p21 as a Predictor and Prognostic Indicator of Clinical Outcome in Rectal Cancer Patients
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics of Entire Cohort
2.2. Patient Characteristics of Subcohort Who Received Neoadjuvant Therapy
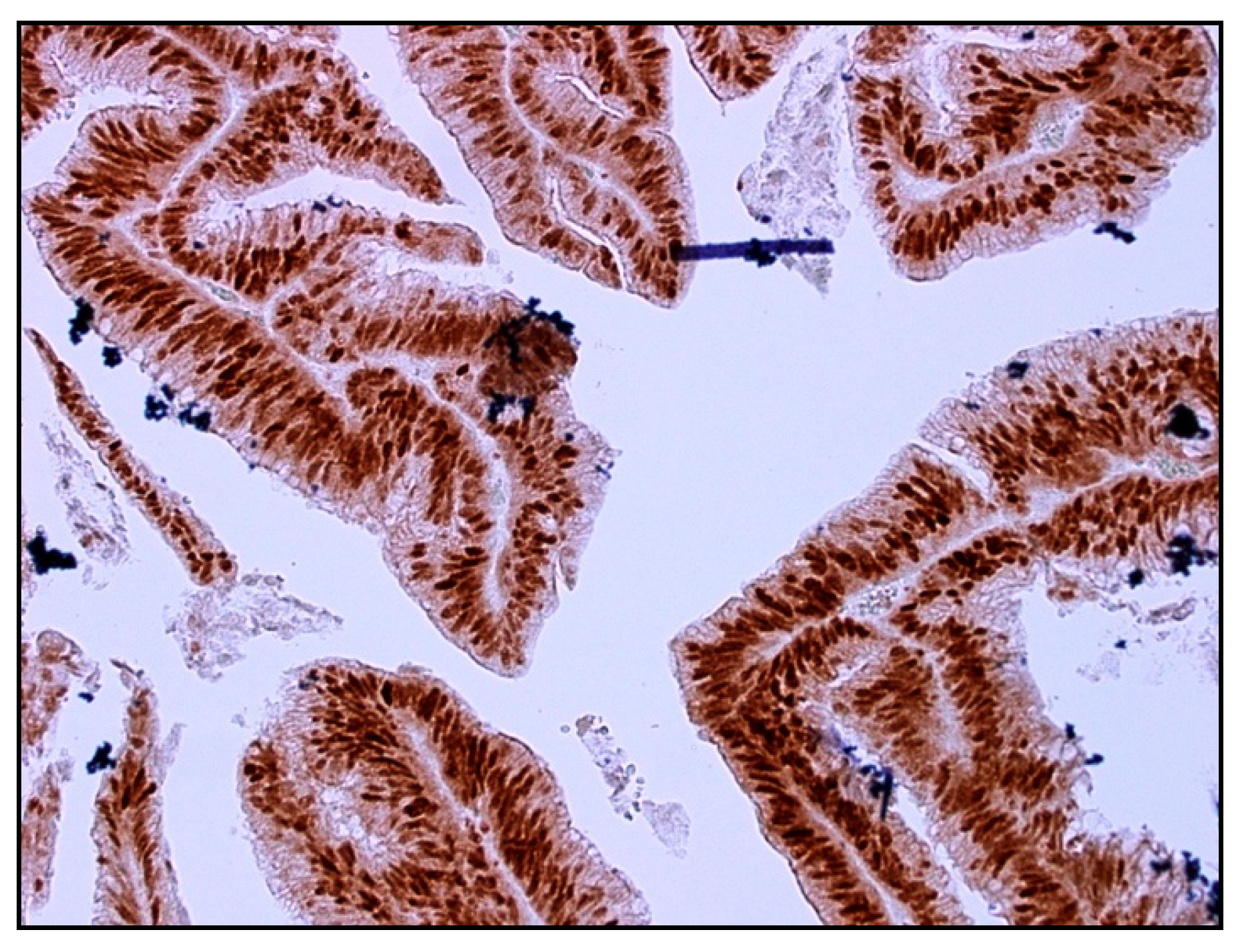
2.3. p21 Expression
2.4. Association of p21 Expression with Clinicopathological Variables
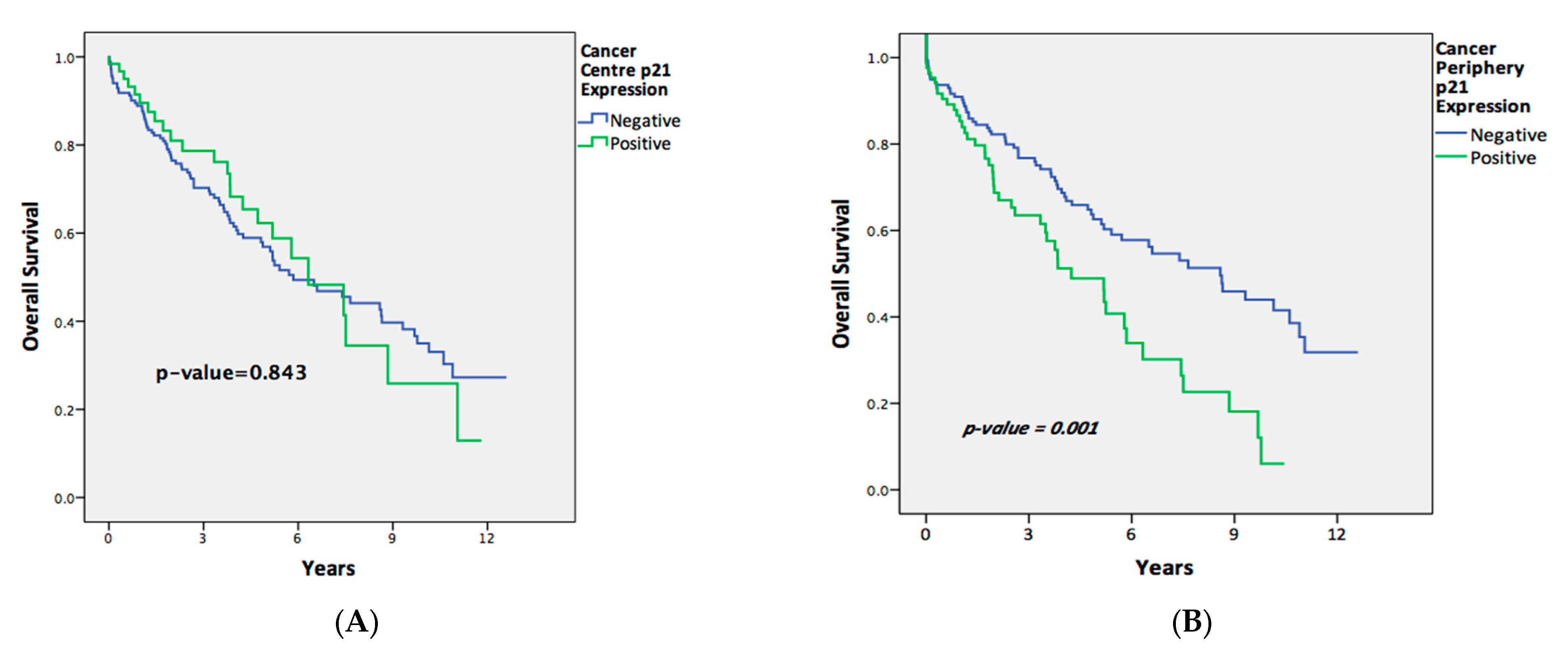
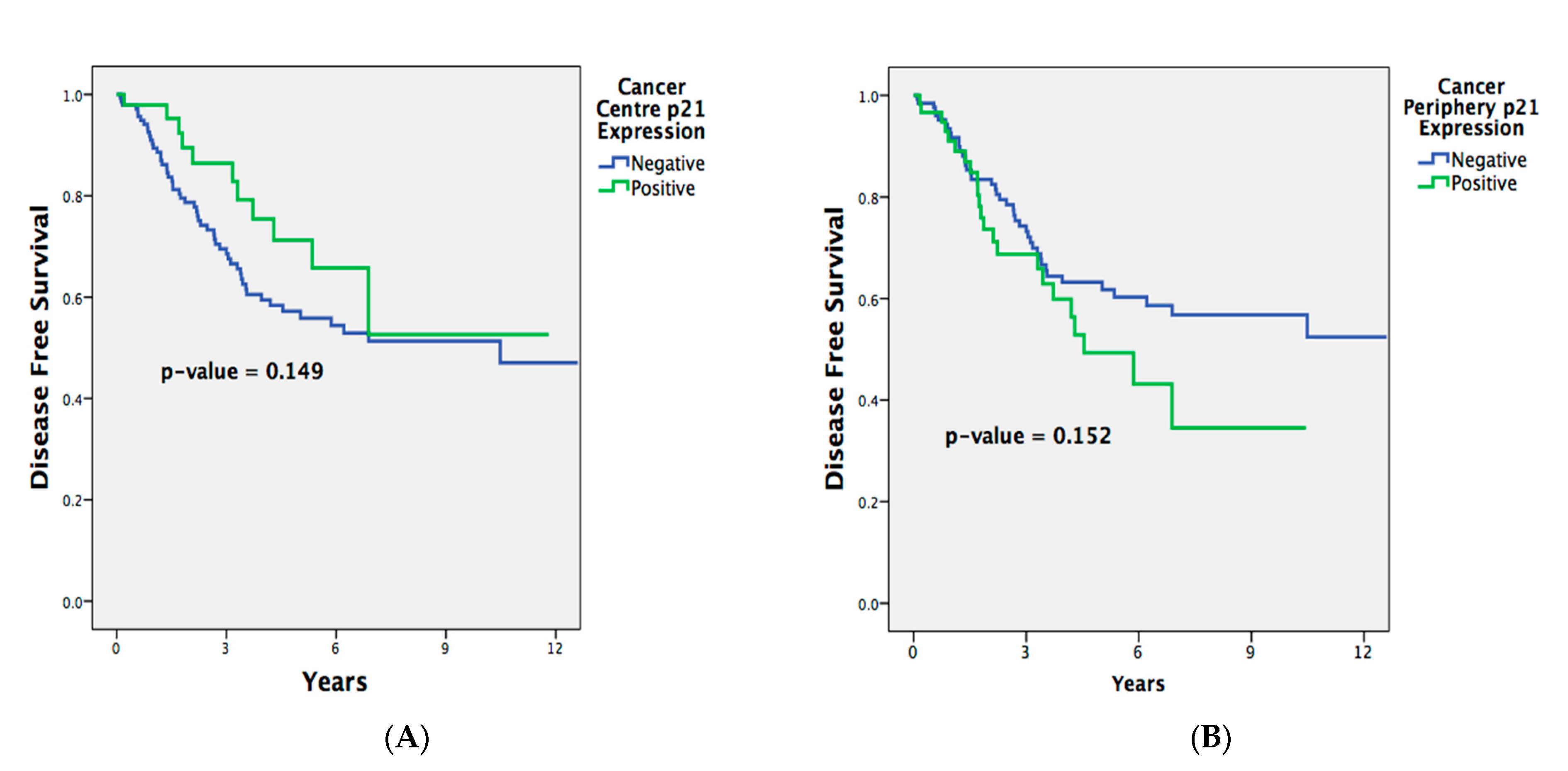
2.5. Association of p21 Expression with Disease-Free Survival and Overall Survival in Entire Rectal Cancer Cohort
2.6. Association of P21 Expression with Neoadjuvant Treatment and Response
3. Discussion
4. Materials and Methods
4.1. Patient Samples
4.2. Tissue Microarrays
4.3. Immunohistochemistry
4.4. Immunohistochemical Scoring
4.5. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Australian Institute of Health and Welfare 2016. Australia’s Health 2016; Australia’s Health Series No. 15. Cat. No. AUS 199; AIHW: Canberra, Australia, 2016.
