Increased Plasma Levels of Thrombin-Cleaved Osteopontin in Patients with Delayed Cerebral Infarction After Aneurysmal Subarachnoid Hemorrhage
Abstract
1. Introduction
2. Results
2.1. Clinical Variables of Patients With or Without DCIn
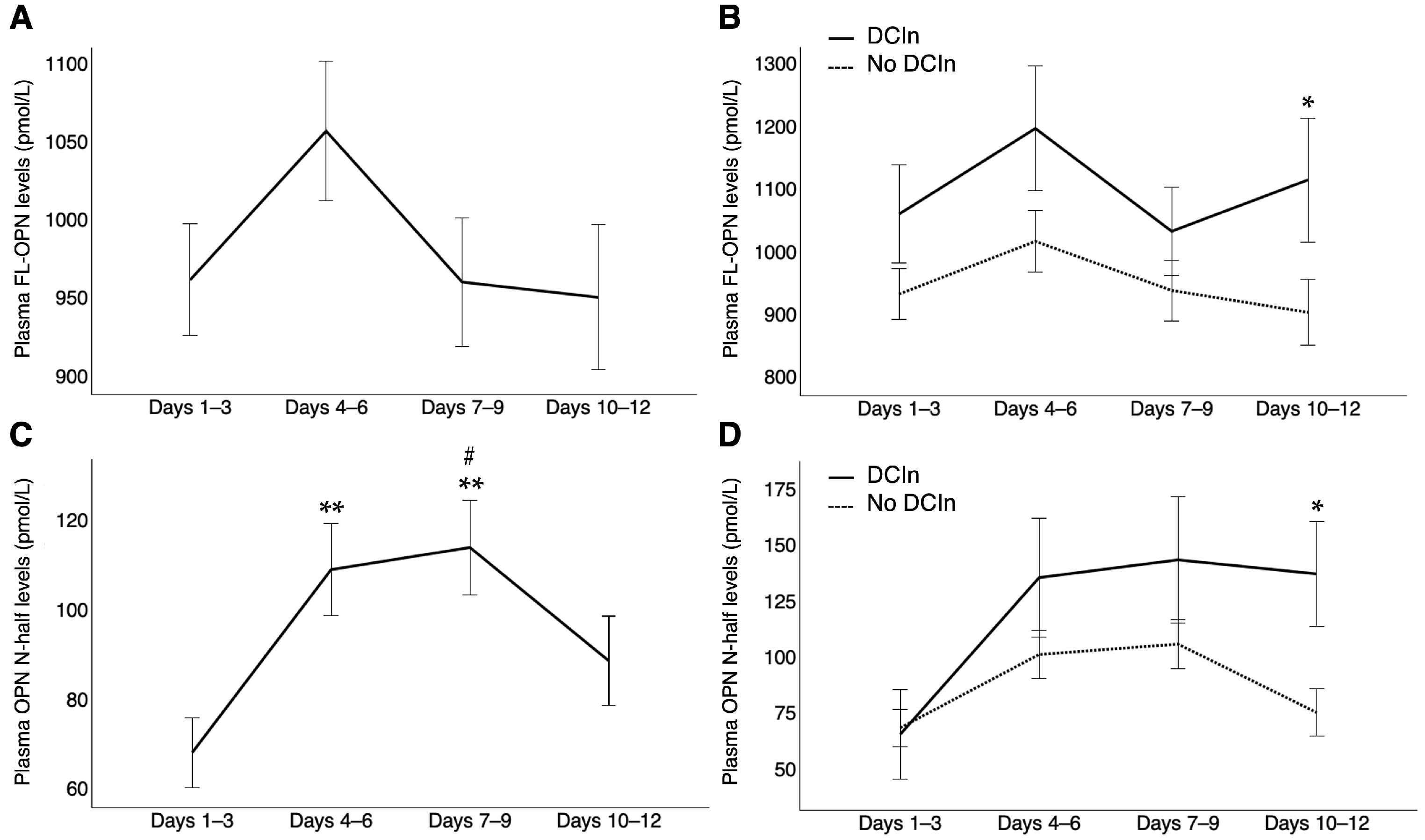
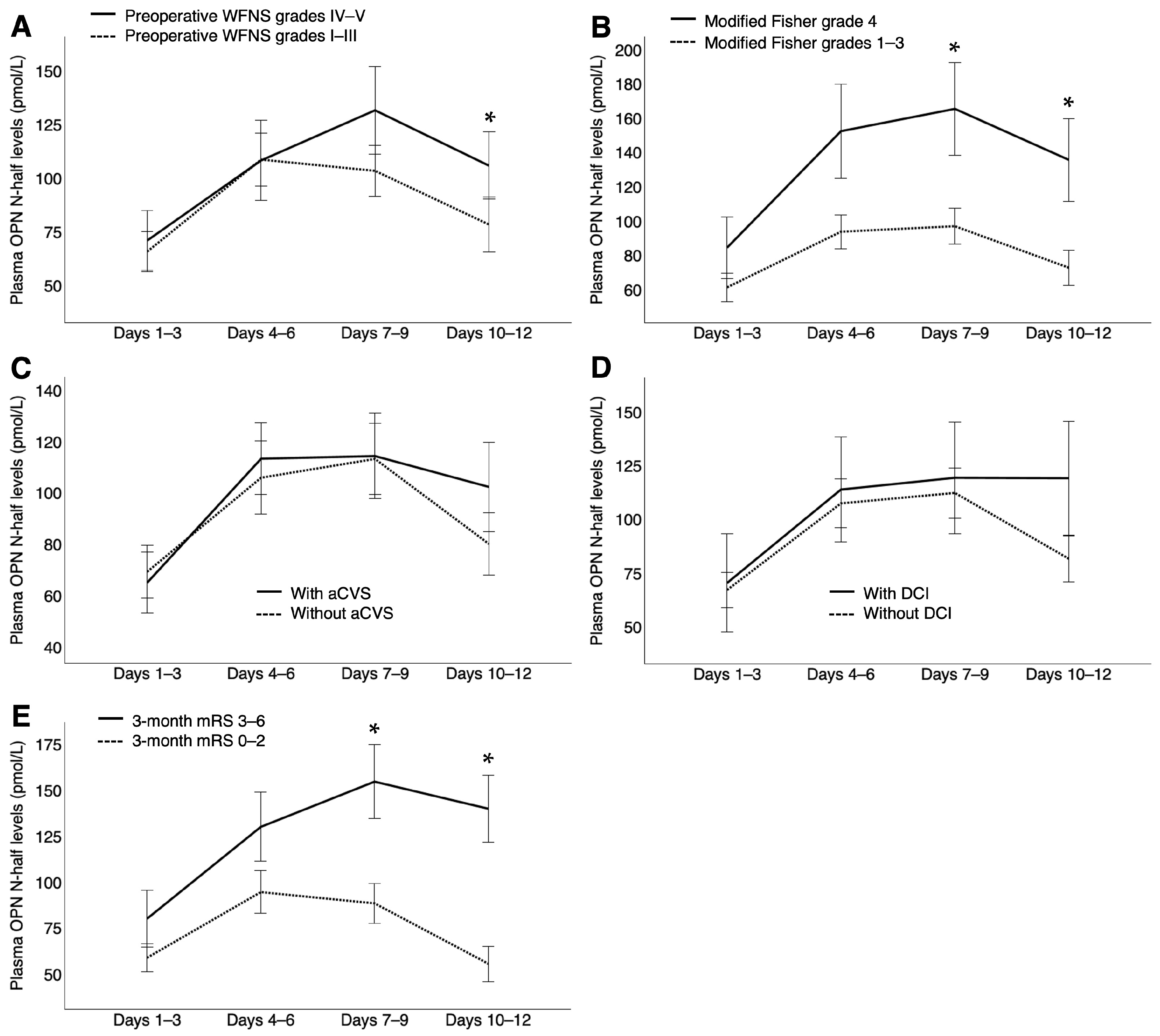
2.2. Plasma FL-OPN and OPN N-Half Levels
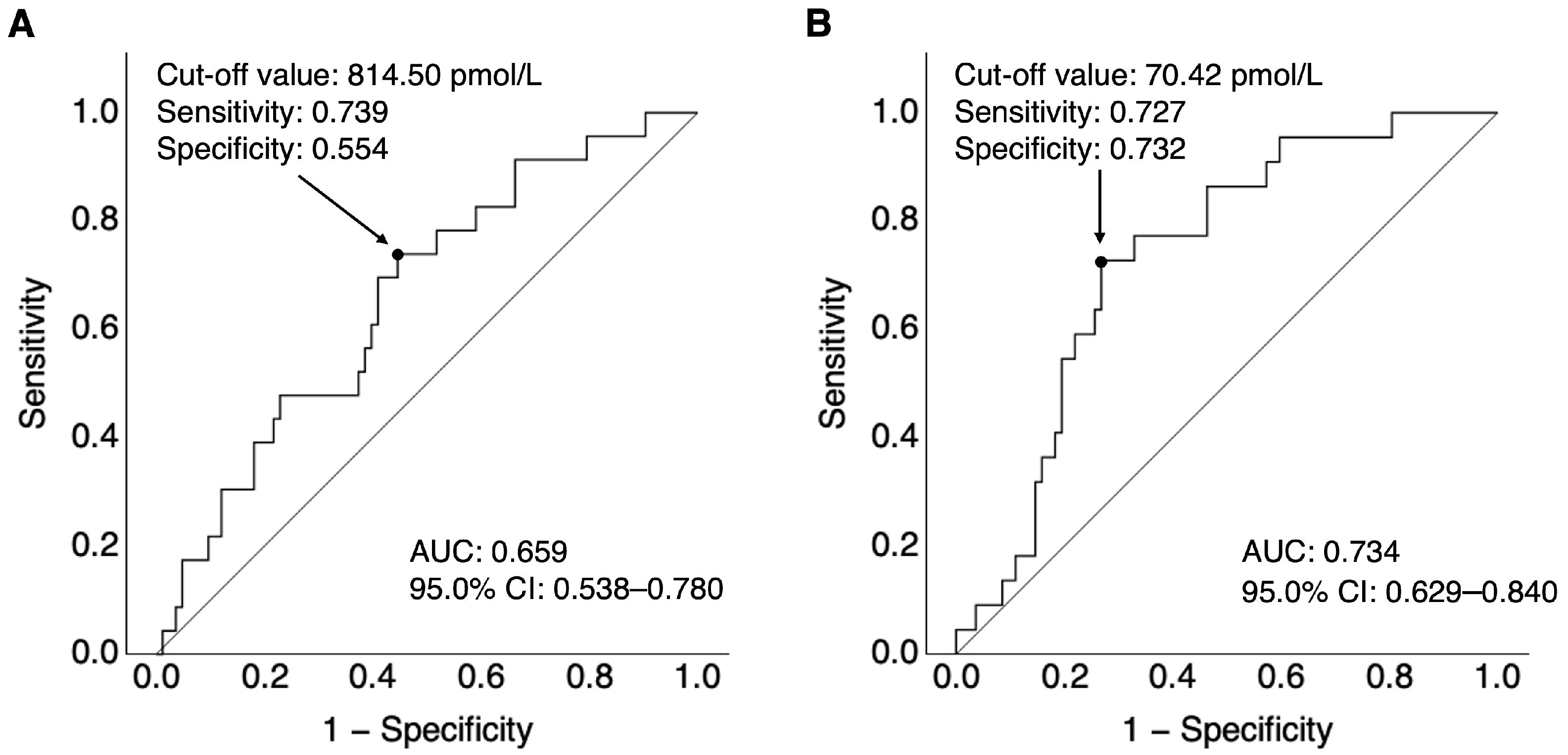
2.3. Multivariate Analysis Using DCIn as the Dependent Variable
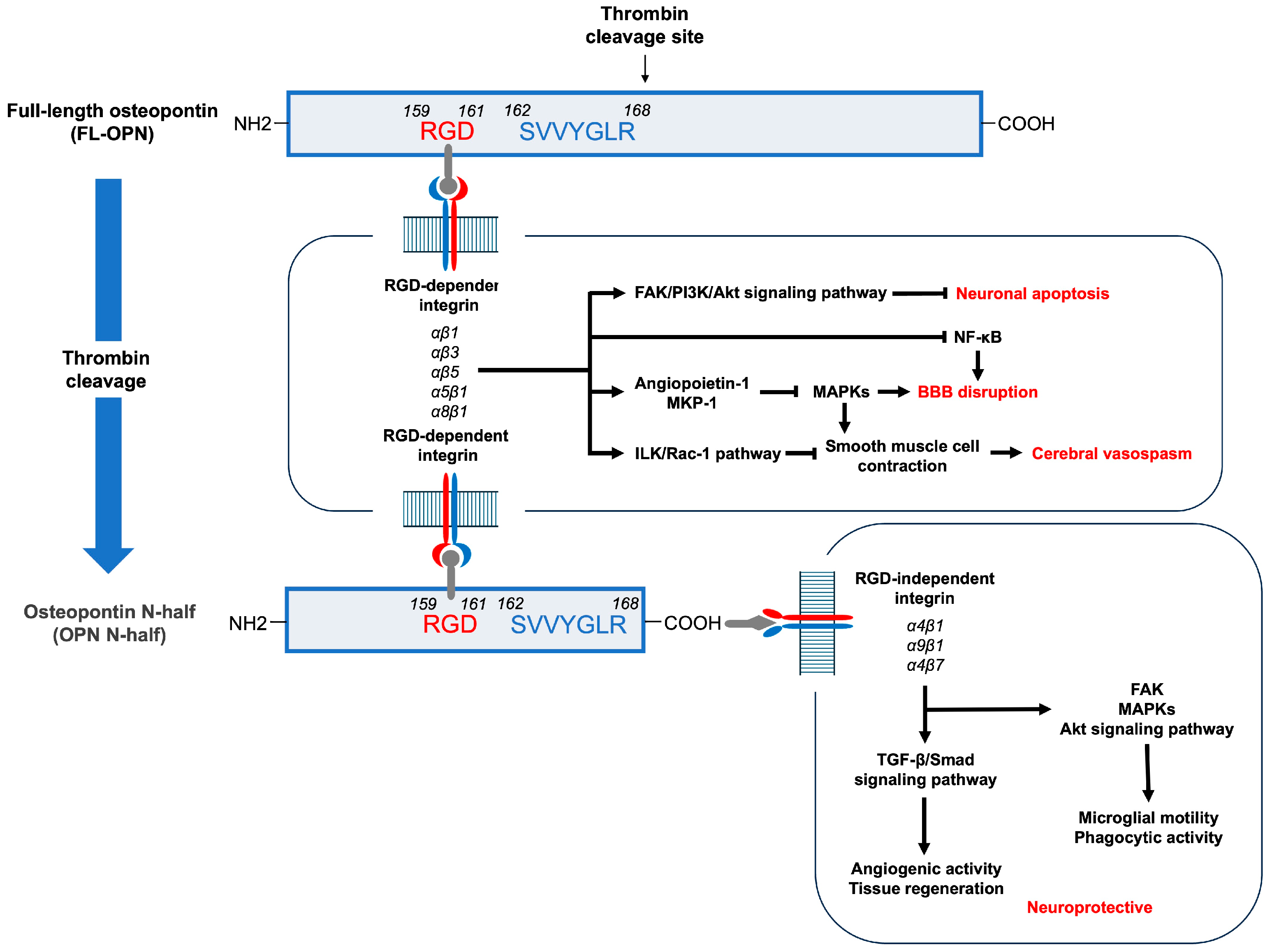
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Clinical Variables
4.3. Measurement of Plasma OPN Levels
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A
References
- Toyoda, K.; Yoshimura, S.; Nakai, M.; Koga, M.; Sasahara, Y.; Sonoda, K.; Kamiyama, K.; Yazawa, Y.; Kawada, S.; Sasaki, M.; et al. Twenty-year change in severity and outcome of ischemic and hemorrhagic strokes. JAMA Neurol. 2022, 79, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Charalambous, L.T.; Adil, S.M.; Rajkumar, S.; Gramer, R.; Kirsch, E.; Liu, B.; Zomorodi, A.; McClellan, M.; Lad, S.P. A nationwide analysis of aneurysmal subarachnoid hemorrhage mortality, complications, and health economics in the USA. Transl. Stroke Res. 2023, 14, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Schupper, A.J.; Hardigan, T.A.; Mehta, A.; Yim, B.; Yaeger, K.A.; De Leacy, R.; Fifi, J.T.; Mocco, J.; Majidi, S. Sex and racial disparity in outcome of aneurysmal subarachnoid hemorrhage in the United States: A 20-year analysis. Stroke 2023, 54, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Vergouwen, M.D.I.; Vermeulen, M.; van Gijn, J.; Rinkel, G.J.E.; Wijdicks, E.F.; Muizelaar, J.P.; Mendelow, A.D.; Juvela, S.; Yonas, H.; Terbrugge, K.G.; et al. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: Proposal of a multidisciplinary research group. Stroke 2010, 41, 2391–2395. [Google Scholar] [CrossRef]
- Lauzier, D.C.; Jayaraman, K.; Yuan, J.Y.; Diwan, D.; Vellimana, A.K.; Osbun, J.W.; Chatterjee, A.R.; Athiraman, U.; Dhar, R.; Zipfel, G.J. Early brain injury after subarachnoid hemorrhage: Incidence and mechanisms. Stroke 2023, 54, 1426–1440. [Google Scholar] [CrossRef]
