Variation of the 2D Pattern of Brain Proteins in Mice Infected with Taenia crassiceps ORF Strain
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adamo, S.A. Modulating the Modulators: Parasites, Neuromodulators and Host Behavioral Change. Brain Behav. Evol. 2002, 60, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Hughes, D.P.; Libersat, F. Parasite manipulation of host behavior. Curr. Biol. 2019, 29, R45–R47. [Google Scholar] [CrossRef] [PubMed]
- Libersat, F.; Kaiser, M.; Emanuel, S. Mind Control: How Parasites Manipulate Cognitive Functions in Their Insect Hosts. Front. Psychol. 2018, 9, 572. [Google Scholar] [CrossRef] [PubMed]
- Berdoy, M.; Webster, J.P.; Macdonald, D.W. Fatal attraction in rats infected with Toxoplasma gondii. Proc. R. Soc. Lond. B Biol. Sci. 2000, 267, 1591–1594. [Google Scholar] [CrossRef] [PubMed]
- Peón, A.N.; Espinoza-Jiménez, A.; Terrazas, L.I. Immunoregulation by Taenia crassiceps and Its Antigens. BioMed Res. Int. 2012, 2013, 498583. [Google Scholar] [CrossRef]
- Gourbal, B.; Lacroix, A.; Gabrion, C. Behavioural dominance and Taenia crassiceps parasitism in BALB/c male mice. Parasitol. Res. 2002, 88, 912–917. [Google Scholar] [CrossRef]
- Morales-Montor, J.; Arrieta, I.; Del Castillo, L.I.; Rodríguez-Dorantes, M.; Cerbón, M.A.; Larralde, C. Remote sensing of intraperitoneal parasitism by the host’s brain: Regional changes of c-fos gene expression in the brain of feminized cysticercotic male mice. Parasitology 2004, 128 Pt 3, 343–351. [Google Scholar] [CrossRef]
- Morales-Montor, J.; Picazo, O.; Besedovsky, H.; Hernández-Bello, R.; López-Griego, L.; Becerril-Villanueva, E.; Moreno, J.; Pavón, L.; Nava-Castro, K.; Camacho-Arroyo, I. Helminth Infection Alters Mood and Short-Term Memory as well as Levels of Neurotransmitters and Cytokines in the Mouse Hippocampus. Neuroimmunomodulation 2014, 21, 195–205. [Google Scholar] [CrossRef]
- Ntoukas, V.; Tappe, D.; Pfütze, D.; Simon, M.; Holzmann, T. Cerebellar Cysticercosis Caused by Larval Taenia crassiceps Tapeworm in Immunocompetent Woman, Germany. Emerg. Infect. Dis. 2013, 19, 2008–2011. [Google Scholar] [CrossRef]
- Tsugita, A.; Kawakami, T.; Uchida, T.; Sakai, T.; Kamo, M.; Matsui, T.; Watanabe, Y.; Morimasa, T.; Hosokawa, K.; Toda, T. Proteome analysis of mouse brain: Two-dimensional electrophoresis profiles of tissue proteins during the course of aging. Electrophoresis 2000, 21, 1853–1871. [Google Scholar] [CrossRef]
