The Differential Effect of a Shortage of Thyroid Hormone Compared with Knockout of Thyroid Hormone Transporters Mct8 and Mct10 on Murine Macrophage Polarization
Abstract
1. Introduction
2. Results
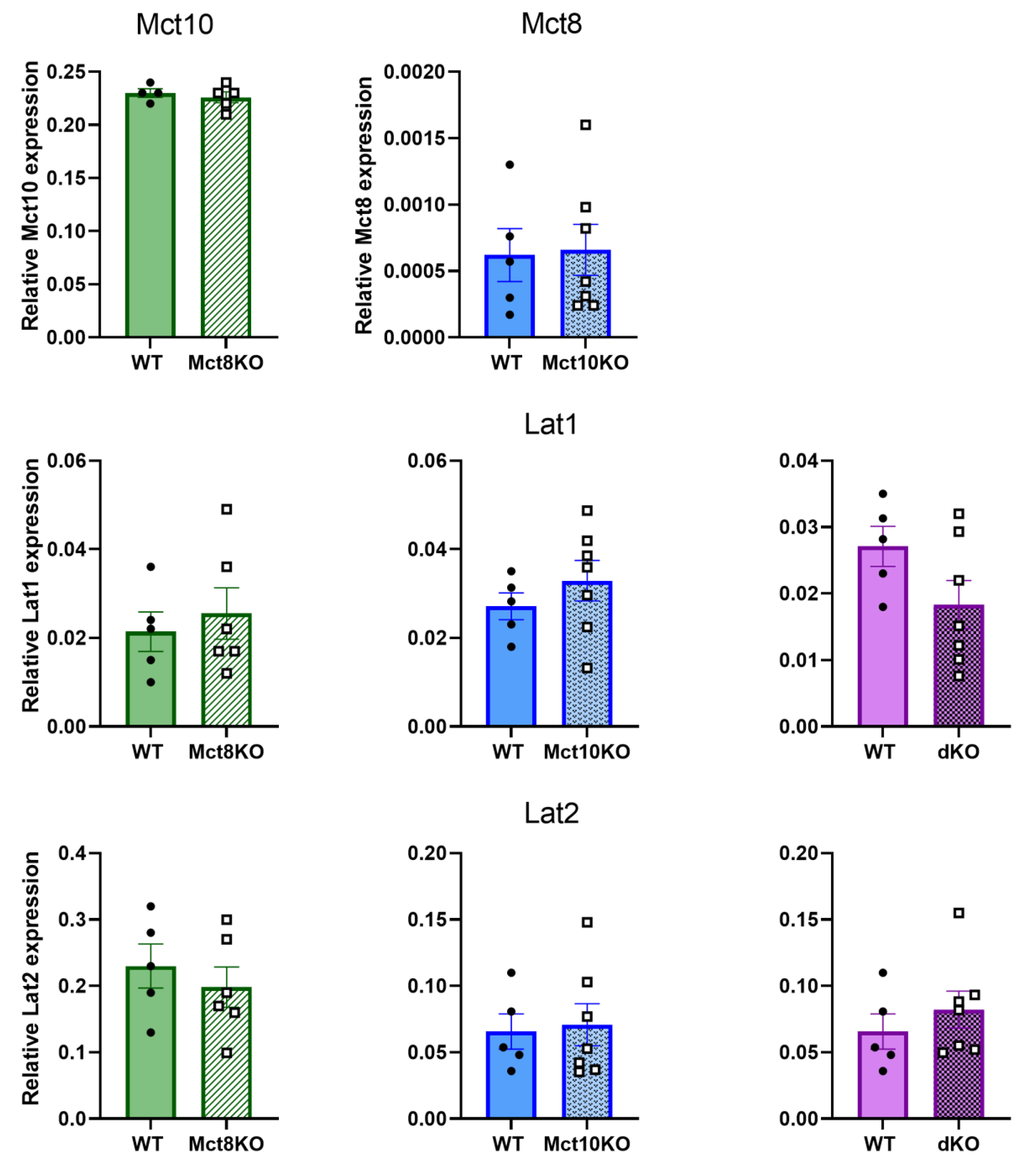
2.1. Transporter Expression in the KO BMDMs
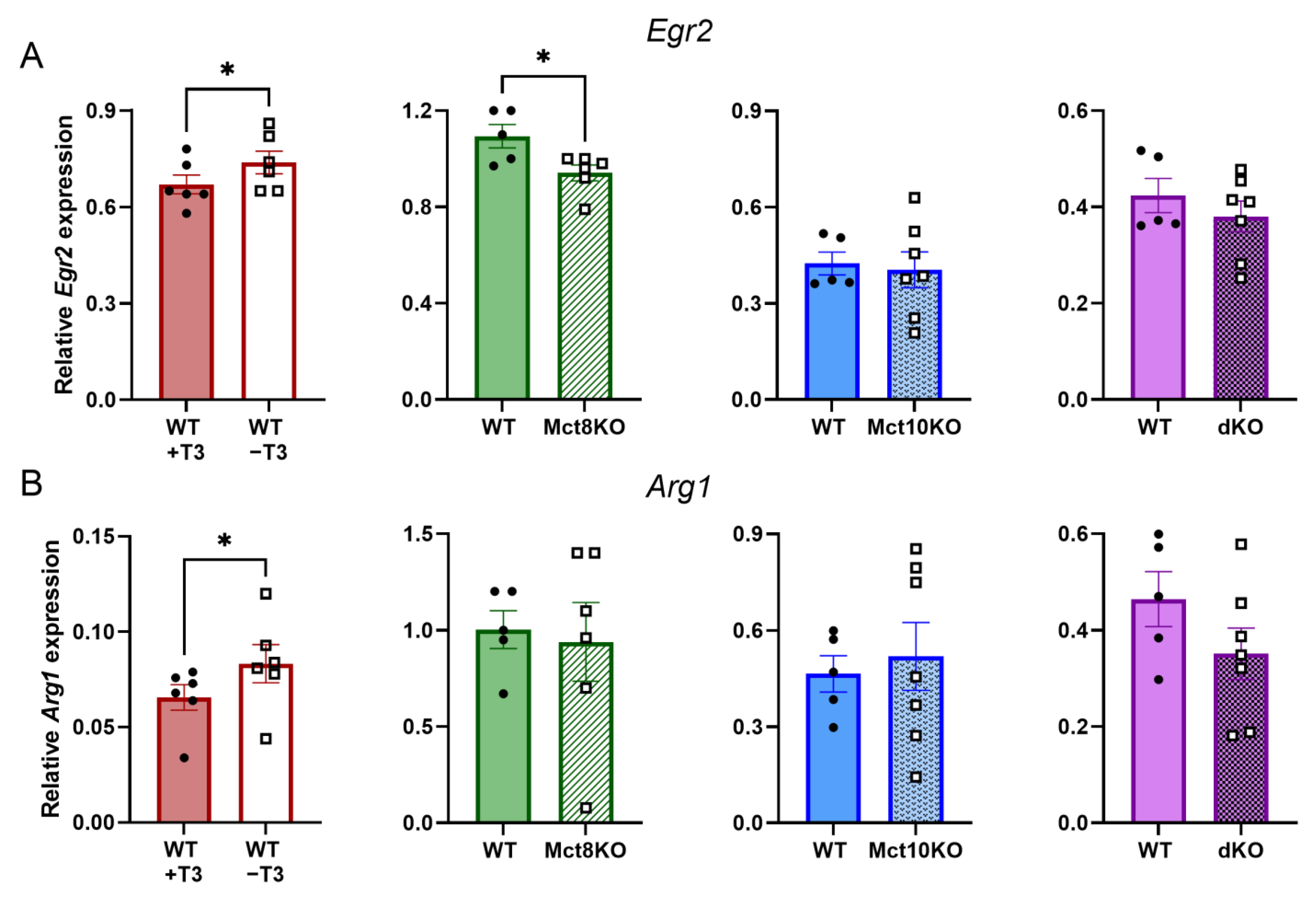
2.2. T3-Responsive Gene Expression in BMDMs
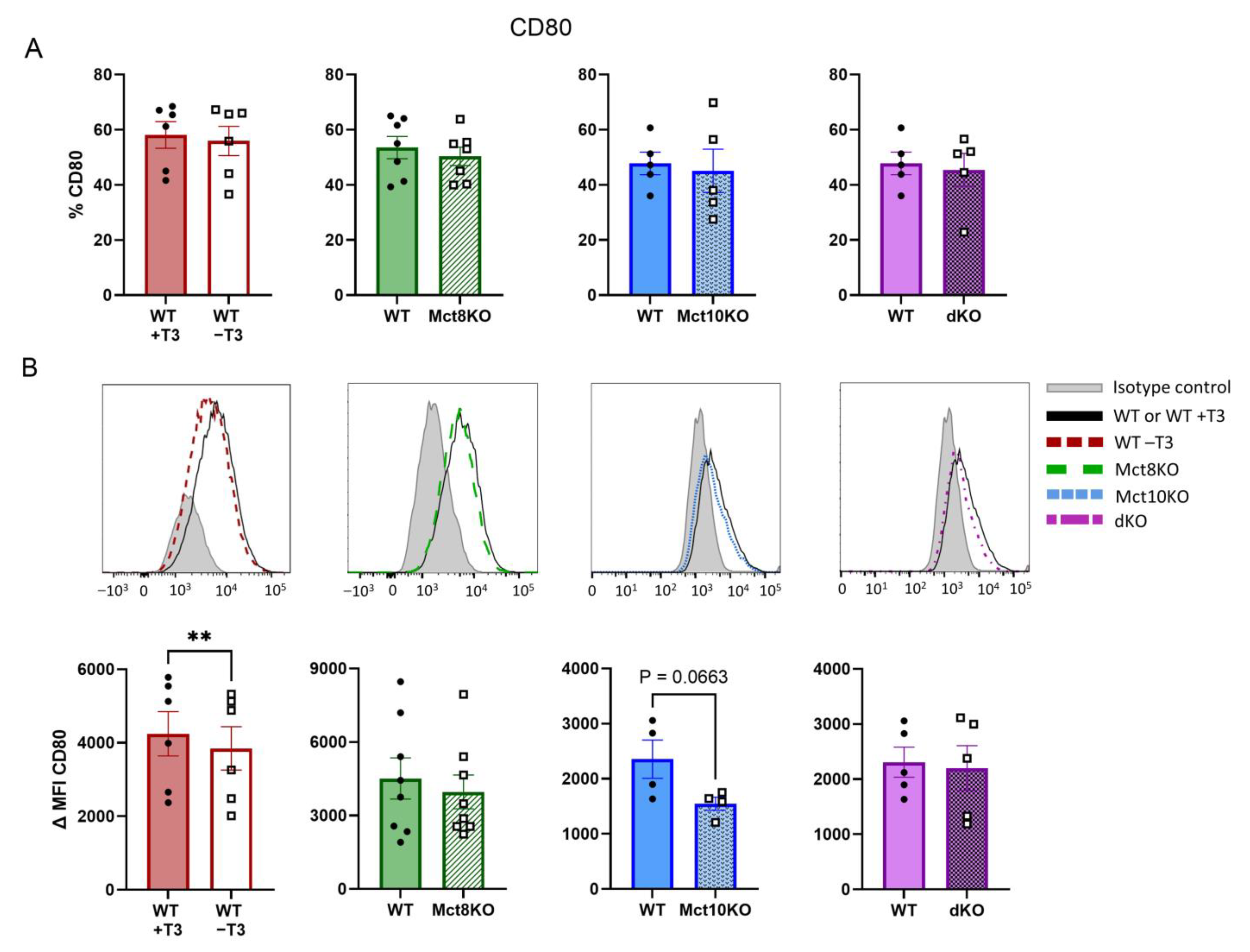
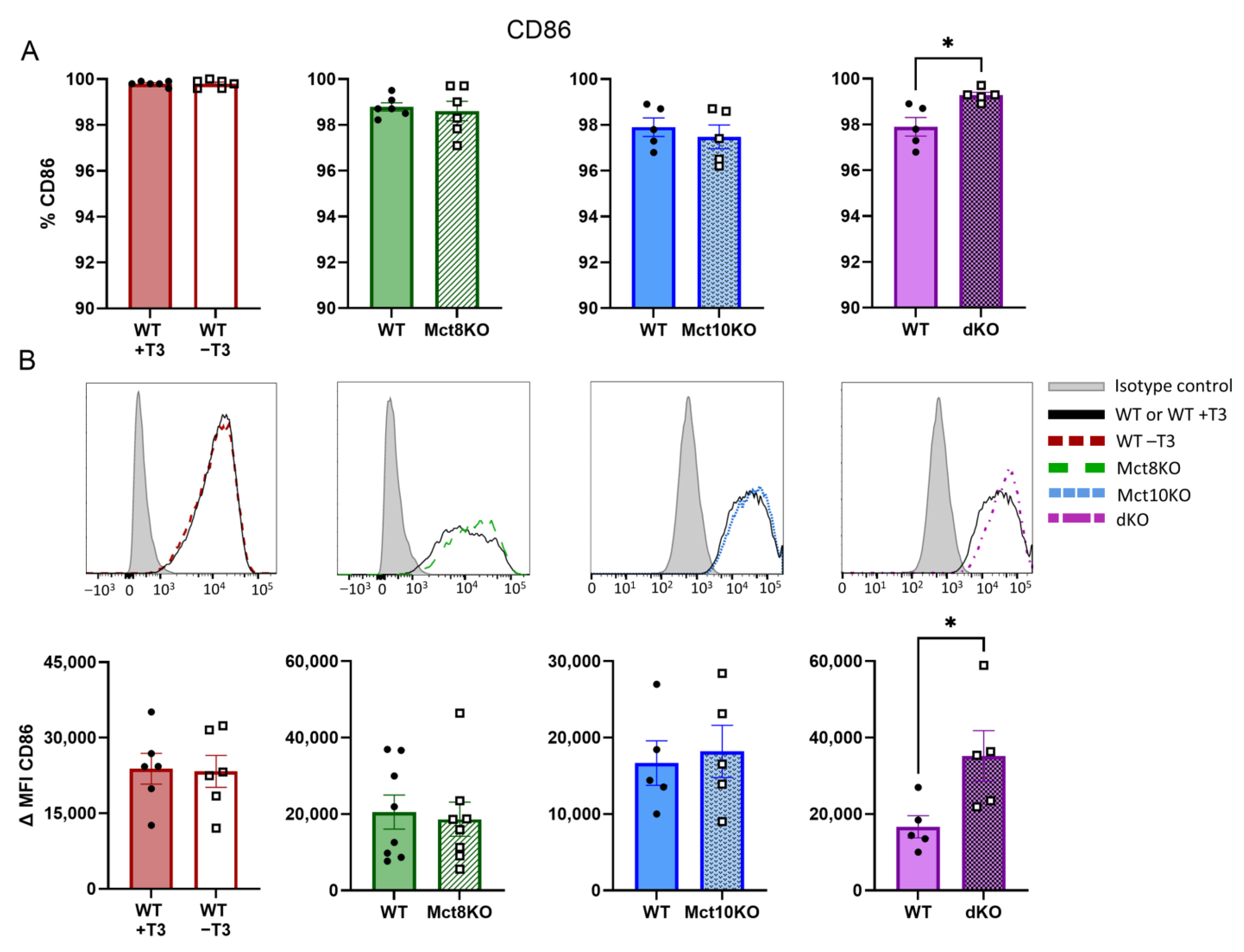
2.3. M1 Membrane Marker Expression of CD80 and CD86 in BMDMs
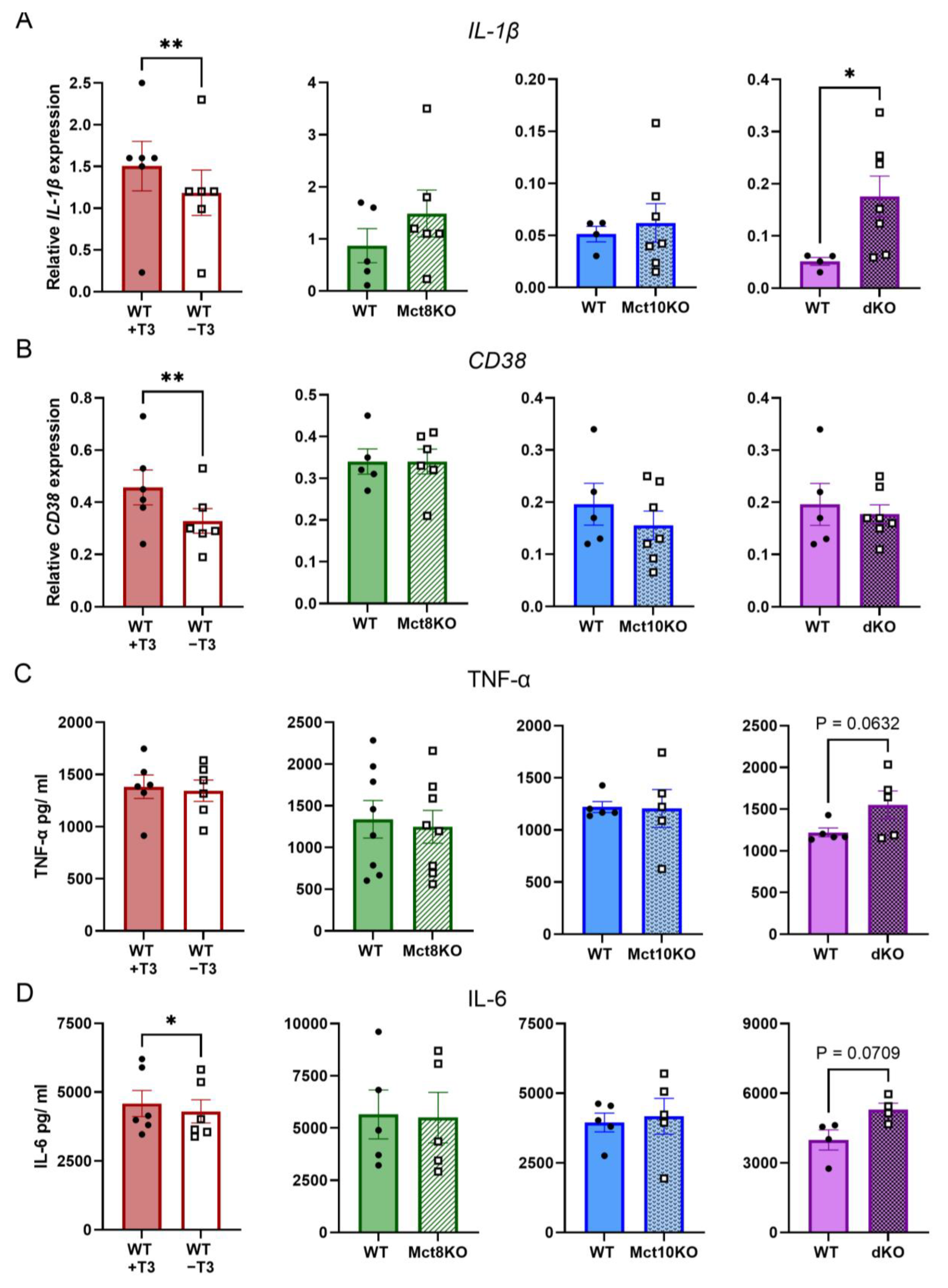
2.4. M1 mRNA Expression and Cytokine Secretion in BMDMs
2.5. M2 Membrane Marker Expression of CD206 in BMDMs
2.6. mRNA Expression of M2 Markers in BMDMs
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Flow Cytometry
4.3. RNA Isolation and Quantitative Real-Time PCR
4.4. Cytokine Measurement
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mendoza, A.; Hollenberg, A.N. New insights into thyroid hormone action. Pharmacol. Ther. 2017, 173, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Mullur, R.; Liu, Y.Y.; Brent, G.A. Thyroid hormone regulation of metabolism. Physiol. Rev. 2014, 94, 355–382. [Google Scholar] [CrossRef] [PubMed]
- De Vito, P.; Pedersen, J.Z.; Luly, P.; Davis, F.B.; Davis, P.J. Thyroid hormones as modulators of immune activities at the cellular level. Thyroid 2011, 21, 879–890. [Google Scholar] [CrossRef] [PubMed]
- Jara, E.L.; Muñoz-Durango, N.; Llanos, C.; Fardella, C.; González, P.A.; Bueno, S.M.; Riedel, C.A. Modulating the function of the immune system by thyroid hormones and thyrotropin. Immunol. Lett. 2017, 184, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Montesinos, M.D.M.; Pellizas, C.G. Thyroid hormone action on innate immunity. Front. Endocrinol. 2019, 10, 350. [Google Scholar] [CrossRef]
