Glycogen Synthase Kinase-3 Inhibition by CHIR99021 Promotes Alveolar Epithelial Cell Proliferation and Lung Regeneration in the Lipopolysaccharide-Induced Acute Lung Injury Mouse Model
Abstract
1. Introduction
2. Results
2.1. GSK-3 Inhibition by CHIR99021 Attenuates Lung Injury in LPS-Induced ALI
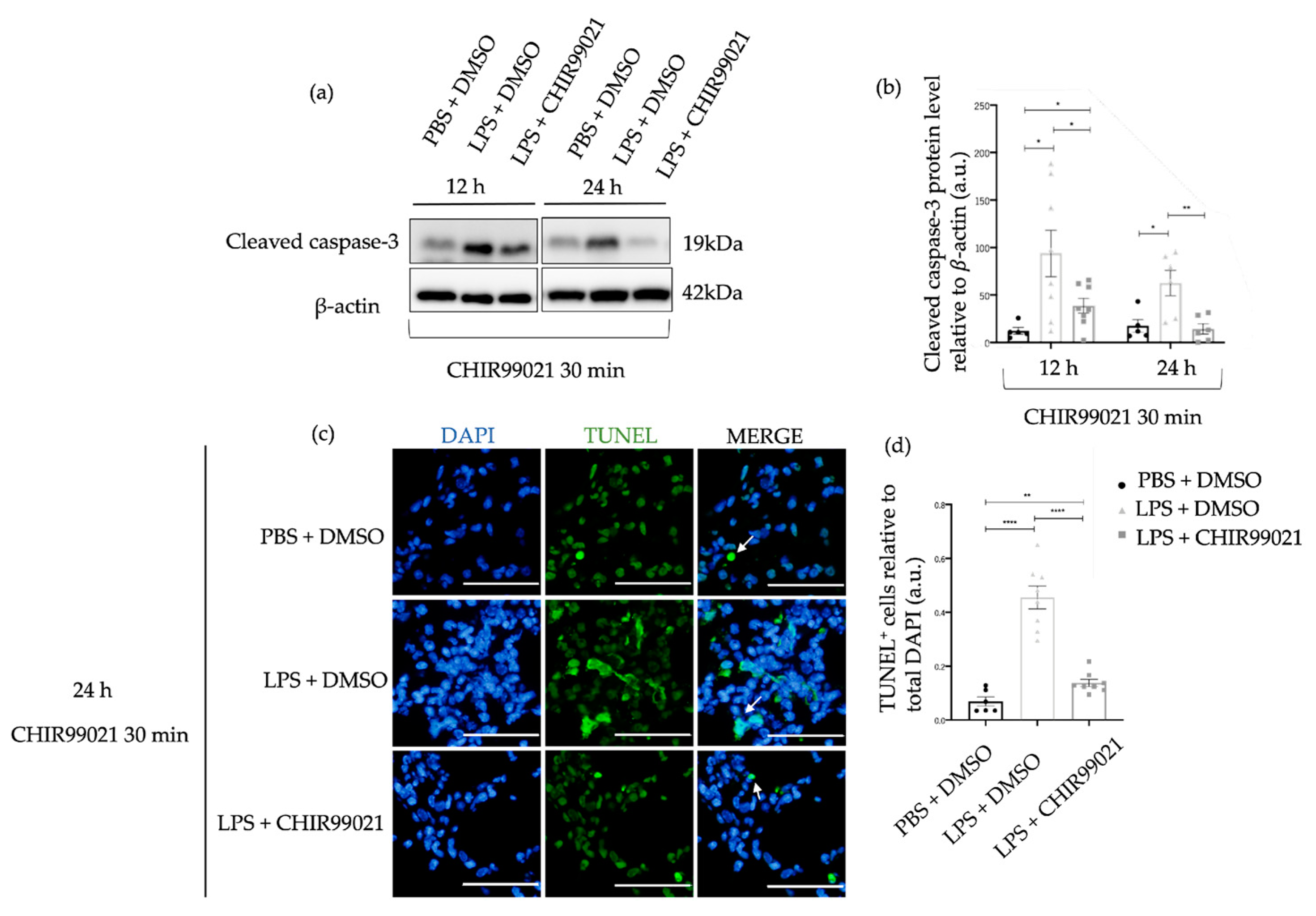
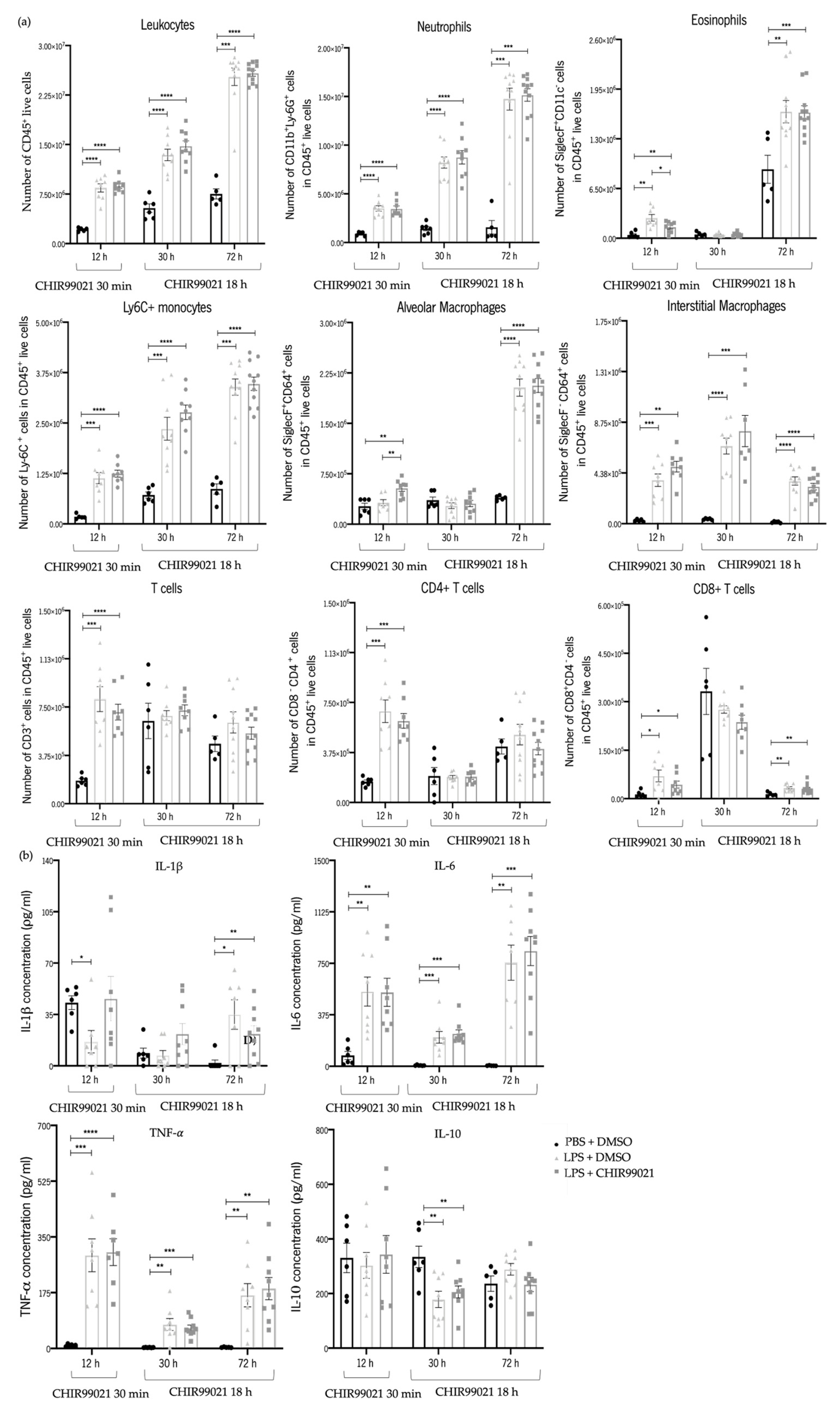
2.2. GSK-3 Inhibition by CHIR99021 Does Not Affect the Recruitment of Inflammatory Cells and the Production of Pro- and Anti-Inflammatory Cytokines in LPS-Induced ALI
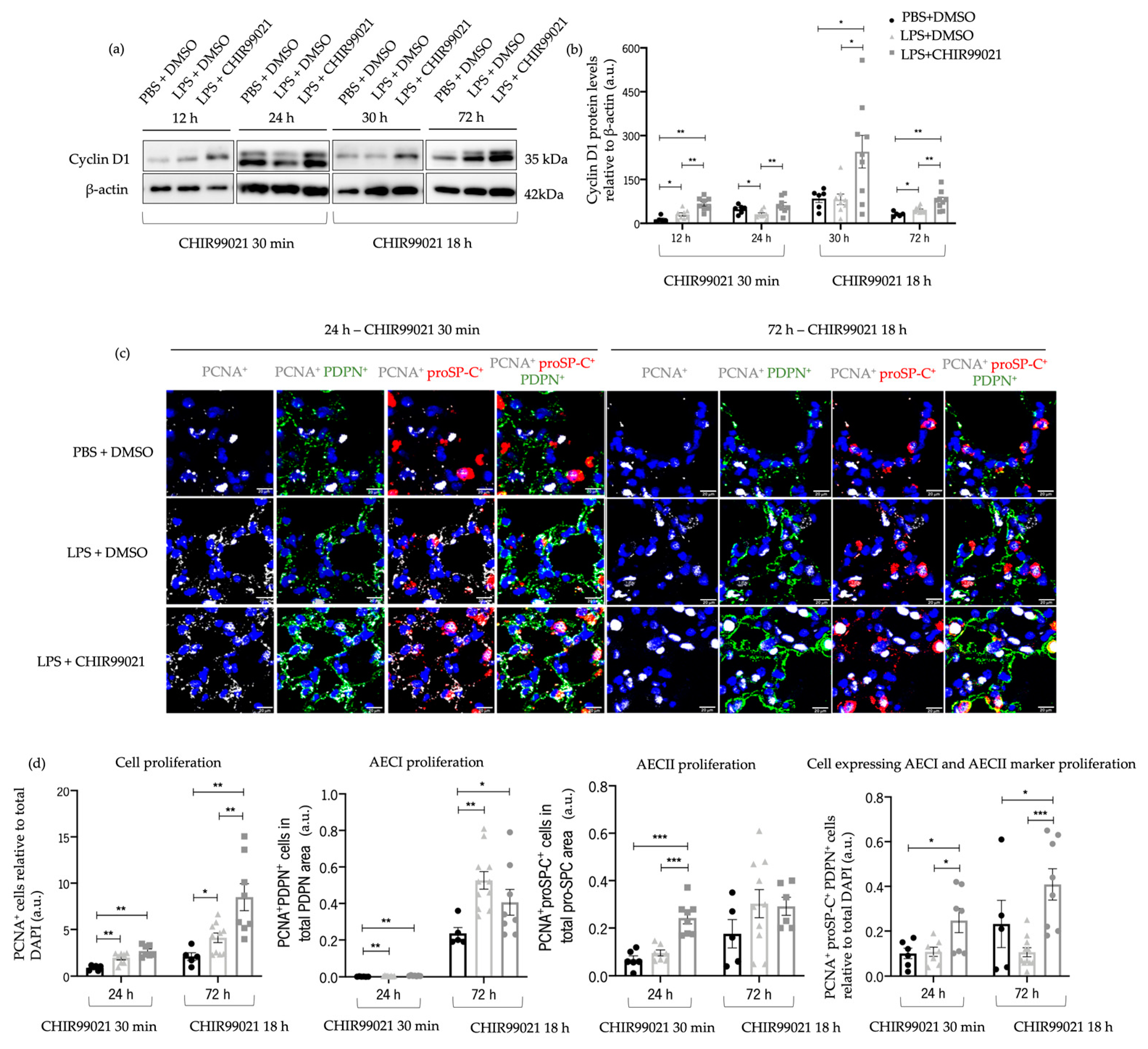
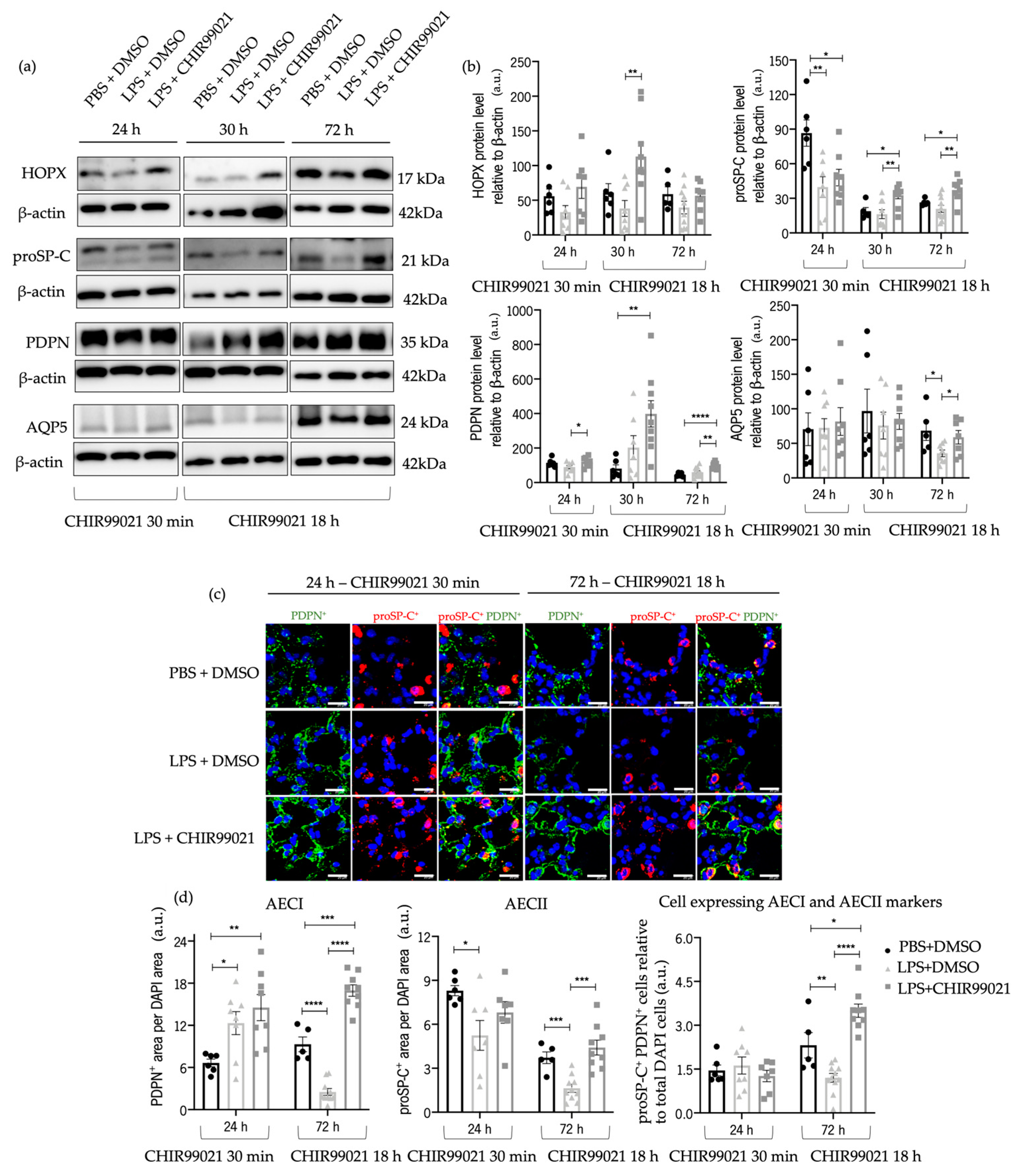
2.3. GSK-3 Inhibition by CHIR99021 Promotes the Proliferation of Cells Expressing AECI and AECII Markers in LPS-Induced ALI
2.4. GSK-3 Inhibition by CHIR99021 Promotes Alveolar Epithelial Population Renewal in LPS-Induced ALI
3. Discussion
4. Materials and Methods
4.1. Mice
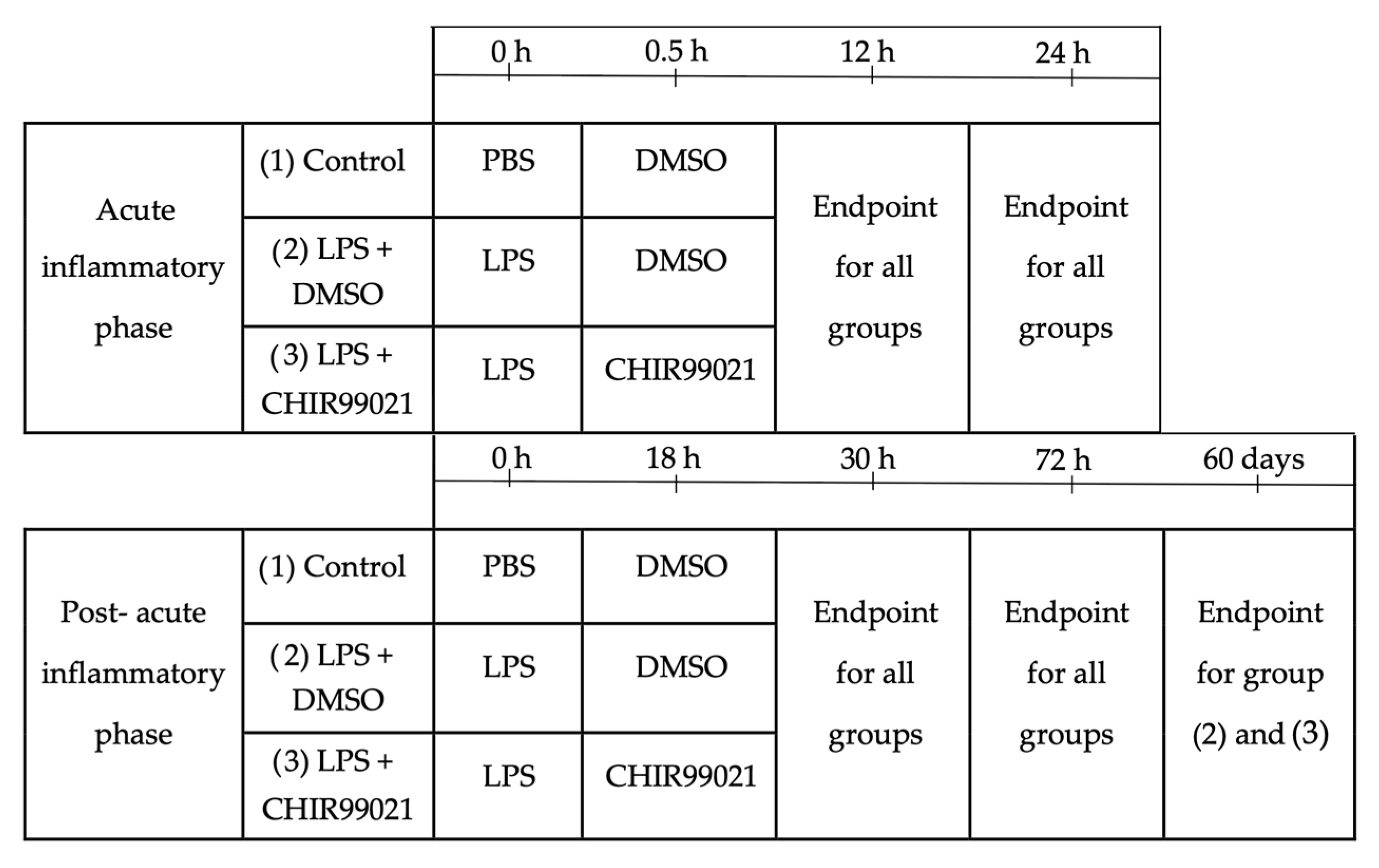
4.2. Experimental Design
4.3. LPS-Induced ALI Mouse Model
4.4. Histological Examination
4.5. Immunofluorescence
4.6. Bronchoalveolar Lavage Fluid Collection
4.7. Flow Cytometry
4.8. ELISA
4.9. Western Blotting
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bellani, G.; Laffey, J.G.; Pham, T.; Fan, E.; Brochard, L.; Esteban, A.; Gattinoni, L.; Van Haren, F.M.P.; Larsson, A.; McAuley, D.F.; et al. Epidemiology, Patterns of Care, and Mortality for Patients with Acute Respiratory Distress Syndrome in Intensive Care Units in 50 Countries. JAMA—J. Am. Med. Assoc. 2016, 315, 788–800. [Google Scholar] [CrossRef] [PubMed]
- Matthay, M.A.; Zemans, R.L.; Zimmerman, G.A.; Arabi, Y.M.; Beitler, J.R.; Mercat, A.; Herridge, M.; Randolph, A.G.; Calfee, C.S. Acute Respiratory Distress Syndrome. Nat. Rev. Dis. Prim. 2018, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.; Rubenfeld, G.D. Fifty Years of Research in Ards the Epidemiology of Acute Respiratory Distress Syndrome a 50th Birthday Review. Am. J. Respir. Crit. Care Med. 2017, 195, 860–870. [Google Scholar] [CrossRef] [PubMed]
