Phytocannabinoids: Exploring Pharmacological Profiles and Their Impact on Therapeutical Use
Abstract
1. Introduction
2. Pharmacological Profiles of Key Phytocannabinoids
2.1. Δ9-Tetrahydrocannabinol (THC)
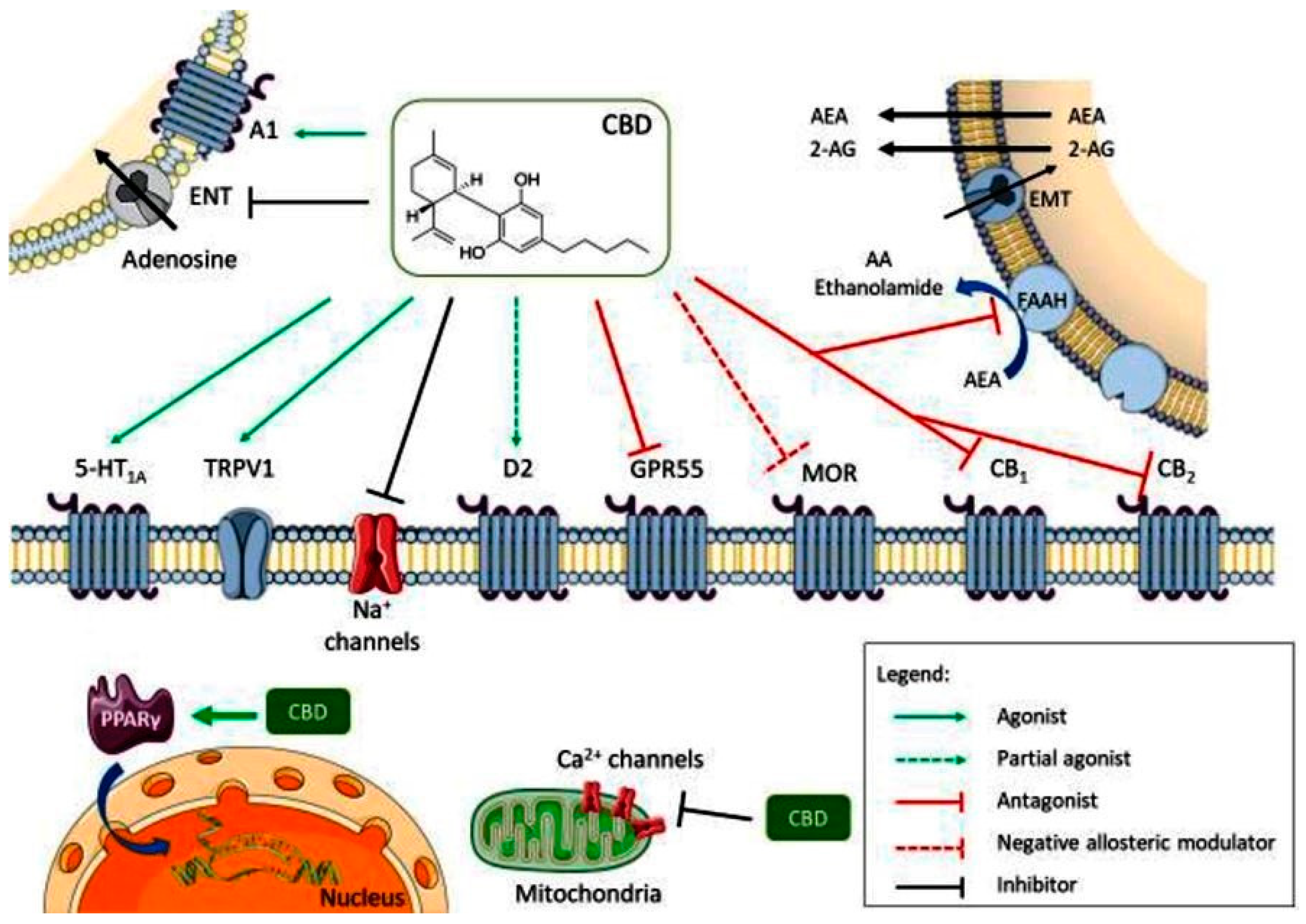
2.2. Cannabidiol (CBD)
2.3. Cannabigerol (CBG)
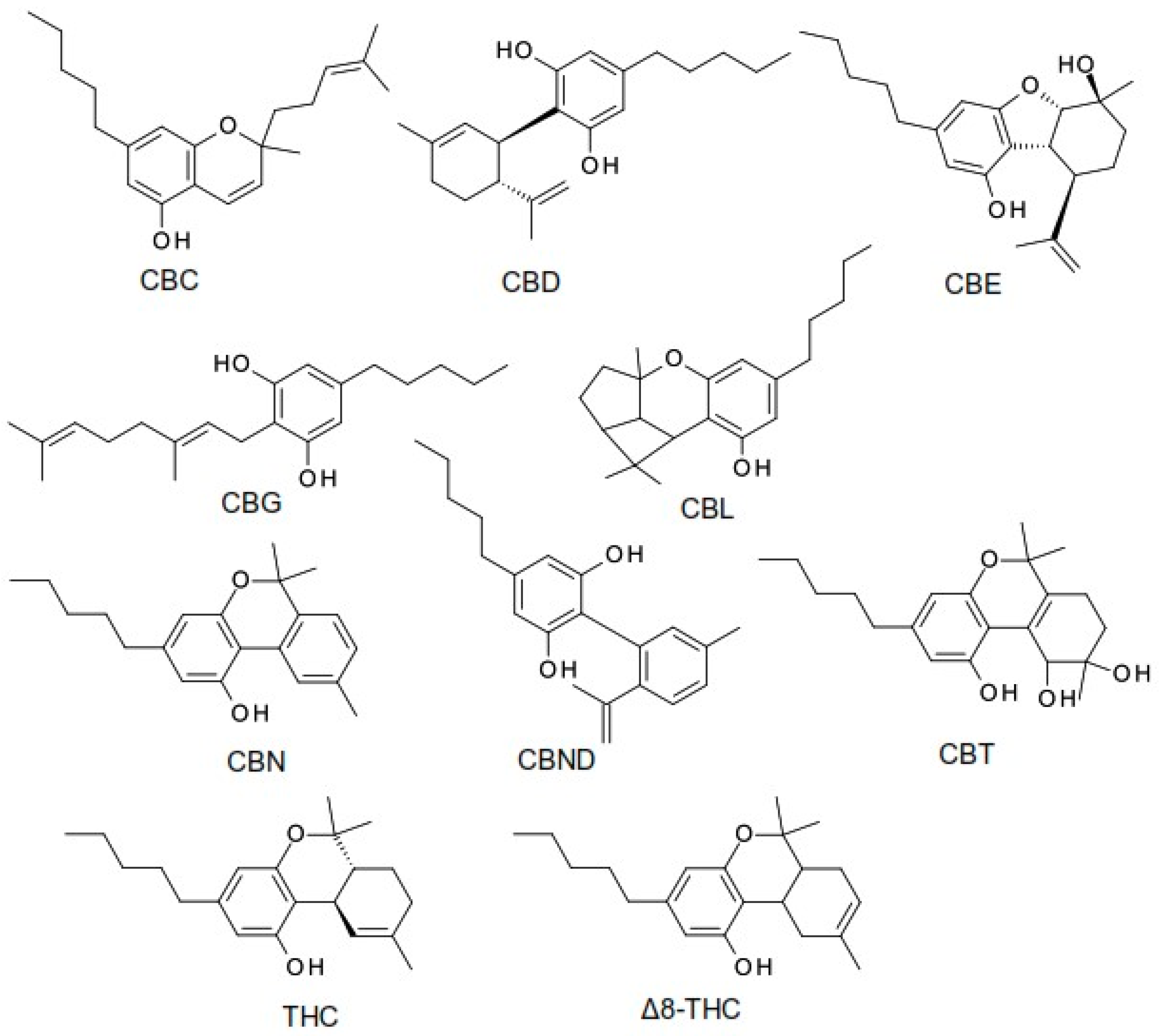
2.4. Other Phytocannabinoids
2.4.1. Cannabichromene (CBC)
2.4.2. Cannabinol (CBN)
2.4.3. Tetrahydrocannabivarin (THCV)
3. Therapeutic Applications
3.1. Anti-Inflammatory Effects
3.2. Antioxidant Effects
3.3. Pain Management
3.4. Neurological Disorders
3.5. Psychiatric Conditions
3.6. Cancer Treatment Support
3.7. Other Emerging Areas
4. Challenges and Controversies
4.1. Legal and Regulatory Issues
4.2. Standardization of Phytocannabinoid Products
4.3. Potential for Abuse and Dependence
5. Future Directions
5.1. Ongoing Research and Preclinical Trials
5.2. Exploration of Novel Phytocannabinoids
5.3. Integration into Mainstream Medicine
5.4. Implications for the Future of Pharmacotherapy
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef]
- Zuardi, A.W. History of cannabis as a medicine: A review. Braz. J. Psychiatry 2006, 28, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Klumpers, L.E.; Thacker, D.L. A brief background on cannabis: From plant to medical indications. J. AOAC Int. 2019, 102, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Grof, C.P. Cannabis, from plant to pill. Br. J. Clin. Pharmacol. 2018, 84, 2463–2467. [Google Scholar] [CrossRef] [PubMed]
- Reekie, T.A.; Scott, M.P.; Kassiou, M. The evolving science of phytocannabinoids. Nat. Rev. Chem. 2017, 2, 0101. [Google Scholar] [CrossRef]
- Morales, P.; Reggio, P.H.; Jagerovic, N. An overview on medicinal chemistry of synthetic and natural derivatives of cannabidiol. Front. Pharmacol. 2017, 8, 422. [Google Scholar] [CrossRef] [PubMed]
- Gülck, T.; Møller, B.L. Phytocannabinoids: Origins and biosynthesis. Trends Plant Sci. 2020, 25, 985–1004. [Google Scholar] [CrossRef] [PubMed]
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: A unified critical inventory. Nat. Prod. Rep. 2016, 33, 1357–1392. [Google Scholar] [CrossRef] [PubMed]
- Koltai, H.; Namdar, D. Cannabis phytomolecule ‘entourage’: From domestication to medical use. Trends Plant Sci. 2020, 25, 976–984. [Google Scholar] [CrossRef]
- Marzo, V.D.; Bifulco, M.; Petrocellis, L.D. The endocannabinoid system and its therapeutic exploitation. Nat. Rev. Drug Discov. 2004, 3, 771–784. [Google Scholar] [CrossRef]
- Lu, H.C.; Mackie, K. Review of the endocannabinoid system. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2021, 6, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Mackie, K. Cannabinoid receptors: Where they are and what they do. J. Neuroendocrinol. 2008, 20, 10–14. [Google Scholar] [CrossRef] [PubMed]
- de Almeida, D.L.; Devi, L.A. Diversity of molecular targets and signaling pathways for CBD. Pharmacol. Res. Perspect. 2020, 8, e00682. [Google Scholar] [CrossRef] [PubMed]