- Wenzek, C.; Boelen, A.; Westendorf, A.M.; Engel, D.R.; Moeller, L.C.; Führer, D. The interplay of thyroid hormones and the immune system—Where we stand and why we need to know about it. Eur. J. Endocrinol. 2022, 186, R65–R77. [Google Scholar] [CrossRef]
- Wynn, T.A.; Chawla, A.; Pollard, J.W. Macrophage biology in development, homeostasis and disease. Nature 2013, 496, 445–455. [Google Scholar] [CrossRef]
- Ginhoux, F.; Jung, S. Monocytes and macrophages: Developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 2014, 14, 392–404. [Google Scholar] [CrossRef]
- Hashimoto, D.; Chow, A.; Noizat, C.; Teo, P.; Beasley, M.B.; Leboeuf, M.; Becker, C.D.; See, P.; Price, J.; Lucas, D.; et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 2013, 38, 792–804. [Google Scholar] [CrossRef] [PubMed]
- Mass, E.; Ballesteros, I.; Farlik, M.; Halbritter, F.; Günther, P.; Crozet, L.; Jacome-Galarza, C.E.; Händler, K.; Klughammer, J.; Kobayashi, Y.; et al. Specification of tissue-resident macrophages during organogenesis. Science 2016, 353(aaf4238). [Google Scholar] [CrossRef] [PubMed]
- Hume, D.A. The Many Alternative Faces of Macrophage Activation. Front. Immunol. 2015, 6, 370. [Google Scholar] [CrossRef]
- Shapouri-Moghaddam, A.; Mohammadian, S.; Vazini, H.; Taghadosi, M.; Esmaeili, S.-A.; Mardani, F.; Seifi, B.; Mohammadi, A.; Afshari, J.T.; Sahebkar, A. Macrophage plasticity, polarization, and function in health and disease. J. Cell. Physiol. 2018, 233, 6425–6440. [Google Scholar] [CrossRef]
- Murray, P.J.; Wynn, T.A. Protective and pathogenic functions of macrophage subsets. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef] [PubMed]
- Jablonski, K.A.; Amici, S.A.; Webb, L.M.; de Dios Ruiz-Rosado, J.; Popovich, P.G.; Partida-Sanchez, S.; Guerau-de-Arellano, M. Novel Markers to Delineate Murine M1 and M2 Macrophages. PLoS ONE 2015, 10, e0145342. [Google Scholar] [CrossRef] [PubMed]
- Ying, W.; Cheruku, P.S.; Bazer, F.W.; Safe, S.H.; Zhou, B. Investigation of macrophage polarization using bone marrow derived macrophages. J. Vis. Exp. 2013, 76, e50323. [Google Scholar]
