Anti-Herpes Simplex Virus Type 1 Activity of Rosa damascena Mill Essential Oil and Floral Water in Retinal Infection In Vitro and In Silico
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Host Cell Lines
4.2. Viruses
4.3. Reference Compound
4.4. Candidates for Therapeutic Substances for Complementary Therapy of Retinal Infection with HSV-1–Rosa damascena Mill Essential Oil and Rose Water
4.5. Gas Chromatographic (GC) Analysis
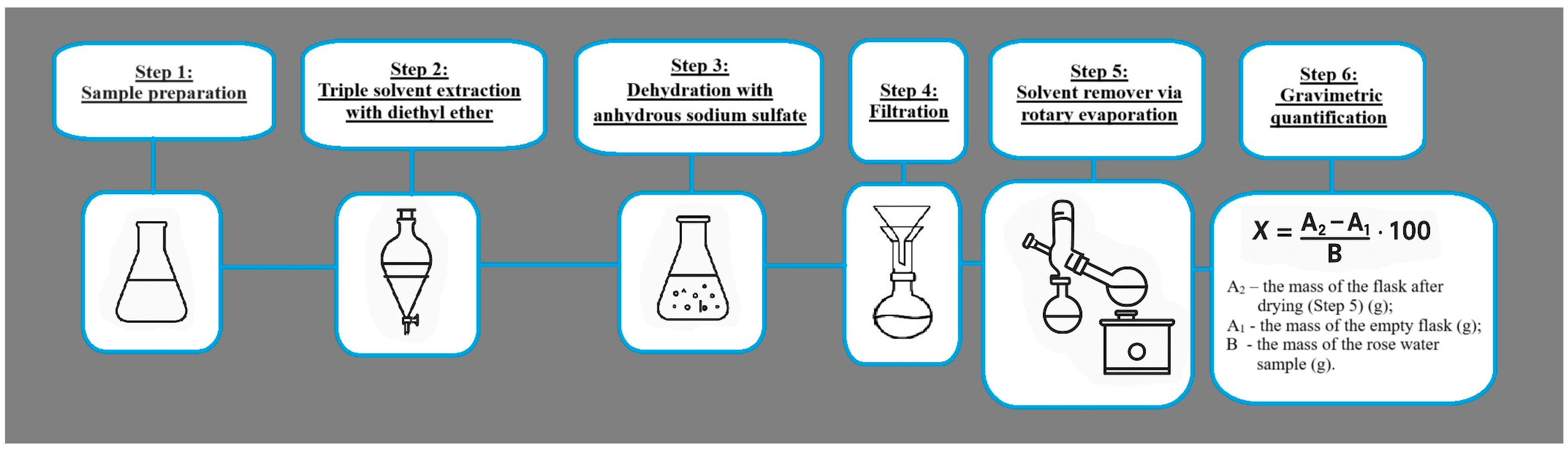
4.6. Determining the Essential Oil Content in Rose Water
4.7. Determining the Ethyl Alcohol Content in Rose Water
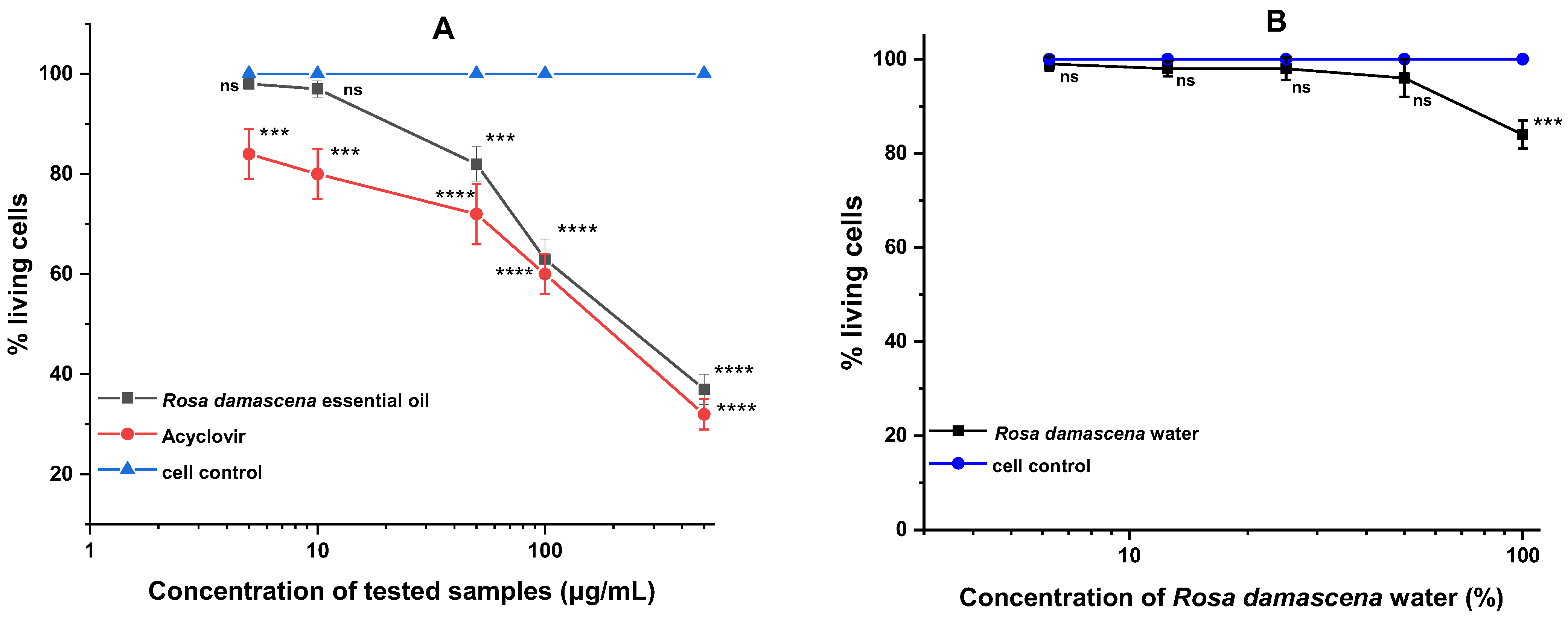
4.8. Cytotoxicity Assay
4.9. Determination of Infectious Viral Titers
4.10. Antiviral Activity Assay
4.11. Virucidal Assay
4.12. Effect on Viral Adsorption
4.13. Pre-Treatment of Healthy Cells
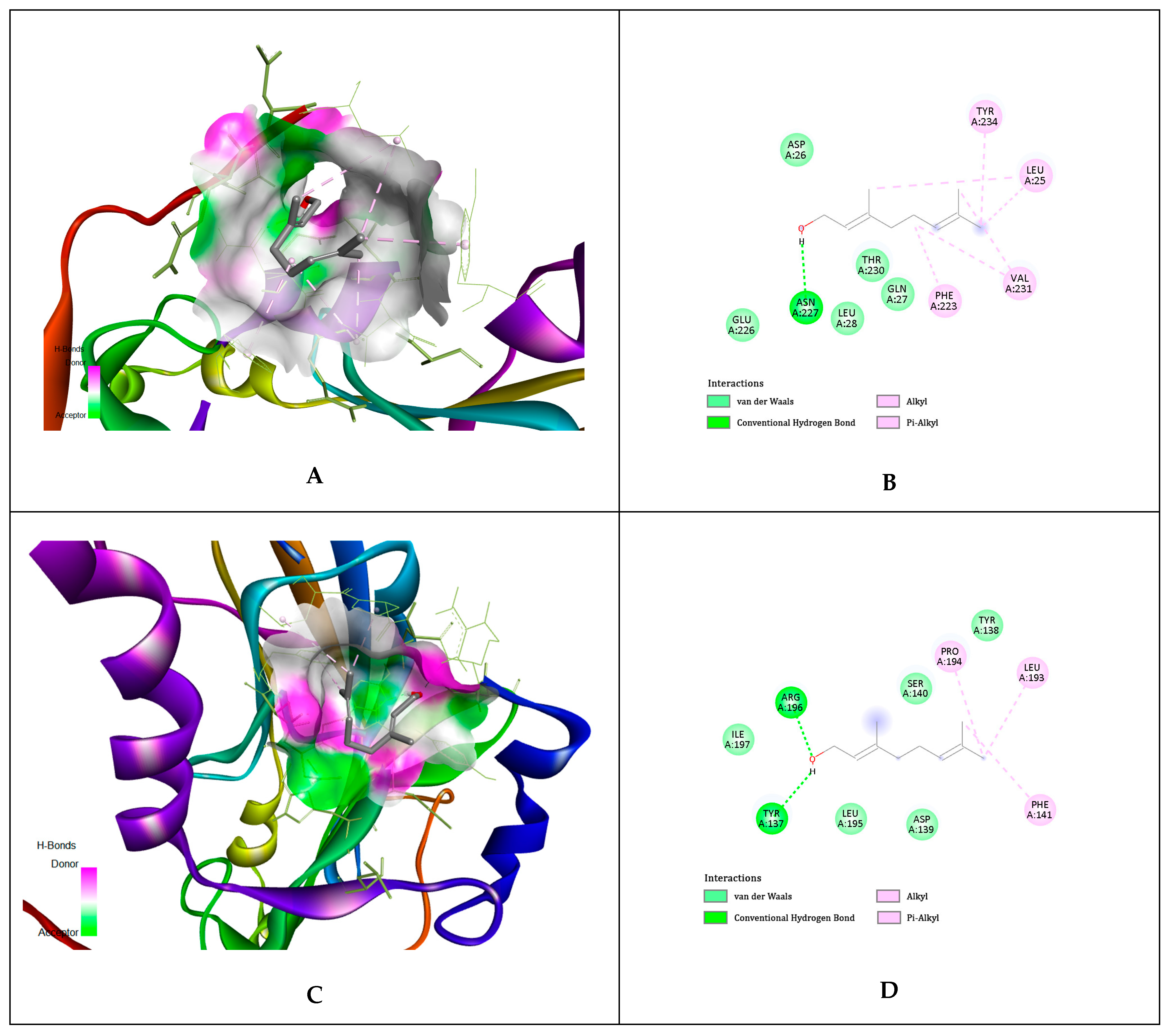
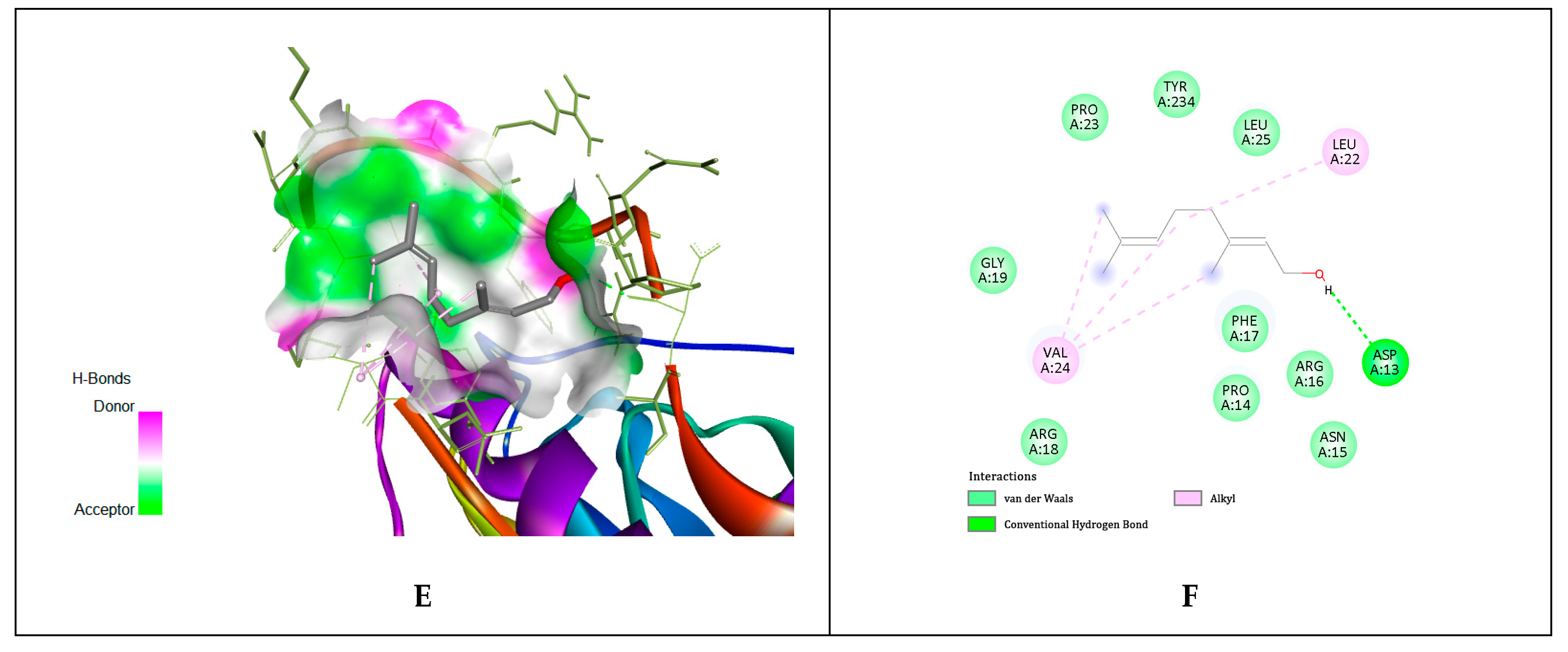
4.14. In Silico Molecular Docking Simulations
4.15. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patil, S.; Beck, P.; Nelson, T.; Bran, A.; Roland, W. Herpes Simplex Virus-2 Meningoencephalitis With Abducens Nerve Palsy With Literature Review. Cureus 2021, 13, e15523. [Google Scholar] [CrossRef]
- Arshad, S.; Petsoglou, C.; Lee, T.; Al-Tamimi, A.; Carnt, N. 20 years since the Herpetic Eye Disease Study: Lessons, developments and applications to clinical practice. Clin. Exp. Optom. 2021, 104, 396–405. [Google Scholar] [CrossRef]
- Shahnazaryan, D.; Khalil, R.; Wynne, C.; Jefferies, C.; Gabhann-Dromgoole, J.; Murphy, C. Herpes simplex virus 1 targets IRF7 via ICP0 to limit type I IFN induction. Sci. Rep. 2020, 10, 22216. [Google Scholar] [CrossRef]
- Arduino, P.G.; Porter, S.R. Herpes Simplex Virus Type 1 infection: Overview on relevant clinico-pathological features. J. Oral Pathol. Med. 2008, 37, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Hilterbrand, A.; Daly, R.; Heldweina, E. Contributions of the Four Essential Entry Glycoproteins to HSV-1 Tropism and the Selection of Entry Routes. mBio 2021, 12, e00143-21. [Google Scholar] [CrossRef]
- Madavaraju, K.; Koganti, R.; Volety, I.; Yadavalli, T.; Shukla, D. Herpes Simplex Virus Cell Entry Mechanisms: An Update. Front. Cell Infect. Microbiol. 2021, 10, 617578. [Google Scholar] [CrossRef]
- Roizman, B.; Knipe, D.M. Herpes Simplex Viruses and Their Replication. In Fields Virology, 3rd ed.; Lippincott-Williams & Wilkins: Philadelphia, PA, USA, 2001; Volume 2, pp. 2399–2459. [Google Scholar]
- Jambunathan, N.; Clark, C.M.; Musarrat, F.; Chouljenko, V.N.; Rudd, J.; Kousoulas, K.G. Two Sides to Every Story: Herpes Simplex Type-1 Viral Glycoproteins gB, gD, gH/gL, gK, and Cellular Receptors Function as Key Players in Membrane Fusion. Viruses 2021, 13, 1849. [Google Scholar] [CrossRef] [PubMed]
- Agelidis, A.; Shukla, D. Cell entry mechanisms of HSV: What we have learned in recent years. Future Virol. 2015, 10, 1145–1154. [Google Scholar] [CrossRef]
- Koujah, L.; Suryawanshi, R.; Shukla, D. Pathological processes activated by herpes simplex virus-1 (HSV-1) infection in the cornea. Cell. Mol. Life Sci. 2019, 76, 405–419. [Google Scholar] [CrossRef]
- Farooq, A.; Shukla, D. Corneal latency and transmission of herpes simplex virus-1. Future Virol. 2011, 6, 101–108. [Google Scholar] [CrossRef]
- Grinde, B. Herpesviruses: Latency and reactivation—Viral strategies and host response. J. Oral Microbiol. 2013, 5, 22766. [Google Scholar] [CrossRef]
- Musa, M.; Enaholo, E.; Aluyi-Osa, G.; Atuanya, G.; Spadea, L.; Salati, C.; Zeppieri, M. Herpes simplex keratitis: A brief clinical overview. World J. Virol. 2024, 13, 89934. [Google Scholar] [CrossRef]
- Lobo, A.; Agelidis, A.; Shukla, D. Pathogenesis of herpes simplex keratitis: The host cell response and ocular sur-face sequelae to infection and inflammation. Ocul. Surf. 2019, 17, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Liesegang, T. Herpes simplex virus epidemiology and ocular importance. Cornea 2001, 20, 1–13. [Google Scholar] [CrossRef]
- Kaye, S.; Choudhary, A. Herpes simplex keratitis. Prog. Retin. Eye Res. 2006, 25, 355–380. [Google Scholar] [CrossRef]