- Abu-Sawwa, R.; Scutt, B.; Park, Y. Emerging use of Epidiolex (cannabidiol) in epilepsy. J. Pediatr. Pharmacol. Ther. 2020, 25, 485–499. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M.P. Sativex®: Clinical efficacy and tolerability in the treatment of symptoms of multiple sclerosis and neuropathic pain. Expert Opin. Pharmacother. 2006, 7, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Costa, B. On the pharmacological properties of Δ9-tetrahydrocannabinol (THC). Chem. Biodivers. 2007, 4, 1664–1677. [Google Scholar] [CrossRef] [PubMed]
- Dinis-Oliveira, R.J. Metabolomics of Δ9-tetrahydrocannabinol: Implications in toxicity. Drug Metab. Rev. 2016, 48, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Kosersky, D.S.; Dewey, W.L.; Harris, L.S. Antipyretic, analgesic, and anti-inflammatory effects of Δ9-tetrahydrocannabinol in the rat. Eur. J. Pharmacol. 1973, 24, 1–7. [Google Scholar] [CrossRef]
- Sweet, D.L.; Miller, N.J.; Weddington, W.; Senay, E.; Sushelsky, L. Δ9-tetrahydrocannabinol as an antiemetic for patients receiving cancer chemotherapy: A pilot study. J. Clin. Pharmacol. 1981, 21, 70S–75S. [Google Scholar] [CrossRef]
- Vlad, R.A.; Hancu, G.; Ciurba, A.; Antonoaea, P.; Rédai, E.M.; Todoran, N.; Muntean, D.L. Cannabidiol-therapeutic and legal aspects. Pharm. Int. J. Pharm. Sci. 2020, 75, 463–469. [Google Scholar]
- Mechoulam, R.; Hanuš, L. Cannabidiol: An overview of some chemical and pharmacological aspects. Part I Chem. Asp. Chem. Phys. Lipids 2002, 121, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and anti-inflammatory properties of cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Nachnani, R.; Raup-Konsavage, W.M.; Vrana, K.E. The pharmacological case for cannabigerol. J. Pharmacol. Exp. Ther. 2021, 376, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Jastrząb, A.; Jarocka-Karpowicz, I.; Skrzydlewska, E. The origin and biomedical relevance of cannabigerol. Int. J. Mol. Sci. 2022, 23, 7929. [Google Scholar] [CrossRef] [PubMed]
- Gaoni, Y.; Mechoulam, R. Cannabichromene, a new active principle in hashish. Chem. Commun. 1966, 20–21. [Google Scholar] [CrossRef]
- Pollastro, F.; Caprioglio, D.; Del Prete, D.; Rogati, F.; Minassi, A.; Taglialatela-Scafati, O.; Appendino, G. Cannabichromene. Nat. Prod. Commun. 2018, 13, 1189–1194. [Google Scholar] [CrossRef]
- Corroon, J. Cannabinol and sleep: Separating fact from fiction. Cannabis Cannabinoid Res. 2021, 6, 366–371. [Google Scholar] [CrossRef] [PubMed]
- McPartland, J.M.; Duncan, M.; Di Marzo, V.; Pertwee, R.G. Are cannabidiol and Δ9-tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review. Br. J. Pharmacol. 2015, 172, 737–753. [Google Scholar] [CrossRef]
- Nagarkatti, P.; Pandey, R.; Rieder, S.A.; Hegde, V.L.; Nagarkatti, M. Cannabinoids as novel anti-inflammatory drugs. Future Med. Chem. 2009, 1, 1333–1349. [Google Scholar] [CrossRef]
- Croxford, J.L.; Yamamura, T. Cannabinoids and the immune system: Potential for the treatment of inflammatory diseases? J. Neuroimmunol. 2005, 166, 3–18. [Google Scholar] [CrossRef]
