Current Perspectives on the Molecular and Clinical Relationships between Primary Biliary Cholangitis and Hepatocellular Carcinoma
Abstract
1. Introduction
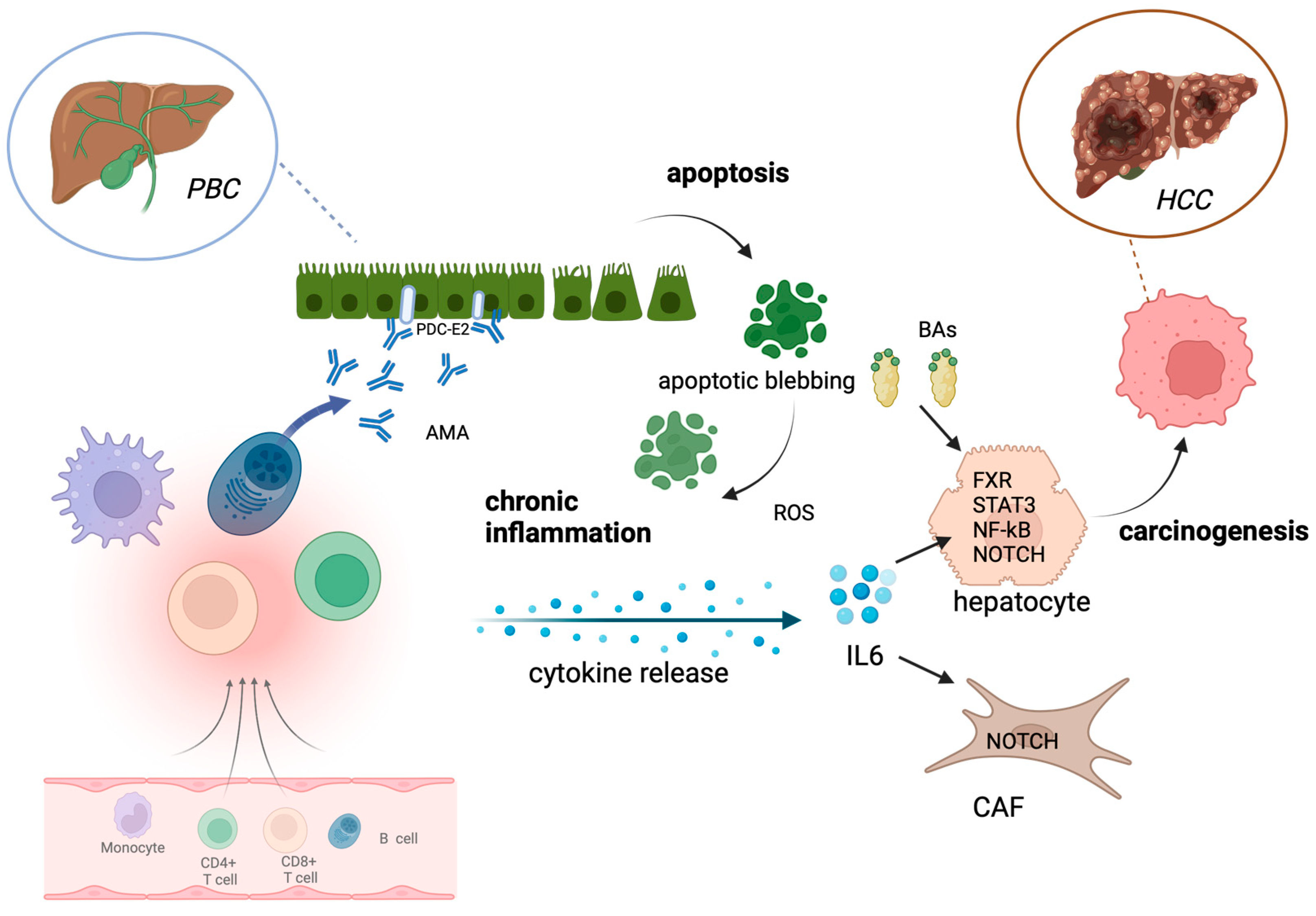
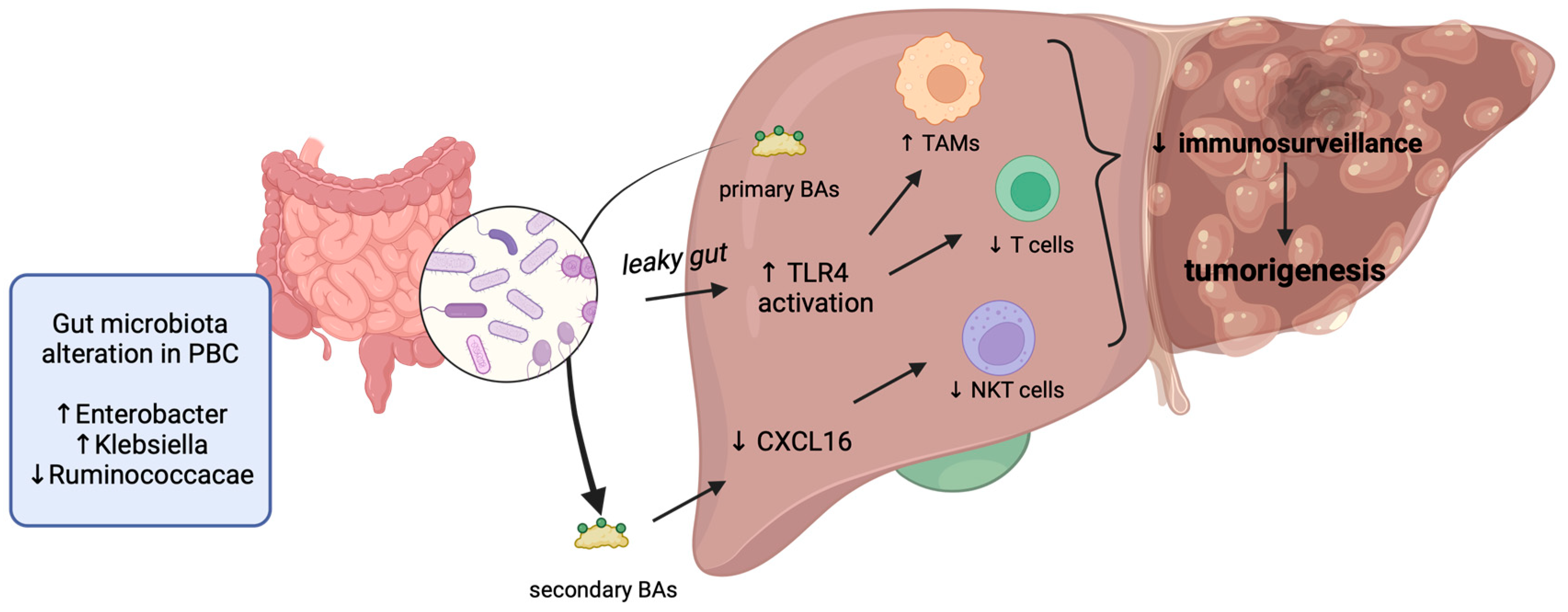
2. Shared Molecular Alterations in PBC and HCC
3. Hepatocellular Carcinoma in PBC Patients
3.1. Risk Factors for HCC in PBC Patients
3.1.1. Male Gender
3.1.2. Hepatitis Viruses
3.1.3. Lack of UDCA Response
3.1.4. Alcohol Intake
3.1.5. Metabolic and Age-Related Risk Factors
4. HCC Screening and Treatment in PBC Patients
5. Future Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tanaka, A. Current Understanding of Primary Biliary Cholangitis. Clin. Mol. Hepatol. 2021, 27, 1–21. [Google Scholar] [CrossRef]
- Colapietro, F.; Bertazzoni, A.; Lleo, A. Contemporary Epidemiology of Primary Biliary Cholangitis. Clin. Liver Dis. 2022, 26, 555–570. [Google Scholar] [CrossRef] [PubMed]
- Lleo, A.; Jepsen, P.; Morenghi, E.; Carbone, M.; Moroni, L.; Battezzati, P.M.; Podda, M.; Mackay, I.R.; Gershwin, M.E.; Invernizzi, P. Evolving Trends in Female to Male Incidence and Male Mortality of Primary Biliary Cholangitis. Sci. Rep. 2016, 6, 25906. [Google Scholar] [CrossRef] [PubMed]
- Sy, A.M.; Ferreira, R.D.; John, B.V. Hepatocellular Carcinoma in Primary Biliary Cholangitis. Clin. Liver Dis. 2022, 26, 691–704. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.G.; Lampertico, P.; Nahon, P. Epidemiology and Surveillance for Hepatocellular Carcinoma: New Trends. J. Hepatol. 2020, 72, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, G.N.; Splan, M.F.; Weiss, N.S.; McDonald, G.B.; Beretta, L.; Lee, S.P. Incidence and Predictors of Hepatocellular Carcinoma in Patients with Cirrhosis. Clin. Gastroenterol. Hepatol. 2007, 5, 938–945.e4. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Willoughby, C.E.; Singal, A.G.; Greten, T.F.; Heikenwälder, M.; El-Serag, H.B.; Finn, R.S.; Friedman, S.L. Nonalcoholic Steatohepatitis-Related Hepatocellular Carcinoma: Pathogenesis and Treatment. Nat. Rev. Gastroenterol. Hepatol. 2023, 20, 487–503. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-C.A.; Cohet, C.; Yang, Y.-C.; Stayner, L.; Hashibe, M.; Straif, K. Meta-Analysis of Epidemiologic Studies on Cigarette Smoking and Liver Cancer. Int. J. Epidemiol. 2009, 38, 1497–1511. [Google Scholar] [CrossRef] [PubMed]
- Rich, N.E.; Hester, C.; Odewole, M.; Murphy, C.C.; Parikh, N.D.; Marrero, J.A.; Yopp, A.C.; Singal, A.G. Racial and Ethnic Differences in Presentation and Outcomes of Hepatocellular Carcinoma. Clin. Gastroenterol. Hepatol. 2019, 17, 551–559.e1. [Google Scholar] [CrossRef]
- Watanabe, T.; Soga, K.; Hirono, H.; Hasegawa, K.; Shibasaki, K.; Kawai, H.; Aoyagi, Y. Features of Hepatocellular Carcinoma in Cases with Autoimmune Hepatitis and Primary Biliary Cirrhosis. World J. Gastroenterol. 2009, 15, 231–239. [Google Scholar] [CrossRef]
- Cazzagon, N.; Sarcognato, S.; Floreani, A.; Corrà, G.; De Martin, S.; Guzzardo, V.; Russo, F.P.; Guido, M. Cholangiocyte Senescence in Primary Sclerosing Cholangitis Is Associated with Disease Severity and Prognosis. JHEP Rep. 2021, 3, 100286. [Google Scholar] [CrossRef]
- Paillet, J.; Plantureux, C.; Lévesque, S.; Le Naour, J.; Stoll, G.; Sauvat, A.; Caudana, P.; Tosello Boari, J.; Bloy, N.; Lachkar, S.; et al. Autoimmunity Affecting the Biliary Tract Fuels the Immunosurveillance of Cholangiocarcinoma. J. Exp. Med. 2021, 218, e20200853. [Google Scholar] [CrossRef]
- Liang, Y.; Yang, Z.; Zhong, R. Primary Biliary Cirrhosis and Cancer Risk: A Systematic Review and Meta-Analysis. Hepatology 2012, 56, 1409–1417. [Google Scholar] [CrossRef]
- Floreani, A.; Franceschet, I.; Cazzagon, N.; Spinazzè, A.; Buja, A.; Furlan, P.; Baldo, V.; Gershwin, M.E. Extrahepatic Autoimmune Conditions Associated with Primary Biliary Cirrhosis. Clin. Rev. Allergy Immunol. 2015, 48, 192–197. [Google Scholar] [CrossRef]
- Braga, M.H.; Cançado, G.G.L.; Bittencourt, P.L.; Couto, C.A.; Guedes, L.V.; Lima, A.M.C.; Ferraz, M.L.G.; Villela-Nogueira, C.A.; Nardelli, M.J.; Faria, L.C.; et al. Risk Factors for Cancer in Patients with Primary Biliary Cholangitis and Autoimmune Hepatitis and Primary Biliary Cholangitis Overlap Syndrome. Ann. Hepatol. 2023, 28, 101105. [Google Scholar] [CrossRef] [PubMed]