- Tarantino, I.; Muller, S.A.; Warschkow, R.; Kulu, Y.; Schmied, B.M.; Buchler, M.W.; Ulrich, A. Baseline mortality-adjusted survival in resected rectal cancer patients. J. Gastrointest. Surg. 2014, 18, 1837–1844. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Abbas, T.; Dutta, A. DNA replication and genomic instability. Adv. Exp. Med. Biol. 2005, 570, 249–279. [Google Scholar] [PubMed]
- Xiong, Y.; Hannon, G.J.; Zhang, H.; Casso, D.; Kobayashi, R.; Beach, D. p21 is a universal inhibitor of cyclin kinases. Nature 1993, 366, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Kreis, N.N.; Louwen, F.; Yuan, J. Less understood issues: p21(Cip1) in mitosis and its therapeutic potential. Oncogene 2015, 34, 1758–1767. [Google Scholar] [CrossRef]
- Schafer, K.A. The cell cycle: A review. Vet. Pathol. 1998, 35, 461–478. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, K.; Van Bockstaele, D.R.; Berneman, Z.N. The cell cycle: A review of regulation, deregulation and therapeutic targets in cancer. Cell Prolif. 2003, 36, 131–149. [Google Scholar] [CrossRef] [PubMed]
- Abbas, T.; Dutta, A. p21 in cancer: Intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.A.; Rahman, M.; Ayad, A.A.; Warrington, A.E.; Burns, T.C. P21 Overexpression Promotes Cell Death and Induces Senescence in Human Glioblastoma. Cancers 2023, 15, 1279. [Google Scholar] [CrossRef]
- Moldovan, G.L.; Pfander, B.; Jentsch, S. PCNA, the maestro of the replication fork. Cell 2007, 129, 665–679. [Google Scholar] [CrossRef]
- Zhou, B.P.; Liao, Y.; Xia, W.; Spohn, B.; Lee, M.H.; Hung, M.C. Cytoplasmic localization of p21Cip1/WAF1 by Akt-induced phosphorylation in HER-2/neu-overexpressing cells. Nat. Cell. Biol. 2001, 3, 245–252. [Google Scholar] [CrossRef]
- Winters, Z.E.; Leek, R.D.; Bradburn, M.J.; Norbury, C.J.; Harris, A.L. Cytoplasmic p21WAF1/CIP1 expression is correlated with HER-2/neu in breast cancer and is an independent predictor of prognosis. Breast Cancer Res. 2003, 5, R242–R249. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Chen, J.S.; Zhou, X.; Sun, P.R.; Lee, D.F.; Liao, Y.; Zhou, B.P.; Hung, M.C. Phosphorylation/cytoplasmic localization of p21Cip1/WAF1 is associated with HER2/neu overexpression and provides a novel combination predictor for poor prognosis in breast cancer patients. Clin. Cancer Res. 2004, 10, 3815–3824. [Google Scholar] [CrossRef]
- Cheung, T.H.; Lo, K.W.; Yu, M.M.; Yim, S.F.; Poon, C.S.; Chung, T.K.; Wong, Y.F. Aberrant expression of p21(WAF1/CIP1) and p27(KIP1) in cervical carcinoma. Cancer Lett. 2001, 172, 93–98. [Google Scholar] [CrossRef]
- Baretton, G.B.; Klenk, U.; Diebold, J.; Schmeller, N.; Lohrs, U. Proliferation- and apoptosis-associated factors in advanced prostatic carcinomas before and after androgen deprivation therapy: Prognostic significance of p21/WAF1/CIP1 expression. Br. J. Cancer 1999, 80, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Bae, D.S.; Cho, S.B.; Kim, Y.J.; Whang, J.D.; Song, S.Y.; Park, C.S.; Kim, D.S.; Lee, J.H. Aberrant expression of cyclin D1 is associated with poor prognosis in early stage cervical cancer of the uterus. Gynecol. Oncol. 2001, 81, 341–347. [Google Scholar] [CrossRef]
- Sulzyc-Bielicka, V.; Domagala, P.; Bielicki, D.; Safranow, K.; Domagala, W. Thymidylate synthase expression and p21(WAF1)/p53 phenotype of colon cancers identify patients who may benefit from 5-fluorouracil based therapy. Cell. Oncol. 2014, 37, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Sulzyc-Bielicka, V.; Domagala, P.; Urasinska, E.; Bielicki, D.; Safranow, K.; Domagala, W. Expression of p21WAF1 in Astler-Coller stage B2 colorectal cancer is associated with survival benefit from 5FU-based adjuvant chemotherapy. Virchows Arch. 2011, 458, 431–438. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bukholm, I.K.; Nesland, J.M. Protein expression of p53, p21 (WAF1/CIP1), bcl-2, Bax, cyclin D1 and pRb in human colon carcinomas. Virchows Arch. 2000, 436, 224–228. [Google Scholar] [CrossRef]
