Acerola (Malpighia emarginata) Anti-Inflammatory Activity—A Review
Abstract
1. Introduction
2. Botanical Characteristics and Habitat
3. Biochemical Properties of Acerola
4. Antioxidant and Anti-Inflammatory Effects of Acerola
5. Effects of Acerola on the Composition of the Gut Microbiome
6. Gastro Protective Effects
7. Hepatoprotective and Hepato-Regenerative Effects
8. Impact on Detoxification of the System
9. Stimulating Effect on the Synthesis of Steroid Hormones (Hormone-Forming Effect)
10. Anticancer Activity
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Belwal, T.; Devkota, H.P.; Hassan, H.A.; Ahluwalia, S.; Ramadan, M.F.; Mocan, A.; Atanasov, A.G. Phytopharmacology of acerola (Malpighia spp.) and its potential as functional food. Trends Food Sci. Technol. 2018, 74, 99–106. [Google Scholar] [CrossRef]
- Poletto, P. Recovery of ascorbic acid, phenolic compounds and carotenoids from acerola by-products: An opportunity for their valorization. LWT-Food Sci. Technol. 2021, 146, 111654. [Google Scholar] [CrossRef]
- Ayala-Zavala, J.F.; Vega-Vega, V.; Rosas-Domínguez, C.; Palafox-Carlos, H.; Villa-Rodriguez, J.A.; Siddiqui, M.W.; Dávila-Aviña, J.E.; González-Aguilar, G.A. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 2011, 44, 1866–1874. [Google Scholar] [CrossRef]
- Tena, M.M. Distribution of Malpighia mexicana in Mexico and its implications for Barranca del Río Santiago. J. For. Res. 2021, 32, 1095–1103. [Google Scholar] [CrossRef]
- Carneiro Ferreira, I. Brazilian varieties of acerola (Malpighia emarginata DC.) produced under tropical semi-arid conditions: Bioactive phenolic compounds, sugars, organic acids, and antioxidant capacity. J. Food Biochem. 2021, 8, e13829. [Google Scholar]
- Jankiewicz, L.; i Lipecki, J. Fizjologia Roślin Sadowniczych Strefy Umiarkowanej (Physiology of Temperatezonefruitplants). T.2. Plonowanie roślin i udział w Tym procesie różnych czynników (Plant Yield and the Contribution of Various Factors to this Process); Wydawnictwo Naukowe PWN: Warsaw, Poland, 2012. [Google Scholar]
- Rutkowski, L. Klucz do Oznaczania Roślin Naczyniowych Polski Niżowej (Key to the Identification of Vascularplants of Lower Poland); Wydawnictwo Naukowe PWN: Warsaw, Poland, 2006. [Google Scholar]
- Carvalho, G.N.; Oliveira, C.S. Bioactive compounds and antioxidant activities in the agro-industrial residues of acerola (Malpighia emarginata L.), guava (Psidium guajava L.), genipap (Genipa americana L.) and umbu (Spondias tuberosa L.) fruits assisted by ultrasonic or shaker extraction. Food Res. Int. 2021, 147, 110538. [Google Scholar]