- de Prost, N.; Pham, T.; Carteaux, G.; Dessap, A.M.; Brun-Buisson, C.; Fan, E.; Bellani, G.; Laffey, J.; Mercat, A.; Brochard, L.; et al. Etiologies, Diagnostic Work-up and Outcomes of Acute Respiratory Distress Syndrome with No Common Risk Factor: A Prospective Multicenter Study. Ann. Intensive Care 2017, 7, 69. [Google Scholar] [CrossRef]
- Yang, C.Y.; Chen, C.S.; Yiang, G.T.; Cheng, Y.L.; Yong, S.B.; Wu, M.Y.; Li, C.J. New Insights into the Immune Molecular Regulation of the Pathogenesis of Acute Respiratory Distress Syndrome. Int. J. Mol. Sci. 2018, 19, 588. [Google Scholar] [CrossRef]
- Confalonieri, M.; Salton, F.; Fabiano, F. Acute Respiratory Distress Syndrome. Eur. Respir. Rev. 2017, 26, 160116. [Google Scholar] [CrossRef]
- Tsushima, K.; King, L.S.; Aggarwal, N.R.; De Gorordo, A.; D’Alessio, F.R.; Kubo, K. Acute Lung Injury Review. Intern. Med. 2009, 48, 621–630. [Google Scholar] [CrossRef]
- Aspal, M.; Zemans, R.L. Mechanisms of ATII-to-ATI Cell Differentiation during Lung Regeneration. Int. J. Mol. Sci. 2020, 21, 3188. [Google Scholar] [CrossRef]
- Bos, L.D.; Martin-Loeches, I.; Schultz, M.J. ARDS: Challenges in Patient Care and Frontiers in Research. Eur. Respir. Rev. 2018, 27, 170107. [Google Scholar] [CrossRef]
- Bick, E. The 3 R’s of Lung, Health and Disease. Autom. Gramm. Anal. 2000, 121, 505. [Google Scholar] [CrossRef]
- Hogan, B.L.M.; Barkauskas, C.E.; Chapman, H.A.; Epstein, J.A.; Jain, R.; Hsia, C.C.W.; Niklason, L.; Calle, E.; Le, A.; Randell, S.H.; et al. Repair and Regeneration of the Respiratory System: Complexity, Plasticity, and Mechanisms of Lung Stem Cell Function. Cell Stem Cell 2014, 15, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Desai, T.J.; Brownfield, D.G.; Krasnow, M.A. Alveolar Progenitor and Stem Cells in Lung Development, Renewal and Cancer. Nature 2014, 507, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.B.; Peng, T.; Zepp, J.A.; Snitow, M.; Vincent, T.L.; Penkala, I.J.; Cui, Z.; Herriges, M.J.; Morley, M.P.; Zhou, S.; et al. Emergence of a Wave of Wnt Signaling That Regulates Lung Alveologenesis by Controlling Epithelial Self-Renewal and Differentiation. Cell Rep. 2016, 17, 2312–2325. [Google Scholar] [CrossRef] [PubMed]