- Suzuki, H.; Kanamaru, H.; Kawakita, F.; Asada, R.; Fujimoto, M.; Shiba, M. Cerebrovascular pathophysiology of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. Histol. Histopathol. 2021, 36, 143–158. [Google Scholar]
- Suzuki, H.; Nakajima, H.; Ichikawa, T.; Yasuda, R.; Okada, T.; Goto, F.; Ito, S.; Horiuchi, Y.; Kitano, Y.; Nishikawa, H.; et al. Beneficial effects of clazosentan add-on treatment on delayed cerebral microcirculatory disturbances after aneurysmal subarachnoid hemorrhage. Brain Hemorrhages 2024, 5, 62–68. [Google Scholar] [CrossRef]
- Suzuki, H.; Kawakita, F.; Asada, R. Neuroelectric mechanisms of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. Int. J. Mol. Sci. 2022, 23, 3102. [Google Scholar] [CrossRef]
- Asada, R.; Suzuki, H. Osteopontin in post-subarachnoid hemorrhage pathologies. J. Integr. Neurosci. 2022, 21, 62. [Google Scholar] [CrossRef]
- Icer, M.A.; Gezmen-Karadag, M. The multiple functions and mechanisms of osteopontin. Clin. Biochem. 2018, 59, 17–24. [Google Scholar] [CrossRef]
- Christensen, B.; Petersen, T.E.; Sørensen, E.S. Post-translational modification and proteolytic processing of urinary osteopontin. Biochem. J. 2008, 411, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Yokosaki, Y.; Tanaka, K.; Higashikawa, F.; Yamashita, K.; Eboshida, A. Distinct structural requirements for binding of the integrins αvβ6, αvβ3, αvβ5, α5β1 and α9β1 to osteopontin. Matrix Biol. 2005, 24, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Kon, S.; Nakayama, Y.; Kurotaki, D.; Saito, Y.; Kanayama, M.; Kimura, C.; Diao, H.; Morimoto, J.; Matsui, Y.; et al. The differential amino acid requirement within osteopontin in α4 and α9 integrin-mediated cell binding and migration. Matrix Biol. 2009, 28, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Topkoru, B.C.; Altay, O.; Duris, K.; Krafft, P.R.; Yan, J.; Zhang, J.H. Nasal administration of recombinant osteopontin attenuates early brain injury after subarachnoid hemorrhage. Stroke 2013, 44, 3189–3194. [Google Scholar] [CrossRef]
