A Plant-Based Strategy for MASLD: Desmodium caudatum (Thunb.) DC. Extract Reduces Hepatic Lipid Accumulation and Improves Glycogen Storage In Vitro and In Vivo
Abstract
1. Introduction
2. Results
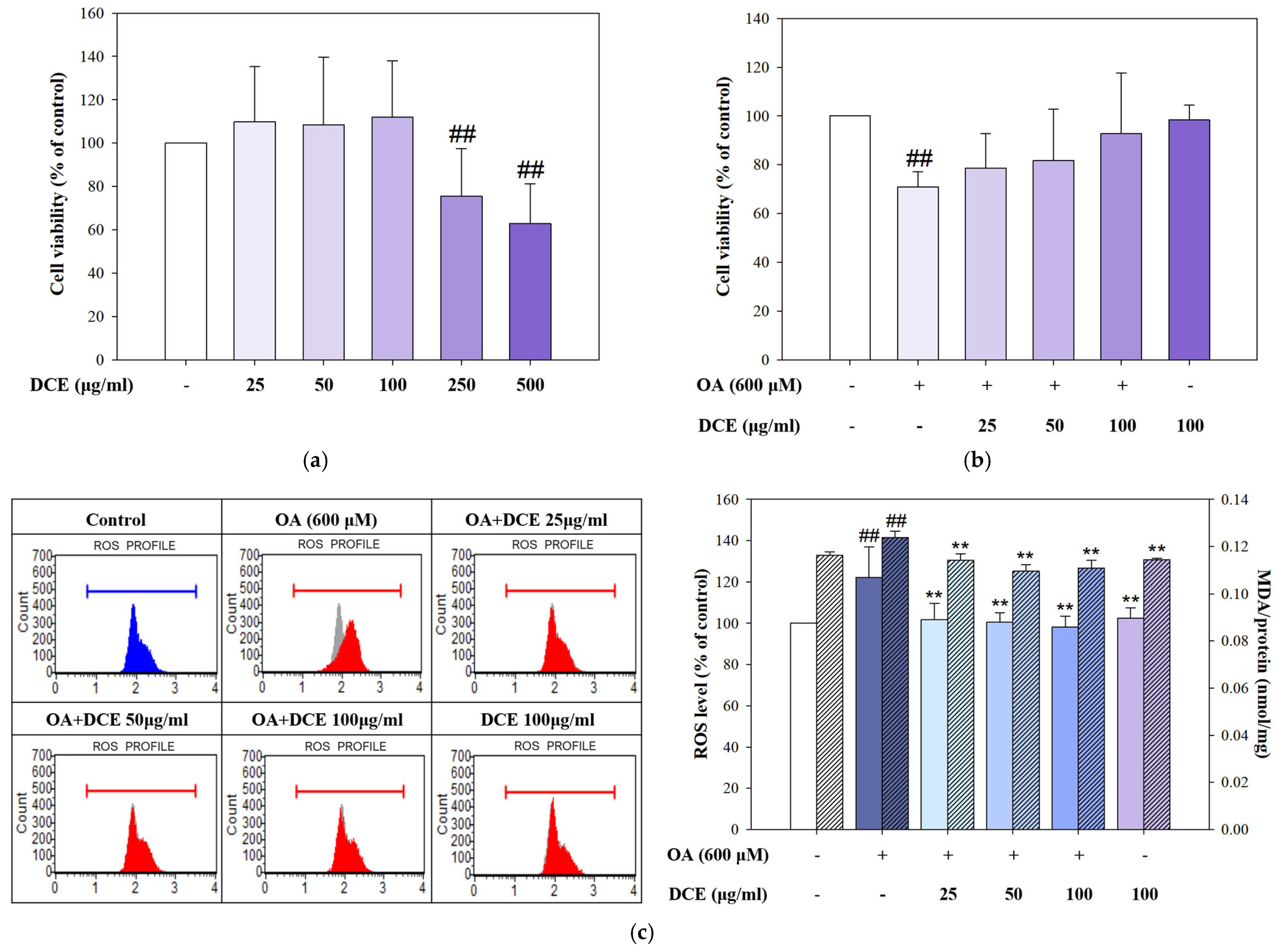
2.1. DCE Attenuates OA-Induced Cytotoxicity and Oxidative Stress in HepG2 Cells
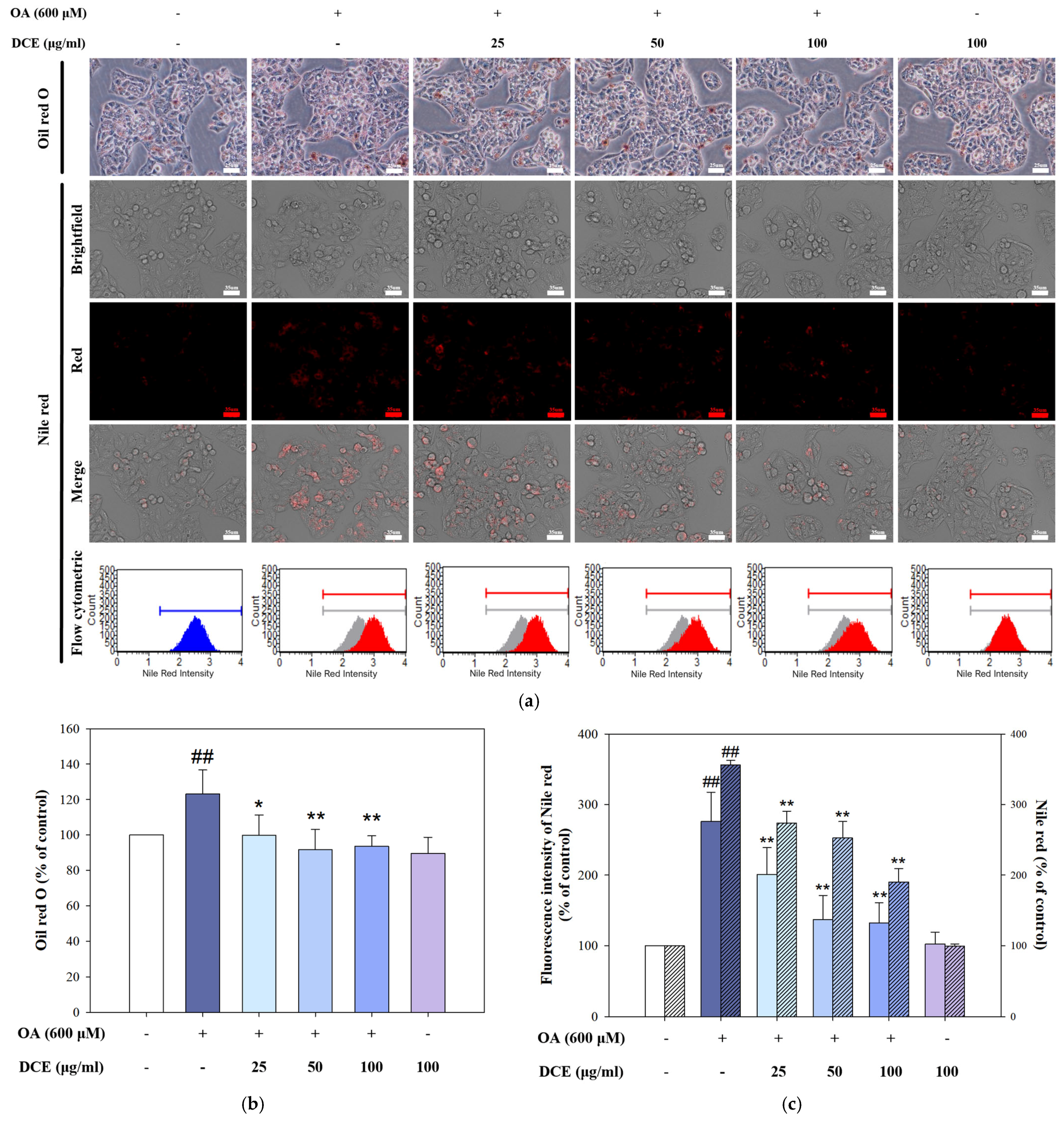
2.2. DCE Reduced OA-Induced Lipid Accumulation in HepG2 Cells
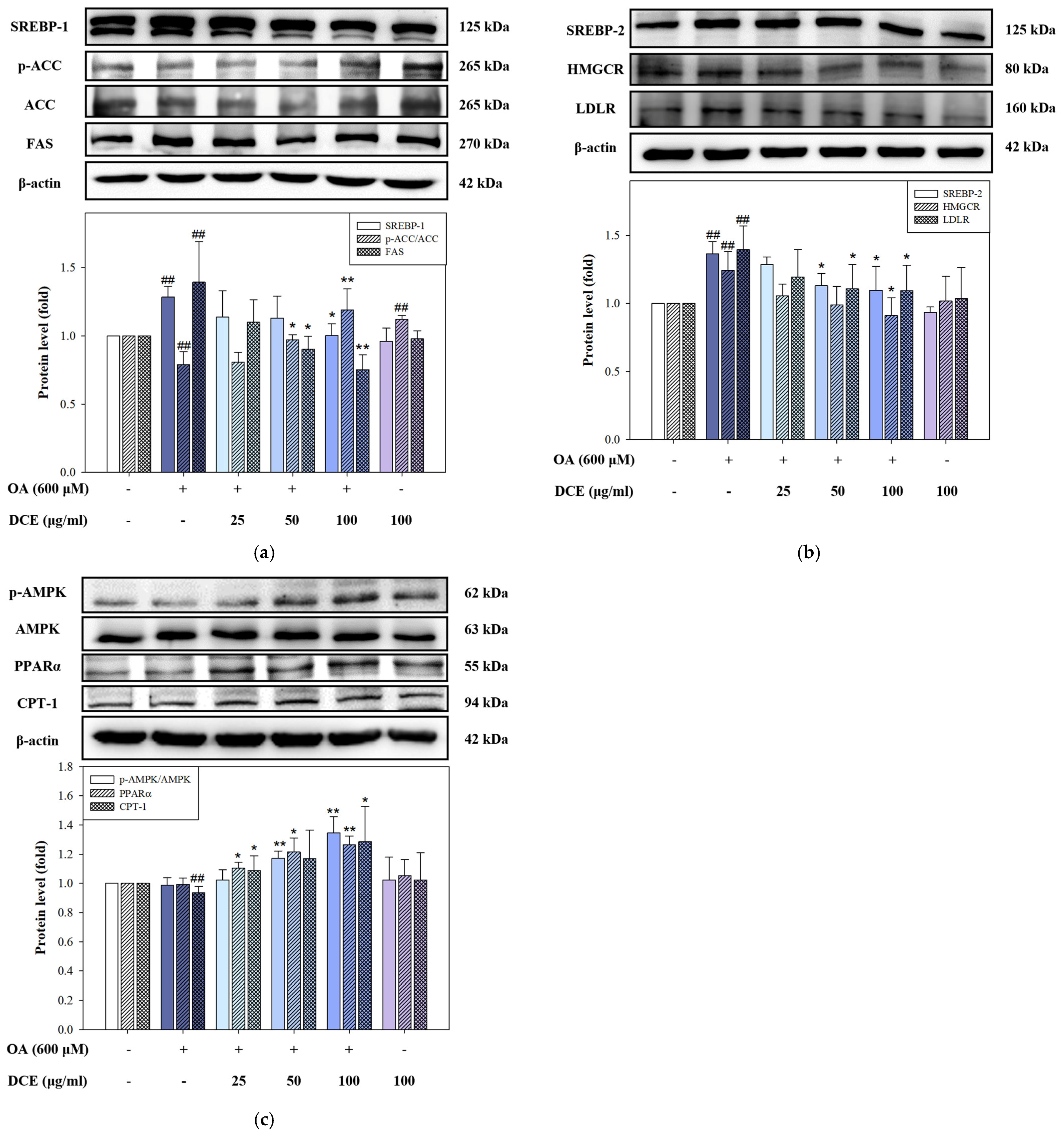
2.3. DCE Modulates Lipid Metabolism-Related Protein Expression in OA-Induced HepG2 Cells
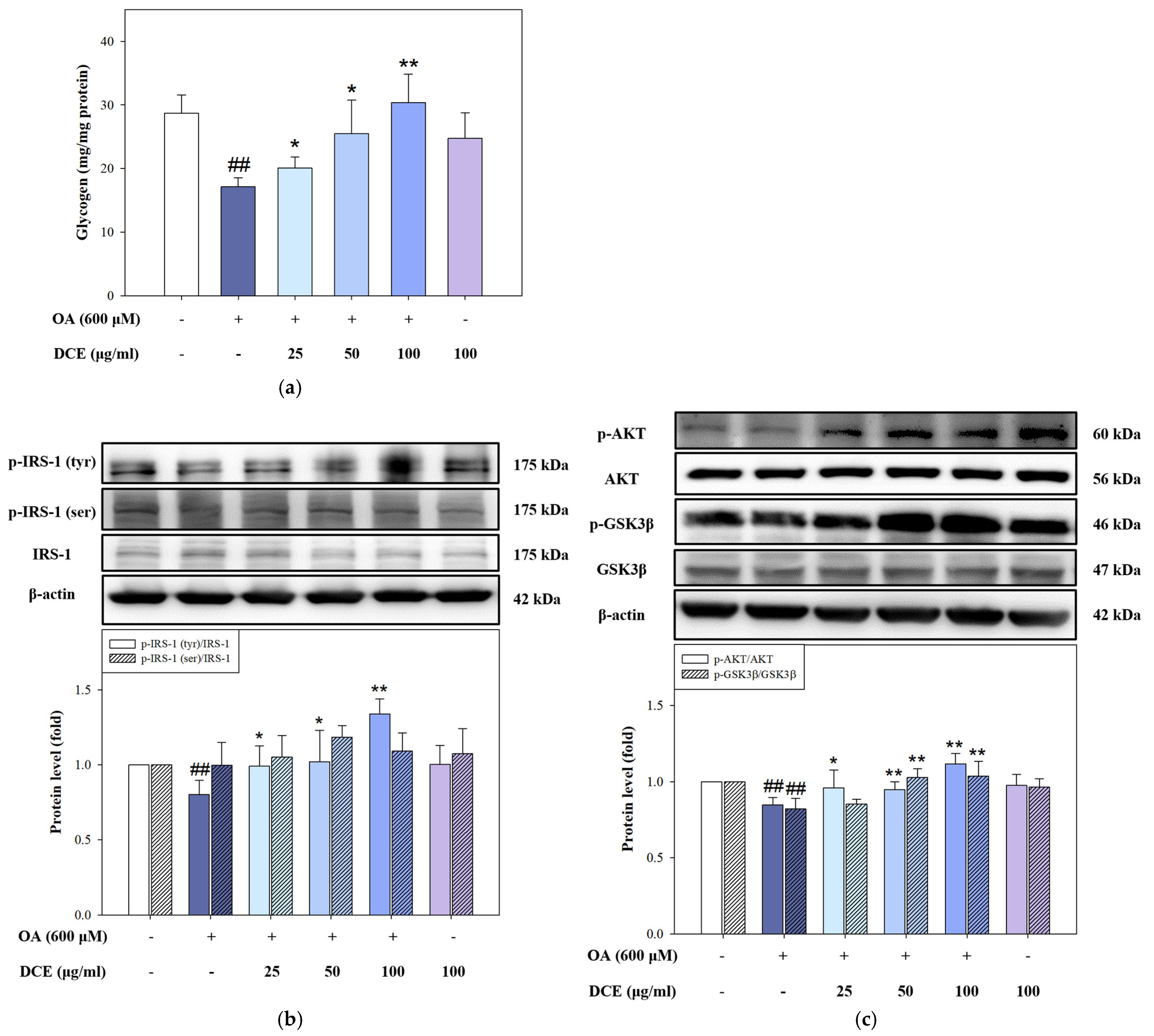
2.4. DCE Improves Glycogen Storage by Restoring Insulin Signaling in OA-Challenged HepG2 Cells
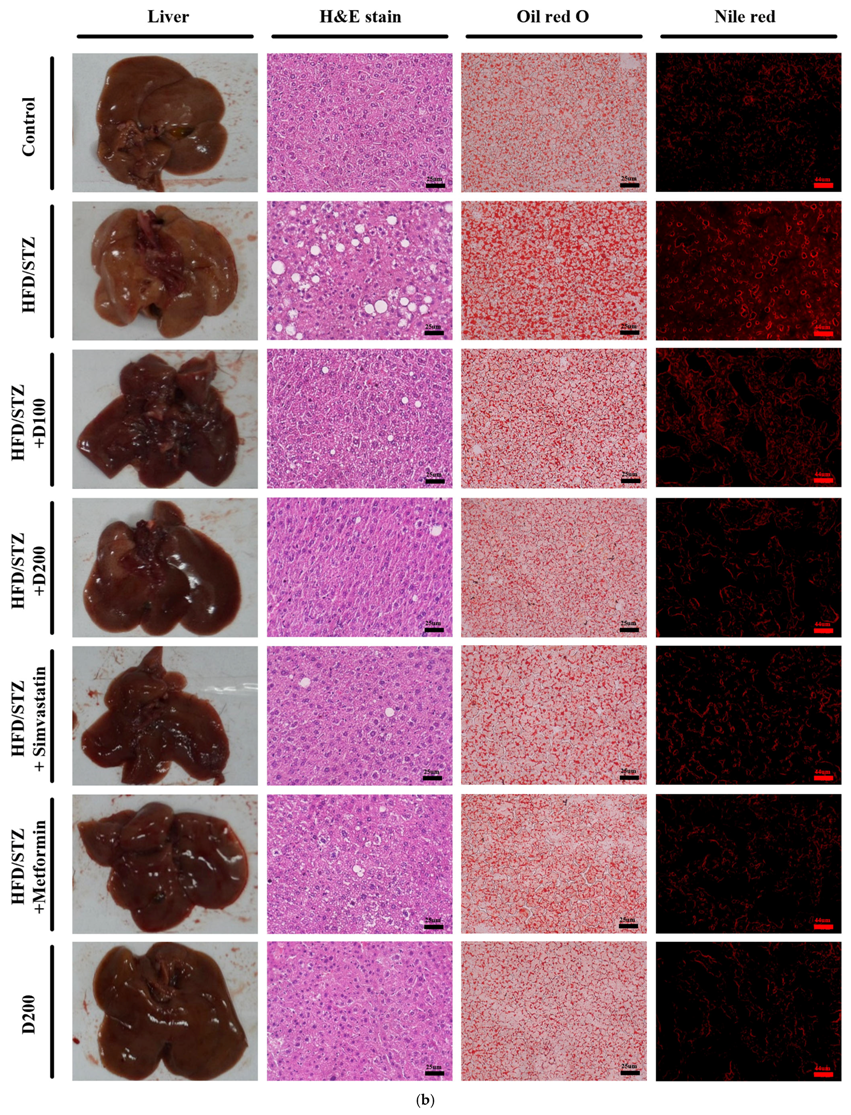
2.5. DCE Improves Hepatic Lipid Accumulation and Serum Biochemical Parameters in HFD/STZ-Induced MASLD Mice
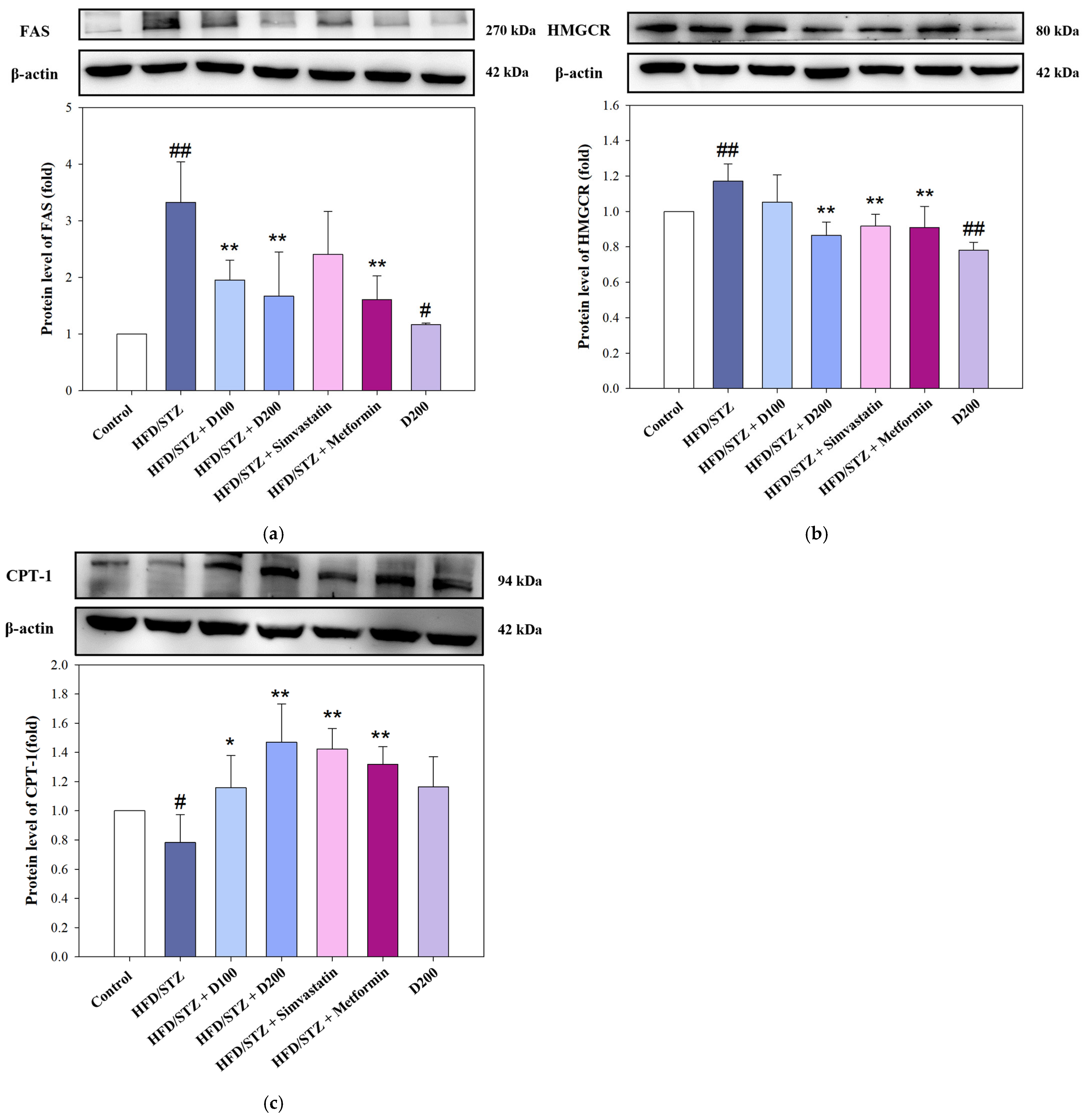
2.6. The Expression of DCE on Lipid Metabolic Regulators in HFD/STZ-Induced MASLD Mice
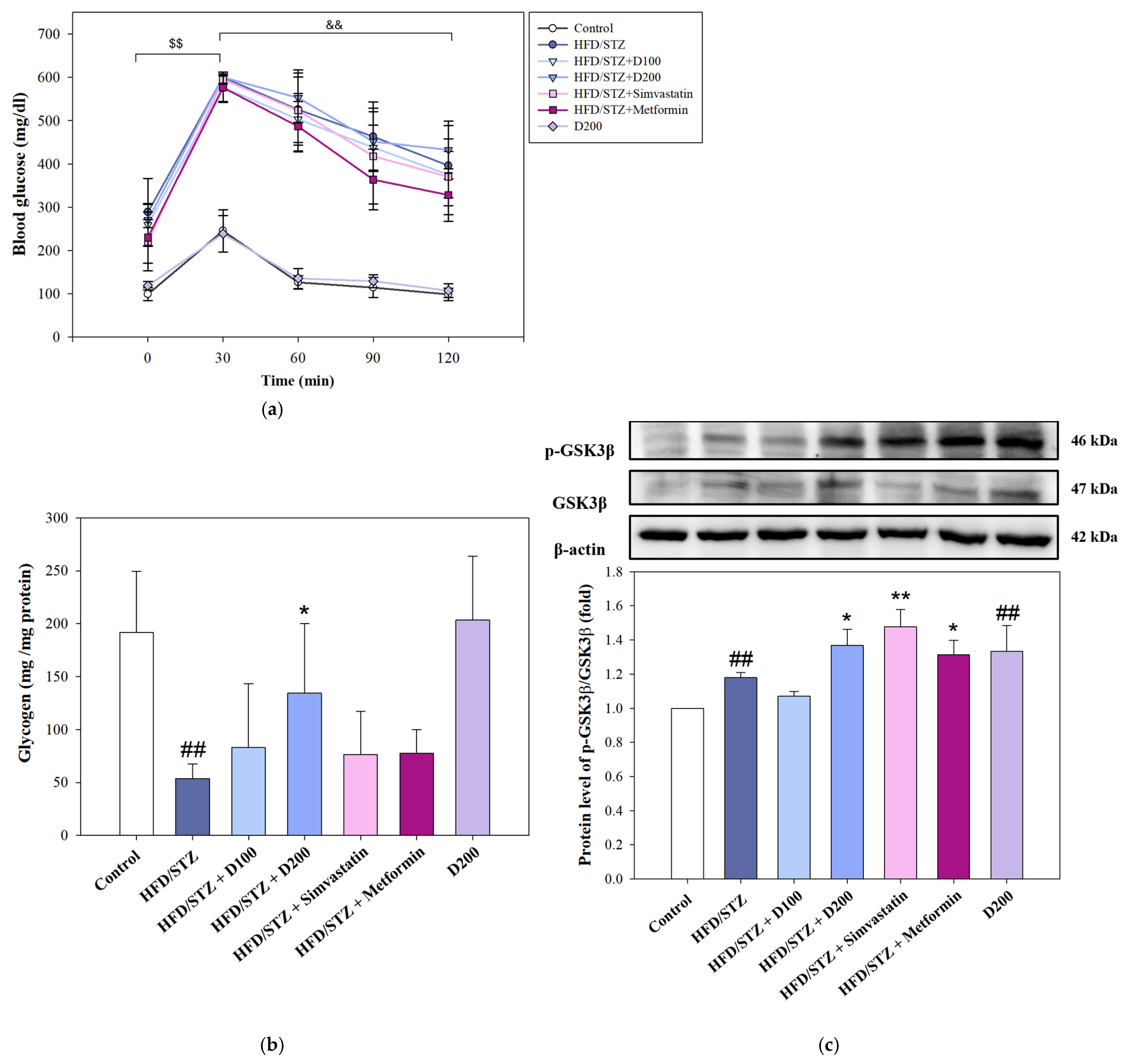
2.7. The Effect of DCE on Glucose Tolerance and Hepatic Glycogen Synthesis in HFD/STZ-Induced MASLD Mice
3. Discussion
4. Materials and Methods
4.1. Desmodium caudatum (Thunb.) DC. Extract (DCE)
4.2. Cell Line and Treatment
4.3. Cell Viability Assay
4.4. Assessment of Intracellular ROS
4.5. Thiobarbituric Acid Reactive Substances (TBARS) Assay
4.6. Oil Red O Staining
4.7. Nile Red Staining
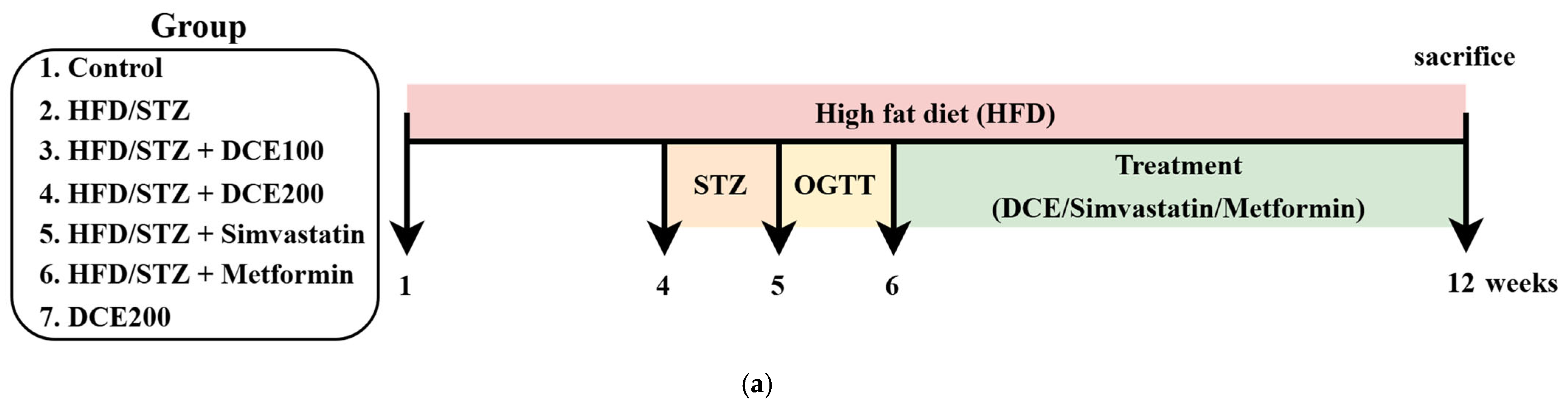
4.8. Animal and Experimental Design
4.9. Oral Glucose Tolerance Test (OGTT)
4.10. Histopathological Examination
4.11. Western Blotting
4.12. Glycogen Assay
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACC | Acetyl-CoA carboxylase |
| AKT | Serine/threonine kinase (protein kinase B) |
| ALD | Alcoholic liver disease |
| ALT | Alanine aminotransferase |
