Bone-Differentiation-Associated Circ-Spen Regulates Death of Mouse Bone Marrow Mesenchymal Stem Cells by Inhibiting Apoptosis and Promoting Autophagy
Abstract
1. Introduction
2. Results
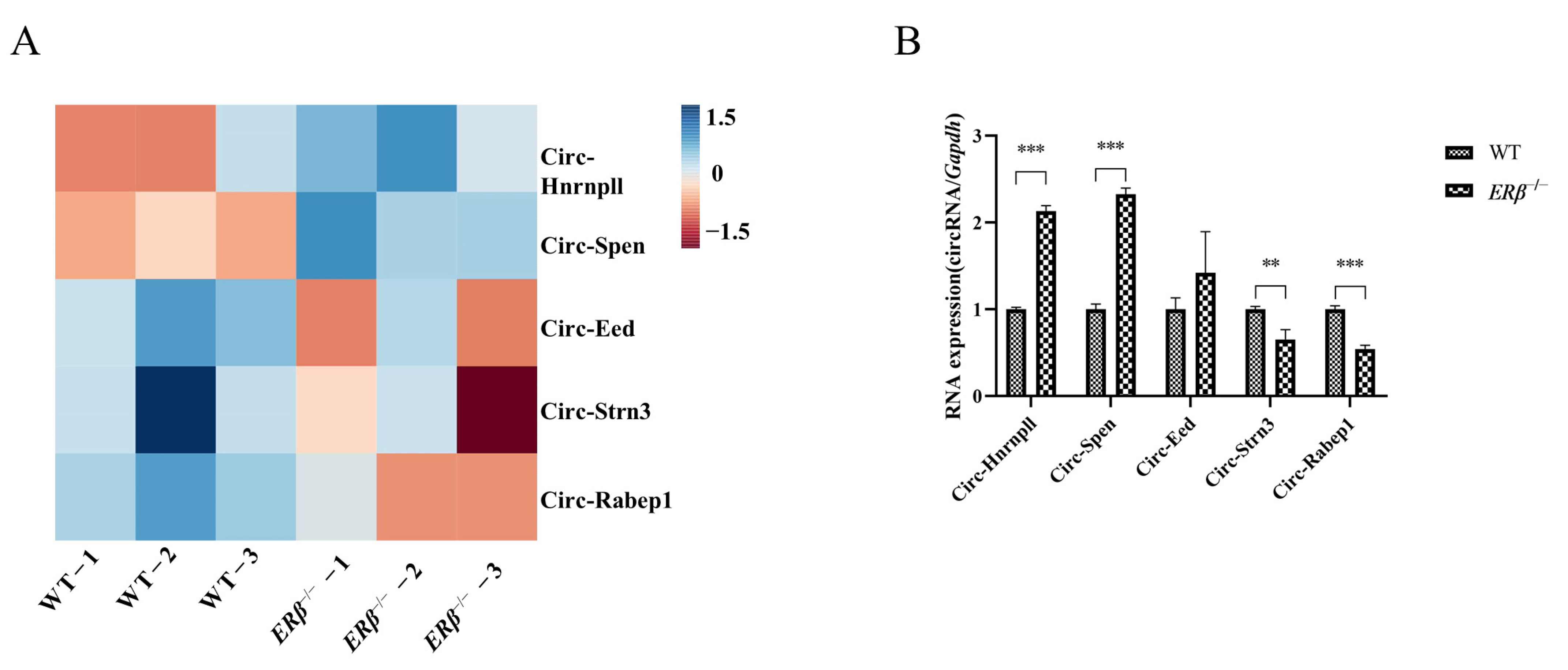
2.1. The Differential Expressions of circRNAs in ERβ−/− Mice
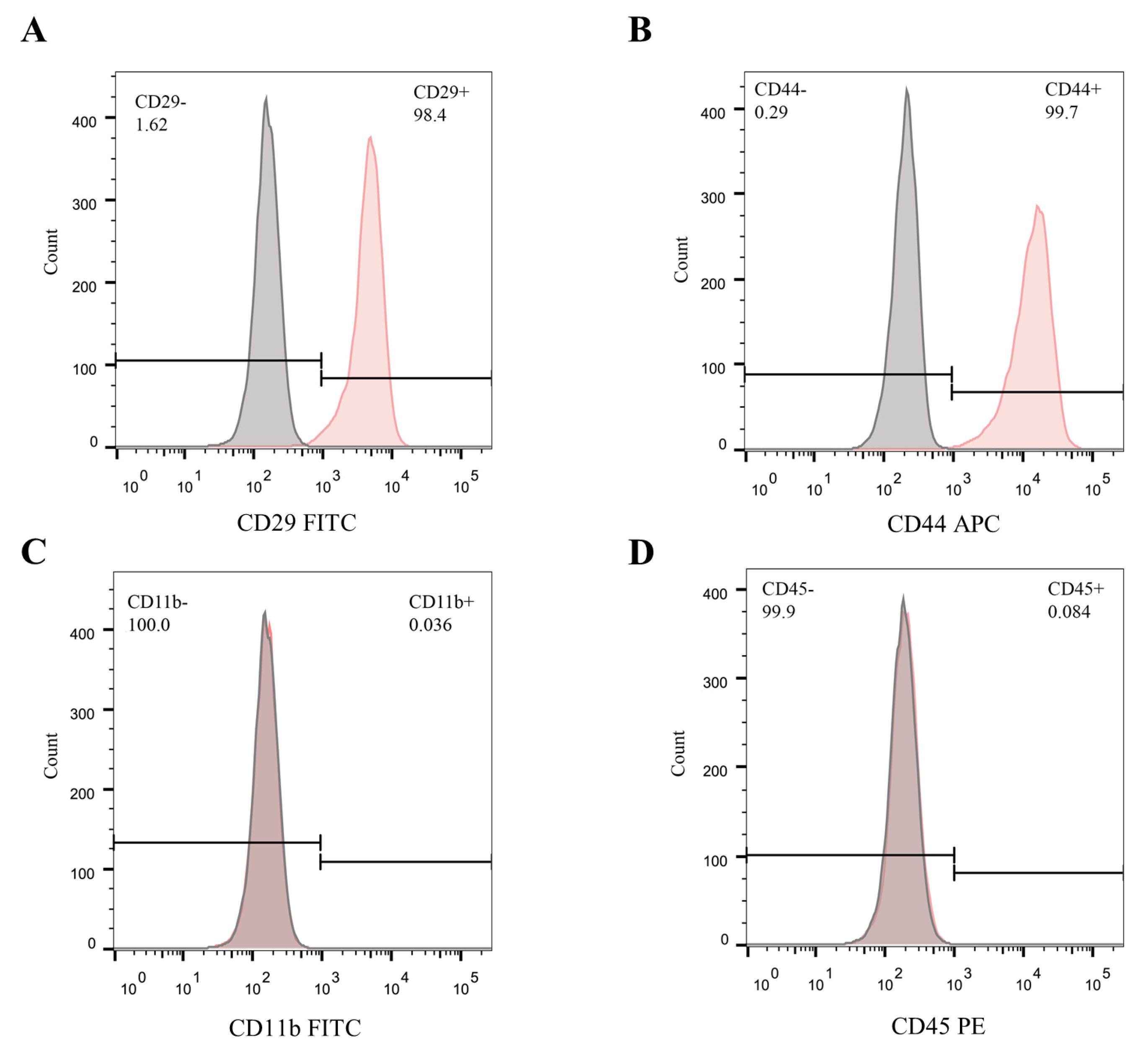
2.2. Identification of mBMSCs
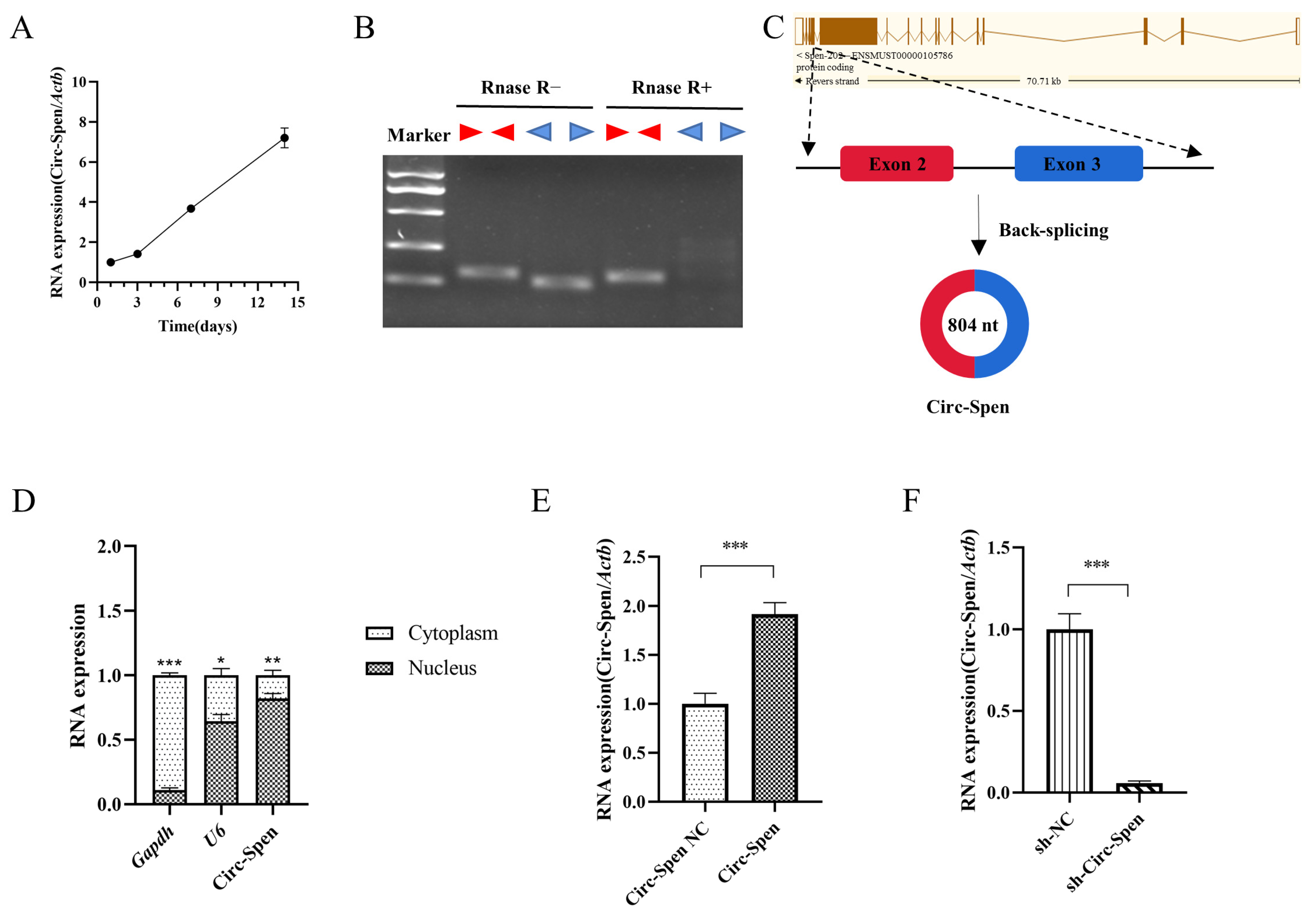
2.3. The Expression of Circ-Spen Increases in Osteogenesis-Induced mBMSCs and Has Been Identified as a Circular RNA in the Nucleus
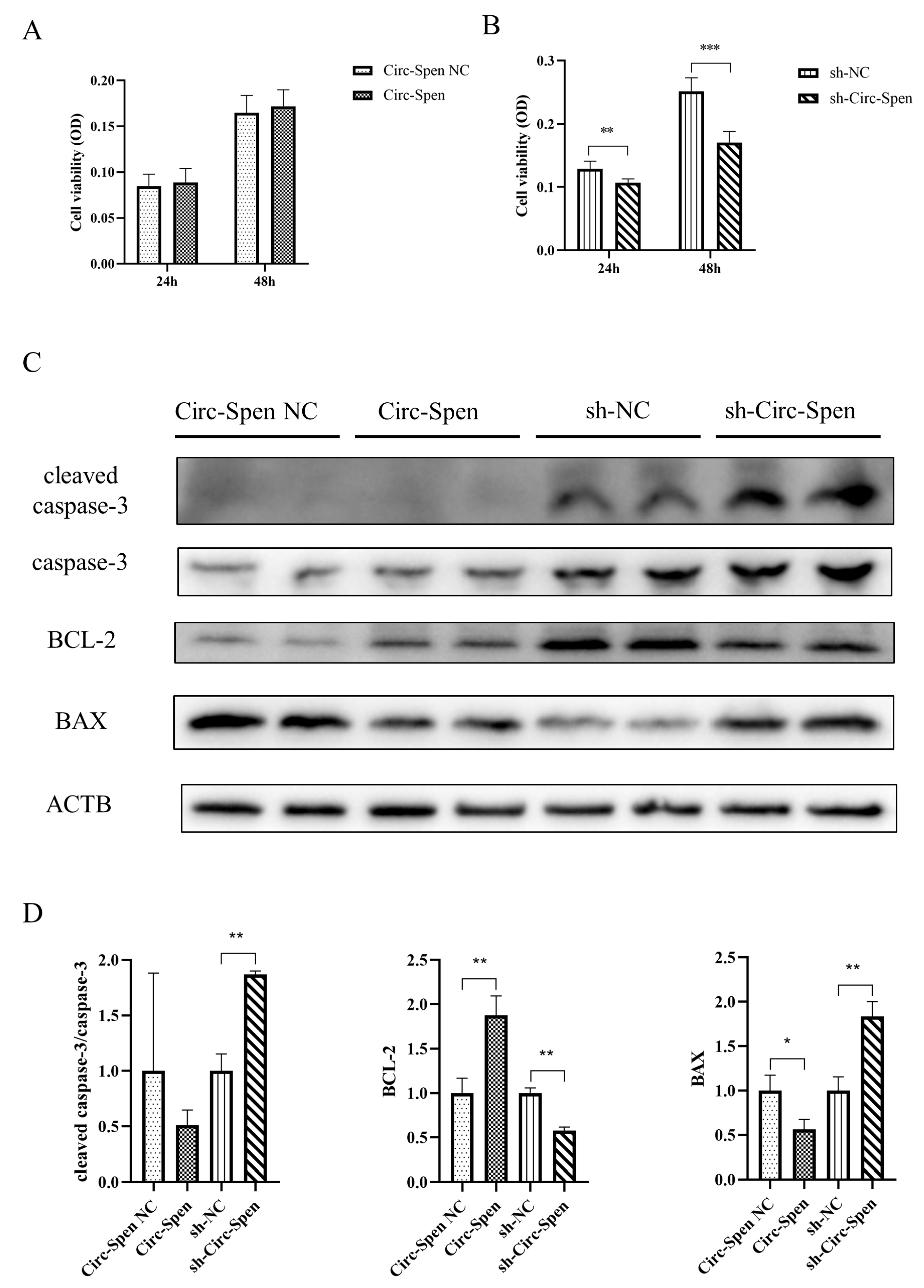
2.4. Circ-Spen Inhibits Cell Apoptosis in mBMSCs
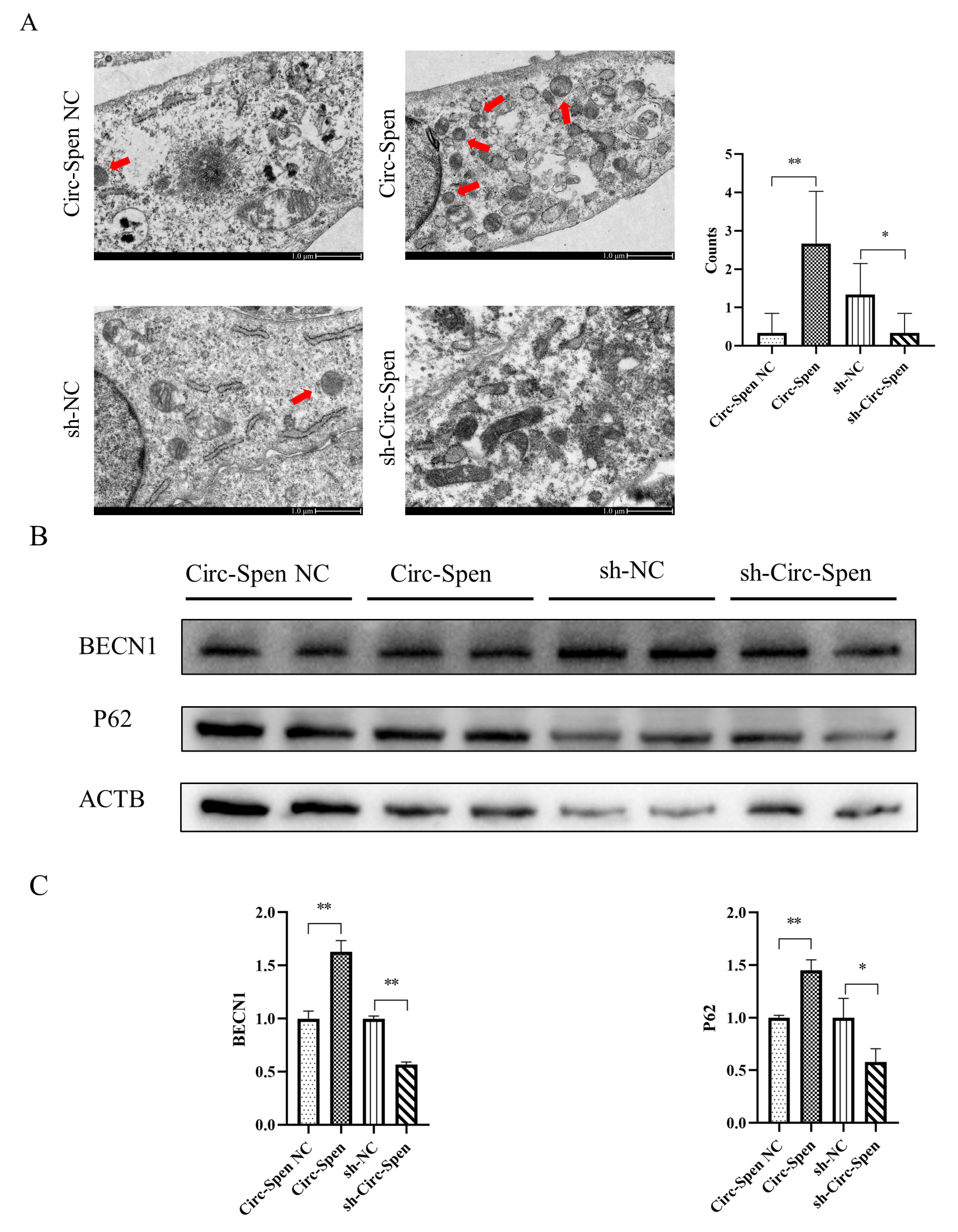
2.5. Circ-Spen Promotes Autophagy in mBMSCs
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. RNA-Seq
4.3. Cell Culture
4.4. Flow Cytometry Analysis
4.5. Cell Transfection
4.6. RNase R Treatment
4.7. CircRNA Localization
4.8. Transmission Electron Microscopy (TEM)
4.9. Methylthiazol Tetrazolium (MTT) Assays
4.10. RNA Isolation and qRT-PCR
4.11. Western Blotting
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bord, S.; Horner, A.; Beavan, S.; Compston, J. Estrogen receptors alpha and beta are differentially expressed in developing human bone. J. Clin. Endocrinol. Metab. 2001, 86, 2309–2314. [Google Scholar] [PubMed]
- Emmanuelle, N.E.; Marie-Cécile, V.; Florence, T.; Jean-François, A.; Françoise, L.; Coralie, F.; Alexia, V. Critical Role of Estrogens on Bone Homeostasis in Both Male and Female: From Physiology to Medical Implications. Int. J. Mol. Sci. 2021, 22, 1568. [Google Scholar] [PubMed]