- Floreani, A.; Gabbia, D.; De Martin, S. Primary Biliary Cholangitis: Primary Autoimmune Disease or Primary Secretory Defect. Expert Rev. Gastroenterol. Hepatol. 2023, 17, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; He, X.; Rojas, M.; Leung, P.S.C.; Gao, L. Mechanism-Based Target Therapy in Primary Biliary Cholangitis: Opportunities before Liver Cirrhosis? Front. Immunol. 2023, 14, 1184252. [Google Scholar] [CrossRef] [PubMed]
- Gulamhusein, A.F.; Hirschfield, G.M. Primary Biliary Cholangitis: Pathogenesis and Therapeutic Opportunities. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 93–110. [Google Scholar] [CrossRef] [PubMed]
- Refolo, M.G.; Messa, C.; Guerra, V.; Carr, B.I.; D’Alessandro, R. Inflammatory Mechanisms of HCC Development. Cancers 2020, 12, 641. [Google Scholar] [CrossRef] [PubMed]
- Gabbia, D.; De Martin, S. Tumor Mutational Burden for Predicting Prognosis and Therapy Outcome of Hepatocellular Carcinoma. Int. J. Mol. Sci. 2023, 24, 3441. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Sharma, H.; Khanna, S.; Sadhu Balasundaram, T.; Chowdhury, S.; Chowdhury, R.; Mukherjee, S. Interleukin-6 Induced Proliferation Is Attenuated by Transforming Growth Factor-β-Induced Signaling in Human Hepatocellular Carcinoma Cells. Front. Oncol. 2022, 11, 811941. [Google Scholar] [CrossRef]
- Xu, J.; Lin, H.; Wu, G.; Zhu, M.; Li, M. IL-6/STAT3 Is a Promising Therapeutic Target for Hepatocellular Carcinoma. Front. Oncol. 2021, 11, 760971. [Google Scholar] [CrossRef]
- Naugler, W.E.; Sakurai, T.; Kim, S.; Maeda, S.; Kim, K.; Elsharkawy, A.M.; Karin, M. Gender Disparity in Liver Cancer Due to Sex Differences in MyD88-Dependent IL-6 Production. Science 2007, 317, 121–124. [Google Scholar] [CrossRef]
- Sayaf, K.; Gabbia, D.; Russo, F.P.; De Martin, S. The Role of Sex in Acute and Chronic Liver Damage. Int. J. Mol. Sci. 2022, 23, 10654. [Google Scholar] [CrossRef]
- Prieto, J. Inflammation, HCC and Sex: IL-6 in the Centre of the Triangle. J. Hepatol. 2008, 48, 380–381. [Google Scholar] [CrossRef]
- Terziroli Beretta-Piccoli, B.; Mieli-Vergani, G.; Vergani, D.; Vierling, J.M.; Adams, D.; Alpini, G.; Banales, J.M.; Beuers, U.; Björnsson, E.; Bowlus, C.; et al. The Challenges of Primary Biliary Cholangitis: What Is New and What Needs to Be Done. J. Autoimmun. 2019, 105, 102328. [Google Scholar] [CrossRef]
- Carpino, G.; Cardinale, V.; Folseraas, T.; Overi, D.; Floreani, A.; Franchitto, A.; Onori, P.; Cazzagon, N.; Berloco, P.B.; Karlsen, T.H.; et al. Hepatic Stem/Progenitor Cell Activation Differs between Primary Sclerosing and Primary Biliary Cholangitis. Am. J. Pathol. 2018, 188, 627–639. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, A.; Alsinet, C.; Yanger, K.; Hoshida, Y.; Zong, Y.; Toffanin, S.; Rodriguez–Carunchio, L.; Solé, M.; Thung, S.; Stanger, B.Z.; et al. Notch Signaling Is Activated in Human Hepatocellular Carcinoma and Induces Tumor Formation in Mice. Gastroenterology 2012, 143, 1660–1669. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Li, J.; Zheng, J.; Wei, A. The Carcinogenic Role of the Notch Signaling Pathway in the Development of Hepatocellular Carcinoma. J. Cancer 2019, 10, 1570–1579. [Google Scholar] [CrossRef] [PubMed]
- Fung, E.; Tang, S.-M.T.; Canner, J.P.; Morishige, K.; Arboleda-Velasquez, J.F.; Cardoso, A.A.; Carlesso, N.; Aster, J.C.; Aikawa, M. Delta-Like 4 Induces Notch Signaling in Macrophages: Implications for Inflammation. Circulation 2007, 115, 2948–2956. [Google Scholar] [CrossRef] [PubMed]
- Arvanitakis, K.; Koletsa, T.; Mitroulis, I.; Germanidis, G. Tumor-Associated Macrophages in Hepatocellular Carcinoma Pathogenesis, Prognosis and Therapy. Cancers 2022, 14, 226. [Google Scholar] [CrossRef] [PubMed]
- Mazzocca, A.; Fransvea, E.; Dituri, F.; Lupo, L.; Antonaci, S.; Giannelli, G. Down-Regulation of Connective Tissue Growth Factor by Inhibition of Transforming Growth Factor β Blocks the Tumor-Stroma Cross-Talk and Tumor Progression in Hepatocellular Carcinoma. Hepatology 2010, 51, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Xiong, S.; Wang, R.; Chen, Q.; Luo, J.; Wang, J.; Zhao, Z.; Li, Y.; Wang, Y.; Wang, X.; Cheng, B. Cancer-Associated Fibroblasts Promote Stem Cell-like Properties of Hepatocellular Carcinoma Cells through IL-6/STAT3/Notch Signaling. Am. J. Cancer Res. 2018, 8, 302–316. [Google Scholar] [PubMed]
- Floreani, A.; Gabbia, D.; De Martin, S. Obeticholic Acid for Primary Biliary Cholangitis. Biomedicines 2022, 10, 2464. [Google Scholar] [CrossRef] [PubMed]