- Souza, P.; Holland, E.; Huang, A. Bilateral herpetic keratoconjunctivitis. Ophthalmology 2003, 110, 493–496. [Google Scholar] [CrossRef]
- Remeijer, L.; Osterhaus, A.; Verjans, G. Human herpes simplex virus keratitis: The pathogenesis revisited. Ocul. Immunol. Inflamm. 2004, 12, 255–285. [Google Scholar] [CrossRef]
- Farooq, A.; Shukla, D. Herpes Simplex Epithelial and Stromal Keratitis: An Epidemiologic Update. Surv. Ophthalmol. 2012, 57, 448–462. [Google Scholar] [CrossRef]
- Greenan, E.; Gallagher, S.; Khalil, R.; Murphy, C.; Gabhann-Dromgoole, J. Advancing Our Understanding of Corneal Herpes Simplex Virus-1 Immune Evasion Mechanisms and Future Therapeutics. Viruses 2021, 13, 1856. [Google Scholar] [CrossRef]
- Buzzetti, M.; Silva, V.; Dendi, A.; Vidal, G.; Sobrero, H. Neonatal ophthalmia caused by Herpes Simplex Virus type I. Andes Pediatr. 2022, 93, 749–754. [Google Scholar] [CrossRef]
- Harbiyeli, I.; Oruz, O.; Erdem, E.; Cam, B.; Demirkazik, M.; Acikalin, A.; Kibar, F.; Ilkit, M.; Yarkin, F.; Yagmur, M. Clinical aspects and prognosis of polymicrobial keratitis caused by different microbial combinations: A retrospective comparative case study. Int. Ophthalmol. 2021, 41, 3849–3860. [Google Scholar] [CrossRef] [PubMed]
- Roels, D.; Coorevits, L.; Lagrou, K. Tintelnotia destructans as an emerging opportunistic pathogen: First case of T. destructans superinfection in herpetic keratitis. Am. J. Ophthalmol. Case Rep. 2020, 19, 100791. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, W.; Wang, M.; Xing, Q.; Sun, X. Clinical diagnosis and treatment of rare painless keratitis caused by three pathogens: Clinical practice and experiential discussion. J. Int. Med. Res. 2020, 48, 300060519895671. [Google Scholar] [CrossRef] [PubMed]
- Elion, G. Mechanism of action and selectivity of acyclovir. Am. J. Med. 1982, 73, 7–13. [Google Scholar] [CrossRef]
- Beck, R.; Asbell, P.; Cohen, E.; Dawson, C.; Hyndiuk, R.; Jones, D.; Kaufman, H.; Kip, K.; Kurinij, N.; Moke, P.; et al. Oral acyclovir for herpes simplex virus eye disease: Effect on prevention of epithelial keratitis and stromal keratitis. Arch. Ophthalmol. 2000, 118, 1030–1036. [Google Scholar] [CrossRef]
- Kaufman, H. Treatment of viral diseases of the cornea and external eye. Prog. Retin. Eye Res. 2000, 19, 69–85. [Google Scholar] [CrossRef]
- Chou, T.; Hong, B. Ganciclovir ophthalmic gel 0.15% for the treatment of acute herpetic keratitis: Background, effectiveness, tolerability, safety, and future applications. Ther. Clin. Risk Manag. 2014, 10, 665–681. [Google Scholar] [CrossRef]
- Tyring, S.; Engst, R.; Corriveau, C.; Robillard, N.; Trottier, S.; Van Slycken, S.; Crann, A.; Locke, L.; Saltzman, R.; Palestine, A. Famciclovir for ophthalmic zoster: A randomised aciclovir controlled study. Br. J. Ophthalmol. 2001, 85, 576–581. [Google Scholar] [CrossRef]
- Boskabady, M.H.; Shafei, M.N.; Saberi, Z.; Amini, S. Pharmacological effects of rosa damascena. Iran. J. Basic Med. Sci. 2011, 14, 295–307. [Google Scholar]
- Mahboubi, M. Rosa damascena as holy ancient herb with novel applications. J. Tradit. Complement. Med. 2015, 6, 10–16. [Google Scholar] [CrossRef]
- Lee, Y.; Park, E.; Jang, B.; Hwang, J.; Lee, J.; Oh, E.S. Antifungal Activity of Bulgarian Rose Damascena Oil against Vaginitis-Causing Opportunistic Fungi. Evid. Based Complement. Altern. Med. 2023, 1, 5054865. [Google Scholar] [CrossRef]
- Chishti, M.A.; Akram, M.; Laila, U.; Zainab, R.; Ifthikar, M.; Ahmet, F.; Ozdemir, I.A.K. Rosa Damescana: A review of its conventional uses Phytochemistry and Pharmacology. Glob. Acad. J. Agric. Biosci. 2023, 5, 61–67. [Google Scholar] [CrossRef]
- Verešová, A.; Vukic, M.D.; Vukovic, N.L.; Terentjeva, M.; Ban, Z.; Li, L.; Bianchi, A.; Kollár, J.; Ben Saad, R.; Ben Hsouna, A.; et al. Chemical Composition, Biological Activity, and Application of Rosa damascena Essential Oil as an Antimicrobial Agent in Minimally Processed Eggplant Inoculated with Salmonella enterica. Foods 2024, 13, 3579. [Google Scholar] [CrossRef] [PubMed]
