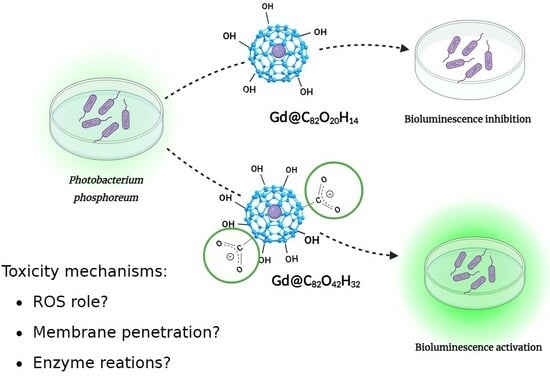
Effects of Endohedral Gd-Containing Fullerenols with a Different Number of Oxygen Substituents on Bacterial Bioluminescence
Abstract
1. Introduction
2. Results and Discussion
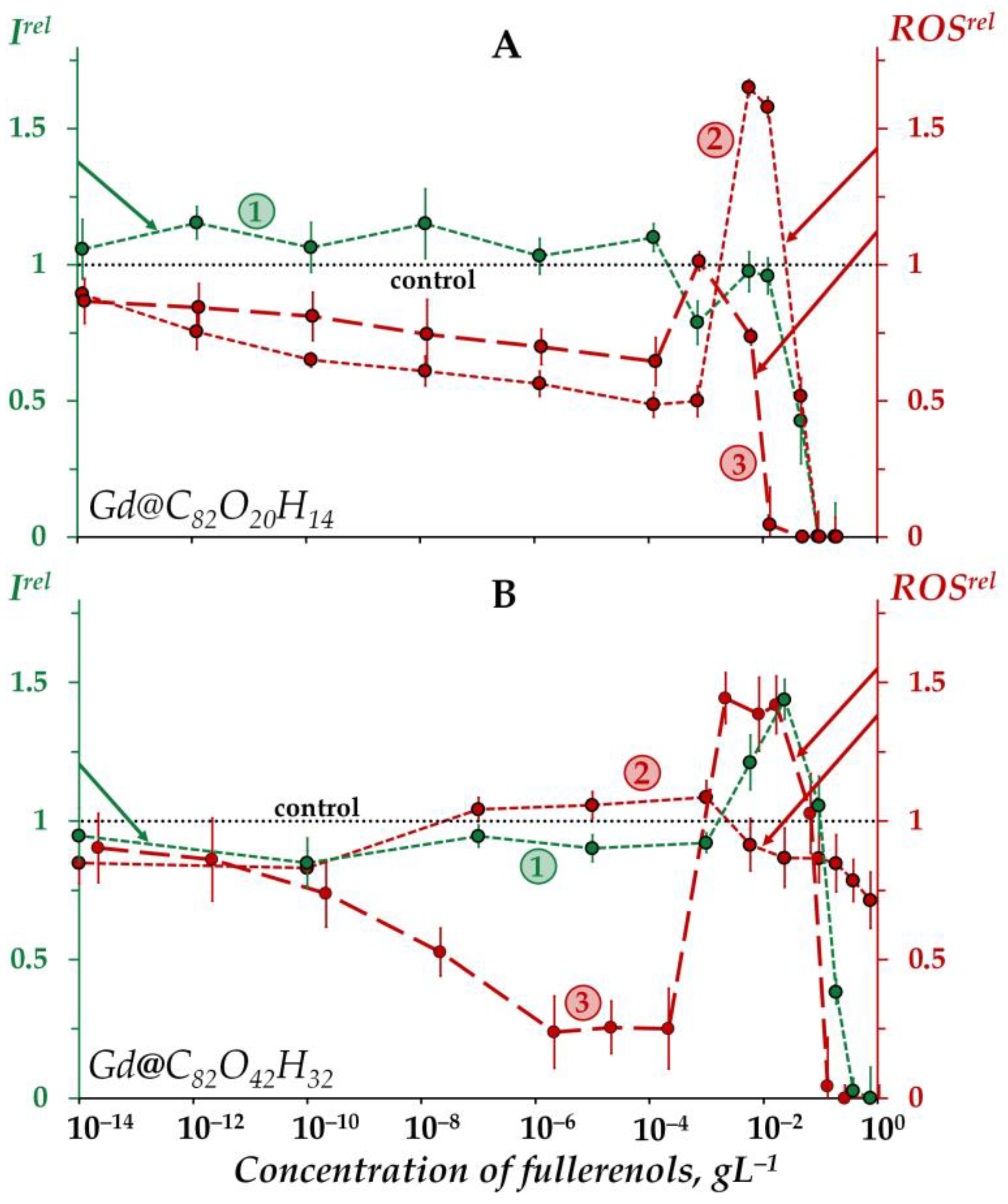
2.1. Effects of Gd@C82O20H14 and Gd@C82O42H32 on Bioluminescence and on the ROS Content in the Bacterial Suspensions
2.1.1. Effects of Gd@C82O20H14 and Gd@C82O42H32 on Bacterial Bioluminescence
2.1.2. Effects of Gd@C82O20H14 and Gd@C82O42H32 on the ROS Content in Bacterial Suspensions
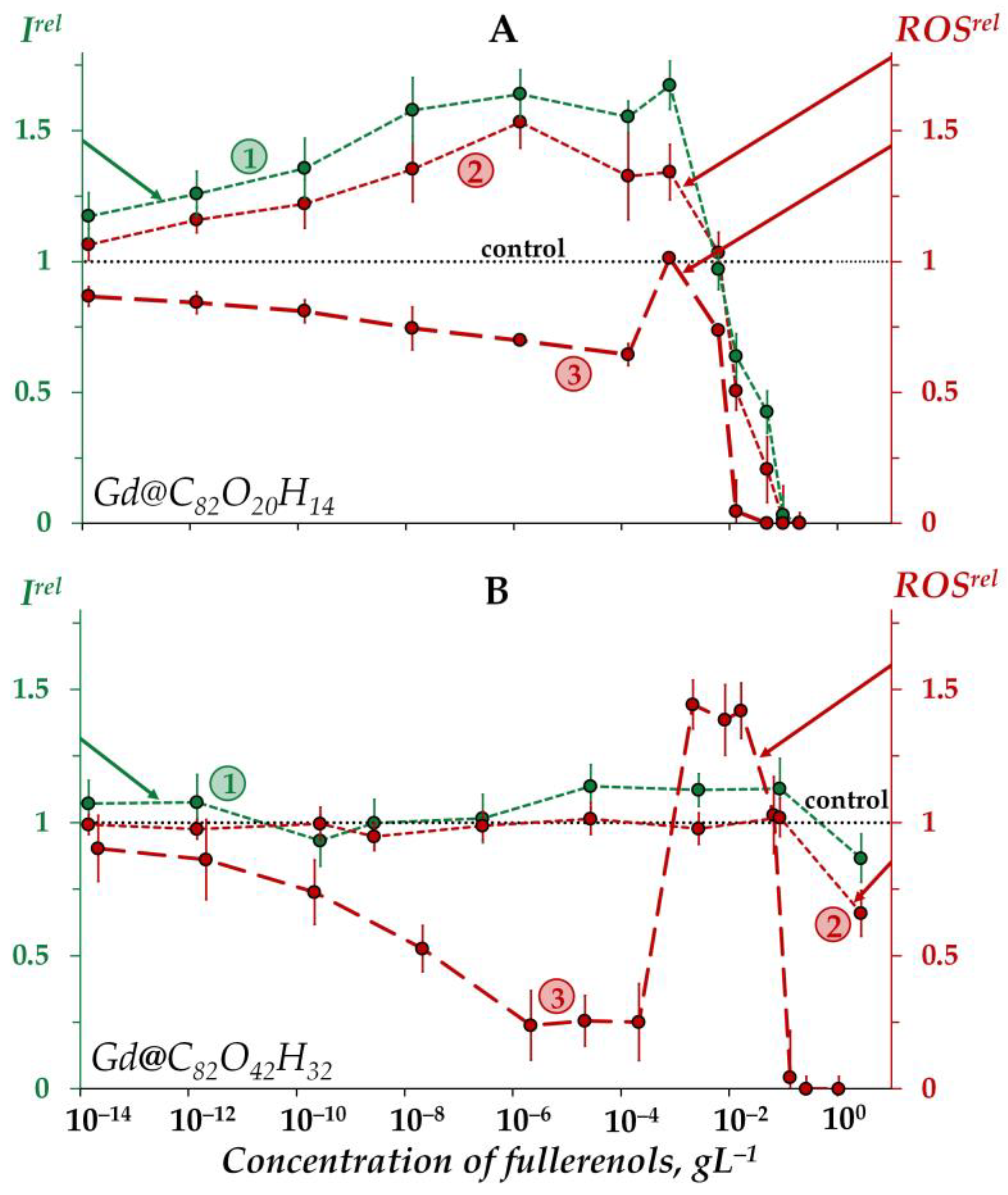
2.2. Effects of Gd@C82O20H14 and Gd@C82O42H32 on Bioluminescence and on the ROS Content in an Enzymatic Solution
2.2.1. Effects of Gd@C82O20H14 and Gd@C82O42H32 on Bioluminescence in Enzymatic Solutions
2.2.2. Effects of Gd@C82O20H14 and Gd@C82O42H32 on the ROS Content in the Enzymatic Solution
2.3. Theoretical Results
2.3.1. Theoretical Calculations of C82O20H14
2.3.2. Theoretical Calculations of C82O42H32
2.3.3. Theoretical Infrared (IR) Spectra of C82O42H32
2.3.4. Theoretical IR Spectra of C82O20H14
2.3.5. Theoretical Discussion
3. Materials and Methods
3.1. Preparation of Fullerenols
3.2. Bioluminescence Assay Systems and Experimental Data Processing
3.3. Luminol Chemiluminescence Assay
3.4. Equipment
3.5. Statistical Processing
3.6. Quantum-Chemical Calculations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DFTB3 | Density Functional Tight Binding method |
| EC50 | Effective concentration of fullerenol which inhibited bioluminescence intensity by 50% |
| FMN | Flavinmononucleotide |
| FTIR | Fourier-transform infrared spectroscopy |
| Gd | Gadolinium |
| I | Bioluminescence intensity |
| IR | Infrared spectroscopy |
| MRI | Magnetic resonance imaging |
| NADH | Nicotinamide adenine dinucleotide, disodium salt, reduced |
| ROS | Reactive oxygen species |
| XPS | X-ray photoelectron spectroscopy |
References
- Fatouros, P.P.; Corwin, F.D.; Chen, Z.J.; Broaddus, W.C.; Tatum, J.L.; Kettenmann, B.; Ge, Z.; Gibson, H.W.; Russ, J.L.; Leonard, A.P.; et al. In Vitro and In Vivo Imaging Studies of a New Endohedral Metallofullerene Nanoparticle. Radiology 2006, 240, 756–764. [Google Scholar] [CrossRef]
- Kobayashi, T.; Yasuno, T.; Takahashi, K.; Nakamura, S.; Mashino, T.; Ohe, T. Novel Pyridinium-Type Fullerene Derivatives as Multitargeting Inhibitors of HIV-1 Reverse Transcriptase, HIV-1 Protease, and HCV NS5B Polymerase. Bioorg. Med. Chem. Lett. 2021, 49, 128267. [Google Scholar] [CrossRef]
- Ren, L.; Jing, Z.; Xia, F.; Zhang, J.Z.; Li, Y. Toxic Effect of Fullerene and Its Derivatives upon the Transmembrane β2-Adrenergic Receptors. Molecules 2022, 27, 4562. [Google Scholar] [CrossRef]
- Cui, X.; Xu, S.; Wang, X.; Chen, C. The nano-bio interaction and biomedical applications of carbon nanomaterials. Carbon 2018, 138, 436–450. [Google Scholar] [CrossRef]
- Ye, L.; Kollie, L.; Liu, X.; Guo, W.; Ying, X.; Zhu, J.; Yang, S.; Yu, M. Antitumor Activity and Potential Mechanism of Novel Fullerene Derivative Nanoparticles. Molecules 2021, 26, 3252. [Google Scholar] [CrossRef]
- Pochkaeva, E.I.; Podolsky, N.E.; Zakusilo, D.N.; Petrov, A.V.; Charykov, N.A.; Vlasov, T.D.; Penkova, A.V.; Vasina, L.V.; Murin, I.V.; Sharoyko, V.V.; et al. Fullerene Derivatives with Amino Acids, Peptides and Proteins: From Synthesis to Biomedical Application. Prog. Solid State Chem. 2020, 57, 100255. [Google Scholar] [CrossRef]