- He, J.; Liu, M.; Liu, Z.; Luo, L. Recombinant osteopontin attenuates experimental cerebral vasospasm following subarachnoid hemorrhage in rats through an anti-apoptotic mechanism. Brain Res. 2015, 1611, 74–83. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, Y.; Yang, P.; Enkhjargal, B.; Manaenko, A.; Tang, J.; Pearce, W.J.; Hartman, R.; Obenaus, A.; Chen, G.; et al. Recombinant osteopontin stabilizes smooth muscle cell phenotype via integrin receptor/integrin-linked kinase/Rac-1 pathway after subarachnoid hemorrhage in rats. Stroke 2016, 47, 1319–1327. [Google Scholar] [CrossRef]
- Sun, C.M.; Enkhjargal, B.; Reis, C.; Zhou, K.R.; Xie, Z.Y.; Wu, L.Y.; Zhang, T.Y.; Zhu, Q.Q.; Tang, J.P.; Jiang, X.D.; et al. Osteopontin attenuates early brain injury through regulating autophagy-apoptosis interaction after subarachnoid hemorrhage in rats. CNS Neurosci. Ther. 2019, 25, 1162–1172. [Google Scholar] [CrossRef]
- Sun, C.; Enkhjargal, B.; Reis, C.; Zhang, T.; Zhu, Q.; Zhou, K.; Xie, Z.; Wu, L.; Tang, J.; Jiang, X.; et al. Osteopontin-enhanced autophagy attenuates early brain injury via FAK-ERK pathway and improves long-term outcome after subarachnoid hemorrhage in rats. Cells 2019, 8, 980. [Google Scholar] [CrossRef]
- Sun, C.; Rahman, M.S.U.; Enkhjargal, B.; Peng, J.; Zhou, K.; Xie, Z.; Wu, L.; Zhang, T.; Zhu, Q.; Tang, J.; et al. Osteopontin modulates microglial activation states and attenuates inflammatory responses after subarachnoid hemorrhage in rats. Exp. Neurol. 2024, 371, 114585. [Google Scholar] [CrossRef]
- Asada, R.; Nakatsuka, Y.; Kanamaru, H.; Kawakita, F.; Fujimoto, M.; Miura, Y.; Shiba, M.; Yasuda, R.; Toma, N.; Suzuki, H.; et al. Higher plasma osteopontin concentrations associated with subsequent development of chronic shunt-dependent hydrocephalus after aneurysmal subarachnoid jemorrhage. Transl. Stroke Res. 2021, 12, 808–816. [Google Scholar] [CrossRef]