- Martinez, F.O.; Sica, A.; Mantovani, A.; Locati, M. Macrophage activation and polarization. Front. Biosci. 2008, 13, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, M.; Wen, Y.; Zhang, H.; Zhao, G.-N.; Gao, Q. Signaling pathways in macrophages: Molecular mechanisms and therapeutic targets. MedComm 2023, 4, e349. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Zhou, M.; Yang, H.; Qu, R.; Qiu, Y.; Hao, J.; Bi, H.; Guo, D. Regulatory Mechanism of M1/M2 Macrophage Polarization in the Development of Autoimmune Diseases. Mediat. Inflamm. 2023, 2023, 8821610. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, C.; Buldorini, M.; Assi, E.; Cazzato, D.; De Palma, C.; Clementi, E.; Cervia, D. The thyroid hormone triiodothyronine controls macrophage maturation and functions: Protective role during inflammation. Am. J. Pathol. 2014, 184, 230–247. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Sjölinder, M.; Wang, X.; Altenbacher, G.; Hagner, M.; Berglund, P.; Gao, Y.; Lu, T.; Jonsson, A.-B.; Sjölinder, H. Thyroid hormone enhances nitric oxide-mediated bacterial clearance and promotes survival after meningococcal infection. PLoS ONE 2012, 7, e41445. [Google Scholar] [CrossRef]
- Van der Spek, A.H.; Surovtseva, O.V.; Jim, K.K.; van Oudenaren, A.; Brouwer, M.C.; Vandenbroucke-Grauls, C.M.J.E.; Leenen, P.J.M.; van de Beek, D.; Hernandez, A.; Fliers, E.; et al. Regulation of Intracellular Triiodothyronine Is Essential for Optimal Macrophage Function. Endocrinology 2018, 159, 2241–2252. [Google Scholar] [CrossRef]
- Friesema, E.C.; Jansen, J.; Jachtenberg, J.-W.; Visser, W.E.; Kester, M.H.A.; Visser, T.J. Effective cellular uptake and efflux of thyroid hormone by human monocarboxylate transporter 10. Mol. Endocrinol. 2008, 22, 1357–1369. [Google Scholar] [CrossRef]
