Carbon–TiO2 Hybrid Quantum Dots for Photocatalytic Inactivation of Gram-Positive and Gram-Negative Bacteria
Abstract
1. Introduction
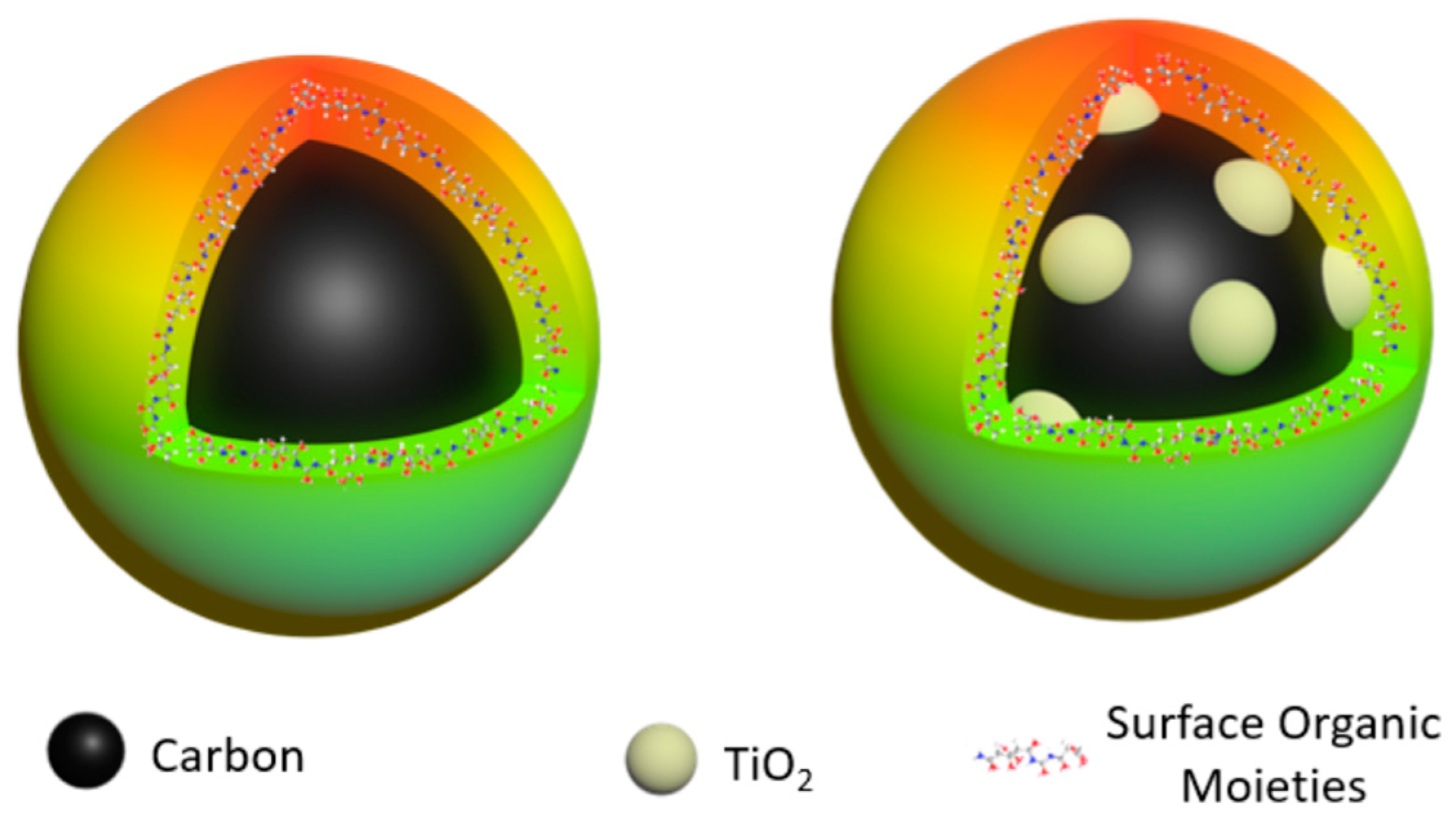
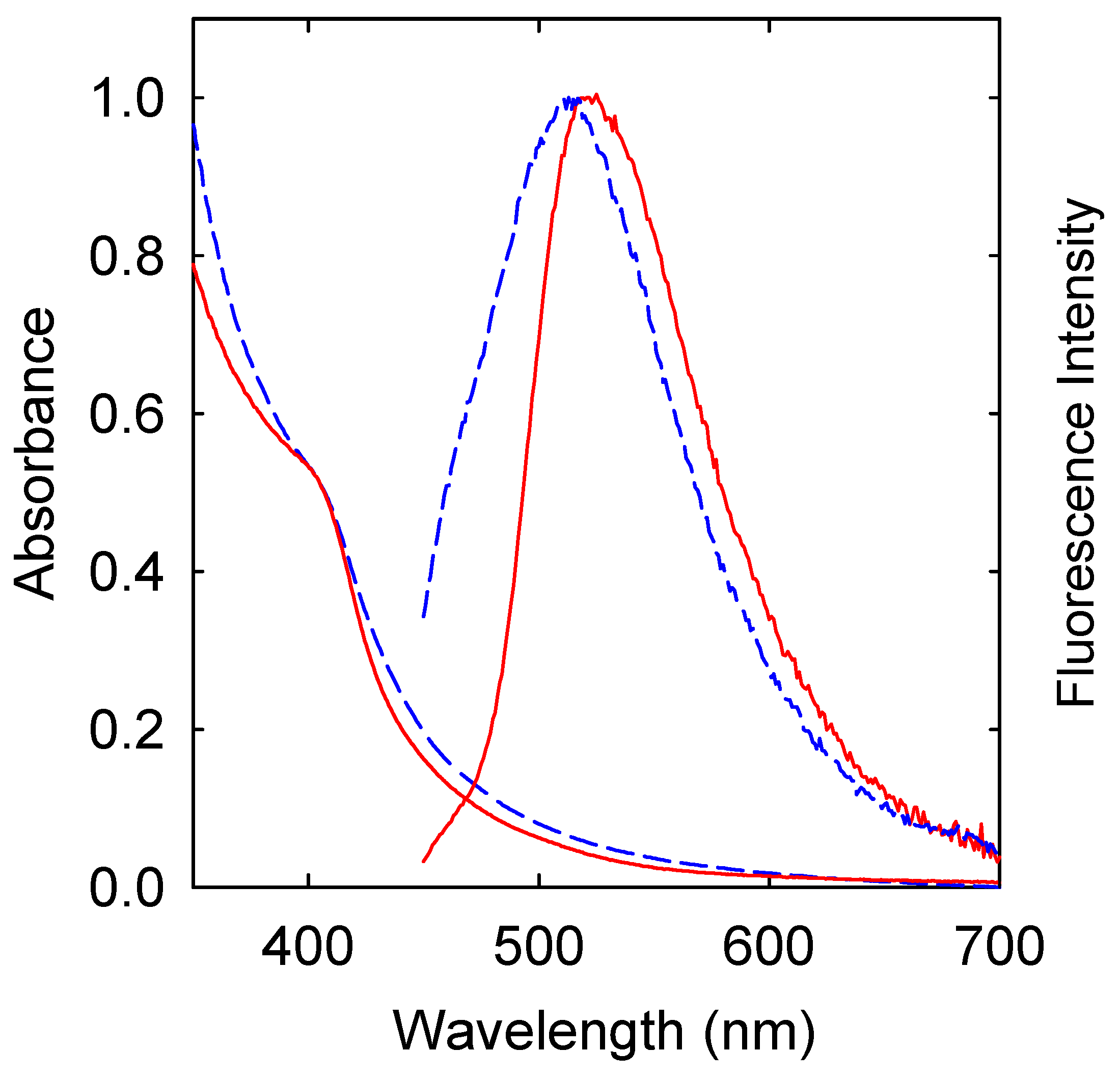
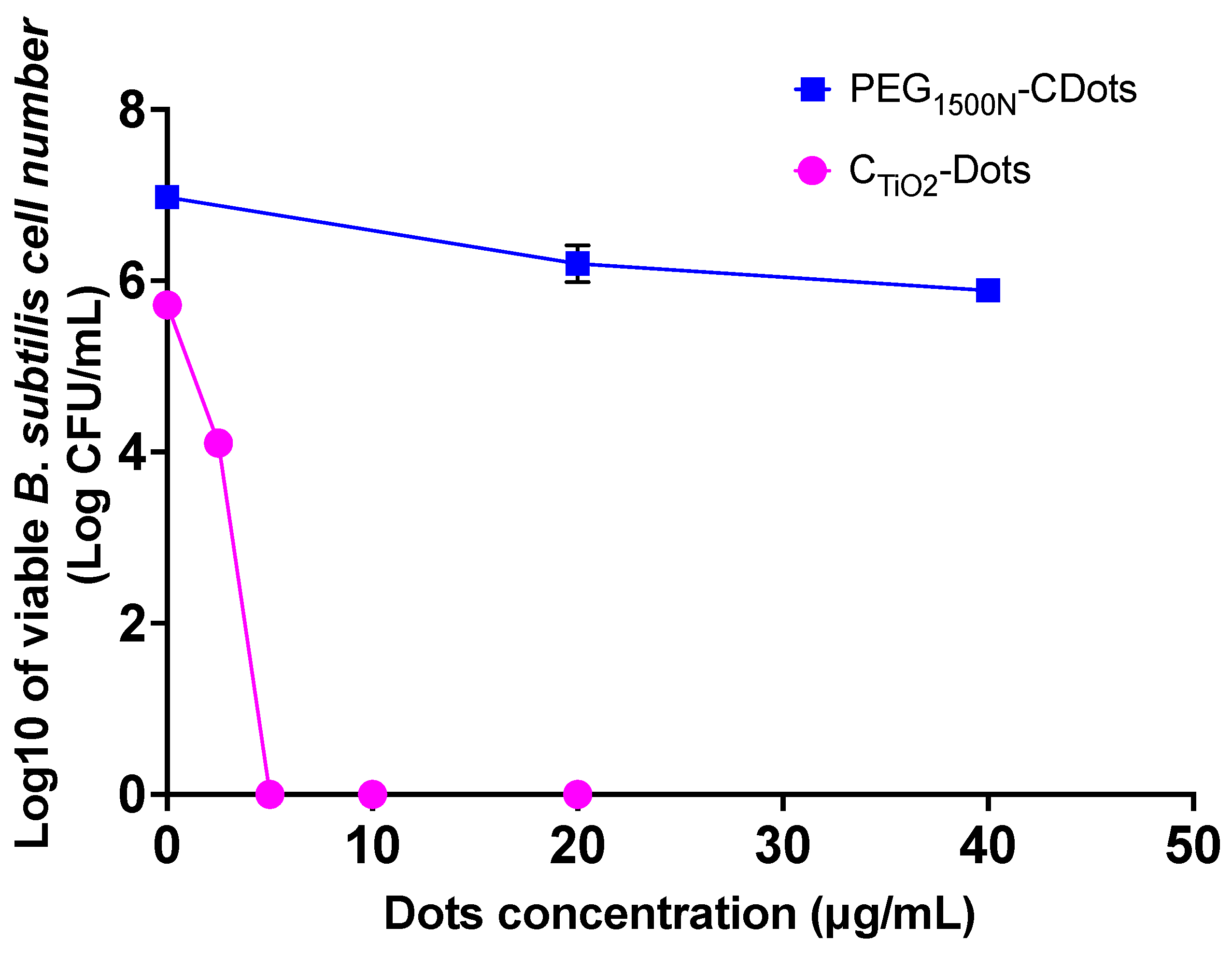
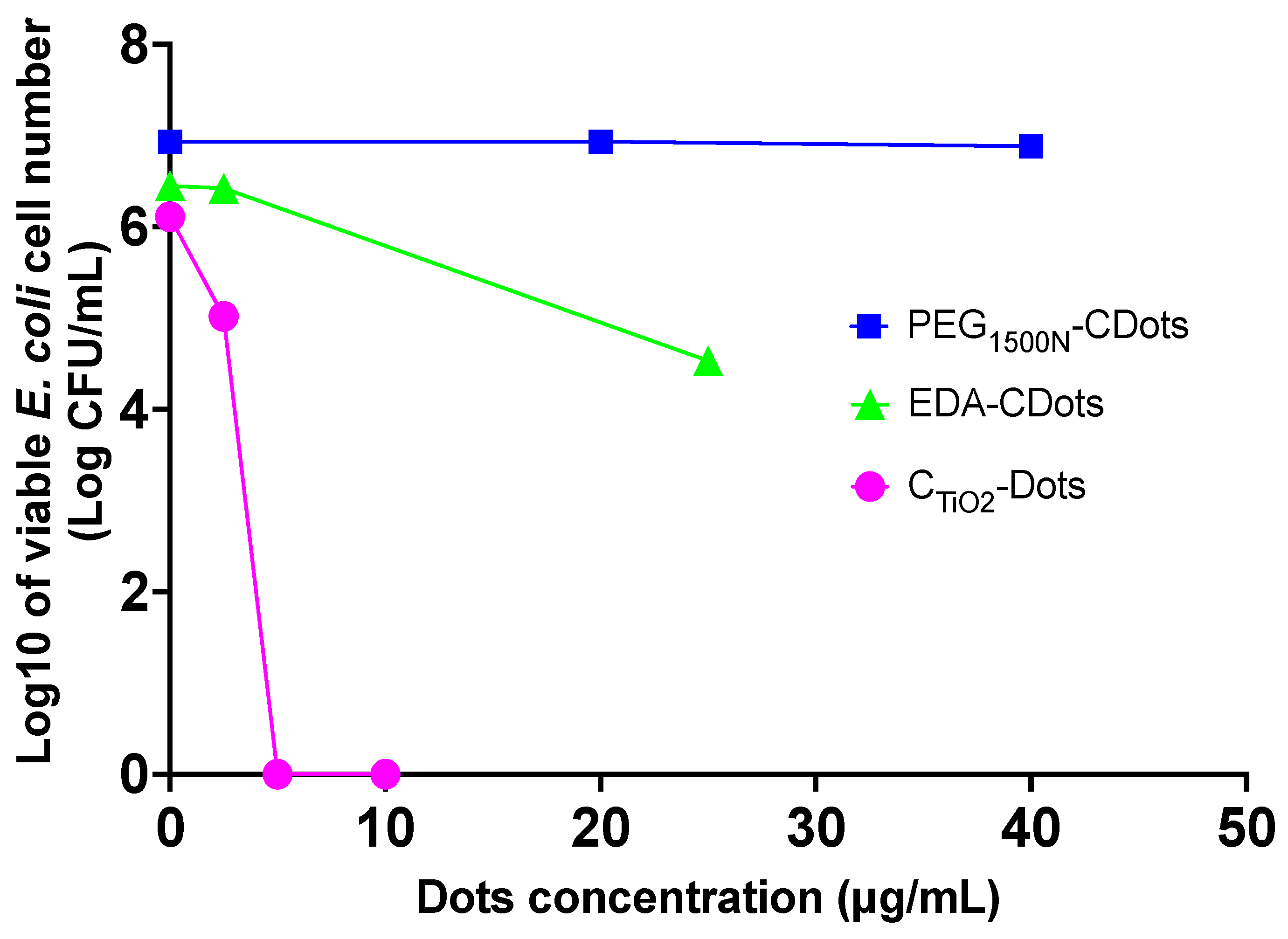
2. Results
3. Discussion
4. Material and Methods
4.1. Materials
4.2. Measurement
4.3. CTiO2-Dots and PEG1500N-CDots
4.4. Bacterial Strains and Cultures
4.5. Treatment of Bacterial Cells
4.6. Assessment of Antibacterial Activity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sun, Y.-P. Carbon Dots-Exploring Carbon at Zero-Dimension; Springer International Publishing: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Cao, L.; Meziani, M.J.; Sahu, S.; Sun, Y.-P. Photoluminescence Properties of Graphene versus Other Carbon Nanomaterials. Acc. Chem. Res. 2013, 46, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Bunker, C.E.; Sun, Y.-P. Carbon Dots: Zero-Dimensional Carbon Allotrope with Unique Photoinduced Redox Characteristics. ACS Omega 2020, 5, 965–971. [Google Scholar] [CrossRef]
- Sun, Y.-P.; Zhou, B.; Lin, Y.; Wang, W.; Fernando, K.A.S.; Pathak, P.; Meziani, M.J.; Harruff, B.A.; Wang, X.; Wang, H.; et al. Quantum-Sized Carbon Dots for Bright and Colorful Photoluminescence. J. Am. Chem. Soc. 2006, 128, 7756–7757. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-P. Fluorescent Carbon Nanoparticles. U.S. Patent 7,829,772, 9 November 2010. [Google Scholar]
- Cao, L.; Anilkumar, P.; Wang, X.; Liu, J.H.; Sahu, S.; Meziani, M.J.; Myers, E.; Sun, Y.-P. Reverse Stern-Volmer Behavior for Luminescence Quenching in Carbon Nanoparticles. Can. J. Chem. 2011, 89, 104–109. [Google Scholar] [CrossRef]
