The Role of Cannabidiol in Liver Disease: A Systemic Review
Abstract
1. Introduction
2. CBD and Liver Disease: Molecular Mechanisms
2.1. Hepatic Steatosis
2.2. Liver Fibrosis
2.3. Complications of Liver Fibrosis
2.4. Hepatocellular Carcinoma and Metastasis
2.5. Viral Infection
2.6. Chemical Liver Injury
2.7. Autoimmune Hepatitis
2.8. Liver Ischemia-Reperfusion Injury
2.9. Hepatic Glucose Metabolism and Diabetes
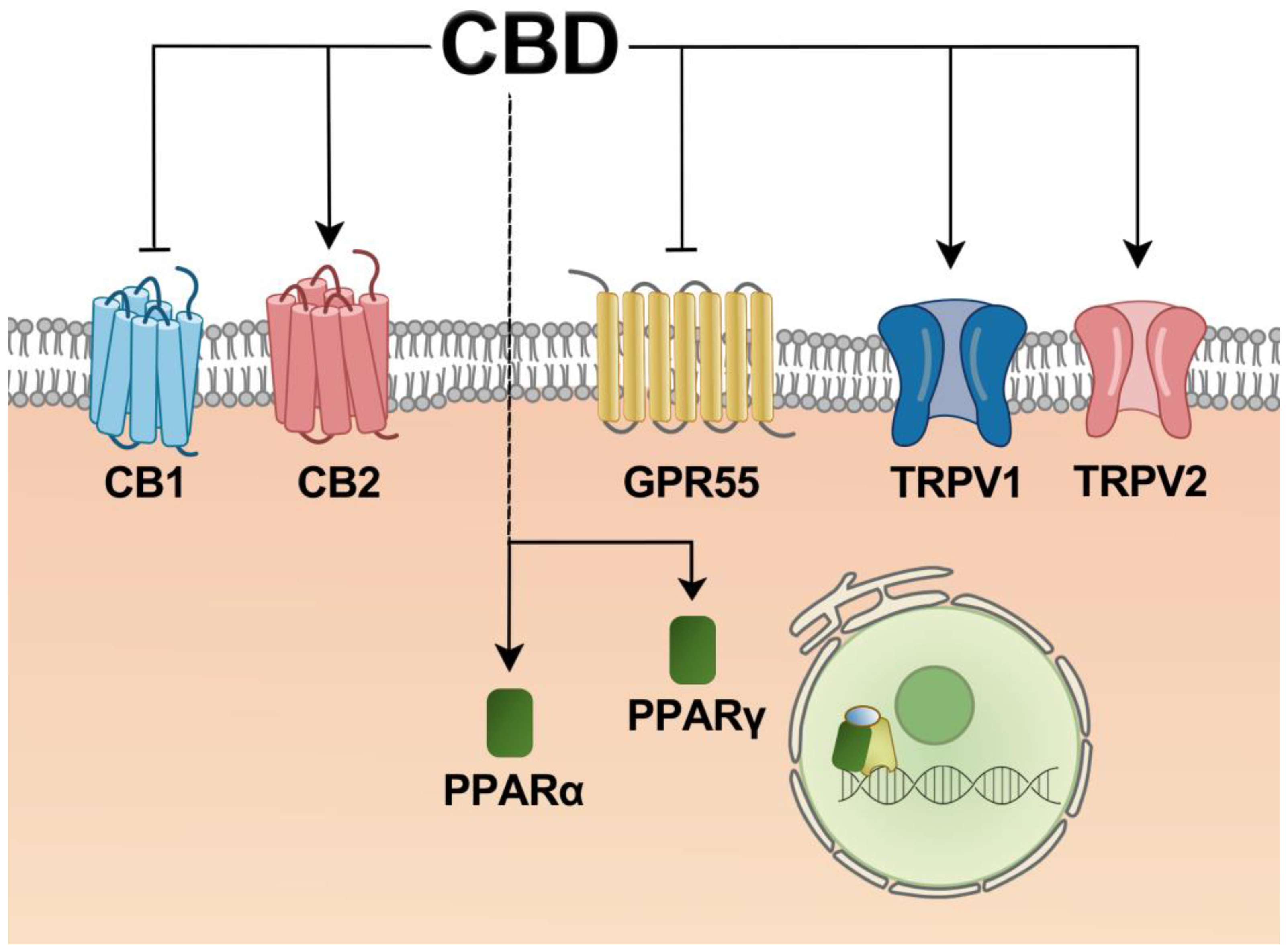
3. Molecular Pathways through which CBD May Affect Liver Disease
3.1. Cannabinoid Receptor 1
3.2. Cannabinoid Receptor 2
3.3. Peroxisome Proliferator-Activated Receptor
3.4. G Protein-Coupled Receptor 55
3.5. Transient Receptor Potential Channels
4. Liver Disease and CBD: Clinical Trials
4.1. MASLD and CBD
4.2. Cancer and CBD
4.3. Obesity and CBD
4.4. CBD Pharmacokinetics
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CBD | Cannabidiol |
| MASLD | Metabolic dysfunction-associated steatotic liver disease |
| NAFLD | non-alcoholic fatty liver disease |
| CB1 | cannabinoid receptor 1 |
| CB2 | cannabinoid receptor 2 |
| PPARs | peroxisome proliferator-activated receptors |
| TRPs | transient receptor potential channels |
| HCC | hepatocellular carcinoma |
| HFD | high-fat diet |
| TGF-β | transforming growth factor-β |
| PPAR γ | peroxisome proliferator-activated receptor γ |
| PPARα | peroxisome proliferator-activated receptor α |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| AMPK | 5′AMP-activated protein kinase |
| ACC | Acetyl-coenzyme A carboxylase |
| NLRP3 | NLR family pyrin domain containing 3 |
| TRPV1 | Transient receptor potential vanilloid 1 |
| TRPV2 | Transient receptor potential vanilloid 2 |
| TRPA1 | Transient receptor potential ankyrin 1 |
| TRPM8 | Transient receptor potential melastatin 8 |
| GPR55 | G protein-coupled receptor 55 |
| 2H2-CBD | deuterium-labelled cannabidiol |
| HSCs | hepatic stellate cells |
| GSDME | gasdermin E |
| COX-2 | cyclooxygenase-2 |
| LSECs | liver sinusoidal cells |
| AEA | anandamide |
| 2-AG | 2-arachidonoylglycerol |
| AA | arachidonic acid |
| LPI | L-α-lysophosphatidylinositol |
| Abn-CBD | abnormal cannabidiol |
| APAP | acetaminophen |
| NASH | non-alcoholic steatohepatitis |
| RA-FLS | Rheumatoid Arthritis Fibroblast-like Synoviocytes |
| LTs | liver tests |
References
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. Burden of liver diseases in the world. J. Hepatol. 2019, 70, 151–171. [Google Scholar] [CrossRef]
- Devarbhavi, H.; Asrani, S.K.; Arab, J.P.; Nartey, Y.A.; Pose, E.; Kamath, P.S. Global burden of liver disease: 2023 update. J. Hepatol. 2023, 79, 516–537. [Google Scholar] [CrossRef]
- Castillo-Arellano, J.; Canseco-Alba, A.; Cutler, S.J.; León, F. The Polypharmacological Effects of Cannabidiol. Molecules 2023, 28, 3271. [Google Scholar] [CrossRef]
- Britch, S.C.; Babalonis, S.; Walsh, S.L. Cannabidiol: Pharmacology and therapeutic targets. Psychopharmacology 2021, 238, 9–28. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, H.; Liu, Y.; Xu, Y.; Yang, B.; Li, H.; Chen, L. An overview on synthetic and biological activities of cannabidiol (CBD) and its derivatives. Bioorg. Chem. 2023, 140, 106810. [Google Scholar] [CrossRef]
- Alswat, K.A. The role of endocannabinoids system in fatty liver disease and therapeutic potentials. Saudi J. Gastroenterol. 2013, 19, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, H.M.; Osman, M.; Elshabrawy, O.; Abdallah, H.M.I.; Khairallah, A. AM-1241 CB2 Receptor Agonist Attenuates Inflammation, Apoptosis and Stimulate Progenitor Cells in Bile Duct Ligated Rats. Open Access Maced. J. Med. Sci. 2019, 7, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Chen, D.; Zhan, P.; Jiang, J. MDA19, a novel CB2 agonist, inhibits hepatocellular carcinoma partly through inactivation of AKT signaling pathway. Biol. Direct. 2019, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Mukhopadhyay, P.; Cao, Z.; Wang, H.; Feng, D.; Haskó, G.; Mechoulam, R.; Gao, B.; Pacher, P. Cannabidiol attenuates alcohol-induced liver steatosis, metabolic dysregulation, inflammation and neutrophil-mediated injury. Sci. Rep. 2017, 7, 12064. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J.; Kumar, V.; Shafi, S.; Khare, P.; Mahajan, N.; Bhadada, S.K.; Kondepudi, K.K.; Bhunia, R.K.; Kuhad, A.; Bishnoi, M. Combination of TRP channel dietary agonists induces energy expending and glucose utilizing phenotype in HFD-fed mice. Int. J. Obes. 2022, 46, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Neumann-Raizel, H.; Shilo, A.; Lev, S.; Mogilevsky, M.; Katz, B.; Shneor, D.; Shaul, Y.D.; Leffler, A.; Gabizon, A.; Karni, R.; et al. 2-APB and CBD-Mediated Targeting of Charged Cytotoxic Compounds into Tumor Cells Suggests the Involvement of TRPV2 Channels. Front. Pharmacol. 2019, 10, 1198. [Google Scholar] [CrossRef]
- Silvestri, C.; Paris, D.; Martella, A.; Melck, D.; Guadagnino, I.; Cawthorne, M.; Motta, A.; Di Marzo, V. Two non-psychoactive cannabinoids reduce intracellular lipid levels and inhibit hepatosteatosis. J. Hepatol. 2015, 62, 1382–1390. [Google Scholar] [CrossRef]
- Huang, Y.; Wan, T.; Pang, N.; Zhou, Y.; Jiang, X.; Li, B.; Gu, Y.; Huang, Y.; Ye, X.; Lian, H.; et al. Cannabidiol protects livers against nonalcoholic steatohepatitis induced by high-fat high cholesterol diet via regulating NF-κB and NLRP3 inflammasome pathway. J. Cell Physiol. 2019, 234, 21224–21234. [Google Scholar] [CrossRef] [PubMed]
- Mridha, A.R.; Wree, A.; Robertson, A.A.B.; Yeh, M.M.; Johnson, C.D.; Van Rooyen, D.M.; Haczeyni, F.; Teoh, N.C.; Savard, C.; Ioannou, G.N.; et al. NLRP3 inflammasome blockade reduces liver inflammation and fibrosis in experimental NASH in mice. J. Hepatol. 2017, 66, 1037–1046. [Google Scholar] [CrossRef]
- Barré, T.; Di Marzo, V.; Marcellin, F.; Burra, P.; Carrieri, P. Expanding Research on Cannabis-Based Medicines for Liver Steatosis: A Low-Risk High-Reward Way Out of the Present Deadlock? Cannabis Cannabinoid Res. 2023, 8, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Ben-Cnaan, E.; Permyakova, A.; Azar, S.; Hirsch, S.; Baraghithy, S.; Hinden, L.; Tam, J. The Metabolic Efficacy of a Cannabidiolic Acid (CBDA) Derivative in Treating Diet- and Genetic-Induced Obesity. Int. J. Mol. Sci. 2022, 23, 5610. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Rozenfeld, R.; Wu, D.; Devi, L.A.; Zhang, Z.; Cederbaum, A. Cannabidiol protects liver from binge alcohol-induced steatosis by mechanisms including inhibition of oxidative stress and increase in autophagy. Free Radic. Biol. Med. 2014, 68, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Gu, Y.; Huang, Y.; Zhou, Y.; Pang, N.; Luo, J.; Tang, Z.; Zhang, Z.; Yang, L. CBD Alleviates Liver Injuries in Alcoholics with High-Fat High-Cholesterol Diet Through Regulating NLRP3 Inflammasome-Pyroptosis Pathway. Front. Pharmacol. 2021, 12, 724747. [Google Scholar] [CrossRef] [PubMed]