- Ke, H.Z.; Brown, T.A.; Qi, H.; Crawford, D.T.; Simmons, H.A.; Petersen, D.N.; Allen, M.R.; McNeish, J.D.; Thompson, D.D. The role of estrogen receptor-beta, in the early age-related bone gain and later age-related bone loss in female mice. J. Musculoskelet. Neuronal Interact. 2002, 2, 479–488. [Google Scholar] [PubMed]
- Wu, G.; Xu, R.; Zhang, P.; Xiao, T.; Fu, Y.; Zhang, Y.; Du, Y.; Ye, J.; Cheng, J.; Jiang, H. Estrogen regulates stemness and senescence of bone marrow stromal cells to prevent osteoporosis via ERβ-SATB2 pathway. J. Cell. Physiol. 2018, 233, 4194–4204. [Google Scholar] [CrossRef] [PubMed]
- Shu, H.S.; Liu, Y.L.; Tang, X.T.; Zhang, X.S.; Zhou, B.; Zou, W.; Zhou, B.O. Tracing the skeletal progenitor transition during postnatal bone formation. Cell Stem Cell. 2021, 28, 2122–2136.e3. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Bo, X.; Ma, X.; Hou, K.; Li, D.; Geng, W.; Zeng, J. Craniomaxillofacial derived bone marrow mesenchymal stem/stromal cells (BMSCs) for craniomaxillofacial bone tissue engineering: A literature review. J. Stomatol. Oral Maxillofac. Surg. 2022, 123, e650–e659. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Peng, W.; Zhang, J.; Dong, W.; Wu, J.; Wang, T.; Xie, Z. P53 and Parkin co-regulate mitophagy in bone marrow mesenchymal stem cells to promote the repair of early steroid-induced osteonecrosis of the femoral head. Cell Death Dis. 2020, 11, 42. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L. The biogenesis and emerging roles of circular RNAs. Nat. Rev. Mol. Cell Biol. 2016, 17, 205–211. [Google Scholar] [CrossRef]