- Suzuki, H.; Nishikawa, H.; Kawakita, F. Matricellular proteins as possible biomarkers for early brain injury after aneurysmal subarachnoid hemorrhage. Neural Regen. Res. 2018, 13, 1175–1178. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuka, Y.; Shiba, M.; Nishikawa, H.; Terashima, M.; Kawakita, F.; Fujimoto, M.; Suzuki, H.; pSEED group. Acute-phase plasma osteopontin as an independent predictor for poor outcome after aneurysmal subarachnoid hemorrhage. Mol. Neurobiol. 2018, 55, 6841–6849. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Wang, J.; Chen, J.; Lin, W.; Shen, H.; Fang, Y.; Yu, W. Multifaceted role of thrombin in subarachnoid hemorrhage: Focusing on cerebrospinal fluid circulation disorder. Exp. Neurol. 2025, 383, 115036. [Google Scholar] [CrossRef] [PubMed]
- Nampei, M.; Suzuki, Y.; Nakajima, H.; Oinaka, H.; Kawakita, F.; Suzuki, H.; pSEED Group. Acute-phase plasma pigment epithelium-derived factor predicting outcomes after aneurysmal subarachnoid hemorrhage in the elderly. Int. J. Mol. Sci. 2024, 25, 1701. [Google Scholar] [CrossRef]
- Oinaka, H.; Kawakita, F.; Nakajima, H.; Suzuki, Y.; Nampei, M.; Okada, T.; Yasuda, R.; Toma, N.; Suzuki, H.; pSEED Group. Increased plasma periostin concentration predicts angiographic vasospasm development in non-severe aneurysmal subarachnoid hemorrhage. Brain Hemorrhages 2024, 5, 1–7. [Google Scholar] [CrossRef]
- Nampei, M.; Kawakita, F.; Asada, R.; Suzuki, Y.; Nakajima, H.; Oinaka, H.; Suzuki, H.; pSEED group. Increased plasma pigment epithelium-derived factor (PEDF) concentrations as a negative predictor of angiographic vasospasm after aneurysmal subarachnoid hemorrhage. Brain Hemorrhages 2024, 5, 169–176. [Google Scholar] [CrossRef]
- Zhou, Y.; Yao, Y.; Sheng, L.; Zhang, J.; Zhang, J.H.; Shao, A. Osteopontin as a candidate of therapeutic application for the acute brain injury. J. Cell. Mol. Med. 2020, 24, 8918–8929. [Google Scholar] [CrossRef]
- Chen, S.; Li, L.; Peng, C.; Bian, C.; Ocak, P.E.; Zhang, J.H.; Yang, Y.; Zhou, D.; Chen, G.; Luo, Y. Targeting oxidative stress and inflammatory response for blood-brain barrier protection in intracerebral hemorrhage. Antioxid. Redox Signal. 2022, 37, 115–134. [Google Scholar] [CrossRef]
- Clarke, J.V.; Suggs, J.M.; Diwan, D.; Lee, J.V.; Lipsey, K.; Vellimana, A.K.; Zipfel, G.J. Microvascular platelet aggregation and thrombosis after subarachnoid hemorrhage: A review and synthesis. J. Cereb. Blood Flow Metab. 2020, 40, 1565–1575. [Google Scholar] [CrossRef]