- de Aquino Souza Miskinis, R.; Ávila do Nascimento, L.; Colussi, R. Bioactive compounds from acerola pomace: A review. Food Chem. 2023, 404, 134613. [Google Scholar] [CrossRef]
- Xu, M.; Shen, C.; Zheng, H.; Xu, Y.; Xue, C.; Zhu, B.; Hu, J. Metabolomic analysis of acerola cherry (Malpighia emarginata) fruit during ripening development via UPLC-Q-TOF and contribution to the antioxidant activity. Food Res. Int. 2020, 130, 108915. [Google Scholar] [CrossRef]
- de Oliveira, S.D.; de Souza, E.L.; Araújo, C.M. Spontaneous fermentation improves the physicochemical characteristics, bioactive compounds, and antioxidant activity of acerola (Malpighia emarginata D.C.) and guava (Psidium guajava L.) fruit processing by-products. Biotechnology 2023, 13, 315. [Google Scholar] [CrossRef]
- Monteiro, S.A. Preparation, phytochemical and bromatological evaluation of flour obtained from the acerola (Malpighia punicifolia) agroindustrial residue with potential use as fiber source. LWT-Food Sci. Technol. 2020, 134, 110142. [Google Scholar] [CrossRef]
- Souza, N.C. Anti-inflammatory and antixidant properties of blend formulated with compounds of Malpighiae marginata D.C. (acerola) and Camellia sinensis L. (green tea) in lipopolysaccharide-stimulated RAW 264.7 macrophages. Biomed. Pharmacother. 2020, 128, 110277. [Google Scholar] [CrossRef]
- Hwang, J.; Hodis, H.N.; Sevanian, A. Soy and alfalfa phytoestrogen extracts become potent low-density lipoprotein antioxidants in the presence of acerola cherry extract. J. Agric. Food Chem. 2001, 49, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Dias, F.M.; Leffa, D.D.; Daumann, F.; Marques, S.O.; Luciano, T.F.; Possato, J.C. Acerola (Malpighia emarginata DC.) juice intake protects against alterations to proteins involved in inflammatory and lipolysis pathways in the adipose tissue of obese mice fed a cafeteria diet. Lipids Health Dis. 2014, 13, 24. [Google Scholar] [CrossRef]
- El-Hawary, S.S.; El-Fitiany, R.A.; El-Fitiany, R.A.; Mousa, O.M.; Salama, A.A.; El Gedaily, R.A. Metabolic profiling and in vivo hepatoprotective activity of Malpighia glabra L. leaves. J. Food Biochem. 2021, 45, e13588. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Yin, F.; Liu, Z.; Zhou, D.; Zhu, B. Acerola polysaccharides ameliorate high-fat diet-induced non-alcoholic fatty liver disease through reduction of lipogenesis and improvement of mitochondrial functions in mice. Food Funct. 