- Gabbia, D.; Roverso, M.; Guido, M.; Sacchi, D.; Scaffidi, M.; Carrara, M.; Orso, G.; Russo, F.P.; Floreani, A.; Bogialli, S.; et al. Western Diet-Induced Metabolic Alterations Affect Circulating Markers of Liver Function before the Development of Steatosis. Nutrients 2019, 11, 1602. [Google Scholar] [CrossRef]
- Dilger, K.; Hohenester, S.; Winkler-Budenhofer, U.; Bastiaansen, B.A.J.; Schaap, F.G.; Rust, C.; Beuers, U. Effect of Ursodeoxycholic Acid on Bile Acid Profiles and Intestinal Detoxification Machinery in Primary Biliary Cirrhosis and Health. J. Hepatol. 2012, 57, 133–140. [Google Scholar] [CrossRef]
- Altamirano-Barrera, A.; Uribe, M.; Lammert, F.; Méndez-Sánchez, N. Bile Acids and the Risk for Hepatocellular Carcinoma in Primary Biliary Cholangitis. Ann. Hepatol. 2016, 15, 453–454. [Google Scholar] [CrossRef]
- Xie, G.; Wang, X.; Huang, F.; Zhao, A.; Chen, W.; Yan, J.; Zhang, Y.; Lei, S.; Ge, K.; Zheng, X.; et al. Dysregulated Hepatic Bile Acids Collaboratively Promote Liver Carcinogenesis. Int. J. Cancer 2016, 139, 1764–1775. [Google Scholar] [CrossRef]
- Gabbia, D.; Dalla Pozza, A.; Albertoni, L.; Lazzari, R.; Zigiotto, G.; Carrara, M.; Baldo, V.; Baldovin, T.; Floreani, A.; De Martin, S. Pregnane X Receptor and Constitutive Androstane Receptor Modulate Differently CYP3A-Mediated Metabolism in Early- and Late-Stage Cholestasis. World J. Gastroenterol. 2017, 23, 7519–7530. [Google Scholar] [CrossRef]
- Colosimo, S.; Tomlinson, J.W. Bile Acids as Drivers and Biomarkers of Hepatocellular Carcinoma. World J. Hepatol. 2022, 14, 1730–1738. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Feng, J.; Li, J.; Yu, Q.; Ji, J.; Wu, J.; Dai, W.; Guo, C. The Gut Microbiome-Bile Acid Axis in Hepatocarcinogenesis. Biomed. Pharmacother. 2021, 133, 111036. [Google Scholar] [CrossRef]
- Kotiya, D.; Jaiswal, B.; Ghose, S.; Kaul, R.; Datta, K.; Tyagi, R.K. Role of PXR in Hepatic Cancer: Its Influences on Liver Detoxification Capacity and Cancer Progression. PLoS ONE 2016, 11, e0164087. [Google Scholar] [CrossRef] [PubMed]
- Fiorucci, S.; Biagioli, M.; Zampella, A.; Distrutti, E. Bile Acids Activated Receptors Regulate Innate Immunity. Front. Immunol. 2018, 9, 1853. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhu, Y.; Tawfik, O.; Kong, B.; Williams, J.A.; Zhan, L.; Kassel, K.M.; Luyendyk, J.P.; Wang, L.; Guo, G.L. Mechanisms of STAT3 Activation in the Liver of FXR Knockout Mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, G829–G837. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Pardoll, D.; Jove, R. STATs in Cancer Inflammation and Immunity: A Leading Role for STAT3. Nat. Rev. Cancer 2009, 9, 798–809. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Xu, Z.; Zhang, Y.; Jiang, P.; Huang, G.; Chen, S.; Lyu, X.; Zheng, P.; Zhao, X.; Zeng, Y.; et al. FXR Induces SOCS3 and Suppresses Hepatocellular Carcinoma. Oncotarget 2015, 6, 34606–34616. [Google Scholar] [CrossRef]
- Attia, Y.M.; Tawfiq, R.A.; Ali, A.A.; Elmazar, M.M. The FXR Agonist, Obeticholic Acid, Suppresses HCC Proliferation & Metastasis: Role of IL-6/STAT3 Signalling Pathway. Sci. Rep. 2017, 7, 12502. [Google Scholar] [CrossRef]
- Gabbia, D.; De Martin, S. Targeting the Adipose Tissue–Liver–Gut Microbiota Crosstalk to Cure MASLD. Biology 2023, 12, 1471. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, L.; Chu, H. Targeting Gut Microbiota for the Treatment of Primary Biliary Cholangitis: From Bench to Bedside. J. Clin. Transl. Hepatol. 2023, 11, 958–966. [Google Scholar] [CrossRef]
- Korbecki, J.; Bajdak-Rusinek, K.; Kupnicka, P.; Kapczuk, P.; Simińska, D.; Chlubek, D.; Baranowska-Bosiacka, I. The Role of CXCL16 in the Pathogenesis of Cancer and Other Diseases. Int. J. Mol. Sci. 2021, 22, 3490. [Google Scholar] [CrossRef]
- Ma, C.; Han, M.; Heinrich, B.; Fu, Q.; Zhang, Q.; Sandhu, M.; Agdashian, D.; Terabe, M.; Berzofsky, J.A.; Fako, V.; et al. Gut Microbiome-Mediated Bile Acid Metabolism Regulates Liver Cancer via NKT Cells. Science 2018, 360, eaan5931. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.M.; Mohs, A.; Gui, W.; Galvez, E.J.C.; Candels, L.S.; Hoenicke, L.; Muthukumarasamy, U.; Holland, C.H.; Elfers, C.; Kilic, K.; et al. Imbalanced Gut Microbiota Fuels Hepatocellular Carcinoma Development by Shaping the Hepatic Inflammatory Microenvironment. Nat. Commun. 2022, 13, 3964. [Google Scholar] [CrossRef] [PubMed]