- Eckerskorn, C.; Jungblut, P.; Mewes, W.; Klose, J.; Lottspeich, F. Identification of mouse brain proteins after two-dimensional electrophoresis and electroblotting by microsequence analysis and amino acid composition analysis. Electrophoresis 1988, 9, 830–838. [Google Scholar] [CrossRef]
- Gauss, C.; Kalkum, M.; Löwe, M.; Lehrach, H.; Klose, J. Analysis of the mouse proteome. (I) Brain proteins: Separation by two-dimensional electrophoresis and identification by mass spectrometry and genetic variation. Electrophoresis 1999, 20, 575–600. [Google Scholar] [CrossRef]
- Balestrino, M.; Lensman, M.; Parodi, M.; Perasso, L.; Rebaudo, R.; Melani, R.; Polenov, S.; Cupello, A. Role of creatine and phosphocreatine in neuronal protection from anoxic and ischemic damage. Amino Acids 2002, 23, 221–229. [Google Scholar] [CrossRef]
- Suzuki, T.; Mitake, S.; Okumura-Noji, K.; Shimizu, H.; Tada, T.; Fujii, T. Excitable membranes and synaptic transmission: Postsynaptic mechanisms.: Localization of α-internexin in the postsynaptic density of the rat brain. Brain Res. 1997, 765, 74–80. [Google Scholar] [CrossRef]
- Kuhla, B.; Boeck, K.; Lüth, H.-J.; Schmidt, A.; Weigle, B.; Schmitz, M.; Ogunlade, V.; Münch, G.; Arendt, T. Age-dependent changes of glyoxalase I expression in human brain. Neurobiol. Aging 2006, 27, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Bu, J.; Yao, X.; Liu, C.; Shen, H.; Li, X.; Li, H.; Chen, G. Phosphorylation at S153 as a Functional Switch of Phosphatidylethanolamine Binding Protein 1 in Cerebral Ischemia-Reperfusion Injury in Rats. Front. Mol. Neurosci. 2017, 10, 358. [Google Scholar] [CrossRef]
- Matoba, K.; Dohi, E.; Choi, E.Y.; Kano, S.-I. Glutathione S-transferases Control astrocyte activation and neuronal health during neuroinflammation. Front. Mol. Biosci. 2023, 9, 1080140. [Google Scholar] [CrossRef]
- Bezek, S.; Biberthaler, P.; Martinez-Espina, I.; Bogner-Flatz, V. Pathophysiology and clinical implementation of traumatic brain injury biomarkers: Neuron-specific enolase. In Biomarkers for Traumatic Brain Injury; Elsevier: Amsterdam, The Netherlands, 2020; pp. 169–182. [Google Scholar] [CrossRef]
- Hua, L.V.; Green, M.; Warsh, J.J.; Li, P.P. Molecular cloning of a novel isoform of diphosphoinositol polyphosphate phosphohydrolase: A potential target of lithium therapy. Neuropsychopharmacology 2001, 24, 640–651. [Google Scholar] [CrossRef] [PubMed]
- Austin, R.; Chander, P.; Zimmerman, A.J.; Overby, M.; Digilio, L.; Yap, C.C.; Linsenbardt, D.N.; Müller, H.K.; Weick, J.P. Global loss of Neuron-specific gene 1 causes alterations in motor coordination, increased anxiety, and diurnal hyperactivity in male mice. Genes Brain Behav. 2022, 21, e12816. [Google Scholar] [CrossRef]