2020, 11, 1037–1048. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.D.S.; de Souza, C.B.; Padilha, M.; Saad, S.M.I.; Venema, K. Impact of a fermented soy beverage supplemented with acerola by-product on the gut microbiota from lean and obese subjects using an in vitro model of the human colon. Appl. Microbiol. Biotechnol. 2021, 105, 3771–3785. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Cui, X.; Xiang, Q.; Guo, M.; Liang, Y.; Qu, Y.; Yang, X. Protective effect of against ethanol-induced gastric ulcer and its mechanism. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2021, 50, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Batista, K.S.; Soares, N.L.; Dorand, V.A.M.; Alves, A.F.; Dos Santos Lima, M.; de Alencar Pereira, R.; Leite de Souza, E.; Magnani, M.; Persuhn, D.C.; de Souza Aquino, J. Acerola fruit by-product alleviates lipid, glucose, and inflammatory changes in the enterohepatic axis of rats fed a high-fat diet. Food Chem. 2023, 403, 134322. [Google Scholar] [CrossRef] [PubMed]
- González, P.; Lozano, P.; Ros, G.; Solano, F. Hyperglycemia and Oxidative Stress: An Integral, Updated and Critical Overview of Their Metabolic Interconnections. Int. J. Mol. Sci. 2023, 24, 9352. [Google Scholar] [CrossRef]
- Olakowski, M. The role of growth factors in pathogenesis of pancreatic cancer. Part II: Transforming growth factor beta (TGF- β), fibroblast growth factor (FGF), nerve growth factor (NGF). Gastroenterol. Rev./Przegląd Gastroenterologiczny 2007, 2, 175–180. [Google Scholar]
- Yao, Q.; Li, S.; Li, X.; Wang, F.; Tu, C. Myricetin modulates macrophage polarization and mitigates liver inflammation and fibrosis in a murine model of nonalcoholic steatohepatitis. Front. Med. 2020, 7, 71. [Google Scholar] [CrossRef] [PubMed]
- da Silva, E.S.; Nunes, A.O.; Hoskin, R.T. Ultrasound-assisted polyphenol extraction of acerola and jambolan pomaces: Comparison of extraction protocols, kinetic modeling, and life cycle assessment. Chem. Eng. Process. Process Intensif. 2023, 191, 109443. [Google Scholar] [CrossRef]
- Mangoni, A.A.; Jackson, S.H. Age-related changes in pharmacokinetics and pharmacodynamics: Basic principles and practical applications. Br. J. Clin. Pharmacol. 2004, 57, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Bai, X.; Liang, Q.; Li, C.; Qiao, Z. Effect of Crushing Time on Physicochemical Properties and Active Ingredient Content of the Whole Fruit Pulp of Acerola Cherry. Sci. Technol. Food Indust. 2023, 44, 81–87. [Google Scholar]