| AMPK | AMP-activated protein kinase |
| AST | Aspartate aminotransferase |
| CPT-1 | Carnitine palmitoyltransferase I |
| DCE | Desmodium caudatum (Thunb.) DC. extract |
| DCF-DA | 2’,7’-dichlorofluorescein diacetate |
| FAS | Fatty acid synthase |
| GLC | Fasting blood glucose |
| GS | Glycogen synthase |
| GSK3β | Glycogen synthase kinase 3 beta |
| HDL-C | High-density lipoprotein-cholesterol |
| HFD | High-fat diet |
| HMGCR | Hydroxymethylglutaryl-CoA reductase |
| IRS-1 | Insulin receptor substrate 1 |
| IRS-2 | Insulin receptor substrate 2 |
| LDH | Lactate dehydrogenase |
| LDL-C | Low-density lipoprotein-cholesterol |
| LDLR | Low-density lipoprotein receptor |
| MASH | Metabolic dysfunction-associated steatohepatitis |
| MASLD | Metabolic dysfunction-associated steatotic liver disease |
| MDA | Malondialdehyde |
| OA | Oleic acid |
| OGTT | Oral glucose tolerance test |
| PI3K | Phosphoinositide 3-kinase |
| PPARα | Peroxisome proliferator-activated receptor α |
| ROS | Reactive oxygen species |
| Ser | Serine |
| SREBP-2 | Sterol regulatory element-binding protein 2 |
| STZ | Streptozotocin |
| TBARS | Thiobarbituric acid reactive substances |
| TC | Total cholesterol |
| TCM | Traditional Chinese Medicine |
| TG | Triglyceride |
| Tyr | Tyrosine |
References
- Hsu, W.-F.; Sheen, L.-Y.; Lin, H.-J.; Chang, H.-H.; Thounaojam, M.C. A review of Western and traditional Chinese medical approaches to managing nonalcoholic fatty liver disease. Evid. Based Complement. Altern. Med. 2016, 2016, 6491420. [Google Scholar] [CrossRef]
- Shah, P.A.; Patil, R.; Harrison, S.A. NAFLD-related hepatocellular carcinoma: The growing challenge. Hepatology 2023, 77, 323–338. [Google Scholar] [CrossRef]
