Abstract
Underwater activities are characterized by an imbalance between reactive oxygen/nitrogen species (RONS) and antioxidant mechanisms, which can be associated with an inflammatory response, depending on O2 availability. This review explores the oxidative stress mechanisms and related inflammation status (Oxy-Inflammation) in underwater activities such as breath-hold (BH) diving, Self-Contained Underwater Breathing Apparatus (SCUBA) and Closed-Circuit Rebreather (CCR) diving, and saturation diving. Divers are exposed to hypoxic and hyperoxic conditions, amplified by environmental conditions, hyperbaric pressure, cold water, different types of breathing gases, and air/non-air mixtures. The “diving response”, including physiological adaptation, cardiovascular stress, increased arterial blood pressure, peripheral vasoconstriction, altered blood gas values, and risk of bubble formation during decompression, are reported.
1. Introduction
SCUBA (Self-Contained Underwater Breathing Apparatus) and BH (breath-hold breathing) diving are breathtaking adventures that allow us to explore the world beneath the seas. However, diving comes with its own set of inherent risks and challenges. From time to time, thankfully rarely, SCUBA diving accidents or illnesses may occur. These can range from minor discomfort to life-threatening situations. One essential tool that plays a crucial role in mitigating the consequences of diving-related diseases is oxygen (O2) [1].
Regarding diving-related diseases, time is another vital parameter in diving activities. The inhalation of O2 (normobaric or hyperbaric, and with various types of masks, depending on whether the diver is conscious or unconscious) works after decompression sickness (DCS) by accelerating the diffusion and elimination of the excess nitrogen absorbed during SCUBA diving [2]. Diving-related activities are well known to produce partial O2 pressure (pO2) variations since the ambient pressure changes during underwater excursions; these changes can go from hyperoxia during hyperbaric phases and reach hypoxia on return from depths during some BH diving activities. Prolonged hyperoxia is known during SCUBA diving but does not always reach levels above 1 absolute atmosphere of O2 in the breathing mix [3]. Prolonged hyperoxia (apart from in medical settings) is not easy to reach under physiological conditions. However, it is still possible, for instance, if people are living below sea level around the “Dead Sea” in Israel (located 402 m below sea level, where the barometric pressure reaches 800 mmHg) [4]. The other setting to achieve a prolonged hyperoxia above 1 atmosphere of pure O2 breathing can be reached during Closed-Circuit Rebreather diving since the O2 set point for such dives is commonly set between 1.2 and 1.4 ATA during several hours of the excursion [5,6,7,8,9]. A chronic, less important form of hyperoxia is encountered during other saturation diving procedures where, depending on the methods used, around 0.49 ATA of partial O2 pressure (pO2) levels are kept during several days of decompression needed to reach sea level [10].
This review will discuss the impact of diving activities on oxidative/inflammatory stress (Oxy-Inflammation).
2. Oxy-Inflammation-Related Mechanisms and Outcomes
Humans and most terrestrial animal organisms have adapted to live in the Earth’s atmosphere, where the fraction of inspired oxygen (fiO2) equals approximately 0.21. O2 is the third most abundant element in the universe, after hydrogen (H2) and helium (He) as compounds, including oxides, which constitute around 50% of Earth’s crust [11]. Furthermore, O2 is one of the main components of biological macromolecules (proteins, carbohydrates, nucleic acids) and the last acceptor of electrons in oxidative phosphorylation, providing most of the natural energy in living organisms.
2.1. Oxidative Stress in Hypoxia and Hyperoxia
The redox system plays a crucial role in maintaining cell homeostasis and balancing the generation/elimination of reactive oxygen (ROS) and nitrogen (RNS) species [12]. A continuous flux of oxidant species guarantees redox homeostasis originated by mitochondria to deal with the electron consumption during the electron chain, resulting in the release of superoxide anions (O22−) [13]. Being O2-derived compounds, ROS and RNS are “double-faced” elements because they carry out an essential function in several physiological mechanisms, including pathogen phagocytosis, modulating activities, and regulatory ability during the transduction of intercellular information [14]. On the other hand, an imbalance between ROS/RNS generation and detoxification leads to a condition of “oxidative stress” and the damage of biological macromolecules like lipids, proteins, and DNA [15] (see Figure 1). Following one of the most complete definitions, oxidative stress is “the consequence of the failure to maintain the physiological redox steady state, which is the self-correcting physiological response to different challenges” [16]. ROS such as O22− act both as a signaling molecule, contributing to the release of proinflammatory cytokine [17], and inflammatory mediator, reacting with nitric oxide (NO) to give peroxynitrite (ONOO−), an aggressive RNS that induces nitrosative stress, exacerbating the inflammation status [18]. Furthermore, the overproduction of ROS has been implicated in the development and progression of some diseases: cardiovascular, neurodegenerative, and metabolic diseases and cancer [19]. Elevated ROS levels contribute to stabilizing hypoxia-inducible factor-1α (HIF-1α), a key regulator of cellular responses to low oxygen. Additionally, ROS are essential messenger and signaling molecules in muscle adaptations to exercise and disuse, as documented by various studies [20]. The interplay between hypoxia-induced signaling pathways and oxidative stress is intricated [1,21].
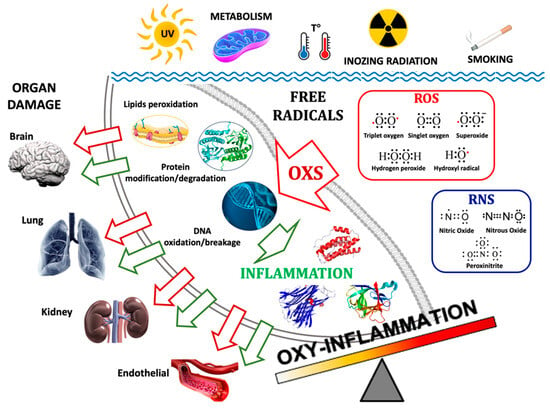
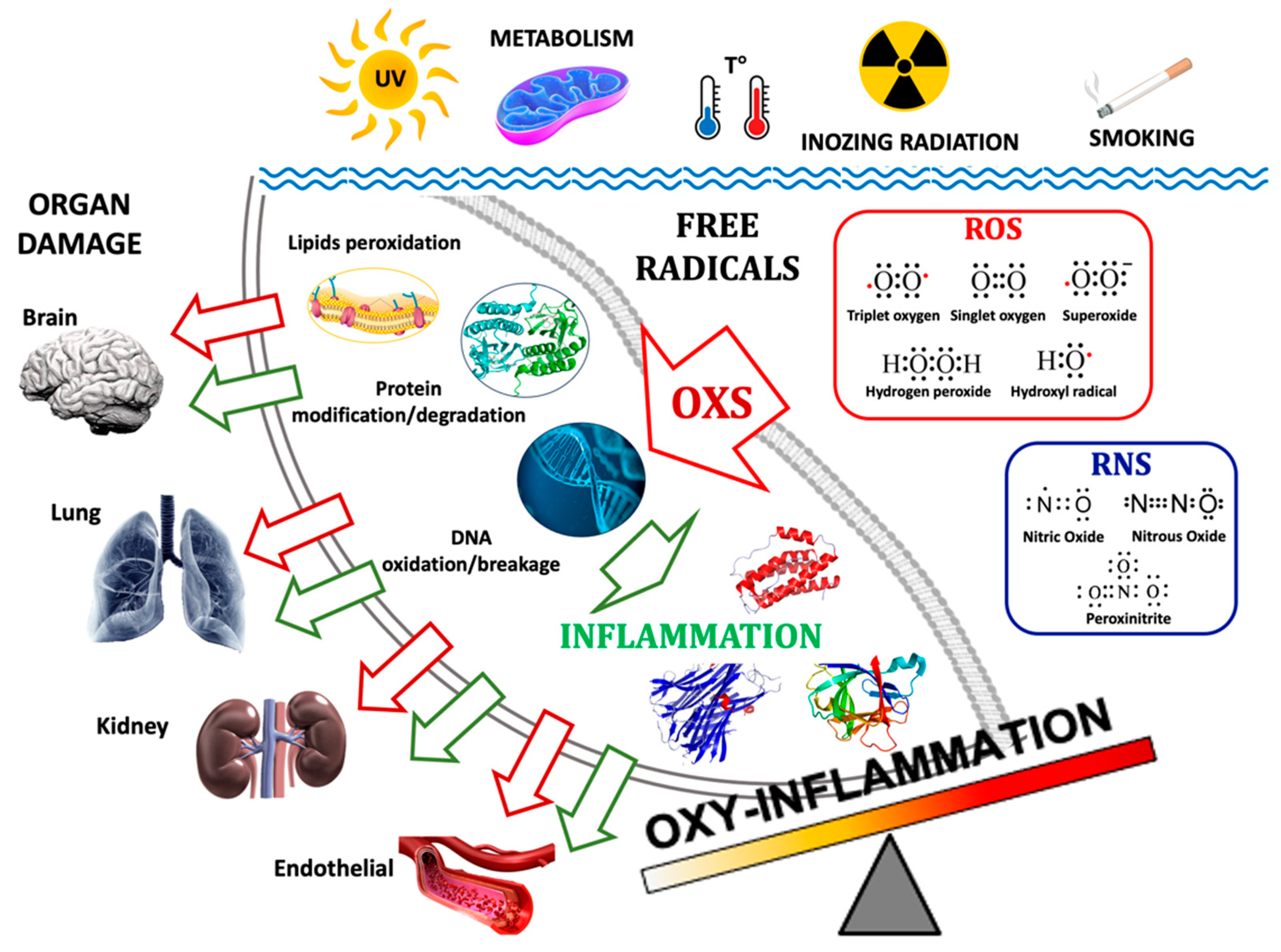
Figure 1.
The main pathways of the formation of Oxy-Inflammation and organ damage. Environmental triggers such as exposure to UV and ionizing radiation, metabolism, water/body temperature, and smoking can produce free radicals: ROS (e.g., 3O2, O2, O2−, H2O2, HO·) and RNS (e.g., NO, N2O, ONOO−). Their accumulations can lead to lipids’ cellular membrane degradation, misfolded proteins, and DNA oxidation/breakage. The inflammation triggered by Oxi-Inflammation is the cause of many types of organ damage and acute/chronic diseases.
On the other hand, an overdose of oxygen can harm cell membranes, lungs, and overall health by increasing oxidative stress and inflammation. For example, it damages the lungs, affecting surfactant proteins and mucociliary clearance, leading to lung collapse, reduced compliance, and higher infection risk [22]. Also, hyperoxemia constricts vessels, reducing coronary blood flow and cardiac output and potentially altering microvascular circulation [23].
2.2. Inflammation in Hypoxia and Hyperoxia
Inflammation may be influenced by fiO2 level, as reported by some authors investigating subjects exposed to hypoxia that showed pulmonary edema caused by inflammatory markers release, including interleukin-6 (IL-6) and protein C-reactive (CRP) [24,25,26]. To adapt body tissues to hypoxia, hypoxia-inducible factors (HIFs), a family of heterodimers that constitute a transcription complex that is stabilized by hypoxia [26] or a return from hyperoxia [27,28,29,30], are activated to coordinate several transcriptional pathways to optimize metabolic and vascular functions toward low fiO2 [31]. Furthermore, HIFs are controlled by prolyl hydroxylase domain (PHD) proteins, a class of “O2 sensors” that are involved in HIF level regulation and O2 homeostasis [32]. Nuclear factor κB (NF-κB) is involved in inflammation regulation and immune response [33], interacting with the PHD-HIF pathway [34], confirming the link between hypoxia and inflammation [35]. Other hypoxia–inflammation interactions involve the IκB kinase complex, an NF-κB regulatory component [31], and the regulation of HIF transcription before and during the inflammation [36]. Under inflammatory status, NF-κB activation stimulates immune defense by neutrophil release [37]. At the same time, HIF-α promotes type 2 helper T-cell (Th2) production [38] and macrophage activation with a ROS-dependent mechanism [39]. Inflammation onset against hypoxia can be clinically significant (e.g., chronic respiratory/renal/kidney/cardiovascular/neurological disease, mitochondrial/endothelial dysfunction, tumors) [21].
Hyperoxia represents the other side of the coin of fiO2 value. In this case, tissues are exposed to an excess supply of O2, with the arterial partial pressure of O2 (paO2) value being greater than 100 mmHg [40]. As mentioned above, exposure to prolonged hyperoxia exacerbates oxidative stress, leading to cellular damage via lipid peroxidation, enzyme inactivation, biological macromolecule oxidation, and apoptosis or necrosis [41]. Exposure to high fiO2 levels during significant time lapse results in O2 lung toxicity, characterized by a pulmonary inflammatory response [42] and a release of proinflammatory cytokines implicated in mediating neutrophil action into hyperoxic lungs [43] in the early stages of inflammatory response [44]. On the other hand, short-term hyperoxia seems to attenuate cytokine production [45], β2-integrin expression necessary for leukocyte adhesion [46], and macrophage phagocytosis and killing [47]. In animal models, some authors found that hyperoxia could mitigate the inflammatory response after glucan intake [48,49].
