Synergistic Effect of Nilavembu Choornam–Gold Nanoparticles on Antibiotic-Resistant Bacterial Susceptibility and Contact Lens Contamination-Associated Infectious Pathogenicity
Abstract
1. Introduction
2. Results
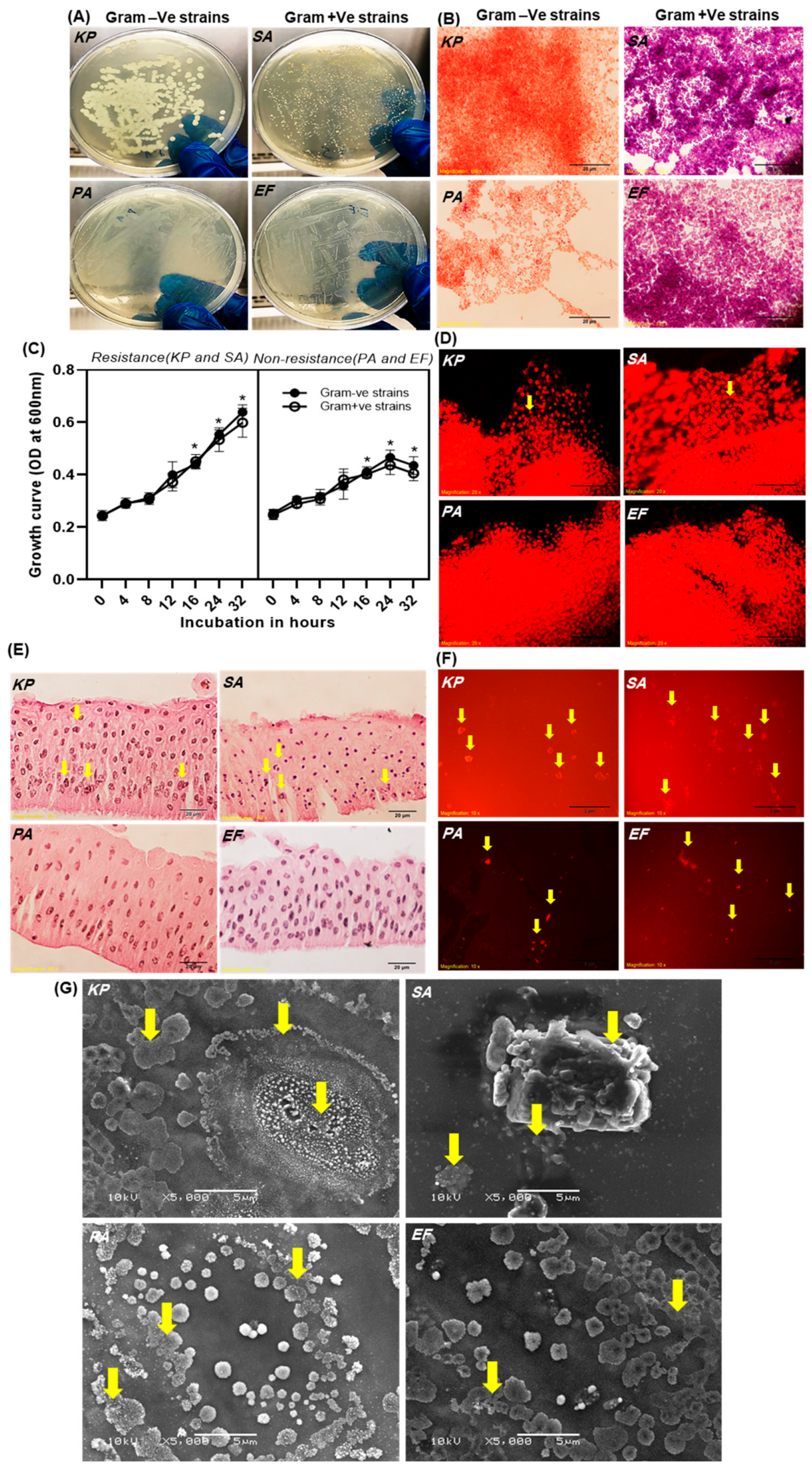
2.1. Determination of Bacterial Infection and Biofilm Formation in Contact Lenses
2.2. Antibacterial Growth Inhibition of Nilavembu Choornam Decoction (NCt)
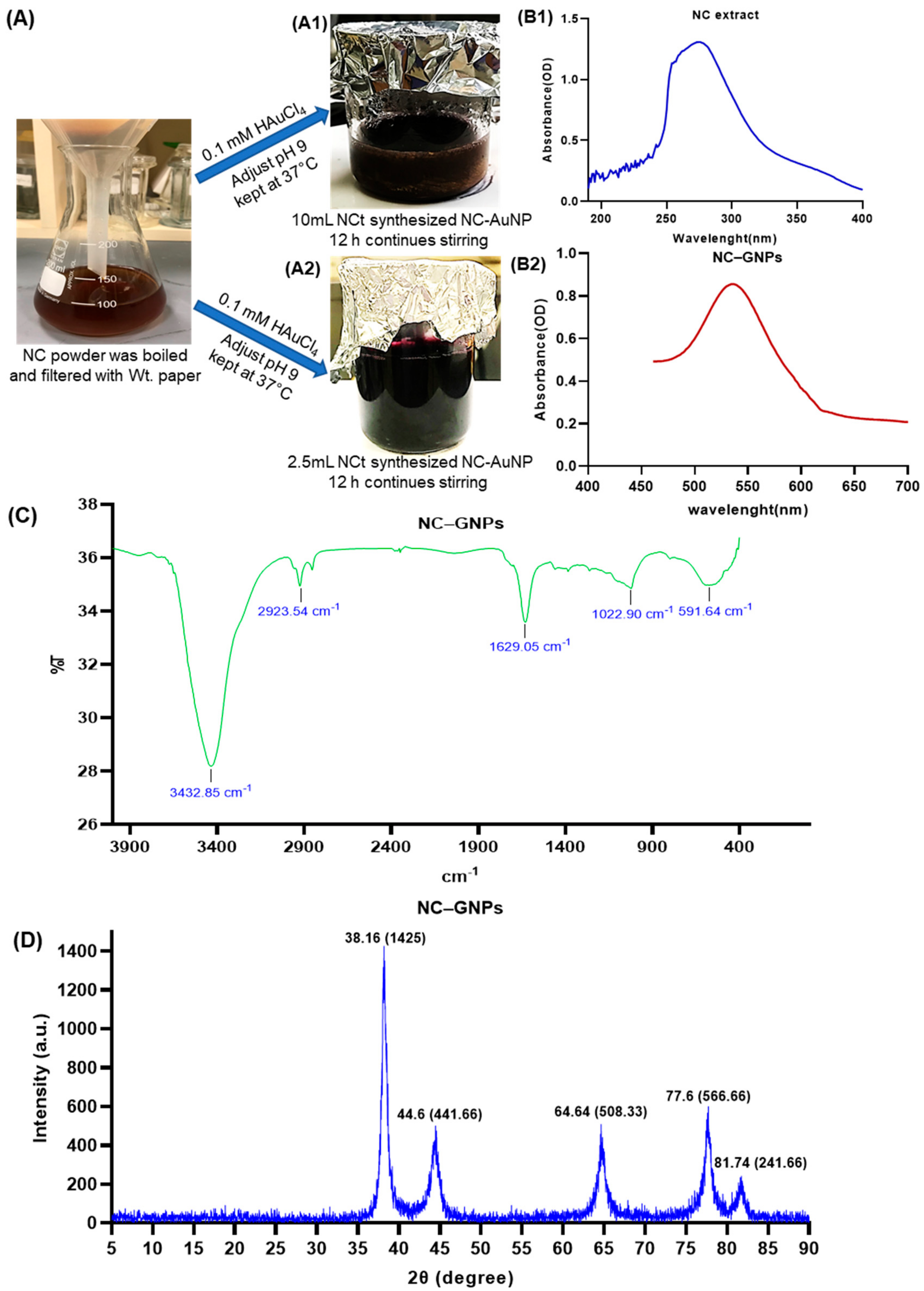
2.3. Synthesis of NC–GNPs
2.4. Characterization of Synthesized NC–GNPs
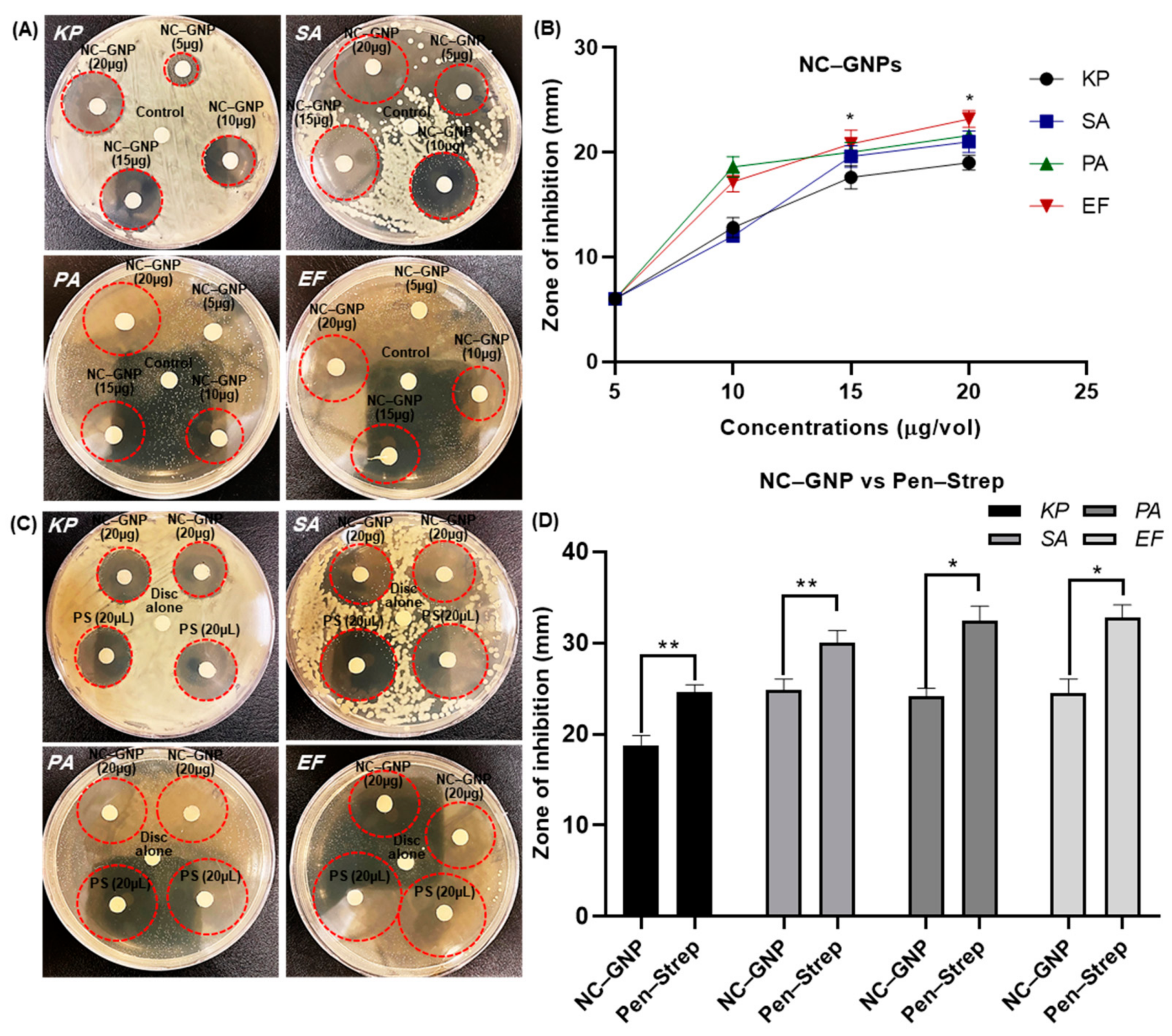
2.5. Growth Inhibition of NC–GNPs in Antibiotic Resistance Bacterial Strains
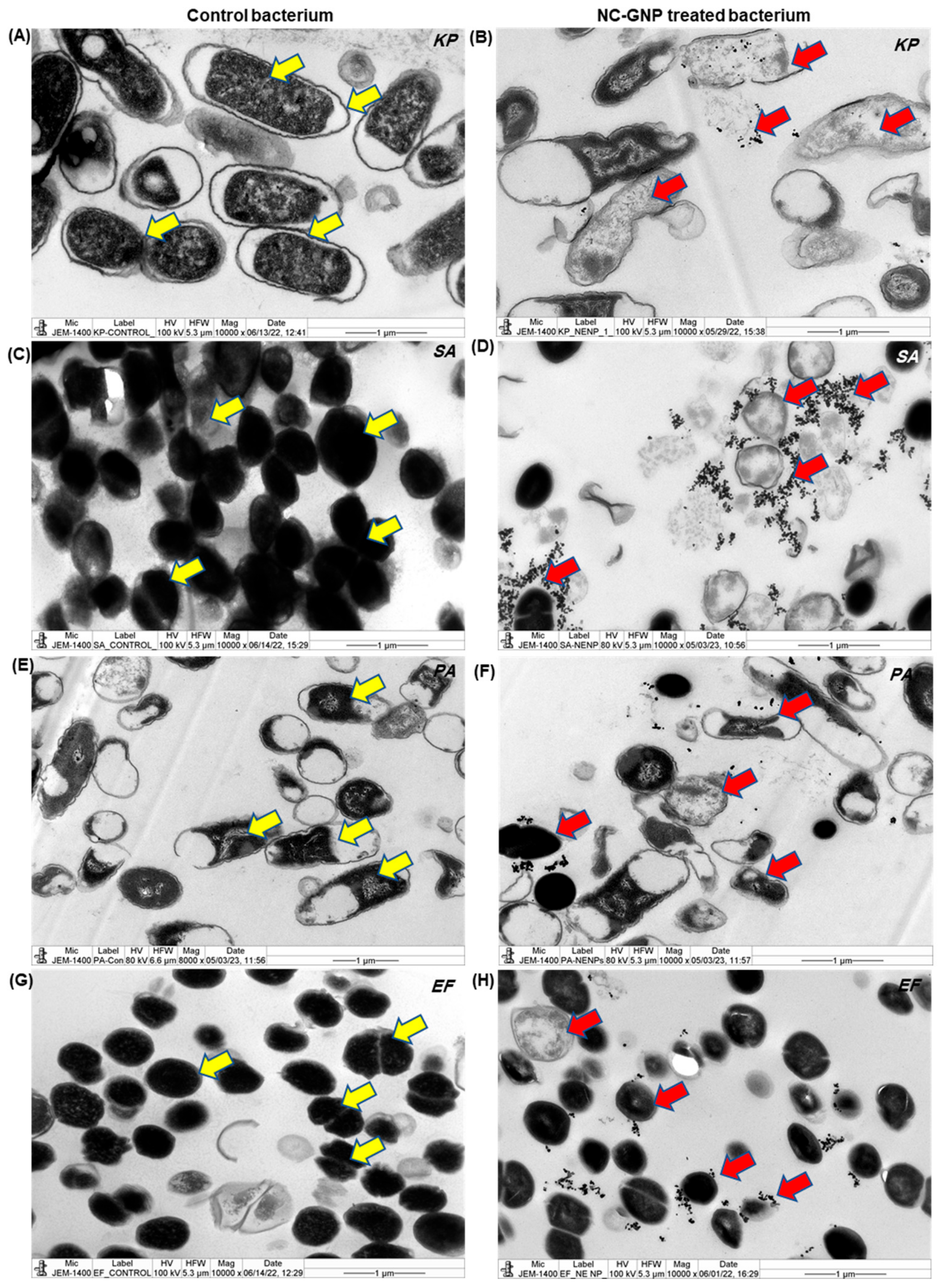
2.6. Ultrastructural and Cellular Degeneration of NC–GNPs
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Bacterial Strains and Contact Lenses
4.3. Biofilm Formation and Quantification
4.4. Assessment of Bacterial Infection in Bovine Cornea and Corneal Epithelial Cells
4.5. Scanning Electron Microscopy (SEM) Analysis
4.6. Preparation and Synthesis of Nilavembu Choornam Extracts (NCt) and NC–GNPs
4.7. Characterization of NC–GNPs
4.8. Antimicrobial Sustainability Assay
4.9. Transmission Electron Microscopy Analysis
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weiss, J.S.; Møller, H.U.; Aldave, A.J.; Seitz, B.; Bredrup, C.; Kivelä, T.; Busin, M.; Labbé, A.; Kenyon, K.R.; Lisch, W.; et al. IC3D classification of corneal dystrophies—Edition 2. Cornea 2015, 34, 117–159. [Google Scholar] [CrossRef]
- Rhee, M.K.; Jacobs, D.S.; Dhaliwal, D.K.; Szczotka-Flynn, L.; Prescott, C.R.; Jhanji, V.; Steinemann, T.L.; Koffler, B.H.; Jeng, B.H. Contact lens safety for the correction of refractive error in healthy eyes. Eye Contact Lens 2022, 48, 449. [Google Scholar] [CrossRef]
- Fleiszig, S.M.J.; Kroken, A.R.; Nieto, V.; Grosser, M.R.; Wan, S.J.; Metruccio, M.M.E.; Evans, D.J. Contact lens-related corneal infection: Intrinsic resistance and its compromise. Prog. Retin. Eye Res. 2020, 76, 100804. [Google Scholar] [CrossRef]