- Friesema, E.C.; Kuiper, G.G.J.M.; Jansen, J.; Visser, T.J.; Kester, M.H.A. Thyroid hormone transport by the human monocarboxylate transporter 8 and its rate-limiting role in intracellular metabolism. Mol. Endocrinol. 2006, 20, 2761–2772. [Google Scholar] [CrossRef]
- Groeneweg, S.; van Geest, F.S.; Peeters, R.P.; Heuer, H.; Visser, W.E. Thyroid Hormone Transporters. Endocr. Rev. 2020, 41, 146–201. [Google Scholar] [CrossRef]
- Müller, J.; Mayerl, S.; Visser, T.J.; Darras, V.M.; Boelen, A.; Frappart, L.; Mariotta, L.; Verrey, F.; Heuer, H. Tissue-Specific Alterations in Thyroid Hormone Homeostasis in Combined Mct10 and Mct8 Deficiency. Endocrinology 2014, 155, 315–325. [Google Scholar] [CrossRef]
- Kwakkel, J.; Surovtseva, O.V.; de Vries, E.M.; Stap, J.; Fliers, E.; Boelen, A. A novel role for the thyroid hormone-activating enzyme type 2 deiodinase in the inflammatory response of macrophages. Endocrinology 2014, 155, 2725–2734. [Google Scholar] [CrossRef]
- Lopez-Mateo, I.; Rodríguez-Muñoz, D.; de La Rosa, J.V.; Castrillo, A.; Alemany, S.; Aranda, A. Regulation of metabolic and transcriptional responses by the thyroid hormone in cellular models of murine macrophages. Front. Immunol. 2022, 13, 923727. [Google Scholar] [CrossRef]
- Wang, N.; Liang, H.; Zen, K. Molecular mechanisms that influence the macrophage M1-M2 polarization balance. Front. Immunol. 2014, 5, 614. [Google Scholar] [CrossRef] [PubMed]
- Sikorski, K.; Chmielewski, S.; Olejnik, A.; Wesoly, J.Z.; Heemann, U.; Baumann, M.; Bluyssen, H. STAT1 as a central mediator of IFNgamma and TLR4 signal integration in vascular dysfunction. JAKSTAT 2012, 1, 241–249. [Google Scholar] [PubMed]
- Furuya, F.; Ishii, T.; Tamura, S.; Takahashi, K.; Kobayashi, H.; Ichijo, M.; Takizawa, S.; Kaneshige, M.; Suzuki-Inoue, K. The ligand-bound thyroid hormone receptor in macrophages ameliorates kidney injury via inhibition of nuclear factor-kappaB activities. Sci. Rep. 2017, 7, 43960. [Google Scholar] [CrossRef] [PubMed]