- Tata, P.R.; Mou, H.; Pardo-Saganta, A.; Zhao, R.; Prabhu, M.; Law, B.M.; Vinarsky, V.; Cho, J.L.; Breton, S.; Sahay, A.; et al. Dedifferentiation of Committed Epithelial Cells into Stem Cells In Vivo. Nature 2013, 503, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Rawlins, E.L.; Okubo, T.; Xue, Y.; Brass, D.M.; Auten, R.L.; Hasegawa, H.; Wang, F.; Hogan, B.L.M. The Role of Scgb1a1+ Clara Cells in the Long-Term Maintenance and Repair of Lung Airway, but Not Alveolar, Epithelium. Cell Stem Cell 2009, 4, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.F.B.; Jackson, E.L.; Woolfenden, A.E.; Lawrence, S.; Babar, I.; Vogel, S.; Crowley, D.; Bronson, R.T.; Jacks, T. Identification of Bronchioalveolar Stem Cells in Normal Lung and Lung Cancer. Cell 2005, 121, 823–835. [Google Scholar] [CrossRef]
- Jain, R.; Barkauskas, C.E.; Takeda, N.; Bowie, E.J.; Aghajanian, H.; Wang, Q.; Padmanabhan, A.; Manderfield, L.J.; Gupta, M.; Li, D.; et al. Plasticity of Hopx+ Type I Alveolar Cells to Regenerate Type II Cells in the Lung. Nat. Commun. 2015, 6, 6727. [Google Scholar] [CrossRef]
- McCauley, K.B.; Hawkins, F.; Serra, M.; Thomas, D.C.; Jacob, A.; Kotton, D.N. Efficient Derivation of Functional Human Airway Epithelium from Pluripotent Stem Cells via Temporal Regulation of Wnt Signaling. Cell Stem Cell 2017, 20, 844–857.e6. [Google Scholar] [CrossRef]
- de Carvalho, A.L.R.T.; Liu, H.Y.; Chen, Y.W.; Porotto, M.; Moscona, A.; Snoeck, H.W. The In Vitro Multilineage Differentiation and Maturation of Lung and Airway Cells from Human Pluripotent Stem Cell–Derived Lung Progenitors in 3D. Nat. Protoc. 2021, 16, 1802–1829. [Google Scholar] [CrossRef]
- de Carvalho, A.L.R.T.; Strikoudis, A.; Liu, H.-Y.; Chen, Y.-W.; Dantas, T.J.; Vallee, R.B.; Correia-Pinto, J.; Snoeck, H.-W. Glycogen Synthase Kinase 3 Induces Multilineage Maturation of Human Pluripotent Stem Cell-Derived Lung Progenitors in 3D Culture. Development 2018, 146, dev.171652. [Google Scholar] [CrossRef]