- Henshaw, F.R.; Dewsbury, L.S.; Lim, C.K.; Steiner, G.Z. The effects of cannabinoids on pro-and anti-inflammatory cytokines: A systematic review of in vivo studies. Cannabis Cannabinoid Res. 2021, 6, 177–195. [Google Scholar] [CrossRef] [PubMed]
- Solinas, M.; Massi, P.; Cinquina, V.; Valenti, M.; Bolognini, D.; Gariboldi, M.; Monti, E.; Rubino, T.; Parolaro, D. Cannabidiol, a non-psychoactive cannabinoid compound, inhibits proliferation and invasion in U87-MG and T98G glioma cells through a multitarget effect. PLoS ONE 2013, 8, e76918. [Google Scholar] [CrossRef] [PubMed]
- Khodadadi, H.; Salles, É.L.; Jarrahi, A.; Chibane, F.; Costigliola, V.; Yu, J.C.; Vaibhav, K.; Hess, D.C.; Dhandapani, K.M.; Baban, B. Cannabidiol modulates cytokine storm in acute respiratory distress syndrome induced by simulated viral infection using synthetic RNA. Cannabis Cannabinoid Res. 2020, 5, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Van Niekerk, G.; Mabin, T.; Engelbrecht, A.M. Anti-inflammatory mechanisms of cannabinoids: An immunometabolic perspective. Inflammopharmacology 2019, 27, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Britch, S.C.; Goodman, A.G.; Wiley, J.L.; Pondelick, A.M.; Craft, R.M. Antinociceptive and immune effects of delta-9-tetrahydrocannabinol or cannabidiol in male versus female rats with persistent inflammatory pain. J. Pharmacol. Exp. Ther. 2020, 373, 416–428. [Google Scholar] [CrossRef] [PubMed]
- Neelakantan, H.; Tallarida, R.J.; Reichenbach, Z.W.; Tuma, R.F.; Ward, S.J.; Walker, E.A. Distinct interactions of cannabidiol and morphine in three nociceptive behavioral models in mice. Behav. Pharmacol. 2015, 26, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Zurier, R.B. Prospects for cannabinoids as anti-inflammatory agents. J. Cell. Biochem. 2003, 88, 462–466. [Google Scholar] [CrossRef]
- Klein, T.W. Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat. Rev. Immunol. 2005, 5, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Katz-Talmor, D.; Katz, I.; Porat-Katz, B.S.; Shoenfeld, Y. Cannabinoids for the treatment of rheumatic diseases—Where do we stand? Nat. Rev. Rheumatol. 2018, 14, 488–498. [Google Scholar] [CrossRef]
- Gamble, L.J.; Boesch, J.M.; Frye, C.W.; Schwark, W.S.; Mann, S.; Wolfe, L.; Wakshlag, J.J. Pharmacokinetics, safety, and clinical efficacy of cannabidiol treatment in osteoarthritic dogs. Front. Vet. Sci. 2018, 5, 165. [Google Scholar] [CrossRef]
- Biernacki, M.; Jastrząb, A.; Skrzydlewska, E. Changes in hepatic phospholipid metabolism in rats under UV irradiation and topically treated with cannabidiol. Antioxidants 2021, 10, 1157. [Google Scholar] [CrossRef] [PubMed]
- Hampson, A.J.; Grimaldi, M.; Lolic, M.; Wink, D.; Rosenthal, R.; Axelrod, J. Neuroprotective antioxidants from marijuana. Ann. N. Y. Acad. Sci. 2000, 899, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Scotter, E.L.; Abood, M.E.; Glass, M. The endocannabinoid system as a target for the treatment of neurodegenerative disease. Br. J. Pharmacol. 2010, 160, 480–498. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.J.; Diemel, L.T.; Pryce, G.; Baker, D. Cannabinoids and neuroprotection in CNS inflammatory disease. J. Neurol. Sci. 2005, 233, 21–25. [Google Scholar] [CrossRef]