- Zirbes, T.K.; Baldus, S.E.; Moenig, S.P.; Nolden, S.; Kunze, D.; Shafizadeh, S.T.; Schneider, P.M.; Thiele, J.; Hoelscher, A.H.; Dienes, H.P. Prognostic impact of p21/waf1/cip1 in colorectal cancer. Int. J. Cancer 2000, 89, 14–18. [Google Scholar] [CrossRef]
- Suzuki, T.; Sadahiro, S.; Fukasawa, M.; Ishikawa, K.; Kamijo, A.; Yasuda, S.; Makuchi, H.; Ohizumi, Y.; Murayama, C. Predictive factors of tumor shrinkage and histological regression in patients who received preoperative radiotherapy for rectal cancer. JPN J. Clin. Oncol. 2004, 34, 740–746. [Google Scholar] [CrossRef]
- Charara, M.; Edmonston, T.B.; Burkholder, S.; Walters, R.; Anne, P.; Michell, E.; Fry, R.; Boman, B.; Rose, D.; Fishel, R.; et al. Microsatellite status and cell cycle associated markers in rectal cancer patients undergoing a combined regimen of 5-FU and CPT-11 chemotherapy and radiotherapy. Anticancer. Res. 2004, 24, 3161–3167. [Google Scholar]
- Dotto, G.P. p21(WAF1/Cip1): More than a break to the cell cycle? Biochim. Biophys. Acta. 2000, 1471, M43–M56. [Google Scholar] [PubMed]
- Roninson, I.B. Oncogenic functions of tumour suppressor p21(Waf1/Cip1/Sdi1): Association with cell senescence and tumour-promoting activities of stromal fibroblasts. Cancer Lett. 2002, 179, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Noske, A.; Lipka, S.; Budczies, J.; Muller, K.; Loddenkemper, C.; Buhr, H.J.; Kruschewski, M. Combination of p53 expression and p21 loss has an independent prognostic impact on sporadic colorectal cancer. Oncol. Rep. 2009, 22, 3–9. [Google Scholar] [PubMed]
- Sturm, I.; Rau, B.; Schlag, P.M.; Wust, P.; Hildebrandt, B.; Riess, H.; Hauptmann, S.; Dorken, B.; Daniel, P.T. Genetic dissection of apoptosis and cell cycle control in response of colorectal cancer treated with preoperative radiochemotherapy. BMC Cancer 2006, 6, 124. [Google Scholar] [CrossRef] [PubMed]
- Rau, B.; Sturm, I.; Lage, H.; Berger, S.; Scheider, U.; Hauptmann, S.; Wust, P.; Riess, H.; Schlag, P.M.; Dorken, B.; et al. Dynamic expression profile of p21WAF1/CIP1 and Ki-67 predicts survival in rectal carcinoma treated with preoperative radiochemotherapy. J. Clin. Oncol. 2003, 21, 3391–3401. [Google Scholar] [CrossRef] [PubMed]
- Kreis, N.N.; Louwen, F.; Zimmer, B.; Yuan, J. Loss of p21Cip1/CDKN1A renders cancer cells susceptible to Polo-like kinase 1 inhibition. Oncotarget 2015, 6, 6611–6626. [Google Scholar] [CrossRef] [PubMed]
- Moore, H.G.; Shia, J.; Klimstra, D.S.; Ruo, Y.; Mazumdar, M.; Schwartz, G.K.; Minsky, B.D.; Saltz, L.; Guillem, J.G. Expression of p27 in residual rectal cancer after preoperative chemoradiation predicts long-term outcome. Ann Surg Oncol. 2004, 11, 955–961. [Google Scholar] [CrossRef] [PubMed]
- Waldman, T.; Lengauer, C.; Kinzler, K.W.; Vogelstein, B. Uncoupling of S phase and mitosis induced by anticancer agents in cells lacking p21. Nature 1996, 381, 713–716. [Google Scholar] [CrossRef]
- Wouters, B.G.; Giaccia, A.J.; Denko, N.C.; Brown, J.M. Loss of p21Waf1/Cip1 sensitizes tumors to radiation by an apoptosis-independent mechanism. Cancer Res. 1997, 57, 4703–4706. [Google Scholar]
- Doglioni, C.; Pelosio, P.; Laurino, L.; Macri, E.; Meggiolaro, E.; Favretti, F.; Barbareschi, M. p21/WAF1/CIP1 expression in normal mucosa and in adenomas and adenocarcinomas of the colon: Its relationship with differentiation. J. Pathol. 1996, 179, 248–253. [Google Scholar] [CrossRef]
- Polyak, K.; Hamilton, S.R.; Vogelstein, B.; Kinzler, K.W. Early alteration of cell-cycle-regulated gene expression in colorectal neoplasia. Am. J. Pathol. 1996, 149, 381–387. [Google Scholar] [PubMed]
- Lee, C.S. The significance of autophagy in colorectal cancer pathogenesis and implications for therapy. J. Clin. Pathol. 2014, 67, 854. [Google Scholar]