2.3. Endothelial Dysfunction
Increased partial O2 pressure (pO2) promotes oxidative stress, which triggers endothelial dysfunction. There are two mechanisms strongly related to the decrease in nitric oxide availability (NO). First, hyperoxia-related oxidative stress leads to the generation of the superoxide anion (O2−), which reacts with NO to produce peroxynitrite (ONOO−) [50]. ROS can also oxidize nitric oxide synthase (NOS) cofactor tetrahydrobiopterin (BH4) into dihydrobiopterin (BH2), which causes a reduced liberation of NO uncoupling endothelial NO synthase [51], leading to endothelial dysfunctions [52]. NO can be preserved by the activation of antioxidant defenses, as observed by several authors that found NO increases during the deep phases of both SCUBA and BH dives [53]. The same tendencies have been shown in SCUBA [54] or CCR diving [55]. After exiting saturation diving, endothelial recovery is fast, showing no long-term adverse effect on vascular function [56]. Furthermore, circulating bubbles may induce endothelial dysfunctions [57] through direct contact between microbubbles and endothelial cells [58] and the activation of a coagulation cascade [59,60]. However, we want to highlight that both deep and repetitive BH diving lead to endothelial dysfunction that may play an essential role in the genesis of neurological decompression sickness [61].
2.4. Hypoxia, Reoxygenation, and Hyperoxia
Redox balance/unbalance is significantly modulated by external factors such as environmental hypoxia [62,63,64,65,66,67,68], hyperoxia [69,70,71,72,73], and physical activity [74,75,76,77]. Moreover, both environmental hypoxia and physical activity have been shown to augment oxidative stress in a dose-dependent manner [77,78,79].
Reduced O2 tension during ischemia–reperfusion (IRI) episodes activates cellular pathways that upregulate proinflammatory signaling and promote oxidant generation [80]. Reperfusion after ischemia recruits inflammatory cells to the vascular wall, further exacerbating oxidant production and ultimately resulting in cell death, tissue injury, and organ dysfunction. As an IRI consequence, the activation of the NF-κB pathway leads to the release of proinflammatory cytokines (IL-1, IL-6, IL-8) [81] and major histocompatibility complex (MHC) upregulation [82]. Observing the correlation of the renal expression of Toll-like receptor (TLR) 4 with IRI degree [83], a link between IRI and innate/adaptive immunity has been hypothesized, as reported by some authors in transplant models [84,85].
Physical exercise has been proposed as a model of “intermittent hypoxia” (IH) or “intermittent hyperoxia” since oxidative stress increases during exercise, which can be defined as a combination of cycles of hypoxia and normoxia, hypercapnia, and hyperoxia, depending on the training type and duration [70,86,87]. Physical exercise, mainly moderate activity, improves health by reducing the risk of obesity and cardiovascular and chronic degenerative disease onset [88], raising NO availability, decreasing oxidative stress, and fostering antioxidant defenses [89]. On the other hand, exercise at altitude exposes tissue to hypoxia, which may increase oxidative stress by reducing antioxidant defense efficacy [90].
2.5. Oxy-Inflammation Biomarkers
The simultaneous measurement of a panel of biomarkers has been frequently chosen, as this can also provide a comprehensive assessment of oxidative stress effects on vascular function. Protein biomarkers were selected to cover critical features of vascular function, i.e., inflammation, endothelial function, and fibrinolysis. CRP is a biomarker of inflammation and endothelial function [91]; its increase is associated with impaired endothelium-dependent vasodilatation [92]. IL-6 increases the adhesiveness of the endothelial cells for lymphocytes by upregulating Intercellular Adhesion Molecule-1 (ICAM-1) for a proinflammatory reaction [93].
In this review, we use the term “Oxy-Inflammation”, proposed by Valacchi et al. [94], to describe “a condition characterized by a combination of systemic oxidative stress associated with an inflammatory condition”. This review summarizes the “Oxy-Inflammation” in underwater activities such as breath-hold (BH) and SCUBA diving.
The main mechanisms of the formation of Oxy-Inflammation and relative organ damage are illustrated in Figure 1.
3. The Oxy-Inflammation Mechanism in Underwater Activity
3.1. Breath-Hold (BH) Diving
BH diving elicits a physiological adaptation collectively termed the “diving response”, which includes changes such as bradycardia, reduced cardiac output, increased arterial blood pressure, peripheral vasoconstriction, and altered blood gas values [95]. Hyperoxia occurs during descent and bottom time in BH divers due to increased environmental pressure and thorax compression [96,97]. Hypoxia-induced endothelial damage might be less pronounced because hypoxia appears in the last portion of the ascent phase of a dive [96,97,98,99].
Hyperoxia-induced changes observed in BH diving may influence arterial blood gas (ABG) parameters, especially PaO2 and arterial pCO2 (PaCO2). At the maximum depth, PaO2 increases due to hyperoxia-induced effects caused by the rise in environmental pressure [99,100,101,102], but this trend was not observed in all BH divers. This unexpected PaO2 decrease is related to a lung volume reduction leading to a right-to-left shunt through lung atelectasis [101,102,103,104]. Furthermore, Bosco et al. [105] reported that BH diving-related exercise breaks down PaO2 levels due to muscle O2 consumption [105], which may increase the risk of injuries [106] during the ascent phase. PaCO2 values do not change much, probably due to the body’s capacity to store soluble CO2 [101] and hyperventilation that decreases PaO2 [98].
Similarly to PaO2, PaCO2 variation may be related to BH diving type, as reported by Bosco et al., who found higher PaCO2 values after static apnea concerning those after a deep dive [105], which reduced from initial values. In the case of deep diving, PaCO2 rises in BH divers who do muscular work when returning to the surface using fins. The muscular work contribution may be confirmed by the higher augmented lactate value that was not recorded in other conditions [105].
In BH diving, IH can be a powerful trigger for endothelial dysfunction [106,107]. More severe diving stress in repetitive BH dives could lead to strains on the cardiovascular system, vascular function, and endothelial integrity. Endothelial dysfunctions in BH diving have been investigated by Theunissen et al. [54], where the reduction in NO-dependent vascular function resulted from low NO bioavailability. The same authors also showed a decrease in this endothelial dysfunction when supplementing antioxidants to the divers [108].
High pO2 may also promote oxidative stress due to the hyperbaric environment, increasing free O2 that may generate ROS [109]. Oxidative stress has been investigated in BH diving [54,107,108,109], confirming its role in the development of endothelial dysfunction [110]. The overproduction of ROS and consequent oxidative damage to membrane lipids and total antioxidant capacity (TAC) reflects a hypoxic condition in the last few meters while ascending to the surface [109]. Neopterin and creatinine levels increase, which suggests an alteration in renal function as a physiological response to pO2 variations during the dive [109,110].
Changes in polyunsaturated fatty acids (PUFAs) and eicosanoids after a single apnea event revealed different kinetics of pro- and anti-inflammatory regulations and changes in oxidative stress levels [111].
Adaptation to hypoxia may involve amino acid metabolism [112,113], and hypoxia might be a stimulus for amino acids’ release in the bloodstream because serine and taurine may be engaged in hypoxia adaptation, especially during the ascent phase of a BH dive [113].
Furthermore, prolonged exposure to hypoxia, such as diving at high altitudes, may affect platelet activity, as reported by some authors [114], but this aspect seems to be related to pO2 [115]. Platelet activation can be induced by exercise-related inflammation [116] engaging with leukocytes via soluble factors and physical interaction with several receptors, including platelet P-selectin (CD62P), CD40 ligand (CD40L), as well as PSGL-1, CD40, and Mac-1 (integrin αMβ2, CD11b/CD18) on leukocytes [117,118]. Platelet–leukocyte interactions facilitate leukocyte recruitment and extravasation to inflammation sites, leading to leukocyte proinflammatory mediator release but, in some cases, damping inflammation [119,120]. Furthermore, some authors found a link between gene activity and immune cell activation against inflammation, observing a temporary increase in leucocytes and an upregulation of the anti-inflammatory response genes [121].
As reported by Wang et al. [122], ultrasound lung comets (ULCs) may be associated with lung inflammation and represent a tool to evaluate heart alteration [122], and some mechanical pressure changes in the chest provoked diaphragmatic spasms with a closed glottis (during the struggling phase of breath-holding), leading to lung alveolo-vascular stress [123]. Paganini et al. [101] investigated ULC during a BH dive at increasing depths, observing the onset of lung comets in some subjects; this may have been due to an altered recovery from atelectasis [103] that seemed to have been caused by the alternating closing and opening of bronchioles and alveoli during the dives [102]. As reported by Tojo et al. [124], atelectasis may lead to alveolar hypoxia-induced inflammation, releasing NF-κB-dependent CXCL-1 from epithelial cells and activating HIF-1α toward alveolar hypoxia [124].
Interestingly, cetaceans exhibit physiological adaptations that allow the transition to aquatic life, including a robust antioxidant defense system that prevents injury from repeated exposure to ischemia–reperfusion events associated with BH diving. x. Heme oxygenase (HO) activity increased in dolphin cells but not human cells. On the contrary, TNF-α expression increased in human cells but not in dolphin cells [125].
HO is a cytoprotective protein with anti-inflammatory properties, catalyzing the first step in the oxidative degradation of heme. Various stimuli, including hypoxia, oxidant stress, and inflammatory cytokines, regulate the inducible HO-1 isoform.
Some authors investigated microparticle (MP) release after a BH-diving training protocol, observing that the alterations in endothelial reactivity were coupled to increased levels of Endothelial MPs (EMPs) (CD31+/41−), markers of endothelial apoptosis [126,127]. Intermittent hypoxia could increase cellular stress [61]. An increase in CD31+/annexin+EMP level was observed in some volunteers exposed to normobaric hypoxia in a chamber with oxygen saturation (SaO2) reduced to 73% ± 10% [3]. A similar increase in CD144 EMPs was found in some BH divers who performed single maximal dry BH (SaO2 of 72% ± 12%). Both experiments were conducted in dry conditions but showed that short-term hypoxic situations could trigger endothelial cell stress. Nevertheless, diving stress in BH diving is only partly related to hypoxia [127].
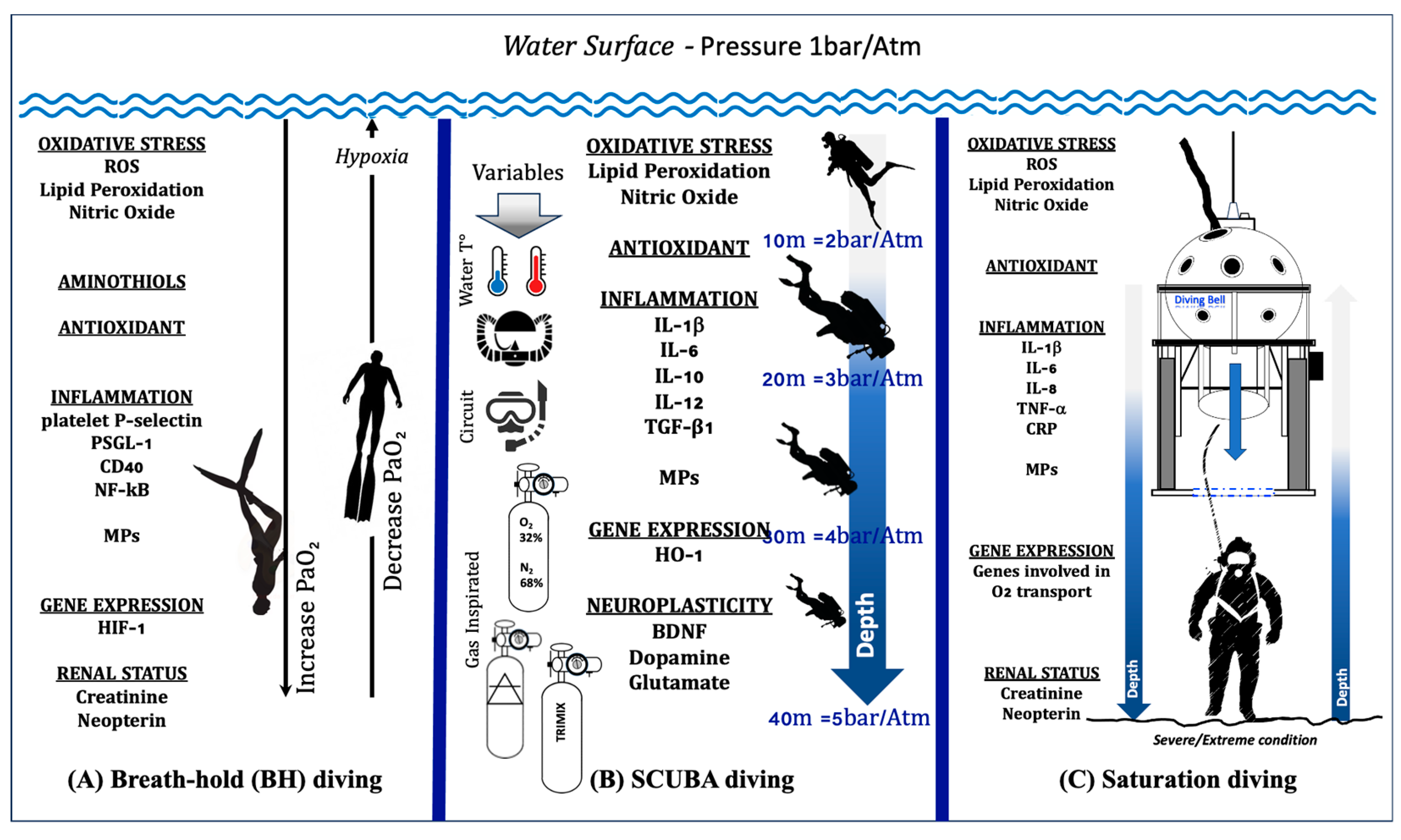
In Figure 2A, we illustrate up–down regulation biomarkers in breath-hold diving.