- Mbaye, B.; Borentain, P.; Magdy Wasfy, R.; Alou, M.T.; Armstrong, N.; Mottola, G.; Meddeb, L.; Ranque, S.; Gérolami, R.; Million, M.; et al. Endogenous Ethanol and Triglyceride Production by Gut Pichia kudriavzevii, Candida albicans and Candida glabrata Yeasts in Non-Alcoholic Steatohepatitis. Cells 2022, 11, 3390. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Coker, O.O.; Chu, E.S.; Fu, K.; Lau, H.C.H.; Wang, Y.X.; Chan, A.W.H.; Wei, H.; Yang, X.; Sung, J.J.Y.; et al. Dietary cholesterol drives fatty liver-associated liver cancer by modulating gut microbiota and metabolites. Gut 2021, 70, 761–774. [Google Scholar] [CrossRef]
- Gan, L.; Feng, Y.; Du, B.; Fu, H.; Tian, Z.; Xue, G.; Yan, C.; Cui, X.; Zhang, R.; Cui, J.; et al. Bacteriophage targeting microbiota alleviates non-alcoholic fatty liver disease induced by high alcohol-producing Klebsiella pneumoniae. Nat. Commun. 2023, 14, 3215. [Google Scholar] [CrossRef] [PubMed]
- Gorelick, J.; Assa-Glazer, T.; Zandani, G.; Altberg, A.; Sela, N.; Nyska, A.; Madar, Z. THC and CBD affect metabolic syndrome parameters including microbiome in mice fed high fat-cholesterol diet. J. Cannabis Res. 2022, 4, 27. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Qu, F.; Chen, L.; Liu, C.; Zhang, M.; Ren, F.; Guo, H.; Zhang, H.; Ge, S.; Wu, C.; et al. SCFAs alleviated steatosis and inflammation in mice with NASH induced by MCD. J. Endocrinol. 2020, 245, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Assa-Glazer, T.; Gorelick, J.; Sela, N.; Nyska, A.; Bernstein, N.; Madar, Z. Cannabis Extracts Affected Metabolic Syndrome Parameters in Mice Fed High-Fat/Cholesterol Diet. Cannabis Cannabinoid Res. 2020, 5, 202–214. [Google Scholar] [CrossRef]
- Del Río, C.; Ruiz-Pino, F.; Prados, M.E.; Fiebich, B.L.; Tena-Sempere, M.; Muñoz, E. Cannabidiol markedly alleviates skin and liver fibrosis. Front. Pharmacol. 2022, 13, 981817. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Zhang, J.; Yin, H.; Li, S.; Xu, S.; Li, S. Cannabidiol alleviates perfluorooctane sulfonate-induced macrophage extracellular trap mediate inflammation and fibrosis in mice liver. Ecotoxicol. Environ. Saf. 2023, 263, 115374. [Google Scholar] [CrossRef]
- Patil, A.S.; Mahajan, U.B.; Agrawal, Y.O.; Patil, K.R.; Patil, C.R.; Ojha, S.; Sharma, C.; Goyal, S.N. Plant-derived natural therapeutics targeting cannabinoid receptors in metabolic syndrome and its complications: A review. Biomed. Pharmacother. 2020, 132, 110889. [Google Scholar] [CrossRef] [PubMed]
- Sunda, F.; Arowolo, A. A molecular basis for the anti-inflammatory and anti-fibrosis properties of cannabidiol. FASEB J. 2020, 34, 14083–14092. [Google Scholar] [CrossRef]
- Lim, M.P.; Devi, L.A.; Rozenfeld, R. Cannabidiol causes activated hepatic stellate cell death through a mechanism of endoplasmic reticulum stress-induced apoptosis. Cell Death Dis. 2011, 2, e170. [Google Scholar] [CrossRef]
- De Ternay, J.; Naassila, M.; Nourredine, M.; Louvet, A.; Bailly, F.; Sescousse, G.; Maurage, P.; Cottencin, O.; Carrieri, P.M.; Rolland, B. Therapeutic Prospects of Cannabidiol for Alcohol Use Disorder and Alcohol-Related Damages on the Liver and the Brain. Front. Pharmacol. 2019, 10, 627. [Google Scholar] [CrossRef]
- Magen, I.; Avraham, Y.; Ackerman, Z.; Vorobiev, L.; Mechoulam, R.; Berry, E.M. Cannabidiol ameliorates cognitive and motor impairments in mice with bile duct ligation. J. Hepatol. 2009, 51, 528–534. [Google Scholar] [CrossRef]
- Sadowska, O.; Baranowska-Kuczko, M.; Gromotowicz-Popławska, A.; Biernacki, M.; Kicman, A.; Malinowska, B.; Kasacka, I.; Krzyżewska, A.; Kozłowska, H. Cannabidiol Ameliorates Monocrotaline-Induced Pulmonary Hypertension in Rats. Int. J. Mol. Sci. 2020, 21, 7077. [Google Scholar] [CrossRef]
- Krzyżewska, A.; Baranowska-Kuczko, M.; Kasacka, I.; Kozłowska, H. Cannabidiol alleviates right ventricular fibrosis by inhibiting the transforming growth factor β pathway in monocrotaline-induced pulmonary hypertension in rats. Biochim. Biophys. Acta Mol. Basis Dis. 2023, 1869, 166753. [Google Scholar] [CrossRef]
- García-Martín, A.; Navarrete, C.; Garrido-Rodríguez, M.; Prados, M.E.; Caprioglio, D.; Appendino, G.; Muñoz, E. EHP-101 alleviates angiotensin II-induced fibrosis and inflammation in mice. Biomed. Pharmacother. 2021, 142, 112007. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, N.; Far, N.P.; Fakhr, S.S.; Faghihkhorasani, F.; Miraghel, S.A.; Chaleshtori, S.R.; Rezaei-Tazangi, F.; Beiranvand, S.; Baziyar, P.; Manavi, M.S.; et al. The endocannabinoid system, a new gatekeeper in the pharmacology of human hepatocellular carcinoma. Environ. Res. 2023, 228, 115914. [Google Scholar] [CrossRef] [PubMed]
- ElTelbany, A.; Khoudari, G.; Al-Khadra, Y.; McCullough, A.; Alkhouri, N. Lower Rates of Hepatocellular Carcinoma Observed Among Cannabis Users: A Population-Based Study. Cureus 2022, 14, e24576. [Google Scholar] [CrossRef] [PubMed]
- Ramer, R.; Hinz, B. Cannabinoids as Anticancer Drugs. Adv. Pharmacol. 2017, 80, 397–436. [Google Scholar] [CrossRef] [PubMed]
- Kis, B.; Ifrim, F.C.; Buda, V.; Avram, S.; Pavel, I.Z.; Antal, D.; Paunescu, V.; Dehelean, C.A.; Ardelean, F.; Diaconeasa, Z.; et al. Cannabidiol-from Plant to Human Body: A Promising Bioactive Molecule with Multi-Target Effects in Cancer. Int. J. Mol. Sci. 2019, 20, 5905. [Google Scholar] [CrossRef]
- Tajik, T.; Baghaei, K.; Moghadam, V.E.; Farrokhi, N.; Salami, S.A. Extracellular vesicles of cannabis with high CBD content induce anticancer signaling in human hepatocellular carcinoma. Biomed. Pharmacother. 2022, 152, 113209. [Google Scholar] [CrossRef] [PubMed]
- Viereckl, M.J.; Krutsinger, K.; Apawu, A.; Gu, J.; Cardona, B.; Barratt, D.; Han, Y. Cannabidiol and Cannabigerol Inhibit Cholangiocarcinoma Growth In Vitro via Divergent Cell Death Pathways. Biomolecules 2022, 12, 854. [Google Scholar] [CrossRef]
- Kosgodage, U.S.; Mould, R.; Henley, A.B.; Nunn, A.V.; Guy, G.W.; Thomas, E.L.; Inal, J.M.; Bell, J.D.; Lange, S. Cannabidiol (CBD) Is a Novel Inhibitor for Exosome and Microvesicle (EMV) Release in Cancer. Front. Pharmacol. 2018, 9, 889. [Google Scholar] [CrossRef] [PubMed]
- Akinloye, D.I.; Metibemu, D.S.; Shittu, M.T.; Lawal, M.A.; Olatunji, F.O.; Oyediran, M.A.; Akinloye, O.A. Cannabis sativa demonstrates anti-hepatocellular carcinoma potentials in animal model: In silico and in vivo studies of the involvement of Akt. J. Cannabis Res. 2023, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Buchtova, T.; Lukac, D.; Skrott, Z.; Chroma, K.; Bartek, J.; Mistrik, M. Drug-Drug Interactions of Cannabidiol with Standard-of-Care Chemotherapeutics. Int. J. Mol. Sci. 2023, 24, 2885. [Google Scholar] [CrossRef] [PubMed]
- Shangguan, F.; Zhou, H.; Ma, N.; Wu, S.; Huang, H.; Jin, G.; Wu, S.; Hong, W.; Zhuang, W.; Xia, H.; et al. A Novel Mechanism of Cannabidiol in Suppressing Hepatocellular Carcinoma by Inducing GSDME Dependent Pyroptosis. Front. Cell Dev. Biol. 2021, 9, 697832. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.; Kim, T.; Kwon, H.; Kim, J.K.; Park, Y.T.; Ham, J.; Kim, Y.J. Cannabidiol Enhances Cabozantinib-Induced Apoptotic Cell Death via Phosphorylation of p53 Regulated by ER Stress in Hepatocellular Carcinoma. Cancers 2023, 15, 3987. [Google Scholar] [CrossRef] [PubMed]