- Meziani, M.J.; Dong, X.; Zhu, L.; Jones, L.P.; LeCroy, G.E.; Yang, F.; Wang, S.; Wang, P.; Zhao, Y.; Yang, L.; et al. Visible-Light-Activated Bactericidal Functions of Carbon “Quantum” Dots. ACS Appl. Mater. Interfaces 2016, 8, 10761–10766. [Google Scholar] [CrossRef]
- Dong, X.; Liang, W.; Meziani, M.J.; Sun, Y.-P.; Yang, L. Carbon Dots as Potent Antimicrobial Agents. Theranostics 2020, 10, 671–686. [Google Scholar] [CrossRef] [PubMed]
- Bing, W.; Sun, H.; Yan, Z.; Ren, J.; Qu, X. Programmed Bacteria Death Induced by Carbon Dots with Different Surface Charge. Small 2016, 12, 4713–4718. [Google Scholar] [CrossRef]
- Stanković, N.K.; Bodik, M.; Šiffalovič, P.; Kotlar, M.; Mičušik, M.; Špitalsky, Z.; Danko, M.; Milivojević, D.D.; Kleinova, A.; Kubat, P.; et al. Antibacterial and Antibiofouling Properties of Light Triggered Fluorescent Hydrophobic Carbon Quantum Dots Langmuir-Blodgett Thin Films. ACS Sustain. Chem. Eng. 2018, 6, 4154–4163. [Google Scholar] [CrossRef]
- Kováčová, M.; Marković, Z.M.; Humpolíček, P.; Mičušík, M.; Švajdlenková, H.; Kleinová, A.; Danko, M.; Kubát, P.; Vajďák, J.; Capáková, Z.; et al. Carbon Quantum Dots Modified Polyurethane Nanocomposite as Effective Photocatalytic and Antibacterial Agents. ACS Biomater. Sci. Eng. 2018, 4, 3983–3993. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Parker, S.; Chiniforush, N.; Bahador, A. Photoexcitation Triggering via Semiconductor Graphene Quantum Dots by Photochemical Doping with Curcumin versus Perio-Pathogens Mixed Biofilms. Photodiagn. Photodyn. 2019, 28, 125–131. [Google Scholar] [CrossRef]