- Jacob, A.; Morley, M.; Hawkins, F.; McCauley, K.B.; Jean, J.C.; Heins, H.; Na, C.-L.; Weaver, T.E.; Vedaie, M.; Hurley, K.; et al. Differentiation of Human Pluripotent Stem Cells into Functional Lung Alveolar Epithelial Cells. Cell Stem Cell 2017, 21, 472–488.e10. [Google Scholar] [CrossRef] [PubMed]
- Force, T.; Woodgett, J.R. Unique and Overlapping Functions of GSK-3 Isoforms in Cell Differentiation and Proliferation and Cardiovascular Development. J. Biol. Chem. 2009, 284, 9643–9647. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhang, Y.; Bersenev, A.; O’Brien, W.T.; Tong, W.; Emerson, S.G.; Klein, P.S. Pivotal Role for Glycogen Synthase Kinase-3 in Hematopoietic Stem Cell Homeostasis in Mice. J. Clin. Investig. 2009, 119, 3519–3529. [Google Scholar] [CrossRef]
- Hoeflich, K.P.; Luo, J.; Rubie, E.A.; Tsao, M.-S.; Jin, O.; Woodgett, J.R. Requirement for Glycogen Synthase Kinase-3β in Cell Survival and NF-ΚB Activation. Nature 2000, 406, 86–90. [Google Scholar] [CrossRef] [PubMed]
- MacAulay, K.; Doble, B.W.; Patel, S.; Hansotia, T.; Sinclair, E.M.; Drucker, D.J.; Nagy, A.; Woodgett, J.R. Glycogen Synthase Kinase 3α-Specific Regulation of Murine Hepatic Glycogen Metabolism. Cell Metab. 2007, 6, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Kerkela, R.; Kockeritz, L.; Macaulay, K.; Zhou, J.; Doble, B.W.; Beahm, C.; Greytak, S.; Woulfe, K.; Trivedi, C.M.; Woodgett, J.R.; et al. Deletion of GSK-3beta in Mice Leads to Hypertrophic Cardiomyopathy Secondary to Cardiomyoblast Hyperproliferation. J. Clin. Investig. 2008, 118, 3609–3618. [Google Scholar] [CrossRef] [PubMed]
- Hur, E.-M.; Zhou, F.-Q. GSK3 Signalling in Neural Development. Nat. Rev. Neurosci. 2010, 11, 539–551. [Google Scholar] [CrossRef]
- Patel, P.; Woodgett, J.R. Glycogen Synthase Kinase 3: A Kinase for All Pathways? Curr. Top. Dev. Biol. 2017, 123, 277–302. [Google Scholar] [CrossRef]
