet aggregation and eicosanoid production by phenolic components of olive oil. Thromb. Res 1995, 78, 151–160. [Google Scholar]
- Léger, C.L.; Carbonneau, M.A.; Michel, F.; Mas, E.; Monnier, L.; Cristol, J.P.; Descomps, B. A thromboxane effect of a hydroxytyrosol-rich olive oil wastewater extract in patients with uncomplicated type I diabetes. Eur. J. Clin. Nutr 2005, 59, 727–730. [Google Scholar]
- de La Puerta, R.; Ruiz-Gutierrez, V.; Hoult, J.R. Inhibition of leukocyte 5-lipoxygenase by phenolics from virgin olive oil. Biochem. Pharmacol 1999, 57, 445–449. [Google Scholar]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A.; Massaro, M.; Scoditti, E.; Storelli, C.; Visioli, F.; Distante, A.; De Caterina, R. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: antiatherogenic properties of Mediterranean diet phytochemicals. Arterioscler. Thromb. Vasc. Biol 2003, 23, 622–629. [Google Scholar]
- Manna, C.; Migliardi, V.; Golino, P.; Scognamiglio, A.; Galletti, P.; Chiariello, M.; Zappia, V. Oleuropein prevents oxidative myocardial injury induced by ischemia and reperfusion. J. Nutr. Biochem 2004, 15, 461–466. [Google Scholar]
- Gouedard, C.; Barouki, R.; Morel, Y. Dietary polyphenols increase paraoxonase 1 gene expression by an aryl hydrocarbon receptor-dependent mechanism. Mol. Cell Biol 2004, 24, 5209–5222. [Google Scholar]
- Nair, M.P.; Kandaswami, C.; Mahajan, S.; Chadha, K.C.; Chawda, R.; Nair, H.; Kumar, N.; Nair, R.E.; Schwartz, S.A. The flavonoid, quercetin, differentially regulates Th-1 (IFN gamma) and Th-2 (IL4) cytokine gene expression by normal peripheral blood mononuclear cells. Biochim. Biophys. Acta 2002, 1593, 29–36. [Google Scholar]
- Myhrstad, M.C.; Carlsen, H.; Nordstrom, O.; Blomhoff, R.; Moskaug, J.O. Flavonoids increase the intracellular glutathione level by transactivation of the gamma-glutamylcysteine synthetase catalytical subunit promoter. Free Radic. Biol. Med 2002, 32, 386–393. [Google Scholar]
- Lo, H.M.; Hung, C.F.; Huang, Y.Y.; Wu, W.B. Tea polyphenols inhibit rat vascular smooth muscle cell adhesion and migration on collagen and laminin via interference with cell-ECM interaction. J. Biomed. Sci 2007, in press. [Google Scholar]
- Mizushige, T.; Mizushige, K.; Miyatake, A.; Kishida, T.; Ebihara, K. Inhibitory effects of soy isoflavones on cardiovascular collagen accumulation in rats. J. Nutr. Sci. Vitaminol. (Tokyo) 2007, 53, 48–52. [Google Scholar]
- Tikkanen, M.J.; Adlercreutz, H. Dietary soy-derived isoflavone phytoestrogens. Could they have a role in coronary heart disease prevention? Biochem.Pharmacol 2000, 60, 1–5. [Google Scholar]
- Schramm, D.D.; Wang, J.F.; Holt, R.R.; Ensunsa, J.L.; Gonsalves, J.L.; Lazarus, S.A.; Schmitz, H.H.; German, J.B.; Keen, C.L. Chocolate procyanidins decrease the leukotriene-prostacyclin ratio in humans and human aortic endothelial cells. Am. J. Clin. Nutr 2001, 73, 36–40. [Google Scholar]
- Dedoussis, G.V.; Kaliora, A.C.; Psarras, S.; Chiou, A.; Mylona, A.; Papadopoulos, N.G.; Andrikopoulos, N.K. Antiatherogenic effect of Pistacia lentiscus via GSH restoration and downregulation of CD36 mRNA expression. Atherosclerosis 2004, 174, 293–303. [Google Scholar]
- Sato, M.; Bagchi, D.; Tosaki, A.; Das, D.K. Grape seed proanthocyanidin reduces cardiomyocyte apoptosis by inhibiting ischemia–reperfusion-induced activation of JNK-1 and c-JUN. Free Radic. Biol. Med 2001, 31, 729–737. [Google Scholar]
- Dasgupta, B.; Milbrandt, J. Resveratrol stimulates AMP kinase activity in neurons. Proc. Natl. Acad. Sci. U. S. A 2007, 104, 7217–7222. [Google Scholar]
- Chavez, E.; Reyes-Gordillo, K.; Segovia, J.; Shibayama, M.; Tsutsumi, V.; Vergara, P.; Moreno, M.G.; Muriel, P. Resveratrol prevents fibrosis, NF-kappaB activation and TGF-beta increases induced by chronic CCl (4) treatment in rats. J. Appl. Toxicol 2007, in press. [Google Scholar]
- Bastianetto, S.; Brouillette, J.; Quirion, R. Neuroprotective effects of natural products: interaction with intracellular kinases, amyloid peptides and a possible role for transthyretin. Neurochem. Res 2007, in press. [Google Scholar]
- Okawara, M.; Katsuki, H.; Kurimoto, E.; Shibata, H.; Kume, T.; Akaike, A. Resveratrol protects dopaminergic neurons in midbrain slice culture from multiple insults. Biochem. Pharmacol 2007, 73, 550–560. [Google Scholar] [Green Version]
- Kim, S.J.; Jeong, H.J.; Lee, K.M.; Myung, N.Y.; An, N.H.; Mo Yang, W; Kyu Park, S.; Lee, H.J.; Hong, S.H.; Kim, H.M.; Um, J.Y. Epigallocatechin-3-gallate suppresses NF-kappaB activation and phosphorylation of p38 MAPK and JNK in human astrocytoma U373MG cells. J. Nutr. Biochem 2007, in press. [Google Scholar]