- Ortiz, T.; Illanes, F.A.; Talero, J.M.G.; Macías-García, L.; Alcudia, A.; Vázquez-Román, V.; Motilva, V.; De-Miguel, M. Polyphenolic maqui extract as a potential nutraceutical to treat TNBS-induced Crohn’s disease by the regulation of antioxidant and anti-inflammatory pathways. Nutrients 2020, 12, 1752. [Google Scholar] [CrossRef] [PubMed]
- Righetto, A.M.; Netto, F.M.; Carraro, F. Chemical Composition and Antioxidant Activity of Juices from Mature and Immature Acerola (Malpighia emarginata DC). Food Sci. Technol. Int. 2005, 11, 315–321. [Google Scholar] [CrossRef]
- Alali, M.; Alqubaisy, M.; Aljaafari, M.N.; Alali, A.O.; Baqais, L.; Molouki, A.; Abushelaibi, A.; Lai, K.S.; Lim, S.H.E. Nutraceuticals: Transformation of conventional foods into health promoters/disease preventers and safety considerations. Molecules 2021, 26, 2540. [Google Scholar] [CrossRef]
- Lima, V.L.; Melo, E.A.; Maciel, M.I.S.; Prazeres, F.G.; Musser, R.S.; Lima, D.E.S. Total phenolic and carotenoid contents in acerola genotypes harvested at three ripening stages. Food Chem. 2005, 90, 565–568. [Google Scholar] [CrossRef]
- De Rosso, V.V.; Mercadante, A.Z. Carotenoid composition of two Brazilian genotypes of acerola (Malpighia punicifolia L.) from two harvests. Food Res. Int. 2005, 38, 1073–1077. [Google Scholar] [CrossRef]
- Fernandes, F.A.N.; Santos, V.O.; Gomes, W.F.; Rodrigues, S. Application of High-Intensity Ultrasound on Acerola (Malpighia emarginata) Juice Supplemented with Fructooligosaccharides and Its Effects on Vitamins, Phenolics, Carotenoids, and Antioxidant Capacity. Processes 2023, 11, 2243. [Google Scholar] [CrossRef]
- Prakash, A.; Baskaran, R. Acerola, an untapped functional superfruit: A review on latest frontiers. J. Food Sci. Technol. 2018, 55, 3373–3384. [Google Scholar] [CrossRef]
- Delva, L.; Schneider, R.G. Acerola (Malpighia emarginata DC): Production, Postharvest Handling, Nutrition, and Biological Activity. Food Rev. Int. 2013, 29, 107–126. [Google Scholar] [CrossRef]
- Rajoka, M.S.R.; Thirumdas, R.; Mehwish, H.M.; Umair, M.; Khurshid, M.; Hayat, H.F.; Phimolsiripol, Y.; Pallarés, N.; Martí-Quijal, F.J.; Barba, F.J. Role of food antioxidants in modulating gut microbial communities: Novel understandings in intestinal oxidative stress damage and their impact on host health. Antioxidants 2021, 10, 1563. [Google Scholar] [CrossRef]