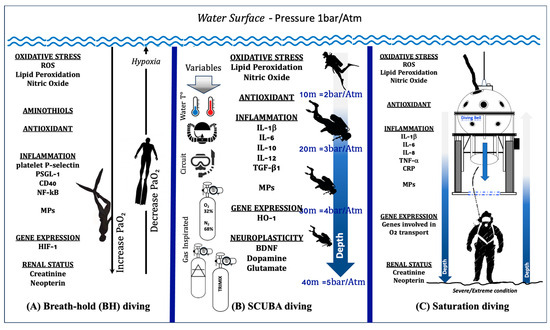
Figure 2.
Illustration of up–down regulation biomarkers in different human underwater activities: (A) Breath-hold diving. The physiological changes in PaO2 during the descent and ascent of a deep breath-hold dive are one of the causes of biological changes at a systemic and molecular level. (B) SCUBA diving. The response in SCUBA diving varies depending on number of dives carried out, depth, water temperature, type of breathing circuit (open vs. CCR), gases inspired, and exercise. (C) Saturation diving. In the sketch, the saturation worksite is shown. The working depth corresponds to the maximum depth reached by the divers, where the “scenario” is considered a severe/extreme condition. All these activities involve changes in levels of Oxy-Inflammation, redox states (aminothiols), gene expression, antioxidants, markers of neuroplasticity, and renal status.
3.2. Self-Contained Underwater Breathing Apparatus (SCUBA) and Closed-Circuit Rebreather (CCR) Diving
One of the most commonly known activities that leads to hyperoxia is SCUBA diving. SCUBA diving exposes the human body to environmental stress represented by increased pO2, physical effort, and elevated breathing resistance, which alter the endothelial function [128,129,130]. SCUBA diving-related physical activity can induce oxidative and cardiovascular stress, amplified by environmental conditions, including hyperoxia, hyperbaric pressure, and cold water [131,132].
The mechanisms that lead to endothelial dysfunction in SCUBA diving may result from increased hyperoxia-induced oxidative stress and peripheral vasoconstriction [129]. The hyperoxia associated with SCUBA diving results in ROS production, which activates the antioxidant defenses, leading to an increased expression of several antioxidant enzymes, such as catalase (CAT) [133], glutathione peroxidase (GPx) [134], and superoxide dismutase (SOD) [135]. As CAT activity rises in lymphocytes within a few hours after the dive, while GPx activity rises after surfacing, ROS production stimulates lymphocyte mobilization because GPx is one of the first antioxidants activated to detoxify ROS [134]. SOD activity increases to protect against ONOO− and to preserve NO generation [135]. Due to the mobilization of endogenous antioxidant systems, the total antioxidant capacity increases after surfacing to help control vascular oxidative stress, activating a signaling cascade that stimulates resistance against diving-related oxidative stress [136].
Oxidative stress may also impair cognitive functions during a dive due to brain-derived neurotrophic factor (BDNF) loss, followed by a decrease in dopamine and glutamate, especially during decompression [137]. This BDNF deficit seems to alter excitatory/inhibitory neurotransmission, including GABA receptors [138], whose interaction with N2 has been proposed as a mechanism of nitrogen narcosis [137,138,139,140,141,142,143].
Pressure changes during a dive or decompression significantly influence the hematocrit component level, including leukocytes and platelets [144,145]. Some authors observed that SCUBA diving did not affect the number of lymphocytes but their functions were affected, while no changes in H2O2 production in lymphocytes and GPx were found. Additionally, CAT activities increased [135]. On the other hand, Morabito et al. [146] showed significantly decreased levels of H2O2 in lymphocytes, probably due to increased CAT activity and endogenous antioxidant enzymes.
Dumić et al. [147] reported that regular recreational SCUBA diving promotes an anti-inflammatory status, thus contributing to cardioprotection and conferring multiple health benefits. Divers who performed five dives, one per week, at a depth of 20–30 m that lasted 30 min, manifested IgG and TPP N-glycosylation alterations toward anti-inflammatory status over the whole study period with an increase in monogalyctosylated and core-fucosylated IgG N-glycans and a decrease in galactosylated TPP N-glycans. On the other hand, military combat swimmers (O2 divers) are regularly exposed to hyperbaric hyperoxia in addition to intensive endurance training intervals and, therefore, are exposed to extreme levels of oxidative stress. Compared to controls, they exhibited a proinflammatory immune status exemplified by an elevated number of CD4+CD25+ T cells, the elevated expression of proinflammatory cytokine IL-12, and the diminished expression of anti-inflammatory TGF-β1 [148]. Supported by decreased basal gene expression and the prolonged upregulation of anti-oxidative HO-1, these data suggest that higher oxidative stress levels, as those present under intermitted hyperbaric hyperoxia conditions, e.g., through oxygen diving, promote a higher inflammatory immune status.
Breathing air in hyperbaric condition leads to the formation of venous gas emboli (VGE) that interact with the endothelium [149], leading to platelet aggregation to the bubble surface [150] and resulting in the formation of microthrombi in lung vessels after decompression in the DCS model [151,152].
In addition to a mechanical explanation for DCS, an inflammatory mechanism has been suggested [153]. In an observational study carried out on a similarly well-matched control group consisting of equally fit non-divers with identical physical training and living conditions before and after a 2-month period of daily diving, no changes in IL-6 and IL-1 receptor antagonist (IL-1ra), but an increase in IL-8 and neutrophil gelatinase-associated lipocalin (NGAL), together with a decrease in secretory leukocyte protease inhibitor (SLPI), were found [154]. The findings suggest inflammatory activation that is not severe because no changes in IL-6 or IL-1ra were found. The increase in NGAL and IL-8 levels was interpreted as a sign of leukocyte activation. The decreased SLPI levels suggest an influence on the inflammatory defense mechanism. Therefore, the compensated activation of the inflammatory defense mechanism without the loss of homeostasis of the inflammatory system was shown. The formation of bubbles during decompression may trigger this inflammatory response. It is also probable that the interplay between pro- and anti-inflammatory substances is a fine-tuned balancing act and that it is only when this balance collapses that morbidity ensues.
Different types of breathing gas are used in SCUBA diving: air and non-air mixtures [155]. The latter contains mainly oxygen, nitrogen, and helium that form nitrox (nitrogen + oxygen), heliox (helium + oxygen), and trimix (oxygen, helium, and nitrogen) with different advantages and features [9,155]. Nitrox is mainly used for shallow recreational dives, heliox for deep diving, and trimix for deep but short dives to avoid neurological side effects. The use of trimix in SCUBA diving has recently become more widespread to reduce nitrogen narcosis compared to air diving [140,141,142].
Some experimental studies, although conflicting, invoked a possible anti-inflammatory role of helium [156]. This gas could increase NO, induce nuclear factor erythroid 2-related factor 2 activation, and cause consequent reductions in systemic inflammation and oxidative stress. Recently, increases in extracellular vesicles and inflammatory status (IL-1) were found in CCR SCUBA divers, highlighting a decrease in plasma gelsolin (pGSN) [157]. It has been demonstrated that pGSN modulates the production of IL-1β-containing microparticles following high-pressure exposure and decompression [158].
Another study [159] found no difference in most of the inflammatory factors (including monocyte chemoattractant protein 1) measured between the trimix and air groups, casting doubt on the protective effect of helium. Following the dive, IL-6 values slightly increased, while IL-8 and epithelial growth factor (EGF) decreased in both groups without significant variation.
To reduce bubble formation and platelet activation, some authors proposed a hyperbaric pretreatment with O2 before the dive, observing a significant decrease in decompression-induced bubbles [160,161] and the activation of antioxidant enzymes such as CAT and SOD [162]. The increased O2 diffuses in micronuclei in substitution of N2, which is eliminated through the lungs and then absorbed, reducing micronuclei formation [163]. Furthermore, the reduction in pN2 and breathing enriched air nitrox (EAN) can reduce platelet activation [164], as well as breathing normobaric O2 before a dive [115]. On the other hand, exposure to EAN mixtures and diving-related physical activity can raise ROS generation [129]; oxidative stress biomarkers including 8-isoprostane and 8-deoxyguanosine; and inflammatory markers such as IL-1β, IL-6, and TNFα [165].
Bosco et al. [165] observed oxidative stress markers’ increase after a dive using close-circuit rebreather (CCR) apparatus, probably originating from increased fiO2, amplified by additional variables such as physical exercise [166] and breathing resistance [167]. On the other hand, mild exercise during a dive may be enough to allow VGE transit, suggesting a potential benefit of exercise before a dive [168].
No association between the decompression model chosen for a 50 msw dive and the likelihood of DCS was established [169]. The dive profiles induced only minor changes in inflammatory markers. The significant increase in IL-6 and decreased IL-8 and EGF levels observed in groups following the ratio decompression strategy (RDS) or compartmental decompression models (CDMs) were within the normal range and were consistent with exercise-induced changes. However, compared to CDM, the RDS-controlled model has the disadvantage of the increased secretion of chemokines in developing vascular damage. This increased secretion of proinflammatory chemokines seems related to the decompression system rather than the more prolonged exposure to high partial pressures of oxygen that RDS divers undergo.
A short-term ketogenic diet has been observed to be effective in weight loss, decreasing inflammation, and protecting against lipid peroxidation during a dive [165].
In Figure 2B, we illustrate up–down regulation biomarkers in SCUBA diving.
3.3. Saturation Diving
A particular case of diving activity is “saturation diving”, which describes a diving method that enables the diver to stay under pressure for a long time (several hours or even days), practiced by military forces and commercial divers. Saturation diving is an established way to conduct subsea operations with human intervention. Saturation divers are exposed to severe/extreme environmental conditions, exposing themselves to higher risks and accidents [170,171,172,173,174,175].
Saturation diving involves divers staying under pressure until their tissues are saturated with breathing gas, exposing them to increased oxygen pressure, potential toxic gases, and the risk of bubble formation during decompression. Prolonged exposure to hyperoxia can generate ROS, damaging cell structures like lipids, proteins, and nucleic acids. All these factors, coupled with vascular gas bubble formation, may lead to endothelial dysfunction. The diver’s antioxidant status, influenced by diet and genetics, plays a crucial role in protecting against injuries caused by these conditions [174].
While working, divers must acclimatize to hyperbaric environments [175,176,177]. In saturation diving, hyperoxia, partial gas pressure changes, and inert gas exchange during decompression are likely sources of excess ROS [178]. Mrakic-Sposta et al. [178] found an increase in oxidative stress-related biomarkers, including 8-deoxyguanosine, 3-nitrotyrosine, and neopterin, due to prolonged exposure to higher pO2 value. Furthermore, saturation divers showed elevated concentrations of IL-6, the first cytokine released into the circulation during inflammation response [10,178,179,180,181,182], and creatinine in the post-dive stage, reflecting the systemic effects induced by hyperoxia, hyperbaric pressure, and exercise at depth [150].
Moreover, Krog et al. [183] demonstrated the increase in Natural Killer (NK) cell cytotoxicity during the compression phase accompanied by alterations in the cytotoxic NK cell subsets (CD32CD16brightCD56dim) in saturation divers’ peripheral blood, with a reduction in glutathione levels [184,185].
Previous studies have concluded that high oxidative stress in divers may be reversed through antioxidant vitamin supplementation [186,187]. In an earlier experiment, the supplementation of vitamin C, α-tocopherol (vitamin E), and tea catechins given to divers during a 400 msw deep saturation dive every day for 40 days prevented hepatic disturbances [188]. A more recent publication demonstrated that divers’ endogenous antioxidant mechanisms counteracted the effects of hyperbaric hyperoxia after 200 msw saturation diving [184]. Sparse data showing the impact of antioxidant vitamins on vascular function in diving are available; however, Deb et al.’s [189] review on antioxidant effects in saturation diving subjects indicates that care is warranted when choosing whether and how to give antioxidant supplements [190].