- de Boer, V.C.; de Goffau, M.C.; Arts, I.C.; Hollman, P.C.; Keijer, J. SIRT1 stimulation by polyphenols is affected by their stability and metabolism. Mech. Ageing Dev 2006, 127, 618–627. [Google Scholar]
- Levites, Y.; Amit, T.; Youdim, M.B.; Mandel, S. Involvement of protein kinase C activation and cell survival/cell cycle genes in green tea polyphenol (−)-epigallocatechin-3-gallate neuron-protective action. J. Biol. Chem 2002, 277, 30574–30580. [Google Scholar]
- Mercer, L. D.; Kelly, B.L.; Horne, M. K.; Beart, P.M. Dietary polyphenols protect dopamine neurons from oxidative insults and apoptosis: investigations in primary rat mesencephalic cultures. Biochem. Pharmacol 2005, 69, 339–345. [Google Scholar]
- Schroeter, H.; Spencer, J.P.; Rice-Evans, C.; Williams, R.J. Flavonoids protect neurons from oxidized low-density lipoprotein-induced apoptosis involving c-Jun N-terminal kinase (JNK), cjun and caspase-3. Biochem. J 2001, 358, 547–557. [Google Scholar]
- Garcia-Alloza, M.; Borrelli, L.A.; Rozkalne, A.; Hyman, B.T.; Bacskai, B.J. Curcumin labels amyloid pathology in vivo, disrupts existing plaques, and partially restores distorted neurites in an Alzheimer mouse model. J. Neurochem 2007, 102, 1095–1104. [Google Scholar]
- Mao, T.K.; Powell, J.; Van de Water, J.; Keen, C.L.; Schmitz, H.H.; Hammerstone, J.F.; Eric Gershwin, M. The effect of cocoa procyanidins on the transcription and secretion of interleukin lβ in peripheral blood mononuclear cells. Life Sci 2000, 66, 1377–1386. [Google Scholar]
- Kawai, K.; Tsuno, N.H.; Kitayama, J.; Okaji, Y.; Yazawa, K.; Asakage, M.; Sasaki, S.; Watanabe, T.; Takahashi, K.; Nagawa, H. Epigallocatechin gallate induces apoptosis of monocytes. J. Allergy Clin. Immunol 2005, 115, 186–191. [Google Scholar]
- Kawai, K.; Tsuno, N.H.; Kitayama, J.; Okaji, Y.; Yazawa, K.; Asakage, M.; Hori, N.; Watanabe, T.; Takahashi, K.; Nagawa, H. Epigallocatechin gallate attenuates adhesion and migration of CD8+ T cells by binding to CD11b. J. Allergy Clin. Immunol 2004, 113, 1211–1217. [Google Scholar]
- Shakibaei, M.; John, T.; Seifarth, C.; Mobasheri, A. Resveratrol inhibits IL-1beta-induced stimulation of caspase-3 and cleavage of PARP in human articular chondrocytes in vitro. Ann. N. Y. Acad. Sci 2007, 1095, 554–563. [Google Scholar]
- Tsai, S.H.; Lin-Shiau, S.Y.; Lin, J.K. Suppression of nitric oxide synthase and the down-regulation of the activation of NF-κB in macrophages by resveratrol. Br. J. Pharmacol 1999, 126, 673–680. [Google Scholar]
- Nonn, L.; Duong, D.; Peehl, D.M. Chemopreventive anti-inflammatory activities of curcumin and other phytochemicals mediated by MAP kinase phosphatase-5 in prostate cells. Carcinogenesis 2007, 28, 1188–1196. [Google Scholar]
- Gerritsen, M.E.; Carley, W.W.; Ranges, G.E.; Shen, C.P.; Phan, S.A.; Ligon, G.F.; Perry, C.A. Flavonoids inhibit cytokine-induced endothelial cell adhesion protein gene expression. Am. J. Pathol 1995, 147, 278–292. [Google Scholar]
- Choi, J.S.; Choi, Y.J.; Park, S.H.; Kang, J.S.; Kang, Y.H. Flavones mitigate tumor necrosis factor-alpha-induced adhesion molecule upregulation in cultured human endothelial cells: role of nuclear factor-kappa B. J. Nutr 2004, 134, 1013–1019. [Google Scholar]
- van Meeteren, M.E.; Hendriks, J.J.; Dijkstra, C.D.; van Tol, E.A. Dietary compounds prevent oxidative damage and nitric oxide production by cells involved in demyelinating disease. Biochem. Pharmacol 2004, 67, 967–975. [Google Scholar]
- Youdim, K.A.; McDonald, J.; Kalt, W.; Joseph, J.A. Potential role of dietary flavonoids in reducing microvascular endothelium vulnerability to oxidative and inflammatory insults. J. Nutr. Biochem 2002, 13, 282–288. [Google Scholar]
- Camacho-Barquero, L.; Villegas, I.; Sanchez-Calvo, J.M.; Talero, E.; Sanchez-Fidalgo, S.; Motilva, V.; Alarcon de la Lastra, C. Curcumin, a Curcuma longa constituent, acts on MAPK p38 pathway modulating COX-2 and iNOS expression in chronic experimental colitis. Int. Immunopharmacol 2007, 7, 333–342. [Google Scholar]
- Rahman, I.; Biswas, SK.; Kirkham, PA. Regulation of inflammation and redox signaling by dietary polyphenols. Biochem. Pharmacol 2006, 72, 1439–1452. [Google Scholar]
- Kim, H.Y.; Park, E.J.; Joe, E.H.; Jou, I. Curcumin suppresses Janus kinase-STAT inflammatory signaling through activation of src homology 2 domain-containing tyrosine phosphatase 2 in brain microglia. J. Immunol 2003, 171, 6072–6079. [Google Scholar]