- Mushtaq, S.; Yasin, T.; Saleem, M.; Dai, T.; Muhammad, Y. Potentiation of Antimicrobial Photodynamic Therapy by Curcumin-Loaded Graphene Quantum Dots. Photochem. Photobiol. 2021, 98, 202–210. [Google Scholar] [CrossRef]
- Nocito, G.; Sciuto, E.L.; Franco, D.; Nastasi, F.; Pulvirenti, L.; Petralia, S.; Spinella, C.; Calabrese, G.; Guglielmino, S.; Conoci, S. Physicochemical Characterization and Antibacterial Properties of Carbon Dots from Two Mediterranean Olive Solid Waste Cultivars. Nanomaterials 2022, 12, 885. [Google Scholar] [CrossRef]
- Courtney, C.M.; Goodman, S.M.; McDaniel, J.A.; Madinger, N.E.; Chatterjee, A.; Nagpal, P. Photoexcited Quantum Dots for Killing Multidrug-Resistant Bacteria. Nat. Mater. 2016, 15, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Carré, G.; Hamon, E.; Ennahar, S.; Estner, M.; Lett, M.-C.; Horvatovich, P.; Gies, J.-P.; Valérie, K.; Keller, N.; Andre, P. TiO2 Photocatalysis Damages Lipids and Proteins in Escherichia coli. Appl. Environ. Microbiol. 2014, 80, 2573–2581. [Google Scholar] [CrossRef] [PubMed]
- Oetiker, N.; Muñoz-Villagrán, C.; Vásquez, C.C.; Bravo, D.; Pérez-Donoso, J.M. Bacterial Phototoxicity of Biomimetic CdTe-GSH Quantum Dots. J. Appl. Microbiol. 2021, 131, 155–168. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Moon, J.; Lee, H.; Koh, S.; Kim, G.-M.; Gauthé, L.; Stellacci, F.; Huh, Y.S.; Kim, P.; Lee, D.C. Photodynamic Treatment of Multidrug-Resistant Bacterial Infection Using Indium Phosphide Quantum Dots. Biomater. Sci. 2022, 10, 7149–7161. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-P.; Wang, X.; Lu, F.; Cao, L.; Meziani, M.J.; Luo, P.G.; Gu, L.; Veca, L.M. Doped Carbon Nanoparticles as a New Platform for Highly Photoluminescent Dots. J. Phys. Chem. C 2008, 112, 18295–18298. [Google Scholar] [CrossRef] [PubMed]