- Ponziani, F.R.; Bhoori, S.; Castelli, C.; Putignani, L.; Rivoltini, L.; Del Chierico, F.; Sanguinetti, M.; Morelli, D.; Paroni Sterbini, F.; Petito, V.; et al. Hepatocellular Carcinoma Is Associated with Gut Microbiota Profile and Inflammation in Nonalcoholic Fatty Liver Disease. Hepatology 2019, 69, 107. [Google Scholar] [CrossRef] [PubMed]
- Khor, S.-S.; Ueno, K.; Nishida, N.; Kawashima, M.; Kawai, Y.; Aiba, Y.; Hitomi, Y.; Nagasaki, M.; Nakamura, M.; Tokunaga, K. Novel HLA Allele Associations with Susceptibility, Staging, Symptomatic State, Autoimmune Hepatitis and Hepatocellular Carcinoma Events for Primary Biliary Cholangitis in the Japanese Population. Front. Immunol. 2023, 14, 1151502. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zheng, X.; Tang, R.; Han, C.; Jiang, Y.; Wu, J.; Shao, Y.; Gao, Y.; Yu, J.; Hu, Z.; et al. Fine Mapping of the MHC Region Identifies Major Independent Variants Associated with Han Chinese Primary Biliary Cholangitis. J. Autoimmun. 2020, 107, 102372. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, P.T.; Baragiotta, A.; Heneghan, M.A.; Floreani, A.; Venturi, C.; Underhill, J.A.; Jones, D.E.J.; James, O.F.W.; Bassendine, M.F. HLA Class II Alleles, Genotypes, Haplotypes, and Amino Acids in Primary Biliary Cirrhosis: A Large-Scale Study. Hepatology 2006, 44, 667–674. [Google Scholar] [CrossRef]
- Invernizzi, P.; Selmi, C.; Poli, F.; Frison, S.; Floreani, A.; Alvaro, D.; Almasio, P.; Rosina, F.; Marzioni, M.; Fabris, L.; et al. Human Leukocyte Antigen Polymorphisms in Italian Primary Biliary Cirrhosis: A Multicenter Study of 664 Patients and 1992 Healthy Controls. Hepatology 2008, 48, 1906–1912. [Google Scholar] [CrossRef]
- Yasunami, M.; Nakamura, H.; Tokunaga, K.; Kawashima, M.; Nishida, N.; Hitomi, Y.; Nakamura, M. Principal Contribution of HLA-DQ Alleles, DQB1*06:04 and DQB1*03:01, to Disease Resistance against Primary Biliary Cholangitis in a Japanese Population. Sci. Rep. 2017, 7, 11093. [Google Scholar] [CrossRef]
- Terziroli Beretta-Piccoli, B.; Mieli-Vergani, G.; Vergani, D. HLA, Gut Microbiome and Hepatic Autoimmunity. Front. Immunol. 2022, 13, 980768. [Google Scholar] [CrossRef]
- Lin, Z.-H.; Xin, Y.-N.; Dong, Q.-J.; Wang, Q.; Jiang, X.-J.; Zhan, S.-H.; Sun, Y.; Xuan, S.-Y. Association between HLA-DRB1 Alleles Polymorphism and Hepatocellular Carcinoma: A Meta-Analysis. BMC Gastroenterol. 2010, 10, 145. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Guo, W.; Zhang, J. Association of HLA-DRB1 Gene Polymorphisms with Hepatocellular Carcinoma Risk: A Meta-Analysis. Minerva Med. 2017, 108, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Pan, T.; Ye, Y.; Ge, G.; Chen, L.; Wen, D.; Zou, S. Serum MicroRNAs as Potential Biomarkers of Primary Biliary Cirrhosis. PLoS ONE 2014, 9, e111424. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiao, Z.; Chen, M.; Shen, B.; Shuai, Z. Roles of Non-Coding RNAs in Primary Biliary Cholangitis. Front. Mol. Biosci. 2022, 9, 915993. [Google Scholar] [CrossRef]
- Coulouarn, C.; Factor, V.M.; Andersen, J.B.; Durkin, M.E.; Thorgeirsson, S.S. Loss of miR-122 Expression in Liver Cancer Correlates with Suppression of the Hepatic Phenotype and Gain of Metastatic Properties. Oncogene 2009, 28, 3526–3536. [Google Scholar] [CrossRef]
- Hayes, C.N.; Chayama, K. MicroRNAs as Biomarkers for Liver Disease and Hepatocellular Carcinoma. Int. J. Mol. Sci. 2016, 17, 280. [Google Scholar] [CrossRef] [PubMed]
- Sayiner, M.; Golabi, P.; Younossi, Z.M. Disease Burden of Hepatocellular Carcinoma: A Global Perspective. Dig. Dis. Sci. 2019, 64, 910–917. [Google Scholar] [CrossRef]
- Goldacre, M.J.; Wotton, C.J.; Yeates, D.; Seagroatt, V.; Collier, J. Liver Cirrhosis, Other Liver Diseases, Pancreatitis and Subsequent Cancer: Record Linkage Study. Eur. J. Gastroenterol. Hepatol. 2008, 20, 384–392. [Google Scholar] [CrossRef]
- Floreani, A.; Baragiotta, A.; Baldo, V.; Menegon, T.; Farinati, F.; Naccarato, R. Hepatic and Extrahepatic Malignancies in Primary Biliary Cirrhosis. Hepatology 1999, 29, 1425–1428. [Google Scholar] [CrossRef]
- Cavazza, A.; Caballería, L.; Floreani, A.; Farinati, F.; Bruguera, M.; Caroli, D.; Parés, A. Incidence, Risk Factors, and Survival of Hepatocellular Carcinoma in Primary Biliary Cirrhosis: Comparative Analysis from Two Centers. Hepatology 2009, 50, 1162–1168. [Google Scholar] [CrossRef]