The iron reserve can also be a valid aid in saturation diving. Data provided by Zwart et al. [179] showed that elevated body iron stores in a hyperoxic environment trigger a DNA damage repair response in peripheral blood mononuclear cells, excluding double-stranded DNA damage. Furthermore, folate levels decrease rapidly in this setting, suggesting higher folate requirements may be necessary when both body iron stores and DNA damage repair responses are elevated. This observation indicates potential interplay between iron status, O2 levels, and folate in cellular responses.
Acclimatization to professional saturation diving was associated with post-saturation gene expression changes to elevated O2 partial pressure by the extensive downregulation of factors involved in O2 transport, including heme, hemoglobin, and erythrocytes. Primary endogenous antioxidants, such as SOD-1, CAT, and glutathione synthetase, were upregulated, and there was increased expression of genes involved in immune activity and inflammatory signaling pathways [186].
The observation that a hyperbaric and hyperoxic environment in saturation diving induces proinflammatory responses that help the body adapt to the inherent oxidative stress in maintaining homeostasis was confirmed recently by Monnoyer et al. [191]. They reported elevated salivary levels of CRP, IL-1β, IL-8, SIgA, and TNF-α during the bottom phase of the hyperbaric saturation, whereas IL-6, cortisol, and alpha-amylase were unchanged. All changes observed during saturation were abolished at the end of decompression. Figure 2C illustrates up–down regulation biomarkers in saturation diving.
4. Conclusions
Several studies encompassed the possibility of mitigating some diving-related impairments; in fact, underwater activities induce oxidative stress and inflammatory-related parameter changes depending on O2 availability. While in SCUBA diving, these changes are related only to hyperoxia onset, in BH diving, they are a consequence of hyperoxia/hypoxia alternation. As described, the presence of ROS, RNS, and inflammation can result in organ dysfunction (e.g., endothelium, lung, kidney, brain) with complex interplay activity [192,193,194,195,196]. In the recreational field, several preconditioning methods have been considered which have challenged previously accepted ideas [197,198,199,200]. One direction was chosen to mitigate diving-related risks: one aiming to endothelial dysfunction, although conduit arteries and microcirculation seem not to behave in parallel [201,202], and supplementation with antioxidant agents before diving [203,204,205,206,207].
Future research should compare and correlate different environmental conditions (e.g., hot/cold water, sea/lake/swimming pool) in different underwater activity scenarios from BH to SCUBA diving. Furthermore, it should also consider the dose-dependent effects of the gas/mixture alongside dive time and depth, associated with decompression times and with the effects of changes in oxygen concentrations. To date, the risk/benefit ratio has not yet been clearly defined.
Author Contributions
Conceptualization, A.V., S.M.-S. and G.B.; methodology, A.V., S.M.-S. and G.B.; writing—original draft preparation, A.V. and A.B.; writing—review and editing, S.M.-S., C.B., E.M.C. and G.B.; visualization, A.B., S.M.-S., C.B., E.M.C. and G.B.; supervision, S.M.-S. and G.B. All authors have read and agreed to the published version of the manuscript.
Funding
This work was primarily supported by the Office of Naval Resource (ONR) Undersea Medicine Program (N00014-23-1-2757).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| AA | Amino acid |
| ABG | Arterial blood gas |
| BH | Breath-hold diving |
| CAT | Catalase |
| CCR | Closed-Circuit Rebreather |
| CDM | Compartmental decompression model |
| CRP | Protein C-reactive |
| DCS | Decompression sickness |
| EGF | Epithelial growth factor |
| HIF | Hypoxia-inducible factor |
| IL- | Interleukin |
| IRI | Ischemia–reperfusion |
| NO | Nitric oxide |
| NK | Natural Killer |
| O2 | Oxygen |
| O22− | Superoxide anion |
| ONOO | Peroxinitrite |
| OxS | Oxidative stress |
| PaCO2 | Arterial partial CO2 pressure |
| pO2 | Partial O2 pressure |
| pGSN | Plasma gelsolin |
| RDS | Ratio decompression strategy |
| ROS | Reactive oxygen species |
| RNS | Reactive nitrogen species |
| SCUBA | Self-Contained Underwater Breathing Apparatus |
| SOD | Superoxide dismutase |
| TAC | Total Antioxidant Capacity |
| 8-OH-dG | 8-OH-2-deoxyguanosine |
| 8-iso-PGF2α | 8-isoprostane |
References
- Wilmshurst, P. ABC of Oxygen. Diving and Oxygen. BMJ 1998, 317, 996–999. [Google Scholar] [CrossRef]
- Lee, Y.I.; Ye, B.J. Underwater and Hyperbaric Medicine as a Branch of Occupational and Environmental Medicine. Ann. Occup. Environ. Med. 2013, 25, 39. [Google Scholar] [CrossRef]
- Balestra, C.; Mrakic-Sposta, S.; Virgili, F. Oxygen Variations—Insights into Hypoxia, Hyperoxia and Hyperbaric Hyperoxia—Is the Dose the Clue? Int. J. Mol. Sci. 2023, 24, 13472. [Google Scholar] [CrossRef]
- Kramer, M.R.; Godfrey, S. Dead Sea: Natural oxygen enrichment at low altitude. Isr. J. Med. Sci. 1996, 32, S20–S23. [Google Scholar]
- Gouin, E.; Balestra, C.; Orsat, J.; Dugrenot, E.; L’Her, E. Pulmonary Effects of One Week of Repeated Recreational Closed-Circuit Rebreather Dives in Cold Water. Medicina 2023, 59, 81. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.R., III; Murphy-Lavoie, H.M. Diving Rebreathers. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024; Bookshelf ID: NBK430743. [Google Scholar]
- Balestra, C.; Guerrero, F.; Theunissen, S.; Germonpre, P.; Lafere, P. Physiology of repeated mixed gas 100-m wreck dives using a closed-circuit rebreather: A field bubble study. Eur. J. Appl. Physiol. 2022, 122, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Tsur, N.; Bar, R.; Hilly, O.; Handzel, O. Balloon Eustachian tuboplasty in a professional Navy SEAL diver: Case report. Undersea Hyperb. Med. 2020, 47, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, S.J.; Doolette, D.J. Recreational technical diving part 1: An introduction to technical diving methods and activities. Diving Hyperb. Med. 2013, 43, 86–93. [Google Scholar] [PubMed]
- Imbert, J.P.; Egi, S.M.; Balestra, C. Vascular Function Recovery Following Saturation Diving. Medicina 2022, 58, 1476. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, R.; Gray, H.B. Preface on making Oxygen. Inorg. Chem. 2008, 47, 1697–1699. [Google Scholar] [CrossRef] [PubMed]
- Kamata, H.; Hirata, H. Redox regulation of cellular signaling. Cell. Signal. 1999, 11, 1–14. [Google Scholar] [CrossRef]
- Rimessi, A.; Previati, M.; Nigro, F.; Wieckowski, M.R.; Pinton, P. Mitochondrial reactive oxygen species and inflammation: Molecular mechanisms, diseases and promising therapies. Int. J. Biochem. Cell Biol. 2016, 81, 281–293. [Google Scholar] [CrossRef]
- Bergendi, L.; Benes, L.; Durackova, Z.; Ferencik, M. Chemistry, physiology and pathology of free radicals. Life Sci. 1999, 65, 1865–1874. [Google Scholar] [CrossRef]
- Caliri, A.W.; Tommasi, S.; Besaratinia, A. Relationships among smoking, oxidative stress, inflammation, macromolecular damage, and cancer. Mutat. Res. Rev. Mutat. Res. 2021, 787, 108365. [Google Scholar] [CrossRef]
- Ursini, F.; Maiorino, M.; Forman, H.J. Redox homeostasis: The Golden Mean of healthy living. Redox Biol. 2016, 8, 205–215. [Google Scholar] [CrossRef]
- Ryter, S.W.; Nakahira, K.; Haspel, J.A.; Choi, A.M. Autophagy in pulmonary diseases. Annu. Rev. Physiol. 2012, 74, 377–401. [Google Scholar] [CrossRef] [PubMed]
- Sunil, V.R.; Shen, J.; Patel-Vayas, K.; Gow, A.J.; Laskin, J.D.; Laskin, D.L. Role of reactive nitrogen species generated via inducible nitric oxide synthase in vesicant-induced lung injury, inflammation and altered lung functioning. Toxicol. Appl. Pharmacol. 2012, 261, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ren, Z.; Zhang, J.; Chuang, C.-C.; Kandaswamy, E.; Zhou, T.; Zou, L. Role of ROS and Nutritional Antioxidants in Human Diseases. Front. Physiol. 2018, 9, 477. [Google Scholar] [CrossRef] [PubMed]
- Debevec, T.; Millet, G.P.; Pialoux, V. Hypoxia-Induced Oxidative Stress Modulation with Physical Activity. Front. Physiol. 2017, 8, 84. [Google Scholar] [CrossRef] [PubMed]
- McGarry, T.; Biniecka, M.; Veale, D.J.; Fearon, U. Hypoxia, oxidative stress and inflammation. Free Radic. Biol. Med. 2018, 125, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.S.; Phuyal, P.; Shah, N. Oxygen Toxicity. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Smit, B.; Smulders, Y.M.; van der Wouden, J.C.; Straaten, H.M.O.-V.; Man, A.M.E.S.-D. Hemodynamic effects of acute hyperoxia: Systematic review and meta-analysis. Crit. Care 2018, 22, 45. [Google Scholar] [CrossRef]
- Hackett, P.H.; Roach, R.C. High-altitude illness. N. Engl. J. Med. 2001, 345, 107–114. [Google Scholar] [CrossRef]
- Hartmann, G.; Tschop, M.; Fischer, R.; Bidlingmaier, C.; Riepl, R.; Tschop, K.; Hautmann, H.; Endres, S.; Toepfer, M. High altitude increases circulating interleukin-6, interleukin-1 receptor antagonist and C-reactive protein. Cytokine 2000, 12, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L.; Nejfelt, M.K.; Chi, S.M.; Antonarakis, S.E. Hypoxia-inducible nuclear factors bind to an enhancer ele-ment located 3′ to the human erythropoietin gene. Proc. Natl. Acad. Sci. USA 1991, 88, 5680–5684. [Google Scholar] [CrossRef]
- Salvagno, M.; Coppalini, G.; Taccone, F.S.; Strapazzon, G.; Mrakic-Sposta, S.; Rocco, M.; Khalife, M.; Balestra, C. The Normobaric Oxygen Paradox-Hyperoxic Hypoxic Paradox: A Novel Expedient Strategy in Hematopoiesis Clinical Issues. Int. J. Mol. Sci. 2022, 24, 82. [Google Scholar] [CrossRef]
- Kiboub, F.Z.; Balestra, C.; Loennechen, O.; Eftedal, I. Hemoglobin and Erythropoietin After Commercial Saturation Diving. Front. Physiol. 2018, 9, 1176. [Google Scholar] [CrossRef]
- Koch, A.; Kähler, W.; Klapa, S.; Grams, B.; van Ooij, P.J.A.M. The conundrum of using hyperoxia in COVID-19 treatment strategies: May intermittent therapeutic hyperoxia play a helpful role in the expression of the surface receptors ACE2 and Furin in lung tissue via triggering of HIF-1α? Intensiv. Care Med. Exp. 2020, 8, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Oxygen sensing, homeostasis, and disease. N. Engl. J. Med. 2011, 365, 537–547. [Google Scholar] [CrossRef]
- Fong, G.H.; Takeda, K. Role and regulation of prolyl hydroxylase domain proteins. Cell Death Differ. 2008, 15, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Naugler, W.E.; Karin, M. NF-kappaB, and cancer-identifying targets and mechanisms. Curr. Opin. Genet. Dev. 2008, 18, 19–26. [Google Scholar] [CrossRef]