- Wang, B.; Kong, Q.; Li, X.; Zhao, J.; Zhang, H.; Chen, W.; Wang, G. A High-Fat Diet Increases Gut Microbiota Biodiversity and Energy Expenditure Due to Nutrient Difference. Nutrients 2020, 12, 3197. [Google Scholar] [CrossRef] [PubMed]
- Menezes, F.N.; de Melo, F.H.C.; Vieira, A.R.S. Acerola (Malpighiaglabra L.) and guava (Psidiumguayaba L.) industrial processing by-products stimulate probiotic Lactobacillus and Bifidobacterium growth and induce beneficial changes in colonic microbiota. J. Appl. Microbiol. 2021, 130, 1323–1336. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.M.R.D.; Lima, J.D.S.S.; Magalhães, F.E.A.; Campos, A.R.; Araújo, J.I.F.; Batista, F.L.A.; Araújo, S.M.B.; Sousa, P.H.M.; Lima, G.C.; Holanda, D.K.R.; et al. Graviola Fruit Bar Added Acerola By-Product Extract Protects Against Inflammation and Nociception in Adult Zebrafish (Danio rerio). J. Med. Food 2020, 23, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Vohra, F.; Raut, A. Comparative efficacy, safety, and tolerability of diclofenac and aceclofenac in musculoskeletal pain management: A systematic review. Indian J. Pain 2016, 30, 3. [Google Scholar]
- Freire, M.A.; Guimarães, J.S.; Leal, W.G.; Pereira, A. Pain modulation by nitric oxide in the spinal cord. Front. Neurosci. 2009, 3, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Fernyhough, P.; Calcutt, N.A. Abnormal calcium homeostasis in peripheral neuropathies. Cell Calcium. 2010, 47, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Hanamura, T.; Mayama, C.; Aoki, H.; Hirayama, Y.; Shimizu, M. Antihyperglycemic effect of polyphenols from Acerola (Malpighia emarginata DC.) fruit. Biosci. Biotechnol. Biochem. 2006, 70, 1813–1820. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Giampieri, F.; Gasparrini, M.; Mazzoni, L.; Santos-Buelga, C.; González-Paramás, A.M.; Forbes-Hernández, T.Y.; Afrin, S.; Páez-Watson, T.; Quilesg, J.L.; et al. The protective effect of acerola (Malpighia emarginata) against oxidative damage in human dermal fibroblasts through the improvement of antioxidant enzyme activity and mitochondrial functionality. Food Funct. 2017, 8, 3250–3258. [Google Scholar] [CrossRef] [PubMed]
- da Costa Lima, M.; Magnani, M.; Dos Santos Lima, M.; de Sousa, C.P.; Dubreuil, J.D.; de Souza, E.L. Phenolic-rich extracts from acerola, cashew apple and mango by-products cause diverse inhibitory effects and cell damages on enterotoxigenic Escherichia coli. Lett. Appl. Microbiol. 2022, 75, 565–577. [Google Scholar] [CrossRef]