- Lin, Z.; Tang, X.; Wan, J.; Zhang, X.; Liu, C.; Liu, T. Functions and mechanisms of circular RNAs in regulating stem cell differentiation. RNA Biol. 2021, 18, 2136–2149. [Google Scholar] [CrossRef]
- Della Bella, E.; Menzel, U.; Basoli, V.; Tourbier, C.; Alini, M.; Stoddart, M.J. Differential Regulation of circRNA, miRNA, and piRNA during Early Osteogenic and Chondrogenic Differentiation of Human Mesenchymal Stromal Cells. Cells 2020, 9, 398. [Google Scholar] [CrossRef]
- Chia, W.; Liu, J.; Huang, Y.G.; Zhang, C. A circular RNA derived from DAB1 promotes cell proliferation and osteogenic differentiation of BMSCs via RBPJ/DAB1 axis. Cell Death Dis. 2020, 11, 372. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Xiang, Q.; Jia, J.; Guo, T.; Liao, Z.; Yang, S.; Cai, X.; Liu, X. CircHGF suppressed cell proliferation and osteogenic differentiation of BMSCs in ONFH via inhibiting miR-25-3p binding to SMAD7. Mol. Ther. Nucleic Acids 2022, 28, 99–113. [Google Scholar] [CrossRef]
- Dossin, F.; Pinheiro, I.; Żylicz, J.J.; Roensch, J.; Collombet, S.; Le Saux, A.; Chelmicki, T.; Attia, M.; Kapoor, V.; Zhan, Y.; et al. SPEN integrates transcriptional and epigenetic control of X-inactivation. Nature 2020, 578, 455–460. [Google Scholar] [CrossRef]