- Taylor, C.T. Interdependent roles for Hypoxia-inducible factor and nuclear factor-kappaB in hypoxic inflammation. J. Physiol. 2008, 586, 4055–4059. [Google Scholar] [CrossRef] [PubMed]
- Leveque, C.; Mrakic Sposta, S.; Theunissen, S.; Germonpré, P.; Lambrechts, K.; Vezzoli, A.; Gussoni, M.; Levenez, M.; Lafère, P.; Guerrero, F.; et al. Oxidative Stress Response Kinetics after 60 Minutes at Different Levels (10% or 15%) of Normobaric Hypoxia Exposure. Int. J. Mol. Sci. 2023, 24, 10188. [Google Scholar] [CrossRef]
- Cummins, E.P.; Berra, E.; Comerford, K.M.; Ginouves, A.; Fitzgerald, K.T.; Seeballuck, F.; Godson, C.; Nielsen, J.E.; Moynagh, P.; Pouyssegur, J.; et al. Prolyl hydroxylase-1 negatively regulates IkappaB kinase-beta, giving in-sight into hypoxia-induced NFkappaB activity. Proc. Natl. Acad. Sci. USA 2006, 103, 18154–18159. [Google Scholar] [CrossRef] [PubMed]
- Rius, J.; Guma, M.; Schachtrup, C.; Akassoglou, K.; Zinkernagel, A.S.; Nizet, V.; Johnson, R.S.; Haddad, G.G.; Karin, M. NF-kappaB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1alpha. Nature 2008, 453, 807–811. [Google Scholar] [CrossRef] [PubMed]
- McDonald, P.P.; Bald, A.; Cassatella, M.A. Activation of the NF-κB Pathway by Inflammatory Stimuli in Human Neutrophils. Blood 1997, 89, 3421–3433. [Google Scholar] [CrossRef]
- Ben-Shoshan, J.; Afek, A.; Maysel-Auslender, S.; Barzelay, A.; Rubinstein, A.; Keren, G.; George, J. HIF-1alpha overex-pression and experimental murine atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 665–670. [Google Scholar] [CrossRef]
- Alonso, D.; Serrano, E.; Bermejo, F.J.; Corral, R.S. HIF-1alpha-regulated MIF activation and Nox2-dependent ROS generation promote Leishmania amazonensis killing by macrophages under Hypoxia. Cell Immunol. 2019, 335, 15–21. [Google Scholar] [CrossRef]
- Damiani, E.; Donati, A.; Girardis, M. Oxygen in the critically ill: Friend or foe? Curr. Opin. Anaesthesiol. 2018, 31, 129–135. [Google Scholar] [CrossRef]
- Jamieson, D.; Chance, B.; Cadenas, E.; Boveris, A. The relation of free radical production to hyperoxia. Annu. Rev. Physiol. 1986, 48, 703–719. [Google Scholar] [CrossRef]
- Jackson, R.M. Pulmonary oxygen toxicity. Chest 1985, 88, 900–905. [Google Scholar] [CrossRef]
- Bhandari, V. Molecular mechanisms of hyperoxia-induced acute lung injury. Front. Biosci. 2008, 13, 6653–6661. [Google Scholar] [CrossRef]
- Li, L.F.; Yang, C.T.; Huang, C.C.; Liu, Y.Y.; Kao, K.C.; Lin, H.C. Low-molecular-weight heparin reduces hyperoxia-augmented ventilator-induced lung injury via serine/threonine kinase-protein kinase B. Respir. Res. 2011, 12, 90. [Google Scholar] [CrossRef] [PubMed]
- Qadan, M.; Battista, C.; Gardner, S.A.; Anderson, G.; Akca, O.; Polk, H.C., Jr. Oxygen and surgical site infection: A study of underlying immunologic mechanisms. Anesthesiology 2010, 113, 369–377. [Google Scholar] [CrossRef]
- McInturff, A.M.; Cody, M.J.; Elliott, E.A.; Glenn, J.W.; Rowley, J.W.; Rondina, M.T.; Yost, C.C. Mammalian target of rapamycin regulates neutrophil extracellular trap formation via induction of hypoxia-inducible factor 1 alpha. Blood 2012, 120, 3118–3125. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, P.J.; Hickman-Davis, J.M.; Davis, I.C.; Matalon, S. Hyperoxia impairs antibacterial function of macrophages through effects on actin. Am. J. Respir. Cell Mol. Biol. 2003, 28, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Xie, K.; Li, N.; Qin, M.; Lu, Y.; Ma, S.; Ji, G.; Xiong, L. 100% oxygen inhalation protects against zymosan-induced sterile sepsis in mice: The roles of inflammatory cytokines and antioxidant enzymes. Shock 2009, 32, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Bai, X.; Du, K.; Huang, Y.; Wang, W.; Zhao, Y.; Pei, Y.; Mu, J.; Han, H.; Hu, S.; et al. Activation of the cholinergic anti-inflammatory pathway contributes to the protective effects of 100% oxygen inhalation on zymosan-induced generalized inflammation in mice. J. Surg. Res. 2012, 174, e75–e83. [Google Scholar] [CrossRef] [PubMed]
- Sureda, A.; Ferrer, M.D.; Batle, J.M.; Tauler, P.; Tur, J.A.; Pons, A. Scuba diving increases erythrocyte and plasma antioxidant defenses and spares NO without oxidative damage. Med. Sci. Sport. Exerc. 2009, 41, 1271–1276. [Google Scholar] [CrossRef]
- Forstermann, U. Nitric oxide and oxidative stress in vascular disease. Pflug. Arch. 2010, 459, 923–939. [Google Scholar] [CrossRef]
- Landmesser, U.; Dikalov, S.; Price, S.R.; McCann, L.; Fukai, T.; Holland, S.M.; Mitch, W.E.; Harrison, D.G. Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J. Clin. Investig. 2003, 111, 1201–1209. [Google Scholar] [CrossRef]
- Solich-Talanda, M.; Żebrowska, A.; Mikołajczyk, R.; Kostorz-Nosal, S.; Ziora, D.; Jastrzębski, D.; Siermontowski, P. Effect of Apnea-Induced Hypoxia on Cardiovascular Adaptation and Circulating Biomarkers of Oxidative Stress in Elite Breath-Hold Divers. Front. Physiol. 2021, 12, 726434. [Google Scholar] [CrossRef]
- Theunissen, S.; Guerrero, F.; Sponsiello, N.; Cialoni, D.; Pieri, M.; Germonpre, P.; Obeid, G.; Tillmans, F.; Papadopoulou, V.; Hemelryck, W.; et al. Nitric oxide-related endothelial changes in breath-hold and scuba divers. Undersea Hyperb. Med. 2013, 40, 135–144. [Google Scholar]
- Arya, A.K.; Balestra, C.; Bhopale, V.M.; Tuominen, L.J.; Raisanen-Sokolowski, A.; Dugrenot, E.; L’Her, E.; Bhat, A.R.; Thom, S.R. Elevations of Extracellular Vesicles and Inflammatory Biomarkers in Closed Circuit SCUBA Divers. Int. J. Mol. Sci. 2023, 24, 5969. [Google Scholar] [CrossRef]
- Sharma, R.I.; Marcinkowska, A.B.; Mankowska, N.D.; Waśkow, M.; Kot, J.; Winklewski, P.J. Cognitive Functions in Scuba, Technical and Saturation Diving. Biology 2023, 12, 229. [Google Scholar] [CrossRef]
- Nossum, V.; Hjelde, A.; Brubakk, A.O. Small amounts of venous gas embolism cause delayed impairment of endothelial function and increase polymorphonuclear neutrophil infiltration. Eur. J. Appl. Physiol. 2002, 86, 209–214. [Google Scholar] [CrossRef]
- Klinger, A.L.; Pichette, B.; Sobolewski, P.; Eckmann, D.M. Mechanotransductional basis of endothelial cell response to intravascular bubbles. Integr. Biol. 2011, 3, 1033–1042. [Google Scholar] [CrossRef]
- Pontier, J.M.; Gempp, E.; Ignatescu, M. Blood platelet-derived microparticles release and bubble formation after an open-sea air dive. Appl. Physiol. Nutr. Metab. 2012, 37, 888–892. [Google Scholar] [CrossRef]
- Brett, K.D.; Nugent, N.Z.; Fraser, N.K.; Bhopale, V.M.; Yang, M.; Thom, S.R. Microparticle and interleukin-1β production with human simulated compressed air diving. Sci. Rep. 2019, 9, 13320. [Google Scholar] [CrossRef] [PubMed]
- Barak, O.F.; Janjic, N.; Drvis, I.; Mijacika, T.; Mudnic, I.; Coombs, G.B.; Thom, S.R.; Madic, D.; Dujic, Z. Vascular dysfunction following breath-hold diving. Can. J. Physiol. Pharmacol. 2020, 98, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, J.; Ascensao, A.; Viscor, G.; Soares, J.; Oliveira, J.; Marques, F.; Duarte, J. Oxidative stress in humans during and after 4 hours of hypoxia at a simulated altitude of 5500 m. Aviat. Space Environ. Med. 2004, 75, 16–22. [Google Scholar] [PubMed]
- Irarrázaval, S.; Allard, C.; Campodónico, J.; Pérez, D.; Strobel, P.; Vásquez, L.; Urquiaga, I.; Echeverría, G.; Leighton, F. Oxidative Stress in Acute Hypobaric Hypoxia. High Alt. Med. Biol. 2017, 18, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Mrakic-Sposta, S.; Gussoni, M.; Dellanoce, C.; Marzorati, M.; Montorsi, M.; Rasica, L.; Pratali, L.; D’Angelo, G.; Martinelli, M.; Bastiani, L.; et al. Effects of acute and sub-acute hypobaric hypoxia on oxidative stress: A field study in the Alps. Eur. J. Appl. Physiol. 2021, 121, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, J.; Liu, G.; Wu, G.; Wang, R.; Zhang, J. High altitude hypoxia and oxidative stress: The new hope brought by free radical scavengers. Life Sci. 2023, 336, 122319. [Google Scholar] [CrossRef] [PubMed]
- Burtscher, J.; Mallet, R.T.; Pialoux, V.; Millet, G.P.; Burtscher, M. Adaptive Responses to Hypoxia and/or Hyperoxia in Humans. Antioxid. Redox Signal. 2022, 37, 887–912. [Google Scholar] [CrossRef] [PubMed]
- Mrakic Sposta, S.; Montorsi, M.; Porcelli, S.; Marzorati, M.; Healey, B.; Dellanoce, C.; Vezzoli, A. Effects of Prolonged Exposure to Hypobaric Hypoxia on Oxidative Stress: Overwintering in Antarctic Concordia Station. Oxid. Med. Cell Longev. 2022, 2022, 4430032. [Google Scholar] [CrossRef]
- Rathor, R.; Suryakumar, G.; Singh, S.N. Diet and redox state in maintaining skeletal muscle health and performance at high altitude. Free. Radic. Biol. Med. 2021, 174, 305–320. [Google Scholar] [CrossRef]
- Verratti, V.; Bondi, D.; Jandova, T.; Camporesi, E.; Paoli, A.; Bosco, G. Sex Hormones Response to Physical Hyperoxic and Hyperbaric Stress in Male Scuba Divers: A Pilot Study. Adv. Exp. Med. Biol. 2019, 1176, 53–62. [Google Scholar] [CrossRef]
- Balestra, C.; Lambrechts, K.; Mrakic-Sposta, S.; Vezzoli, A.; Levenez, M.; Germonpré, P.; Virgili, F.; Bosco, G.; Lafère, P. Hypoxic and Hyperoxic Breathing as a Complement to Low-Intensity Physical Exercise Programs: A Proof-of-Principle Study. Int. J. Mol. Sci. 2021, 22, 9600. [Google Scholar] [CrossRef]
- Leveque, C.; Mrakic-Sposta, S.; Theunissen, S.; Germonpr, P.; Lambrechts, K.; Vezzoli, A.; Bosco, G.; Lévenéz, M.; Lafèvre, P.; Guerrero, F.; et al. Oxidative Stress Response Kinetics after 60 Minutes at Different (1.4 ATA and 2.5 ATA) Hyperbaric Hyperoxia Exposures. Int. J. Mol. Sci. 2023, 24, 12361. [Google Scholar] [CrossRef] [PubMed]
- Brugniaux, J.V.; Coombs, G.B.; Barak, O.F.; Dujic, Z.; Sekhon, M.S.; Ainslie, P.N. Highs and lows of hyperoxia: Physiological, performance, and clinical aspects. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R1–R27. [Google Scholar] [CrossRef] [PubMed]
- Modun, D.; Krnic, M.; Vukovic, J.; Kokic, V.; Kukoc-Modun, L.; Tsikas, D.; Dujic, Z. Plasma nitrite concentration decreases after hyperoxia-induced oxidative stress in healthy humans. Clin. Physiol. Funct. Imaging 2012, 32, 404–408. [Google Scholar] [CrossRef]
- Thom, S.R.; Bhopale, V.; Fisher, D.; Manevich, Y.; Huang, P.L.; Buerk, D.G. Stimulation of nitric oxide synthase in cerebral cortex due to elevated partial pressures of oxygen: An oxidative stress response. J. Neurobiol. 2002, 51, 85–100. [Google Scholar] [CrossRef]
- Mrakic-Sposta, S.; Gussoni, M.; Porcelli, S.; Pugliese, L.; Pavei, G.; Bellistri, G.; Montorsi, M.; Tacchini, P.; Vezzoli, A. Training effects on ROS production determined by electron paramagnetic resonance in master swimmers. Oxid. Med. Cell. Longev. 2015, 2015, 804794. [Google Scholar] [CrossRef] [PubMed]