- Batista, A.C.V.; Ribeiro, M.D.A.; Oliveira, K.A.D.; de Souza, E.L.; de Oliveira, A.C. Effects of consumption of acerola, cashew and guava by-products on adiposity and redox homeostasis of adipose tissue in obese rats. Clin. Nutr. ESPEN 2021, 43, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Turkseven, S.; Kruger, A.; Mingone, C.J.; Kaminski, P.; Inaba, M.; Rodella, L.F.; Ikehara, S.; Wolin, M.S.; Abraham, N.G. Antioxidant mechanism of heme oxygenase-1 involves an increase in superoxide dismutase and catalase in experimental diabetes. Heart Circ. Physiol. 2005, 289, H701–H707. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.A.; Spencer, S.J. Obesity and neuroinflammation: A pathway to cognitive impairment. Brain Behav. Immun. 2014, 42, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wan, X.; Wu, X. Eubacteriumrectale contributes to colorectal cancer initiation via promoting colitis. Gut Pathog. 2021, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Assis, B.B.T. Biotransformation of the Brazilian Caatinga fruit-derived phenolics by Lactobacillus acidophilus La-5 and Lacticaseibacilluscasei 01 impacts bioaccessibility and antioxidant activity. Food Res. Int. 2021, 146, 110435. [Google Scholar] [CrossRef]
- El-Hawary, S.S.; Mousa, O.M.; El-Fitiany, R.A.; Gedaily, R.A.E. Cytotoxic, Antimicrobial Activities, and Phytochemical Investigation of Three Peach Cultivars and Acerola Leaves. J. Rep. Pharm. Sci. 2020, 9, 221–236. [Google Scholar] [CrossRef]
- Grattan, B.J. Plant sterols as anticancer nutrients: Evidence for their role in breast cancer. Nutrients 2013, 5, 359–387. [Google Scholar] [CrossRef]
- Liu, J.Q.; Deng, Y.Y.; Li, T.Z.; Han, Q.; Li, Y.; Qiu, M.H. Three new tetranorditerpenes from aerial parts of acerola cherry (Malpighia emarginata). Molecules 2014, 19, 2629–2636. [Google Scholar] [CrossRef]
- Xie, Y.; Zhu, G.; Yi, J.; Ji, Y.; Xia, Y.; Zheng, Y.; Ye, C. A new product of multi-plant extracts improved skin photoaging: An oral intake in vivo study. J. Cosmet. Dermatol. 2022, 21, 3406–3415. [Google Scholar] [CrossRef] [PubMed]
- Parker, E.R. The influence of climate change on skin cancer incidence—A review of the evidence. Int. J. Womens Dermatol. 2020, 7, 17–27, Erratum in: Int. J. Womens Dermatol. 2021, 7, 867. [Google Scholar] [CrossRef] [PubMed]