- Robert-Finestra, T.; Tan, B.F.; Mira-Bontenbal, H.; Timmers, E.; Gontan, C.; Merzouk, S.; Giaimo, B.D.; Dossin, F.; van IJcken, W.F.J.; Martens, J.W.M.; et al. SPEN is required for Xist upregulation during initiation of X chromosome inactivation. Nat. Commun. 2021, 12, 7000. [Google Scholar] [CrossRef]
- Légaré, S.; Chabot, C.; Basik, M. SPEN, a new player in primary cilia formation and cell migration in breast cancer. Breast Cancer Res. 2017, 19, 104. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lv, Y.; Cheng, C.; Huang, Y.; Yang, L.; He, J.; Tao, X.; Hu, Y.; Ma, Y.; Su, Y.; et al. SPEN induces miR-4652-3p to target HIPK2 in nasopharyngeal carcinoma. Cell Death Dis. 2020, 11, 509. [Google Scholar] [CrossRef]
- Rattka, M.; Westphal, S.; Gahr, B.M.; Just, S.; Rottbauer, W. Spen deficiency interferes with Connexin 43 expression and leads to heart failure in zebrafish. J. Mol. Cell Cardiol. 2021, 155, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.; Boyette, L.B.; Tuan, R.S. Characterization of bone marrow-derived mesenchymal stem cells in aging. Bone 2015, 70, 37–47. [Google Scholar] [CrossRef]
- Luo, X.; Li, H.; Sun, X.; Zuo, Q.; Li, B.; Zhu, Y.; Wei, W.; Gu, X. Promotion of Differentiating Bone Marrow Mesenchymal Stromal Cells (BMSCs) into Cardiomyocytes via HCN2 and HCN4 Cotransfection. BioMed Res. Int. 2021, 2021, 5529276. [Google Scholar] [CrossRef]