- Yasuno, T.; Ohe, T.; Kataoka, H.; Hashimoto, K.; Ishikawa, Y.; Furukawa, K.; Tateishi, Y.; Kobayashi, T.; Takahashi, K.; Nakamura, S.; et al. Fullerene Derivatives as Dual Inhibitors of HIV-1 Reverse Transcriptase and Protease. Bioorg. Med. Chem. Lett. 2021, 31, 127675. [Google Scholar] [CrossRef]
- Torres, V.M.; Srdjenovic, B. Biomedical Application of Fullerenes. In Handbook on Fullerene: Synthesis, Properties and Applications; Nova Science Publishers: New York, NY, USA, 2012; pp. 197–238. [Google Scholar]
- Troshin, P.A.; Lyubovskaya, R.N. Organic Chemistry of Fullerenes: The Major Reactions, Types of Fullerene Derivatives and Prospects for Practical Use. Russ. Chem. Rev. 2008, 77, 323–369. [Google Scholar] [CrossRef]
- Mroz, P.; Tegos, G.P.; Gali, H.; Wharton, T.; Sarna, T.; Hamblin, M.R. Photodynamic Therapy with Fullerenes. Photochem. Photobiol. Sci. 2007, 6, 1139–1149. [Google Scholar] [CrossRef]
- Bartelmess, J.; Milcovich, G.; Maffeis, V.; d’Amora, M.; Bertozzi, S.M.; Giordani, S. Modulation of Efficient Diiodo-BODIPY in Vitro Phototoxicity to Cancer Cells by Carbon Nano-Onions. Front. Chem. 2020, 8, 573211. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, C.; Xie, J.; Ji, C.; Gu, Z. Eco-Friendly and Scalable Synthesis of Fullerenols with High Free Radical Scavenging Ability for Skin Radioprotection. Small 2021, 17, 2102035. [Google Scholar] [CrossRef]
- Lee, C.-W.; Chi, M.-C.; Peng, K.-T.; Chiang, Y.-C.; Hsu, L.-F.; Yan, Y.-L.; Li, H.-Y.; Chen, M.-C.; Lee, I.-T.; Lai, C.-H. Water-Soluble Fullerenol C60(OH)36 toward Effective Anti-Air Pollution Induced by Urban Particulate Matter in HaCaT Cell. Int. J. Mol. Sci. 2019, 20, 4259. [Google Scholar] [CrossRef]
- Chen, X.; Yang, J.; Li, M.; Zhu, S.; Zhao, M.; Yang, C.; Liu, B.; Gao, H.; Lu, A.; Ge, L.; et al. Fullerenol Protects Cornea from Ultraviolet B Exposure. Redox Biol. 2022, 54, 102360. [Google Scholar] [CrossRef]
- Shafiq, F.; Iqbal, M.; Ali, M.; Ashraf, M.A. Fullerenol Regulates Oxidative Stress and Tissue Ionic Homeostasis in Spring Wheat to Improve Net-Primary Productivity under Salt-Stress. Ecotoxicol. Environ. Saf. 2021, 211, 111901. [Google Scholar] [CrossRef]
- Chen, C.; Xing, G.; Wang, J.; Zhao, Y.; Li, B.; Tang, J.; Jia, G.; Wang, T.; Sun, J.; Xing, L.; et al. Multihydroxylated [Gd@C82(OH)22]n Nanoparticles: Antineoplastic Activity of High Efficiency and Low Toxicity. Nano Lett. 2005, 5, 2050–2057. [Google Scholar] [CrossRef]
- Anderson, S.A.; Lee, K.K.; Frank, J.A. Gadolinium-Fullerenol as a Paramagnetic Contrast Agent for Cellular Imaging. Investig. Radiol. 2006, 41, 332–338. [Google Scholar] [CrossRef]
- Chen, K.; Wang, Y.; Liang, H.; Huang, H.; Liang, Y.; Zhang, J.; Chang, Y.; Li, J.; Fang, M.; Xing, G. Fullerenols Boosting the Therapeutic Effect of Anti-CD47 Antibody to Trigger Robust Anti-Tumor Immunity by Inducing Calreticulin Exposure. Nano Today 2021, 37, 101070. [Google Scholar] [CrossRef]
- Grebowski, J.; Kazmierska, P.; Krokosz, A. Fullerenols as a new therapeutic approach in nanomedicine. Biomed. Res. Int. 2013, 2013, 751913. [Google Scholar] [CrossRef]
- Cai, X.; Hao, J.; Zhang, X.; Yu, B.; Ren, J.; Luo, C.; Li, Q.; Huang, Q.; Shi, X.; Li, W.; et al. The polyhydroxylated fullerene derivative C60(OH)24 protects mice from ionizing-radiation-induced immune and mitochondrial dysfunction. Toxicol. Appl. Pharmacol. 2010, 243, 27–34. [Google Scholar] [CrossRef]