- Jones, D.E.; Metcalf, J.V.; Collier, J.D.; Bassendine, M.F.; James, O.F. Hepatocellular Carcinoma in Primary Biliary Cirrhosis and Its Impact on Outcomes. Hepatology 1997, 26, 1138–1142. [Google Scholar] [CrossRef]
- Nijhawan, P.K.; Therneau, T.M.; Dickson, E.R.; Boynton, J.; Lindor, K.D. Incidence of Cancer in Primary Biliary Cirrhosis: The Mayo Experience. Hepatology 1999, 29, 1396–1398. [Google Scholar] [CrossRef]
- Shibuya, A.; Tanaka, K.; Miyakawa, H.; Shibata, M.; Takatori, M.; Sekiyama, K.; Hashimoto, N.; Amaki, S.; Komatsu, T.; Morizane, T. Hepatocellular Carcinoma and Survival in Patients with Primary Biliary Cirrhosis. Hepatology 2002, 35, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- Howel, D.; Metcalf, J.V.; Gray, J.; Newman, W.L.; Jones, D.E.; James, O.F. Cancer Risk in Primary Biliary Cirrhosis: A Study in Northern England. Gut 1999, 45, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Jackson, H.; Solaymani-Dodaran, M.; Card, T.R.; Aithal, G.P.; Logan, R.; West, J. Influence of Ursodeoxycholic Acid on the Mortality and Malignancy Associated with Primary Biliary Cirrhosis: A Population-Based Cohort Study. Hepatology 2007, 46, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Su, C.-W.; Hung, H.-H.; Huo, T.-I.; Huang, Y.-H.; Li, C.-P.; Lin, H.-C.; Lee, P.-C.; Lee, S.-D.; Wu, J.-C. Natural History and Prognostic Factors of Primary Biliary Cirrhosis in Taiwan: A Follow-up Study up to 18 Years. Liver Int. 2008, 28, 1305–1313. [Google Scholar] [CrossRef] [PubMed]
- Deutsch, M.; Papatheodoridis, G.V.; Tzakou, A.; Hadziyannis, S.J. Risk of Hepatocellular Carcinoma and Extrahepatic Malignancies in Primary Biliary Cirrhosis. Eur. J. Gastroenterol. Hepatol. 2008, 20, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, E.M.M.; Hansen, B.E.; Adang, R.P.R.; van Nieuwkerk, C.M.J.; Timmer, R.; Drenth, J.P.H.; Spoelstra, P.; Brouwer, H.T.; Kuyvenhoven, J.P.H.; van Buuren, H.R.; et al. Relatively High Risk for Hepatocellular Carcinoma in Patients with Primary Biliary Cirrhosis Not Responding to Ursodeoxycholic Acid. Eur. J. Gastroenterol. Hepatol. 2010, 22, 1495–1502. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Nakanuma, Y. Prevalence and Risk Factors of Hepatocellular Carcinoma in Japanese Patients with Primary Biliary Cirrhosis. Hepatol. Res. 2014, 44, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.J.; Lammers, W.J.; van Buuren, H.R.; Parés, A.; Floreani, A.; Janssen, H.L.A.; Invernizzi, P.; Battezzati, P.M.; Ponsioen, C.Y.; Corpechot, C.; et al. Stratification of Hepatocellular Carcinoma Risk in Primary Biliary Cirrhosis: A Multicentre International Study. Gut 2016, 65, 321–329. [Google Scholar] [CrossRef]
- Rong, G.; Wang, H.; Bowlus, C.L.; Wang, C.; Lu, Y.; Zeng, Z.; Qu, J.; Lou, M.; Chen, Y.; An, L.; et al. Incidence and Risk Factors for Hepatocellular Carcinoma in Primary Biliary Cirrhosis. Clin. Rev. Allergy Immunol. 2015, 48, 132–141. [Google Scholar] [CrossRef]
- Zhang, X.-X.; Wang, L.-F.; Jin, L.; Li, Y.-Y.; Hao, S.-L.; Shi, Y.-C.; Zeng, Q.-L.; Li, Z.-W.; Zhang, Z.; Lau, G.K.; et al. Primary Biliary Cirrhosis-Associated Hepatocellular Carcinoma in Chinese Patients: Incidence and Risk Factors. World J. Gastroenterol. 2015, 21, 3554–3563. [Google Scholar] [CrossRef]
- Cheng, J.-S.; Chen, W.-T.; Ku, H.-P.; Chien, R.-N.; Chang, M.-L. Characteristic Geoepidemiology of Primary Biliary Cholangitis in Taiwan: A Nationwide Population-Based Study. Hepatol. Res. 2023, 53, 866–877. [Google Scholar] [CrossRef]
- Boonstra, K.; Bokelaar, R.; Stadhouders, P.H.; Tuynman, H.A.; Poen, A.C.; van Nieuwkerk, K.M.; Witteman, E.M.; Hamann, D.; Witteman, B.J.; Beuers, U.; et al. Increased Cancer Risk in a Large Population-Based Cohort of Patients with Primary Biliary Cirrhosis: Follow-up for up to 36 Years. Hepatol. Int. 2014, 8, 266–274. [Google Scholar] [CrossRef]
- Natarajan, Y.; Tansel, A.; Patel, P.; Emologu, K.; Shukla, R.; Qureshi, Z.; El-Serag, H.B.; Thrift, A.P.; Kanwal, F. Incidence of Hepatocellular Carcinoma in Primary Biliary Cholangitis: A Systematic Review and Meta-Analysis. Dig. Dis. Sci. 2021, 66, 2439–2451. [Google Scholar] [CrossRef]
- Anstee, Q.M.; Reeves, H.L.; Kotsiliti, E.; Govaere, O.; Heikenwalder, M. From NASH to HCC: Current Concepts and Future Challenges. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 411–428. [Google Scholar] [CrossRef]