- Kopajtich, R.; Nicholls, T.J.; Rorbach, J.; Metodiev, M.D.; Freisinger, P.; Mandel, H.; Vanlander, A.; Ghezzi, D.; Carrozzo, R.; Taylor, R.W.; et al. Mutations in GTPBP3 Cause a Mitochondrial Translation Defect Associated with Hypertrophic Cardiomyopathy, Lactic Acidosis, and Encephalopathy. Am. J. Hum. Genet. 2014, 95, 708–720. [Google Scholar] [CrossRef]
- Chebbok, E. Basic Leucine Zipper and W2 Domaincontaining Protein 2 (BZW2): A Novel Cardiac WNT Component. Ph.D. Thesis, Georg-August University Göttingen, Göttingen, Germany, September 2015. [Google Scholar] [CrossRef]
- Chapman, G.; Shanmugalingam, U.; Smith, P.D. The Role of Neuronal Pentraxin 2 (NP2) in Regulating Glutamatergic Signaling and Neuropathology. Front. Cell. Neurosci. 2020, 13, 575. [Google Scholar] [CrossRef]
- Peddada, N.; Sagar, A.; Ashish; Garg, R. Plasma gelsolin: A general prognostic marker of health. Med. Hypotheses 2012, 78, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.-T.; Yu, J.; Grass, D.; de Beer, F.C.; Kindy, M.S. Inflammation-Dependent Cerebral Deposition of Serum Amyloid A Protein in a Mouse Model of Amyloidosis. J. Neurosci. 2002, 22, 5900–5909. [Google Scholar] [CrossRef] [PubMed]
- Watson, W.; Srivastava, M.; Leighton, X.; Glasman, M.; Faraday, M.; Fossam, L.; Pollard, H.; Verma, A. Annexin 7 mobilizes calcium from endoplasmic reticulum stores in brain. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2004, 1742, 151–160. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Petroff, O.A.C. Book Review: GABA and Glutamate in the Human Brain. Neuroscientist 2002, 8, 562–573. [Google Scholar] [CrossRef] [PubMed]
- Okui, M.; Ito, F.; Ogita, K.; Kuramoto, N.; Kudoh, J.; Shimizu, N.; Ide, T. Expression of APG-2 protein, a member of the heat shock protein 110 family, in developing rat brain. Neurochem. Int. 2000, 36, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, D.A. Ubiquitin carboxyl-terminal hydrolase L-1 in brain: Focus on its oxidative/nitrosative modification and role in brains of subjects with Alzheimer disease and mild cognitive impairment. Free Radic. Biol. Med. 2021, 177, 278–286. [Google Scholar] [CrossRef]
- Huang, C.; Chen, M.; Pang, D.; Bi, D.; Zou, Y.; Xia, X.; Yang, W.; Luo, L.; Deng, R.; Tan, H.; et al. Developmental and Activity-Dependent Expression of LanCL1 Confers Antioxidant Activity Required for Neuronal Survival. Dev. Cell 2014, 30, 479–487. [Google Scholar] [CrossRef]