- Thirupathi, A.; Pinho, R.A.; Ugbolue, U.C.; He, Y.; Meng, Y.; Gu, Y. Effect of Running Exercise on Oxidative Stress Biomarkers: A Systematic Review. Front. Physiol. 2021, 11, 610112. [Google Scholar] [CrossRef]
- Brizzolari, A.; Bosco, G.; Vezzoli, A.; Dellanoce, C.; Barassi, A.; Paganini, M.; Cialoni, D.; Mrakic-Sposta, S. Seasonal Oxy-Inflammation and Hydration Status in Non-Elite Freeskiing Racer: A Pilot Study by Non-Invasive Analytic Method. Int. J. Environ. Res. Public Health 2023, 20, 3157. [Google Scholar] [CrossRef]
- Goto, C.; Higashi, Y.; Kimura, M.; Noma, K.; Hara, K.; Nakagawa, K.; Kawamura, M.; Chayama, K.; Yoshizumi, M.; Nara, I. Effect of different intensities of exercise on endothelium-dependent vasodilation in humans: Role of endothelium-dependent nitric oxide and oxidative stress. Circulation 2003, 108, 530–535. [Google Scholar] [CrossRef]
- Vezzoli, A.; Dellanoce, C.; Mrakic-Sposta, S.; Montorsi, M.; Moretti, S.; Tonini, A.; Pratali, L.; Accinni, R. Oxidative Stress Assessment in Response to Ultraendurance Exercise: Thiols Redox Status and ROS Production according to Duration of a Competitive Race. Oxid. Med. Cell. Longev. 2016, 2016, 6439037. [Google Scholar] [CrossRef]
- Granger, D.N.; Kvietysb, P.R. Reperfusion injury, and reactive oxygen species: The evolution of a concept. Redox Biol. 2015, 6, 524–551. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Y.; Nunez, G. Sterile inflammation: Sensing and reacting to damage. Nat. Rev. Immunol. 2010, 10, 826–837. [Google Scholar] [CrossRef]
- Mills, K.H. TLR-dependent T cell activation in autoimmunity. Nat. Rev. Immunol. 2011, 11, 807–822. [Google Scholar] [CrossRef] [PubMed]
- Kruger, B.; Krick, S.; Dhillon, N.; Lerner, S.M.; Ames, S.; Bromberg, J.S.; Lin, M.; Walsh, L.; Vella, J.; Fischereder, M.; et al. Donor Toll-like receptor 4 contributes to ischemia and reperfusion injury following human kidney transplantation. Proc. Natl. Acad. Sci. USA 2009, 106, 3390–3395. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yin, H.; Han, J.; Huang, B.; Xu, J.; Zheng, F.; Tan, Z.; Fang, M.; Rui, L.; Chen, D.; et al. Extracellular hmgb1 functions as an innate immune mediator implicated in murine cardiac allograft acute rejection. Am. J. Transplant. 2007, 7, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Moser, B.; Szabolcs, M.J.; Ankersmit, H.J.; Lu, Y.; Qu, W.; Weinberg, A.; Herold, K.C.; Schmidt, A.M. Blockade of RAGE suppresses alloimmune reactions in vitro and delays allograft rejection in murine heart transplantation. Am. J. Transplant. 2007, 7, 293–302. [Google Scholar] [CrossRef]
- Rybnikova, E.A.; Nalivaeva, N.N.; Zenko, M.Y.; Baranova, K.A. Intermittent Hypoxic Training as an Effective Tool for Increasing the Adaptive Potential, Endurance and Working Capacity of the Brain. Front. Neurosci. 2022, 16, 941740. [Google Scholar] [CrossRef] [PubMed]
- Lakka, T.A.; Venalainen, J.M.; Rauramaa, R.; Salonen, R.; Tuomilehto, J.; Salonen, J.T. Relation of leisure-time physical activity and cardiorespiratory fitness to the risk of acute myocardial infarction. N. Engl. J. Med. 1994, 330, 1549–1554. [Google Scholar] [CrossRef] [PubMed]
- Briones, A.M.; Touyz, R.M. Moderate exercise decreases inflammation and oxidative stress in hypertension: But what are the mechanisms? Hypertension 2009, 54, 1206–1208. [Google Scholar] [CrossRef] [PubMed]
- Moller, P.; Loft, S.; Lundby, C.; Olsen, N.V. Acute hypoxia and hypoxic exercise induce DNA strand breaks and oxidative DNA damage in humans. FASEB J. 2001, 15, 1181–1186. [Google Scholar] [CrossRef]
- Wozniak, A.; Drewa, G.; Chesy, G.; Rakowski, A.; Rozwodowska, M.; Olszewska, D. Effect of altitude training on sportsmen’s peroxidation and antioxidant enzymes. Med. Sci. Sport. Exerc. 2001, 33, 1109–1113. [Google Scholar] [CrossRef]
- Ridker, P.M.; Cushman, M.; Stampfer, M.J.; Tracy, R.P.; Hennekens, C.H. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N. Engl. J. Med. 1997, 336, 973–979. [Google Scholar] [CrossRef]
- Verma, S.; Buchanan, M.R.; Anderson, T.J. Endothelial function testing as a biomarker of vascular disease. Circulation 2003, 108, 2054–2059. [Google Scholar] [CrossRef]
- Watson, C.; Whittaker, S.; Smith, N.; Vora, A.J.; Dumonde, D.C.; Brown, K.A. IL-6 acts on endothelial cells to preferentially increase their lymphocyte adherence. Clin. Exp. Immunol. 1996, 105, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Valacchi, G.; Virgili, F.; Cervellati, C.; Pecorelli, A. OxInflammation: From Subclinical Condition to Pathological Biomarker. Front. Physiol. 2018, 9, 858. [Google Scholar] [CrossRef] [PubMed]
- Heusser, K.; Dzamonja, G.; Tank, J.; Palada, I.; Valic, Z.; Bakovic, D.; Obad, A.; Ivancev, V.; Breskovic, T.; Diedrich, A.; et al. Cardiovascular regulation during apnea in elite divers. Hypertension 2009, 53, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Bosco, G.; Rizzato, A.; Moon, R.E.; Camporesi, E.M. Environmental Physiology and Diving Medicine. Front. Psychol. 2018, 9, 72. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, G. Extreme human breath-hold diving. Eur. J. Appl. Physiol. 2001, 84, 254–271. [Google Scholar] [CrossRef] [PubMed]
- Muth, C.M.; Radermacher, P.; Pittner, A.; Steinacker, J.; Schabana, R.; Hamich, S.; Paulat, K.; Calzia, E. Arterial blood gases during diving in elite apnea divers. Int. J. Sport. Med. 2003, 24, 104–107. [Google Scholar] [CrossRef]
- Garbella, E.; Piarulli, A.; Fornai, E.; Pingitore, A.; Prediletto, R. Preliminary observations on the effect of hypoxic and hyperbaric stress on pulmonary gas exchange in breath-hold divers. Diving Hyperb. Med. 2011, 41, 97–100. [Google Scholar]
- Bosco, G.; Rizzato, A.; Martani, L.; Schiavo, S.; Talamonti, E.; Garetto, G.; Paganini, M.; Camporesi, E.M.; Moon, R.E. Arterial Blood Gas Analysis in Breath-Hold Divers at Depth. Front. Physiol. 2018, 9, 1558. [Google Scholar] [CrossRef]
- Paganini, M.; Moon, R.E.; Giacon, T.A.; Cialoni, D.; Martani, L.; Zucchi, L.; Garetto, G.; Talamonti, E.; Camporesi, E.M.; Bosco, G. Relative hypoxemia at depth during breath-hold diving investigated through arterial blood gas analysis and lung ultrasound. J. Appl. Physiol. 2023, 135, 863–871. [Google Scholar] [CrossRef]
- Barković, I.; Jurilj, Z.; Marinelli, F.; Maričić, V.; Pavlović, M.; Wensveen, T.T.; Peršić, V. Arterial blood gases’ analysis in elite breath-hold divers at extreme depths. Eur. J. Appl Physiol. 2023, 123, 857–865. [Google Scholar] [CrossRef]
- Scott, T.; van Waart, H.; Vrijdag, X.C.E.; Mullins, D.; Mesley, P.; Mitchell, S.J. Arterial blood gas measurements during deep open-water breath-hold dives. J. Appl. Physiol. 2021, 130, 1490–1495. [Google Scholar] [CrossRef]
- Fitz-Clarke, J.R. Mechanics of airway and alveolar collapse in human breath-hold diving. Respir. Physiol. Neurobiol. 2007, 159, 202–210. [Google Scholar] [CrossRef]
- Bosco, G.; Paganini, M.; Rizzato, A.; Martani, L.; Garetto, G.; Lion, J.; Camporesi, E.M.; Moon, R.E. Arterial blood gases in divers at the surface after prolonged breath-hold. Eur. J. Appl. Physiol. 2020, 120, 505–512. [Google Scholar] [CrossRef]
- Patrician, A.; Dujić, Z.; Spajić, B.; Drviš, I.; Ainslie, P.N. Breath-Hold Diving—The Physiology of Diving Deep and Returning. Front. Physiol. 2021, 12, 639377. [Google Scholar] [CrossRef]
- Barlow, M.J.; Elia, A.; Shannon, O.M.; Zacharogianni, A.; Lodin-Sundstrom, A. The Effect of a Dietary Nitrate Supplementation in the Form of a Single Shot of Beetroot Juice on Static and Dynamic Apnea Performance. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Theunissen, S.; Schumacker, J.; Guerrero, F.; Tillmans, F.; Boutros, A.; Lambrechts, K.; Mazur, A.; Pieri, M.; Germonpre, P.; Balestra, C. Dark chocolate reduces endothelial dysfunction after successive breath-hold dives in cool water. Eur. J. Appl. Physiol. 2013, 113, 2967–2975. [Google Scholar] [CrossRef] [PubMed]
- Mrakic-Sposta, S.; Vezzoli, A.; Rizzato, A.; Della Noce, C.; Malacrida, S.; Montorsi, M.; Paganini, M.; Cancellara, P.; Bosco, G. Oxidative stress assessment in breath-hold diving. Eur. J. Appl. Physiol. 2019, 119, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Harrison, D.G. Endothelial dysfunction in cardiovascular diseases: The role of oxidant stress. Circ. Res. 2000, 87, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Dolscheid-Pommerich, R.C.; Weikert, J.; Reinicke, M.; Fimmers, R.; Stoffel-Wagner, B.; Erdfelder, F.; Ceglarek, U.; Eichhorn, L. Changes in PUFA and eicosanoid metabolism during/after apnea diving: A prospective single-center study. Undersea Hyperb. Med. 2020, 47, 539–549. [Google Scholar] [CrossRef]
- Data, P.G.; Di Tano, G.; Arborelius, M., Jr.; Polidoro, G.; Arduini, A. Change in plasma amino acid concentrations during breath-hold diving at high altitude. Clin. Physiol. Biochem. 1988, 6, 327–333. [Google Scholar]
- Cialoni, D.; Brizzolari, A.; Sponsiello, N.; Lancellotti, V.; Bosco, G.; Marroni, A.; Barassi, A. Serum Amino Acid Profile Changes After Repetitive Breath-Hold Dives: A Preliminary Study. Sport. Med. Open 2022, 8, 80. [Google Scholar] [CrossRef]
- Zaccaria, M.; Borea, P.A.; Opocher, G.; Ponchia, A.; Varani, K.; Fraccarollo, D.; Scandellari, C. Effects of high-altitude chronic Hypoxia on platelet alpha 2-receptors in man. Eur. J. Clin. Investig. 1997, 27, 316–321. [Google Scholar] [CrossRef]
- Bosco, G.; Yang, Z.J.; Savini, F.; Nubile, G.; Data, P.G.; Wang, J.P.; Camporesi, E.M. Environmental stress on diving-induced platelet activation. Undersea Hyperb. Med. 2001, 28, 207–211. [Google Scholar]
- Goebel, M.U.; Mills, P.J.; Irwin, M.R.; Ziegler, M.G. Interleukin-6 and tumor necrosis factor-alpha production after acute psychological stress, exercise, and infused isoproterenol: Differential effects and pathways. Psychosom. Med. 2000, 62, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Kurose, I.; Anderson, D.C.; Miyasaka, M.; Tamatani, T.; Paulson, J.C.; Todd, R.F.; Rusche, J.R.; Granger, D.N. Molecular determinants of reperfusion-induced leukocyte adhesion and vascular protein leakage. Circ. Res. 1994, 74, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Lievens, D.; Zernecke, A.; Seijkens, T.; Soehnlein, O.; Beckers, L.; Munnix, I.C.; Wijnands, E.; Goossens, P.; van Kruchten, R.; Thevissen, L.; et al. Platelet CD40L mediates thrombotic and inflammatory processes in atherosclerosis. Blood 2010, 116, 4317–4327. [Google Scholar] [CrossRef] [PubMed]
