Effects of Brassinosteroid on the Physiological Changes on Two Varieties of Tea Plants Under Salt Stress
Abstract
1. Introduction
2. Results
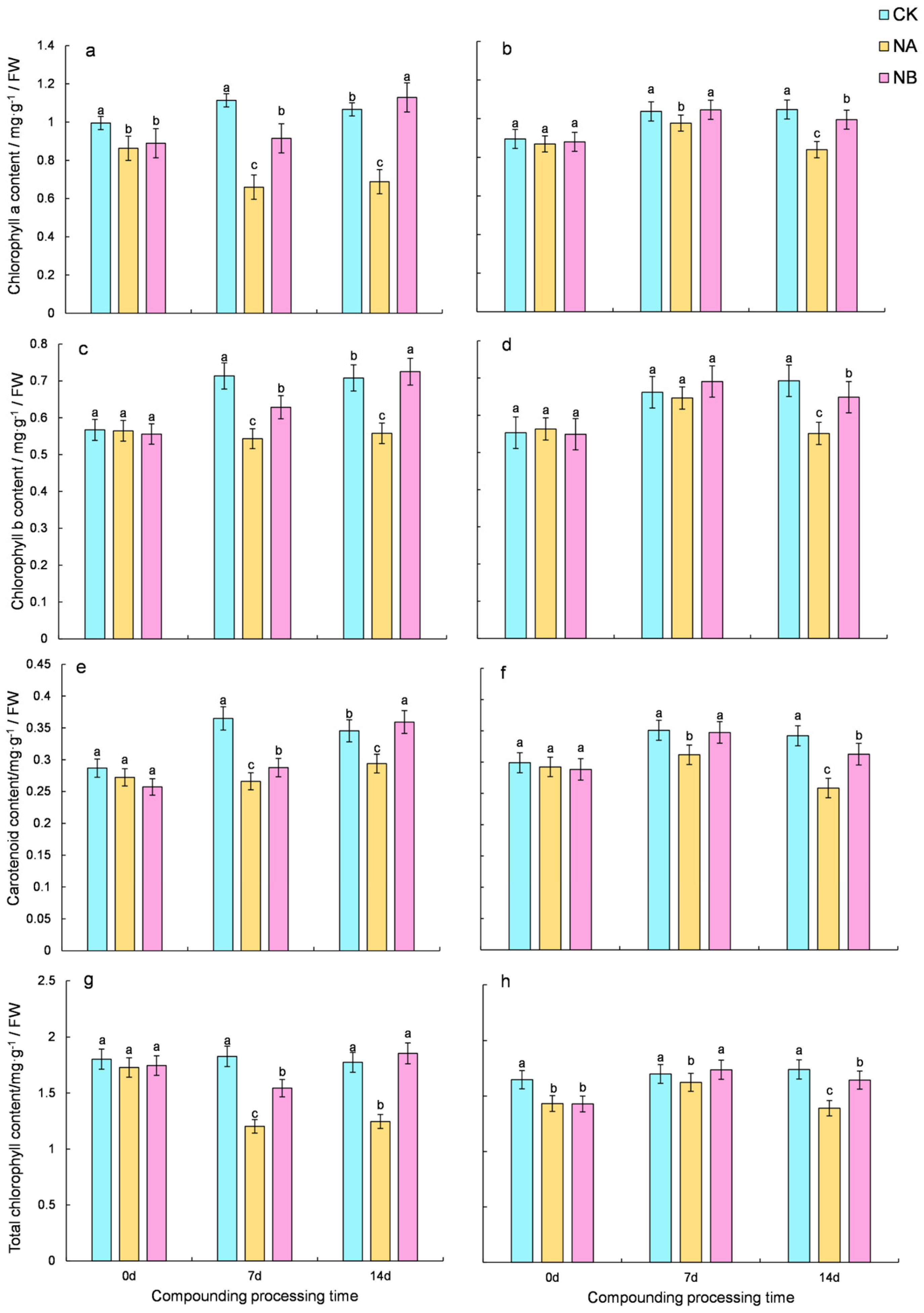
2.1. Effects of Brassinosteroids on Photosynthetic Pigments Under Salt Stress in C. sinensis
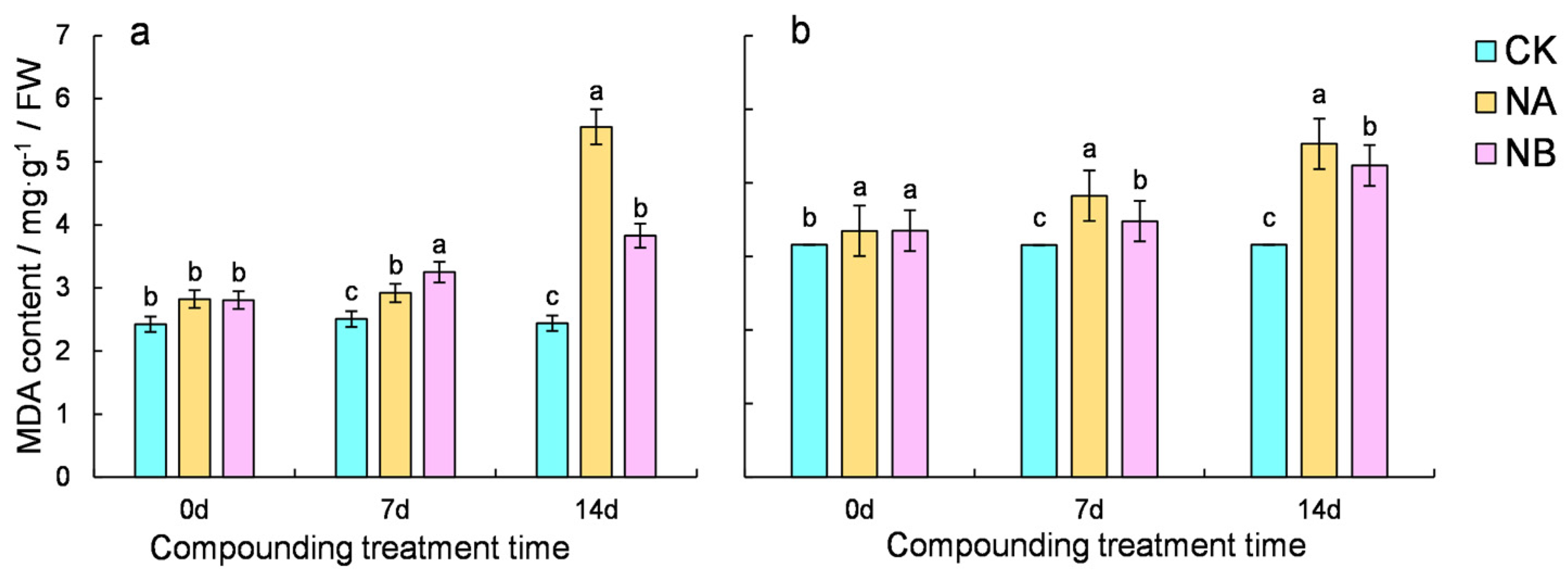
2.2. Effect of Brassinosteroids on MDA in Tea Plants Under Salt Stress
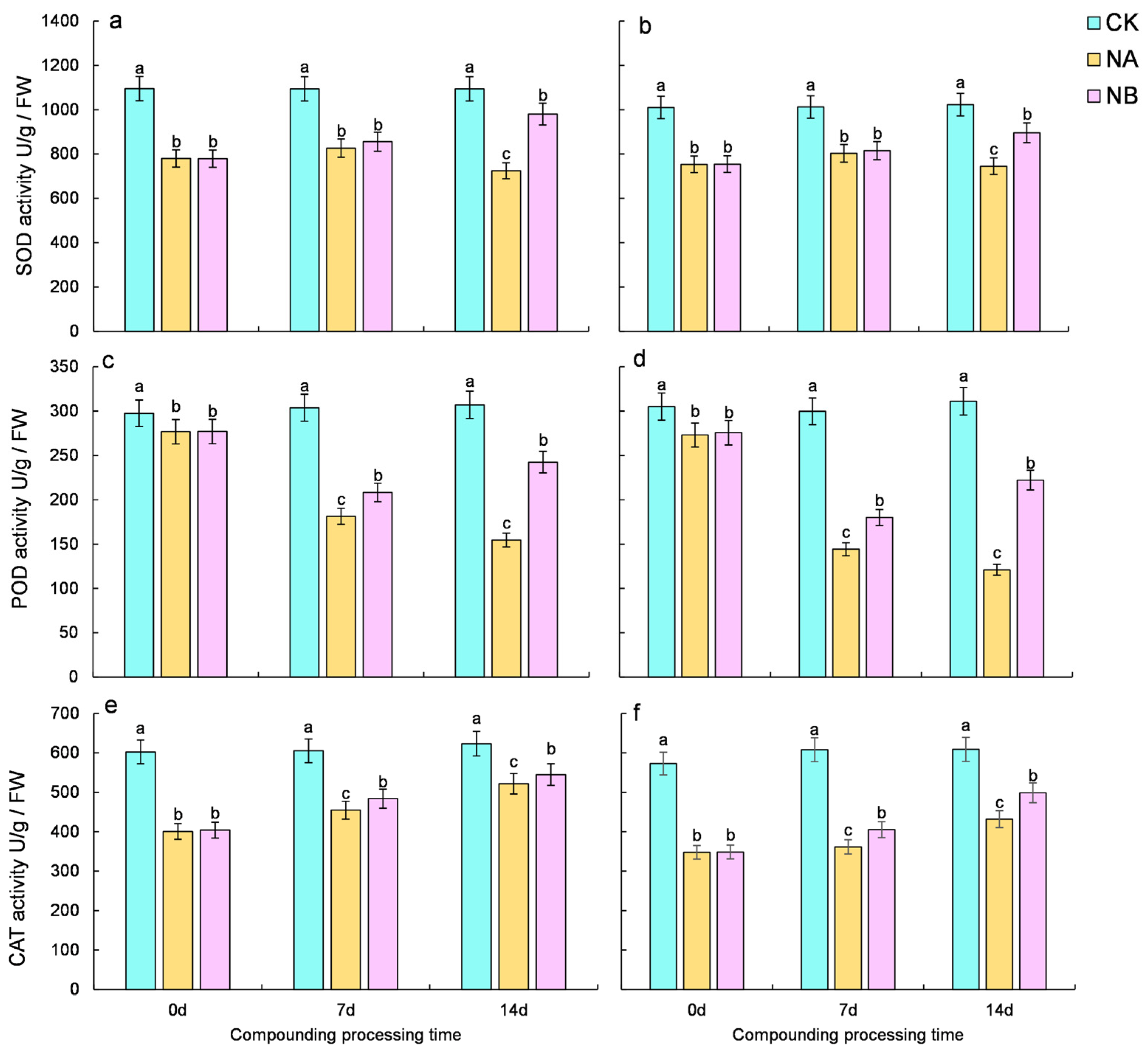
2.3. Effect of Brassinosteroids on the Enzyme Antioxidant System of Camellia Sinensis Under Salt Stress
2.4. Changes in Contents of Soluble Sugars, Soluble Proteins, and Proline
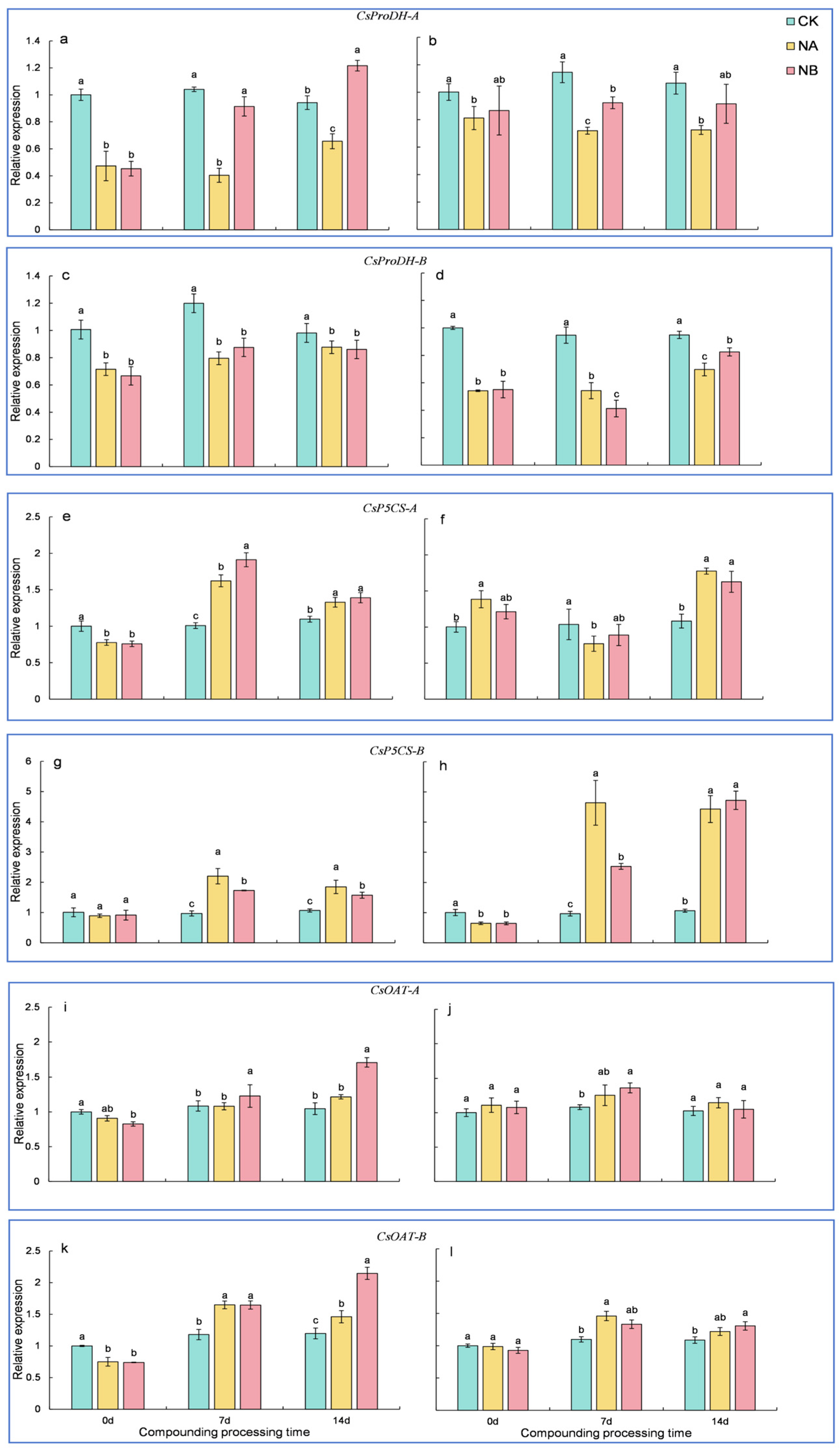
2.5. The Effect of Exogenous Brassinosteroids (BRs) on the Expression of Genes Related to the Proline Metabolism Pathway in Tea Plants Under Salt Stress
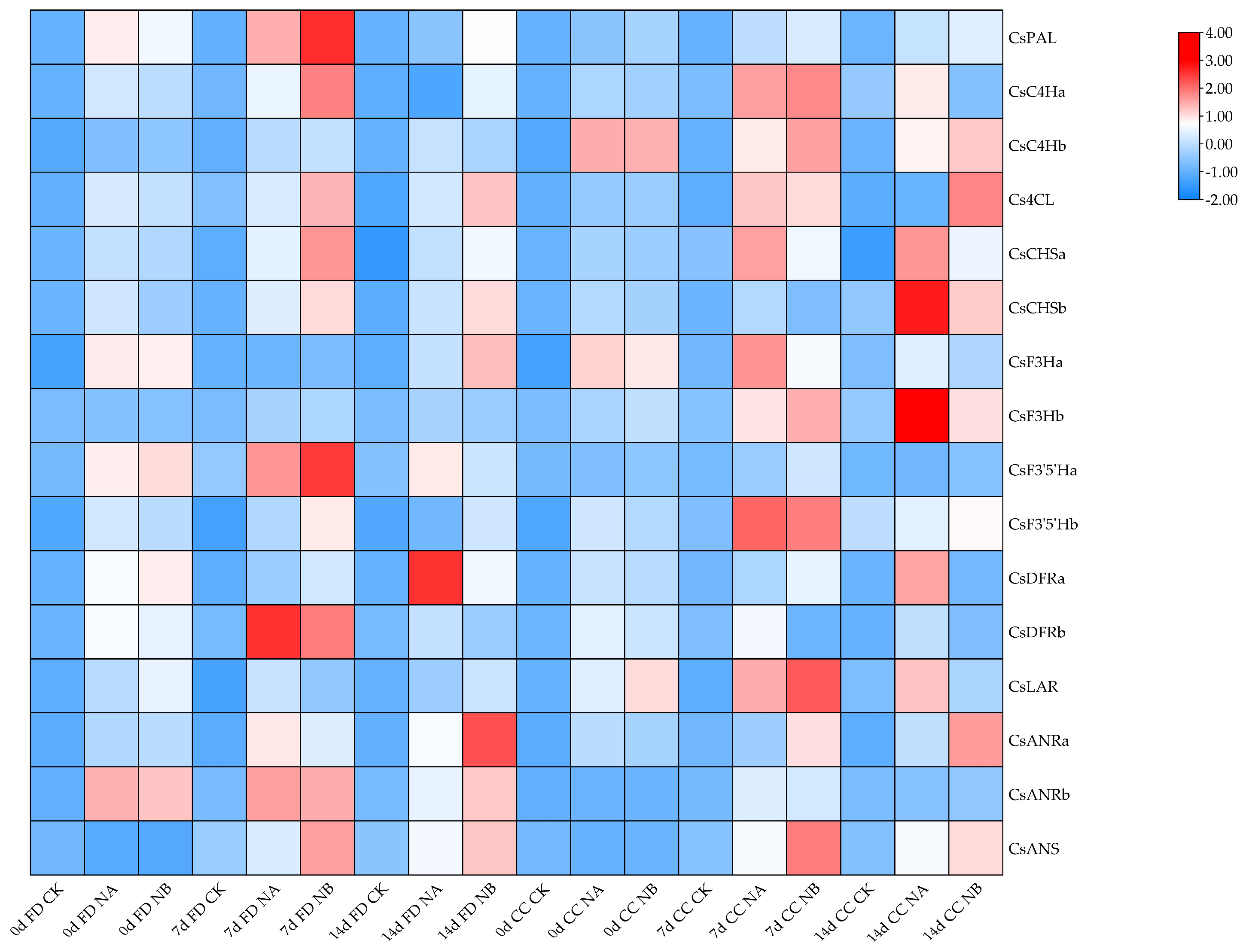
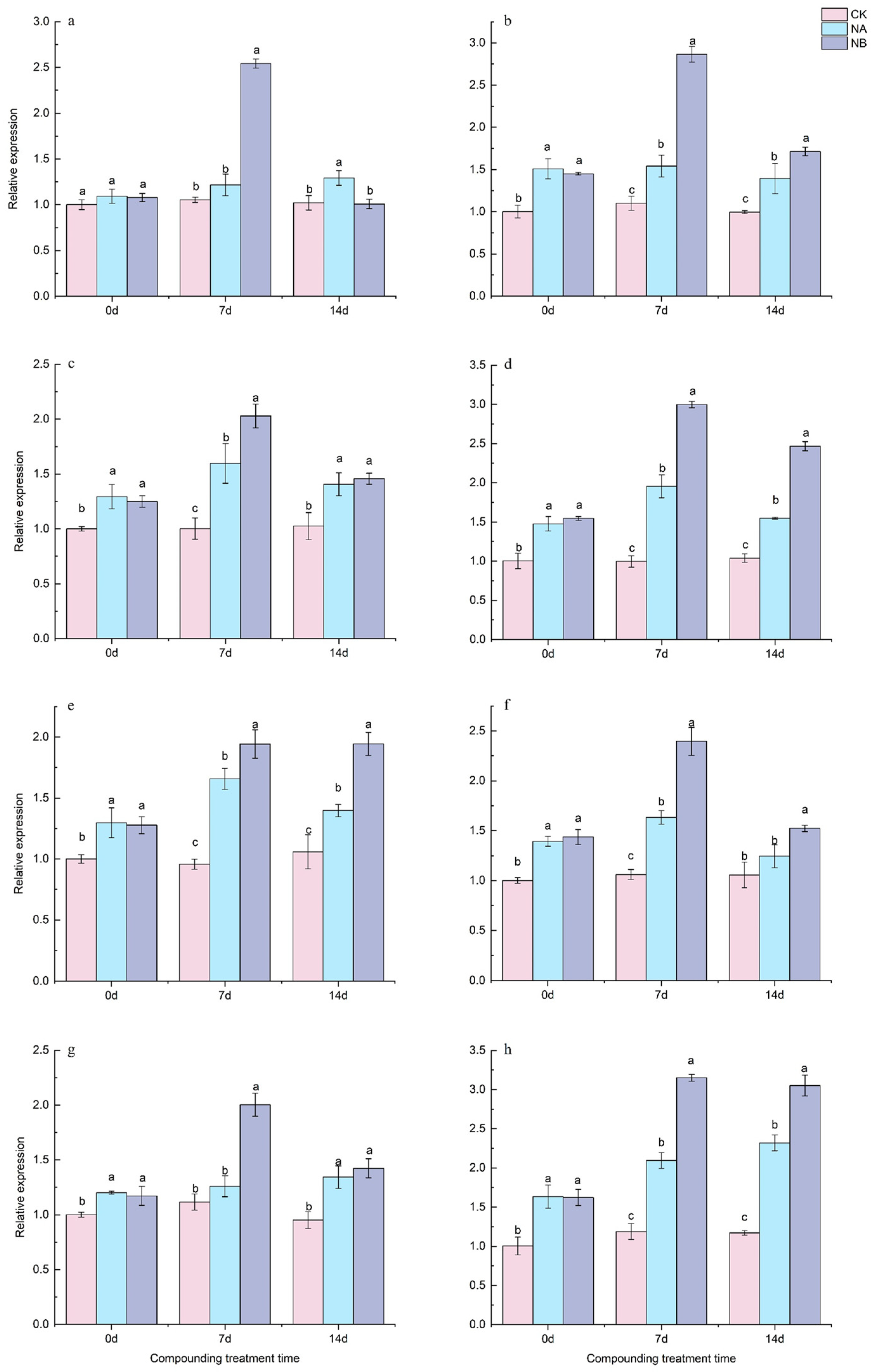
2.6. The Effect of Exogenous Brassinosteroids (BRs) on the Expression of Genes Related to the Flavonoid Synthesis Pathway in Tea Plants Under Salt Stress
2.7. The Effect of Exogenous Brassinosteroids (BRs) on the Expression of Genes Related to the Theanine Synthesis Pathway in Tea Plants Under Salt Stress
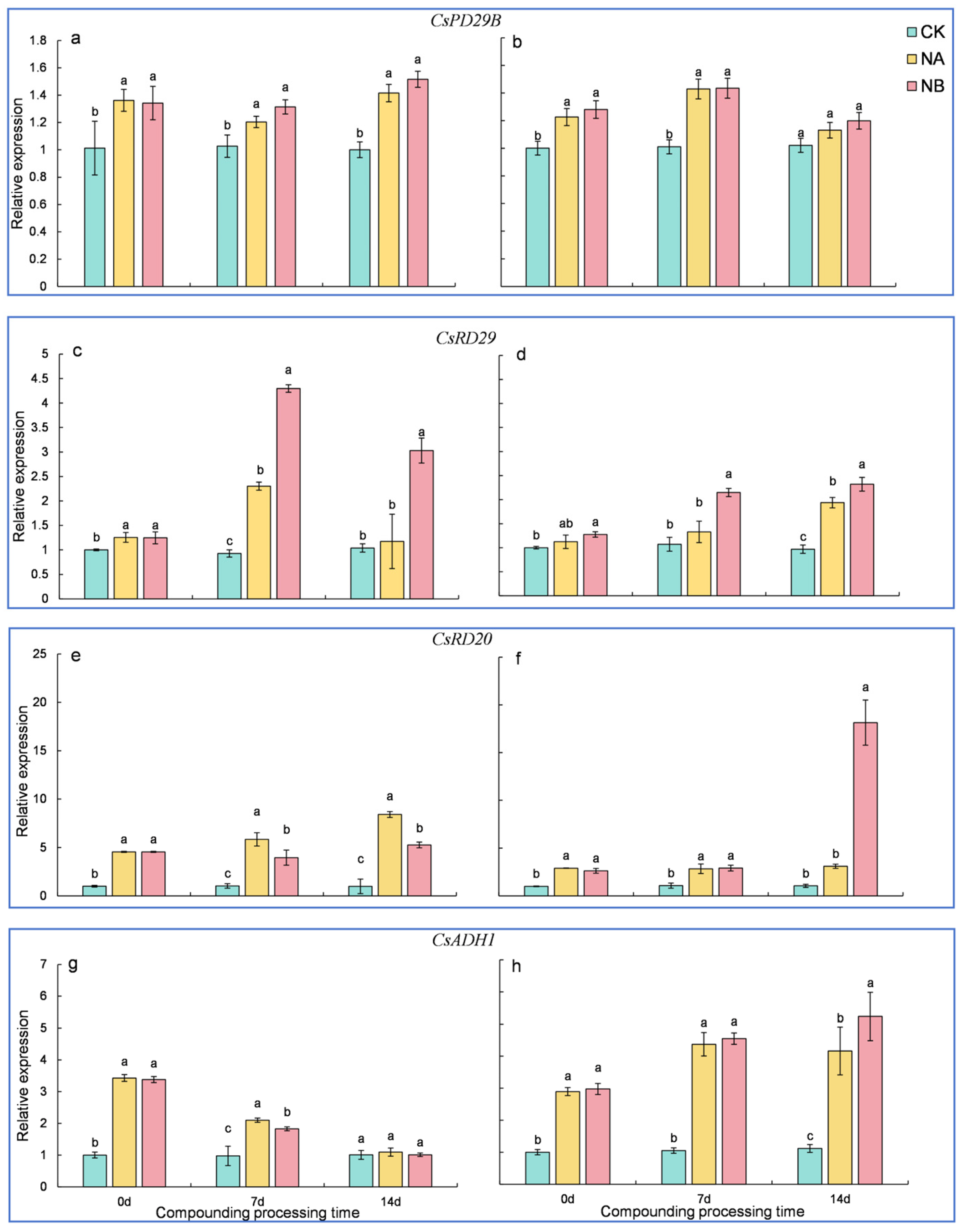
2.8. Genes Related to Stress
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Salt Stress Treatments
4.2. Determination of Physiological Indicators