- Akram, M.; Riaz, M.; Munir, N.; Akhter, N.; Zafar, S.; Jabeen, F.; Shariati, M.A.; Akhtar, N.; Riaz, Z.; Altaf, S.H.; et al. Chemical constituents, experimental and clinical pharmacology of Rosa damascena: A literature review. J. Pharm. Pharmacol. 2020, 72, 161–174. [Google Scholar] [CrossRef]
- Karadağ, A.E.; Biltekin, S.N.; Demirci, B.; Demirci, F.; Ghani, U. Comparative In Vitro and In Silico Enzyme Inhibitory Screening of Rosa x damascena and Pelargonium graveolens Essential Oils and Geraniol. Plants 2023, 12, 3296. [Google Scholar] [CrossRef]
- Hongratanaworakit, T. Relaxing effect of rose oil on humans. Nat. Prod. Commun. 2009, 4, 1934578X0900400226. [Google Scholar] [CrossRef]
- Kwon, E.K.; Lee, D.Y.; Lee, H.; Kim, D.O.; Baek, N.I.; Kim, Y.E.; Kim, H.Y. Flavonoids from the buds of Rosa damascena inhibit the activity of 3-hydroxy-3-methylglutarylcoenzyme a reductase and angiotensin Iconverting enzyme. J. Agric. Food Chem. 2010, 58, 882–886. [Google Scholar] [CrossRef]
- Rakhshandah, H.; Boskabadi, M.H.; Mousavi, Z.; Gholami, M.; Saberi, Z. The differences in the relaxant effects of different fractions of Rosa damascena on guinea pig tracheal smooth muscle. Iran. J. Basic Med. Sci. 2010, 13, 126–132. [Google Scholar]
- Krajian, H.; Odeh, A. Polycyclic aromatic hydrocarbons in medicinal plants from Syria. Toxic. Environ. Chem. 2013, 95, 942–953. [Google Scholar] [CrossRef]
- Baser, K.H.C.; Altintas, A.; Kurkcuoglu, M. Turkish Rose: A review of the history, ethnobotany and modern uses of rose petals, rose oil, rose water and other rose products. HerbalGram 2012, 96, 40–53. [Google Scholar]
- Bhattacharya, S.; Roy, B. Preliminary investigation on antipyretic activity of Cuscuta Reflexa in rats. J. Adv. Pharm. Technol. Res. 2010, 1, 83. [Google Scholar] [CrossRef]
- Preedy, V.R. Essential Oils in Food Preservation, Flavor, and Safety; Academic Press: London, UK, 2015. [Google Scholar]
- Vilhelmova-Ilieva, N.; Dobreva, A.; Doynovska, R.; Krastev, D.; Mileva, M. Antiviral Activity of Rosa damascena Mill. and Rosa alba L. Essential Oils against the Multiplication of Herpes Simplex Virus Type 1 Strains Sensitive and Resistant to Acyclovir. Biology 2021, 10, 746. [Google Scholar] [CrossRef]
- Gochev, V.; Jirovetz, L.; Wlcek, K.; Buchbauer, G.; Schmidt, E.; Stoyanova, A.; Dobreva, A. Chemical Composition and Antimicrobial Activity of Historical Rose Oil from Bulgaria. J. Essent. Oil Bear. Plants 2009, 12, 1–6. [Google Scholar] [CrossRef]
- Ulusoy, S.; Boşgelmez-Tinaz, G.; Seçilmiş-Canbay, H. Tocopherol, carotene, phenolic contents and antibacterial properties of rose essential oil, hydrosol and absolute. Curr. Microbiol. 2009, 59, 554–558. [Google Scholar] [CrossRef]
- Basim, E.; Basim, H. Antibacterial activity of Rosa damascena essential oil. Fitoterapia 2003, 74, 394–396. [Google Scholar] [CrossRef] [PubMed]
- Mahboubi, M.; Kazempour, N.; Khamechian, T.; Fallah, M.H.; Kermani, M.M. Chemical composition and antimicrobial activity of Rosa damascena Mill essential oil. J. Biol. Act. Prod. Nat. 2011, 1, 19–26. [Google Scholar] [CrossRef]
- Biswas, N.R.; Gupta, S.K.; Das, G.K.; Kumar, N.; Mongre, P.K.; Haldar, D.; Beri, S. Evaluation of Ophthacare eye drops--a herbal formulation in the management of various ophthalmic disorders. Phytother. Res. 2001, 15, 618–620. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.K.; Sundaram, R.; Venkataranganna, M.V.; Gopumadhavan, S.; Prakash, N.S.; Jayaram, H.D.; Sarma, D.N. Anti-inflammatory, antioxidant and antimicrobial activity of Ophthacare brand, an herbal eye drops. Phytomedicine 2000, 7, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Nedeltcheva-Antonova, D.; Stoicheva, P.; Antonov, L. Chemical profiling of Bulgarian rose absolute (Rosa damascena Mill.) using gas chromatography–mass spectrometry and trimethylsilyl derivatives. Ind. Crops Prod. 2017, 108, 36–43. [Google Scholar] [CrossRef]
- Guenter, E.; Vol, V. Individual essential oils of the plant families. In The Essential Oils; Publishing House D. Van Nostrand Company: New York, NY, USA, 1952; Volume 5, pp. 3–47. [Google Scholar]