- Alipour, F.; Khaheshi, S.; Soleimanzadeh, M.; Heidarzadeh, S.; Heydarzadeh, S. Contact Lens-related Complications: A Review. J. Ophthalmic. Vis. Res. 2017, 12, 193–204. [Google Scholar] [PubMed]
- Shin, H.; Price, K.; Albert, L.; Dodick, J.; Park, L.; Dominguez-Bello, M.G. Changes in the eye microbiota associated with contact lens wearing. MBio 2016, 7, 1110–1128. [Google Scholar] [CrossRef] [PubMed]
- Udomwech, L.; Karnjana, K.; Jewboonchu, J.; Rattanathamma, P.; Narkkul, U.; Juhong, J.; Mordmuang, A. Bacterial microbiota of the contact lens surface and associated care behaviours. Heliyon 2022, 8, e09038. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, A.B.; Nixon, A.D.; Rueff, E.M. Contact lens associated microbial keratitis: Practical considerations for the optometrist. Clin. Optom. 2016, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, R.L.; Wagner, H.; Mitchell, G.L.; Lam, D.Y.; Kinoshita, B.T.; Jansen, M.E.; Richdale, K.; Sorbara, L.; McMahon, T.T. Age and other risk factors for corneal infiltrative and inflammatory events in young soft contact lens wearers from the Contact Lens Assessment in Youth (CLAY) study. Investig. Ophthalmol. Vis. Sci. 2011, 52, 6690–6696. [Google Scholar] [CrossRef]
- Masters, J.; Kocak, M.; Waite, A. Risk for microbial keratitis: Comparative metaanalysis of contact lens wearers and post-laser in situ keratomileusis patients. J. Cataract. Refract. Surg. 2017, 43, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Maier, P.; Betancor, P.K.; Reinhard, T. Contact Lens–Associated Keratitis—An Often Underestimated Risk. Dtsch. Ärzteblatt Int. 2022, 119, 669. [Google Scholar] [CrossRef]
- Wang, J.J.; Lai, C.H.; Chen, C.Y.; Liu, C.Y.; Lin, M.H.; Yang, Y.H.; Wu, P.L. Trends in Infectious Keratitis in Taiwan: An Update on Predisposing Factors, Microbiological and Antibiotic Susceptibility Patterns. Diagnostics 2022, 12, 2095. [Google Scholar] [CrossRef]
- Ting, D.S.J.; Ho, C.S.; Deshmukh, R.; Said, D.G.; Dua, H.S. Infectious keratitis: An update on epidemiology, causative microorganisms, risk factors, and antimicrobial resistance. Eye 2021, 35, 1084–1101. [Google Scholar] [CrossRef]