4.3. Total RNA Isolation and cDNA Synthesis
4.4. Real-Time Quantitative PCR (qPCR) Detection of Genes Involved in Proline, Flavonoid, and Theanine Metabolism Pathway and Stress Response Markers
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, F.; Lu, K.; Gu, Y.; Zhang, L.; Li, W.; Li, Z. Effects of Low-Temperature Stress and Brassinolide Application on the Photosynthesis and Leaf Structure of Tung Tree Seedlings. Front. Plant Sci. 2019, 10, 1767. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.L.; Dong, X.Y.; Yan, D.H.; Qu, Y.C.; Liu, Y.L.; Hu, H.T. The effect of 2,4-electrobrassinolide on lotus under high temperature stress. J. Zhejiang Agric. Sci. 2022, 63, 63–68. [Google Scholar] [CrossRef]
- Xian, X.L.; Zhang, D.; Zhang, Z.X.; Wang, S.C.; Gao, Y.L.; Wang, Y.X. The effect of 2,4-electrobrassinolide on the physiological characteristics of drooping crabapple under drought stress. Agric. Res. Arid. Areas 2022, 40, 37–45. [Google Scholar]
- Waisi, H.; Janković, B.; Janković, M.; Nikolić, B.; Dimkić, I.; Lalević, B.; Raičević, V. New insights in dehydration stress behavior of two maize hybrids using advanced distributed reactivity model (DRM). Responses to the impact of 24-epibrassinolide. PLoS ONE 2017, 12, e0179650. [Google Scholar] [CrossRef]