- Dashdulam, D.; Kim, I.D.; Lee, H.; Lee, H.K.; Kim, S.W.; Lee, J.K. Osteopontin heptamer peptide containing the RGD motif enhances the phagocytic function of microglia. Biochem. Biophys. Res. Commun. 2020, 524, 371–377. [Google Scholar] [CrossRef]
- Lee, H.; Jin, Y.C.; Kim, S.W.; Kim, I.D.; Lee, H.K.; Lee, J.K. Proangiogenic functions of an RGD-SLAY-containing osteopontin icosamer peptide in HUVECs and in the postischemic brain. Exp. Mol. Med. 2018, 50, e430. [Google Scholar] [CrossRef] [PubMed]
- Davaanyam, D.; Kim, I.D.; Lee, J.K. Intranasal delivery of RGD-containing osteopontin heptamer peptide confers neuroprotection in the ischemic brain and augments microglia M2 polarization. Int. J. Mol. Sci. 2021, 22, 9999. [Google Scholar] [CrossRef] [PubMed]
- Doyle, K.P.; Yang, T.; Lessov, N.S.; Ciesielski, T.M.; Stevens, S.L.; Simon, R.P.; King, J.S.; Stenzel-Poore, M.P. Nasal administration of osteopontin peptide mimetics confers neuroprotection in stroke. J. Cereb. Blood Flow Metab. 2008, 28, 1235–1248. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Hamada, Y.; Yokoyama, Y.; Yamamoto, H.; Kogo, M. Osteopontin-derived synthetic peptide SVVYGLR upregulates functional regeneration of oral and maxillofacial soft-tissue injury. Jpn. Dent. Sci. Rev. 2021, 57, 174–181. [Google Scholar] [CrossRef]
- Kim, I.D.; Lee, H.; Jin, Y.C.; Lee, J.K. Osteopontin peptide icosamer containing RGD and SLAYGLR motifs enhances the motility and phagocytic activity of microglia. Exp. Neurobiol. 2017, 26, 339–349. [Google Scholar] [CrossRef]
- Abate, M.G.; Moretto, L.; Licari, I.; Esposito, T.; Capuano, L.; Olivieri, C.; Benech, A.; Brucoli, M.; Avanzi, G.C.; Cammarota, G.; et al. Osteopontin in the cerebrospinal fluid of patients with severe aneurysmal subarachnoid hemorrhage. Cells 2019, 8, 695. [Google Scholar] [CrossRef]
- Ji, Y.; Meng, Q.H.; Wang, Z.G. Changes in the coagulation and fibrinolytic system of patients with subarachnoid hemorrhage. Neurol. Med. Chir. 2014, 54, 457–464. [Google Scholar] [CrossRef]