- Elikottil, J.; Gupta, P.; Gupta, K. The analgesic potential of cannabinoids. J. Opioid Manag. 2009, 5, 341. [Google Scholar] [CrossRef]
- Walker, J.M.; Huang, S.M. Cannabinoid analgesia. Pharmacol. Ther. 2002, 95, 127–135. [Google Scholar] [CrossRef]
- Păunescu, H.; Coman, O.A.; Coman, L.; Ghiţă, I.; Georgescu, S.R.; Drăia, F.; Fulga, I. Cannabinoid system and cyclooxygenases inhibitors. J. Med. Life 2011, 4, 11. [Google Scholar] [PubMed]
- Vanegas, H.; Vazquez, E.; Tortorici, V. NSAIDs, opioids, cannabinoids and the control of pain by the central nervous system. Pharmaceuticals 2010, 3, 1335–1347. [Google Scholar] [CrossRef]
- Stevens, A.J.; Higgins, M.D. A systematic review of the analgesic efficacy of cannabinoid medications in the management of acute pain. Acta Anaesthesiol. Scand. 2017, 61, 268–280. [Google Scholar] [CrossRef]
- Russo, E.B. Cannabinoids in the management of difficult to treat pain. Ther. Clin. Risk Manag. 2008, 4, 245–259. [Google Scholar] [CrossRef]
- Pergolizzi, J.V., Jr.; LeQuang, J.A.; Taylor, R., Jr.; Raffa, R.B.; Colucci, D. The role of cannabinoids in pain control: The good, the bad, and the ugly. Minerva Anestesiol. 2018, 84, 955–969. [Google Scholar] [CrossRef] [PubMed]
- Vučković, S.; Srebro, D.; Vujović, K.S.; Vučetić, Č.; Prostran, M. Cannabinoids and pain: New insights from old molecules. Front. Pharmacol. 2018, 9, 1259. [Google Scholar] [CrossRef]
- Johal, H.; Devji, T.; Chang, Y.; Simone, J.; Vannabouathong, C.; Bhandari, M. Cannabinoids in chronic non-cancer pain: A systematic review and meta-analysis. Clin. Med. Insights Arthritis Musculoskelet. Disord. 2020, 13, 1179544120906461. [Google Scholar] [CrossRef]
- Abu-Sawwa, R.; Stehling, C. Epidiolex (cannabidiol) primer: Frequently asked questions for patients and caregivers. J. Pediatr. Pharmacol. Ther. 2020, 25, 75–77. [Google Scholar] [PubMed]
- Giacoppo, S.; Bramanti, P.; Mazzon, E. Sativex in the management of multiple sclerosis-related spasticity: An overview of the last decade of clinical evaluation. Mult. Scler. Relat. Dis. 2017, 17, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Stampanoni Bassi, M.; Sancesario, A.; Morace, R.; Centonze, D.; Iezzi, E. Cannabinoids in Parkinson’s disease. Cannabis Cannabinoid Res. 2017, 2, 21–29. [Google Scholar] [CrossRef]
- Boychuk, D.G.; Goddard, G.; Mauro, G.; Orellana, M.F. The effectiveness of cannabinoids in the management of chronic nonmalignant neuropathic pain: A systematic review. J. Oral Facial Pain Headache 2015, 29, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Aso, E.; Ferrer, I. Cannabinoids for treatment of Alzheimer’s disease: Moving toward the clinic. Front. Pharmacol. 2014, 5, 37. [Google Scholar] [CrossRef] [PubMed]
- Cristino, L.; Bisogno, T.; Di Marzo, V. Cannabinoids and the expanded endocannabinoid system in neurological disorders. Nat. Rev. Neurol. 2020, 16, 9–29. [Google Scholar] [CrossRef]
- Moreira, F.A.; Wotjak, C.T. Cannabinoids and anxiety. Behav. Neurobiol. Anxiety Treat. 2010, 2, 429–450. [Google Scholar]