- Moeller, L.C.; Wardrip, C.; Niekrasz, M.; Refetoff, S.; Weiss, R.E. Comparison of thyroidectomized calf serum and stripped serum for the study of thyroid hormone action in human skin fibroblasts in vitro. Thyroid 2009, 19, 639–644. [Google Scholar] [CrossRef]
- Siemes, D.; Vancamp, P.; Markova, B.; Spangenberg, P.; Shevchuk, O.; Siebels, B.; Schlüter, H.; Mayerl, S.; Heuer, H.; Engel, D.R. Proteome Analysis of Thyroid Hormone Transporter Mct8/Oatp1c1-Deficient Mice Reveals Novel Dysregulated Target Molecules Involved in Locomotor Function. Cells 2023, 12, 2487. [Google Scholar] [CrossRef]
- Gopinath, A.; Mackie, P.M.; Phan, L.T.; Mirabel, R.; Smith, A.R.; Miller, E.; Franks, S.; Syed, O.; Riaz, T.; Law, B.K.; et al. Who Knew? Dopamine Transporter Activity Is Critical in Innate and Adaptive Immune Responses. Cells 2023, 12, 269. [Google Scholar] [CrossRef]
- Han, X.; Ni, J.; Wu, Z.; Wu, J.; Li, B.; Ye, X.; Dai, J.; Chen, C.; Xue, J.; Wan, R.; et al. Myeloid-specific dopamine D2 receptor signalling controls inflammation in acute pancreatitis via inhibiting M1 macrophage. Br. J. Pharmacol. 2020, 177, 2991–3008. [Google Scholar] [CrossRef]
- Kavelaars, A.; PCobelens, M.; Teunis, M.A.T.; Heijnen, C.J. Changes in innate and acquired immune responses in mice with targeted deletion of the dopamine transporter gene. J. Neuroimmunol. 2005, 161, 162–168. [Google Scholar] [CrossRef]
- Yan, Y.; Jiang, W.; Liu, L.; Wang, X.; Ding, C.; Tian, Z.; Zhou, R. Dopamine controls systemic inflammation through inhibition of NLRP3 inflammasome. Cell 2015, 160, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Mariotta, L.; Ramadan, T.; Singer, D.; Guetg, A.; Herzog, B.; Stoeger, C.; Palacín, M.; Lahoutte, T.; Camargo, S.M.R.; Verrey, F. T-type amino acid transporter TAT1 (Slc16a10) is essential for extracellular aromatic amino acid homeostasis control. J. Physiol. 2012, 590, 6413–6424. [Google Scholar] [CrossRef] [PubMed]