- Dobreva, A.; Velcheva, A.; Bardarov, B.; Bardarov, K. Chemical composition of different genotypes oil-bearing roses. Bulg. J. Agric. Sci. 2013, 19, 1213–1218. [Google Scholar]
- Raeber, J.; Favrod, S.; Steuer, C. Determination of Major, Minor and Chiral Components as Quality and Authenticity Markers of Rosa damascena Oil by GC-FID. Plants 2023, 12, 506. [Google Scholar] [CrossRef]
- Blanco, P.L.; Marshall, J.C.A.; Antecka, E.; Callejo, S.; Souza Filho, J.P.; Saraiva, V.; Burnier, M.N. Characterization of ocular and metastatic uveal melanoma in an animal model. Investig. Ophthalmol. Vis. Sci. 2005, 46, 4376–4382. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Grossniklaus, H. Rabbit Model of Retinoblastoma. J. Biomed. Biotechnol. 2011, 2011, 394730. [Google Scholar] [CrossRef] [PubMed]
- Gochev, V.; Wlcek, K.; Buchbauer, G.; Stoyanova, A.; Dobreva, A.; Schmidt, E.; Jirovetz, L. Comparative Evaluation of Antimicrobial Activity and Composition of Rose Oils From Various Geographic Origins, in Particular Bulgarian Rose Oil. Nat. Prod. Commun. 2008, 3, 1063–1068. [Google Scholar] [CrossRef]
- Rusanov, K.; Kovacheva, N.; Rusanova, M.; Atanassov, I. Low variability of flower volatiles of Rosa damascena Mill. plants from rose plantations along the Rose Valley, Bulgaria. Industr. Crops. Prod. 2012, 37, 6–10. [Google Scholar] [CrossRef]
- Mirzaei, M.; Ahmadi, N.; Sefidkon, F.; Shojaeiyan, A.; Mazaheri, A. Evaluation of some postharvest storage approaches on essential oil characteristics of fresh organic damask rose (Rosa damascena Mill.) flowers. Horticulturae 2017, 3, 16. [Google Scholar] [CrossRef]
- Thakur, M.; Sharma, S.; Sharma, U.; Kumar, R. Study on effect of pruning time on growth, yield and quality of scented rose (Rosa damascena Mill) varieties under acidic conditions of western Himalayas. J. Appl. Res. Med. Arom. Plants 2019, 13, 100202. [Google Scholar] [CrossRef]
- Yassa, N.; Masoomi, F.; Rankouhi, S.; Hadjiakhoondi, A. Chemical composition and antioxidant activity of the extract and essential oil of Rosa damascena from Iran, population of Guilan. DARU J. Pharm. Sci. 2009, 17, 175–180. [Google Scholar]
- Katsukawa, M.; Nakata, R.; Koeji, S.; Hori, K.; Takahashi, S.; Inoue, H. Citronellol and geraniol, components of rose oil, activate peroxisome proliferator-activated receptor α and γ and suppress cyclooxygenase-2 expression. Biosci. Biotechnol. Biochem. 2011, 75, 1010–1012. [Google Scholar] [CrossRef]
- Jelyani, A.Z.; Khademipour, N.; Aghajani, A. Identification of volatile alcoholic compound in rosewater by GC-MS analysis: A method to differentiate original and artificial samples. J. Food Bioprocess Eng. 2021, 4, 99–104. [Google Scholar] [CrossRef]
- Verma, S.; Padalia, C.; Chauhan, A. Chemical investigation of the volatile components of shade-dried petals of damask rose (Rosa damascena Mill.). Arch. Biol. Sci. 2011, 63, 1111–1115. [Google Scholar] [CrossRef]
- Nenova, R.; Nedeva, D.; Dobreva, A.; Vilhelmova-Ilieva, N.; Popova, R.; Ilieva, I.; Iliev, I. Evaluation of the safety and antiproliferative activity of Bulgarian rose oil in an in vitro model of colorectal adenocarcinoma. In Proceedings of the 2nd International Applied Scientific Conference in Medicinal, Aromatic and Edible Plants (MAEPs) and Their by-Products: Utilization and Valorization for a Sustainable and Circular Economy (MAEP & WASTE 2025), Plovdiv, Bulgaria, 16–17 May 2025. [Google Scholar]
- Heydarirad, G.; Keyhanmehr, A.; Mofid, B.; Nikfarjad, H.; Mosavat, S. Efficacy of aromatherapy with Rosa damascena in the improvement of sleep quality of cancer patients: A randomized controlled clinical trial. Complem. Therap. Clin. Pract. 2019, 35, 57–61. [Google Scholar] [CrossRef]
- Mahmood, N.; Piacente, S.; Pizza, C.; Burke, A.; Khan, A.; Hay, A. The anti- HIV activity and mechanisms of action of pure compounds isolated from Rosa damascena. Biochem. Biophys. Res. Commun. 1996, 229, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.; Nagarajan, S.; Ramesh, V.; Palaniyandi, V.; Selvam, S.; Madhavan, T. Computational evaluation of major components from plant essential oils as potent inhibitors of SARS-CoV-2 spike protein. J. Mol. Struct. 2020, 1221, 128823. [Google Scholar] [CrossRef]