- Horibata, Y.; Elpeleg, O.; Eran, A.; Hirabayashi, Y.; Savitzki, D.; Tal, G.; Mandel, H.; Sugimoto, H. EPT1 (selenoprotein I) is critical for the neural development and maintenance of plasmalogen in humans. J. Lipid Res. 2018, 59, 1015–1026. [Google Scholar] [CrossRef]
- Ellis, J.M.; Wong, G.W.; Wolfgang, M.J. Acyl Coenzyme A Thioesterase 7 Regulates Neuronal Fatty Acid Metabolism to Prevent Neurotoxicity. Mol. Cell. Biol. 2013, 33, 1869–1882. [Google Scholar] [CrossRef]
- Murshid, A.; Srivastava, A.; Kumar, R.; Presley, J.F. Characterization of the localization and function of NECAP 1 in neurons. J. Neurochem. 2006, 98, 1746–1762. [Google Scholar] [CrossRef] [PubMed]
- Kleopa, K.A.; Orthmann-Murphy, J.; Sargiannidou, I. Gap Junction Disorders of Myelinating Cells. Rev. Neurosci. 2010, 21, 397–419. [Google Scholar] [CrossRef] [PubMed]
- Bartesaghi, S.; Betts-Henderson, J.; Cain, K.; Dinsdale, D.; Zhou, X.; Karlsson, A.; Salomoni, P.; Nicotera, P. Loss of thymidine kinase 2 alters neuronal bioenergetics and leads to neurodegeneration. Hum. Mol. Genet. 2010, 19, 1669–1677. [Google Scholar] [CrossRef] [PubMed]
- Tatton, W.G.; Chalmers-Redman, R.M.E.; Elstner, M.; Leesch, W.; Jagodzinski, F.B.; Stupak, D.P.; Sugrue, M.M.; Tatton, N.A. Glyceraldehyde-3-phosphate dehydrogenase in neurodegeneration and apoptosis signaling. In Advances in Research on Neurodegeneration; Springer: Vienna, Austria, 2000; pp. 77–100. [Google Scholar] [CrossRef]
- Liu, G.; Ruan, Y.; Zhang, J.; Wang, X.; Wu, W.; He, P.; Wang, J.; Xiong, J.; Cheng, Y.; Liu, L.; et al. ABHD11 Is Critical for Embryonic Stem Cell Expansion, Differentiation and Lipid Metabolic Homeostasis. Front. Cell Dev. Biol. 2020, 8, 570. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Cheng, N.; Xu, X.; Yang, Z.; Hoffman, R.M.; Zhu, J. AMMECR1 Inhibits Apoptosis and Promotes Cell-cycle Progression and Proliferation of the A549 Human Lung Cancer Cell Line. Anticancer Res. 2019, 39, 4637–4642. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.T.; Balogh, S.A.; Davydov, I.V.; Kashina, A.S.; Yoon, J.K.; Xie, Y.; Gaur, A.; Hyde, L.; Denenberg, V.H.; Varshavsky, A. Altered activity, social behavior, and spatial memory in mice lacking the NTAN1p amidase and the asparagine branch of the N-end rule pathway. Mol. Cell. Biol. 2000, 20, 4135–4148. [Google Scholar] [CrossRef] [PubMed]
- Balog, J.; Mehta, S.L.; Vemuganti, R. Mitochondrial fission and fusion in secondary brain damage after CNS insults. J. Cereb. Blood Flow Metab. 2016, 36, 2022–2033. [Google Scholar] [CrossRef]
- Westergard, L.; Christensen, H.M.; Harris, D.A. The cellular prion protein (PrPC): Its physiological function and role in disease. Biochim. Biophys. Acta (BBA)—Mol. Basis Dis. 2007, 1772, 629–644. [Google Scholar] [CrossRef]