- Ogino, S.; Nosho, K.; Shima, K.; Baba, Y.; Irahara, N.; Kirkner, G.J.; Hazra, A.; De Vivo, I.; Giovannucci, E.L.; Meyerhardt, J.A.; et al. p21 expression in colon cancer and modifying effects of patient age and body mass index on prognosis. Cancer Epidemiol. Biomark. Prev. 2009, 18, 2513–2521. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.F.; Sharpless, N.E. Telomeres, p21 and the cancer-aging hypothesis. Nat. Genet. 2007, 39, 11–12. [Google Scholar] [CrossRef] [PubMed]
- Shoji, M.; Dobashi, Y.; Morinaga, S.; Jiang, S.X.; Kameya, T. Tumor extension and cell proliferation in adenocarcinomas of the lung. Am. J. Pathol. 1999, 154, 909–918. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dobashi, Y.; Shoji, M.; Jiang, S.X.; Kobayashi, M.; Kawakubo, Y.; Kameya, T. Active cyclin A-CDK2 complex, a possible critical factor for cell proliferation in human primary lung carcinomas. Am. J. Pathol. 1998, 153, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Thoma, O.M.; Neurath, M.F.; Waldner, M.J. Cyclin-Dependent Kinase Inhibitors and Their Therapeutic Potential in Colorectal Cancer Treatment. Front. Pharmacol. 2021, 12, 757120. [Google Scholar] [CrossRef]
- Coqueret, O. New roles for p21 and p27 cell-cycle inhibitors: A function for each cell compartment? Trends Cell Biol. 2003, 13, 65–70. [Google Scholar] [CrossRef]
- Wendt, J.; Radetzki, S.; von Haefen, C.; Hemmati, P.G.; Guner, D.; Schulze-Osthoff, K.; Dorken, B.; Daniel, P.T. Induction of p21CIP/WAF-1 and G2 arrest by ionizing irradiation impedes caspase-3-mediated apoptosis in human carcinoma cells. Oncogene 2006, 25, 972–980. [Google Scholar] [CrossRef]
- Teyssier, F.; Bay, J.O.; Dionet, C.; Verrelle, P. Cell cycle regulation after exposure to ionizing radiation. Bull Cancer 1999, 86, 345–357. [Google Scholar]
- Reerink, O.; Karrenbeld, A.; Plukker, J.T.; Verschueren, R.C.J.; Szabo, B.G.; Sluiter, W.J.; Hospers, G.A.P.; Mulder, N.H. Molecular prognostic factors in locally irresectable rectal cancer treated preoperatively by chemo-radiotherapy. Anticancer. Res. 2004, 24, 1217–1221. [Google Scholar] [PubMed]
- Sim, S.H.; Kang, M.H.; Kim, Y.J.; Lee, K.W.; Kim, D.W.; Kang, S.B.; Eom, K.Y.; Kim, J.S.; Lee, H.S.; Kim, J.H. P21 and CD166 as predictive markers of poor response and outcome after fluorouracil-based chemoradiotherapy for the patients with rectal cancer. BMC Cancer 2014, 14, 241. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Sadahiro, S.; Tanaka, A.; Okada, K.; Saito, G.; Kamijo, A.; Akiba, T.; Kawada, S. Predictive markers of chemoradiotherapy for rectal cancer: Comparison of biopsy specimens taken before and about 1 week after the start of chemoradiotherapy. Int. J. Clin. Oncol. 2015, 20, 1130–1139. [Google Scholar] [CrossRef] [PubMed]
- Schwandner, O.; Bruch, H.P.; Broll, R. p21, p27, cyclin D1, and p53 in rectal cancer: Immunohistology with prognostic significance? Int. J. Colorectal. Dis. 2002, 17, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.C.; Lee, H.H.; Hwang, W.S.; Li, C.F.; Huang, C.T.; Que, J.; Lin, K.L.; Lin, F.C.; Lu, C.L. p53 and p27 as predictors of clinical outcome for rectal-cancer patients receiving neoadjuvant therapy. Surg. Oncol. 2006, 15, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Leibold, T.; Hui, V.W.; Shia, J.; Ruby, J.A.; Riedel, E.R.; Guillem, J.G. p27 expression in post-treatment rectal cancer: A potential novel approach for predicting residual nodal disease. Am. J. Surg. 2014, 208, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Helfman, D.M. Cytoplasmic p21Cip1 is involved in Ras-induced inhibition of the ROCK/LIMK/cofilin pathway. J. Biol. Chem. 2004, 279, 1885–1891. [Google Scholar] [CrossRef]
- Gartel, A.L.; Tyner, A.L. The role of the cyclin-dependent kinase inhibitor p21 in apoptosis. Mol. Cancer Ther. 2002, 1, 639–649. [Google Scholar]
- Shen, H.; Ding, J.; Ji, J.; Jiang, B.; Wang, X.; Yang, P. Overcoming MTDH and MTDH-SND1 complex: Driver and potential therapeutic target of cancer. Cancer Insight 2024, 3, 29. [Google Scholar] [CrossRef]