- Kaidanovich-Beilin, O.; Woodgett, J.R. GSK-3: Functional Insights from Cell Biology and Animal Models. Front. Mol. Neurosci. 2011, 4, 40. [Google Scholar] [CrossRef]
- Beurel, E.; Grieco, S.F.; Jope, R.S. Glycogen Synthase Kinase-3 (GSK3): Regulation, Actions, and Diseases. Pharmacol. Ther. 2015, 148, 114–131. [Google Scholar] [CrossRef]
- Cohen, P.; Frame, S. The Renaissance of GSK3. Nat. Rev. Mol. Cell Biol. 2001, 2, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Gadjeva, M.; Tomczak, M.F.; Zhang, M.; Wang, Y.Y.; Dull, K.; Rogers, A.B.; Erdman, S.E.; Fox, J.G.; Carroll, M.; Horwitz, B.H. A Role for NF-ΚB Subunits P50 and P65 in the Inhibition of Lipopolysaccharide-Induced Shock. J. Immunol. 2004, 173, 5786–5793. [Google Scholar] [CrossRef]
- Ko, R.; Lee, S.Y. Glycogen Synthase Kinase 3β in Toll-like Receptor Signaling. BMB Rep. 2016, 49, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Rehani, K.; Jope, R.S.; Michalek, S.M. Toll-like Receptor—Mediated Cytokine Production Is Differentially Regulated by Glycogen Synthase Kinase 3. Nat. Immunol. 2005, 6, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Park, D.W.; Jiang, S.; Liu, Y.; Siegal, G.P.; Inoki, K.; Abraham, E.; Zmijewski, J.W. GSK3β-Dependent Inhibition of AMPK Potentiates Activation of Neutrophils and Macrophages and Enhances Severity of Acute Lung Injury. Am. J. Physiol.—Lung Cell. Mol. Physiol. 2014, 307, L735–L745. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Yang, X.; Zhao, K.; Zhao, L.; Chen, C.; Yu, J. Glycogen Synthase Kinase-3 Beta Inhibitors Protectagainst the Acute Lung Injuries Resulting from Acute Necrotizing Pancreatitis1. Acta Cir. Bras. 2019, 34, e201900609. [Google Scholar] [CrossRef] [PubMed]
- Cinetto, F.; Ceccato, J.; Caputo, I.; Cangiano, D.; Montini, B.; Lunardi, F.; Piazza, M.; Agostini, C.; Calabrese, F.; Semenzato, G.; et al. GSK-3 Inhibition Modulates Metalloproteases in a Model of Lung Inflammation and Fibrosis. Front. Mol. Biosci. 2021, 8, 633054. [Google Scholar] [CrossRef]