- Wei, L.J.; Deng, X.G.; Zhu, T.; Zheng, T.; Li, P.X.; Wu, J.Q.; Zhang, D.W.; Lin, H.H. Ethylene is Involved in Brassinosteroids Induced Alternative Respiratory Pathway in Cucumber (Cucumis sativus L.) Seedlings Response to Abiotic Stress. Front. Plant Sci. 2015, 6, 982. [Google Scholar] [CrossRef]
- Wang, A.L.; Wang, Y.Y. The effect of brassinolide on stress resistance of wetland pine seedlings. J. Nanjing For. Univ. 1993, 3, 27. [Google Scholar]
- Su, Q.; Zheng, X.; Tian, Y.; Wang, C. Exogenous Brassinolide Alleviates Salt Stress in Malus hupehensis Rehd. by Regulating the Transcription of NHX-Type Na(+)(K(+))/H(+) Antiporters. Front. Plant Sci. 2020, 11, 38. [Google Scholar] [CrossRef]
- Wang, X.F.; Wang, L.B.; Wang, X.G.; Yu, Y.H.; Wang, J.L.; Ma, Y.Q. Study on the induction of salt resistance in pepper seedlings by exogenous brassinosteroids. J. China Capsicum 2012, 10, 23–25. [Google Scholar] [CrossRef]
- Zhang, L.Q. The effect of brassinosteroids on physiological indicators of tomato seedlings under salt stress. N. Hortic. 2013, 1, 1–3. [Google Scholar]
- Li, W.; Sun, J.; Zhang, X.; Ahmad, N.; Hou, L.; Zhao, C.; Pan, J.; Tian, R.; Wang, X.; Zhao, S. The Mechanisms Underlying Salt Resistance Mediated by Exogenous Application of 24-Epibrassinolide in Peanut. Int. J. Mol. Sci. 2022, 23, 6376. [Google Scholar] [CrossRef]