- Taube, M.A.; del Mar Cendra, M.; Elsahn, A.; Christodoulides, M.; Hossain, P. Pattern recognition receptors in microbial keratitis. Eye 2015, 29, 1399–1415. [Google Scholar] [CrossRef]
- Alkatan, H.M.; Al-Essa, R.S. Challenges in the diagnosis of microbial keratitis: A detailed review with update and general guidelines. Saudi J. Ophthalmol. 2019, 33, 268–276. [Google Scholar] [CrossRef]
- Vestby, L.K.; Grønseth, T.; Simm, R.; Nesse, L.L. Bacterial biofilm and its role in the pathogenesis of disease. Antibiotics 2020, 9, 59. [Google Scholar] [CrossRef]
- Hildebrandt, C.; Wagner, D.; Kohlmann, T.; Kramer, A. In-vitro analysis of the microbicidal activity of 6 contact lens care solutions. BMC Infect. Dis. 2012, 12, 241. [Google Scholar] [CrossRef]
- Nie, L.; Li, Y.; Liu, Y.; Shi, L.; Chen, H. Recent applications of contact lenses for bacterial corneal keratitis therapeutics: A review. Pharmaceutics 2022, 14, 2635. [Google Scholar] [CrossRef]
- Cumurcu, T.; Firat, P.; Özsoy, E.; Cavdar, M.; Yakupogullari, Y. Contact-lens-related corneal ulcer caused by klebsiella pneumoniae. Clinics 2011, 66, 1509–1510. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dutt, Y.; Dhiman, R.; Singh, T.; Vibhuti, A.; Gupta, A.; Pandey, R.P.; Chang, C.-M.; Raj, V.S.; Priyadarshini, A. The association between biofilm formation and antimicrobial resistance with possible ingenious bio-remedial approaches. Antibiotics 2022, 11, 930. [Google Scholar] [CrossRef] [PubMed]
- Szczotka-Flynn, L.B.; Imamura, Y.; Chandra, J.; Yu, C.; Mukherjee, P.K.; Pearlman, E.; Ghannoum, M.A. Increased resistance of contact lens related bacterial biofilms to antimicrobial activity of soft contact lens care solutions. Cornea 2009, 28, 918. [Google Scholar] [CrossRef] [PubMed]
- Khan, J.; Tarar, S.M.; Gul, I.; Nawaz, U.; Arshad, M. Challenges of antibiotic resistance biofilms and potential combating strategies: A review. 3 Biotech 2021, 11, 1–15. [Google Scholar] [CrossRef]
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Health 2015, 109, 309–318. [Google Scholar] [CrossRef]