- Chou, S.H.; Macdonald, R.L.; Keller, E. Biospecimens and molecular and cellular biomarkers in aneurysmal subarachnoid hemorrhage studies: Common data elements and standard reporting recommendations. Neurocrit. Care 2019, 30, S46–S59. [Google Scholar] [CrossRef]
- Batista, S.; Bocanegra-Becerra, J.E.; Claassen, B.; Rubião, F.; Rabelo, N.N.; Figueiredo, E.G.; Oberman, D.Z. Biomarkers in aneurysmal subarachnoid hemorrhage: A short review. World Neurosurg. X 2023, 19, 100205. [Google Scholar] [CrossRef]
- Hattori, T.; Iwasaki-Hozumi, H.; Bai, G.; Chagan-Yasutan, H.; Shete, A.; Telan, E.F.; Takahashi, A.; Ashino, Y.; Matsuba, T. Both full-length and protease-cleaved products of osteopontin are elevated in infectious diseases. Biomedicines 2021, 9, 1006. [Google Scholar] [CrossRef]
- Frontera, J.A.; Claassen, J.; Schmidt, J.M.; Wartenberg, K.E.; Temes, R.; Connolly, E.S., Jr.; MacDonald, R.L.; Mayer, S.A. Prediction of symptomatic vasospasm after subarachnoid hemorrhage: The modified fisher scale. Neurosurgery 2006, 59, 21–27. [Google Scholar] [PubMed]
- Kim, H.Y. Statistical notes for clinical researchers: Chi-squared test and Fisher’s exact test. Restor. Dent. Endod. 2017, 42, 152–155. [Google Scholar] [CrossRef] [PubMed]


| Variables | Total (n = 108) | DCIn (n = 23) | No DCIn (n = 85) | p Value |
|---|---|---|---|---|
| Age, mean ± SD (years) | 63.6 ± 14.0 | 68.6 ± 12.2 | 62.3 ± 14.2 | 0.054 a |
| Age ≥ 75 years old | 23 (21.3%) | 7 (30.4%) | 16 (18.8%) | 0.256 b |
| Sex, female | 76 (70.4%) | 17 (73.9%) | 59 (69.4%) | 0.871 c |
| Underlying condition | ||||
| Cerebral infarction | 6 (5.6%) | 1 (4.3%) | 5 (5.9%) | 1.000 b |
| Hypertension | 43 (39.8%) | 9 (39.1%) | 34 (40.0%) | 1.000 c |
| Dyslipidemia | 12 (11.1%) | 1 (4.3%) | 11 (12.9%) | 0.455 b |
| Diabetes mellitus | 6 (5.6%) | 0 (0.0%) | 6 (7.1%) | 0.388 b |
| Family history of SAH | 11 (10.2%) | 0 (0.0%) | 11 (12.9%) | 0.116 b |
| Smoking history | 25 (23.1%) | 6 (26.1%) | 19 (22.4%) | 0.992 c |
| Regular alcohol | 18 (16.7%) | 4 (17.4%) | 14 (16.5%) | 1.000 b |
| mRS before onset | ||||