- Tanveer, M.; Shahzad, B.; Sharma, A.; Biju, S.; Bhardwaj, R. 24-Epibrassinolide; an active brassinolide and its role in salt stress tolerance in plants: A review. Plant Physiol. Biochem. 2018, 130, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.W.; Yang, N.; Li, Y.M. The Effect of Exogenous 24 Epibrassinolide on the Photosynthetic Characteristics of Tea Trees. J. Tea Sci. 2021, 41, 58–70. [Google Scholar] [CrossRef]
- Jin, Q.; Wang, Z.; Chen, Y.; Luo, Y.; Tian, N.; Liu, Z.; Huang, J.; Liu, S. Transcriptomics analysis reveals the signal transduction mechanism of brassinolides in tea leaves and its regulation on the growth and development of Camellia sinensis. BMC Genom. 2022, 23, 29. [Google Scholar] [CrossRef]
- Zhang, L.W.; Xie, L.J.; Yang, J.C.; Pai, N.C. Research Progress on the Response of Plant Secondary Metabolites to Abiotic Stress. For. Sci. Technol. 2024, 8, 1–6. [Google Scholar]
- Golam Jalal, A.; Youxin, Y. Anthocyanin-mediated arsenic tolerance in plants. Environ. Pollut. 2022, 292, 118475. [Google Scholar] [CrossRef]
- Clayton, W.A.; Albert, N.W.; Thrimawithana, A.H.; McGhie, T.K.; Deroles, S.C.; Schwinn, K.E.; Warren, B.A.; McLachlan, A.R.G.; Bowman, J.L.; Jordan, B.R.; et al. UVR8-mediated induction of flavonoid biosynthesis for UVB tolerance is conserved between the liverwort Marchantia polymorpha and flowering plants. Plant J. 2018, 96, 503–517. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Liu, H.; Luo, J.; Zhang, S.; Xiang, P.; Wang, W.; Cai, J.; Lu, Z.; Zhou, Z.; Hu, J.; et al. Partial root-zone simulated drought induces greater flavonoid accumulation than full root-zone simulated water deficiency in the leaves of Ginkgo biloba. Environ. Exp. Bot. 2022, 201, 104998. [Google Scholar] [CrossRef]
- Dong, N.-Q.; Sun, Y.; Guo, T.; Shi, C.-L.; Zhang, Y.-M.; Kan, Y.; Xiang, Y.-H.; Zhang, H.; Yang, Y.-B.; Li, Y.-C.; et al. UDP-glucosyltransferase regulates grain size and abiotic stress tolerance associated with metabolic flux redirection in rice. Nat. Commun. 2020, 11, 2629. [Google Scholar] [CrossRef]
- Chen, S.; Wu, F.; Li, Y.; Qian, Y.; Pan, X.; Li, F.; Wang, Y.; Wu, Z.; Fu, C.; Lin, H.; et al. NtMYB4 and NtCHS1 Are Critical Factors in the Regulation of Flavonoid Biosynthesis and Are Involved in Salinity Responsiveness. Front. Plant Sci. 2019, 10, 178. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, W.; Zhan, X.; Zhang, M.; Xiao, Y.; Hou, X.; Gao, M.; Xiao, B.; Gao, Y. CsCHLI plays an important role in chlorophyll biosynthesis of tea plant (Camellia sinensis). Beverage Plant Res. 2024, 4, e004. [Google Scholar] [CrossRef]
- Jia, C.; Zhao, S.; Bao, T.; Zhao, P.; Peng, K.; Guo, Q.; Gao, X.; Qin, J. Tomato BZR/BES transcription factor SlBZR1 positively regulates BR signaling and salt stress tolerance in tomato and Arabidopsis. Plant Sci. 2021, 302, 110719. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.X.; Gao, L.Y.; Zhang, R.; Zhao, T.; Zhang, Z.X.; Wang, S.C.; Wang, Y.X. The effect of 2,4-electrobrassinolide on photosynthesis and physiological characteristics of drooping crabapple under salt alkali stress. J. Fruit Sci. 2021, 38, 1479–1490. [Google Scholar] [CrossRef]
- Wu, C.X.; Hu, Z.H.; Lu, H.Y.; Ke, Y.Z.; Wang, Y.J.; Gong, Z.Y. The effect of Ebr on photosynthesis and antioxidant system of maize seedlings under salt stress. Jiangsu Agric. Sci. 2023, 51, 109–116. [Google Scholar] [CrossRef]
- Chen, Z.; Lin, S.; Li, J.; Chen, T.; Gu, Q.; Yang, T.; Zhang, Z. Theanine Improves Salt Stress Tolerance via Modulating Redox Homeostasis in Tea Plants (Camellia sinensis L.). Front. Plant Sci. 2021, 12, 770398. [Google Scholar] [CrossRef] [PubMed]
- Zong, X.F.; Yang, A.J.; Anjum, S.; Lv, J.; Li, N.J.; He, X.J.; Xu, Y.; Wu, X.; Wang, S.G. Effects of brassinolide application on antioxidantcharacteristics and endogenous hormones ofLeymus chinensis (Trin.) Tzvelev under differentlight intensity regimes. Chil. J. Agric. Res. 2018, 78, 539–548. [Google Scholar] [CrossRef]
- Zhou, Y.K. Advances in Brassinolide Regulation of Plant Growth and Development and Stress Resistance. Hans J. Agric. Sci. 2020, 10, 407–418. [Google Scholar] [CrossRef]