- Zheng, L.; Liang, H.; Zhang, Q.; Shen, Z.; Sun, Y.; Zhao, X.; Gong, J.; Hou, Z.; Jiang, K.; Wang, Q.; et al. circPTEN1, a circular RNA generated from PTEN, suppresses cancer progression through inhibition of TGF-β/Smad signaling. Mol. Cancer 2022, 21, 41. [Google Scholar] [CrossRef]
- Patop, I.L.; Wüst, S.; Kadener, S. Past, present, and future of circRNAs. EMBO J. 2019, 38, e100836. [Google Scholar] [CrossRef]
- Kristensen, L.S.; Andersen, M.S.; Stagsted, L.V.W.; Ebbesen, K.K.; Hansen, T.B.; Kjems, J. The biogenesis, biology and characterization of circular RNAs. Nat. Rev. Genet. 2019, 20, 675–691. [Google Scholar] [CrossRef]
- Chen, Z.; Lin, W.; Zhao, S.; Mo, X.; Wen, Z.; Cheung, W.H.; Fu, D.; Chen, B. Identification of circRNA Expression Profiles in BMSCs from Glucocorticoid-Induced Osteoporosis Model. Stem Cells Int. 2022, 2022, 3249737. [Google Scholar] [CrossRef] [PubMed]
- Xin, W.; Yuan, S.; Wang, B.; Qian, Q.; Chen, Y. Hsa_circ_0066523 promotes the proliferation and osteogenic differentiation of bone mesenchymal stem cells by repressing PTEN. Bone Joint Res. 2021, 10, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Arthur, A.; Gronthos, S. Clinical Application of Bone Marrow Mesenchymal Stem/Stromal Cells to Repair Skeletal Tissue. Int. J. Mol. Sci. 2020, 21, 9759. [Google Scholar] [CrossRef] [PubMed]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Shahali, A.; Ghanadian, M.; Jafari, S.M.; Aghaei, M. Mitochondrial and caspase pathways are involved in the induction of apoptosis by nardosinen in MCF-7 breast cancer cell line. Res. Pharm. Sci. 2018, 13, 12–21. [Google Scholar] [PubMed]