- Schrader, M.; Reuber, B.E.; Morrell, J.C.; Jimenez-Sanchez, G.; Obie, C.; Stroh, T.A.; Valle, D.; Schroer, T.A.; Gould, S.J. Expression of PEX11β Mediates Peroxisome Proliferation in the Absence of Extracellular Stimuli. J. Biol. Chem. 1998, 273, 29607–29614. [Google Scholar] [CrossRef]
- Amorim, I.S.; Lach, G.; Gkogkas, C.G. The Role of the Eukaryotic Translation Initiation Factor 4E (eIF4E) in Neuropsychiatric Disorders. Front. Genet. 2018, 9, 561. [Google Scholar] [CrossRef]
- Tabe, S.; Hikiji, H.; Ariyoshi, W.; Hashidate-Yoshida, T.; Shindou, H.; Shimizu, T.; Okinaga, T.; Seta, Y.; Tominaga, K.; Nishihara, T. Lysophosphatidylcholine acyltransferase 4 is involved in chondrogenic differentiation of ATDC5 cells. Sci. Rep. 2017, 7, 16701. [Google Scholar] [CrossRef]
- López-Griego, L.; Nava-Castro, K.E.; López-Salazar, V.; Hernández-Cervantes, R.; Guzmán, N.T.; Muñiz-Hernández, S.; Hernández-Bello, R.; Besedovsky, H.O.; Pavón, L.; Villanueva, L.E.B.; et al. Gender-Associated Differential Expression of Cytokines in Specific Areas of the Brain During Helminth Infection. J. Interf. Cytokine Res. 2015, 35, 116–125. [Google Scholar] [CrossRef]
- Chavarria, A.; Alcocer-Varela, J. Is damage in central nervous system due to inflammation? Autoimmun. Rev. 2004, 3, 251–260. [Google Scholar] [CrossRef]
- Mishra, B.B.; Mishra, P.K.; Teale, J.M. Expression and distribution of Toll-like receptors in the brain during murine neurocysticercosis. J. Neuroimmunol. 2006, 181, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, L.; Díaz-Zaragoza, M.; Hernández, M.; Navarro, L.; Hernández-Ávila, R.; Encarnación-Guevara, S.; Ostoa-Saloma, P.; Landa, A. Differential Protein Expression of Taenia crassiceps ORF Strain in the Murine Cysticercosis Model Using Resistant (C57BL/6) Mice. Pathogens 2023, 12, 678. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Zaragoza, M.; Jiménez, L.; Hernández, M.; Hernández-Ávila, R.; Navarro, L.; Ochoa-Sánchez, A.; Encarnación-Guevara, S.; Ostoa-Saloma, P.; Landa, A. Protein expression profile of Taenia crassiceps cysticerci related to Th1- and Th2-type responses in the mouse cysticercosis model. Acta Trop. 2020, 212, 105696. [Google Scholar] [CrossRef] [PubMed]
- Taraslia, V.K.; Kouskoukis, A.; Anagnostopoulos, A.K.; Stravopodis, D.; Margaritis, L.H.; Tsangaris, G.T. Proteomic Analysis of Normal Murine Brain Parts. Cancer Genom. Proteom. 2013, 10, 125–154. [Google Scholar]