- Farinati, F.; Floreani, A.; De Maria, N.; Fagiuoli, S.; Naccarato, R.; Chiaramonte, M. Hepatocellular Carcinoma in Primary Biliary Cirrhosis. J. Hepatol. 1994, 21, 315–316. [Google Scholar] [CrossRef] [PubMed]
- Giannini, E.G.; Pieri, G.; Labanca, S.; Plaz Torres, M.C.; Gasbarrini, A.; Biasini, E.; Campani, C.; Cazzagon, N.; Foschi, F.G.; Mega, A.; et al. Characteristics and Survival of Patients with Primary Biliary Cholangitis and Hepatocellular Carcinoma. Dig. Liver Dis. 2022, 54, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Rigopoulou, E.I.; Dalekos, G.N. Current Trends and Characteristics of Hepatocellular Carcinoma in Patients with Autoimmune Liver Diseases. Cancers 2021, 13, 1023. [Google Scholar] [CrossRef] [PubMed]
- Kulik, L.; El-Serag, H.B. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology 2019, 156, 477–491.e1. [Google Scholar] [CrossRef]
- Yeh, S.-H.; Chen, P.-J. Gender Disparity of Hepatocellular Carcinoma: The Roles of Sex Hormones. Oncology 2010, 78 (Suppl. S1), 172–179. [Google Scholar] [CrossRef] [PubMed]
- Kido, T.; Lo, R.C.-L.; Li, Y.; Lee, J.; Tabatabai, Z.L.; Ng, I.O.; Lau, Y.-F.C. The Potential Contributions of a Y-Located Protooncogene and Its X Homologue in Sexual Dimorphisms in Hepatocellular Carcinoma. Hum. Pathol. 2014, 45, 1847–1858. [Google Scholar] [CrossRef]
- Li, Z.; Tuteja, G.; Schug, J.; Kaestner, K.H. Foxa1 and Foxa2 Are Essential for Sexual Dimorphism in Liver Cancer. Cell 2012, 148, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Ji, F.; Zhang, J.; Liu, N.; Gu, Y.; Zhang, Y.; Huang, P.; Zhang, N.; Lin, S.; Pan, R.; Meng, Z.; et al. Blocking Hepatocarcinogenesis by a Cytochrome P450 Family Member with Female-Preferential Expression. Gut 2022, 71, 2313–2324. [Google Scholar] [CrossRef] [PubMed]
- Gajos-Michniewicz, A.; Czyz, M. WNT/β-Catenin Signaling in Hepatocellular Carcinoma: The Aberrant Activation, Pathogenic Roles, and Therapeutic Opportunities. Genes Dis. 2024, 11, 727–746. [Google Scholar] [CrossRef] [PubMed]
- Natri, H.M.; Wilson, M.A.; Buetow, K.H. Distinct Molecular Etiologies of Male and Female Hepatocellular Carcinoma. BMC Cancer 2019, 19, 951. [Google Scholar] [CrossRef]
- Leung, R.W.H.; Lee, T.K.W. Wnt/β-Catenin Signaling as a Driver of Stemness and Metabolic Reprogramming in Hepatocellular Carcinoma. Cancers 2022, 14, 5468. [Google Scholar] [CrossRef] [PubMed]
- Bhat, M.; Pasini, E.; Pastrello, C.; Angeli, M.; Baciu, C.; Abovsky, M.; Coffee, A.; Adeyi, O.; Kotlyar, M.; Jurisica, I. Estrogen Receptor 1 Inhibition of Wnt/β-Catenin Signaling Contributes to Sex Differences in Hepatocarcinogenesis. Front. Oncol. 2021, 11, 4878. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Sanchez, E.; Vaquero, J.; Férnandez-Barrena, M.G.; Lasarte, J.J.; Avila, M.A.; Sarobe, P.; Reig, M.; Calvo, M.; Fabregat, I. The TGF-β Pathway: A Pharmacological Target in Hepatocellular Carcinoma? Cancers 2021, 13, 3248. [Google Scholar] [CrossRef]
- Xu, L.; Yuan, Y.; Che, Z.; Tan, X.; Wu, B.; Wang, C.; Xu, C.; Xiao, J. The Hepatoprotective and Hepatotoxic Roles of Sex and Sex-Related Hormones. Front. Immunol. 2022, 13, 939631. [Google Scholar] [CrossRef]
- Rizzo, G.E.M.; Cabibbo, G.; Craxì, A. Hepatitis B Virus-Associated Hepatocellular Carcinoma. Viruses 2022, 14, 986. [Google Scholar] [CrossRef]
- Kaur, S.P.; Talat, A.; Karimi-Sari, H.; Grees, A.; Chen, H.W.; Lau, D.T.Y.; Catana, A.M. Hepatocellular Carcinoma in Hepatitis B Virus-Infected Patients and the Role of Hepatitis B Surface Antigen (HBsAg). J. Clin. Med. 2022, 11, 1126. [Google Scholar] [CrossRef]
- Kumada, T.; Toyoda, H.; Yasuda, S.; Ito, T.; Tsuji, K.; Fujioka, S.; Hiraoka, A.; Kariyama, K.; Nouso, K.; Ishikawa, T.; et al. Factors Linked to Hepatocellular Carcinoma Development beyond 10 Years after Viral Eradication in Patients with Hepatitis C Virus. J. Viral Hepat. 2022, 29, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Floreani, A.; Baragiotta, A.; Leone, M.G.; Baldo, V.; Naccarato, R. Primary Biliary Cirrhosis and Hepatitis C Virus Infection. Off. J. Am. Coll. Gastroenterol. ACG 2003, 98, 2757. [Google Scholar] [CrossRef]