- Quy, P.; My, T.; Bui, T.; Loan, H.; Van Anh, T.; Triet, N.; Quang, D.; Nhung, N. Molecular docking prediction of carvone and trans-geraniol inhibitability towards SARS-CoV-2. Viet. J. Chem. 2021, 59, 457–466. [Google Scholar] [CrossRef]
- Kumar, K.; Vani, M.; Wang, C.; Chen, C.; Chen, Y.; Lu, L.; Huang, C.; Lai, C.; Wang, S. Geranium and lemon essential oils and their active compounds downregulate angiotensin-converting enzyme 2 (ACE2), a SARS-CoV-2 spike receptor-binding domain, in epithelial cells. Plants 2020, 9, 770. [Google Scholar] [CrossRef]
- Orhan, I.; Ozcelik, B.; Kartal, M.; Kan, Y. Antimicrobial, and antiviral effects of essential oils from selected Umbelliferae and Labiatae plants and individual essential oil components. Turk. J. Biol. 2012, 36, 239–246. [Google Scholar] [CrossRef]
- Flechas, M.; Ocazionez, R.; Stashenko, E. Evaluation of in vitro antiviral activity of essential oil compounds against dengue virus. Pharmacogn. J. 2018, 10, 55–59. [Google Scholar] [CrossRef]
- Gómez, L.; Stashenko, E.; Ocazionez, R. Comparative study on in vitro activities of citral, limonene and essential oils from Lippia citriodora and L. alba on yellow fever virus. Nat. Prod. Commun. 2013, 8, 249–252. [Google Scholar] [CrossRef]
- Gilling, D.; Kitajima, M.; Torrey, J.; Bright, K. Mechanisms of antiviral action of plant antimicrobials against murine norovirus. Appl. Environ. Microbiol. 2014, 80, 4898–4910. [Google Scholar] [CrossRef]
- Benencia, F.; Courreges, M. In vitro and in vivo Activity of Eugenol on Human Herpesvirus. Phytother. Res. 2000, 14, 495–500. [Google Scholar] [CrossRef]
- Tragoolpua, Y.; Jatisatienr, A. Anti-Herpes Simplex Virus Activities of Eugenia caryophyllus (Spreng.) Bullock & SG Harrison and Essential Oil, Eugenol. Phytother. Res. 2007, 21, 1153–1158. [Google Scholar]
- Ilieva, Y.; Dimitrova, L.; Georgieva, A.; Vilhelmova-Ilieva, N.; Zaharieva, M.; Kokanova-Nedialkova, Z.; Dobreva, A.; Paraskev, N.; Kussovski, V.; Najdenski, H.; et al. In Vitro Study of the Biological Potential of Wastewater Obtained after the Distillation of Four Bulgarian Oil-Bearing Roses. Plants 2022, 11, 1073. [Google Scholar] [CrossRef]
- Krummenacher, C.; Supekar, V.M.; Whitbeck, J.C.; Lazear, E.; Connolly, S.A.; Eisenberg, R.J.; Cohen, G.H.; Wiley, D.C.; Carf, A. Structure of unliganded HSV gD reveals a mechanism for receptor-mediated activation of virus entry. EMBO J. 2005, 24, 4144–4153. [Google Scholar] [CrossRef] [PubMed]
- Yue, D.; Chen, Z.; Yang, F.; Ye, F.; Lin, S.; He, B.; Cheng, Y.; Wang, J.; Chen, Z.; Lin, X.; et al. Crystal structure of bovine herpesvirus 1 glycoprotein D bound to nectin-1 reveals the basis for its low-affinity binding to the receptor. Sci. Adv. 2020, 6, eaba5147. [Google Scholar] [CrossRef]
- Staykov, V.; Decheva, R.; Balinova-Tsvetkova, A. Studies on the composition of rose oil obtained from the flowers in different stages of their development. Riv. Ital. 1975, 57, 192–195. [Google Scholar]
- Clevenger, J.F. Apparatus for the determination of volatile oil. J. Am. Pharm. Assoc. 1928, 17, 345–349. [Google Scholar] [CrossRef]
- Balinova-Cvetkova, A.; Dyakov, D. Improved apparatus for microdistillation of oil-bearing rose. Plant Sci. 1974, 11, 79–86. (In Bulgarian) [Google Scholar]
- ISO 9842:2024; Essential Oil of Rose (Rosa x damascena Miller). International Organization for Standardization: Geneva, Switzerland, 2024. Available online: https://www.iso.org/standard/86897.html (accessed on 4 October 2024).
- Borenfreund, E.; Puerner, J. Toxicity determination in vitro by morphological alterations and neutral red absorption. Toxicol. Lett. 1985, 24, 119–124. [Google Scholar] [CrossRef]
- Creative Diagnostics. Cytopathic Effect Inhibition Assay. 2025. Available online: https://antiviral.creative-diagnostics.com/cytopathic-effect-inhibition-assay.html (accessed on 15 January 2025).