- Gadiparthi, C.; Spatz, M.; Greenberg, S.; Iqbal, U.; Kanna, S.; Satapathy, S.K.; Broder, A.; Ahmed, A. NAFLD epidemiology, emerging pharmacotherapy, liver transplantation implications and the trends in the United States. J. Clin. Transl. Hepatol. 2020, 8, 215–221. [Google Scholar] [CrossRef]
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. Burden of liver diseases in the world. J. Hepatol. 2019, 70, 151–171. [Google Scholar] [CrossRef]
- Kamata, S.; Honda, A.; Ishii, I. Current clinical trial status and future prospects of PPAR-targeted drugs for treating nonalcoholic fatty liver disease. Biomolecules 2023, 13, 1264. [Google Scholar] [CrossRef] [PubMed]
- Alves-Bezerra, M.; Cohen, D.E. Triglyceride metabolism in the liver. Compr. Physiol. 2017, 8, 1–8. [Google Scholar] [CrossRef]
- Goedeke, L.; Fernández-Hernando, C. Regulation of cholesterol homeostasis. Cell. Mol. Life Sci. 2012, 69, 915–930. [Google Scholar] [CrossRef]
- Espenshade, P.J. SREBPs: Sterol-regulated transcription factors. J. Cell Sci. 2006, 119, 973–976. [Google Scholar] [CrossRef] [PubMed]
- Softic, S.; Cohen, D.E.; Kahn, C.R. Role of dietary fructose and hepatic de novo lipogenesis in fatty liver disease. Dig. Dis. Sci. 2016, 61, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Grabacka, M.; Reiss, K.; Panigrahy, D. Anticancer Properties of PPARα-Effects on Cellular Metabolism and Inflammation. PPAR Res. 2008, 2008, 930705. [Google Scholar] [CrossRef]
- Qu, Q.; Zeng, F.; Liu, X.; Wang, Q.J.; Deng, F. Fatty acid oxidation and carnitine palmitoyltransferase I: Emerging therapeutic targets in cancer. Cell Death Dis. 2016, 7, e2226. [Google Scholar] [CrossRef] [PubMed]
- Schreurs, M.; Kuipers, F.; Van Der Leij, F.R. Regulatory enzymes of mitochondrial β-oxidation as targets for treatment of the metabolic syndrome. Obes. Rev. 2010, 11, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Krishan, S.; Richardson, D.R.; Sahni, S. Amp kinase (prkaa1). J. Clin. Pathol. 2014, 67, 758–763. [Google Scholar] [CrossRef]