- Mayerl, S.; Schmidt, M.; Doycheva, D.; Darras, V.M.; Hüttner, S.S.; Boelen, A.; Visser, T.J.; Kaether, C.; Heuer, H.; von Maltzahn, J. Thyroid Hormone Transporters MCT8 and OATP1C1 Control Skeletal Muscle Regeneration. Stem Cell Rep. 2018, 10, 1959–1974. [Google Scholar] [CrossRef] [PubMed]
- Trajkovic, M.; Visser, T.J.; Mittag, J.; Horn, S.; Lukas, J.; Darras, V.M.; Raivich, G.; Bauer, K.; Heuer, H. Abnormal thyroid hormone metabolism in mice lacking the monocarboxylate transporter 8. J. Clin. Investig. 2007, 117, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Heap, R.E.; Marín-Rubio, J.L.; Peltier, J.; Heunis, T.; Dannoura, A.; Moore, A.; Trost, M. Proteomics characterisation of the L929 cell supernatant and its role in BMDM differentiation. Life Sci. Alliance 2021, 4, e202000957. [Google Scholar] [CrossRef] [PubMed]
- Ruijter, J.M.; Barnewall, R.J.; Marsh, I.B.; Szentirmay, A.N.; Quinn, J.C.; van Houdt, R.; Gunst, Q.D.; van den Hoff, M.J.B. Efficiency Correction Is Required for Accurate Quantitative PCR Analysis and Reporting. Clin. Chem. 2021, 67, 829–842. [Google Scholar] [CrossRef] [PubMed]
- Bouaboula, M.; Legoux, P.; Pességué, B.; Delpech, B.; Dumont, X.; Piechaczyk, M.; Casellas, P.; Shire, D. Standardization of Messenger-RNA Titration Using a Polymerase Chain-Reaction Method Involving Coamplification with a Multispecific Internal Control. J. Biol. Chem. 1992, 267, 21830–21838. [Google Scholar] [CrossRef] [PubMed]
- Efstathiou, C.E. Estimation of type I error probability from experimental Dixon’s “Q” parameter on testing for outliers within small size data sets. Talanta 2006, 69, 1068–1071. [Google Scholar] [CrossRef] [PubMed]


| Surface Marker Target | Fluorescent Conjugate | Species Raised In; Mono- or Polyclonal | Clone | Catalog Number | Manufacturer | Dilution |