- Reed, L.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Eberhardt, J.; Santos-Martins, D.; Tillack, A.F.; Forli, S. AutoDock Vina 1.2.0: New Docking Methods, Expanded Force Field, and Python Bindings. J. Chem. Inf. Model. 2021, 61, 3891–3898. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
- Agu, P.C.; Afiukwa, C.A.; Orji, O.U.; Ezeh, E.M.; Ofoke, I.H.; Ogbu, C.O.; Ugwuja, E.I.; Aja, P.M. Molecular docking as a tool for the discovery of molecular targets of nutraceuticals in diseases management. Sci. Rep. 2023, 13, 13398. [Google Scholar] [CrossRef]
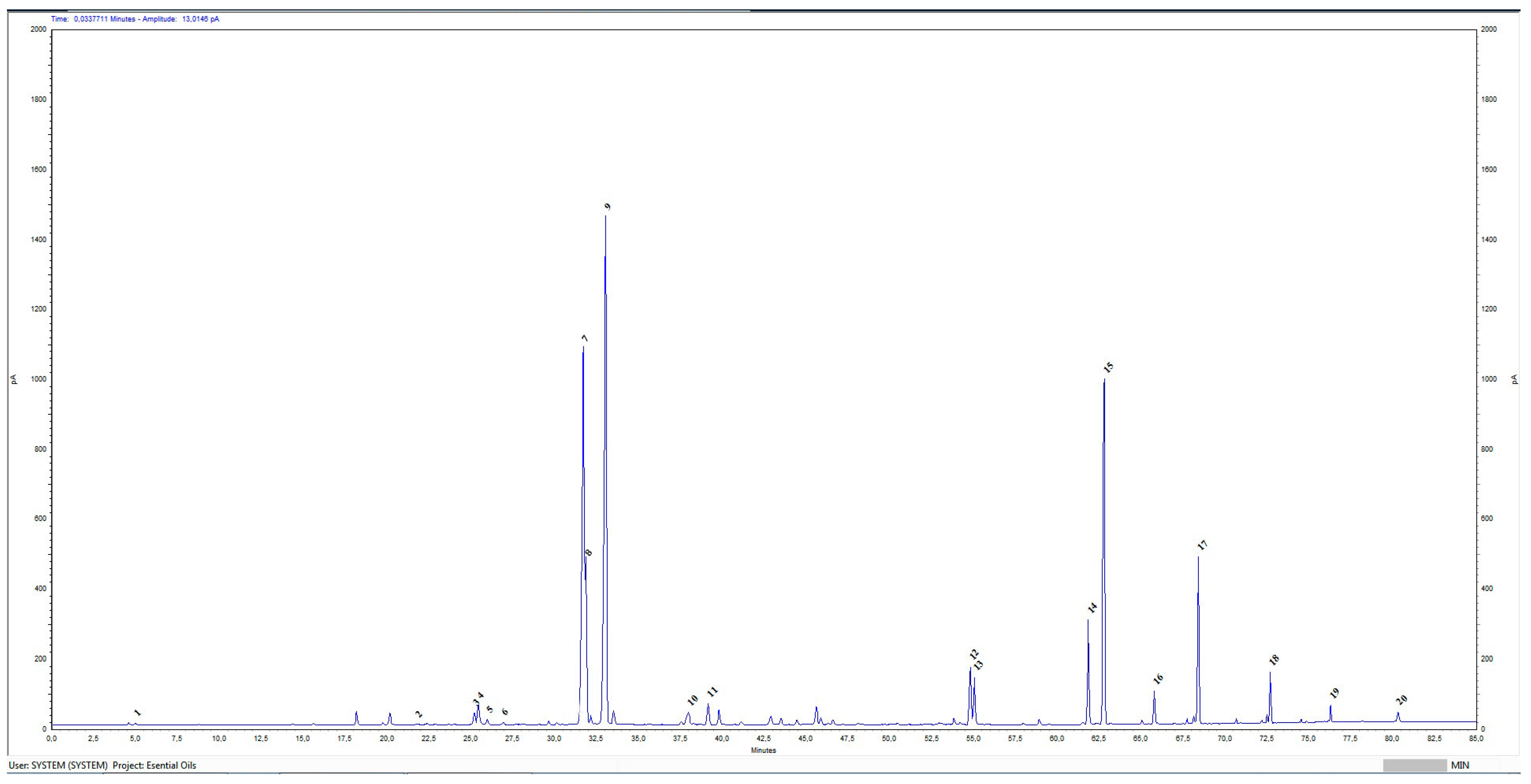



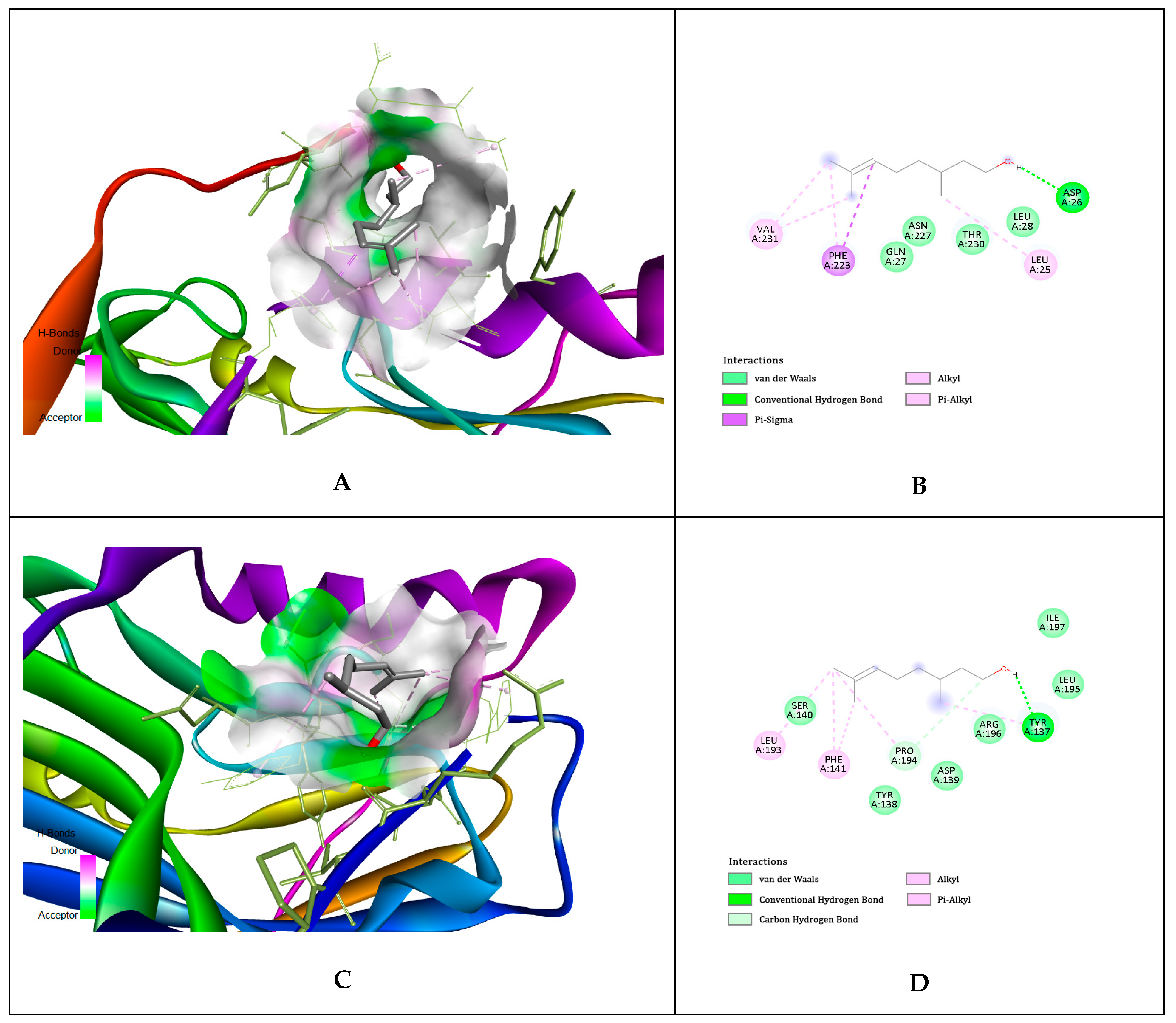
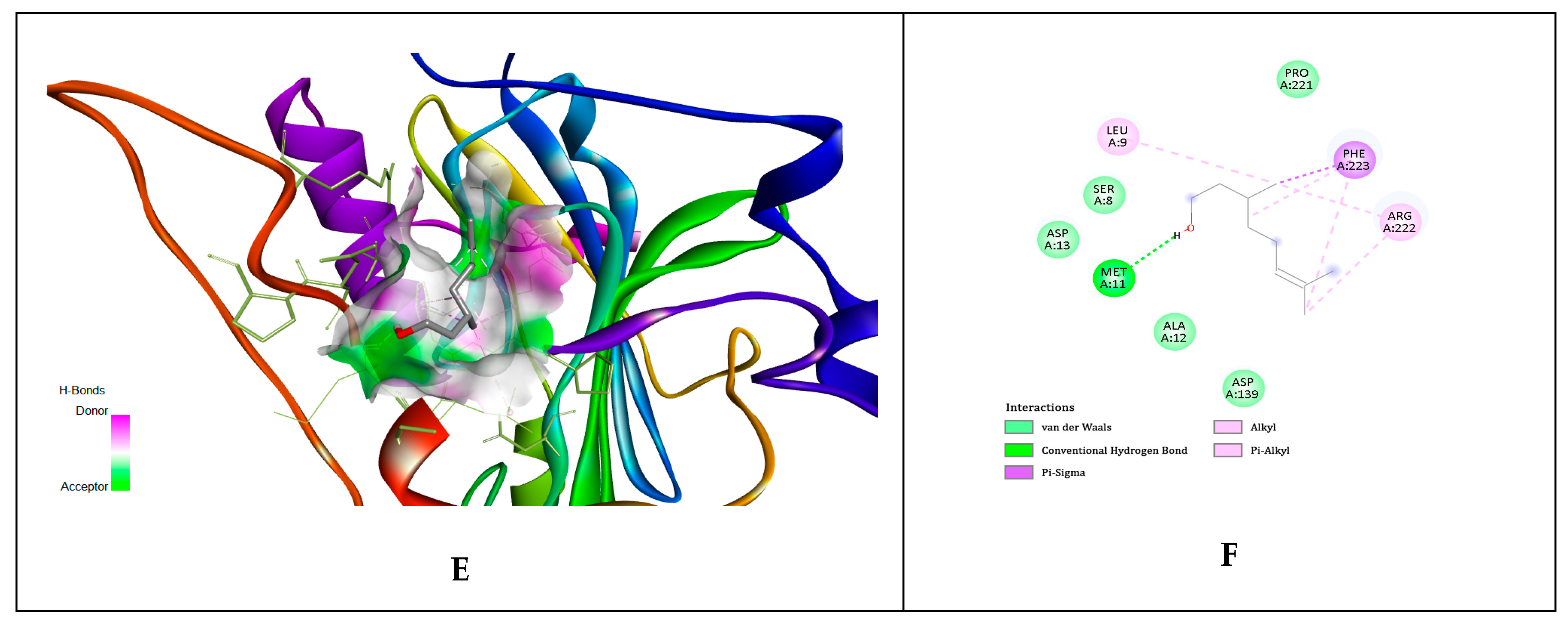
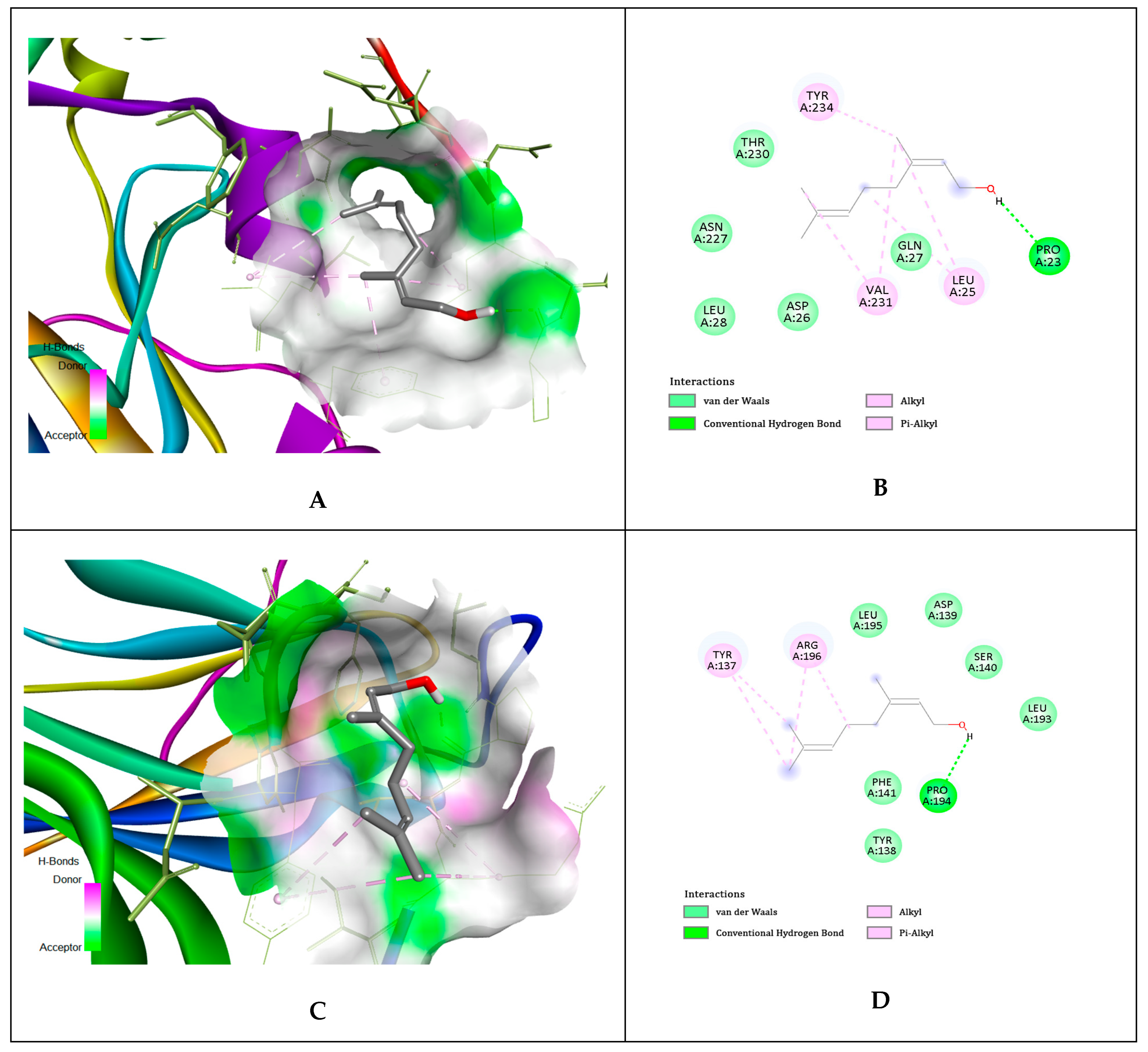
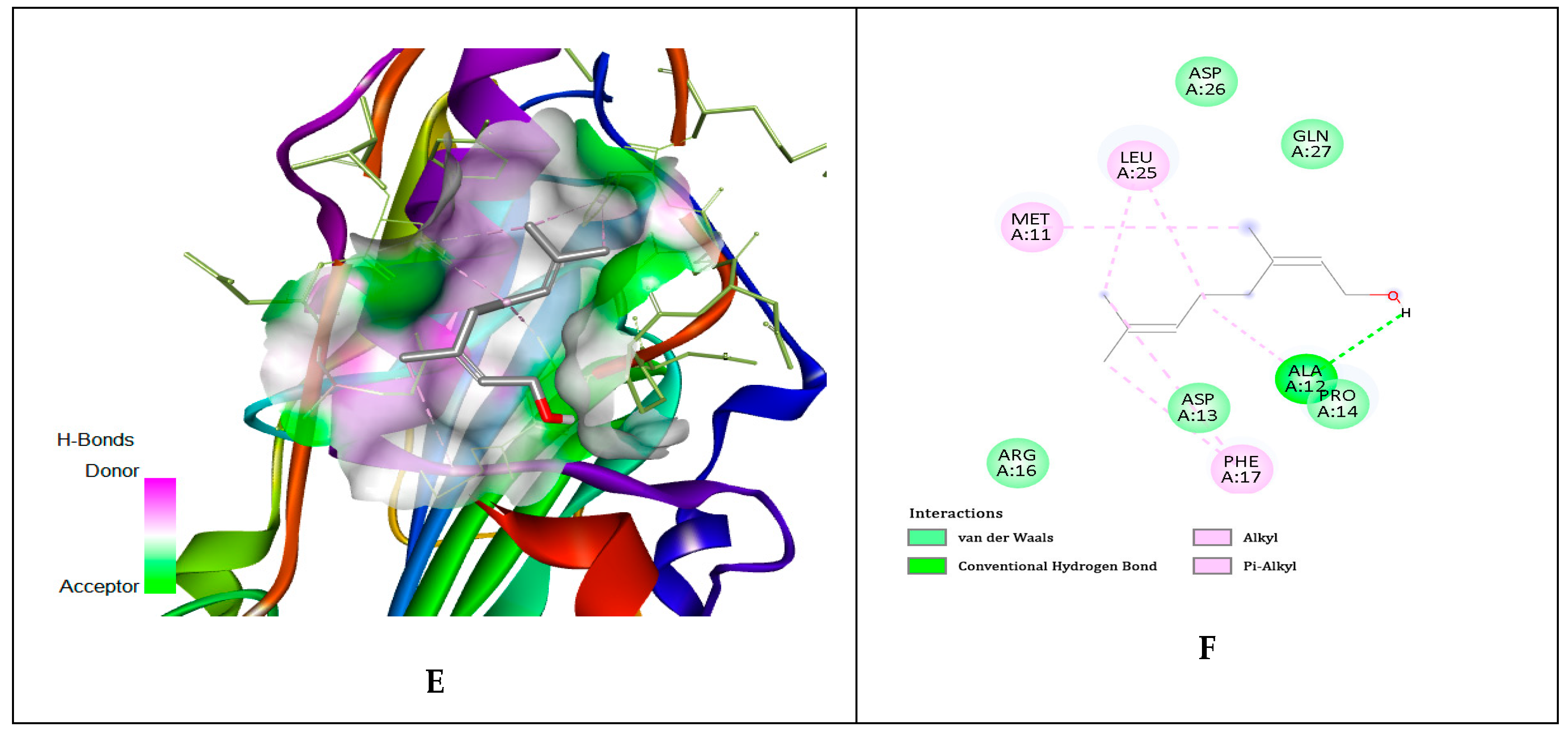
| No. | Compounds Identified Agilent 7890A GC-FID | Results Relative% | Standard ISO 9842:2024 Relative% |
|---|---|---|---|
| 1 | Ethanol | 0.06 | ≤3.0 |
| 2 | Limonene | 0.05 | |
| 3 | Linalool | 0.53 | |
| 4 | Phenylethanol | 0.96 | ≤2.5 |
| 5 | Cis-rose oxide | 0.23 | |
| 6 | Trans-rose oxide | 0.13 | |
| 7 | Citronellol | 21.50 | 20.0–34.0 |
| 8 | Nerol | 5.51 | 5.0–12.0 |
| 9 | Geraniol | 28.73 | 14.0–22.0 |
| 10 | Eugenol | 0.85 | |
| 11 | Methyl eugenol | 0.70 | 0.8–3.0 |
| 12 | Heptadecane | 2.29 | 1.0–2.5 |
| 13 | Farnesol | 2.11 | |
| 14 | Nonadecene | 3.93 | 1.5–4.0 |
| 15 | Nonadecane | 13.13 | 8.0–15.0 |
| 16 | Eicosane | 1.01 | |
| 17 | Heneicosane | 4.87 | 3.0–5.5 |
| 18 | Tricosane | 1.29 | |
| 19 | Pentacosane | 0.46 | |
| 20 | Heptacosane | 0.39 |
| No. | Tested Indicators | Results | Standard ISO 9842:2024 |
|---|---|---|---|
| 1. | Appearance | Colorless liquid | Colorless liquid |