- Kargl, J.; Andersen, L.; Hasenöhrl, C.; Feuersinger, D.; Stančić, A.; Fauland, A.; Magnes, C.; El-Heliebi, A.; Lax, S.; Uranitsch, S.; et al. GPR55 promotes migration and adhesion of colon cancer cells indicating a role in metastasis. Br. J. Pharmacol. 2016, 173, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Sea, Y.L.; Gee, Y.J.; Lal, S.K.; Choo, W.S. Cannabis as antivirals. J. Appl. Microbiol. 2023, 134, lxac036. [Google Scholar] [CrossRef]
- Tomer, S.; Mu, W.; Suryawanshi, G.; Ng, H.; Wang, L.; Wennerberg, W.; Rezek, V.; Martin, H.; Chen, I.; Kitchen, S.; et al. Cannabidiol modulates expression of type I IFN response genes and HIV infection in macrophages. Front. Immunol. 2022, 13, 926696. [Google Scholar] [CrossRef]
- Nguyen, L.C.; Yang, D.; Nicolaescu, V.; Best, T.J.; Gula, H.; Saxena, D.; Gabbard, J.D.; Chen, S.N.; Ohtsuki, T.; Friesen, J.B.; et al. Cannabidiol inhibits SARS-CoV-2 replication through induction of the host ER stress and innate immune responses. Sci. Adv. 2022, 8, eabi6110. [Google Scholar] [CrossRef]
- Vallée, A. Cannabidiol and SARS-CoV-2 Infection. Front. Immunol. 2022, 13, 870787. [Google Scholar] [CrossRef]
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic. Br. J. Pharmacol. 2020, 177, 4967–4970. [Google Scholar] [CrossRef]
- Maor, Y.; Yu, J.; Kuzontkoski, P.M.; Dezube, B.J.; Zhang, X.; Groopman, J.E. Cannabidiol inhibits growth and induces programmed cell death in kaposi sarcoma-associated herpesvirus-infected endothelium. Genes. Cancer 2012, 3, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Lowe, H.I.; Toyang, N.J.; McLaughlin, W. Potential of Cannabidiol for the Treatment of Viral Hepatitis. Pharmacogn. Res. 2017, 9, 116–118. [Google Scholar] [CrossRef]
- Bickerstaff, C. The cost-effectiveness of novel direct acting antiviral agent therapies for the treatment of chronic hepatitis C. Expert. Rev. Pharmacoecon Outcomes Res. 2015, 15, 787–800. [Google Scholar] [CrossRef] [PubMed]
- Fouad, A.A.; Al-Mulhim, A.S.; Gomaa, W. Protective effect of cannabidiol against cadmium hepatotoxicity in rats. J. Trace Elem. Med. Biol. 2013, 27, 355–363. [Google Scholar] [CrossRef]
- Vilela, L.R.; Gomides, L.F.; David, B.A.; Antunes, M.M.; Diniz, A.B.; Moreira, F.d.A.; Menezes, G.B. Cannabidiol rescues acute hepatic toxicity and seizure induced by cocaine. Mediat. Inflamm. 2015, 2015, 523418. [Google Scholar] [CrossRef] [PubMed]
- Erukainure, O.L.; Matsabisa, M.G.; Salau, V.F.; Oyedemi, S.O.; Oyenihi, O.R.; Ibeji, C.U.; Islam, M.S. Cannabis sativa L. (var. indica) Exhibits Hepatoprotective Effects by Modulating Hepatic Lipid Profile and Mitigating Gluconeogenesis and Cholinergic Dysfunction in Oxidative Hepatic Injury. Front. Pharmacol. 2021, 12, 705402. [Google Scholar] [CrossRef]
- Mathur, K.; Vuppalanchi, V.; Gelow, K.; Vuppalanchi, R.; Lammert, C. Cannabidiol (CBD) Consumption and Perceived Impact on Extrahepatic Symptoms in Patients with Autoimmune Hepatitis. Dig. Dis. Sci. 2020, 65, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Hegde, V.L.; Nagarkatti, P.S.; Nagarkatti, M. Role of myeloid-derived suppressor cells in amelioration of experimental autoimmune hepatitis following activation of TRPV1 receptors by cannabidiol. PLoS ONE 2011, 6, e18281. [Google Scholar] [CrossRef]
- Fouad, A.A.; Jresat, I. Therapeutic potential of cannabidiol against ischemia/reperfusion liver injury in rats. Eur, J. Pharmacol. 2011, 670, 216–223. [Google Scholar] [CrossRef]
- Mukhopadhyay, P.; Rajesh, M.; Horváth, B.; Bátkai, S.; Park, O.; Tanchian, G.; Gao, R.Y.; Patel, V.; Wink, D.A.; Liaudet, L.; et al. Cannabidiol protects against hepatic ischemia/reperfusion injury by attenuating inflammatory signaling and response, oxidative/nitrative stress, and cell death. Free Radic. Biol. Med. 2011, 50, 1368–1381. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lin, C.; Jin, S.; Wang, H.; Wang, Y.; Du, X.; Hutchinson, M.R.; Zhao, H.; Fang, L.; Wang, X. The pharmacology and therapeutic role of cannabidiol in diabetes. Exploration 2023, 3, 20230047. [Google Scholar] [CrossRef]
- Booz, G.W. Cannabidiol as an emergent therapeutic strategy for lessening the impact of inflammation on oxidative stress. Free Radic. Biol. Med. 2011, 51, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.; Zeira, M.; Reich, S.; Har-Noy, M.; Mechoulam, R.; Slavin, S.; Gallily, R. Cannabidiol lowers incidence of diabetes in non-obese diabetic mice. Autoimmunity. 2006, 39, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Berk, K.; Konstantynowicz-Nowicka, K.; Charytoniuk, T.; Harasim-Symbor, E.; Chabowski, A. Distinct Effects of Cannabidiol on Sphingolipid Metabolism in Subcutaneous and Visceral Adipose Tissues Derived from High-Fat-Diet-Fed Male Wistar Rats. Int. J. Mol. Sci. 2022, 23, 5382. [Google Scholar] [CrossRef] [PubMed]
- Cinar, R.; Godlewski, G.; Liu, J.; Tam, J.; Jourdan, T.; Mukhopadhyay, B.; Harvey-White, J.; Kunos, G. Hepatic cannabinoid-1 receptors mediate diet-induced insulin resistance by increasing de novo synthesis of long-chain ceramides. Hepatology 2014, 59, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Jadoon, K.A.; Ratcliffe, S.H.; Barrett, D.A.; Thomas, E.L.; Stott, C.; Bell, J.D.; O’Sullivan, S.E.; Tan, G.D. Efficacy and Safety of Cannabidiol and Tetrahydrocannabivarin on Glycemic and Lipid Parameters in Patients with Type 2 Diabetes: A Randomized, Double-Blind, Placebo-Controlled, Parallel Group Pilot Study. Diabetes Care 2016, 39, 1777–1786. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.Y.; Alexa, K.; Cortes, M.; Schatzman-Bone, S.; Kim, A.J.; Mukhopadhyay, B.; Cinar, R.; Kunos, G.; North, T.E. Cannabinoid receptor signaling regulates liver development and metabolism. Development 2016, 143, 609–622. [Google Scholar] [CrossRef]
- Shi, D.; Zhan, X.; Yu, X.; Jia, M.; Zhang, Y.; Yao, J.; Hu, X.; Bao, Z. Inhibiting CB1 receptors improves lipogenesis in an in vitro non-alcoholic fatty liver disease model. Lipids Health Dis. 2014, 13, 173. [Google Scholar] [CrossRef]
- De Nunzio, V.; Carrieri, L.; Scavo, M.P.; Lippolis, T.; Cofano, M.; Caponio, G.R.; Tutino, V.; Rizzi, F.; Depalo, N.; Osella, A.R.; et al. Plasma-Derived Exosomes from NAFLD Patients Modulate the Cannabinoid Receptors’ Expression in Cultured HepaRG Cells. Int. J. Mol. Sci. 2023, 24, 1739. [Google Scholar] [CrossRef]
- Yang, L.; Tian, L.; Zhang, Z.; Zhou, X.; Ji, X.; Liu, F.; Dong, C.; Hou, L.; Zhao, X.; Chang, N.; et al. Cannabinoid Receptor 1/miR-30b-5p Axis Governs Macrophage NLRP3 Expression and Inflammasome Activation in Liver Inflammatory Disease. Mol. Ther. Nucleic Acids 2020, 20, 725–738. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.P.; Laudermilk, L.; Ware, J.; Rosa, T.; Mathews, K.; Gay, E.; Amato, G.; Maitra, R. Peripherally Selective CB1 Receptor Antagonist Improves Symptoms of Metabolic Syndrome in Mice. ACS Pharmacol. Transl. Sci. 2021, 4, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Dibba, P.; Li, A.; Cholankeril, G.; Iqbal, U.; Gadiparthi, C.; Khan, M.A.; Kim, D.; Ahmed, A. Mechanistic Potential and Therapeutic Implications of Cannabinoids in Nonalcoholic Fatty Liver Disease. Medicines 2018, 5, 47. [Google Scholar] [CrossRef] [PubMed]