- Kang, Y.C.; Hsiao, C.H.; Yeh, L.K.; Ma, D.H.K.; Chen, P.Y.F.; Lin, H.C.; Tan, H.-Y.; Chen, H.-C.; Chen, S.-Y.; Huang, Y.C.; et al. Methicillin-Resistant Staphylococcus aureus ocular infection in Taiwan: Clinical features, Genotying, and antibiotic susceptibility. Medicine 2015, 94, e1620. [Google Scholar] [CrossRef]
- Franco, P.; De Marco, I. Contact lenses as ophthalmic drug delivery systems: A review. Polymers 2021, 13, 1102. [Google Scholar] [CrossRef]
- Dewi, M.K.; Chaerunisaa, A.Y.; Muhaimin, M.; Joni, I.M. Improved Activity of Herbal Medicines through Nanotechnology. Nanomaterials 2022, 12, 4073. [Google Scholar] [CrossRef]
- Kumar, D.T.; Devi, M.S.S.; Kumar, S.U.; Sherlin, A.; Mathew, A.; Lakshmipriya, M.; Sathiyarajeswaran, P.; Gnanasambandan, R.; Siva, R.; Magesh, R.; et al. Understanding the activating mechanism of the immune system against COVID-19 by Traditional Indian Medicine: Network pharmacology approach. Adv. Protein Chem. Struct. Biol. 2022, 129, 275–379. [Google Scholar]
- Srivastava, A.; Rengaraju, M.; Srivastava, S.; Narayanan, V.; Gupta, V.; Upadhayay, R.; Kumar, J.; Parameswaran, S.; Kadarkarai, K.; Velmurugan, A.; et al. Efficacy of two siddha polyherbal decoctions, Nilavembu Kudineer and Kaba Sura Kudineer, along with standard allopathy treatment in the management of mild to moderate symptomatic COVID-19 patients—A double-blind, placebo-controlled, clinical trial. Trials 2021, 22, 570. [Google Scholar] [CrossRef]
- Jain, J.; Kumar, A.; Narayanan, V.; Ramaswamy, R.S.; Sathiyarajeswaran, P.; Devi, M.S.S.; Kannan, M.; Sunil, S. Antiviral activity of ethanolic extract of Nilavembu Kudineer against dengue and chikungunya virus through in vitro evaluation. J. Ayurveda Integr. Med. 2020, 11, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Alagu Lakshmi, S.; Shafreen, R.M.B.; Priya, A.; Shunmugiah, K.P. Ethnomedicines of Indian origin for combating COVID-19 infection by hampering the viral replication: Using structure-based drug discovery approach. J. Biomol. Struct. Dyn. 2021, 39, 4594–4609. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Zahiruddin, S.; Parveen, B.; Basist, P.; Parveen, A.; Parveen, R.; Ahmad, M. Indian medicinal plants and formulations and their potential against COVID-19–preclinical and clinical research. Front. Pharmacol. 2021, 11, 2470. [Google Scholar] [CrossRef] [PubMed]
- Mekala, P.; Murthy, T.R.G.K. Phytochemical screening and pharmacological update on Kabasura Kudineer Choornam and Nilavembu Kudineer Choornam. J. Pharmacogn. Phytochem. 2020, 9, 1031–1036. [Google Scholar] [CrossRef]
- Ayehubizu, Z.; Mulu, W.; Biadglegne, F. Common bacterial causes of external ocular infections, associated risk factors and antibiotic resistance among patients at ophthalmology unit of Felege Hiwot Referral Hospital, Northwest Ethiopia: A cross-sectional study. J. Ophthalmic Inflamm. Infect. 2021, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Al-Mujaini, A.; Al-Kharusi, N.; Thakral, A.; Wali, U.K. Bacterial keratitis: Perspective on epidemiology, clinico-pathogenesis, diagnosis and treatment. Sultan Qaboos Univ. Med. J. 2009, 9, 184. [Google Scholar] [PubMed]
- Rändler, C.; Matthes, R.; McBain, A.J.; Giese, B.; Fraunholz, M.; Sietmann, R.; Kohlmann, T.; Hübner, N.-O.; Kramer, A. A three-phase in-vitro system for studying Pseudomonas aeruginosa adhesion and biofilm formation upon hydrogel contact lenses. BMC Microbiol. 2010, 10, 282. [Google Scholar] [CrossRef] [PubMed]
- Fey, P.D.; Olson, M.E. Current concepts in biofilm formation of Staphylococcus epidermidis. Future Microbiol. 2010, 5, 917–933. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Gao, X.; Chen, Z.; Liu, G.; Zhang, X.; Yan, H.; Zhao, H.; Zheng, L. The high yield synthesis and characterization of gold nanoparticles with superior stability and their catalytic activity. CrystEngComm 2012, 14, 7600–7606. [Google Scholar] [CrossRef]
- Krishnamurthy, S.; Esterle, A.; Sharma, N.C.; Sahi, S.V. Yucca-derived synthesis of gold nanomaterial and their catalytic potential. Nanoscale Res. Lett. 2014, 9, 627. [Google Scholar] [CrossRef]