| 2. | Color | Colorless | Colorless |
| 3. | Smell | Typical of rose flowers | Typical of rose flowers |
| 4. | pH | 5.0 | From 4.00 to 7.50 |
| 5. | Content of essential oil, % | 0.33 | No less than 0.025 |
| 6. | Content of ethyl alcohol, % | 0.00 | Not more than 4.00 |
| Compound Tested | Cytotoxicity | |
|---|---|---|
| CC50 Mean ± SD | MTC | |
| R. damascena essential oil (μg/mL) | 223.8 ± 5.3 *** | 10.0 |
| R. damascena water (%) | >50 | 50.0 |
| ACV (μg/mL) | 182.4 ± 6.8 | 100.0 |
| Compounds | Δlg ± SD | ||||
|---|---|---|---|---|---|
| Virus Treatment Time | |||||
| 15 min | 30 min | 60 min | 90 min | 120 min | |
| Rosa damascena essential oil | 1.5 ± 0.20 | 2.0 ± 0.17 * | 3.0 ± 0.40 **** | 3.0 ± 0.38 **** | 3.0 ± 0.28 **** |
| Rosa damascena water | 1.5 ± 0.18 | 2.0 ± 0.25 * | 3.25 ± 0.24 **** | 3.25 ± 0.30 **** | 3.25 ± 0.34 **** |
| 70% ethanol | 5.0 ± 0.12 | 5.0 ± 0.13 ns | 5.0 ± 0.13 ns | 5.0 ± 0.12 ns | 5.0 ± 0.13 ns |
| Compounds | Δlg ± SD | |||
|---|---|---|---|---|
| Virus Treatment Time | ||||
| 15 min | 30 min | 45 min | 60 min | |
| Rosa damascena essential oil | 1.25 ± 0.22 | 1.5 ± 0.22 ns | 1.75 ± 0.24 * | 2.0 ± 0.26 ** |
| Rosa damascena water | 1.5 ± 0.18 | 1.5 ± 0.22 ns | 2.0 ± 0.19 * | 2.25 ± 0.20 ** |
| Compounds | Δlg ± SD | ||||
|---|---|---|---|---|---|
| Treatment Time for Healthy Cells | |||||
| 15 min | 30 min | 60 min | 90 min | 120 min | |
| Rosa damascena essential oil | 0.5 ± 0.18 | 1.0 ± 0.20 * | 1.75 ± 0.22 **** | 2.0 ± 0.23 **** | 2.5 ± 0.24 **** |
| Rosa damascena water | 0.5 ± 0.19 | 1.25 ± 0.22 *** | 1.5 ± 0.21 **** | 1.75 ± 0.23 **** | 2.0 ± 0.25 **** |