- Chen, H.-W.; Huang, H.-H.; Lai, C.-H.; Chang, W.-E.; Shih, Y.-L.; Chang, W.-K.; Hsieh, T.-Y.; Chu, H.-C. Hepatitis C Virus Infection in Patients with Primary Biliary Cirrhosis. Ann. Hepatol. 2013, 12, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, E.M.M.; Hansen, B.E.; de Vries, R.A.; den Ouden–Muller, J.W.; van Ditzhuijsen, T.J.M.; Haagsma, E.B.; Houben, M.H.M.G.; Witteman, B.J.M.; van Erpecum, K.J.; van Buuren, H.R. Improved Prognosis of Patients with Primary Biliary Cirrhosis That Have a Biochemical Response to Ursodeoxycholic Acid. Gastroenterology 2009, 136, 1281–1287. [Google Scholar] [CrossRef]
- John, B.V.; Khakoo, N.S.; Schwartz, K.B.; Aitchenson, G.; Levy, C.; Dahman, B.; Deng, Y.; Goldberg, D.S.; Martin, P.; Kaplan, D.E.; et al. Ursodeoxycholic Acid Response Is Associated with Reduced Mortality in Primary Biliary Cholangitis with Compensated Cirrhosis. Off. J. Am. Coll. Gastroenterol. ACG 2021, 116, 1913. [Google Scholar] [CrossRef]
- The Lancet. Alcohol and Cancer. Lancet 2017, 390, 2215. [Google Scholar] [CrossRef]
- Pöschl, G.; Seitz, H.K. Alcohol and Cancer. Alcohol Alcohol. 2004, 39, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, C.J.P. Genetic-Epidemiological Evidence for the Role of Acetaldehyde in Cancers Related to Alcohol Drinking. Adv. Exp. Med. Biol. 2015, 815, 41–58. [Google Scholar] [CrossRef]
- Meadows, G.G.; Zhang, H. Effects of Alcohol on Tumor Growth, Metastasis, Immune Response, and Host Survival. Alcohol Res. 2015, 37, 311–322. [Google Scholar]
- Híndi, M.; Levy, C.; Couto, C.A.; Bejarano, P.; Mendes, F. Primary Biliary Cirrhosis Is More Severe in Overweight Patients. J. Clin. Gastroenterol. 2013, 47, e28–e32. [Google Scholar] [CrossRef] [PubMed]
- Doycheva, I.; Zhang, T.; Amjad, W.; Thuluvath, P.J. Diabetes and Hepatocellular Carcinoma: Incidence Trends and Impact of Liver Disease Etiology. J. Clin. Exp. Hepatol. 2020, 10, 296–303. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of Hepatocellular Carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Omata, M.; Cheng, A.-L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.-H.; Chawla, Y.K.; Shiina, S.; et al. Asia–Pacific Clinical Practice Guidelines on the Management of Hepatocellular Carcinoma: A 2017 Update. Hepatol. Int. 2017, 11, 317–370. [Google Scholar] [CrossRef] [PubMed]
- Marrero, J.A.; Kulik, L.M.; Sirlin, C.B.; Zhu, A.X.; Finn, R.S.; Abecassis, M.M.; Roberts, L.R.; Heimbach, J.K. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 2018, 68, 723–750. [Google Scholar] [CrossRef] [PubMed]
- Lleo, A.; de Boer, Y.S.; Liberal, R.; Colombo, M. The Risk of Liver Cancer in Autoimmune Liver Diseases. Ther. Adv. Med. Oncol. 2019, 11, 1758835919861914. [Google Scholar] [CrossRef] [PubMed]
- Tzartzeva, K.; Obi, J.; Rich, N.E.; Parikh, N.D.; Marrero, J.A.; Yopp, A.; Waljee, A.K.; Singal, A.G. Surveillance Imaging and Alpha Fetoprotein for Early Detection of Hepatocellular Carcinoma in Patients with Cirrhosis: A Meta-Analysis. Gastroenterology 2018, 154, 1706–1718.e1. [Google Scholar] [CrossRef]
- Surdea-Blaga, T.; Cărăguț, R.L.; Caraiani, C.; Spârchez, Z.; Al Hajjar, N.; Dumitrașcu, D.L. Overlap Syndrome of Autoimmune Hepatitis and Primary Biliary Cholangitis Complicated with Atypical Hepatocellular Carcinoma: A Case Report. J. Med. Case Rep. 2023, 17, 328. [Google Scholar] [CrossRef]
- Carbone, M.; Neuberger, J. Liver Transplantation in PBC and PSC: Indications and Disease Recurrence. Clin. Res. Hepatol. Gastroenterol. 2011, 35, 446–454. [Google Scholar] [CrossRef]
- Imam, M.H.; Silveira, M.G.; Sinakos, E.; Gossard, A.A.; Jorgensen, R.; Keach, J.; DeCook, A.C.; Lindor, K.D. Long-Term Outcomes of Patients with Primary Biliary Cirrhosis and Hepatocellular Carcinoma. Clin. Gastroenterol. Hepatol. 2012, 10, 182–185. [Google Scholar] [CrossRef]
- Mijic, M.; Saric, I.; Delija, B.; Lalovac, M.; Sobocan, N.; Radetic, E.; Martincevic, D.; Filipec Kanizaj, T. Pretransplant Evaluation and Liver Transplantation Outcome in PBC Patients. Can. J. Gastroenterol. Hepatol. 2022, 2022, 7831165. [Google Scholar] [CrossRef]
- Trivella, J.; John, B.V.; Levy, C. Primary Biliary Cholangitis: Epidemiology, Prognosis, and Treatment. Hepatol. Commun. 2023, 7, e0179. [Google Scholar] [CrossRef]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular Carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef] [PubMed]