- Ahmad, T.; Irfan, M.; Bustam, M.A.; Bhattacharjee, S. Effect of reaction time on green synthesis of gold nanoparticles by using aqueous extract of Elaise guineensis (oil palm leaves). Procedia Eng. 2016, 148, 467–472. [Google Scholar] [CrossRef]
- Ahmad, T.; Bustam, M.A.; Irfan, M.; Moniruzzaman, M.; Asghar, H.M.A.; Bhattacharjee, S. Mechanistic investigation of phytochemicals involved in green synthesis of gold nanoparticles using aqueous Elaeis guineensis leaves extract: Role of phenolic compounds and flavonoids. Biotechnol. Appl. Biochem. 2019, 66, 698–708. [Google Scholar] [CrossRef]
- Lata, K.; Arvind, K.J.; Laxmana, N.; Rajan, S. Gold nanoparticles: Preparation, characterization and its stability in buffer. Nano Trends A J. Nanotechnol. Its Appl. 2014, 17, 1–10. [Google Scholar]
- Elavazhagan, T.; Arunachalam, K.D. Memecylon edule leaf extract mediated green synthesis of silver and gold nanoparticles. Int. J. Nanomed. 2011, 6, 1265–1278. [Google Scholar] [CrossRef]
- Lekha, G.S.; Deepika, E.; Swetha, S.; Kanagarajan, A.; Gayathridevi, V.; Santhy, K.S. In vivo Evaluation of Antimicrobial, Antipyretic, Analgesic, and Anti-Inflammatory Activities of Nilavembu Kudineer Capsule in Comparison with Siddha Classical Nilavembu Kudineer. Pharmacogn. Res. 2020, 12, 387–393. [Google Scholar] [CrossRef]
- Biswas, B.; Rogers, K.; McLaughlin, F.; Daniels, D.; Yadav, A. Antimicrobial activities of leaf extracts of guava (Psidium guajava L.) on two gram-negative and gram-positive bacteria. Int. J. Microbiol. 2013, 2013, 746165. [Google Scholar] [CrossRef]
- Balderrama-González, A.S.; Piñón-Castillo, H.A.; Ramírez-Valdespino, C.A.; Landeros-Martínez, L.L.; Orrantia-Borunda, E.; Esparza-Ponce, H.E. Antimicrobial resistance and inorganic nanoparticles. Int. J. Mol. Sci. 2021, 22, 12890. [Google Scholar] [CrossRef]
- Anantharaman, S.; Rego, R.; Muthakka, M.; Anties, T.; Krishna, H. Andrographis paniculata-mediated synthesis of silver nanoparticles: Antimicrobial properties and computational studies. SN Appl. Sci. 2020, 2, 1618. [Google Scholar] [CrossRef]