- Anilkumar, P.; Wang, X.; Cao, L.; Sahu, S.; Liu, J.-H.; Wang, P.; Korch, K.; Tackett, K.N., II; Parenzan, A.; Sun, Y.-P. Toward Quantitatively Fluorescent Carbon-Based “Quantum” Dots. Nanoscale 2011, 3, 2023–2027. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Y.; Qian, H.; Wang, P.; LeCroy, G.E.; Bunker, C.E.; Fernando, K.A.S.; Yang, L.; Reibold, M.; Sun, Y.-P. Carbon-TiO2 Hybrid Dots in Different Configurations—Optical Properties, Redox Characteristics, and Mechanistic Implications. New J. Chem. 2018, 42, 10798–10806. [Google Scholar] [CrossRef]
- Ruppé, É.; Woerther, P.L.; Barbier, F. Mechanisms of Antimicrobial Resistance in Gram-negative Bacilli. Ann. Intensive Care 2015, 5, 61. [Google Scholar] [CrossRef]
- Gauba, A.; Rahman, K.M. Evaluation of Antibiotic Resistance Mechanisms in Gram-Negative Bacteria. Antibiotics 2023, 12, 1590. [Google Scholar] [CrossRef]
- Miller, S.I. Antibiotic Resistance and Regulation of the Gram-Negative Bacterial Outer Membrane Barrier by Host Innate Immune Molecules. mBio 2016, 7, e01541. [Google Scholar] [CrossRef]
- Le Moual, H.; Gruenheid, S. Resistance to Antimicrobial Peptides in Gram-negative Bacteria. FEMS Microbiol. Lett. 2012, 330, 81–89. [Google Scholar]
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial Resistance: A Global Multifaceted Phenomenon. Pathog. Glob. Health 2015, 109, 309–318. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, D.M.P.; Forde, B.M.; Kidd, T.J.; Harris, P.N.A.; Schembri, M.A.; Beatson, S.A.; Paterson, D.L.; Walker, M.J. Antimicrobial Resistance in ESKAPE Pathogens. Clin. Microbiol. Rev. 2020, 33, e00181. [Google Scholar] [CrossRef]