- Li, S.; Zhang, Y.; Yao, Q.; Bai, L.Q.; Hou, L.P.; Shi, Y. The effect of BR on tomato growth and osmotic regulation characteristics under isotonic salt stress. J. Northwest A F Univ. (Nat. Sci. Ed.) 2020, 48, 130–136, 145. [Google Scholar] [CrossRef]
- Li, X.B.; Li, Z.; Dai, S.J.; Pan, J.W.; Wang, Q.G.; Guan, Y.A.; Ding, G.H.; Liu, Y. Response of SiRLK35, a receptor protein kinase gene in foxtail millet, to salt stress in rice. Sci. Agric. Sin. 2019, 52, 3976. [Google Scholar] [CrossRef]
- Gu, M.; Wang, P.; Chen, X.; Zheng, Y.; Guo, Y.; Lin, X.; Gao, T.; Hou, B.; Ye, N. Identification of the Alcohol Dehydrogenase Gene Family in Tea Plants and Its Expression Analysis during the Withering Process of White Tea. J. Tea Sci. 2021, 41, 302. [Google Scholar] [CrossRef]
- Li, H.S. Principles and Techniques of Plant Physiology and Biochemistry Experiments; Higher Education Press: Beijing, China, 2000. [Google Scholar]
- Xiong, Q.E. Plant Physiology Laboratory Course; Sichuan Science and Technology Press: Chengdu, China, 2003. [Google Scholar]
- Wang, X.K. Principles and Techniques of Plant Physiology and Biochemistry Experiments; Higher Education Press: Beijing, China, 2006. [Google Scholar]
- Chen, C.; Wu, Y.; Li, J.; Wang, X.; Zeng, Z.; Xu, J.; Liu, Y.; Feng, J.; Chen, H.; He, Y.; et al. TBtools-II: A “one for all, all for one” bioinformatics platform for biological big-data mining. Mol. Plant 2023, 16, 1733–1742. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data using Real-Time Quantitative PCR. Methods 2002, 4, 402–408. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Ma, X.; Tang, D.; Chen, Y.; Chen, G.; Zou, J.; Tan, L.; Tang, Q.; Chen, W. Effects of Brassinosteroid on the Physiological Changes on Two Varieties of Tea Plants Under Salt Stress. Int. J. Mol. Sci. 2024, 25, 13445. https://doi.org/10.3390/ijms252413445
Zhang Z, Ma X, Tang D, Chen Y, Chen G, Zou J, Tan L, Tang Q, Chen W. Effects of Brassinosteroid on the Physiological Changes on Two Varieties of Tea Plants Under Salt Stress. International Journal of Molecular Sciences. 2024; 25(24):13445. https://doi.org/10.3390/ijms252413445
Chicago/Turabian StyleZhang, Zhuolu, Xiru Ma, Dandan Tang, Yiduo Chen, Guo Chen, Juanfen Zou, Liqiang Tan, Qian Tang, and Wei Chen. 2024. "Effects of Brassinosteroid on the Physiological Changes on Two Varieties of Tea Plants Under Salt Stress" International Journal of Molecular Sciences 25, no. 24: 13445. https://doi.org/10.3390/ijms252413445
APA StyleZhang, Z., Ma, X., Tang, D., Chen, Y., Chen, G., Zou, J., Tan, L., Tang, Q., & Chen, W. (2024). Effects of Brassinosteroid on the Physiological Changes on Two Varieties of Tea Plants Under Salt Stress. International Journal of Molecular Sciences, 25(24), 13445. https://doi.org/10.3390/ijms252413445