- Mardilovich, K.; Pankratz, S.L.; Shaw, L.M. Expression and function of the insulin receptor substrate proteins in cancer. Cell Commun. Signal. 2009, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Boura-Halfon, S.; Zick, Y. Phosphorylation of IRS proteins, insulin action, and insulin resistance. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E581–E591. [Google Scholar] [CrossRef]
- Atkins, R.J.; Dimou, J.; Paradiso, L.; Morokoff, A.P.; Kaye, A.H.; Drummond, K.J.; Hovens, C.M. Regulation of glycogen synthase kinase-3 beta (GSK-3β) by the Akt pathway in gliomas. J. Clin. Neurosci. 2012, 19, 1558–1563. [Google Scholar] [CrossRef]
- Flory, J.; Lipska, K. Metformin in 2019. JAMA 2019, 321, 1926–1927. [Google Scholar] [CrossRef]
- Ma, X.; Zheng, C.; Hu, C.; Rahman, K.; Qin, L. The genus Desmodium (Fabaceae)-traditional uses in Chinese medicine, phytochemistry and pharmacology. J. Ethnopharmacol. 2011, 138, 314–332. [Google Scholar] [CrossRef]
- Ma, K.-J.; Zhu, Z.-Z.; Yu, C.-H.; Zhang, H.; Liu, J.; Qin, L.-P. Analgesic, anti-inflammatory, and antipyretic activities of the ethanol extract from Desmodium caudatum. Pharm. Biol. 2011, 49, 403–407. [Google Scholar] [CrossRef]
- Cao, M.; Miao, J.; Wang, L.; Liu, H.; Cui, H.; Bian, Y. The advances of traditional Chinese medicine in the treatment of liver diseases in 2019. Tradit. Med. Res. 2020, 5, 261–271. [Google Scholar] [CrossRef]
- Irimia, J.M.; Meyer, C.M.; Segvich, D.M.; Surendran, S.; DePaoli-Roach, A.A.; Morral, N.; Roach, P.J. Lack of liver glycogen causes mice hepatic insulin resistance and steatosis. J. Biol. Chem. 2017, 292, 10455–10464. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Amin, M.J.; Ahmed, M.U.; Taj, A.; Aasim, M.; Tabrez, E. Frequency of non-alcoholic fatty liver disease (NAFLD) and its associated risk factors among Type-2 diabetics. Pak. J. Med. Sci. 2022, 38, 28. [Google Scholar] [CrossRef]