- Nikaido, H.; Nakae, T. The Outer Membrane of Gram-Negative Bacteria. Adv. Microb. Physiol. 1979, 20, 163–250. [Google Scholar]
- Hancock, R.E.W.; Bell, A. Antibiotic Uptake into Gram-Negative Bacteria. Eur. J. Clin. Microbiol. Infect. Dis. 1988, 7, 713–720. [Google Scholar] [CrossRef]
- Breijyeh, Z.; Jubeh, B.; Karaman, R. Resistance of Gram-Negative Bacteria to Current Antibacterial Agents and Approaches to Resolve It. Molecules 2020, 25, 1340. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ma, W.; Fang, Y.; Liang, H.; Yang, H.; Wang, Y.; Dong, X.; Zhan, Y.; Wang, X. Core Oligosaccharide Portion of Lipopolysaccharide Plays Important Roles in Multiple Antibiotic Resistance in Escherichia Coli. Antimicrob. Agents Chemother. 2021, 65, e00341-21. [Google Scholar] [CrossRef] [PubMed]
- Silhavy, T.J.; Kahne, D.; Walker, S. The Bacterial Cell Envelope. Cold Spring Harb. Perspect. Boil. 2010, 2, a000414. [Google Scholar] [CrossRef] [PubMed]
- Exner, M.; Bhattacharya, S.; Christiansen, B.; Gebel, J.; Goroncy-Bermes, P.; Hartemann, P.; Heeg, P.; Ilschner, C.; Kramer, A.; Larson, E.; et al. Antibiotic Resistance: What Is So Special about Multidrug-Resistant Gram-Negative Bacteria? GMS Hyg. Infect. Control 2017, 12, Doc05. [Google Scholar]