- Baarsma, H.A.; Engelbertink, L.H.; van Hees, L.J.; Menzen, M.H.; Meurs, H.; Timens, W.; Postma, D.S.; Kerstjens, H.A.; Gosens, R. Glycogen Synthase Kinase-3 (GSK-3) Regulates TGF-Β₁-Induced Differentiation of Pulmonary Fibroblasts. Br. J. Pharmacol. 2013, 169, 590–603. [Google Scholar] [CrossRef]
- Hoffman, O.; Burns, N.; Vadász, I.; Eltzschig, H.K.; Edwards, M.G.; Vohwinkel, C.U. Detrimental ELAVL-1/HuR-Dependent GSK3β MRNA Stabilization Impairs Resolution in Acute Respiratory Distress Syndrome. PLoS ONE 2017, 12, e0172116. [Google Scholar] [CrossRef]
- Yi, L.; Huang, X.; Guo, F.; Zhou, Z.; Chang, M.; Huan, J. GSK-3beta-Dependent Activation of GEF-H1/ROCK Signaling Promotes LPS-Induced Lung Vascular Endothelial Barrier Dysfunction and Acute Lung Injury. Front. Cell. Infect. Microbiol. 2017, 7, 357. [Google Scholar] [CrossRef]
- Noh, K.T.; Son, K.H.; Jung, I.D.; Kang, H.K.; Hwang, S.A.; Lee, W.S.; You, J.C.; Park, Y.M. Protein Kinase C δ (PKCδ)-Extracellular Signal-Regulated Kinase 1/2 (ERK1/2) Signaling Cascade Regulates Glycogen Synthase Kinase-3 (GSK-3) Inhibition-Mediated Interleukin-10 (IL-10) Expression in Lipopolysaccharide (LPS)-Induced Endotoxemia. J. Biol. Chem. 2012, 287, 14226–14233. [Google Scholar] [CrossRef] [PubMed]
- Vohwinkel, C.U.; Buchackert, Y.; Al-Tamari, H.M.; Mazzocchi, L.C.; Eltzschig, H.K.; Mayer, K.; Morty, R.E.; Herold, S.; Seeger, W.; Pullamsetti, S.S.; et al. Restoration of Megalin-Mediated Clearance of Alveolar Protein as a Novel Therapeutic Approach for Acute Lung Injury. Am. J. Respir. Cell Mol. Biol. 2017, 57, 589–602. [Google Scholar] [CrossRef] [PubMed]
- Wagner, F.F.; Bishop, J.A.; Gale, J.P.; Shi, X.; Walk, M.; Ketterman, J.; Patnaik, D.; Barker, D.; Walpita, D.; Campbell, A.J.; et al. Inhibitors of Glycogen Synthase Kinase 3 with Exquisite Kinome-Wide Selectivity and Their Functional Effects. ACS Chem. Biol. 2016, 11, 1952–1963. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, K.; Obermayer, B.; Hönzke, K.; Fatykhova, D.; Demir, Z.; Löwa, A.; Alves, L.G.T.; Wyler, E.; Lopez-Rodriguez, E.; Mieth, M.; et al. Human Alveolar Progenitors Generate Dual Lineage Bronchioalveolar Organoids. Commun. Biol. 2022, 5, 875. [Google Scholar] [CrossRef] [PubMed]