- Poleszak, E.; Wośko, S.; Sławińska, K.; Szopa, A.; Wróbel, A.; Serefko, A. Cannabinoids in depressive disorders. Life Sci. 2018, 213, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.A.B.; Filev, R.; Fidalgo, T.M. Cannabinoids for Substance Use Disorder Treatment: What Does the Current Evidence Say? Cannabis Cannabinoid Res. 2023, 8, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Sarfaraz, S.; Adhami, V.M.; Syed, D.N.; Afaq, F.; Mukhtar, H. Cannabinoids for cancer treatment: Progress and promise. Cancer Res. 2008, 68, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Dariš, B.; Verboten, M.T.; Knez, Ž.; Ferk, P. Cannabinoids in cancer treatment: Therapeutic potential and legislation. Bosn. J. Basic Med. Sci. 2019, 19, 14. [Google Scholar] [CrossRef] [PubMed]
- Alexander, A.; Smith, P.F.; Rosengren, R.J. Cannabinoids in the treatment of cancer. Cancer Lett. 2009, 285, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Bifulco, M.; Laezza, C.; Pisanti, S.; Gazzerro, P. Cannabinoids and cancer: Pros and cons of an antitumour strategy. Br. J. Pharmacol. 2006, 148, 123–135. [Google Scholar] [CrossRef]
- Karas, J.A.; Wong, L.J.; Paulin, O.K.; Mazeh, A.C.; Hussein, M.H.; Li, J.; Velkov, T. The antimicrobial activity of cannabinoids. Antibiotics 2020, 9, 406. [Google Scholar] [CrossRef] [PubMed]
- Aqawi, M.; Sionov, R.V.; Friedman, M.; Steinberg, D. The antibacterial effect of cannabigerol toward Streptococcus mutans is influenced by the autoinducers 21-CSP and AI-2. Biomedicines 2023, 11, 668. [Google Scholar] [CrossRef] [PubMed]
- Horváth, B.; Mukhopadhyay, P.; Haskó, G.; Pacher, P. The endocannabinoid system and plant-derived cannabinoids in diabetes and diabetic complications. Am. J. Pathol. 2012, 180, 432–442. [Google Scholar] [CrossRef]
- Hall, W.; Lynskey, M. Evaluating the public health impacts of legalizing recreational cannabis use in the United States. Addiction 2016, 111, 1764–1773. [Google Scholar] [CrossRef]
- Cohen, P.A.; Sharfstein, J. The opportunity of CBD—Reforming the law. N. Engl. J. Med. 2019, 381, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Corroon, J.; Kight, R. Regulatory status of cannabidiol in the United States: A perspective. Cannabis Cannabinoid Res. 2018, 3, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Smart, R.; Pacula, R.L. Early evidence of the impact of cannabis legalization on cannabis use, cannabis use disorder, and the use of other substances: Findings from state policy evaluations. Am. J. Drug Alcohol Abus. 2019, 45, 644–663. [Google Scholar] [CrossRef]
- Hammond, D.; Goodman, S.; Wadsworth, E.; Rynard, V.; Boudreau, C.; Hall, W. Evaluating the impacts of cannabis legalization: The International Cannabis Policy Study. Int. J. Drug Policy 2020, 77, 102698. [Google Scholar] [CrossRef]
- Walker, L.A.; Koturbash, I.; Kingston, R.; ElSohly, M.A.; Yates, C.R.; Gurley, B.J.; Khan, I. Cannabidiol (CBD) in dietary supplements: Perspectives on science, safety, and potential regulatory approaches. J. Diet. Suppl. 2020, 17, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Bartončíková, M.; Lapčíková, B.; Lapčík, L.; Valenta, T. Hemp-Derived CBD Used in Food and Food Supplements. Molecules 2023, 28, 8047. [Google Scholar] [CrossRef]