- El Rayes, J.; Rodríguez-Alonso, R.; Collet, J.F. Lipoproteins in Gram-Negative Bacteria: New Insights into Their Biogenesis, Subcellular Targeting and Functional Roles. Curr. Opin. Microbiol. 2021, 61, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Blair, J.M.; Richmond, G.E.; Piddock, L.J. Multidrug Efflux Pumps in Gram-Negative Bacteria and Their Role in Antibiotic Resistance. Future Microbiol. 2014, 9, 1165–1177. [Google Scholar] [CrossRef] [PubMed]
- Richmond, M.H.; Wotton, S. Comparative Study of Seven Cephalosporins: Susceptibility to Betalactamases and Ability to Penetrate the Surface Layers of Escherichia coli. Antimicrob. Agents Chemother. 1976, 10, 219–222. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhnmerman, W.; Rosselet, A. Function of the Outer Membrane of Escherichia coli as a Permeability Barrier to Beta-Lactam Antibiotics. Antimicrob. Agents Chemother. 1977, 12, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Dik, D.A.; Fisher, J.F.; Mobashery, S. Cell-Wall Recycling of the Gram-Negative Bacteria and the Nexus to Antibiotic Resistance. Chem. Rev. 2018, 118, 5952–5984. [Google Scholar] [CrossRef] [PubMed]
- Lima, L.M.; da Silva, B.N.M.; Barbosa, G.; Barreiro, E.J. β-Lactam Antibiotics: An Overview from a Medicinal Chemistry Perspective. Eur. J. Med. Chem. 2020, 208, 112829. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Jeong, S.H.; Cha, S.; Lee, S.H. A Lack of Drugs for Antibiotic-resistant Gram-negative Bacteria. Nat. Rev. Drug Discov. 2007, 6, 29–40. [Google Scholar] [CrossRef]
- Xu, Z.; Flavin, M.T.; Flavin, J. Combating Multidrug-resistant Gram-negative Bacterial Infections. Exp. Opin. Investig. Drugs 2014, 23, 163–182. [Google Scholar] [CrossRef]
- Datta, P.; Gupta, V. Next-generation Strategy for Treating Drug Resistant Bacteria: Antibiotic Hybrids. Indian J. Med. Res. 2019, 149, 97–106. [Google Scholar] [CrossRef]