- Jorgačević, B.; Vučević, D.; Samardžić, J.; Mladenović, D.; Vesković, M.; Vukićević, D.; Ješić, R.; Radosavljević, T. The Effect of CB1 Antagonism on Hepatic Oxidative/Nitrosative Stress and Inflammation in Nonalcoholic Fatty Liver Disease. Curr. Med. Chem. 2021, 28, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Jourdan, T.; Nicoloro, S.M.; Zhou, Z.; Shen, Y.; Liu, J.; Coffey, N.J.; Cinar, R.; Godlewski, G.; Gao, B.; Aouadi, M.; et al. Decreasing CB1 receptor signaling in Kupffer cells improves insulin sensitivity in obese mice. Mol. Metab. 2017, 6, 1517–1528. [Google Scholar] [CrossRef]
- Liu, J.; Godlewski, G.; Jourdan, T.; Liu, Z.; Cinar, R.; Xiong, K.; Kunos, G. Cannabinoid-1 Receptor Antagonism Improves Glycemic Control and Increases Energy Expenditure Through Sirtuin-1/Mechanistic Target of Rapamycin Complex 2 and 5′Adenosine Monophosphate-Activated Protein Kinase Signaling. Hepatology 2019, 69, 1535–1548. [Google Scholar] [CrossRef] [PubMed]
- Drori, A.; Gammal, A.; Azar, S.; Hinden, L.; Hadar, R.; Wesley, D.; Nemirovski, A.; Szanda, G.; Salton, M.; Tirosh, B.; et al. CB1R regulates soluble leptin receptor levels via CHOP, contributing to hepatic leptin resistance. Elife 2020, 9, e60771. [Google Scholar] [CrossRef]
- Moreira, F.P.; Wiener, C.D.; Oliveira, J.F.; Souza, L.D.M.; da Silva, R.A.; Portela, L.V.; Lara, D.R.; Jansen, K.; Oses, J.P. Gender differences of cannabis smoking on serum leptin levels: Population-based study. Braz. J. Psychiatry 2018, 40, 216–219. [Google Scholar] [CrossRef]
- Wagner, J.D.; Zhang, L.; Kavanagh, K.; Ward, G.M.; Chin, J.E.; Hadcock, J.R.; Auerbach, B.J.; Harwood, H.J. A selective cannabinoid-1 receptor antagonist, PF-95453, reduces body weight and body fat to a greater extent than pair-fed controls in obese monkeys. J. Pharmacol. Exp. Ther. 2010, 335, 103–113. [Google Scholar] [CrossRef]
- Riggs, P.K.; Vaida, F.; Rossi, S.S.; Sorkin, L.S.; Gouaux, B.; Grant, I.; Ellis, R.J. A pilot study of the effects of cannabis on appetite hormones in HIV-infected adult men. Brain Res. 2012, 1431, 46–52. [Google Scholar] [CrossRef]
- Muniyappa, R.; Sable, S.; Ouwerkerk, R.; Mari, A.; Gharib, A.M.; Walter, M.; Courville, A.; Hall, G.; Chen, K.Y.; Volkow, N.D.; et al. Metabolic effects of chronic cannabis smoking. Diabetes Care 2013, 36, 2415–2422. [Google Scholar] [CrossRef]
- Tian, L.; Li, W.; Yang, L.; Chang, N.; Fan, X.; Ji, X.; Xie, J.; Yang, L.; Li, L. Cannabinoid Receptor 1 Participates in Liver Inflammation by Promoting M1 Macrophage Polarization via RhoA/NF-κB p65 and ERK1/2 Pathways, Respectively, in Mouse Liver Fibrogenesis. Front. Immunol. 2017, 8, 1214. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, Q.; Liang, G.; Franks, T.; Boucher, M.; Bence, K.K.; Lu, M.; Castorena, C.M.; Zhao, S.; Elmquist, J.K.; et al. Cannabinoid receptor 1 signaling in hepatocytes and stellate cells does not contribute to NAFLD. J. Clin. Investig. 2021, 131, e152242. [Google Scholar] [CrossRef]
- Patsenker, E.; Stoll, M.; Millonig, G.; Agaimy, A.; Wissniowski, T.; Schneider, V.; Mueller, S.; Brenneisen, R.; Seitz, H.K.; Ocker, M.; et al. Cannabinoid receptor type I modulates alcohol-induced liver fibrosis. Mol. Med. 2011, 17, 1285–1294. [Google Scholar] [CrossRef] [PubMed]
- Jeong, W.I.; Osei-Hyiaman, D.; Park, O.; Liu, J.; Bátkai, S.; Mukhopadhyay, P.; Horiguchi, N.; Harvey-White, J.; Marsicano, G.; Lutz, B.; et al. Paracrine activation of hepatic CB1 receptors by stellate cell-derived endocannabinoids mediates alcoholic fatty liver. Cell Metab. 2008, 7, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Amato, G.S.; Manke, A.; Harris, D.L.; Wiethe, R.W.; Vasukuttan, V.; Snyder, R.W.; Lefever, T.W.; Cortes, R.; Zhang, Y.; Wang, S.; et al. Blocking Alcoholic Steatosis in Mice with a Peripherally Restricted Purine Antagonist of the Type 1 Cannabinoid Receptor. J. Med. Chem. 2018, 61, 4370–4385. [Google Scholar] [CrossRef] [PubMed]
- Roger, C.; Buch, C.; Muller, T.; Leemput, J.; Demizieux, L.; Passilly-Degrace, P.; Cinar, R.; Iyer, M.R.; Kunos, G.; Vergès, B.; et al. Simultaneous Inhibition of Peripheral CB1R and iNOS Mitigates Obesity-Related Dyslipidemia Through Distinct Mechanisms. Diabetes 2020, 69, 2120–2132. [Google Scholar] [CrossRef] [PubMed]
- van der Poorten, D.; Shahidi, M.; Tay, E.; Sesha, J.; Tran, K.; McLeod, D.; Milliken, J.S.; Ho, V.; Hebbard, L.W.; Douglas, M.W.; et al. Hepatitis C virus induces the cannabinoid receptor 1. PLoS ONE 2010, 5, e12841. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Gautam, S.; Aseer, K.R.; Kim, J.; Chandrasekaran, P.; Mazucanti, C.H.; Ghosh, P.; O’Connell, J.F.; Doyle, M.E.; Appleton, A.; et al. Hepatocyte cannabinoid 1 receptor nullification alleviates toxin-induced liver damage via NF-κB signaling. Cell Death Dis. 2020, 11, 1044. [Google Scholar] [CrossRef] [PubMed]
- Müller, G.A.; Wied, S.; Herling, A.W. Analysis of Direct Effects of the CB1 Receptor Antagonist Rimonabant on Fatty Acid Oxidation and Glycogenolysis in Liver and Muscle Cells in vitro. Biochemistry 2019, 84, 954–962. [Google Scholar] [CrossRef] [PubMed]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.; Denovan-Wright, E.M. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br. J. Pharmacol. 2015, 172, 4790–4805. [Google Scholar] [CrossRef]
- Louvet, A.; Teixeira-Clerc, F.; Chobert, M.N.; Deveaux, V.; Pavoine, C.; Zimmer, A.; Pecker, F.; Mallat, A.; Lotersztajn, S. Cannabinoid CB2 receptors protect against alcoholic liver disease by regulating Kupffer cell polarization in mice. Hepatology 2011, 54, 1217–1226. [Google Scholar] [CrossRef]
- Deveaux, V.; Cadoudal, T.; Ichigotani, Y.; Teixeira-Clerc, F.; Louvet, A.; Manin, S.; Nhieu, J.T.; Belot, M.P.; Zimmer, A.; Even, P.; et al. Cannabinoid CB2 receptor potentiates obesity-associated inflammation, insulin resistance and hepatic steatosis. PLoS ONE 2009, 4, e5844. [Google Scholar] [CrossRef] [PubMed]
- Agudo, J.; Martin, M.; Roca, C.; Molas, M.; Bura, A.S.; Zimmer, A.; Bosch, F.; Maldonado, R. Deficiency of CB2 cannabinoid receptor in mice improves insulin sensitivity but increases food intake and obesity with age. Diabetologia 2010, 53, 2629–2640. [Google Scholar] [CrossRef]
- De Gottardi, A.; Spahr, L.; Ravier-Dall’Antonia, F.; Hadengue, A. Cannabinoid receptor 1 and 2 agonists increase lipid accumulation in hepatocytes. Liver Int. 2010, 30, 1482–1489. [Google Scholar] [CrossRef] [PubMed]
- Kamikubo, R.; Kai, K.; Tsuji-Naito, K.; Akagawa, M. β-Caryophyllene attenuates palmitate-induced lipid accumulation through AMPK signaling by activating CB2 receptor in human HepG2 hepatocytes. Mol. Nutr. Food Res. 2016, 60, 2228–2242. [Google Scholar] [CrossRef]
- Varga, Z.V.; Matyas, C.; Erdelyi, K.; Cinar, R.; Nieri, D.; Chicca, A.; Nemeth, B.T.; Paloczi, J.; Lajtos, T.; Corey, L.; et al. β-Caryophyllene protects against alcoholic steatohepatitis by attenuating inflammation and metabolic dysregulation in mice. Br. J. Pharmacol. 2018, 175, 320–334. [Google Scholar] [CrossRef]
- Wang, H.Q.; Wan, Z.; Zhang, Q.; Su, T.; Yu, D.; Wang, F.; Zhang, C.; Li, W.; Xu, D.; Zhang, H. Schisandrin B targets cannabinoid 2 receptor in Kupffer cell to ameliorate CCl4-induced liver fibrosis by suppressing NF-κB and p38 MAPK pathway. Phytomedicine 2022, 98, 153960. [Google Scholar] [CrossRef]