- Stefkov, G.; Cvetkovikj Karanfilova, I.; Stoilkovska Gjorgievska, V.; Trajkovska, A.; Geskovski, N.; Karapandzova, M.; Kulevanova, S. Analytical techniques for phytocannabinoid profiling of cannabis and cannabis-based products—A comprehensive review. Molecules 2022, 27, 975. [Google Scholar] [CrossRef]
- Nahler, G.; Jones, T.M.; Russo, E.B. Cannabidiol and contributions of major hemp phytocompounds to the “entourage effect”; possible mechanisms. J. Altern. Complement. Integr. Med. 2019, 5, 070. [Google Scholar] [CrossRef]
- Blebea, N.M.; Rambu, D.; Costache, T.; Negreș, S. Very fast RP–UHPLC–PDA method for identification and quantification of the cannabinoids from hemp oil. Appl. Sci. 2021, 11, 9414. [Google Scholar] [CrossRef]
- Kubilius, R.A.; Kaplick, P.M.; Wotjak, C.T. Highway to hell or magic smoke? The dose-dependence of Δ9-THC in place conditioning paradigms. Learn. Mem. 2018, 25, 446–454. [Google Scholar] [CrossRef]
- Batalla, A.; Janssen, H.; Gangadin, S.S.; Bossong, M.G. The potential of cannabidiol as a treatment for psychosis and addiction: Who benefits most? A systematic review. J. Clin. Med. 2019, 8, 1058. [Google Scholar] [CrossRef] [PubMed]
- Pennypacker, S.D.; Romero-Sandoval, E.A. CBD and THC: Do they complement each other like Yin and Yang? Pharmacotherapy: J. Hum. Pharmacol. Drug Ther. 2020, 40, 1152–1165. [Google Scholar] [CrossRef] [PubMed]
- Dei Cas, M.; Casagni, E.; Casiraghi, A.; Minghetti, P.; Fornasari, D.M.M.; Ferri, F.; Roda, G. Phytocannabinoids profile in medicinal cannabis oils: The impact of plant varieties and preparation methods. Front. Pharmacol. 2020, 11, 570616. [Google Scholar] [CrossRef] [PubMed]
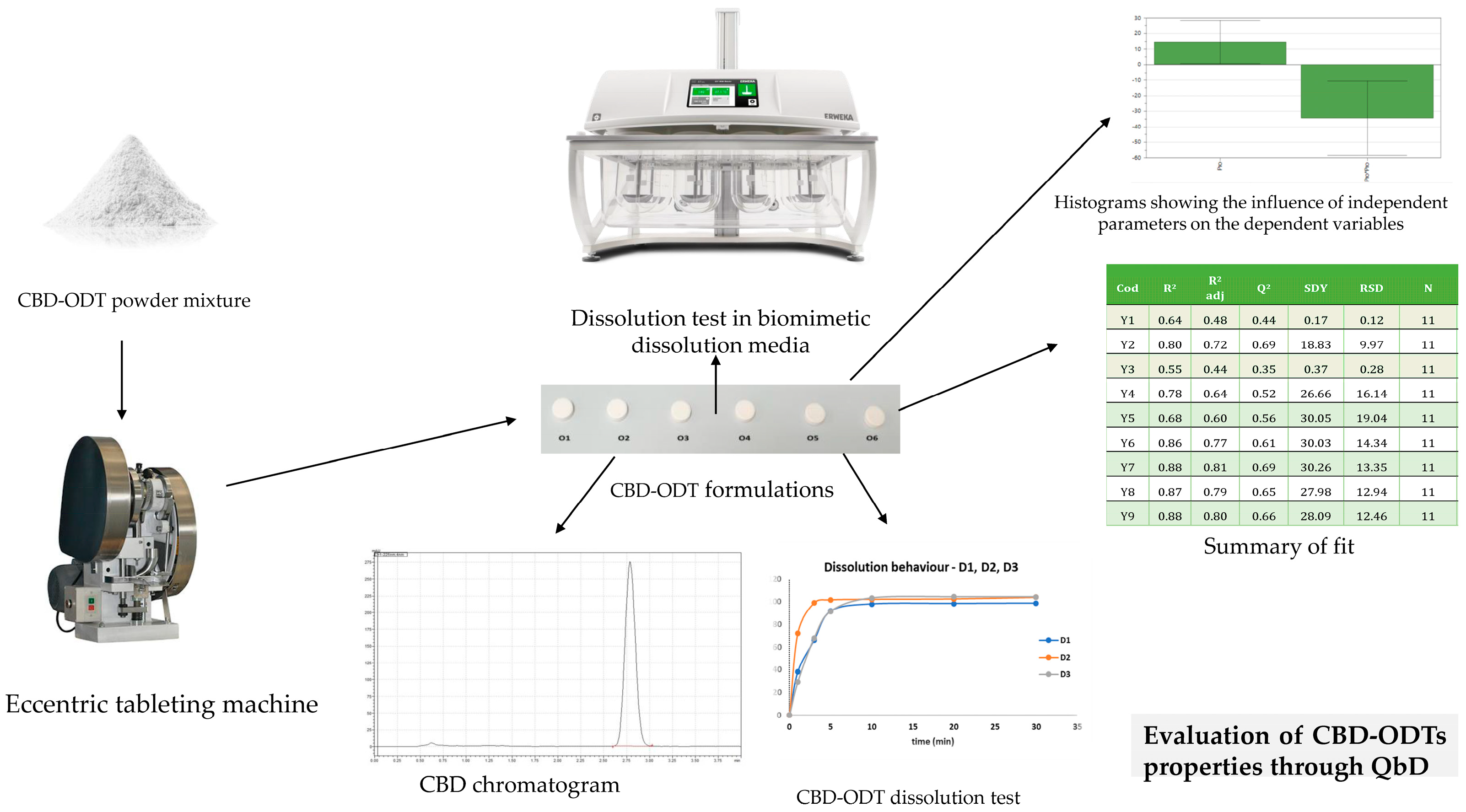
- Vlad, R.A.; Antonoaea, P.; Todoran, N.; Muntean, D.L.; Redai, E.M.; Silași, O.-A.; Tătaru, A.; Bîrsan, M.; Imre, S.; Ciurba, A. Pharmacotechnical and analytical preformulation studies for cannabidiol orodispersible tablets. SPJ 2021, 29, 1029–1042. [Google Scholar] [CrossRef] [PubMed]
- Vlad, R.A.; Antonoaea, P.; Todoran, N.; Rédai, E.M.; Bîrsan, M.; Muntean, D.L.; Imre, S.; Hancu, G.; Farczádi, L.; Ciurba, A. Development and Evaluation of Cannabidiol Orodispersible Tablets Using a 23-Factorial Design. Pharmaceutics 2022, 14, 1467. [Google Scholar] [CrossRef] [PubMed]
- Limpongsa, E.; Tabboon, P.; Pongjanyakul, T.; Jaipakdee, N. Preparation and Evaluation of Directly Compressible Orally Disintegrating Tablets of Cannabidiol Formulated Using Liquisolid Technique. Pharmaceutics 2022, 14, 2407. [Google Scholar] [CrossRef] [PubMed]
- Vlad, R.-A.; Farczadi, L.; Toma, C.-M.; Imre, S.; Antonoaea, P.; Redai, E.M.; Muntean, D.-L.; Ciurba, A. Cannabidiol content evaluation in commercial dietary supplements and stability in oil vehicle. Stud. Univ. Babes-Bolyai Chem. 2021, 66, 299. [Google Scholar]