- Fabiani, R.; De Bartolomeo, A.; Rosignoli, P.; Servili, M.; Montedoro, G.F.; Morozzi, G. Cancer chemoprevention by hydroxytyrosol isolated from virgin olive oil through G1 cell cycle arrest and apoptosis. Eur. J. Cancer Prev 2002, 11, 351–358. [Google Scholar]
- Fuggetta, M.P.; Lanzilli, G.; Tricarico, M.; Cottarelli, A.; Falchetti, R.; Ravagnan, G.; Bonmassar, E. Effect of resveratrol on proliferation and telomerase activity of human colon cancer cells in vitro. J. Exp. Clin. Cancer Res 2006, 25, 189–193. [Google Scholar]
- Kuo, P.L.; Chiang, L.C.; Lin, C.C. Resveratrol-induced apoptosis is mediated by p53-dependent pathway in HepG2 cells. Life Sci 2002, 72, 23–34. [Google Scholar]
- Pozo-Guisado, E.; Lorenzo-Benayas, M.J.; Fernandez-Salguero, P.M. Resveratrol modulates the phosphoinositide 3-kinase pathway through an estrogen receptor alpha-dependent mechanism: relevance in cell proliferation. Int. J. Cancer 2004, 109, 167–173. [Google Scholar]
- Kundu, J.K.; Chun, K.S.; Kim, S.O.; Surh, Y.J. Resveratrol inhibits phorbol ester-induced cyclooxygenase-2 expression in mouse skin: MAPKs and AP-1 as potential molecular targets. Biofactors 2004, 21, 33–39. [Google Scholar]
- Yoon, S.H.; Kim, Y.S.; Ghim, S.Y.; Song, B.H.; Bae, Y.S. Inhibition of protein kinase CKII activity by resveratrol, a natural compound in red wine and grapes. Life Sci 2002, 71, 2145–2152. [Google Scholar]
- Slater, S.J.; Seiz, J.L.; Cook, A.C.; Stagliano, B.A.; Buzas, C.J. Inhibition of protein kinase C by resveratrol. Biochim. Biophys. Acta 2003, 1637, 59–69. [Google Scholar]
- Li, H.; Cheng, Y.; Wang, H.; Sun, H.; Liu, Y.; Liu, K.; Peng, S. Inhibition of nitrobenzene-induced DNA and hemoglobin adductions by dietary constituents. Appl. Radiat. Isot 2003, 58, 291–298. [Google Scholar]
- Grace, S.C.; Salgo, M.G.; Pryor, W.A. Scavenging of peroxynitrite by a phenolic/peroxidase system prevents oxidative damage to DNA. FEBS Lett 1998, 426, 24–28. [Google Scholar]
- Lee, L.T.; Huang, Y.T.; Hwang, J.J.; Lee, P.P.; Ke, F.C.; Nair, M.P.; Kanadaswam, C.; Lee, M.T. Blockade of the epidermal growth factor receptor tyrosine kinase activity by quercetin and luteolin leads to growth inhibition and apoptosis of pancreatic tumor cells. Anticancer Res 2002, 22, 1615–1627. [Google Scholar]
- Cooray, H.C.; Janvilisri, T.; van Veen, H.W.; Hladky, S.B.; Barrand, M.A. Interaction of the breast cancer resistance protein with plant polyphenols. Biochem. Biophys. Res. Commun 2004, 317, 269–275. [Google Scholar]
- Kunnumakkara, A.B.; Guha, S.; Krishnan, S.; Diagaradjane, P.; Gelovani, J.; Aggarwal, B.B. Curcumin potentiates antitumor activity of gemcitabine in an orthotopic model of pancreatic cancer through suppression of proliferation, angiogenesis, and inhibition of nuclear factor-kappaB-regulated gene products. Cancer Res 2007, 67, 3853–3861. [Google Scholar]
- Collett, G.P.; Campbell, F.C. Curcumin induces c-jun N-terminal kinase-dependent apoptosis in HCT116 human colon cancer cells. Carcinogenesis 2004, 25, 2183–2189. [Google Scholar]
- Anto, R.J.; Mukhopadhyay, A.; Denning, K.; Aggarwal, B.B. Curcumin (diferuloylmethane) induces apoptosis through activation of caspase-8, BID cleavage and cytochrome c release: its suppression by ectopic expression of Bcl-2 and Bcl-xL. Carcinogenesis 2002, 23, 143–150. [Google Scholar]
- Aoki, H.; Takada, Y.; Kondo, S.; Sawaya, R.; Aggarwal, B.; Kondo, Y.; Aoki, H.; Takada, Y.; Kondo, S.; Sawaya, R.; Aggarwal, B.; Kondo, Y. Evidence that curcumin suppresses the growth of malignant gliomas in vitro and in vivo through induction of autophagy: role of Akt and ERK signaling pathways. Mol. Pharmacol 2007, 72, 29–39. [Google Scholar]
- Nardini, M.; Leonardi, F.; Scaccini, C.; Virgili, F. Modulation of ceramide-induced NF-κB binding activity and apoptotic response by caffeic acid in U937 cells: comparison with other antioxidants. Free Radic. Biol. Med 2001, 30, 722–733. [Google Scholar]
- Naasani, I.; Oh-Hashi, F.; Oh-Hara, T.; Feng, W.Y.; Johnston, J.; Chan, K.; Tsuruo, T. Blocking telomerase by dietary polyphenols is a major mechanisms for limiting the growth of human cancer cells in vitro and in vivo. Cancer Res 2003, 63, 824–830. [Google Scholar]
- Jung, J.Y.; Mo, H.C.; Yang, K.H.; Jeong, Y.J.; Yoo, H.G.; Choi, N.K.; Oh, W.M.; Oh, H.K.; Kim, S.H.; Lee, J.H.; Kim, H.J.; Kim, W.J. Inhibition by epigallocatechin gallate of CoCl2-induced apoptosis in rat PC12 cells. Life Sci 2007, 80, 1355–1363. [Google Scholar]