- Denaës, T.; Lodder, J.; Chobert, M.N.; Denaës, T.; Lodder, J.; Chobert, M.N.; Ruiz, I.; Pawlotsky, J.M.; Lotersztajn, S.; Teixeira-Clerc, F. The Cannabinoid Receptor 2 Protects Against Alcoholic Liver Disease Via a Macrophage Autophagy-Dependent Pathway. Sci. Rep. 2016, 6, 28806. [Google Scholar] [CrossRef] [PubMed]
- Trebicka, J.; Racz, I.; Siegmund, S.V.; Cara, E.; Granzow, M.; Schierwagen, R.; Klein, S.; Wojtalla, A.; Hennenberg, M.; Huss, S.; et al. Role of cannabinoid receptors in alcoholic hepatic injury: Steatosis and fibrogenesis are increased in CB2 receptor-deficient mice and decreased in CB1 receptor knockouts. Liver Int. 2011, 31, 860–870. [Google Scholar] [CrossRef]
- Wu, Y.; Ma, R.; Long, C.; Shu, Y.; He, P.; Zhou, Y.; Xiang, Y.; Wang, Y. The protective effect of cannabinoid type II receptor agonist AM1241 on ConA-induced liver injury in mice via mitogen-activated protein kinase signalling pathway. Int. J. Immunopathol. Pharmacol. 2021, 35, 20587384211035251. [Google Scholar] [CrossRef]
- Coppola, N.; Zampino, R.; Bellini, G.; Macera, M.; Marrone, A.; Pisaturo, M.; Boemio, A.; Nobili, B.; Pasquale, G.; Maione, S.; et al. Association between a polymorphism in cannabinoid receptor 2 and severe necroinflammation in patients with chronic hepatitis C. Clin. Gastroenterol. Hepatol. 2014, 12, 334–340. [Google Scholar] [CrossRef]
- Sagnelli, C.; Uberti-Foppa, C.; Hasson, H.; Bellini, G.; Minichini, C.; Salpietro, S.; Messina, E.; Barbanotti, D.; Merli, M.; Punzo, F.; et al. Cannabinoid receptor 2-63 RR variant is independently associated with severe necroinflammation in HIV/HCV coinfected patients. PLoS ONE 2017, 12, e0181890. [Google Scholar] [CrossRef]
- Gui, H.; Liu, X.; Wang, Z.W.; He, D.Y.; Su, D.F.; Dai, S.M. Expression of cannabinoid receptor 2 and its inhibitory effects on synovial fibroblasts in rheumatoid arthritis. Rheumatology 2014, 53, 802–809. [Google Scholar] [CrossRef]
- Thomas, A.; Baillie, G.L.; Phillips, A.M.; Razdan, R.K.; Ross, R.A.; Pertwee, R.G. Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br. J. Pharmacol. 2007, 150, 613–623. [Google Scholar] [CrossRef]
- Pawlak, M.; Lefebvre, P.; Staels, B. Molecular mechanism of PPARα action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J. Hepatol. 2015, 62, 720–733. [Google Scholar] [CrossRef] [PubMed]
- Abdelmegeed, M.A.; Yoo, S.H.; Henderson, L.E.; Gonzalez, F.J.; Woodcroft, K.J.; Song, B.J. PPARalpha expression protects male mice from high fat-induced nonalcoholic fatty liver. J. Nutr. 2011, 141, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Montagner, A.; Polizzi, A.; Fouché, E.; Ducheix, S.; Lippi, Y.; Lasserre, F.; Barquissau, V.; Régnier, M.; Lukowicz, C.; Benhamed, F.; et al. Liver PPARα is crucial for whole-body fatty acid homeostasis and is protective against NAFLD. Gut 2016, 65, 1202–1214. [Google Scholar] [CrossRef] [PubMed]
- Gervois, P.; Kleemann, R.; Pilon, A.; Percevault, F.; Koenig, W.; Staels, B.; Kooistra, T. Global suppression of IL-6-induced acute phase response gene expression after chronic in vivo treatment with the peroxisome proliferator-activated receptor-alpha activator fenofibrate. J. Biol. Chem. 2004, 279, 16154–16160. [Google Scholar] [CrossRef] [PubMed]
- Toyama, T.; Nakamura, H.; Harano, Y.; Yamauchi, N.; Morita, A.; Kirishima, T.; Minami, M.; Itoh, Y.; Okanoue, T. PPARalpha ligands activate antioxidant enzymes and suppress hepatic fibrosis in rats. Biochem. Biophys. Res. Commun. 2004, 324, 697–704. [Google Scholar] [CrossRef]
- Chen, L.; Li, L.; Chen, J.; Li, L.; Zheng, Z.; Ren, J.; Qiu, Y. Oleoylethanolamide, an endogenous PPAR-α ligand, attenuates liver fibrosis targeting hepatic stellate cells. Oncotarget 2015, 6, 42530–42540. [Google Scholar] [CrossRef]
- Scandiffio, R.; Bonzano, S.; Cottone, E.; Shrestha, S.; Bossi, S.; De Marchis, S.; Maffei, M.E.; Bovolin, P. Beta-Caryophyllene Modifies Intracellular Lipid Composition in a Cell Model of Hepatic Steatosis by Acting through CB2 and PPAR Receptors. Int. J. Mol. Sci. 2023, 24, 6060. [Google Scholar] [CrossRef]
- Hoekstra, M.; Kruijt, J.K.; Van Eck, M.; Van Berkel, T.J. Specific gene expression of ATP-binding cassette transporters and nuclear hormone receptors in rat liver parenchymal, endothelial, and Kupffer cells. J. Biol. Chem. 2003, 278, 25448–25453. [Google Scholar] [CrossRef]
- Lee, C.H.; Olson, P.; Hevener, A.; Mehl, I.; Chong, L.W.; Olefsky, J.M.; Gonzalez, F.J.; Ham, J.; Kang, H.; Peters, J.M.; et al. PPARdelta regulates glucose metabolism and insulin sensitivity. Proc. Natl. Acad. Sci. USA 2006, 103, 3444–3449. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hatano, B.; Zhao, M.; Yen, C.C.; Kang, K.; Reilly, S.M.; Gangl, M.R.; Gorgun, C.; Balschi, J.A.; Ntambi, J.M.; et al. Role of peroxisome proliferator-activated receptor {delta}/{beta} in hepatic metabolic regulation. J. Biol. Chem. 2011, 286, 1237–1247. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, L.M.; Boekschoten, M.V.; Desvergne, B.; Müller, M.; Kersten, S. Transcriptional profiling reveals divergent roles of PPARalpha and PPARbeta/delta in regulation of gene expression in mouse liver. Physiol. Genom. 2010, 41, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhang, S.; Chu, E.S.; Go, M.Y.; Lau, R.H.; Zhao, J.; Wu, C.W.; Tong, L.; Zhao, J.; Poon, T.C.; et al. Peroxisome proliferator-activated receptors gamma reverses hepatic nutritional fibrosis in mice and suppresses activation of hepatic stellate cells in vitro. Int. J. Biochem. Cell Biol. 2010, 42, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Deng, F.; Zhang, Y.; Zhang, X.; Chen, J.; Jiang, Y. PPARγ attenuates hepatic inflammation and oxidative stress of non-alcoholic steatohepatitis via modulating the miR-21-5p/SFRP5 pathway. Mol. Med. Rep. 2021, 24, 823. [Google Scholar] [CrossRef] [PubMed]
- Gavrilova, O.; Haluzik, M.; Matsusue, K.; Cutson, J.J.; Johnson, L.; Dietz, K.R.; Nicol, C.J.; Vinson, C.; Gonzalez, F.J.; Reitman, M.L. Liver peroxisome proliferator-activated receptor gamma contributes to hepatic steatosis, triglyceride clearance, and regulation of body fat mass. J. Biol. Chem. 2003, 278, 34268–34276. [Google Scholar] [CrossRef]
- Boelsterli, U.A.; Bedoucha, M. Toxicological consequences of altered peroxisome proliferator-activated receptor gamma (PPARgamma) expression in the liver: Insights from models of obesity and type 2 diabetes. Biochem. Pharmacol. 2002, 63, 1–10. [Google Scholar] [CrossRef]
- Rahimian, R.; Masih-Khan, E.; Lo, M.; van Breemen, C.; McManus, B.M.; Dubé, G.P. Hepatic over-expression of peroxisome proliferator activated receptor gamma2 in the ob/ob mouse model of non-insulin dependent diabetes mellitus. Mol. Cell Biochem. 2001, 224, 29–37. [Google Scholar] [CrossRef]
- Pettinelli, P.; Videla, L.A. Up-regulation of PPAR-gamma mRNA expression in the liver of obese patients: An additional reinforcing lipogenic mechanism to SREBP-1c induction. J. Clin. Endocrinol. Metab. 2011, 96, 1424–1430. [Google Scholar] [CrossRef] [PubMed]