- Fraguas-Sánchez, A.I.; Fernández-Carballido, A.; Martin-Sabroso, C.; Torres-Suárez, A.I. Stability characteristics of cannabidiol for the design of pharmacological, biochemical and pharmaceutical studies. J. Chromatogr. B 2020, 1150, 122188. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Tajima, M.; Sugiyama, E.; Sato, V.H.; Sato, H. Development of a novel nano-emulsion formulation to improve intestinal absorption of cannabidiol. Med. Cannabis Cannabinoids 2019, 2, 35–42. [Google Scholar] [CrossRef]
- Stan, D.; Enciu, A.M.; Mateescu, A.L.; Ion, A.C.; Brezeanu, A.C.; Tanase, C. Natural compounds with antimicrobial and antiviral effect and nanocarriers used for their transportation. Front. Pharmacol. 2021, 12, 723233. [Google Scholar] [CrossRef]
- Fu, J.; Zhang, K.; Lu, L.; Li, M.; Han, M.; Guo, Y.; Wang, X. Improved Therapeutic Efficacy of CBD with Good Tolerance in the Treatment of Breast Cancer through Nanoencapsulation and in Combination with 20 (S)-Protopanaxadiol (PPD). Pharmaceutics 2022, 14, 1533. [Google Scholar] [CrossRef] [PubMed]
- Shilo-Benjamini, Y.; Cern, A.; Zilbersheid, D.; Hod, A.; Lavy, E.; Barasch, D.; Barenholz, Y. A case report of subcutaneously injected liposomal cannabidiol formulation used as a compassion therapy for pain management in a dog. Front. Vet. Sci. 2022, 9, 892306. [Google Scholar] [CrossRef] [PubMed]
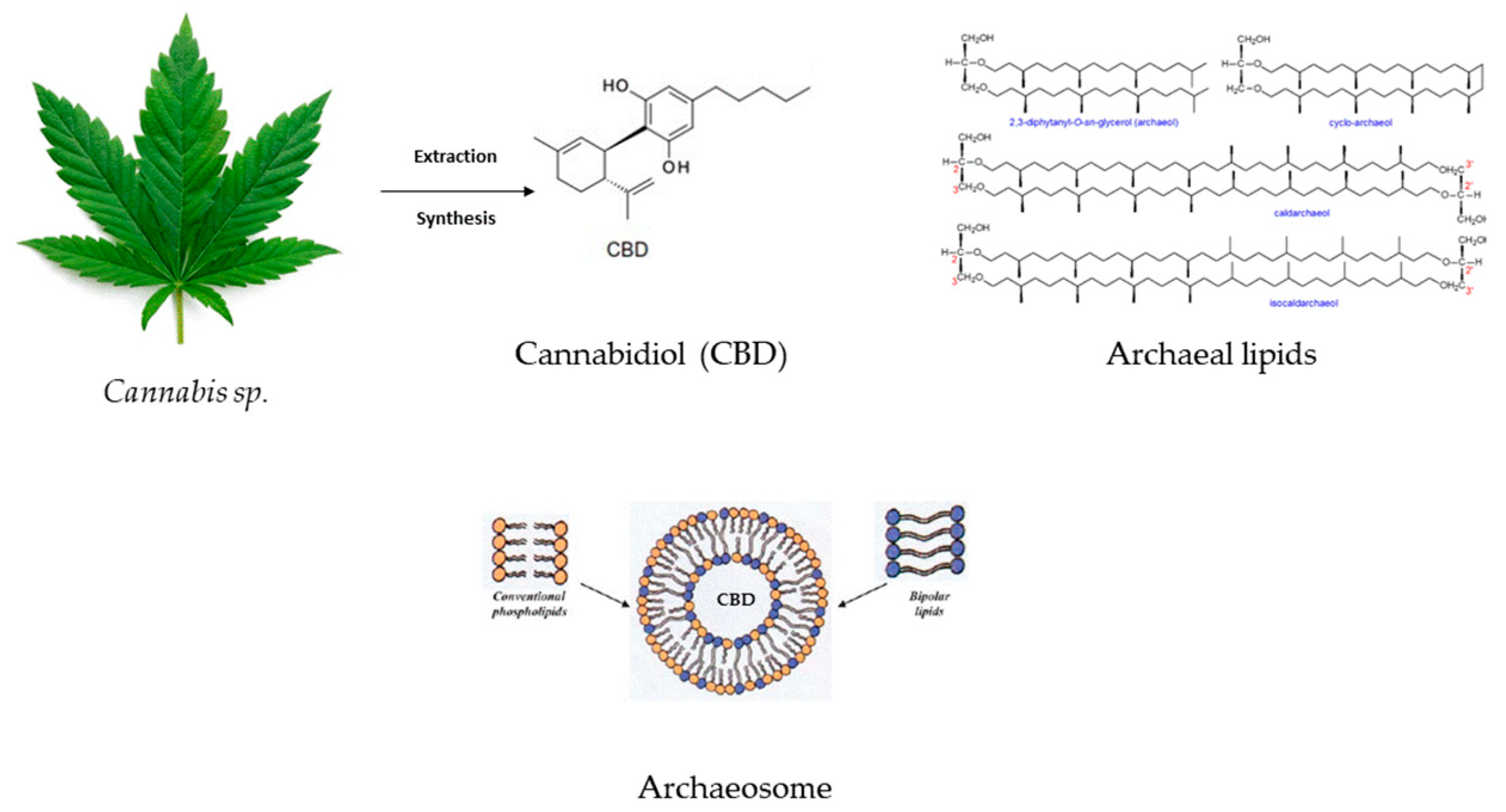
- Sedlmayr, V.; Horn, C.; Wurm, D.J.; Spadiut, O.; Quehenberger, J. Archaeosomes facilitate storage and oral de-livery of cannabidiol. Int. J. Pharm. 2023, 645, 123434. [Google Scholar] [CrossRef] [PubMed]
- Volmajer Valh, J.; Peršin, Z.; Vončina, B.; Vrezner, K.; Tušek, L.; Fras Zemljič, L. Microencapsulation of Canna-bidiol in Liposomes as Coating for Cellulose for Potential Advanced Sanitary Material. Coatings 2021, 11, 3. [Google Scholar] [CrossRef]
- Blair, E.; Miller, A. Liposomal hemp extract for the management of cachexia. Bioact. Compd. Health Dis. 2023, 6, 56–62. [Google Scholar] [CrossRef]
- Franzè, S.; Angelo, L.; Casiraghi, A.; Minghetti, P.; Cilurzo, F. Design of Liposomal Lidocaine/Cannabidiol Fixed Combinations for Local Neuropathic Pain Treatment. Pharmaceutics 2022, 14, 1915. [Google Scholar] [CrossRef] [PubMed]
- Filipiuc, L.E.; Ababei, D.C.; Alexa-Stratulat, T.; Pricope, C.V.; Bild, V.; Stefanescu, R.; Tamba, B.I. Major phytocannabinoids and their related compounds: Should we only search for drugs that act on cannabinoid receptors? Pharmaceutics 2021, 13, 1823. [Google Scholar] [CrossRef]
- Scott, C.; Neira Agonh, D.; Lehmann, C. Antibacterial Effects of Phytocannabinoids. Life 2022, 12, 1394. [Google Scholar] [CrossRef]
- Leinen, Z.J.; Mohan, R.; Premadasa, L.S.; Acharya, A.; Mohan, M.; Byrareddy, S.N. Therapeutic Potential of Cannabis: A Comprehensive Review of Current and Future Applications. Biomedicines 2023, 11, 2630. [Google Scholar] [CrossRef]
| Phytocannabinoids | Interactions | Effects |
|---|---|---|
| Δ9-Tetrahydrocannabinol (THC) |
|
|
| Cannabidiol (CBD) |
|
|
| Cannabigerol (CBG) |
|
|
| Cannabichromene (CBC) |
|
|
| Cannabinol (CBN) |
|
|