- Galanos, P.; Vougas, K.; Walter, D.; Polyzos, A.; Maya-Mendoza, A.; Haagensen, E.J.; Kokkalis, A.; Roumelioti, F.M.; Gagos, S.; Tzetis, M.; et al. p53-independent p21 expression causes genomic instability by deregulating replication licensing. Nat. Cell Biol. 2016, 18, 777–789. [Google Scholar] [CrossRef]
- Kreis, N.N.; Louwen, F.; Yuan, J. The Multifaceted p21 (Cip1/Waf1/CDKN1A) in Cell Differentiation, Migration and Cancer Therapy. Cancers 2019, 11, 1220. [Google Scholar] [CrossRef] [PubMed]
- Mokou, M.; Mischak, H.; Frantzi, M. Statistical determination of cancer biomarkers: Moving forward clinically. Expert Rev. Mol. Diagn. 2023, 23, 187–189. [Google Scholar] [CrossRef] [PubMed]
- Vickerstaff, V.; Omar, R.Z.; Ambler, G. Methods to adjust for multiple comparisons in the analysis and sample size calculation of randomised controlled trials with multiple primary outcomes. BMC Med. Res. Methodol. 2019, 19, 129. [Google Scholar]
- Yang, K.; Yu, G.; Yang, Z.; Yue, L.; Zhang, X.; Sun, C.; Wei, J.; Rao, L.; Chen, X.; Wang, R. Supramolecular Polymerization-Induced Nanoassemblies for Self-Augmented Cascade Chemotherapy and Chemodynamic Therapy of Tumor. Angew. Chem. Int. Ed. Engl. 2021, 60, 17570–17578. [Google Scholar] [CrossRef] [PubMed]
- Edge, S.B.; Compton, C.C. The American Joint Committee on Cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Ann. Surg. Oncol. 2010, 17, 1471–1474. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, P.C.; Penny, S.A.; Dolan, R.T.; Kelly, C.M.; Madden, S.F.; Rexhepaj, E.; Brennan, D.J.; McCann, A.H.; Ponten, F.; Uhlen, M.; et al. Systematic antibody generation and validation via tissue microarray technology leading to identification of a novel protein prognostic panel in breast cancer. BMC Cancer 2013, 13, 175. [Google Scholar]
- Hoos, A.; Urist, M.J.; Stojadinovic, A.; Mastorides, S.; Dudas, M.E.; Leung, D.H.Y.; Kuo, D.; Brennan, M.F.; Lewis, J.J.; Cordon-Cardo, C. Validation of tissue microarrays for immunohistochemical profiling of cancer specimens using the example of human fibroblastic tumors. Am. J. Pathol. 2001, 158, 1245–1251. [Google Scholar] [CrossRef]
- Kim, S.W.; Roh, J.; Park, C.S. Immunohistochemistry for Pathologists: Protocols, Pitfalls, and Tips. J. Pathol. Transl. Med. 2016, 50, 411–418. [Google Scholar] [CrossRef]
| All Patients | |
|---|---|
| Total, n | 266 |
| Sex, n (%) | |
| Male | 176 (66.2) |
| Female | 90 (33.8) |
| Mean age, yrs | 70.9 |
| pT category, n (%) | |
| T1–2 | 87/260 (33.5) |
| T3–4 | 173/260 (66.5) |
| pN category, n (%) | |
| N0 | 139/259 (53.7) |
| N1–2 | 120/259 (46.3) |
| pM category, n (%) | |
| M0 | 223/240 (92.9) |
| M1 | 17/240 (7.1) |
| Grade, n (%) | |
| 1–2 | 246/266 (92.5) |
| 3 | 20/266 (7.5) |
| Vascular invasion, | |
| n (%) | |
| Absent | 201/263 (76.4) |
| Present | 62/263 (23.6) |
| Perineural invasion, | |
| n (%) | |
| Absent | 220/263 (83.7) |
| Present | 43/263 (16.3) |
| Neoadjuvant Treatment, n (%) | |
| Negative | 191/245 (78) |
| Positive | 54/245 (22) |
| Adjuvant Treatment, n (%) | |
| Negative | 153/221 (69.2) |
| Positive | 68/221 (30.8) |
| Recurrence Status | |
| Recurred | 83/218 (38.1) |
| Did not recur | 135/218 (61.9) |
| Death Status | |
| Alive | 142/251 (56.6) |
| Dead | 109/251 (43.4) |
| Median time to recurrence (years) | 2.78 |
| Median time to death (years) | 3.19 |
| Patients Who Received Neoadjuvant Therapy | |
|---|---|
| Total, n | 54 |
| Sex, n (%) | |
| Male | 37/54 (68.5) |
| Female | 17/54 (31.5) |
| Mean age, yrs | 66.41 |
| pT category, n (%) | |
| T1–2 | 16/54 (29.6) |
| T3–4 | 38/54 (70.4) |
| pN category, n (%) | |
| N0 | 28/54 (51.9) |
| N1–2 | 26/54 (48.1) |
| pM category, n (%) | |
| M0 | 52/53 (98.1) |
| M1 | 1/53 (1.9) |
| Grade, n (%) | |
| 1–2 | 50/54 (92.6) |
| 3 | 4/54 (7.4) |
| Vascular invasion, n (%) | |
| Absent | 46/54 (85.2) |
| Present | 8/54 (14.8) |
| Perineural invasion, n (%) | |
| Absent | 40/54 (74.1) |
| Present | 14/54 (25.9) |