- Khanduja, K.L.; Avti, P.K.; Kumar, S.; Mittal, N.; Sohi, K.K.; Pathak, C.M. Anti-apoptotic activity of caffeic acid, ellagic acid and ferulic acid in normal human peripheral blood mononuclear cells: A Bcl-2 independent mechanism. Biochim. Biophys. Acta 2006, 1760, 283–289. [Google Scholar]
- Choi, Y.J.; Jeong, Y.J.; Lee, Y.J.; Kwon, H.M.; Kang, Y.H. (−) Epigallocatechin gallate and quercetin enhance survival signaling in response to oxidant-induced human endothelial apoptosis. J. Nutr 2005, 135, 707–713. [Google Scholar]
- Bharti, A.C.; Donato, N.; Aggarwal, B.B. Curcumin (diferuloylmethane) inhibits constitutive and IL-6-inducible STAT3 phosphorylation in human multiple myeloma cells. J. Immunol 2003, 171, 3863–3871. [Google Scholar]
- Yuste, P.; Longstaff, M.; McCorquodale, C. The effect of proanthocyanidin-rich hulls and proanthocyanidin extracts from bean (Vicia faba L.) hulls on nutrient digestibility and digestive enzyme activities in young chicks. Br. J. Nutr 1992, 67, 57–65. [Google Scholar]
- Longstaff, M.; McNab, J.M. The inhibitory effects of hull polysaccharides and tannins of field beans (Vicia faba L.) on the digestion of amino acids, starch and lipid and on digestive enzyme activities in young chicks. Br. J. Nutr 1991, 65, 199–216. [Google Scholar]
- Sbarra, V.; Ristorcelli, E.; Petit-Thevenin, J.L.; Teissedre, P.L.; Lombardo, D.; Verine, A. In vitro polyphenol effects on activity, expression and secretion of pancreatic bile salt-dependent lipase. Biochim. Biophys. Acta 2005, 1736, 67–76. [Google Scholar]
- Aggarwal, B.B.; Shisodia, S. Suppression of the nuclear factorkappaB activation pathway by spice-derived phytochemicals: reasoning for seasoning. Ann. N.Y. Acad. Sci 2004, 1030, 434– 441. [Google Scholar]
- Hanamura, T.; Hagiwara, T.; Kawagishi, H. Structural and functional characterization of polyphenols isolated from acerola (Malpighia emarginata DC.) fruit. Biosci. Biotechnol. Biochem 2005, 69, 280–286. [Google Scholar]
- Youn, H.S.; Saitoh, S.I.; Miyake, K.; Hwang, D.H. Inhibition of homodimerization of Toll-like receptor 4 by curcumin. Biochem. Pharmacol 2006, 72, 62–69. [Google Scholar]
- Pan, M.H.; Lin-Shiau, S.Y.; Lin, J.K. Comparative studies on the suppression of nitric oxide synthase by curcumin and its hydrogenated metabolites through down-regulation of IkappaB kinase and NFkappaB activation in macrophages. Biochem. Pharmacol 2000, 60, 1665–1676. [Google Scholar]
- Kang, G.; Kong, P.J.; Yuh, Y.J.; Lim, S.Y.; Yim, S.V.; Chun, W.; Kim, S.S. Curcumin suppresses lipopolysaccharide-induced cyclooxygenase-2 expression by inhibiting activator protein 1 and nuclear factor kappaB bindings in BV2 microglial cells. J. Pharmacol. Sci 2004, 94, 325–328. [Google Scholar]
- Kluth, D.; Banning, A.; Paur, I.; Blomhoff, R.; Brigelius-Flohe, R. Modulation of pregnane X receptor-and electrophile responsive element-mediated gene expression by dietary polyphenolic compounds. Free Radic. Biol. Med 2007, 42, 315–325. [Google Scholar]
- Meeran, S.M.; Katiyar, S.K. Grape seed proanthocyanidins promote apoptosis in human epidermoid carcinoma A431 cells through alterations in Cdki-Cdk-cyclin cascade, and caspase-3 activation via loss of mitochondrial membrane potential. Exp. Dermatol 2007, 16, 405–415. [Google Scholar]
- Sharma, S.D.; Meeran, S.M.; Katiyar, S.K. Dietary grape seed proanthocyanidins inhibit UVB-induced oxidative stress and activation of mitogen-activated protein kinases and nuclear factor-kappaB signaling in in vivo SKH-1 hairless mice. Mol. Cancer Ther 2007, 6, 995–1005. [Google Scholar]
- Spencer, J.P.; Rice-Evans, C.; Williams, R.J. Modulation of pro-survival Akt/PKB and ERK1/2 signalling cascades by quercetin and its in vivo metabolites underlie their action on neuronal viability. J. Biol. Chem 2003, 278, 34783–34793. [Google Scholar]
- Joy, S.; Siow, R.C.; Rowlands, D.J.; Becker, M.; Wyatt, A.W.; Aaronson, P.I.; Coen, C.W.; Kallo, I.; Jacob, R.; Mann, G.E. The isoflavone Equol mediates rapid vascular relaxation: Ca2+-independent activation of endothelial nitric-oxide synthase/Hsp90 involving ERK1/2 and Akt phosphorylation in human endothelial cells. J. Biol. Chem 2006, 281, 27335–27345. [Google Scholar]
- Chen, C.Y.; Jang, J.H.; Li, M.H.; Surh, Y.J. Resveratrol upregulates heme oxygenase-1 expression via activation of NF-E2-related factor 2 in PC12 cells. Biochem. Biophys. Res. Commun 2005, 331, 993–1000. [Google Scholar]