- Ando, Y.; Oku, T.; Tsuji, T. Platelets attenuate production of cytokines and nitric oxide by macrophages in response to bacterial endotoxin. Platelets 2016, 27, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Gudbrandsdottir, S.; Hasselbalch, H.C.; Nielsen, C.H. Activated platelets enhance IL-10 secretion and reduce TNF-alpha secretion by monocytes. J. Immunol. 2013, 191, 4059–4067. [Google Scholar] [CrossRef] [PubMed]
- Eftedal, I.; Flatberg, A.; Davis, I.; Dujic, Z. Immune and inflammatory responses to freediving calculated from leukocyte gene expression profiles. Physiol. Genom. 2016, 48, 795–802. [Google Scholar] [CrossRef]
- Wang, Z.L.; Liu, J.Y.; Zhou, C.J.; Wang, M.B.; Wang, H.Y.; Xu, Y. Risk factors and impacts on prognosis of ultrasound lung comets in patients undergoing hemodialysis. Zhonghua Yi Xue Za Zhi 2017, 97, 3796–3801. [Google Scholar] [CrossRef]
- Boussuges, A.; Coulange, M.; Bessereau, J.; Gargne, O.; Ayme, K.; Gavarry, O.; Fontanari, P.; Joulia, F. Ultrasound lung comets induced by repeated breath-hold diving, a study in underwater fishermen. Scand. J. Med. Sci. Sports 2011, 21, e384–e392. [Google Scholar] [CrossRef] [PubMed]
- Tojo, K.; Nagamine, Y.; Yazawa, T.; Mihara, T.; Baba, Y.; Ota, S.; Goto, T.; Kurahashi, K. Atelectasis causes alveolar hypoxia-induced inflammation during uneven mechanical ventilation in rats. Intensive Care Med. Exp. 2015, 3, 56. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Ramos, C.A.; Ramírez-Jirano, L.J.; Bitzer-Quintero, O.K.; Vázquez-Medina, J.P.; Gaxiola-Robles, R.; Zenteno-Savín, T. Dolphin leukocytes exhibit an attenuated cytokine response and increase heme oxygenase activity upon exposure to lipopolysaccharides. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2023, 281, 111438. [Google Scholar] [CrossRef] [PubMed]
- Eichhorn, L.; Dolscheid-Pommerich, R.; Erdfelder, F.; Ayub, M.A.; Schmitz, T.; Werner, N.; Jansen, F. Sustained apnea induces endothelial activation. Clin. Cardiol. 2017, 40, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Lichtenauer, M.; Goebel, B.; Fritzenwanger, M.; Foster, M.; Betge, S.; Laute, A.; Figulla, H.-R.; Jung, C. Simulated temporary hypoxia triggers the release of CD31+/Annexin+ endothelial microparticles: A prospective pilot study in humans. Clin. Hemorheol. Microcirc. 2015, 61, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Brubakk, A.O.; Duplancic, D.; Valic, Z.; Palada, I.; Obad, A.; Bakovic, D.; Wisloff, U.; Dujic, Z. A single air dive reduces arterial endothelial function in man. J. Physiol. 2005, 566, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Obad, A.; Marinovic, J.; Ljubkovic, M.; Breskovic, T.; Modun, D.; Boban, M.; Dujic, Z. Successive deep dives impair endothelial function and enhance oxidative stress in man. Clin. Physiol. Funct. Imaging 2010, 30, 432–438. [Google Scholar] [CrossRef]
- Perovic, A.; Nikolac, N.; Braticevic, M.N.; Milcic, A.; Sobocanec, S.; Balog, T.; Dabelic, S.; Dumic, J. Does recreational scuba diving have clinically significant effect on routine haematological parameters? Biochem. Med. 2017, 27, 325–331. [Google Scholar] [CrossRef]
- Sureda, A.; Batle, J.M.; Ferrer, M.D.; Cases, N.; Aguiló, A.; Pons, A. Neutrophil tolerance to oxidative stress induced by hypoxia/reoxygenation. Free Radic. Res. 2004, 38, 1003–1009. [Google Scholar] [CrossRef]
- Bosco, G.; Yang, Z.J.; Di Tano, G.; Camporesi, E.M.; Faralli, F.; Savini, F.; Landolfi, A.; Doria, C.; Fano, G. Effect of in-water Oxygen prebreathing at different depths on decompression-induced bubble formation and platelet activation. J. Appl. Physiol. 2010, 108, 1077–1083. [Google Scholar] [CrossRef]
- Ferrer, M.D.; Sureda, A.; Batle, J.M.; Tauler, P.; Tur, J.A.; Pons, A. Scuba diving enhances endogenous antioxidant defenses in lymphocytes and neutrophils. Free Radic. Res. 2007, 41, 274–281. [Google Scholar] [CrossRef]
- Perovic, A.; Sobocanec, S.; Dabelic, S.; Balog, T.; Dumic, J. Effect of scuba diving on the oxidant/antioxidant status, SIRT1 and SIRT3 expression in recreational divers after a winter nondive period. Free Radic. Res. 2018, 52, 188–197. [Google Scholar] [CrossRef]
- Faraci, F.M.; Didion, S.P. Vascular protection: Superoxide dismutase isoforms in the vessel wall. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Sureda, A.; Batle, J.M.; Ferrer, M.D.; Mestre-Alfaro, A.; Tur, J.A.; Pons, A. Scuba diving activates vascular antioxidant system. Int. J. Sport. Med. 2012, 33, 531–536. [Google Scholar] [CrossRef]
- Bosco, G.; Giacon, T.A.; Paolocci, N.; Vezzoli, A.; Noce, C.D.; Paganini, M.; Agrimi, J.; Garetto, G.; Cialoni, D.; D’Alessandro, N.; et al. Dopamine/BDNF loss underscores narcosis cognitive impairment in divers: A proof of concept in a dry condition. Eur. J. Appl. Physiol. 2022, 123, 143–158. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, S.; Kang, S.; Kim, S.H.; Kim, J.C.; Yang, M.; Moon, C. Brain-derived neurotropic factor and GABAergic transmission in neurodegeneration and neuroregeneration. Neural. Regen. Res. 2017, 12, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Lavoute, C.; Weiss, M.; Rostain, J.C. Alterations in nigral NMDA and GABAA receptor control of the striatal dopamine level after repetitive exposures to nitrogen narcosis. Exp. Neurol. 2008, 212, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Lemaitre, F.; Costalat, G.; Allinger, J.; Balestra, C. Possible causes of narcosis-like symptoms in freedivers. Undersea Hyperb. Med. 2023, 50, 85–93. [Google Scholar] [CrossRef]
- Di Pumpo, F.; Gualtiero, M.; Paganini, M.; Cialoni, D.; Giacomo, G.; Alessandro Cipriano, A.; Giacon, T.A.; Martani, L.; Camporesi, E.; Bosco, G. Comparison between Arterial Blood Gases and Oxygen Reserve Index™ in a SCUBA Diver: A Case Report. Healthcare 2023, 11, 1102. [Google Scholar] [CrossRef]
- Rocco, M.; Pelaia, P.; Di Benedetto, P.; Conte, G.; Maggi, L.; Fiorelli, S.; Mercieri, M.; Balestra, C.; De Blasi, R.A.; on behalf of ROAD Project Investigators. Inert gas narcosis in scuba diving, different gases different reactions. Eur. J. Appl. Physiol. 2019, 119, 247–255. [Google Scholar] [CrossRef]
- Lafere, P.; Balestra, C.; Hemelryck, W.; Guerrero, F.; Germonpre, P. Do Environmental Conditions Contribute to Narcosis Onset and Symptom Severity? Int. J. Sport. Med. 2016, 37, 1124–1128. [Google Scholar] [CrossRef] [PubMed]
- Ersoz, G.; Ocakcioglu, B.; Bastug, M.; Ficicilar, H.; Yavuzer, S. Platelet aggregation and release function in hyperbaric oxygenation. Undersea Hyperb. Med. 1998, 25, 229–232. [Google Scholar] [PubMed]
- Hjelde, A.; Bergh, K.; Brubakk, A.O.; Iversen, O.J. Complement activation in divers after repeated air/heliox dives and its possible relevance to DCS. J. Appl. Physiol. 1995, 78, 1140–1144. [Google Scholar] [CrossRef]
- Morabito, C.; Bosco, G.; Pilla, R.; Corona, C.; Mancinelli, R.; Yang, Z.; Camporesi, E.M.; Fano, G.; Mariggio, M.A. Effect of pre-breathing Oxygen at different depth on oxidative status and calcium concentration in lymphocytes of scuba divers. Acta Physiol. 2011, 202, 69–78. [Google Scholar] [CrossRef]
- Dumić, J.; Cvetko, A.; Abramović, I.; Goreta, S.S.; Perović, A.; Bratičević, M.N.; Kifer, D.; Sinčić, N.; Gornik, O.; Žarak, M. Changes in Specific Biomarkers Indicate Cardiac Adaptive and Anti-inflammatory Response of Repeated Recreational SCUBA Diving. Front. Cardiovasc. Med. 2022, 9, 855682. [Google Scholar] [CrossRef]
- Tillmans, F.; Sharghi, R.; Noy, T.; Kähler, W.; Klapa, S.; Sartisohn, S.; Sebensm, S.; Kochm, A. Effect of Hyperoxia on the immune status of oxygen divers and endurance athletes. Free Radic. Res. 2019, 53, 522–534. [Google Scholar] [CrossRef]
- Eckmann, D.M.; Armstead, S.C. Influence of endothelial glycocalyx degradation and surfactants on air embolism adhesion. Anesthesiology 2006, 105, 1220–1227. [Google Scholar] [CrossRef] [PubMed]
- Philp, R.B.; Inwood, M.J.; Warren, B.A. Interactions between gas bubbles and components of the blood: Implications in decompression sickness. Aerosp. Med. 1972, 43, 946–953. [Google Scholar]
- Philp, R.B.; Schacham, P.; Gowdey, C.W. Involvement of platelets and microthrombi in experimental decompression sickness: Similarities with disseminated intravascular coagulation. Aerosp. Med. 1971, 42, 494–502. [Google Scholar]
- Kuroiwa, K. The functional and biochemical changes of platelets in experimental decompression sickness of rabbits. Bull. Tokyo Med. Dent. Univ. 1984, 31, 73–84. [Google Scholar]
- Bennett, M.; Mitchell, S.; Dominguez, A. Adjunctive treatment of decompression illness with a non-steroidal anti-inflammatory drug (tenoxicam) reduces compression requirement. Undersea Hyperb. Med. 2003, 30, 195–205. [Google Scholar] [PubMed]
- Ersson, A.; Walles, M.; Ohlsson, K.; Ekholm, A. Chronic hyperbaric exposure activates proinflammatory mediators in humans. J. Appl. Physiol. 2002, 92, 2375–2380. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.R.; Palombo, L.J.; Jensen, A.E.; Bernards, J.R. Efficacy of closed cell wet-suit at various depths and gas mixtures for thermoprotection during military training dives. Front. Physiol. 2023, 14, 1165196. [Google Scholar] [CrossRef]
- Li, Y.; Liu, K.; Kang, Z.M.; Sun, X.J.; Liu, W.W.; Mao, Y.F. Helium preconditioning protects against neonatal hypoxia-ischemia via nitric oxide-mediated up-regulation of antioxidants in a rat model. Behav. Brain Res. 2016, 300, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Bhopale, V.M.; Ruhela, D.; Brett, K.D.; Nugent, N.Z.; Fraser, N.K.; Levinson, S.L.; DiNubile, M.J.; Thom, S.R. Plasma gelsolin modulates the production and fate of IL-1β-containing microparticles following high-pressure exposure and decompression. J. Appl. Physiol. 2021, 130, 1604–1613. [Google Scholar] [CrossRef] [PubMed]
- Balestra, C.; Arya, A.K.; Leveque, C.; Virgili, F.; Germonpre, P.; Lambrechts, K.; Lafere, P.; Thom, S.R. Varying Oxygen Partial Pressure Elicits Blood-Borne Microparticles Expressing Different Cell-Specific Proteins-Toward a Targeted Use of Oxygen? Int. J. Mol. Sci. 2022, 23, 7888. [Google Scholar] [CrossRef]
- Rocco, M.; Maggi, L.; Loffredo, C.; Pelli, M.; Di Benedetto, P.; Fiorelli, S.; Simmaco, M.; De Blasi, R.A. The impact of different gas mixtures on inflammatory responses in advanced recreational divers. Diving Hyperb. Med. 2021, 51, 140–146. [Google Scholar] [CrossRef]
- Yang, M.; Milovanova, T.N.; Bogush, M.; Uzun, G.; Bhopale, V.M.; Thom, S.R. Microparticle enlargement and altered surface proteins after air decompression are associated with inflammatory vascular injuries. J. Appl. Physiol. 2012, 112, 204–211. [Google Scholar] [CrossRef]
- Landolfi, A.; Yang, Z.J.; Savini, F.; Camporesi, E.M.; Faralli, F.; Bosco, G. Pretreatment with hyperbaric oxygenation reduces bubble formation and platelet activation. Sport Sci. Health 2006, 1, 122–128. [Google Scholar] [CrossRef]