- Petrelli, F.; Manara, M.; Colombo, S.; De Santi, G.; Ghidini, M.; Mariani, M.; Iaculli, A.; Rausa, E.; Rampulla, V.; Arru, M.; et al. Hepatocellular Carcinoma in Patients with Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis. Neoplasia 2022, 30, 100809. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.A.; Patil, R.; Harrison, S.A. NAFLD-related Hepatocellular Carcinoma: The Growing Challenge. Hepatology 2023, 77, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Orci, L.A.; Sanduzzi-Zamparelli, M.; Caballol, B.; Sapena, V.; Colucci, N.; Torres, F.; Bruix, J.; Reig, M.; Toso, C. Incidence of Hepatocellular Carcinoma in Patients with Nonalcoholic Fatty Liver Disease: A Systematic Review, Meta-Analysis, and Meta-Regression. Clin. Gastroenterol. Hepatol. 2022, 20, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Floreani, A.; Cazzagon, N.; Franceschet, I.; Canesso, F.; Salmaso, L.; Baldo, V. Metabolic Syndrome Associated with Primary Biliary Cirrhosis. J. Clin. Gastroenterol. 2015, 49, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Calderaro, J.; Seraphin, T.P.; Luedde, T.; Simon, T.G. Artificial Intelligence for the Prevention and Clinical Management of Hepatocellular Carcinoma. J. Hepatol. 2022, 76, 1348–1361. [Google Scholar] [CrossRef]
| Study | Country | Study Period | n. PBC Patients | n. HCC Cases |
|---|---|---|---|---|
| Goldacre MJ [67] | UK | 1963–1999 | 424 | 8 |
| Floreani A [68] | Italy | 1973–1996 | 175 | 4 |
| Cavazza A [69] | Spain | NA | 389 | 13 |
| Cavazza A [69] | Italy | 1973–2007 | 327 | 11 |
| Jones D [70] | UK | 1975–1995 | 667 | 16 |
| Nijhawan P [71] | USA | 1976–1985 | 1692 | 12 |
| Shibuya A [72] | Japan | 1980–1998 | 396 | 14 |
| Howel D [73] | UK | 1987–1994 | 769 | 7 |
| Jackson H [74] | UK | 1987–2002 | 930 | 7 |
| Su C-W [75] | Taiwan | 1985–2006 | 96 | 5 |
| Deutsch M [76] | Greece | 1987–2005 | 212 | 8 |
| Kuiper EM [77] | The Netherland | 1990–2007 | 375 | 9 |
| Harada H [78] | Japan | 1980–2009 | 2946 | 71 |
| Trivedi PJ [79] | Global PBC | 1959–2012 | 4565 | 123 |
| Rong G [80] | China | 1994–2014 | 1865 | 70 |
| Braga MH [15] | Brasil | 1992–2020 | 752 | 20 |
| Zhang X-X [81] | China | 2002–2013 | 1255 | 52 |
| Cheng J-S [82] | Taiwan | 2002–2015 | 2737 | 146 |
| Boonstra K [83] | The Netherland | 2008–2011 | 992 | 7 |
| Study | % HCC Cases/PBC Patients | Histological Stage | Risk Factors |
|---|---|---|---|
| Cheng J-S [82] | 5.33 | NA | Male sex |
| Zhang X-X [81] | 4.14 | 100% cirrhosis | BMI ≥, alcohol intake |
| Deutsch M [76] | 3.77 | IV | Advanced histological stage |
| Rong G [80] | 3.75 | 80% with III/IV stage | Advanced age, male sex, co-existence of diabetes, history of HBV infection |
| Shibuya A [72] | 3.54 | III-IV | Male gender, age at diagnosis, history of blood transfusion |
| Cavazza A [69] | 3.36 | III-IV | Male gender |
| Trivedi PJ [79] | 2.69 | 42% with advanced disease | Advanced age, male sex, thrombocytopenia at 12 months, non-response to UDCA |
| Braga MH [15] | 2.66 | 95% with cirrhosis | Cirrhosis, obesity, prior azathioprine use |
| Harada H [78] | 2.41 | 10 I/17 II/14 III/8 IV | Male sex, advanced histological stage (in females) |
| Kuiper EM [77] | 2.40 | Lack of response to UDCA | |
| Jones D [70] | 2.40 | IV | Male gender |
| Floreani A [68] | 2.29 | IV | HCV, smoking |
| Jackson H [74] | 0.75 | NA | UDCA seems protective |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Floreani, A.; Gabbia, D.; De Martin, S. Current Perspectives on the Molecular and Clinical Relationships between Primary Biliary Cholangitis and Hepatocellular Carcinoma. Int. J. Mol. Sci. 2024, 25, 2194. https://doi.org/10.3390/ijms25042194
Floreani A, Gabbia D, De Martin S. Current Perspectives on the Molecular and Clinical Relationships between Primary Biliary Cholangitis and Hepatocellular Carcinoma. International Journal of Molecular Sciences. 2024; 25(4):2194. https://doi.org/10.3390/ijms25042194
Chicago/Turabian StyleFloreani, Annarosa, Daniela Gabbia, and Sara De Martin. 2024. "Current Perspectives on the Molecular and Clinical Relationships between Primary Biliary Cholangitis and Hepatocellular Carcinoma" International Journal of Molecular Sciences 25, no. 4: 2194. https://doi.org/10.3390/ijms25042194
APA StyleFloreani, A., Gabbia, D., & De Martin, S. (2024). Current Perspectives on the Molecular and Clinical Relationships between Primary Biliary Cholangitis and Hepatocellular Carcinoma. International Journal of Molecular Sciences, 25(4), 2194. https://doi.org/10.3390/ijms25042194