- Oak, M.H.; Chataigneau, M.; Keravis, T.; Chataigneau, T.; Beretz, A.; Andriantsitohaina, R.; Stoclet, J.C.; Chang, S.J.; Schini-Kerth, V.B. Red wine polyphenolic compounds inhibit vascular endothelial growth factor expression in vascular smooth muscle cells by preventing the activation of the p38 mitogen-activated protein kinase pathway. Arterioscler. Thromb. Vasc. Biol 2003, 23, 1001–1007. [Google Scholar]
- Oak, M.H.; El Bedoui, J.; Anglard, P.; Schini-Kerth, V.B. Red wine polyphenolic compounds strongly inhibit pro-matrix metalloproteinase-2 expression and its activation in response to thrombin via direct inhibition of membrane type 1-matrix metalloproteinase in vascular smooth muscle cells. Circulation 2004, 110, 1861–1867. [Google Scholar]
- Iijima, K.; Yoshizumi, M.; Hashimoto, M.; Akishita, M.; Kozaki, K.; Ako, J.; Watanabe, T.; Ohike, Y.; Son, B.; Yu, J.; Nakahara, K.; Ouchi, Y. Red wine polyphenols inhibit vascular smooth muscle cell migration through two distinct signaling pathways. Circulation 2002, 105, 2404–2410. [Google Scholar]
- Ndiaye, M.; Chataigneau, T.; Chataigneau, M.; Schini-Kerth, V.B. Red wine polyphenols induce EDHF-mediated relaxations in porcine coronary arteries through the redox-sensitive activation of the PI3-kinase/Akt pathway. Br. J. Pharmacol 2004, 142, 1131–1136. [Google Scholar]
- Martin, S.; Andriambeloson, E.; Takeda, K.; Andriantsitohaina, R. Red wine polyphenols increase calcium in bovine aortic endothelial cells: a basis to elucidate signalling pathways leading to nitric oxide production. Br. J. Pharmacol 2002, 135, 1579–1587. [Google Scholar]
- Jiménez, R.; López-Sepúlveda, R.; Kadmiri, M.; Romero, M.; Vera, R.; Sánchez, M.; Vargas, F.; O’valle, F.; Zarzuelo, A.; Dueñas, M.; Santos-Buelga, C.; Duarte, J. Polyphenols restore endothelial function in DOCA-salt hypertension: Role of endothelin-1 and NADPH oxidase. Free Radic. Biol. Med 2007, 43, 462–473. [Google Scholar]
- Khan, N.Q.; Lees, D.M.; Douthwaite, J.A.; Carrier, M.J.; Corder, R. Comparison of red wine extract and polyphenol constituents on endothelin-1 synthesis by cultured endothelial cells. Clin. Sci. (Lond) 2002, 103 Suppl 48, 72S–75S. [Google Scholar]
- Wollny, T.; Chabielska, E.; Malinowska-Zaprzałka, M.; Nazarko, J.; Rozmysłowicz-Szermińska, W.; Buczko, W. Effects of Bulgarian red and white wines on primary hemostasis and experimental thrombosis in rats. Pol. J. Pharmacol 2003, 55, 1089–1096. [Google Scholar]
- Dell’Agli, M.; Busciala, A.; Bosisio, E. Vascular effects of wine polyphenols. Cardiovasc. Res 2004, 63, 593–602. [Google Scholar]
- Xu, J.W.; Ikeda, K.; Yamori, Y. Cyanidin-3-glucoside regulates phosphorylation of endothelial nitric oxide synthase. FEBS Lett 2004, 574, 176–180. [Google Scholar]
- Kim, J.A.; Formoso, G.; Li, Y.; Potenza, M.A.; Marasciulo, F.L.; Montagnani, M.; Quon, M.J. Epigallocatechin gallate, a green tea polyphenol, mediates NO-dependent vasodilation using signaling pathways in vascular endothelium requiring reactive oxygen species and Fyn. J. Biol. Chem 2007, 282, 13736–13745. [Google Scholar]
- Lorenz, M.; Wessler, S.; Follmann, E.; Michaelis, W.; Düsterhöft, T.; Baumann, G.; Stangl, K.; Stangl, V. A constituent of green tea, epigallocatechin-3-gallate, activates endothelial nitric oxide synthase by a phosphatidylinositol-3-OH-kinase-, cAMP-dependent protein kinase-, and Akt-dependent pathway and leads to endothelial-dependent vasorelaxation. J. Biol. Chem 2004, 279, 6190–6195. [Google Scholar]
- Maiti, T.K.; Chatterjee, J.; Dasgupta, S. Effect of green tea polyphenols on angiogenesis induced by an angiogenin-like protein. Biochem. Biophys. Res. Commun 2003, 308, 64–67. [Google Scholar]
- Kalin, R.; Righi, A.; Del Rosso, A.; Bagchi, D.; Generini, S.; Cerinic, M.M.; Das, D.K. Activin, a grape seed-derived proanthocyanidin extract, reduces plasma levels of oxidative stress and adhesion molecules (ICAM-1, VCAM-1 and E-selectin) in systemic sclerosis. Free Radic. Res 2002, 36, 819–825. [Google Scholar]
- Sen, C.K.; Bagchi, D. Regulation of inducible adhesion molecule expression in human endothelial cells by grape seed proanthocyanidin extract. Mol. Cell Biochem 2001, 216, 1–7. [Google Scholar]
- Actis-Goretta, L.; Ottaviani, J.I.; Keen, C.L.; Fraga, C.G. Inhibition of angiotensin converting enzyme (ACE) activity by flavan-3-ols and procyanidins. FEBS Lett 2003, 555, 597–600. [Google Scholar]