|---|---|---|---|---|---|---|
| F4/80 | APC-eFluor780 | Rat IgG2a, κ; Monoclonal | BMB | 47-4801-82 | Invitrogen, Darmstadt, Germany | 1:100 |
| CD11b | FITC | Rat IgG2b, κ; Monoclonal | M1/70 | 11-0112-41 | Invitrogen, Darmstadt, Germany | 1:100 |
| CD206 | APC | Rat IgG2a, κ; Monoclonal | C068C2 | 141707 | Biolegend, Amsterdam, the Netherlands | 1:100 |
| CD86 | APC | Rat IgG2a, κ; Monoclonal | GL1 | 17-0862-81 | Invitrogen, Darmstadt, Germany | 1:200 |
| CD80 | PE | Hamster IgG2, κ; Monoclonal | 16-10A1 | 561955 | BD Bioscience, Eysins, Switzerland | 1:400 |
| CD16/CD32 (Fc-block) | - | Rat IgG2a, λ; Monoclonal | 93 | 14-0161-85 | Invitrogen, Darmstadt, Germany | 1:50 |
| Gene | Protein | Forward (5′-3′) | Reverse (5′-3′) | Amplicon Length | Source | Accession Number |
|---|---|---|---|---|---|---|
| Ppib | Cyclophilin-B | GAGACTTCACCAGGGG | CTGTCTGTCTTGGTGCTCTCC | 253 | [21] | NM_011149.2 |
| Rplp0 | Rplp0 | GGCCCTGCACTCTCGCTTTC | TGCCAGGACGCGCTTGT | 124 | [21] | NM_007475.5 |
| Ubc | Ubc | AGCCCAGTGTTACCACCAAG | CTAAGACACCTCCCCCATCA | 118 | [26] | NM_019639.4 |
| Slc16a10 | Mct10 | GATGAACATGGCCTCCCAACA | CCTTGAAGTGAGTCTGGCTGG | 164 | Newly designed | NM_001114332.1 |
| Slc16a2 | Mct8 | GGTGGAGTCACTGTCCTGTC | GCACACTTATTCTGCCCCCT | 180 | Newly designed | NM_009197.2 |
| Slc7a5 | Lat1 | AAGGGCAGGGATTCATGGTG | GTAGGGGTGTCTTTCAGGGC | 188 | Newly designed | NM_011404.3 |
| Slc7a8 | Lat2 | GCAAGAAAGTACCTGAGCACG | AACAGATCGCCTCCACCTTC | 106 | Newly designed | NM_016972.2 |
| Klf9 | Klf9 | CCACCGAATCTGGGTCGAG | TCCGAGCGCGAGAACTTTT | 265 | Newly designed | NM_010638.5 |
| Il1b | IL-1β | TTGACGGACCCCAAAAGATG | AGAAGGTGCTCATGTCCTCA | 204 | [42] | NM_008361.4 |
| CD38 | CD38 | ACTGGAGAGCCTACCACGAA | AGTGGGGCGTAGTCTTCTCT | 179 | Newly designed | NM_007646.6 |
| Fpr2 | Fpr2 | ATTTACACCACAGGAACCGAAGA | TGATGGAGACAACCACCATTGA | 166 | Newly designed | NM_008039.2 |