- Camporesi, E.M.; Bosco, G. Hyperbaric oxygen pretreatment and preconditioning. Undersea Hyperb. Med. 2014, 41, 259–263. [Google Scholar]
- Dreyer, S.; Schneppendahl, J.; Moeller, F.; Koch, A.; Muth, T.; Schipke, J.D. An Updated Narrative Review on Er-gometric Systems Applied to Date in Assessing Divers’ Fitness. Healthcare 2021, 9, 1044. [Google Scholar] [CrossRef]
- Baj, Z.; Olszanski, R.; Majewska, E.; Konarski, M. The effect of air and nitrox divings on platelet activation tested by flow cytometry. Aviat. Space Environ. Med. 2000, 71, 925–928. [Google Scholar]
- Bosco, G.; Rizzato, A.; Quartesan, S.; Camporesi, E.; Mangar, D.; Paganini, M.; Cenci, L.; Malacrida, S.; Mrakic-Sposta, S.; Moretti, S.; et al. Effects of the Ketogenic diet in overweight divers breathing Enriched Air Nitrox. Sci. Rep. 2018, 8, 2655. [Google Scholar] [CrossRef]
- Gronow, G.; Kahler, W.; Koch, A.; Klause, N. Benzoate hydroxylation: A measure of oxidative stress in divers. Adv. Exp. Med. Biol. 2005, 566, 223–229. [Google Scholar] [CrossRef]
- Doubt, T.J. Cardiovascular and thermal responses to SCUBA diving. Med. Sci. Sport. Exerc. 1996, 28, 581–586. [Google Scholar] [CrossRef]
- Madden, D.; Thom, S.R.; Milovanova, T.N.; Yang, M.; Bhopale, V.M.; Ljubkovic, M.; Dujic, Z. Exercise before SCUBA diving ameliorates decompression-induced neutrophil activation. Med. Sci. Sports Exerc. 2014, 46, 1928–1935. [Google Scholar] [CrossRef] [PubMed]
- Spisni, E.; Marabotti, C.; De Fazio, L.; Valerii, M.C.; Cavazza, E.; Brambilla, S.; Hoxha, K.; L’Abbate, A.; Longobardi, P. A comparative evaluation of two decompression procedures for technical diving using inflammatory responses: Compartmental versus ratio deco. Diving Hyperb. Med. 2017, 47, 9–16. [Google Scholar] [CrossRef]
- Romsbotn, S.; Eftedal, I.; Vaag, J.R. A Work Environment Under Pressure: Psychosocial Job Demands and Resources Among Saturation Divers. Front. Public Health 2022, 10, 765197. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, S.; Deussen, A.; Berndt, D.; Schipke, J.D. How to Survive 33 min after the Umbilical of a Saturation Diver Severed at a Depth of 90 msw? Healthcare 2022, 10, 453. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.C. Rescue of a saturation diver, unconscious due to an explosion underwater. Int. Marit. Health 2021, 72, 46–48. [Google Scholar] [CrossRef]
- Gjerde, K.Ø.; Ryggvik, H. On the Edge, Under Water: Offshore Diving in Norway, 2nd ed.; Wigestrand Forlag: Stavanger, Norway, 2014. [Google Scholar]
- Ramnefjell, M.P.; Morild, I.; Mørk, S.J.; Lilleng, P.K. Fatal diving accidents in western Norway 1983–2007. Forensic Sci. Int. 2012, 223, e22–e26. [Google Scholar] [CrossRef]
- Keatinge, W.R.; Hayward, M.G.; McIver, N.K. Hypothermia during saturation diving in the North Sea. Br. Med. J. 1980, 280, 291. [Google Scholar] [CrossRef]
- Beyerstein, G.; Lang, M.A.; Smith, N.E. Commercial Diving: Surface-Mixed Gas, Sur-D-O2, Bell Bounce, Saturation. In Proceedings of the Advanced Scientific Diving Workshop, Washington, DC, USA, 23–24 February 2006. [Google Scholar]
- Brubakk, A.O.; Ross, J.A.; Thom, S.R. Saturation diving; physiology and pathophysiology. Compr. Physiol. 2014, 4, 1229–1272. [Google Scholar] [CrossRef]
- Mrakic-Sposta, S.; Vezzoli, A.; D’Alessandro, F.; Paganini, M.; Dellanoce, C.; Cialoni, D.; Bosco, G. Change in Oxidative Stress Biomarkers During 30 Days in Saturation Dive: A Pilot Study. Int. J. Environ. Res. Public Health 2020, 17, 7118. [Google Scholar] [CrossRef] [PubMed]
- Zwart, S.R.; Jessup, J.M.; Ji, J.; Smith, S.M. Saturation Diving Alters Folate Status and Biomarkers of DNA Damage and Repair. PLoS ONE 2012, 7, e31058. [Google Scholar] [CrossRef]
- Imbert, J.P.; Balestra, C.; Kiboub, F.Z.; Loennechen, Ø.; Eftedal, I. Commercial Divers’ Subjective Evaluation of Saturation. Front. Psychol. 2019, 9, 2774. [Google Scholar] [CrossRef]
- Monnoyer, R.; Eftedal, I.; Hjelde, A.; Deb, S.; Haugum, K.; Lautridou, J. Functional Profiling Reveals Altered Metabolic Activity in Divers’ Oral Microbiota During Commercial Heliox Saturation Diving. Front. Physiol. 2021, 12, 702634. [Google Scholar] [CrossRef]
- Sureda, A.; Batle, J.M.; Capo, X.; Martorell, M.; Cordova, A.; Tur, J.A.; Pons, A. Scuba diving induces nitric oxide synthesis and the expression of inflammatory and regulatory genes of the immune response in neutrophils. Physiol. Genom. 2014, 46, 647–654. [Google Scholar] [CrossRef]
- Krog, J.; Tønnesen, E.K.; Jepsen, C.F.; Parner, E.; Segadal, K.; Hope, A.; Ulvik, R.J.; Holland, M.E. Nural killer cells as biomarkers of hyperbaric stress during a dry heliox saturation dive. Aviat. Space Environ. Med. 2010, 81, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Domoto, H.; Iwaya, K.; Ikomi, F.; Matsuo, H.; Tadano, Y.; Fujii, S.; Tachi, K.; Itoh, Y.; Sato, M.; Inoue, K.; et al. Upregulation of antioxidant proteins in the plasma proteome during saturation diving: Unique coincidence under hypobaric Hypoxia. PLoS ONE 2016, 11, e0163804. [Google Scholar] [CrossRef] [PubMed]
- Djurhuus, R.; Segadal, K.; Svardal, A.M. Glutathione in blood cells decreases without DNA breaks after a simulated saturation dive to 250 msw. Aviat. Space Environ. Med. 2006, 77, 597–604. [Google Scholar]
- Kiboub, F.Z.; Møllerløkken, A.; Hjelde, A.; Flatberg, A.; Loennechen, Ø.; Eftedal, I. Blood Gene Expression and Vascular Function Biomarkers in Professional Saturation Diving. Front. Physiol. 2018, 9, 937. [Google Scholar] [CrossRef]
- Smith, S.M.; Davis-Street, J.E.; Fesperman, J.V.; Smith, M.D.; Rice, B.L.; Zwart, S.R. Nutritional assessment during a 14-d saturation dive: The NASA Extreme Environment Mission Operations V Project. J. Nutr. 2004, 134, 1765–1771. [Google Scholar] [CrossRef]
- Ikeda, M.; Nakabayashi, K.; Shinkai, M.; Hara, Y.; Kizaki, T.; Oh-Ishi, S.; Ohno, H. Supplementation of antioxidants prevents oxidative stress during a deep saturation dive. Tohoku J. Exp. Med. 2004, 203, 353–357. [Google Scholar] [CrossRef]
- Deb, S.K.; Swinton, P.A.; Dolan, E. Nutritional considerations during prolonged exposure to a confined, hyperbaric, hyperoxic environment: Recommendations for saturation divers. Extrem. Physiol. Med. 2016, 5, 1. [Google Scholar] [CrossRef]
- Deb, S.K.; Dolan, E.; Hambly, C.; Speakman, J.R.; Eftedal, O.; Zariwala, M.G.; Eftedal, I. The Assessment of Daily Energy Expenditure of Commercial Saturation Divers Using Doubly Labelled Water. Front. Physiol. 2021, 12, 687605. [Google Scholar] [CrossRef]
- Monnoyer, R.; Lautridou, J.; Deb, S.; Hjelde, A.; Eftedal, I. Using Salivary Biomarkers for Stress Assessment in Offshore Saturation Diving: A Pilot Study. Front. Physiol. 2021, 12, 791525. [Google Scholar] [CrossRef] [PubMed]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Oxygen Species in Inflammation and Tissue Injury. Antioxid. Redox Signal. 2014, 20, 7. [Google Scholar] [CrossRef] [PubMed]
- Alhayaza, R.; Haque, E.; Karbasiafshar, C.; Sellke, F.W.; Abid, M.R. The Relationship Between Reactive Oxygen Species and Endothelial Cell Metabolism. Front. Chem. 2020, 8, 592688. [Google Scholar] [CrossRef] [PubMed]
- Teleanu, D.M.; Niculescu, A.-G.; Lungu, I.I.; Radu, C.I.; Vladâcenco, O.; Roza, E.; Costăchescu, B.; Grumezescu, A.M.; Teleanu, R.I. An Overview of Oxidative Stress, Neuroinflammation, and Neurodegenerative Diseases. Int. J. Mol. Sci. 2022, 23, 5938. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, E.; Ananiev, J.; Yovchev, Y.; Arabadzhiev, G.; Abrashev, H.; Abrasheva, D.; Atanasov, V.; Kostandieva, R.; Mitev, M.; Petkova-Parlapanska, K.; et al. COVID-19 Complications: Oxidative Stress, Inflammation, and Mitochondrial and Endothelial Dysfunction. Int. J. Mol. Sci. 2023, 24, 14876. [Google Scholar] [CrossRef]
- Comhair, S.A.; Erzurum, S.C. Antioxidant responses to oxidant-mediated lung diseases. Am. J. Physiol. Lung Cell. Mol. Physiol. 2002, 283, 246–255. [Google Scholar] [CrossRef]
- Gempp, E.; Blatteau, J.E. Preconditioning Methods and Mechanisms for Preventing the Risk of Decompression Sickness in Scuba Divers: A Review. Res. Sports Med. 2010, 18, 205–218. [Google Scholar] [CrossRef]
- Winkler, B.; Muth, C.M.; Piepho, T. Hyperbare Therapie und Tauchmedizin—Tauchmedizin: Status quo und Ausblick. Anästhesiol Intensivmed Notfallmed Schmerzther 2015, 50, 638–646. [Google Scholar] [CrossRef]
- Madden, D.; Thom, S.R.; Dujic, Z. Exercise before and after SCUBA diving and the role of cellular microparticles in decompression stress. Med. Hypotheses 2016, 86, 80–84. [Google Scholar] [CrossRef]
- Held, H.E.; Pendergast, D.R. The effects of respiratory muscle training on respiratory mechanics and energy cost. Respir. Physiol. Neurobiol. 2014, 200, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Germonpré, P.; Balestra, C. Preconditioning to Reduce Decompression Stress in Scuba Divers. Aerosp. Med. Hum. Perform. 2017, 88, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Šegrt Ribičić, I.; Valić, M.; Lušić Kalcina, L.; Božić, J.; Obad, A.; Glavaš, D.; Glavičić, I.; Valić, Z. Effects of Oxygen Prebreathing on Bubble Formation, Flow-Mediated Dilatation, and Psychomotor Performance during Trimix Dives. Sports 2024, 12, 35. [Google Scholar] [CrossRef] [PubMed]
- Barak, O.F.; Caljkusic, K.; Hoiland, R.L.; Ainslie, P.N.; Thom, S.R.; Yang, M.; Jovanov, P.; Dujic, Z. Differential influence of vitamin C on the peripheral and cerebral circulation after diving and exposure to hyperoxia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R759–R767. [Google Scholar] [CrossRef]
- Thiele, R.H. Subcellular Energetics and Metabolism: Potential Therapeutic Applications. Obstet. Anesthesia Dig. 2017, 124, 1872–1885. [Google Scholar] [CrossRef]
- Arieli, R. Nanobubbles Form at Active Hydrophobic Spots on the Luminal Aspect of Blood Vessels: Consequences for Decompression Illness in Diving and Possible Implications for Autoimmune Disease—An Overview. Front. Physiol. 2017, 8, 591. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Barak, O.F.; Dujic, Z.; Madden, D.; Bhopale, V.M.; Bhullar, J.; Thom, S.R. Ascorbic acid supplementation diminishes microparticle elevations and neutrophil activation following SCUBA diving. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R338–R344. [Google Scholar] [CrossRef] [PubMed]
- Obad, A.; Valic, Z.; Palada, I.; Brubakk, A.O.; Modun, D.; Dujić, Z. Antioxidant Pretreatment and Reduced Arterial Endothelial Dysfunction After Diving. Aviat. Space Environ. Med. 2007, 78, 1114–1120. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).