- Zelber-Sagi, S.; Ratziu, V.; Oren, R. Nutrition and physical activity in NAFLD: An overview of the epidemiological evidence. World J. Gastroenterol. WJG 2011, 17, 3377–3389. [Google Scholar] [CrossRef]
- Nascimbeni, F.; Pellegrini, E.; Lugari, S.; Mondelli, A.; Bursi, S.; Onfiani, G.; Carubbi, F.; Lonardo, A. Statins and nonalcoholic fatty liver disease in the era of precision medicine: More friends than foes. Atherosclerosis 2019, 284, 66–74. [Google Scholar] [CrossRef]
- Rouabhia, S.; Milic, N.; Abenavoli, L. Abenavoli, Metformin in the treatment of non-alcoholic fatty liver disease: Safety, efficacy and mechanism. Expert Rev. Gastroenterol. Hepatol. 2014, 8, 343–349. [Google Scholar] [CrossRef]
- Li, W.; Sun, Y.N.; Yan, X.T.; Yang, S.Y.; Kim, S.; Chae, D.; Hyun, J.W.; Kang, H.K.; Koh, Y.-S.; Kim, Y.H. Anti-inflammatory and antioxidant activities of phenolic compounds from Desmodium caudatum leaves and stems. Arch. Pharmacal Res. 2014, 37, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Kashiwada, Y.; Shibata, H.; Takaishi, Y. Prenylated flavonoids from Desmodium caudatum and evaluation of their anti-MRSA activity. Phytochemistry 2012, 82, 136–142. [Google Scholar] [CrossRef]
- Shi, L.-j.; Shi, L.; Song, G.-Y.; Zhang, H.-F.; Hu, Z.-J.; Wang, C.; Zhang, D.-H. Oxymatrine attenuates hepatic steatosis in non-alcoholic fatty liver disease rats fed with high fructose diet through inhibition of sterol regulatory element binding transcription factor 1 (Srebf1) and activation of peroxisome proliferator activated receptor alpha (Pparα). Eur. J. Pharmacol. 