- LeCroy, G.E.; Sonkar, S.K.; Yang, F.; Veca, L.M.; Wang, P.; Tackett, K.N., II; Yu, J.-J.; Vasile, E.; Qian, H.; Liu, Y.; et al. Toward Structurally Defined Carbon Dots as Ultracompact Fluorescent Probes. ACS Nano 2014, 8, 4522–4529. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Awak, M.A.; Tomlinson, N.; Tang, Y.; Sun, Y.-P.; Yang, L. Antibacterial Effects of Carbon Dots in Combination with other Antimicrobial Reagents. PLoS ONE 2017, 12, e0185324. [Google Scholar] [CrossRef] [PubMed]
- Al Awak, M.M.; Wang, P.; Wang, S.; Tang, Y.; Sun, Y.-P.; Yang, L. Correlation of Carbon Dots’ Light-Activated Antimicrobial Activities and Fluorescence Quantum Yield. RSC Adv. 2017, 7, 30177–30184. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, P.; Moreira, J.; Gomaa, H.; Ray, A.K. Visible-Solar-Light-Driven Photocatalytic Degradation of Phenol with Dye-Sensitized TiO2: Parametric and Kinetic Study. Ind. Eng. Chem. Res. 2012, 51, 4523–4532. [Google Scholar] [CrossRef]
- Etacheri, V.; Di Valentin, C.; Schneider, J.; Bahnemann, D.; Pillai, S.C. Visible-Light Activation of TiO2 Photocatalysts: Advances in Theory and Experiments. J. Photochem. Photobiol. C 2015, 25, 1–29. [Google Scholar] [CrossRef]
- Giridhar, M.; BhojyaNaik, H.S.; Vishwanath, R.; Sudhamani, C.N.; Prabakar, M.C.; Kenchappa, R. Preparation of Azo-Dye Sensitized TiO2 Photocatalyst for Inhibition of E-Coli Bacteria under Visible Light Irradiation. Mater. Today Proc. 2017, 4, 11671–11678. [Google Scholar] [CrossRef]