- Sharma, K.; Schmitt, S.; Bergner, C.G.; Tyanova, S.; Kannaiyan, N.; Manrique-Hoyos, N.; Kongi, K.; Cantuti, L.; Hanisch, U.-K.; Philips, M.-A.; et al. Cell type- and brain region-resolved mouse brain proteome. Nat. Neurosci. 2015, 18, 1819–1831. [Google Scholar] [CrossRef]
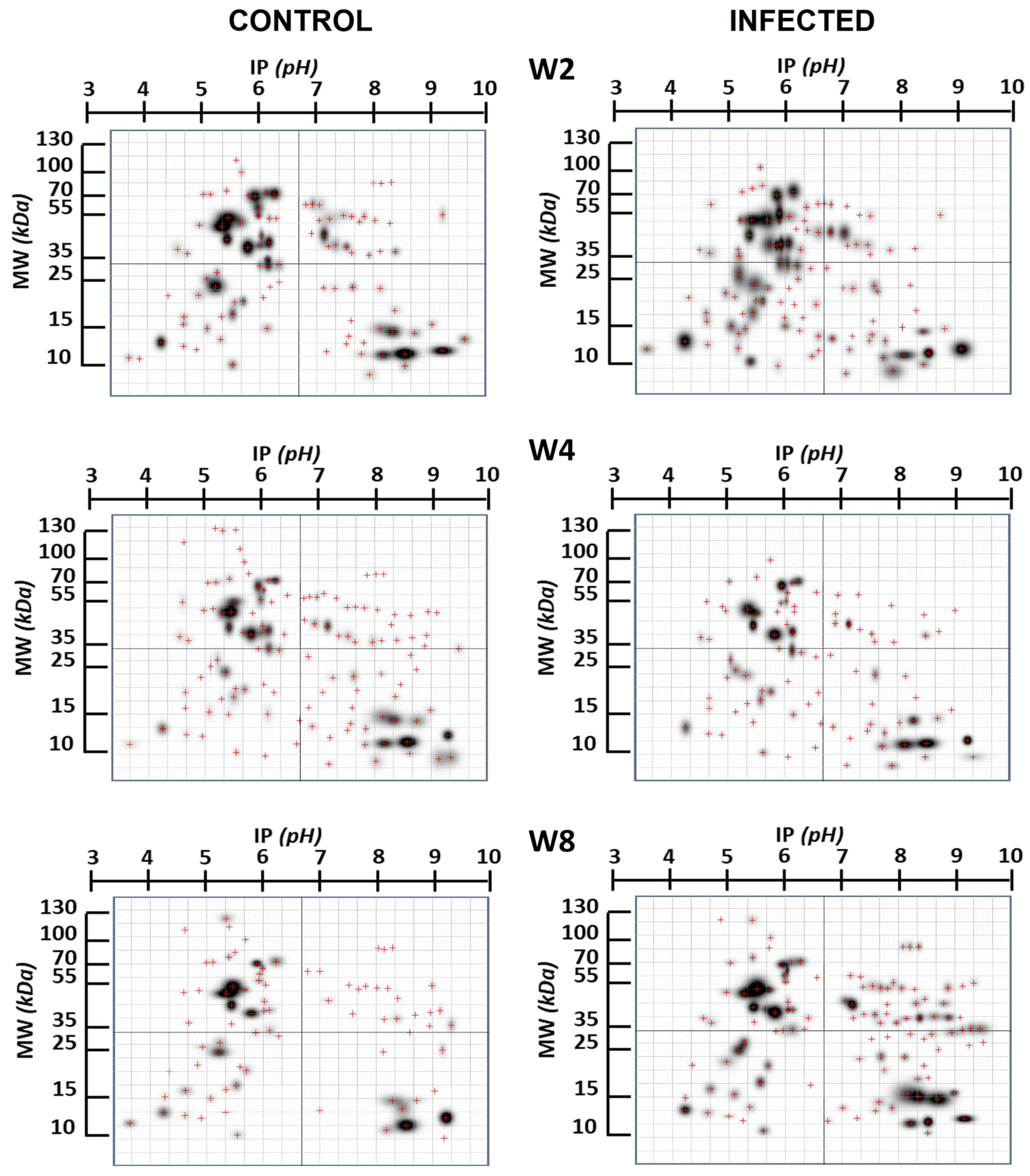
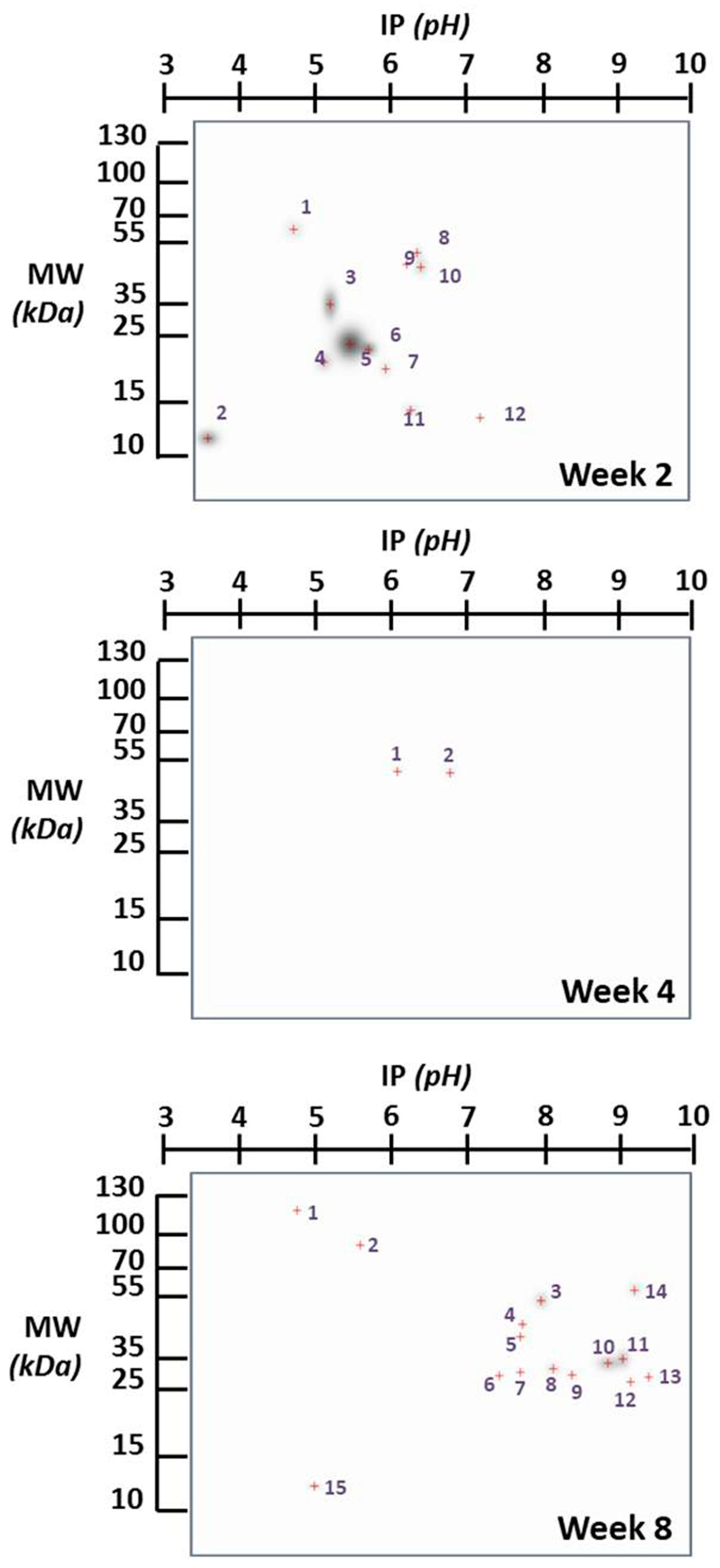
| Infection Time | N° Spot | ~MW Spot | ~IP Spot | Probable Protein | MW Protein | IP Protein | Refs. |
|---|---|---|---|---|---|---|---|
| Week 2 | 1 | 62.24 | 4.7 | Neurofilament protein NF-66 mRNA α-Internexin | 61 | 4.79 | [13] |
| 2 | 11.8 | 3.6 | U6 snRNA-associated Sm-like protein LSm3 | 11.8 | 4.08 | [14] | |
| 3 | 35 | 5.2 | Creatine kinase b-chain | 34.09 | 5.22 | [15] | |
| 4 | 21.11 | 5.1 | Lactoylglutathione Lyase Phosphatidylethanolamine-binding protein mRNA | 21 21 | 5.15 5.11 | [16,17] | |
| 5 | 23.66 | 5.45 | Glutathione S-transferase P 1 | 23.6 | 5.43 | [18] | |
| 6 | 22.78 | 5.7 | γ-Enolase | 25.24 | 5.73 | [19] | |
| 7 | 20 | 5.95 | Diphosphoinositol polyphosphate phosphohydrolase 2 Neuron-specific protein family member 1 | 20.16 20.93 | 5.99 5.99 | [20] | |
| 8 | 52 | 6.35 | tRNA modification GTPase GTPBP3, mitochondrial | 52.1 | 6.37 | [21] | |
| 9 | 48 | 6.2 | Basic leucine zipper and W2 domain-containing protein 2 | 48.06 | 6.26 | [22] | |
| 10 | 47 | 6.4 | Neuronal pentraxin-2 | 47.14 | 6.3 | [23] | |
| 11 | 14.2 | 6.3 | Gelsolin precursor, plasma | 17.13 | 6.21 | [24] | |
| 12 | 13.67 | 7.2 | Serum amyloid A-3 protein | 13.7 | 6.0, 6.4, 7.0, 7.4, 7.5, 8.0 | [25] | |
| Infection Time | N° spot | ~MW spot | IP spot | Probable protein | MW protein | IP protein | |
| Week 4 | 1 2 | 51.25 50.5 | 6.05 6.75 | Annexin VII (synexin) 4-aminobutyrat aminotransferanse, mitochondrial precursor | 50 52.83 | 6.02 6.78 | [26] [27] |
| Infection Time | N° spot | ~MW spot | ~IP spot | Probable protein | MW protein | IP protein | |
| 1 | 118 | 4.79 | Apg-2 mRNA | 120 | 5.0 | [28] | |
| 2 | 96.67 | 5.6 | Ubiquitin carboxyl-terminal hydrolase | 96.7 | 5.42 | [29] | |
| 3 | 57.75 | 7.98 | ------------------------------------------------------------------------------- | ----- | ---- | ||
| 4 | 45.5 | 7.75 | LanC-like protein 1 Ethanolamine phosphotransferase 1 | 45.34 45.35 | 7.57 7.33 | [30] [31] | |
| 5 | 42 | 7.7 | Cytosolic acyl coenzyme A thioester hydrolase mRNA | 42.11 | 7.65 | [32] | |
| 6 | 29.5 | 7.45 | Adaptin ear-binding coat-associated protein 1 | 29.63 | 7.72 | [33] | |
| Week 8 | 7 | 30 | 7.72 | Gap junction gamma-3 protein | 30.29 | 7.71 | [34] |
| 8 | 31.25 | 8.14 | Thymidine kinase 2, mitochondrial | 31.26 | 8.71 | [35] | |