2013, 714, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-H.; Liang, Y.-H.; Chyau, C.-C.; Tseng, C.-Y.; Zhang, J.-Q.; Chen, J.-H. Desmodium caudatum (Thunb.) DC. extract attenuates hyperuricemia-induced renal fibrosis via modulating TGF-β1 pathway and uric acid transporters: Evidence from in vitro and in vivo studies. J. Ethnopharmacol. 2025, 345, 119609. [Google Scholar] [CrossRef] [PubMed]
- Pavasutti, V.; Sinthuvanich, C.; Tayana, N.; Kongkiatpaiboon, S.; Sae-Tan, S. Mung bean seed coat water extract restores insulin sensitivity via upregulation of antioxidant defense system and downregulation of inflammation in insulin-resistant HepG2 cells. NFS J. 2023, 32, 100145. [Google Scholar] [CrossRef]
- Mokashi, P.; Khanna, A.; Pandita, N. Flavonoids from Enicostema littorale blume enhances glucose uptake of cells in insulin resistant human liver cancer (HepG2) cell line via IRS-1/PI3K/Akt pathway. Biomed. Pharmacother. 2017, 90, 268–277. [Google Scholar] [CrossRef]
- Rastogi, S.; Pandey, M.M.; Rawat, A.K.S. An ethnomedicinal, phytochemical and pharmacological profile of Desmodium gangeticum (L.) DC. and Desmodium adscendens (Sw.) DC. J. Ethnopharmacol. 2011, 136, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Comi, L.; Giglione, C.; Klinaku, F.T.; Da Dalt, L.; Ullah, H.; Daglia, M.; Magni, P. Evaluation of metabolic dysfunction-associated fatty liver disease-related pathogenic mechanisms in human steatotic liver cell-based model: Beneficial effects of Prunus domestica L. subsp. syriaca extract. Nutrients 2025, 17, 1249. [Google Scholar] [CrossRef]
- Gu, M.J.; Ahn, Y.; Lee, Y.R.; Yoo, G.; Kim, Y.; Choi, I.; Ha, S.K.; Kim, D. Coriandrum sativum L. Leaf Extract Ameliorates Metabolic Dysfunction-Associated Steatotic Liver Disease by Modulating the AMPK Pathway in High Fat-Fed C57BL/6 Mice. Nutrients 2024, 16, 4165. [Google Scholar] [CrossRef]