- Yu, Q.; Li, J.; Zhang, Y.; Wang, Y.; Liu, L.; Li, M. Inhibition of gold nanoparticles (AuNPs) on pathogenic biofilm formation and invasion to host cells. Sci. Rep. 2016, 6, 26667. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef] [PubMed]
- Mobed, A.; Hasanzadeh, M.; Seidi, F. Anti-bacterial activity of gold nanocomposites as a new nanomaterial weapon to combat photogenic agents: Recent advances and challenges. RSC Adv. 2021, 11, 34688–34698. [Google Scholar] [CrossRef] [PubMed]
- Jacob, T.J.C.; Cruwys, S.; Mackie, I.A. Control of cellular proliferation in the bovine cornea: An in vitro study. Eye 1989, 3, 618–625. [Google Scholar] [CrossRef]


| S. No. | Botanical Name | Active Phytochemicals |
|---|---|---|
| 1. | Andrographis paniculata | Andrographolide, neoandrographolide, 14-deoxyandrographolide, 14-deoxy-12-hydroxy andrographolide, isoandrographolide, andrograpanin β-sitosterol and stigmasterol |
| 2. | Chrysopogon zizanioides L. | Khusimol, α-vetivone, β-vetivone, zizanal, epizizanal, and nootkatone |
| 3. | Santalum album L. | α-Santalol, sandalore, β-santalol, (+)-α-santalene, and isobornyl cyclohexanol |
| 4. | Zingiber officinale R. | Gingerols, shogaols, paradols, quercetin, zingerone, gingerenone-A, 6-dehydrogingerdione β-bisabolene, α-curcumene, zingiberene, α-farnesene, and β-sesquiphellandrene |
| 5. | Piper nigrum L. | Piperine, sarmentosine, piperamide, piperamine, trichosta, sarmentine, and chavicine |
| 6. | Cyperus rotundus L. | Patchoulenone, isopatchoulenone, sugenoyl acetate, sugetriol triacetate, sugeibol, kaempferol, luteolin, and quercetin |
| 7. | Mollugo cerviana L. | C-glycolsyl flavonoids-Orientin and Vitexin |
| 8. | Plectranthus vettiveroides | Androstan-17-one-3-ethyl-3-hydroxy-(5α)-spathulenol, α-bisabolol, Z-valerenyl acetate, megastigma-4,6(E), and 8(Z)-triene |
| 9. | Trichosanthes cucumerina L. | Cucurbitacin B, E, isocucurbitacin, 23,24-dihydroisocucurbitacin B, E, lycopene, and ascorbic acid |
| S. No. | Botanical Name | Local Name (English) | Plant Parts |
|---|---|---|---|