- Cory, S.; Adams, J.M. The Bcl2 family: Regulators of the cellular life-or-death switch. Nat. Rev. Cancer 2002, 2, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Changizi, Z.; Moslehi, A.; Rohani, A.H.; Eidi, A. Chlorogenic acid induces 4T1 breast cancer tumor’s apoptosis via p53, Bax, Bcl-2, and caspase-3 signaling pathways in BALB/c mice. J. Biochem. Mol. Toxicol. 2021, 35, e22642. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Ma, W.; Xiao, J.; Dai, X.; Ling, W. CircRNA_0092516 regulates chondrocyte proliferation and apoptosis in osteoarthritis through the miR-337-3p/PTEN axis. J. Biochem. 2021, 169, 467–475. [Google Scholar] [CrossRef]
- Cao, G.; Meng, X.; Han, X.; Li, J. Exosomes derived from circRNA Rtn4-modified BMSCs attenuate TNF-α-induced cytotoxicity and apoptosis in murine MC3T3-E1 cells by sponging miR-146a. Biosci. Rep. 2020, 40, BSR20193436. [Google Scholar] [CrossRef]
- Li, Z.; Li, D.; Chen, R.; Gao, S.; Xu, Z.; Li, N. Cell death regulation: A new way for natural products to treat osteoporosis. Pharmacol. Res. 2023, 187, 106635. [Google Scholar] [CrossRef] [PubMed]
- Maiuri, M.C.; Zalckvar, E.; Kimchi, A.; Kroemer, G. Self-eating and self-killing: Crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2007, 8, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, Y.; Cao, J.; Wang, Y.; Anwar, N.; Zhang, Z.; Zhang, D.; Ma, Y.; Xiao, Y.; Xiao, L.; et al. The role of autophagy in bone metabolism and clinical significance. Autophagy 2023, 19, 2409–2427. [Google Scholar] [CrossRef] [PubMed]