- Slavic, M.; Djordjevic, A.; Radojicic, R.; Milovanovic, S.; Orescanin-Dusic, Z.; Rakocevic, Z.; Spasic, M.B.; Blagojevic, D. Fullerenol C60(OH)24 nanoparticles decrease relaxing effects of dimethyl sulfoxide on rat uterus spontaneous contraction. J. Nanopart. Res. 2013, 15, 1650. [Google Scholar] [CrossRef]
- Mirkov, S.M.; Djordjevic, A.N.; Andric, N.L.; Andric, S.A.; Kostic, T.S.; Bogdanovic, G.M.; Vojinovic-Miloradov, M.B.; Kovacevic, R.Z. Nitric oxide-scavenging activity of polyhydroxylated fullerenol, C60(OH)24. Nitric Oxide 2004, 11, 201–207. [Google Scholar] [CrossRef]
- Injac, R.; Prijatelj, M.; Strukelj, B. Fullerenol nanoparticles: Toxicity and antioxidant activity. Methods Mol. Biol. 2013, 1028, 75–100. [Google Scholar] [CrossRef]
- Eropkin, M.Y.; Melenevskaya, E.Y.; Nasonova, K.V.; Bryazzhikova, T.S.; Eropkina, E.M.; Danilenko, D.M.; Kiselev, O.I. Synthesis and Biological Activity of Fullerenols with Various Contents of Hydroxyl Groups. Pharm. Chem. J. 2013, 47, 87–91. [Google Scholar] [CrossRef]
- Kovel, E.S.; Sachkova, A.S.; Vnukova, N.G.; Churilov, G.N.; Knyazeva, E.M.; Kudryasheva, N.S. Antioxidant activity and toxicity of fullerenols via bioluminescence signaling: Role of oxygen substituents. Int. J. Mol. Sci. 2019, 20, 2324. [Google Scholar] [CrossRef]
- Kovel, E.S.; Kicheeva, A.G.; Vnukova, N.G.; Churilov, G.N.; Stepin, E.A.; Kudryasheva, N.S. Toxicity and Antioxidant Activity of Fullerenol C60,70 with Low Number of Oxygen Substituents. Int. J. Mol. Sci. 2021, 22, 6382. [Google Scholar] [CrossRef]
- Li, J.; Chen, L.; Yan, L.; Gu, Z.; Chen, Z.; Zhang, A.; Zhao, F. A Novel Drug Design Strategy: An Inspiration from Encaging Tumor by Metallofullerenol Gd@C82(OH)22. Molecules 2019, 24, 2387. [Google Scholar] [CrossRef]
- Li, X.; Wang, C. The potential biomedical platforms based on the functionalized Gd@C82 nanomaterials. View 2020, 1, e7. [Google Scholar] [CrossRef]
- Li, J.; Chen, L.; Su, H.; Yan, L.; Gu, Z.; Chen, Z.; Zhang, A.; Zhao, F.; Zhao, Y. The Pharmaceutical Multi-Activity of Metallofullerenol Invigorates Cancer Therapy. Nanoscale 2019, 11, 14528–14539. [Google Scholar] [CrossRef]
- Fernandes, N.B.; Shenoy, R.U.K.; Kajampady, M.K.; DCruz, C.E.M.; Shirodkar, R.K.; Kumar, L.; Verma, R. Fullerenes for the treatment of cancer: An emerging tool. Environ. Sci. Pollut. Res. Int. 2022, 29, 58607–58627. [Google Scholar] [CrossRef]
- Meng, J.; Liang, X.; Chen, X.; Zhao, Y. Biological characterizations of [Gd@C82(OH)22]n nanoparticles as fullerene derivatives for cancer therapy. Integr. Biol. 2013, 5, 43–47. [Google Scholar] [CrossRef]
- Maravilla, K.R.; San-Juan, D.; Kim, S.J.; Elizondo-Riojas, G.; Fink, J.R.; Escobar, W.; Bag, A.; Roberts, D.R.; Hao, J.; Pitrou, C.; et al. Comparison of Gadoterate Meglumine and Gadobutrol in the MRI Diagnosis of Primary Brain Tumors: A Double-Blind Randomized Controlled Intraindividual Crossover Study (the REMIND Study). AJNR Am. J. Neuroradiol. 2017, 38, 1681–1688. [Google Scholar] [CrossRef]
- Ersoy, H.; Rybicki, F.J. Biochemical Safety Profiles of Gadolinium-Based Extracellular Contrast Agents and Nephrogenic Systemic Fibrosis. J. Magn. Reson. Imaging 2007, 26, 1190–1197. [Google Scholar] [CrossRef]