| 1. | Andrographis paniculata (Burm. f.) Nees | King of bitters | Whole plant |
| 2. | Chrysopogon zizanioides (L.) Roberty | Perennial bunchgrass | Root |
| 3. | Santalum album L. | Sandalwood | Bark |
| 4. | Zingiber officinale Roscoe | Dry Ginger | Rhizome |
| 5. | Piper nigrum L. | Black pepper | Fruit |
| 6. | Cyperus rotundus L. | Purple nutsedge | Root tuber |
| 7. | Mollugo cerviana (L.) Ser. | Threadstem carpetweed | Whole plant |
| 8. | Plectranthus vettiveroides (Jacob) | Coleus vettiveroides root | Root |
| 9. | Trichosanthes cucumerina L. | Snake gourd | Whole plant |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almutleb, E.S.; Ramachandran, S.; Khan, A.A.; El-Hiti, G.A.; Alanazi, S.A. Synergistic Effect of Nilavembu Choornam–Gold Nanoparticles on Antibiotic-Resistant Bacterial Susceptibility and Contact Lens Contamination-Associated Infectious Pathogenicity. Int. J. Mol. Sci. 2024, 25, 2115. https://doi.org/10.3390/ijms25042115
Almutleb ES, Ramachandran S, Khan AA, El-Hiti GA, Alanazi SA. Synergistic Effect of Nilavembu Choornam–Gold Nanoparticles on Antibiotic-Resistant Bacterial Susceptibility and Contact Lens Contamination-Associated Infectious Pathogenicity. International Journal of Molecular Sciences. 2024; 25(4):2115. https://doi.org/10.3390/ijms25042115
Chicago/Turabian StyleAlmutleb, Essam S., Samivel Ramachandran, Adnan A. Khan, Gamal A. El-Hiti, and Saud A. Alanazi. 2024. "Synergistic Effect of Nilavembu Choornam–Gold Nanoparticles on Antibiotic-Resistant Bacterial Susceptibility and Contact Lens Contamination-Associated Infectious Pathogenicity" International Journal of Molecular Sciences 25, no. 4: 2115. https://doi.org/10.3390/ijms25042115
APA StyleAlmutleb, E. S., Ramachandran, S., Khan, A. A., El-Hiti, G. A., & Alanazi, S. A. (2024). Synergistic Effect of Nilavembu Choornam–Gold Nanoparticles on Antibiotic-Resistant Bacterial Susceptibility and Contact Lens Contamination-Associated Infectious Pathogenicity. International Journal of Molecular Sciences, 25(4), 2115. https://doi.org/10.3390/ijms25042115