- Shi, B.; Ma, M.; Zheng, Y.; Pan, Y.; Lin, X. mTOR and Beclin1: Two key autophagy-related molecules and their roles in myocardial ischemia/reperfusion injury. J. Cell. Physiol. 2019, 234, 12562–12568. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Zeh, H.J.; Lotze, M.T.; Tang, D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ. 2011, 18, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Bai, L.; Li, Y.; Wang, K. The functional mechanism of bone marrow-derived mesenchymal stem cells in the treatment of animal models with Alzheimer’s disease: Crosstalk between autophagy and apoptosis. Stem Cell Res. Ther. 2022, 13, 90. [Google Scholar] [CrossRef]
- Moscat, J.; Diaz-Meco, M.T. p62 at the crossroads of autophagy, apoptosis, and cancer. Cell 2009, 137, 1001–1004. [Google Scholar] [CrossRef]
| Gene | Primer Sequence (5′–3′) |
|---|---|
| Gapdh | F: CAAGGCTGAGAACGGGAAG |
| R: TGAAGACGCCAGTGGACTC | |
| Circ-Spen | F: AGTCGATCCTCCAGTAGTGAC |
| R: CAGCCACTCCTCCTTCAGAC | |
| Circ-Hnrnpll | F: GAGATAGAGGAAAGGGTCGCC |
| R: CACAACAGATTCACAGAGCCC | |
| Circ-Eed | F: GATCCTCATAAAGCCAAGCCA |
| R: GCCAGGTTTCCAGCATACAA | |
| Circ-Strn3 | F: CGGAGTTCAGGGGATGGTAC |
| R: CTTGGTTCAGTTCTGTGCCATA | |
| Circ-Rabep1 | F: CGGGAAATAGCTGACTTAAGAAG |
| R: CCAATTCTGCTACCCGTTGC | |
| Actb | F: GCTGTGCTATGTTGCCCTAGACTTC |