- Azar, S.; Udi, S.; Drori, A.; Hadar, R.; Nemirovski, A.; Vemuri, K.V.; Miller, M.; Sherill-Rofe, D.; Arad, Y.; Gur-Wahnon, D.; et al. Reversal of diet-induced hepatic steatosis by peripheral CB1 receptor blockade in mice is p53/miRNA-22/SIRT1/PPARα dependent. Mol. Metab. 2020, 42, 101087. [Google Scholar] [CrossRef] [PubMed]
- Khosropoor, S.; Alavi, M.S.; Etemad, L.; Roohbakhsh, A. Cannabidiol goes nuclear: The role of PPARγ. Phytomedicine 2023, 114, 154771. [Google Scholar] [CrossRef] [PubMed]
- Esposito, G.; Scuderi, C.; Valenza, M.; Togna, G.I.; Latina, V.; De Filippis, D.; Cipriano, M.; Carratù, M.R.; Iuvone, T.; Steardo, L.; et al. Cannabidiol reduces Aβ-induced neuroinflammation and promotes hippocampal neurogenesis through PPARγ involvement. PLoS ONE 2011, 6, e28668. [Google Scholar] [CrossRef] [PubMed]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef]
- Simcocks, A.C.; O’Keefe, L.; Jenkin, K.A.; Mathai, M.L.; Hryciw, D.H.; McAinch, A.J. A potential role for GPR55 in the regulation of energy homeostasis. Drug Discov. Today 2014, 19, 1145–1151. [Google Scholar] [CrossRef]
- McCloskey, A.G.; Miskelly, M.G.; Moore, C.B.T.; Nesbit, M.A.; Christie, K.A.; Owolabi, A.I.; Flatt, P.R.; McKillop, A.M. CRISPR/Cas9 gene editing demonstrates metabolic importance of GPR55 in the modulation of GIP release and pancreatic beta cell function. Peptides 2020, 125, 170251. [Google Scholar] [CrossRef]
- Meadows, A.; Lee, J.H.; Wu, C.S.; Wei, Q.; Pradhan, G.; Yafi, M.; Lu, H.C.; Sun, Y. Deletion of G-protein-coupled receptor 55 promotes obesity by reducing physical activity. Int. J. Obes. 2016, 40, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Lipina, C.; Walsh, S.K.; Mitchell, S.E.; Speakman, J.R.; Wainwright, C.L.; Hundal, H.S. GPR55 deficiency is associated with increased adiposity and impaired insulin signaling in peripheral metabolic tissues. FASEB J. 2019, 33, 1299–1312. [Google Scholar] [CrossRef] [PubMed]
- Vong, C.T.; Tseng, H.H.L.; Kwan, Y.W.; Lee, S.M.; Hoi, M.P.M. Novel protective effect of O-1602 and abnormal cannabidiol, GPR55 agonists, on ER stress-induced apoptosis in pancreatic β-cells. Biomed. Pharmacother. 2019, 111, 1176–1186. [Google Scholar] [CrossRef]
- Fondevila, M.F.; Fernandez, U.; Gonzalez-Rellan, M.J.; Da Silva Lima, N.; Buque, X.; Gonzalez-Rodriguez, A.; Alonso, C.; Iruarrizaga-Lejarreta, M.; Delgado, T.C.; Varela-Rey, M.; et al. The L-α-Lysophosphatidylinositol/G Protein-Coupled Receptor 55 System Induces the Development of Nonalcoholic Steatosis and Steatohepatitis. Hepatology 2021, 73, 606–624. [Google Scholar] [CrossRef]
- Moreno-Navarrete, J.M.; Catalán, V.; Whyte, L.; Díaz-Arteaga, A.; Vázquez-Martínez, R.; Rotellar, F.; Guzmán, R.; Gómez-Ambrosi, J.; Pulido, M.R.; Russell, W.R.; et al. The L-α-lysophosphatidylinositol/GPR55 system and its potential role in human obesity. Diabetes 2012, 61, 281–291. [Google Scholar] [CrossRef]
- Díaz-Arteaga, A.; Vázquez, M.J.; Vazquez-Martínez, R.; Pulido, M.R.; Suarez, J.; Velásquez, D.A.; López, M.; Ross, R.A.; de Fonseca, F.R.; Bermudez-Silva, F.J.; et al. The atypical cannabinoid O-1602 stimulates food intake and adiposity in rats. Diabetes Obes. Metab. 2012, 14, 234–243. [Google Scholar] [CrossRef]
- Kargl, J.; Balenga, N.; Parzmair, G.P.; Brown, A.J.; Heinemann, A.; Waldhoer, M. The cannabinoid receptor CB1 modulates the signaling properties of the lysophosphatidylinositol receptor GPR55. J. Biol. Chem. 2012, 287, 44234–44248. [Google Scholar] [CrossRef]
- Balenga, N.A.; Aflaki, E.; Kargl, J.; Platzer, W.; Schröder, R.; Blättermann, S.; Kostenis, E.; Brown, A.J.; Heinemann, A.; Waldhoer, M. GPR55 regulates cannabinoid 2 receptor-mediated responses in human neutrophils. Cell Res. 2011, 21, 1452–1469, Erratum in Cell Res. 2011, 21, 1641. [Google Scholar] [CrossRef] [PubMed]
- Shore, D.M.; Reggio, P.H. The therapeutic potential of orphan GPCRs, GPR35 and GPR55. Front. Pharmacol. 2015, 6, 69. [Google Scholar] [CrossRef] [PubMed]
- Wargent, E.T.; Kepczynska, M.; Zaibi, M.S.; Hislop, D.C.; Arch, J.R.S.; Stocker, C.J. High fat-fed GPR55 null mice display impaired glucose tolerance without concomitant changes in energy balance or insulin sensitivity but are less responsive to the effects of the cannabinoids rimonabant or Δ(9)-tetrahydrocannabivarin on weight gain. PeerJ 2020, 8, e9811. [Google Scholar] [CrossRef] [PubMed]
- Marichal-Cancino, B.A.; Fajardo-Valdez, A.; Ruiz-Contreras, A.E.; Mendez-Díaz, M.; Prospero-García, O. Advances in the Physiology of GPR55 in the Central Nervous System. Curr. Neuropharmacol. 2017, 15, 771–778. [Google Scholar] [CrossRef] [PubMed]
- McCloskey, A.G.; Miskelly, M.G.; Lafferty, R.A.; Flatt, P.R.; McKillop, A.M. Antidiabetic actions of GPR55 agonist Abn-CBD and sitagliptin in obese-diabetic high fat fed mice. Biochem. Pharmacol. 2023, 208, 115398. [Google Scholar] [CrossRef] [PubMed]
- Li, H. TRP Channel Classification. Adv. Exp. Med. Biol. 2017, 976, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Samanta, A.; Hughes, T.E.T.; Moiseenkova-Bell, V.Y. Transient Receptor Potential (TRP) Channels. Subcell. Biochem. 2018, 87, 141–165. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Liu, F.; Du, S.; Li, M.; Wu, T.; Tan, X.; Cheng, W. Mechanism for Regulation of Melanoma Cell Death via Activation of Thermo-TRPV4 and TRPV2. J. Oncol. 2019, 2019, 7362875. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.Y.; Zhang, Y.Q.; Zeng, Y.Z.; Zhu, J.L.; Chen, H.; Wei, X.L.; Liu, L.J. TRPC1 inhibits the proliferation and migration of estrogen receptor-positive Breast cancer and gives a better prognosis by inhibiting the PI3K/AKT pathway. Breast Cancer Res. Treat. 2020, 182, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Rychkov, G.Y.; Barritt, G.J. Expression and function of TRP channels in liver cells. Adv. Exp. Med. Biol. 2011, 704, 667–686. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Yang, G.; Sun, T.; Jie, T.; Zhu, C.; Yu, H.; Cheng, Y.; Yang, Z.; Xu, M.; Jiang, Y.; et al. Capsaicin receptor TRPV1 maintains quiescence of hepatic stellate cells in the liver via recruitment of SARM1. J. Hepatol. 2023, 78, 805–819. [Google Scholar] [CrossRef] [PubMed]
- Qian, K.; Lei, X.; Liu, G.; Fang, Y.; Xie, C.; Wu, X.; Liu, Q.; Liu, G.; Cao, Z.; Zhang, J.; et al. Transient Receptor Potential Vanilloid-1 (TRPV1) Alleviates Hepatic Fibrosis via TGF-β Signaling. Dis. Markers 2022, 2022, 3100943. [Google Scholar] [CrossRef]
- Xie, C.; Liu, G.; Li, M.; Fang, Y.; Qian, K.; Tang, Y.; Wu, X.; Lei, X.; Li, X.; Liu, Q.; et al. Targeting TRPV1 on cellular plasticity regulated by Ovol 2 and Zeb 1 in hepatocellular carcinoma. Biomed. Pharmacother. 2019, 118, 109270. [Google Scholar] [CrossRef]
- Wang, T.; Chen, Y.; Li, Y.; Wang, Z.; Qiu, C.; Yang, D.; Chen, K. TRPV1 Protect against Hyperglycemia and Hyperlipidemia Induced Liver Injury via OPA1 in Diabetes. Tohoku J. Exp. Med. 2022, 256, 131–139. [Google Scholar] [CrossRef]
- Li, L.; Chen, J.; Ni, Y.; Feng, X.; Zhao, Z.; Wang, P.; Sun, J.; Yu, H.; Yan, Z.; Liu, D.; et al. TRPV1 activation prevents nonalcoholic fatty liver through UCP2 upregulation in mice. Pflugers Arch. 2012, 463, 727–732. [Google Scholar] [CrossRef]