- Lenart, K.; Szyda, A.; Kiełbasiński, M.; Duś, D.; Podolak-Dawidziak, M. Clinical effects of multidrug resistance in neoplasms. Oncol. Clin. Pract. 2005, 1, 18–26, (Kliniczne skutki oporności wielolekowej w nowotworach). Onkologia W Praktyce Klinicznej. [Google Scholar]
- Motohashi, S.; Ishikawa, A.; Ishikawa, E. A phase I study of in vitro expanded natural killer T cells in patients with advanced and recurrent non-small cell lung cancer. Clin. Cancer Res. 2006, 12, 6079–6086. [Google Scholar] [CrossRef] [PubMed]
- Nagamine, I.; Akiyama, T.; Kainuma, M.; Kumagai, H.; Satoh, H.; Yamada, K.; Yano, T.; Sakurai, H. Effect of acerola cherry extract on cell proliferation and activation of Ras signal pathway at the promotion stage of lung tumorigenesis in mice. J. Nutr. Sci. Vitaminol. 2002, 48, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Takino, J.I.; Sato, T.; Nagamine, K.; Hori, T. The inhibition of Bax activation-induced apoptosis by RasGRP2 via R-Ras-PI3K-Akt signaling pathway in the endothelial cells. Sci. Rep. 2019, 9, 16717. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Zhou, J.; Fu, L.; Li, Y.; Zeng, H.; Xu, X.; Lv, C.; Jin, H. Ascorbic Acid Inhibits Liver Cancer Growth and Metastasis in vitro and in vivo, Independent of Stemness Gene Regulation. Front. Pharmacol. 2021, 24, 726015. [Google Scholar] [CrossRef]
- Naidu, K.A. Vitamin C in human health and disease is still a mystery? An overview. Nutr. J. 2003, 2, 7. [Google Scholar] [CrossRef]
- Matta, V.M.; Moretti, R.H.; Cabral, L.M.C. Microfiltration and reverse osmosis for clarification and concentration of acerola juice. J. Food Eng. 2004, 61, 477–482. [Google Scholar] [CrossRef]

| Bioactive Compounds | Malpighia emarginata/Malpighia glabra | Reference |
|---|---|---|
| ascorbic acid | 1.18 to 2.43 g*100 g−1 FW (pomace) | [5] |
| 34 mg*100 g−1 FW (leaves) | [5] | |
| β-carotene | 5.84 mg*g−1 DM | [11] |
| polyphenoliccompounds | 378.69 to 444.05 mg*100 g−1 DM | [11] |
| naringenin | 478 μg*g−1 DM | [11] |
| cyanidin-3-rhamnoside | 149–682 µg*g−1 FW | [5] |
| ferulicacid | 40.96 μg*g−1 DM | [11] |
| p-coumaricacid | 42.25 μg*g−1 DM | [11] |
| catechins | 15.66 μg*g−1 DM | [11] |
| epicatechin | 15.95 μg*g−1 DM | [11] |
| rutoside | 36.22 μg*g−1 DM | [11] |
| total anthocyanins | 19.43 mg/100 g FW | [5] |
| tartaricacid | 1116 mg*100 g−1 DM | [11] |
| succinicacid | 119 mg*100 g−1 DM | [11] |
| fructose | 29.8 mg*g−1 DM | [12] |
| glucose | 48.55 mg*g−1 DM | [12] |