- Li, X.; Shi, J.-L.; Hao, H.; Lang, X. Visible Light-Induced Selective Oxidation of Alcohols with Air by Dye-Sensitized TiO2 Photocatalysis. Appl. Catal. B–Environ. 2018, 232, 260–267. [Google Scholar] [CrossRef]
- Schutte-Smith, M.; Erasmus, E.; Mogale, R.; Marogoa, N.; Jayiya, A.; Visser, H.G. Using Visible Light to Activate Antiviral and Antimicrobial Properties of TiO2 Nanoparticles in Paints and Coatings: Focus on New Developments for Frequent-Touch Surfaces in Hospitals. J. Coat. Technol. Res. 2023, 20, 789–817. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, X.; Liu, Y.; Adcock, A.F.; Sheriff, K.; Liang, W.; Yang, L.; Sun, Y.-P. Carbon–TiO2 Hybrid Quantum Dots for Photocatalytic Inactivation of Gram-Positive and Gram-Negative Bacteria. Int. J. Mol. Sci. 2024, 25, 2196. https://doi.org/10.3390/ijms25042196
Dong X, Liu Y, Adcock AF, Sheriff K, Liang W, Yang L, Sun Y-P. Carbon–TiO2 Hybrid Quantum Dots for Photocatalytic Inactivation of Gram-Positive and Gram-Negative Bacteria. International Journal of Molecular Sciences. 2024; 25(4):2196. https://doi.org/10.3390/ijms25042196
Chicago/Turabian StyleDong, Xiuli, Yamin Liu, Audrey F. Adcock, Kirkland Sheriff, Weixiong Liang, Liju Yang, and Ya-Ping Sun. 2024. "Carbon–TiO2 Hybrid Quantum Dots for Photocatalytic Inactivation of Gram-Positive and Gram-Negative Bacteria" International Journal of Molecular Sciences 25, no. 4: 2196. https://doi.org/10.3390/ijms25042196
APA StyleDong, X., Liu, Y., Adcock, A. F., Sheriff, K., Liang, W., Yang, L., & Sun, Y.-P. (2024). Carbon–TiO2 Hybrid Quantum Dots for Photocatalytic Inactivation of Gram-Positive and Gram-Negative Bacteria. International Journal of Molecular Sciences, 25(4), 2196. https://doi.org/10.3390/ijms25042196