- Abdurehman, D.; Guoruoluo, Y.; Lu, X.; Li, J.; Abudulla, R.; Liu, G.; Xin, X.; Aisa, H.A. Optimization of preparation method of hepatoprotective active components from Coreopsis tinctoria Nutt. and its action mechanism in vivo. Biomed. Pharmacother. 2023, 167, 115590. [Google Scholar] [CrossRef]
- Kastelein, J.J.; Akdim, F.; Stroes, E.S.; Zwinderman, A.H.; Bots, M.L.; Stalenhoef, A.F.; Visseren, F.L.; Sijbrands, E.J.; Trip, M.D.; Stein, E.A.; et al. Simvastatin with or without ezetimibe in familial hypercholesterolemia. N. Engl. J. Med. 2008, 358, 1431–1443, Erratum in N. Engl. J. Med. 2008, 358, 1977. [Google Scholar] [CrossRef] [PubMed]
- Rena, G.; Hardie, D.G.; Pearson, E.R. The mechanisms of action of metformin. Diabetologia 2017, 60, 1577–1585. [Google Scholar] [CrossRef] [PubMed]

| Control | HFD/STZ | HFD/STZ +D100 | HFD/STZ +D200 | HFD/STZ +Simvastatin | HFD/STZ +Metformin | D200 | |
|---|---|---|---|---|---|---|---|
| GLC (mg/dL) | 104.29 ± 10.23 | 291.86 ± 38.51 ## | 164.83 ± 31.74 ** | 269.33 ± 66.40 | 303.50 ± 54.47 | 181.20 ± 51.63 ** | 86.20 ± 28.60 |
| TC (mg/dL) | 81.00 ± 17.51 | 178.00 ± 7.94 ## | 144.50 ± 20.11 * | 122.40 ± 27.53 * | 161.33 ± 4.73 * | 124.60 ± 30.86 * | 82.33 ± 4.04 |
| TG (mg/dL) | 65.00 ± 8.75 | 140.00 ± 8.54 ## | 117.00 ± 11.53 | 102.33 ± 6.11 ** | 154.00 ± 21.21 | 112.75 ± 22.20 * | 59.00 ± 13.23 ## |
| LDL-C (mg/dL) | 5.80 ± 0.75 | 7.87 ± 1.08 # | 4.75 ± 1.32 * | 3.68 ± 1.44 ** | 6.85 ± 2.67 | 4.72 ± 1.80 | 2.33 ± 0.21 |
| HDL-C (mg/dL) | 59.60 ± 2.78 | 40.90 ± 0.71 ## | 76.95 ± 5.93 ** | 65.03 ± 5.87 ** | 73.76 ± 5.24 ** | 68.30 ± 2.60 ** | 53.83 ± 4.63 |
| AST (U/L) | 176.33 ± 7.09 | 246.33 ± 29.48 # | 171.00 ± 20.54 * | 138.50 ± 30.95 * | 166.67 ± 9.24 * | 148.00 ± 31.18 * | 148.00 ± 11.22 # |