- Sosnovik, D.E.; Caravan, P. Molecular MRI of the Cardiovascular System in the Post-NSF Era. Curr. Cardiovasc. Imaging Rep. 2013, 6, 61–68. [Google Scholar] [CrossRef]
- Zhang, J.; Ye, Y.; Chen, Y.; Pregot, C.; Li, T.; Balasubramaniam, S.; Hobart, D.B.; Zhang, Y.; Wi, S.; Davis, R.M.; et al. Gd3N@C84(OH)x: A New Egg-Shaped Metallofullerene Magnetic Resonance Imaging Contrast Agent. J. Am. Chem. Soc. 2014, 136, 2630–2636. [Google Scholar] [CrossRef]
- Yang, S.; Lian, G. ROS and Diseases: Role in Metabolism and Energy Supply. Mol. Cell. Biochem. 2020, 467, 1–12. [Google Scholar] [CrossRef]
- Yang, B.; Chen, Y.; Shi, J. Reactive Oxygen Species (ROS)-Based Nanomedicine. Chem. Rev. 2019, 119, 4881–4985. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive Oxygen Species (ROS) as Pleiotropic Physiological Signalling Agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
- Fan, J.; Ren, D.; Wang, J.; Liu, X.; Zhang, H.; Wu, M.; Yang, G. Bruceine D Induces Lung Cancer Cell Apoptosis and Autophagy via the ROS/MAPK Signaling Pathway in Vitro and in Vivo. Cell Death Dis. 2020, 11, 126. [Google Scholar] [CrossRef]
- Su, L.J.; Zhang, J.H.; Gomez, H.; Murugan, R.; Hong, X.; Xu, D.; Jiang, F.; Peng, Z.Y. Reactive Oxygen Species-Induced Lipid Peroxidation in Apoptosis, Autophagy, and Ferroptosis. Oxid. Med. Cell. Longev. 2019, 2019, 5080843. [Google Scholar] [CrossRef]
- Tavsan, Z.; Kayali, H.A. Flavonoids Showed Anticancer Effects on the Ovarian Cancer Cells: Involvement of Reactive Oxygen Species, Apoptosis, Cell Cycle and Invasion. Biomed. Pharmacother. 2019, 116, 109004. [Google Scholar] [CrossRef]
- Fleury, C.; Mignotte, B.; Vayssière, J.-L. Mitochondrial Reactive Oxygen Species in Cell Death Signaling. Biochimie 2002, 84, 131–141. [Google Scholar] [CrossRef]
- Fernandez-gil, B.I.; Guerra-librero, A.; Shen, Y.; Florido, J.; Martínez-ruiz, L.; García-lópez, S.; Adan, C.; Rodríguez-santana, C.; Acuña-castroviejo, D.; Quiñones-hinojosa, A.; et al. Melatonin Enhances Cisplatin and Radiation Cytotoxicity in Head and Neck Squamous Cell Carcinoma by Stimulating. Oxid. Med. Cell. Longev. 2019, 2019, 7187128. [Google Scholar] [CrossRef]
- Gheena, S.; Ezhilarasan, D. Syringic Acid Triggers Reactive Oxygen Species–mediated Cytotoxicity in HepG2 Cells. Hum. Exp. Toxicol. 2019, 38, 694–702. [Google Scholar] [CrossRef]
- Sies, H.; Belousov, V.V.; Chandel, N.S.; Davies, M.J.; Jones, D.P.; Mann, G.E.; Murphy, M.P.; Yamamoto, M.; Winterbourn, C. Defining Roles of Specific Reactive Oxygen Species (ROS) in Cell Biology and Physiology. Nat. Rev. Mol. Cell Biol. 2022, 23, 499–515. [Google Scholar] [CrossRef]
- Jakubczyk, K.; Dec, K.; Kałduńska, J.; Kawczuga, D.; Kochman, J.; Janda, K. Reactive oxygen species—Sources, functions, oxidative damage. Pol. Merkur. Lekarski. 2020, 48, 124–127. [Google Scholar]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Parvin, K.; Bhuiyan, T.F.; Anee, T.I.; Nahar, K.; Hossen, M.S.; Zulfiqar, F.; Alam, M.M.; Fujita, M. Regulation of ROS Metabolism in Plants under Environmental Stress: A Review of Recent Experimental Evidence. Int. J. Mol. Sci. 2020, 21, 8695. [Google Scholar] [CrossRef]
- Bulich, A.A.; Isenberg, D.L. Use of the luminescent bacterial system for rapid assessment of aquatic toxicity. ISA Trans. 1981, 20, 29–33. [Google Scholar]
- Roda, A.; Pasini, P.; Mirasoni, M.; Michchelini, E.; Guardigli, M. Biotechnological application of bioluminescence and chemiluminescence. Trends Biotechnol. 2004, 22, 295–303. [Google Scholar] [CrossRef]