- Alvarez, P.; Alvarado, C.; Puerto, M.; Schlumberger, A.; Jiménez, L.; De la Fuente, M. Improvement of leukocyte functions in prematurely aging mice after five weeks of diet supplementation with polyphenol-rich cereals. Nutrition 2006, 22, 913–921. [Google Scholar]
- Bhattacharyya, S.; Mandal, D.; Saha, B.; Sen, G.S.; Das, T.; Sa, G. Curcumin prevents tumor-induced T cell apoptosis through Stat-5a-mediated Bcl-2 induction. J.Biol.Chem 2007, 282, 15954–15964. [Google Scholar]
- Akiyama, H.; Sato, Y.; Watanabe, T.; Nagaoka, M.H.; Yoshioka, Y.; Shoji, T.; Kanda, T.; Yamada, K.; Totsuka, M.; Teshima, R.; Sawada, J.; Goda, Y.; Maitani, T. Dietary unripe apple polyphenol inhibits the development of food allergies in murine models. FEBS Lett 2005, 579, 4485–4491. [Google Scholar]
- Kanda, T.; Akiyama, H.; Yanagida, A.; Tanabe, M.; Goda, Y.; Toyoda, M.; Teshima, R.; Saito, Y. Inhibitory effects of apple polyphenol on induced histamine release from RBL-2H3 cells and rat mast cells. Biosci. Biotechnol. Biochem 1998, 62, 1284–1289. [Google Scholar]
- Kowluru, R.A.; Kanwar, M. Effects of curcumin on retinal oxidative stress and inflammation in diabetes. Nutr Metab (London) 2007, 4, 8. [Google Scholar]
- Johnston, K.; Sharp, P.; Clifford, M.; Morgan, L. Dietary polyphenols decrease glucose uptake by human intestinal Caco-2 cells. FEBS Lett 2005, 579, 1653–1657. [Google Scholar]
- Kobayashi, Y.; Suzuki, M.; Hideo, S.; Arai, S.; Yukihiko, H.; Suzuki, K.; Miyamoto, Y.; Shimizu, M. Green tea polyphenols inhibit the sodium-dependent glucose transporter of intestinal epithelial cells by a competitive mechanism. J. Agric. Food Chem 2000, 48, 5618–5623. [Google Scholar]
- Song, J.; Kwon, O.; Chen, S.; Daruwala, R.; Eck, P.; Park, J.B.; Levine, M. Flavonoid inhibition of SVCT1 and GLUT2, intestinal transporters for vitamin C and glucose. J. Biol. Chem 2002, 277, 15252–15260. [Google Scholar]
- Yoshikawa, M.; Nishida, N.; Shimoda, H.; Takada, M.; Kawahara, Y.; Matsuda, H. Polyphenol constituents from Salacia species: quantitative analysis of mangiferin with alpha-glucosidase and aldose reductase inhibitory activities. Yakugaku Zasshi 2001, 121, 371–378. [Google Scholar]
- McDougall, GJ.; Shpiro, F.; Dobson, P.; Smith, P.; Blake, A.; Stewart, D. Different polyphenolic components of soft fruits inhibits alpha-amylase and alpha-glucosidase. J. Agric. Food Chem 2005, 53, 2760–2766. [Google Scholar]
- Bhat, K.P.; Lantvit, D.; Christov, K.; Mehta, R.G.; Moon, R.C.; Pezzuto, J.M. Estrogenic and antiestrogenic properties of resveratrol in mammary tumor models. Cancer Res 2001, 61, 7456–7463. [Google Scholar]
- Bhat, K.P.; Pezzuto, J.M. Resveratrol exhibits cytostatic and antiestrogenic properties with human endometrial adenocarcinoma (Ishikawa) cells. Cancer Res 2001, 61, 6137–6144. [Google Scholar]
- Otake, Y.; Nolan, A.L.; Walle, U.K.; Walle, T. Quercetin and resveratrol potently reduce estrogen sulfotransferase activity in normal human mammary epithelial cells. J. Steroid Biochem. Mol. Biol 2000, 73, 265–270. [Google Scholar]
- Reidenberg, M.M. Environmental inhibition of 11b-hydroxysteroid dehydrogenase. Toxicology 2000, 144, 107–111. [Google Scholar]
- Kuo, P.L.; Chiang, L.C.; Lin, C.C. Resveratrol-induced apoptosis is mediated by p53-dependent pathway in HepG2 cells. Life Sci 2002, 72, 23–34. [Google Scholar]
- Ahmad, N.; Adhami, V. M.; Afaq, F.; Feyes, D. K.; Mukhtar, H. Resveratrol causesWAF-1/p21-mediated G (1)-phase arrest of cell cycle and induction of apoptosis in human epidermoid carcinoma A431 cells. Clin. Cancer Res 2001, 7, 1466–1473. [Google Scholar]
- Wolter, F.; Akoglu, B.; Clausnitzer, A.; Stein, J. Downregulation of the cyclin D1/Cdk4 complex occurs during resveratrol-induced cell cycle arrest in colon cancer cell lines. J. Nutr 2001, 131, 2197–2203. [Google Scholar]
- Adhami, V.M.; Afaq, F.; Ahmad, N. Involvement of the retinoblastoma (pRb)-E2F/DP pathway during antiproliferative effects of resveratrol in human epidermoid carcinoma (A431) cells. Biochem. Biophys. Res. Commun 2001, 288, 579–585. [Google Scholar]
- Joe, A.K.; Liu, H.; Suzui, M.; Vural, M. E.; Xiao, D.; Weinstein, I. B. Resveratrol induces growth inhibition, S-phase arrest, apoptosis, and changes in biomarker expression in several human cancer cell lines. Clin. Cancer Res 2002, 8, 893–903. [Google Scholar]
- Poussier, B.; Cordova, A.C.; Becquemin, J.P.; Sumpio, B.E. Resveratrol inhibits vascular smooth muscle cell proliferation and induces apoptosis. J. Vasc. Surg 2005, 42, 1190–1197. [Google Scholar]