| ALT (U/L) | 29.80 ± 2.95 | 45.67 ± 7.41 ## | 31.67 ± 6.34 * | 33.00 ± 4.97 * | 28.00 ± 8.12 * | 40.67 ± 8.39 | 30.67 ± 3.79 |
| LDH (U/L) | 882.33 ± 156.05 | 1830.67 ± 579.71 # | 745.00 ± 65.65 ** | 697.50 ± 144.34 ** | 687.00 ± 38.11 ** | 676.29 ± 106.39 ** | 685.33 ± 158.45 # |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-C.; Liang, Y.-H.; Wong, Y.; Tseng, C.-Y.; Chang, C.-W.; Lin, H.-H.; Chen, J.-H. A Plant-Based Strategy for MASLD: Desmodium caudatum (Thunb.) DC. Extract Reduces Hepatic Lipid Accumulation and Improves Glycogen Storage In Vitro and In Vivo. Int. J. Mol. Sci. 2025, 26, 8442. https://doi.org/10.3390/ijms26178442
Chen Y-C, Liang Y-H, Wong Y, Tseng C-Y, Chang C-W, Lin H-H, Chen J-H. A Plant-Based Strategy for MASLD: Desmodium caudatum (Thunb.) DC. Extract Reduces Hepatic Lipid Accumulation and Improves Glycogen Storage In Vitro and In Vivo. International Journal of Molecular Sciences. 2025; 26(17):8442. https://doi.org/10.3390/ijms26178442
Chicago/Turabian StyleChen, Yu-Ching, Yu-Hsuan Liang, Yueching Wong, Chiao-Yun Tseng, Chi-Wen Chang, Hui-Hsuan Lin, and Jing-Hsien Chen. 2025. "A Plant-Based Strategy for MASLD: Desmodium caudatum (Thunb.) DC. Extract Reduces Hepatic Lipid Accumulation and Improves Glycogen Storage In Vitro and In Vivo" International Journal of Molecular Sciences 26, no. 17: 8442. https://doi.org/10.3390/ijms26178442
APA StyleChen, Y.-C., Liang, Y.-H., Wong, Y., Tseng, C.-Y., Chang, C.-W., Lin, H.-H., & Chen, J.-H. (2025). A Plant-Based Strategy for MASLD: Desmodium caudatum (Thunb.) DC. Extract Reduces Hepatic Lipid Accumulation and Improves Glycogen Storage In Vitro and In Vivo. International Journal of Molecular Sciences, 26(17), 8442. https://doi.org/10.3390/ijms26178442