- Abbas, M.; Adil, M.; Ehtisham-Ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Shar, G.A.; Tahir, M.A.; Iqbal, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar] [CrossRef]
- Ismailov, A.D.; Aleskerova, L.E. Photobiosensors containing luminescent bacteria. Biochemistry 2015, 80, 733–744. [Google Scholar] [CrossRef]
- Ventura, F.F.; Mendes, L.F.; Oliveira, A.G.; Bazito, R.C.; Bechara, E.J.H.; Freire, R.S.; Stevani, C.V. Evaluation of Phenolic Compound Toxicity Using a Bioluminescent Assay with the Fungus Gerronema viridilucens. Environ. Toxicol. Chem. 2020, 39, 1558–1565. [Google Scholar] [CrossRef]
- Li, Y.; He, X.; Zhu, W.; Li, H.; Wang, W. Bacterial Bioluminescence Assay for Bioanalysis and Bioimaging. Anal. Bioanal. Chem. 2022, 414, 75–83. [Google Scholar] [CrossRef]
- Kudryasheva, N.S.; Kovel, E.S. Monitoring of low-intensity exposures via luminescent bioassays of different complexity: Cells, enzyme reactions and fluorescent proteins. Int. J. Mol. Sci. 2019, 20, 4451. [Google Scholar] [CrossRef]
- Esimbekova, E.N.; Torgashina, I.G.; Kalyabina, V.P. Enzymatic Biotesting: Scientific Basis and Application. Contemp. Probl. Ecol. 2021, 14, 290–304. [Google Scholar] [CrossRef]
- Esimbekova, E.N.; Kalyabina, V.P.; Kopylova, K.V.; Torgashina, I.G.; Kratasyuk, V.A. Design of bioluminescent biosensors for assessing contamination of complex matrices. Talanta 2021, 233, 122509. [Google Scholar] [CrossRef]
- Sushko, E.S.; Vnukova, N.G.; Churilov, G.N.; Kudryasheva, N.S. Endohedral Gd-Containing Fullerenol: Toxicity, Antioxidant Activity, and Regulation of Reactive Oxygen Species in Cellular and Enzymatic Systems. Int. J. Mol. Sci. 2022, 23, 5152. [Google Scholar] [CrossRef]
- Kudryasheva, N.S.; Tarasova, A.S. Pollutant toxicity and detoxification by humic substances: Mechanisms and quantitative assessment via luminescent biomonitoring. Environ. Sci. Pollut. Res. 2015, 22, 155–167. [Google Scholar] [CrossRef]
- Yehia, M.R.; Smolyarova, T.E.; Shabanov, A.V.; Sushko, E.S.; Badun, G.A.; Kudryasheva, N.S. Adaptation of a Bacterial Bioluminescent Assay to Monitor Bioeffects of Gold Nanoparticles. Bioengineering 2022, 9, 61. [Google Scholar] [CrossRef]
- Kudryasheva, N.S.; Kovel, E.S.; Sachkova, A.S.; Vorobeva, A.A.; Isakova, V.G.; Churilov, G.N. Bioluminescent enzymatic assay as a tool for studying antioxidant activity and toxicity of bioactive compounds. Photochem. Photobiol. 2017, 93, 536–540. [Google Scholar] [CrossRef]
- Sachkova, A.S.; Kovel, E.S.; Churilov, G.N.; Stom, D.I.; Kudryasheva, N.S. Biological activity of carbonic nano-structures—Comparison via enzymatic bioassay. J. Soils Sediments 2019, 19, 2689–2696. [Google Scholar] [CrossRef]
- Bondarenko, L.S.; Kovel, E.S.; Kydralieva, K.A.; Dzhardimalieva, G.I.; Illé, E.; Tombácz, E.; Kicheeva, A.G.; Kudryasheva, N.S. Effects of Modified Magnetite Nanoparticles on Bacterial Cells and Enzyme Reactions. Nanomaterials 2020, 10, 1499. [Google Scholar] [CrossRef] [PubMed]
- Kicheeva, A.G.; Sushko, E.S.; Bondarenko, L.S.; Kydralieva, K.A.; Pankratov, D.A.; Tropskaya, N.S.; Dzeranov, A.A.; Dzhardimalieva, G.I.; Zarrelli, M.; Kudryasheva, N.S. Functionalized Magnetite Nanoparticles: Characterization, Bioeffects, and Role of Reactive Oxygen Species in Unicellular and Enzymatic Systems. Int. J. Mol. Sci. 2023, 24, 1133. [Google Scholar] [CrossRef] [PubMed]