- Lacerda, J.T.; Gomes, P.R.L.; Zanetti, G.; Mezzalira, N.; Lima, O.G.; de Assis, L.V.M.; Guler, A.; Castrucci, A.M.; Moraes, M.N. Lack of TRPV1 Channel Modulates Mouse Gene Expression and Liver Proteome with Glucose Metabolism Changes. Int. J. Mol. Sci. 2022, 23, 7014. [Google Scholar] [CrossRef]
- Baskaran, P.; Nazminia, K.; Frantz, J.; O’Neal, J.; Thyagarajan, B. Mice lacking endogenous TRPV1 express reduced levels of thermogenic proteins and are susceptible to diet-induced obesity and metabolic dysfunction. FEBS Lett. 2021, 595, 1768–1781. [Google Scholar] [CrossRef]
- Wang, W.; Liu, P.; Zhang, Y.; Yan, L.; Zhu, M.X.; Wang, J.; Yu, Y. Expression and functions of transient receptor potential channels in liver diseases. Acta Pharm. Sin. B. 2023, 13, 445–459. [Google Scholar] [CrossRef]
- Å Nilsson, J.L.; Mallet, C.; Shionoya, K.; Blomgren, A.; Sundin, A.P.; Grundemar, L.; Boudieu, L.; Blomqvist, A.; Eschalier, A.; Nilsson, U.J.; et al. Paracetamol analogues conjugated by FAAH induce TRPV1-mediated antinociception without causing acute liver toxicity. Eur. J. Med. Chem. 2021, 213, 113042. [Google Scholar] [CrossRef]
- Costa, B.; Giagnoni, G.; Franke, C.; Trovato, A.E.; Colleoni, M. Vanilloid TRPV1 receptor mediates the antihyperalgesic effect of the nonpsychoactive cannabinoid, cannabidiol, in a rat model of acute inflammation. Br. J. Pharmacol. 2004, 143, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Cao, X.; Fang, Y.; Liu, G.; Xie, C.; Qian, K.; Lei, X.; Cao, Z.; Du, H.; Cheng, X.; et al. Transient receptor potential vanilloid-type 2 targeting on stemness in liver cancer. Biomed. Pharmacother. 2018, 105, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Chianese, G.; Lopatriello, A.; Schiano-Moriello, A.; Caprioglio, D.; Mattoteia, D.; Benetti, E.; Ciceri, D.; Arnoldi, L.; De Combarieu, E.; Vitale, R.M.; et al. Cannabitwinol, a Dimeric Phytocannabinoid from Hemp, Cannabis sativa L., Is a Selective Thermo-TRP Modulator. J. Nat. Prod. 2020, 83, 2727–2736. [Google Scholar] [CrossRef] [PubMed]
- Clinicaltrials.gov. Study to Assess the Effect of Cannabidiol on Liver Fat Levels in Subjects with Fatty Liver Disease. 2018. Available online: https://clinicaltrials.gov/ct2/show/results/NCT01284634 (accessed on 1 January 2020).
- Farooqui, M.T.; Khan, M.A.; Cholankeril, G.; Khan, Z.; Mohammed Abdul, M.K.; Li, A.A.; Shah, N.; Wu, L.; Haq, K.; Solanki, S.; et al. Marijuana is not associated with progression of hepatic fibrosis in liver disease: A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2019, 31, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Millar, S.A.; Stone, N.L.; Yates, A.S.; O’Sullivan, S.E. A Systematic Review on the Pharmacokinetics of Cannabidiol in Humans. Front. Pharmacol. 2018, 9, 1365. [Google Scholar] [CrossRef] [PubMed]
- Valenti, C.; Billi, M.; Pancrazi, G.L.; Calabria, E.; Armogida, N.G.; Tortora, G.; Pagano, S.; Barnaba, P.; Marinucci, L. Biological effects of cannabidiol on human cancer cells: Systematic review of the literature. Pharmacol. Res. 2022, 181, 106267. [Google Scholar] [CrossRef]
- Kenyon, J.; Liu, W.; Dalgleish, A. Report of Objective Clinical Responses of Cancer Patients to Pharmaceutical-grade Synthetic Cannabidiol. Anticancer. Res. 2018, 38, 5831–5835. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, Y.; Tong, M.; Pan, H.; Li, D. New Prospect for Cancer Cachexia: Medical Cannabinoid. J. Cancer 2019, 10, 716–720. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.R.; Burnell-Nugent, M.; Lossignol, D.; Ganae-Motan, E.D.; Potts, R.; Fallon, M.T. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J. Pain. Symptom Manag. 2010, 39, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.; Haywood, A.; Gogna, G.; Martin, J.; Yates, P.; Greer, R.; Good, P. Oral medicinal cannabinoids to relieve symptom burden in the palliative care of patients with advanced cancer: A double-blind, placebo-controlled, randomised clinical trial of efficacy and safety of 1:1 delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD). Trials 2020, 21, 611. [Google Scholar] [CrossRef]
- Polyzos, S.A.; Kountouras, J.; Mantzoros, C.S. Obesity and nonalcoholic fatty liver disease: From pathophysiology to therapeutics. Metabolism 2019, 92, 82–97. [Google Scholar] [CrossRef]
- Parray, H.A.; Yun, J.W. Cannabidiol promotes browning in 3T3-L1 adipocytes. Mol. Cell Biochem. 2016, 416, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Bielawiec, P.; Harasim-Symbor, E.; Sztolsztener, K.; Konstantynowicz-Nowicka, K.; Chabowski, A. Attenuation of Oxidative Stress and Inflammatory Response by Chronic Cannabidiol Administration Is Associated with Improved n-6/n-3 PUFA Ratio in the White and Red Skeletal Muscle in a Rat Model of High-Fat Diet-Induced Obesity. Nutrients 2021, 13, 1603. [Google Scholar] [CrossRef]
- Perucca, E.; Bialer, M. Critical Aspects Affecting Cannabidiol Oral Bioavailability and Metabolic Elimination, and Related Clinical Implications. CNS Drugs 2020, 34, 795–800. [Google Scholar] [CrossRef]
- Abbotts, K.S.S.; Ewell, T.R.; Butterklee, H.M.; Bomar, M.C.; Akagi, N.; Dooley, G.P.; Bell, C. Cannabidiol and Cannabidiol Metabolites: Pharmacokinetics, Interaction with Food, and Influence on Liver Function. Nutrients 2022, 14, 2152. [Google Scholar] [CrossRef]
- Chayasirisobhon, S. Mechanisms of Action and Pharmacokinetics of Cannabis. Perm. J. 2020, 25, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.J.; Galettis, P.; Schneider, J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br. J. Clin. Pharmacol. 2018, 84, 2477–2482. [Google Scholar] [CrossRef]
- Varadi, G.; Zhu, Z.; Crowley, H.D.; Moulin, M.; Dey, R.; Lewis, E.D.; Evans, M. Examining the Systemic Bioavailability of Cannabidiol and Tetrahydrocannabinol from a Novel Transdermal Delivery System in Healthy Adults: A Single-Arm, Open-Label, Exploratory Study. Adv. Ther. 2023, 40, 282–293. [Google Scholar] [CrossRef]
- Ohlsson, A.; Lindgren, J.E.; Andersson, S.; Agurell, S.; Gillespie, H.; Hollister, L.E. Single-dose kinetics of deuterium-labelled cannabidiol in man after smoking and intravenous administration. Biomed. Environ. Mass. Spectrom. 1986, 13, 77–83. [Google Scholar] [CrossRef]
- Sholler, D.J.; Spindle, T.R.; Cone, E.J.; Goffi, E.; Kuntz, D.; Mitchell, J.M.; Winecker, R.E.; Bigelow, G.E.; Flegel, R.R.; Vandrey, R. Urinary Pharmacokinetic Profile of Cannabidiol (CBD), Δ9-Tetrahydrocannabinol (THC) and Their Metabolites following Oral and Vaporized CBD and Vaporized CBD-Dominant Cannabis Administration. J. Anal. Toxicol. 2022, 46, 494–503. [Google Scholar] [CrossRef]
- Johnson, D.A.; Funnell, M.P.; Heaney, L.M.; Cable, T.G.; Wheeler, P.C.; Bailey, S.J.; Clifford, T.; James, L.J. Cannabidiol Oil Ingested as Sublingual Drops or Within Gelatin Capsules Shows Similar Pharmacokinetic Profiles in Healthy Males. Cannabis Cannabinoid Res. 2023; ahead of print. [Google Scholar] [CrossRef]
- MacNair, L.; Kulpa, J.; Hill, M.L.; Eglit, G.M.L.; Mosesova, I.; Bonn-Miller, M.O.; Peters, E.N. Sex Differences in the Pharmacokinetics of Cannabidiol and Metabolites Following Oral Administration of a Cannabidiol-Dominant Cannabis Oil in Healthy Adults. Cannabis Cannabinoid Res. 2023; ahead of print. [Google Scholar] [CrossRef]