| Gpr18 | Gpr18 | ATCTGCTTTGCCGTCCTGAT | ACTGCGAAGGTAATTGCGGT | 180 | Newly designed | NM_182806.2 |
| Egr2 | Egr2 | GGGTCTGCATGTGTACAGGA | AAACAAATCAGCGGCAGTGAC | 209 | Newly designed | NM_010118.3 |
| Arg1 | Arg1 | CAGCACTGAGGAAAGCTGGT | CAGACCGTGGGTTCTTCACA | 132 | [21] | NM_007482.3 |
| Il10 | IL-10 | ATGCAGGACTTTAAGGGTTACTTG | TAGACACCTTGGTCTTGGAGCTTA | 254 | [42] | NM_010548.2 |
| c-Myc | c-Myc | GGAACGTCAGAGGAGGAACG | TGCTCGTCTGCTTGAATGGA | 147 | Newly designed | NM_010849.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoen, E.; Goossens, F.M.; Falize, K.; Mayerl, S.; van der Spek, A.H.; Boelen, A. The Differential Effect of a Shortage of Thyroid Hormone Compared with Knockout of Thyroid Hormone Transporters Mct8 and Mct10 on Murine Macrophage Polarization. Int. J. Mol. Sci. 2024, 25, 2111. https://doi.org/10.3390/ijms25042111
Hoen E, Goossens FM, Falize K, Mayerl S, van der Spek AH, Boelen A. The Differential Effect of a Shortage of Thyroid Hormone Compared with Knockout of Thyroid Hormone Transporters Mct8 and Mct10 on Murine Macrophage Polarization. International Journal of Molecular Sciences. 2024; 25(4):2111. https://doi.org/10.3390/ijms25042111
Chicago/Turabian StyleHoen, Esmée, Franka M. Goossens, Kim Falize, Steffen Mayerl, Anne H. van der Spek, and Anita Boelen. 2024. "The Differential Effect of a Shortage of Thyroid Hormone Compared with Knockout of Thyroid Hormone Transporters Mct8 and Mct10 on Murine Macrophage Polarization" International Journal of Molecular Sciences 25, no. 4: 2111. https://doi.org/10.3390/ijms25042111
APA StyleHoen, E., Goossens, F. M., Falize, K., Mayerl, S., van der Spek, A. H., & Boelen, A. (2024). The Differential Effect of a Shortage of Thyroid Hormone Compared with Knockout of Thyroid Hormone Transporters Mct8 and Mct10 on Murine Macrophage Polarization. International Journal of Molecular Sciences, 25(4), 2111. https://doi.org/10.3390/ijms25042111