- Alexandrova, M.; Rozhko, T.; Vydryakova, G.; Kudryasheva, N. Effect of americium-241 on luminous bacteria. Role of peroxides. J. Environ. Radioact. 2011, 102, 407–411. [Google Scholar] [CrossRef]
- Rozhko, T.; Nogovitsyna, E.; Badun, G.; Lukyanchuk, A.; Kudryasheva, N. Reactive Oxygen Species and Low-Dose Effects of Tritium on Bacterial Cells. J. Environ. Radioact. 2019, 208–209, 106035. [Google Scholar] [CrossRef] [PubMed]
- Rozhko, T.V.; Kolesnik, O.V.; Badun, G.A.; Stom, D.I.; Kudryasheva, N.S. Humic Substances Mitigate the Impact of Tritium on Luminous Marine Bacteria. Involvement of Reactive Oxygen Species. Int. J. Mol. Sci. 2020, 21, 6783. [Google Scholar] [CrossRef] [PubMed]
- Kolesnik, O.V.; Rozhko, T.V.; Lapina, M.A.; Solovyev, V.S.; Sachkova, A.S.; Kudryasheva, N.S. Development of Cellular and Enzymatic Bioluminescent Assay Systems to Study Low-Dose Effects of Thorium. Bioengineering 2021, 8, 194. [Google Scholar] [CrossRef]
- Calabrese, E. Hormesis: Path and Progression to Significance. Int. J. Mol. Sci. 2018, 19, 2871. [Google Scholar] [CrossRef]
- Jargin, S.V. Hormesis and Radiation Safety Norms: Comments for an Update. Hum. Exp. Toxicol. 2018, 37, 1233–1243. [Google Scholar] [CrossRef]
- Shibamoto, Y.; Nakamura, H. Overview of Biological, Epidemiological, and Clinical Evidence of Radiation Hormesis. Int. J. Mol. Sci. 2018, 19, 2387. [Google Scholar] [CrossRef]
- Ge, H.; Zhou, M.; Lv, D.; Wang, M.; Xie, D.; Yang, X.; Dong, C.; Li, S.; Lin, P. Novel Segmented Concentration Addition Method to Predict Mixture Hormesis of Chlortetracycline Hydrochloride and Oxytetracycline Hydrochloride to Aliivibrio fischeri. Int. J. Mol. Sci. 2020, 21, 481. [Google Scholar] [CrossRef]
- Kaiser, J. Hormesis: Sipping from a poisoned chalice Science. Science 2003, 302, 376–379. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Hormetic mechanisms. Crit. Rev. Toxicol. 2013, 43, 580–606. [Google Scholar] [CrossRef] [PubMed]
- Balogh, L.P. Caging cancer. Nanomedicine 2015, 11, 867–869. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Kang, S.G.; Wang, P.; Wang, Y.; Lv, X.; Liu, Y.; Wang, F.; Gu, Z.; Yang, Z.; Weber, J.K.; et al. Molecular Mechanism of Gd@C82(OH)22 Increasing Collagen Expression: Implication for Encaging Tumor. Biomaterials 2018, 152, 24–36. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, C.; Qian, P.; Lu, X.; Sun, B.; Zhang, X.; Wang, L.; Gao, X.; Li, H.; Chen, Z.; et al. Gd-Metallofullerenol Nanomaterial as Non-Toxic Breast Cancer Stem Cell-Specific Inhibitor. Nat. Commun. 2015, 6, 5988. [Google Scholar] [CrossRef] [PubMed]
- Rokitskaya, T.I.; Antonenko, Y.N. Fullerenol C60(OH)24 Increases Ion Permeability of Lipid Membranes in a pH-Dependent Manner. Biochim. Biophys. Acta Biomembr. 2016, 1858, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xing, G.; Yuan, H.; Chang, X.; Jing, L.; Zhao, Y.; Zhu, C.; Fang, X. Transmembrane Delivery of Aggregated [Gd@C82(OH)22]n Nanoparticles. J. Nanosci. Nanotechnol. 2010, 10, 8556–8561. [Google Scholar] [CrossRef] [PubMed]
- Rozhko, T.V.; Kudryasheva, N.S.; Aleksandrova, M.A.; Bondareva, L.G.; Bolsunovsky, A.A.; Vydryakova, G.V. Comparison of Effects of Uranium and Americium on Bioluminescent Bacteria. J. Sib. Fed. Univ. Biol. 2008, 1, 60–65. [Google Scholar]