- Nadulski, T.; Pragst, F.; Weinberg, G.; Roser, P.; Schnelle, M.; Fronk, E.M.; Stadelmann, A.M. Randomized, double-blind, placebo-controlled study about the effects of cannabidiol (CBD) on the pharmacokinetics of Delta9-tetrahydrocannabinol (THC) after oral application of THC verses standardized cannabis extract. Ther. Drug Monit. 2005, 27, 799–810. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L.; Gidal, B.; Blakey, G.; Tayo, B.; Morrison, G. A Phase I, Randomized, Double-Blind, Placebo-Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. CNS Drugs 2019, 32, 1053–1067, Erratum in CNS Drugs 2019, 33, 397. [Google Scholar] [CrossRef]
- Martinez Naya, N.; Kelly, J.; Corna, G.; Golino, M.; Polizio, A.H.; Abbate, A.; Toldo, S.; Mezzaroma, E. An Overview of Cannabidiol as a Multifunctional Drug: Pharmacokinetics and Cellular Effects. Molecules 2024, 29, 473. [Google Scholar] [CrossRef] [PubMed]
- Bardhi, K.; Coates, S.; Watson, C.J.W.; Lazarus, P. Cannabinoids and drug metabolizing enzymes: Potential for drug-drug interactions and implications for drug safety and efficacy. Expert. Rev. Clin. Pharmacol. 2022, 15, 1443–1460. [Google Scholar] [CrossRef]
- Schlag, A.K.; O’Sullivan, S.E.; Zafar, R.R.; Nutt, D.J. Current controversies in medical cannabis: Recent developments in human clinical applications and potential therapeutics. Neuropharmacology 2021, 191, 108586. [Google Scholar] [CrossRef]
- Greenwich Biosciences. Epidiolex [Package Insert]; Greenwich Biosciences: Carlsbad, CA, USA, 2018. [Google Scholar]
- Bergamaschi, M.M.; Queiroz, R.H.C.; Zuardi, A.W.; Crippa, J.A. Safety and Side Effects of Cannabidiol, a Cannabis Sativa Constituent. Curr. Drug Saf. 2011, 6, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, R.; Aqua, K.; Lombardo, J.; Lee, M. Observed Impact of Long-Term Consumption of Oral Cannabidiol on Liver Function in Healthy Adults. Cannabis Cannabinoid Res. 2023, 8, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Caputi, T.L. Re: “Observed Impact of Long-Term Consumption of Oral Cannabidiol on Liver Function in Healthy Adults” and a Recent Announcement of a New Cannabidiol Safety Study. Cannabis Cannabinoid Res. 2022, 7, 896–897. [Google Scholar] [CrossRef] [PubMed]
- Chesney, E.; Oliver, D.; Green, A.; Sovi, S.; Wilson, J.; Englund, A.; Freeman, T.P.; McGuire, P. Adverse Effects of Cannabidiol:A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Neuropsychopharmacology 2020, 45, 1799–1806. [Google Scholar] [CrossRef] [PubMed]

| Compounds | Model | Cell Lines/Animals | CBD Concentration | Mechanism | Ref. |
|---|---|---|---|---|---|
| CBD | Alcohol feeding-induced liver steatosis | C57BL/6J | 5 or 10 mg/kg/day i.p. during the ethanol exposure 11 days | TNFα, MCP1, IL-1β, MIP2, E-Selectin mRNA, lipid peroxidation, 3-nitrotyrosine formation, NOX2, ACC-1, FASN protein ↓, PPARα, MCAD, ADIPOR-1, and mCPT-1 protein ↑ | [9] |
| CBD | Co-treatment with doxorubicin on hepatocellular carcinoma | BNL1 ME cells | 10 µM | TRPV2 channels ↑, P-glycoprotein ATPase transporter ↓ | [11] |
| CBD | MASLD | HHL-5 cells, 3T3-L1 cells, Female ob/ob mice, Zebrafish | Female ob/ob mice:3 mg/kg/d oral gavage 4 weeks | CREB, ERK1/2, STAT2, STAT3, STAT6, AMPKa2, PRAS40 ↓ | [12] |
| CBD | HFC diet, LPS + ATP ± CBD (MASLD) | RAW264.7 cells, C57BL/6J | 5 mg/kg/d hand-fed 8 weeks | NF-κB-NLRP3 ↓ | [13] |
| CBDA | HFD-fed mice, GIO mice | C57BL/6J, HepG2, HK-2 cells | 20 or 40 mg/kg/d i.p. 28 days | obesity and its metabolic abnormalities ↓ | [16] |
| CBD | Acute alcohol drinking-induced liver steatosis | C57BL/6, HepG2 cells | 5 mg/kg/12 h i.p. 30 min before each ethanol gavage 5 days | JNK MAPK, autophagy ↑ | [17] |
| CBD | Ethanol plus high-fat, high-cholesterol diet-induced liver steatosis | C57BL/6J | 5 mg/kg/day gavaged 8 weeks | NF-κB–NLRP3–pyroptosis pathway ↓ | [18] |
| CBD | High-fat/cholesterol diet (MASLD) | C57BL/6J | HFCD containing CBD (2.39 mg/kg) | TNF-α, iNOS ↓ | [22] |
| CBD | CCl4-induced liver fibrosis | C57BL/6J | 20 mg/kg/day oral gavage 2 weeks | TGF-β and IL-4 induced fibroblast migration ↓ | [25] |
| CBD | PFOS-induced liver fibrosis | RAW264.7, AML12 and LX-2 cells, C57BL/6J | 10 mg/kg gavage 30 days | CCDC25-ILK-NF-κB ↓ | [26] |
| CBD | Liver Fibrosis | Hepatic stellate cells (HSCs) | 5 µM 0–8 h | PERK-, ATF6-, and IRE1-mediated ER stress ↓, IRE1/ASK1/JNK pathway ↑ in activated HSCs | [29] |
| CBD | Hepatocellular carcinoma | HepG2 and ECACC cells | 5 µM 60 min | Exosome and Micro-vesicle Release, CD63, STAT3 ↓ | [41] |
| C. sativa extract | Diethylnitrosamine-induced hepatocellular carcinoma | Wistar rats | 15 mg/kg or 30 mg/kg of C. sativa extract oral gavage 3 weeks | Akt, COX2, CRP, AFP mRNA ↓ | [42] |
| CBD | Co-treatment with cabozantinib on hepatocellular carcinoma | HepG2 and Hep3B cells | <100 µM 48 h | P53, ER stress ↓ | [45] |
| CBD | HCV/HBV infection | HepG2 2.2.15 cells | 10 µM 6 days | HCV replication ↓ | [53] |
| CBD | Cadmium hepatotoxicity | Rats | 5 mg/kg/day i.p. 5 days | TNF-α, COX-2, NF-kB, caspase-3, and caspase-9 ↓, endothelial nitric oxide synthase↑ | [55] |
| CBD | Cocaine-induced acute Hepatic Toxicity | Swiss mice | 30 mg/kg/day i.p. | Reduced serum ALT and cardio green levels, inflammatory injury ↓ | [56] |
| C. sativa extract | Oxidative-mediated hepatic injury | Albino rats | 15–240 μg/mL | GSH level, SOD, and catalase activities↓, MDA and NO levels ↑, hepatic amylase and lipase activities, hepatic cholesterol and LDL-C, gluconeogenic activity ↓ | [57] |
| CBD | Concanavalin A induced acute hepatitis | C57BL/6 | 5–50 mg/kg i.p. 6, 12, 24 and 48 h | TRPV1, Myeloid-derived suppressor cells (MDSCs) ↑ | [59] |
| CBD | Ischemia/reperfusion (I/R)-induced liver injury | Rats | 5 mg/kg/24 h i.v. (tail vein) 2 days | Nitric oxide synthase, COX-2, NF-κB, Fas ligand and caspase-3 ↓, Surviving protein ↑ | [60] |
| CBD | Ischemia/reperfusion (I/R)-induced liver injury | C57BL/6J | 3 or 10 mg/kg i.p. 60 min before the occlusion of the hepatic artery and the portal vein Or i.v. (femoral vein) before the reocclusion or 90 min after | Oxidative/nitrative stress, NF-κB, TNF-α production in isolated Kupffer cells ↓, adhesion molecule expression ↓ independent of CB1/2 receptors | [61] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Kim, J.-K. The Role of Cannabidiol in Liver Disease: A Systemic Review. Int. J. Mol. Sci. 2024, 25, 2370. https://doi.org/10.3390/ijms25042370
Chen S, Kim J-K. The Role of Cannabidiol in Liver Disease: A Systemic Review. International Journal of Molecular Sciences. 2024; 25(4):2370. https://doi.org/10.3390/ijms25042370
Chicago/Turabian StyleChen, Si, and Jeon-Kyung Kim. 2024. "The Role of Cannabidiol in Liver Disease: A Systemic Review" International Journal of Molecular Sciences 25, no. 4: 2370. https://doi.org/10.3390/ijms25042370
APA StyleChen, S., & Kim, J.-K. (2024). The Role of Cannabidiol in Liver Disease: A Systemic Review. International Journal of Molecular Sciences, 25(4), 2370. https://doi.org/10.3390/ijms25042370