- Larrosa, M; Tomas-Barberan, F.A.; Espin, J.C. Grape polyphenol resveratrol and the related molecule 4-hydroxystilbene induce growth inhibition, apoptosis, S-phase arrest, and upregulation of cyclins A, E, and B1 in human SK-Mel-28 melanoma cells. J. Agric. Food Chem 2003, 51, 4576–4584. [Google Scholar]
- Meeran, S.M.; Katiyar, S.K. Grape seed proanthocyanidins promote apoptosis in human epidermoid carcinoma A431 cells through alterations in Cdki-Cdk-cyclin cascade, and caspase-3 activation via loss of mitochondrial membrane potential. Exp. Dermatol 2007, 16, 405–415. [Google Scholar]
- Donnelly, L.E.; Newton, R.; Kennedy, G.E.; Fenwick, P.S.; Leung, R.H.; Ito, K.; Russell, R.E.; Barnes, P.J. Anti-inflammatory effects of resveratrol in lung epithelial cells: molecular mechanisms. Am. J. Physiol. Lung Cell Mol. Physiol 2004, 287, L774–L783. [Google Scholar]
- Rahman, I.; Kilty, I. Antioxidant therapeutic targets in COPD. Curr. Drug Targets 2006, 7, 707–720. [Google Scholar]
- Xu, M.; Deng, B.; Chow, Y.L.; Zhao, Z.Z.; Hu, B. Effects of curcumin in treatment of experimental pulmonary fibrosis: a comparison with hydrocortisone. J Ethnopharmacol 2007, 112, 292–299. [Google Scholar]
- Sakagami, Y.; Sawabe, A.; Komemushi, S.; All, Z.; Tanaka, T.; Iliya, I.; Iinuma, M. Antibacterial activity of stilbene oligomers against vancomycin-resistant Enterococci (VRE) and methicillin-resistant Staphylococcus aureus (MRSA) and their synergism with antibiotics. Biocontrol Sci 2007, 12, 7–14. [Google Scholar]
- Furneri, P.M.; Piperno, A.; Sajia, A.; Bisignano, G. Antimycoplasmal activity of hydroxytyrosol. Antimicrob. Agents Chemother 2004, 48, 4892–4894. [Google Scholar]
- Nair, M.P.; Kandaswami, C.; Mahajan, S.; Nair, H.N.; Chawda, R.; Shanahan, T.; Schwartz, S.A. Grape seed extract proanthocyanidins downregulate HIV-1 entry coreceptors, CCR2b, CCR3 and CCR5 gene expression by normal peripheral blood mononuclear cells. Biol. Res 2002, 35, 421–431. [Google Scholar]
- Khanna, S.; Roy, S.; Bagchi, D.; Bagchi, M.; Sen, C.K. Upregulation of oxidant-induced VEGF expression in cultured keratinocytes by a grape seed proanthocyanidin extract. Free Radic. Biol. Med 2001, 31, 38–42. [Google Scholar]
- Ray, S.D.; Kumar, M.A.; Bagchi, D. A novel proanthocyanidin IH636 grape seed extract increases in vivo Bcl-XL expression and prevents acetaminophen-induced programmed and unprogrammed cell death in mouse liver. Arch. Biochem. Biophys 1999, 369, 42–58. [Google Scholar]
- Wong, M.C.; Portmann, B.; Sherwood, R.; Niemela, O.; Koivisto, H.; Parkkila, S.; Trick, K.; L’abbe, M.R.; Wilson, J.; Dash, P.R.; Srirajaskanthan, R.; Preedy, V.R.; Wiseman, H. The cytoprotective effect of alpha-tocopherol and daidzein against d-galactosamine-induced oxidative damage in the rat liver. Metabolism 2007, 56, 865–875. [Google Scholar]
- Kuzu, N.; Metin, K.; Dagli, A.F.; Akdemir, F.; Orhan, C.; Yalniz, M.; Ozercan, I.H.; Sahin, K.; Bahcecioglu, I.H. Protective role of genistein in acute liver damage induced by carbon tetrachloride. Mediators Inflamm 2007, 2007, 36381. [Google Scholar]
- Rucinska, A.; Kirko, S.; Gabryelak, T. Effect of the phytoestrogen, genistein-8-C-glucoside, on Chinese hamster ovary cells in vitro. Cell Biol. Int 2007, in press. [Google Scholar]
- Galati, G.; Sabzevari, O.; Wilson, J.X.; O’Brien, P.J. Prooxidant activity and cellular effects of the phenoxyl radicals of dietary flavonoids and other polyphenolics. Toxicology 2002, 177, 91–104. [Google Scholar]
- Rietjens, I.M.C.M.; Boersma, M.G.; de Haan, L.; Spenkelink, B.; Awad, H.M.; Cnubben, N.H.P.; van Zanden, J.J.; van der Woude, H.; Alink, G.M.; Koeman, J. H. The pro-oxidant chemistry of the natural antioxidants vitamin C, vitamin E, carotenoids and flavonoids. Environ. Toxicol. Pharmacol 2002, 11, 321–333. [Google Scholar]
- Huisman, A.; van de Wiel, A.; Rabelink, T.J.; van Faassen, E.E. Wine polyphenols and ethanol do not significantly scavenge superoxide nor affect endothelial nitric oxide production. J. Nutr. Biochem 2004, 15, 426–432. [Google Scholar]
- Sakihama, Y.; Cohen, M.F.; Grace, S.C.; Yamasaki, H. Plant phenolic antioxidant and prooxidant activities: phenolics-induced oxidative damage mediated by metals in plants. Toxicology 2002, 177, 67–80. [Google Scholar]
- Fujisawa, S.; Atsumi, T.; Ishihara, M.; Kadoma, Y. Cytotoxicity, ROS-generation activity and radical-scavenging activity of curcumin and related compounds. Anticancer Res 2004, 24, 563–569. [Google Scholar]