| Tetrahydrocannabivarin (THCV) |
|
|
| Cannabinoid | Therapeutic Applications |
|---|---|
| Δ9-Tetrahydrocannabinol (THC) | pain management, anti-inflammatory, nausea reduction, appetite stimulation, muscle spasm reduction |
| Cannabidiol (CBD) | anti-inflammatory, antioxidant, pain management, neurological disorders (e.g., epilepsy, Parkinson’s disease), psychiatric disorders (e.g., anxiety, depression), cancer treatment support, acne treatment, neuroprotective effects |
| Cannabigerol (CBG) | anti-inflammatory, pain management, neuroprotective, potential treatment for glaucoma, antimicrobial properties |
| Cannabichromene (CBC) | anti-inflammatory, pain management, potential antidepressant effects, neuroprotective properties |
| Cannabinol (CBN) | mild sedative, pain relief, potential antibacterial effects, appetite stimulant |
| Tetrahydrocannabivarin (THCV) | appetite suppression, potential treatment for diabetes, neuroprotective effects |
| Product Name | Pharmaceutical Form | CBD Concentration | References |
|---|---|---|---|
| CBD oil | oily solution | 5, 10, 15, 25, 30% | https://www.legalize-it.ro/ulei-cbd-canabis/ |
| Hand cream | semisolid formulation (cream) | 0.2% | https://www.legalize-it.ro/ulei-cbd-canabis/unguent/crema-de-maini-39-cibdol-39-200mg-cbd-100ml-p25054/ |
| Mouthwash | solution | 0.1% | https://www.legalize-it.ro/search/apa%20de%20gura?q=apa+de+gura |
| Chewable tablets | tablets | 600, 1200, 1800 mg | https://www.legalize-it.ro/search/tablete?q=tablete |
| Toothpaste | paste | 0.133% | https://www.legalize-it.ro/ulei-cbd-canabis/unguent/pasta-39-pharmahemp-39-de-dinti-39-cosmohemps-cbd-39-100mg-cbd-75ml-p24680/ |
| Nasal CBD spray | solution with special administration | 0.8% | https://www.legalize-it.ro/ulei-cbd-canabis/unguent/spray-nasal-39-cibdol-39-50mg-cbd-10ml-p25056/ |
| Soothing/calming gel | semisolid formulation (gel) | 0.1% | https://www.legalize-it.ro/ulei-cbd-canabis/gel-39-justcbd-39-pentru-dureri-musculare-1000mg-cbd-p26872/ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blebea, N.M.; Pricopie, A.I.; Vlad, R.-A.; Hancu, G. Phytocannabinoids: Exploring Pharmacological Profiles and Their Impact on Therapeutical Use. Int. J. Mol. Sci. 2024, 25, 4204. https://doi.org/10.3390/ijms25084204
Blebea NM, Pricopie AI, Vlad R-A, Hancu G. Phytocannabinoids: Exploring Pharmacological Profiles and Their Impact on Therapeutical Use. International Journal of Molecular Sciences. 2024; 25(8):4204. https://doi.org/10.3390/ijms25084204
Chicago/Turabian StyleBlebea, Nicoleta Mirela, Andreea Iulia Pricopie, Robert-Alexandru Vlad, and Gabriel Hancu. 2024. "Phytocannabinoids: Exploring Pharmacological Profiles and Their Impact on Therapeutical Use" International Journal of Molecular Sciences 25, no. 8: 4204. https://doi.org/10.3390/ijms25084204
APA StyleBlebea, N. M., Pricopie, A. I., Vlad, R.-A., & Hancu, G. (2024). Phytocannabinoids: Exploring Pharmacological Profiles and Their Impact on Therapeutical Use. International Journal of Molecular Sciences, 25(8), 4204. https://doi.org/10.3390/ijms25084204