| 0 | 98 (90.7%) | 21 (91.3%) | 77 (90.6%) | 1.000 b |
| 1 | 9 (8.3%) | 1 (4.3%) | 8 (9.4%) | 0.681 b |
| 2 | 1 (0.9%) | 1 (4.3%) | 0 (0.0%) | 0.213 b |
| Preoperative WFNS grade | ||||
| I | 28 (25.9%) | 3 (13.0%) | 25 (29.4%) | 0.187 c |
| II | 31 (28.7%) | 7 (30.4%) | 24 (28.2%) | 1.000 c |
| III | 10 (9.3%) | 3 (13.0%) | 7 (8.2%) | 0.441 b |
| IV | 20 (18.5%) | 4 (17.4%) | 16 (18.8%) | 1.000 b |
| V | 19 (17.6%) | 6 (26.1%) | 13 (15.3%) | 0.231 b |
| IV–V | 39 (36.1%) | 10 (43.5%) | 29 (34.1%) | 0.559 c |
| Modified Fisher grade | ||||
| 1 | 14 (13.0%) | 0 (0.0%) | 14 (16.5%) | 0.038 b |
| 2 | 6 (5.6%) | 0 (0.0%) | 6 (7.1%) | 0.338 b |
| 3 | 60 (55.6%) | 16 (69.6%) | 44 (51.8%) | 0.198 c |
| 4 | 28 (25.9%) | 7 (30.4%) | 21 (24.7%) | 0.773 c |
| 3–4 | 88 (81.5%) | 23 (100%) | 65 (76.4%) | 0.006 b |
| Acute hydrocephalus | 41 (38.0%) | 6 (26.1%) | 35 (41.2%) | 0.280 c |
| Ruptured aneurysm location | ||||
| ICA | 43 (39.8%) | 8 (34.8%) | 35 (41.2%) | 0.752 c |
| MCA | 21 (19.4%) | 8 (34.8%) | 13 (15.3%) | 0.070 b |
| AcomA | 30 (27.8%) | 7 (30.4%) | 23 (27.1%) | 0.954 c |
| ACA | 8 (7.4%) | 0 (0.0%) | 8 (9.4%) | 0.198 b |
| VA-BA | 6 (5.6%) | 0 (0.0%) | 6 (7.1%) | 0.338 b |
| Variables | Total (n = 108) | DCIn (n = 23) | No DCIn (n = 85) | p Value |
|---|---|---|---|---|
| Treatment-related complication | 21 (19.4%) | 6 (26.1%) | 15 (17.6%) | 0.381 a |
| Cerebral infarction | 17 (15.7%) | 4 (17.4%) | 13 (15.3%) | 0.756 a |
| Cerebral contusion | 4 (3.7%) | 2 (8.7%) | 2 (2.4%) | 0.198 a |
| CSF drainage | 47 (43.5%) | 10 (43.5%) | 37 (43.5%) | 1.000 b |
| Ventricular drainage | 35 (32.4%) | 5 (21.7%) | 30 (35.3%) | 0.327 b |
| Cisternal drainage | 23 (21.3%) | 4 (17.4%) | 19 (22.4%) | 0.777 a |
| Spinal drainage | 10 (9.3%) | 4 (17.4%) | 6 (7.1%) | 0.215 a |
| Prophylaxis for CVS or DCI | ||||
| i.v. Fasudil hydrochloride | 107 (99.1%) | 23 (100%) | 84 (98.8%) | 1.000 a |
| Oral or enteral cilostazol | 87 (80.6%) | 17 (73.9%) | 70 (82.4%) | 0.381 a |
| Angiographic CVS | 40 (37.0%) | 16 (69.6%) | 24 (28.2%) | <0.001 b |
| DCI | 18 (16.7%) | 12 (52.2%) | 6 (7.1%) | <0.001 a |
| Endovascular CVS treatment | ||||