| Adjuvant Treatment, n (%) | |
| Negative | 21/44 (47.7) |
| Positive | 23/44 (52.3) |
| Tumor Regression Grade, n (%) | |
| 0–1(Good response) | 6/51 (11.8) |
| 2–3 (Poor response) | 45/51 (88.2) |
| Recurrence Status | |
| Recurred | 24/52 (46.2) |
| Did not recur | 28/52 (53.8) |
| Death Status | |
| Alive | 32/54 (59.3) |
| Dead | 22/54 (40.7) |
| Median time to recurrence (years) | 2.25 |
| Median time to death (years) | 3.08 |
| Tissue Type | p21 Expression | Total | |
|---|---|---|---|
| Positive | Negative | ||
| Tumor Center (TC), n (%) | 66/265 (24.9) | 199/265 (75.1) | 265 |
| Tumor Periphery (TP), n (%) | 92/263 (35) | 171/263 (65) | 263 |
| Normal Tissue Close to Tumor (NCT), n (%) | 156/248 (62.9) | 92/248 (37.1) | 248 |
| Normal Tissue Away from Tumor (NAT), n (%) | 193/256 (75.4) | 63/256 (24.6) | 256 |
| Lymph nodes with metastases, n (%) | 21/115 (18.3) | 94/115 (81.7) | 115 |
| Adenoma, n (%) | 46/58 (79.3) | 12/58 (20.7) | 58 |
| Hyperplastic polyp, n (%) | 40/56 (71.4) | 16/56 (28.6) | 56 |
| All Tissue Types | 614/1261 (48.7) | 647/1261 (51.3) | 1261 |
| Tumor Center (TC) | p | Tumor Periphery (TP) | p | |||
|---|---|---|---|---|---|---|
| Negative | Positive | Negative | Positive | |||
| n (%) | n (%) | n (%) | n (%) | |||
| Sex | ||||||
| Male | 133 (75.6) | 43 (24.4) | 0.802 | 119 (68.4) | 55 (31.6) | 0.109 |
| Female | 66 (74.2) | 23 (25.8) | 52 (58.4) | 37 (41.6) | ||
| Age | ||||||
| ≤70 yrs | 88 (71.5) | 35 (28.5) | 0.214 | 80 (66.1) | 41 (33.9) | 0.731 |
| >70 yrs | 111 (78.2) | 31 (21.8) | 91 (64.1) | 51 (35.9) | ||
| pT category | ||||||
| T1–2 | 61 (70.1) | 26 (29.9) | 0.138 | 58 (67.4) | 28 (32.6) | 0.620 |
| T3–4 | 135 (78.5) | 37 (21.5) | 110 (64.3) | 61 (35.7) | ||
| pN category | ||||||
| N0 | 98 (71) | 40 (29) | 0.046 | 90 (65.2) | 48 (34.8) | 0.995 |
| N1–3 | 98 (81.7) | 22 (18.3) | 77 (65.3) | 41 (34.7) | ||
| pM category | ||||||
| M0 | 165 (74.3) | 57 (25.7) | 0.200 | 148 (67.3) | 72 (32.7) | 0.476 |
| M1 | 15 (88.2) | 2 (11.8) | 10 (58.8) | 7 (41.2) | ||
| Grade | ||||||
| 1–2 | 184 (75.1) | 61 (24.9) | 0.992 | 157 (64.6) | 86 (35.4) | 0.627 |
| 3 | 15 (75) | 5 (25) | 14 (70) | 6 (30) | ||
| Vascular invasion | ||||||
| Absent | 146 (73) | 54 (27) | 0.082 | 132 (66.7) | 66 (33.3) | 0.437 |
| Present | 52 (83.9) | 10 (16.1) | 38 (61.3) | 24 (38.7) | ||
| Perineural invasion | ||||||
| Absent | 166 (75.8) | 53 (24.2) | 0.847 | 144 (66.4) | 73 (33.6) | 0.458 |
| Present | 32 (74.4) | 11 (25.6) | 26 (60.5) | 17 (39.5) | ||
| Recurrence Status | ||||||
| Did not recur | 94 (69.6) | 41 (30.4) | 0.029 | 92 (69.7) | 40 (30.3) | 0.478 |
| Recur | 68 (82.9) | 14 (17.1) | 54 (65.1) | 29 (34.9) | ||
| Overall Survival | Disease-Free Survival | |||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| p21, tumor center (TC) | ||||
| Positive vs. negative | 0.955 (0.606–1.506) | 0.843 | 0.623 (0.325–1.193) | 0.153 |
| p21, tumor periphery (TP) | ||||
| Positive vs. negative | 1.946 (1.313–2.885) | 0.001 | 1.450 (0.870–2.419) | 0.154 |
| Sex | ||||
| Male vs. female | 1.057 (0.711–1.572) | 0.784 | 1.098 (0.658–1.832) | 0.720 |
| Age | ||||
| <70 years vs. ≥70 years | 1.382 (0.930–2.052) | 0.109 | 0.740 (0.456–1.201) | 0.223 |
| pT category | ||||
| T3–4 vs. T1–2 | 1.756 (1.133–2.720) | 0.012 | 2.687 (1.463–4.935) | 0.001 |
| pN category | ||||
| N1–2 vs. N0 | 1.441 (0.980–2.120) | 0.064 | 1.302 (0.803–2.117) | 0.283 |
| pM category | ||||
| M1 vs. M0 | 5.096 (2.700–9.618) | <0.0001 | - | - |
| Grade | ||||
| 3–4 vs. 1–2 | 1.525 (0.817–2.849) | 0.185 | 2.256 (1.030–4.942) | 0.042 |
| Vascular invasion | ||||
| Presence vs. absence | 1.968 (1.301–2.979) | 0.001 | 1.783 (1.015–3.133) | 0.044 |
| Perineural invasion | ||||
| Presence vs. absence | 2.409 (1.550–3.746) | <0.0001 | 3.342 (1.930–5.785) | <0.0001 |
| Neoadjuvant treatment | ||||
| Yes vs. no | 0.944 (0.589–1.512) | 0.810 | 1.815 (1.084–3.039) | 0.023 |
| Adjuvant treatment | ||||