- Girotti, S.; Ferri, E.N.; Fumo, M.G.; Maiolini, E. Monitoring of Environmental Pollutants by Bioluminescent Bacteria. Anal. Chim. Acta 2008, 608, 2–29. [Google Scholar] [CrossRef]
- Nemtseva, E.V.; Kudryasheva, N. The mechanism of electronic excitation in bacterial bioluminescent reaction. Russ. Chem. Rev. 2007, 76, 101–112. [Google Scholar] [CrossRef]
- Hastings, J.W.; Gibson, Q.H. Intermediates in the bioluminescent oxidation of reduced flavin mononucleotide. J. Biol. Chem. 1963, 238, 2537–2554. [Google Scholar] [CrossRef] [PubMed]
- Tomilin, F.N.; Artyushenko, P.V.; Shchugoreva, I.A.; Rogova, A.V.; Vnukova, N.G.; Churilov, G.N.; Shestakov, N.P.; Tchaikovskaya, O.N.; Ovchinnikov, S.G.; Avramov, P.V. Structure and Vibrational Spectroscopy of C82 Fullerenol Valent Isomers: An Experimental and Theoretical Joint Study. Molecules 2023, 28, 1569. [Google Scholar] [CrossRef] [PubMed]
- Popov, A.A.; Yang, S.; Dunsch, L. Endohedral Fullerenes. Chem. Rev. 2013, 113, 5989–6113. [Google Scholar] [CrossRef] [PubMed]
- Churilov, G.; Popov, A.; Vnukova, N.; Dudnik, A.; Samoylova, N.; Glushenko, G. Controlled synthesis of fullerenes and endohedral metallofullerenes in high frequency arc discharge. Fuller. Nanotub. Carbon Nanostruct. 2016, 24, 675–678. [Google Scholar] [CrossRef]
- Akiyama, K.; Hamano, T.; Nakanishi, Y.; Takeuchi, E.; Noda, S.; Wang, Z.; Kubuki, S.; Shinohara, H. Non-HPLC rapid separation of metallofullerenes and empty cages with TiCl4 Lewis acid. J. Am. Chem. Soc. 2012, 134, 9762–9767. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Huang, H.; Yang, S. Synthesis and characterization of a water-soluble endohedral metallofullerol. Chem. Mater. 1999, 11, 1003–1006. [Google Scholar] [CrossRef]
- Li, J.; Zhang, M.; Sun, B.; Xing, G.; Song, Y.; Guo, H.; Chang, Y.; Ge, Y.; Zhao, Y. Separation and purification of fullerenols for improved biocompatibility. Carbon 2012, 50, 460–469. [Google Scholar] [CrossRef]
- Li, J.; Wang, T.; Feng, Y.; Zhang, Y.; Zhen, M.; Shu, C.; Jiang, L.; Wang, Y.; Wang, C. A water-soluble gadolinium metallofullerenol: Facile preparation, magnetic properties and magnetic resonance imaging application. Dalton Trans. 2016, 45, 8696–8699. [Google Scholar] [CrossRef]
- Dudnik, A.I.; Vnukova, N.G.; Drokin, N.A.; Bondarev, V.S.; Shestakov, N.P.; Tomashevich, Y.V.; Churilov, G.N. Electrophysical Properties of Hydroxylated Endohedral Metallofullerene with Gadolinium. J. Phys. Chem. Solids 2019, 135, 109094. [Google Scholar] [CrossRef]
- Kuznetsov, A.M.; Rodicheva, E.K.; Shilova, E.V. Bioassay based on lyophilized bacteria. Biotekhnologiya 1996, 9, 57–61. (In Russian) [Google Scholar]
- Fedorova, E.; Kudryasheva, N.; Kuznetsov, A.; Mogil’naya, O.; Stom, D. Bioluminescent monitoring of detoxification processes: Activity of humic substances in quinone solutions. J. Photochem. Photobiol. B 2007, 88, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Khan, P.; Idrees, D.; Moxley, M.A.; Corbett, J.A.; Ahmad, F.; von Figura, G.; Sly, W.S.; Waheed, A.; Hassan, M.I. Luminol-Based Chemiluminescent Signals: Clinical and Non-Clinical Application and Future Uses. Appl. Biochem. Biotechnol. 2014, 173, 333–355. [Google Scholar] [CrossRef]
- Vasil’ev, R.F.; Veprintsev, T.L.; Dolmatova, L.S.; Naumov, V.V.; Trofimov, A.V.; Tsaplev, Y.B. Kinetics of Ethylbenzene Oxy-Chemiluminescence in the Presence of Antioxidants from Tissues of the Marine Invertebrate Eupentacta Fraudatrix: Estimating the Concentration and Reactivity of the Natural Antioxidants. Kinet. Catal. 2014, 55, 148–153. [Google Scholar] [CrossRef]