- Nemeikaite-Ceniene, A.; Imbrasaite, A.; Sergediene, E.; Cenas, N. Quantitative structure-activity relationships in prooxidant cytotoxicity of polyphenols: role of potential of phenoxyl radical/phenol redox couple. Arch. Biochem. Biophys 2005, 441, 182–190. [Google Scholar]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: food sources and bioavailability. Am. J. Clin. Nutr 2004, 79, 727–747. [Google Scholar]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr 2005, 81, 230S–242S. [Google Scholar]
- Gonthier, M.P.; Donovan, J.L.; Texier, O.; Felgines, C.; Remesy, C.; Scalbert, A. Metabolism of dietary procyanidins in rats. Free Radic. Biol. Med 2003, 35, 837–844. [Google Scholar]
- Seeram, N.P.; Lee, R.; Heber, D. Bioavailability of ellagic acid in human plasma after consumption of ellagitannins from pomegranate (Punica granatum L.) juice. Clin. Chim. Acta 2004, 348, 63–68. [Google Scholar]
- Chow, H.H.; Hakim, I.A.; Vining, D.R.; Crowell, J.A.; Ranger-Moore, J.; Chew, W.M.; Celaya, C.A.; Rodney, S.R.; Hara, Y.; Alberts, D.S. Effects of dosing condition on the oral bioavailability of green tea catechins after single-dose administration of Polyphenon E in healthy individuals. Clin. Cancer Res 2005, 11, 4627–4633. [Google Scholar]
- Henning, S.M.; Niu, Y.; Lee, N.H.; Thames, G.D.; Minutti, R.R.; Wang, H.; Go, V.L.; Heber, D. Bioavailability and antioxidant activity of tea flavanols after consumption of green tea, black tea, or a green tea extract supplement. Am. J. Clin. Nutr 2004, 80, 1558–1564. [Google Scholar]
- Henning, S.M.; Niu, Y.; Liu, Y.; Lee, N.H.; Hara, Y.; Thames, G.D.; Minutti, R.R.; Carpenter, C.L.; Wang, H.; Heber, D. Bioavailability and antioxidant effect of epigallocatechin gallate administered in purified form versus as green tea extract in healthy individuals. J. Nutr. Biochem 2005, 16, 610–616. [Google Scholar]
- Visioli, F.; Galli, C.; Bornet, F.; Mattei, A.; Patelli, R.; Galli, G.; Caruso, D. Olive oil phenolics are dose-dependently absorbed in humans. FEBS Lett 2000, 468, 159–160. [Google Scholar]
- Rasmussen, S.E.; Breinholt, V.M. Non-nutritive bioactive food constituents of plants: bioavailability of flavonoids. Int. J. Vitam. Nutr. Res 2003, 73, 101–111. [Google Scholar]
- Covas, M.I. Olive oil and the cardiovascular system. Pharmacol. Res 2007, 55, 175–186. [Google Scholar]
- Miro-Casas, E.; Covas, M.I.; Farre, M.; Fito, M.; Ortuño, J.; Weinbrenner, T.; Roset, P.; de la Torre, R. Hydroxytyrosol disposition in humans. Clin. Chem 2003, 49, 945–952. [Google Scholar]
- Caruso, D.; Visioli, F.; Patelli, R.; Galli, C.; Galli, G. Urinary excretion of olive oil phenols and their metabolites in humans. Metabolism 2001, 50, 1426–1428. [Google Scholar]
- Tuck, K.L.; Hayball, P.J.; Stupans, I. Structural characterization of the metabolites of hydroxytyrosol, the principal phenolic component in olive oil in rats. J. Agric. Food Chem 2002, 50, 2404–2409. [Google Scholar]
- Corona, G.; Tzounis, X.; Assunta Dessi, M.; Deiana, M.; Debnam, E.S.; Visioli, F.; Spencer, J.P. The fate of olive oil polyphenols in the gastrointestinal tract: implications of gastric and colonic microflora-dependent biotransformation. Free Radic. Res 2006, 40, 647–658. [Google Scholar]
- de la Torre-Carbot, K.; Jauregui, O.; Castellote, A.I.; Lamuela-Raventós, R.M.; Covas, M.I.; Casals, I.; López-Sabater, M.C. Rapid high-performance liquid chromatography-electrospray ionization tandem mass spectrometry method for qualitative and quantitative analysis of virgin olive oil metabolites in human low-density lipoproteins. J. Chromatogr. A 2006, 1116, 69–75. [Google Scholar]
- Carbonaro, M.; Grant, G.; Pusztai, A. Evaluation of polyphenol bioavailability in rat small intestine. Eur. J. Nutr 2001, 40, 84–90. [Google Scholar]
© 2007 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Han, X.; Shen, T.; Lou, H. Dietary Polyphenols and Their Biological Significance. Int. J. Mol. Sci. 2007, 8, 950-988. https://doi.org/10.3390/i8090950
Han X, Shen T, Lou H. Dietary Polyphenols and Their Biological Significance. International Journal of Molecular Sciences. 2007; 8(9):950-988. https://doi.org/10.3390/i8090950
Chicago/Turabian StyleHan, Xiuzhen, Tao Shen, and Hongxiang Lou. 2007. "Dietary Polyphenols and Their Biological Significance" International Journal of Molecular Sciences 8, no. 9: 950-988. https://doi.org/10.3390/i8090950
APA StyleHan, X., Shen, T., & Lou, H. (2007). Dietary Polyphenols and Their Biological Significance. International Journal of Molecular Sciences, 8(9), 950-988. https://doi.org/10.3390/i8090950