| Yes vs. no | 0.506 (0.301–0.850) | 0.010 | 1.124 (0.661–1.910) | 0.666 |
| Overall Survival Period | ||
|---|---|---|
| HR (95% CI) | p | |
| p21, tumor periphery (TP) | ||
| Positive vs. negative | 2.068 (1.290–3.316) | 0.003 |
| pT category | ||
| T3–4 vs. T1–2 | 1.366 (0.816–2.286) | 0.235 |
| pM category | ||
| M1 vs. M0 | 3.444 (1.422–8.342) | 0.006 |
| Vascular invasion | ||
| Presence vs. absence | 1.687 (0.950–2.997) | 0.074 |
| Perineural invasion | ||
| Presence vs. absence | 2.184 (1.194–3.996) | 0.011 |
| Adjuvant treatment | ||
| Yes vs. no | 0.346 (0.196–0.613) | <0.0001 |
| Overall Survival | ||
| HR (95% CI) | p | |
| p21, tumor center (TC) | ||
| Positive vs. negative | 0.888 (0.503–1.566) | 0.682 |
| pT category | ||
| T3–4 vs. T1–2 | 1.576 (0.945–2.628) | 0.081 |
| pM category | ||
| M1 vs. M0 | 3.617 (1.517–8.621) | 0.004 |
| Vascular invasion | ||
| Presence vs. absence | 1.752 (0.999–3.072) | 0.050 |
| Perineural invasion | ||
| Presence vs. absence | 1.951 (1.075–3.541) | 0.028 |
| Adjuvant treatment | ||
| Yes vs. no | 0.332 (0.186–0.590) | <0.0001 |
| Disease-Free Survival | ||
| HR (95% CI) | p | |
| p21, tumor periphery (TP) | ||
| Positive vs. negative | 1.769 (1.027–3.049) | 0.040 |
| pT category | ||
| T3–4 vs. T1–2 | 1.841 (0.970–3.494) | 0.062 |
| Grade | ||
| 3–4 vs. 1–2 | 1.952 (0.819–4.648) | 0.131 |
| Vascular invasion | ||
| Presence vs. absence | 1.370 (0.713–2.633) | 0.344 |
| Perineural invasion | ||
| Presence vs. absence | 2.465 (1.312–4.631) | 0.005 |
| Neoadjuvant treatment | ||
| Yes vs. no | 1.816 (1.034–3.191) | 0.038 |
| Disease-Free Survival | ||
| HR (95% CI) | p | |
| p21, tumor center (TC) | ||
| Positive vs. negative | 1.422 (0.737–2.745) | 0.294 |
| pT category | ||
| T3–4 vs. T1–2 | 2.039 (1.079–3.853) | 0.028 |
| Grade | ||
| 3–4 vs. 1–2 | 1.727 (0.735–4.060) | 0.210 |
| Vascular invasion | ||
| Presence vs. absence | 1.369 (0.714–2.626) | 0.344 |
| Perineural invasion | ||
| Presence vs. absence | 2.182 (1.188–4.006) | 0.012 |
| Neoadjuvant treatment | ||
| Yes vs. no | 1.875 (1.062–3.311) | 0.030 |
| Neoadjuvant Therapy | Tumor Center | p | Tumor Periphery | p | ||
|---|---|---|---|---|---|---|
| Negative, n (%) | Positive, n (%) | Negative, n (%) | Positive, n (%) | |||
| Yes | 40 (74.1) | 14 (25.9) | 0.954 | 36 (67.9) | 17 (32.1) | 0.551 |
| No | 140 (73.7) | 50 (26.3) | 120 (63.5) | 69 (36.5) | ||
| Grade | Description |
|---|---|
| 0 | No viable cancer cells |
| 1 | Moderate response Single or small groups of tumor cells |
| 2 | Minimal response Residual cancer outgrown by fibrosis |
| 3 | Minimal or no tumor cells killed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ooi, L.C.; Ho, V.; Zhu, J.Z.; Lim, S.; Chung, L.; Abubakar, A.; Rutland, T.; Chua, W.; Ng, W.; Lee, M.; et al. p21 as a Predictor and Prognostic Indicator of Clinical Outcome in Rectal Cancer Patients. Int. J. Mol. Sci. 2024, 25, 725. https://doi.org/10.3390/ijms25020725
Ooi LC, Ho V, Zhu JZ, Lim S, Chung L, Abubakar A, Rutland T, Chua W, Ng W, Lee M, et al. p21 as a Predictor and Prognostic Indicator of Clinical Outcome in Rectal Cancer Patients. International Journal of Molecular Sciences. 2024; 25(2):725. https://doi.org/10.3390/ijms25020725
Chicago/Turabian StyleOoi, Li Ching, Vincent Ho, Jing Zhou Zhu, Stephanie Lim, Liping Chung, Askar Abubakar, Tristan Rutland, Wei Chua, Weng Ng, Mark Lee, and et al. 2024. "p21 as a Predictor and Prognostic Indicator of Clinical Outcome in Rectal Cancer Patients" International Journal of Molecular Sciences 25, no. 2: 725. https://doi.org/10.3390/ijms25020725
APA StyleOoi, L. C., Ho, V., Zhu, J. Z., Lim, S., Chung, L., Abubakar, A., Rutland, T., Chua, W., Ng, W., Lee, M., Morgan, M., MacKenzie, S., & Lee, C. S. (2024). p21 as a Predictor and Prognostic Indicator of Clinical Outcome in Rectal Cancer Patients. International Journal of Molecular Sciences, 25(2), 725. https://doi.org/10.3390/ijms25020725