| R: GGAACCGCTCATTGCCGATAGTG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Z.; Luo, B.; Peng, B.; Li, Y.; Hu, X.; Zhong, W.; Li, X.; Wang, P.; Zhu, X.; Zhang, R.; et al. Bone-Differentiation-Associated Circ-Spen Regulates Death of Mouse Bone Marrow Mesenchymal Stem Cells by Inhibiting Apoptosis and Promoting Autophagy. Int. J. Mol. Sci. 2024, 25, 3034. https://doi.org/10.3390/ijms25053034
Liang Z, Luo B, Peng B, Li Y, Hu X, Zhong W, Li X, Wang P, Zhu X, Zhang R, et al. Bone-Differentiation-Associated Circ-Spen Regulates Death of Mouse Bone Marrow Mesenchymal Stem Cells by Inhibiting Apoptosis and Promoting Autophagy. International Journal of Molecular Sciences. 2024; 25(5):3034. https://doi.org/10.3390/ijms25053034
Chicago/Turabian StyleLiang, Ziwen, Bingjie Luo, Bojia Peng, Yunchuan Li, Xueling Hu, Wenqiang Zhong, Xiaoyun Li, Panpan Wang, Xiaofeng Zhu, Ronghua Zhang, and et al. 2024. "Bone-Differentiation-Associated Circ-Spen Regulates Death of Mouse Bone Marrow Mesenchymal Stem Cells by Inhibiting Apoptosis and Promoting Autophagy" International Journal of Molecular Sciences 25, no. 5: 3034. https://doi.org/10.3390/ijms25053034
APA StyleLiang, Z., Luo, B., Peng, B., Li, Y., Hu, X., Zhong, W., Li, X., Wang, P., Zhu, X., Zhang, R., & Yang, L. (2024). Bone-Differentiation-Associated Circ-Spen Regulates Death of Mouse Bone Marrow Mesenchymal Stem Cells by Inhibiting Apoptosis and Promoting Autophagy. International Journal of Molecular Sciences, 25(5), 3034. https://doi.org/10.3390/ijms25053034