| 9 | 29.5 | 8.38 | Glyceraldehide 3-phosphate deshydrogenase | 28.93 | 8.39 | [36] | |
| 10 | 33 | 8.85 | Alpha/beta hydrolase domain-containing protein 11 | 33.56 | 8.86 | [37] | |
| 11 | 34.5 | 9.15 | AMMECR1-like protein Protein N-terminal asparagine amidohydrolase | 34.52 34.59 | 9.18 9.07 | [38] [39] | |
| 12 | 27 | 9.18 | Mitochondrial fission factor Major prion protein | 27.22 27.98 | 9.1 9.13 | [40] [41] | |
| 13 | 28.75 | 9.4 | Peroxisomal membrane protein 11B | 28.72 | 9.68 | [42] | |
| 14 | 57.07 | 9.3 | Lysophospholipid acyltransferase LPCAT4 | 57.1 | 8.65 | [43] | |
| 15 | 12.33 | 5.0 | Eukaryotic translation initiation factor 4E-binding protein 1 | 12.33 | 5.32 | [44] |
| Protein | Pathway | KEEG Code |
|---|---|---|
| Neurofilament protein NF-66 mRNA α-Internexin | Cytoskeleton | --- |
| U6 snRNA-associated Sm-like protein LSm3 | RNA degradation | mmu:67678 |
| Creatine kinase b-chain | Arginine and proline metabolism | mmu:12709 |
| Lactoylglutathione Lyase | Pyruvate metabolism | mmu:109801 |
| Phosphatidylethanolamine-binding protein mRNA | Peptidase inhibitors | mmu:23980 |
| Glutathione S-transferase P 1 | Glutathione metabolism | mmu:14858 |
| γ-Enolase | Glycolysis/Gluconeogenesis | mmu:13807 |
| Diphosphoinositol polyphosphate phosphohydrolase 2 | Hydrolase | mmu:71207 |
| Neuron-specific protein family member 1 | Membrane trafficking | rno:25247 |
| tRNA modification GTPase GTPBP3, mitochondrial | Transfer Rna biogenesis | mmu:70359 |
| Basic leucine zipper and W2 domain-containing protein 2 | Translation regulator | mmu:66912 |
| Neuronal pentraxin-2 | Signaling proteins | --- |
| Gelsolin precursor, plasma | Phagocytosis/Regulation of actin cytoskeleton | mmu:56320 |
| Serum amyloid A-3 protein | Exosome | mmu:20210 |
| Annexin VII (synexin) | Amyotrophic lateral sclerosis | mmu:11750 |
| 4-aminobutyrat aminotransferanse, mitochondrial precursor | Alanine, aspartate, and glutamate metabolism Valine, leucine, and isoleucine degradation Beta-Alanine metabolism Ropanoate metabolism Butanoate metabolism Metabolic pathways GABAergic synapse | mmu:268860 |
| Apg-2 mRNA | Heat shock protein | --- |
| Ubiquitin carboxyl-terminal hydrolase | DNA repair and recombination proteins Ubiquitin system | mmu:230484 |
| LanC-like protein 1 | Glutathione metabolism | mmu:14768 |
| Ethanolamine phosphotransferase 1 | Phosphonate and phosphinate metabolism Glycerophospholipid metabolism Ether lipid metabolism | mmu:99712 |
| Cytosolic acyl coenzyme A thioester hydrolase mRNA | Fatty acid elongation Biosynthesis of unsaturated fatty acids Ovarian steroidogenesis | mmu:26897 |