| Ligands | gD N-Terminal Loop (at Nectin-1 Binding Site) Interactions with Ligand (Kcal/mol) | gD C-Terminal Extention (at Nectin-1 Binding Site) Interactions with Ligand (Kcal/mol) | gD N-Terminal Loop (at HVEM Binding Site) Interactions with Ligand (Kcal/mol) |
|---|---|---|---|
| Geraniol | −5.81 | −5.84 | −5.65 |
| Citronellol | −4.97 | −4.62 | −4.89 |
| Nerol | −4.58 | −4.54 | −6.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilhelmova-Ilieva, N.; Nenova, R.; Kalinov, K.; Dobreva, A.; Peshev, D.; Iliev, I. Anti-Herpes Simplex Virus Type 1 Activity of Rosa damascena Mill Essential Oil and Floral Water in Retinal Infection In Vitro and In Silico. Int. J. Mol. Sci. 2025, 26, 7521. https://doi.org/10.3390/ijms26157521
Vilhelmova-Ilieva N, Nenova R, Kalinov K, Dobreva A, Peshev D, Iliev I. Anti-Herpes Simplex Virus Type 1 Activity of Rosa damascena Mill Essential Oil and Floral Water in Retinal Infection In Vitro and In Silico. International Journal of Molecular Sciences. 2025; 26(15):7521. https://doi.org/10.3390/ijms26157521
Chicago/Turabian StyleVilhelmova-Ilieva, Neli, Rayna Nenova, Kalin Kalinov, Ana Dobreva, Dimitar Peshev, and Ivan Iliev. 2025. "Anti-Herpes Simplex Virus Type 1 Activity of Rosa damascena Mill Essential Oil and Floral Water in Retinal Infection In Vitro and In Silico" International Journal of Molecular Sciences 26, no. 15: 7521. https://doi.org/10.3390/ijms26157521
APA StyleVilhelmova-Ilieva, N., Nenova, R., Kalinov, K., Dobreva, A., Peshev, D., & Iliev, I. (2025). Anti-Herpes Simplex Virus Type 1 Activity of Rosa damascena Mill Essential Oil and Floral Water in Retinal Infection In Vitro and In Silico. International Journal of Molecular Sciences, 26(15), 7521. https://doi.org/10.3390/ijms26157521