- Gmurman, V.E.; Berenblut, I.I. Fundamentals of Probability Theory and Mathematical Statistics; Berenblut, I.I., Ed.; Iliffe Book Ltd.: London, UK, 1968; p. 249. [Google Scholar]
- Rüger, R.; Niehaus, T.; Van Lenthe, E.; Heine, T.; Visscher, L. Vibrationally Resolved UV/Vis Spectroscopy with Time-Dependent Density Functional Based Tight Binding. J. Chem. Phys. 2016, 145, 184102. [Google Scholar] [CrossRef] [PubMed]
- Inakollu, V.S.S.; Yu, H. A Systematic Benchmarking of Computational Vibrational Spectroscopy with DFTB3: Normal Mode Analysis and Fast Fourier Transform Dipole Autocorrelation Function. J. Comput. Chem. 2018, 39, 2067–2078. [Google Scholar] [CrossRef]
- Gaus, M.; Goez, A.; Elstner, M. Parametrization and Benchmark of DFTB3 for Organic Molecules. J. Chem. Theory Comput. 2013, 9, 338–354. [Google Scholar] [CrossRef]
- Barca, G.M.J.; Bertoni, C.; Carrington, L.; Datta, D.; De Silva, N.; Deustua, J.E.; Fedorov, D.G.; Gour, J.R.; Gunina, A.O.; Guidez, E.; et al. Recent Developments in the General Atomic and Molecular Electronic Structure System. J. Chem. Phys. 2020, 152, 154102. [Google Scholar] [CrossRef]
- Afreen, S.; Kokubo, K.; Muthoosamy, K.; Manickam, S. Hydration or hydroxylation: Direct synthesis of fullerenol from pristine fullerene [C60] via acoustic cavitation in the presence of hydrogen peroxide. RSC Adv. 2017, 7, 31930–31939. [Google Scholar] [CrossRef]
- Singh, R.; Goswami, T. Understanding of thermo-gravimetric analysis to calculate number of addends in multifunctional hemi-ortho ester derivatives of fullerenol. Thermochim. Acta 2011, 513, 60–67. [Google Scholar] [CrossRef]
- Wang, B.C.; Wang, H.W.; Tso, H.C.; Chen, T.L.; Chou, Y.M. Theoretical studies of C70(OH)n (n = 14, 16, 18 and 20) fullerols. J. Mol. Struct. Theochem. 2002, 581, 177–186. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stepin, E.A.; Sushko, E.S.; Vnukova, N.G.; Churilov, G.N.; Rogova, A.V.; Tomilin, F.N.; Kudryasheva, N.S. Effects of Endohedral Gd-Containing Fullerenols with a Different Number of Oxygen Substituents on Bacterial Bioluminescence. Int. J. Mol. Sci. 2024, 25, 708. https://doi.org/10.3390/ijms25020708
Stepin EA, Sushko ES, Vnukova NG, Churilov GN, Rogova AV, Tomilin FN, Kudryasheva NS. Effects of Endohedral Gd-Containing Fullerenols with a Different Number of Oxygen Substituents on Bacterial Bioluminescence. International Journal of Molecular Sciences. 2024; 25(2):708. https://doi.org/10.3390/ijms25020708
Chicago/Turabian StyleStepin, Evsei A., Ekaterina S. Sushko, Natalia G. Vnukova, Grigoriy N. Churilov, Anastasia V. Rogova, Felix N. Tomilin, and Nadezhda S. Kudryasheva. 2024. "Effects of Endohedral Gd-Containing Fullerenols with a Different Number of Oxygen Substituents on Bacterial Bioluminescence" International Journal of Molecular Sciences 25, no. 2: 708. https://doi.org/10.3390/ijms25020708
APA StyleStepin, E. A., Sushko, E. S., Vnukova, N. G., Churilov, G. N., Rogova, A. V., Tomilin, F. N., & Kudryasheva, N. S. (2024). Effects of Endohedral Gd-Containing Fullerenols with a Different Number of Oxygen Substituents on Bacterial Bioluminescence. International Journal of Molecular Sciences, 25(2), 708. https://doi.org/10.3390/ijms25020708