| Adaptin ear-binding coat-associated protein 1 | Membrane trafficking | mmu:67602 |
| Gap junction gamma-3 protein | Pores ion channels | mmu:118446 |
| Thymidine kinase 2, mitochondrial | Nucleotido metabolism | --- |
| Glyceraldehide 3-phosphate deshydrogenase | Glycolysis/Gluconeogenesis Carbon metabolism Biosynthesis of amino acids Hif-1 signaling pathway Alzheimer’s disease | mmu:14433 |
| Alpha/beta hydrolase domain-containing protein 11 | Serine peptidases | hsa:83451 |
| Junctional adhesion molecule B | Cell adhesion molecules Tight junction Leukocyte transendothelial migration | mmu:67374 |
| AMMECR1-like protein | Signaling proteins | mmu:225339 |
| Protein N-terminal asparagine amidohydrolase | Ubiquitin system | mmu:18203 |
| Mitochondrial fission factor | Mitochondrial biogenesis | mmu:75734 |
| Major prion protein | Ferroptosis Prion disease—Mus musculus (house mouse) Pathways of neurodegeneration | mmu:19122 |
| Peroxisomal membrane protein 11B | Peroxisome | mmu:18632 |
| Lysophospholipid acyltransferase LPCAT4 | Glycerophospholipid metabolism Ether lipid metabolism | mmu:99010 |
| Eukaryotic translation initiation factor 4E-binding protein 1 | Egfr tyrosine kinase inhibitor resistance Hif-1 signaling pathway Mtor signaling pathway Pi3k-Akt signaling pathway Longevity regulating pathway Insulin signaling pathway | mmu:13684 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz-Zaragoza, M.; Hernández-Ávila, R.; Landa, A.; Ostoa-Saloma, P. Variation of the 2D Pattern of Brain Proteins in Mice Infected with Taenia crassiceps ORF Strain. Int. J. Mol. Sci. 2024, 25, 1460. https://doi.org/10.3390/ijms25031460
Díaz-Zaragoza M, Hernández-Ávila R, Landa A, Ostoa-Saloma P. Variation of the 2D Pattern of Brain Proteins in Mice Infected with Taenia crassiceps ORF Strain. International Journal of Molecular Sciences. 2024; 25(3):1460. https://doi.org/10.3390/ijms25031460
Chicago/Turabian StyleDíaz-Zaragoza, Mariana, Ricardo Hernández-Ávila, Abraham Landa, and Pedro Ostoa-Saloma. 2024. "Variation of the 2D Pattern of Brain Proteins in Mice Infected with Taenia crassiceps ORF Strain" International Journal of Molecular Sciences 25, no. 3: 1460. https://doi.org/10.3390/ijms25031460
APA StyleDíaz-Zaragoza, M., Hernández-Ávila, R., Landa, A., & Ostoa-Saloma, P. (2024). Variation of the 2D Pattern of Brain Proteins in Mice Infected with Taenia crassiceps ORF Strain. International Journal of Molecular Sciences, 25(3), 1460. https://doi.org/10.3390/ijms25031460