| i.a. Fasudil hydrochloride | 9 (8.3%) | 5 (21.7%) | 4 (4.7%) | 0.020 a |
| 3-month mRS | ||||
| 0 | 35 (32.4%) | 4 (17.4%) | 31 (36.5%) | 0.138 b |
| 1 | 16 (14.8%) | 1 (4.3%) | 15 (17.6%) | 0.184 a |
| 2 | 13 (12.0%) | 2 (8.7%) | 11 (12.9%) | 0.731 a |
| 3 | 15 (13.9%) | 5 (21.7%) | 10 (11.8%) | 0.305 a |
| 4 | 9 (8.3%) | 2 (8.7%) | 7 (8.2%) | 1.000 a |
| 5 | 15 (13.9%) | 5 (21.7%) | 10 (11.8%) | 0.305 a |
| 6 | 5 (4.6%) | 4 (17.4%) | 1 (1.2%) | 0.007 a |
| 0–2 | 64 (59.3%) | 7 (30.4%) | 57 (67.1%) | 0.003 b |
| Variables | aOR | 95% CI | p Value |
|---|---|---|---|
| DCI | 10.49 | 2.89–38.12 | <0.001 |
| Plasma OPN N-half ≥ 70.42 pmol/L at days 10–12 | 5.65 | 1.68–18.97 | 0.005 |
| Modified Fisher grades 3–4 | 0.998 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aoki, K.; Kawakita, F.; Hakozaki, K.; Kanamaru, H.; Asada, R.; Suzuki, H.; Group, p. Increased Plasma Levels of Thrombin-Cleaved Osteopontin in Patients with Delayed Cerebral Infarction After Aneurysmal Subarachnoid Hemorrhage. Int. J. Mol. Sci. 2025, 26, 2781. https://doi.org/10.3390/ijms26062781
Aoki K, Kawakita F, Hakozaki K, Kanamaru H, Asada R, Suzuki H, Group p. Increased Plasma Levels of Thrombin-Cleaved Osteopontin in Patients with Delayed Cerebral Infarction After Aneurysmal Subarachnoid Hemorrhage. International Journal of Molecular Sciences. 2025; 26(6):2781. https://doi.org/10.3390/ijms26062781
Chicago/Turabian StyleAoki, Kazuaki, Fumihiro Kawakita, Koichi Hakozaki, Hideki Kanamaru, Reona Asada, Hidenori Suzuki, and pSEED Group. 2025. "Increased Plasma Levels of Thrombin-Cleaved Osteopontin in Patients with Delayed Cerebral Infarction After Aneurysmal Subarachnoid Hemorrhage" International Journal of Molecular Sciences 26, no. 6: 2781. https://doi.org/10.3390/ijms26062781
APA StyleAoki, K., Kawakita, F., Hakozaki, K., Kanamaru, H., Asada, R., Suzuki, H., & Group, p. (2025). Increased Plasma Levels of Thrombin-Cleaved Osteopontin in Patients with Delayed Cerebral Infarction After Aneurysmal Subarachnoid Hemorrhage. International Journal of Molecular Sciences, 26(6), 2781. https://doi.org/10.3390/ijms26062781