| Antioxidant activity (EC50 as the concentration for a 50% reduction of DPPH radicals) | 38.17 μg/mL (pulp) | [2] |
| Experimental Model | The Impact on the Human Organism | Reference |
|---|---|---|
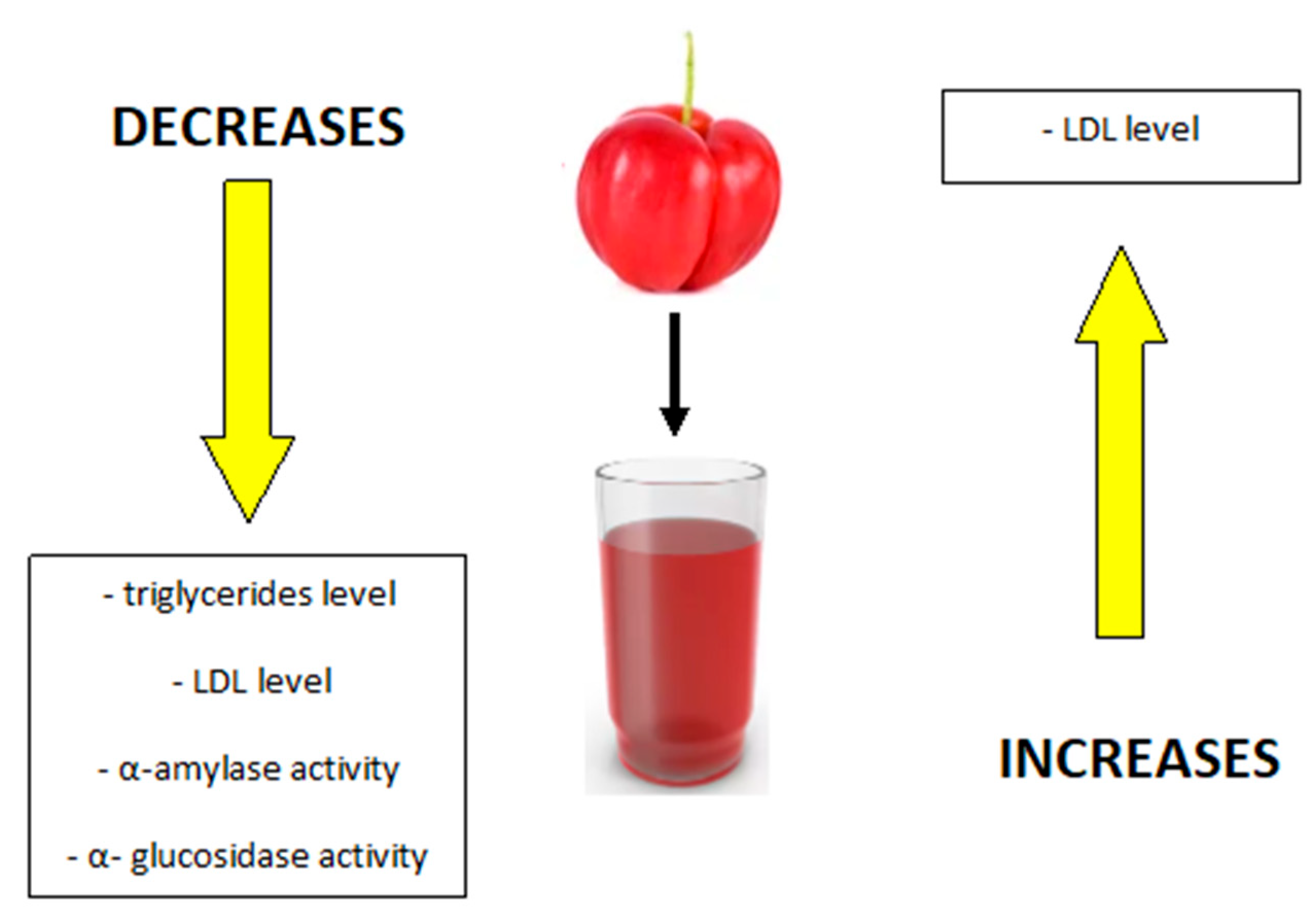
| In vivo | anti-atherosclerotic effect | [14] |
| estrogen-like effect | [14] | |
| anti-inflammatory effects | [13] | |
| supporting the process of weight loss | [15] | |
| protection against lipid disorders | [15] | |
| increase in catalase activity | [16] | |
| reduction of subcutaneous fat | [17] | |
| improving the microbiological balance in the intestines | [18] | |
| gastro-protective effect (stimulation of the renewal of intestinal epithelial cells) | [19] | |
| inhibition of lipid peroxidation in gastric and intestinal tissues | [19] | |
| detoxification effect | [16] | |
| increasing the concentration of glutathione (GSH) | [19] | |
| regeneration of the gastric mucosa | [19] | |
| stimulation of androgen synthesis | [16] | |
| increase in the number of muscle fibers (hyperplasia) and enlargement of the volume of muscle fibers (hypertrophy) | [16] | |
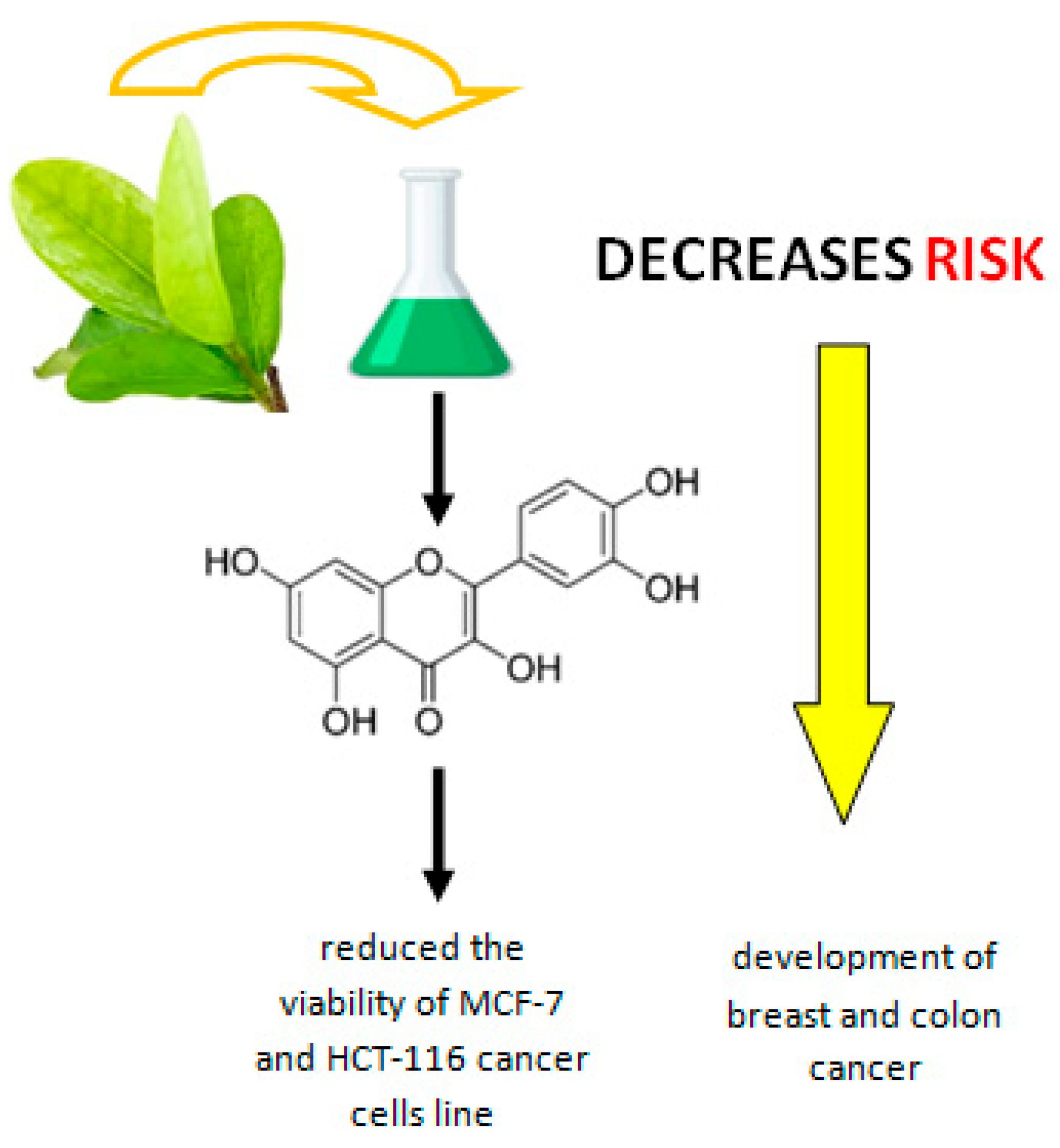
| antitumor effect; reduction of the viability of breast cancer cells (MCF-7) and colon (HCT-116 cell line) | [16] | |
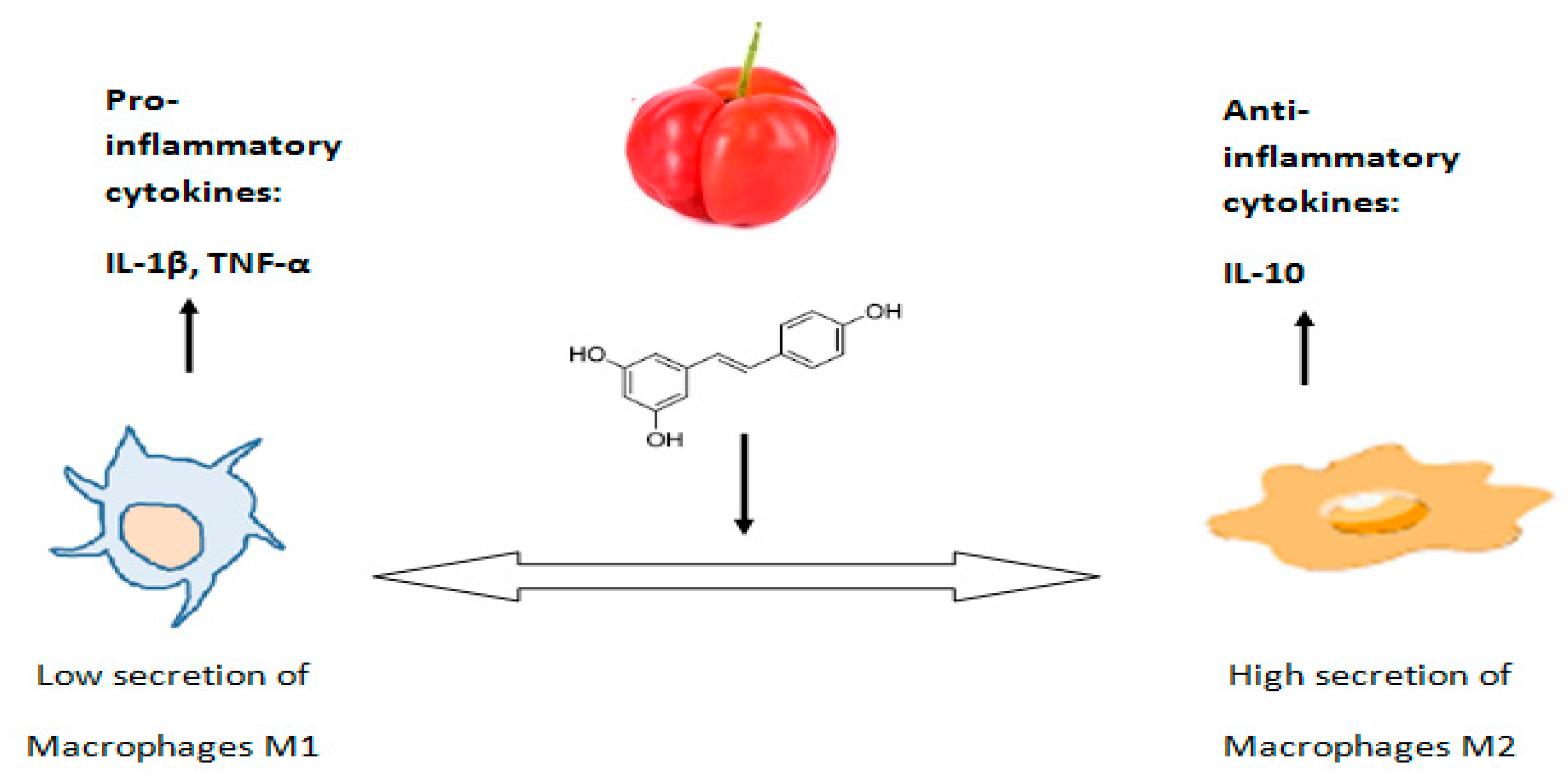
| In vitro | reduction of oxidative stress in macrophages | [20] |
| reduction of the secretion of the pro-inflammatory cytokines IL-1β, IL-6 and TNF-α | [20] | |
| increasing the secretion of anti-inflammatory cytokines such as IL-10 and TGF-β by M2 macrophage cells | [20] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olędzki, R.; Harasym, J. Acerola (Malpighia emarginata) Anti-Inflammatory Activity—A Review. Int. J. Mol. Sci. 2024, 25, 2089. https://doi.org/10.3390/ijms25042089
Olędzki R, Harasym J. Acerola (Malpighia emarginata) Anti-Inflammatory Activity—A Review. International Journal of Molecular Sciences. 2024; 25(4):2089. https://doi.org/10.3390/ijms25042089
Chicago/Turabian StyleOlędzki, Remigiusz, and Joanna Harasym. 2024. "Acerola (Malpighia emarginata) Anti-Inflammatory Activity—A Review" International Journal of Molecular Sciences 25, no. 4: 2089. https://doi.org/10.3390/ijms25042089
APA StyleOlędzki, R., & Harasym, J. (2024). Acerola (Malpighia emarginata) Anti-Inflammatory Activity—A Review. International Journal of Molecular Sciences, 25(4), 2089. https://doi.org/10.3390/ijms25042089