- Alysandratos, K.D.; Herriges, M.J.; Kotton, D.N. Epithelial Stem and Progenitor Cells in Lung Repair and Regeneration. Annu. Rev. Physiol. 2021, 83, 529–550. [Google Scholar] [CrossRef] [PubMed]
- Asti, C.; Ruggieri, V.; Porzio, S.; Chiusaroli, R.; Melillo, G.; Caselli, G.F. Lipopolysaccharide-Induced Lung Injury in Mice. I. Concomitant Evaluation of Inflammatory Cells and Haemorrhagic Lung Damage. Pulm. Pharmacol. Ther. 2000, 13, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Domscheit, H.; Hegeman, M.A.; Carvalho, N.; Spieth, P.M. Molecular Dynamics of Lipopolysaccharide-Induced Lung Injury in Rodents. Front. Physiol. 2020, 11, 36. [Google Scholar] [CrossRef]
- Galani, V.; Tatsaki, E.; Bai, M.; Kitsoulis, P.; Lekka, M.; Nakos, G.; Kanavaros, P. The Role of Apoptosis in the Pathophysiology of Acute Respiratory Distress Syndrome (ARDS): An up-to-Date Cell-Specific Review. Pathol. Res. Pract. 2010, 206, 145–150. [Google Scholar] [CrossRef]
- Z’Graggen, B.R.; Tornic, J.; Müller-Edenborn, B.; Reyes, L.; Booy, C.; Beck-Schimmer, B. Acute Lung Injury: Apoptosis in Effector and Target Cells of the Upper and Lower Airway Compartment. Clin. Exp. Immunol. 2010, 161, 324–331. [Google Scholar] [CrossRef]
- Cuzzocrea, S.; Crisafulli, C.; Mazzon, E.; Esposito, E.; Muià, C.; Abdelrahman, M.; Di Paola, R.; Thiemermann, C. Inhibition of Glycogen Synthase Kinase-3β Attenuates the Development of Carrageenan-Induced Lung Injury in Mice. Br. J. Pharmacol. 2006, 149, 687–702. [Google Scholar] [CrossRef]
- Xaus, J.; Comalada, M.; Valledor, A.F.; Lloberas, J.; López-Soriano, F.; Argilés, J.M.; Bogdan, C.; Celada, A. LPS Induces Apoptosis in Macrophages Mostly through the Autocrine Production of TNF-α. Blood 2000, 95, 3823–3831. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, Y.; Hashimoto, S.; Mizuta, N.; Kobayashi, A.; Kooguchi, K.; Fujiwara, I.; Nakajima, H. Fas/FasL-Dependent Apoptosis of Alveolar Cells after Lipopolysaccharide-Induced Lung Injury in Mice. Am. J. Respir. Crit. Care Med. 2001, 163, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, M.; Kuwano, K.; Hagimoto, N.; Matsuba, T.; Kunitake, R.; Tanaka, T.; Maeyama, T.; Hara, N. Protection from Lethal Apoptosis in Lipopolysaccharide-Induced Acute Lung Injury in Mice by a Caspase Inhibitor. Am. J. Pathol. 2000, 157, 597–603. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; He, S.; Lian, S.; Shen, Y.; Jiang, W.; Zhou, L.; Zhou, L.; Zhang, X. The Wnt/β-Catenin Pathway Regulates Inflammation and Apoptosis in Ventilator-Induced Lung Injury. Biosci. Rep. 2023, 43, BSR20222429. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Liu, G.; Zeng, Y.; Zhu, J.; Liu, Z.; Jiang, J.; Huang, J. Glycogen Synthase Kinase-3β Inhibitor Reduces LPS-Induced Acute Lung Injury in Mice. Mol. Med. Rep. 2017, 16, 6715–6721. [Google Scholar] [CrossRef][Green Version]
- Adhikari, N.K.; Burns, K.E.; Meade, M.O.; Ratnapalan, M. Pharmacologic Therapies for Adults with Acute Lung Injury and Acute Respiratory Distress Syndrome. Cochrane Database Syst. Rev. 2004, 2004, CD004477. [Google Scholar] [CrossRef]
- Baudouin, S.V. Manipulation of Inflammation in ARDS: Achievable Goal or Distant Target? Thorax 2006, 61, 464–465. [Google Scholar] [CrossRef][Green Version]
- Burnham, E.L.; Janssen, W.J.; Riches, D.W.; Moss, M.; Downey, G.P. The Fibroproliferative Response in Acute Respiratory Distress Syndrome: Mechanisms and Clinical Significance. Eur. Respir. J. 2014, 43, 276–285. [Google Scholar] [CrossRef]
- Treutlein, B.; Brownfield, D.G.; Wu, A.R.; Neff, N.F.; Mantalas, G.L.; Espinoza, F.H.; Desai, T.J.; Krasnow, M.A.; Quake, S.R. Reconstructing Lineage Hierarchies of the Distal Lung Epithelium Using Single-Cell RNA-Seq. Nature 2014, 509, 371–375. [Google Scholar] [CrossRef]
- Barkauskas, C.E.; Cronce, M.J.; Rackley, C.R.; Bowie, E.J.; Keene, D.R.; Stripp, B.R.; Randell, S.H.; Noble, P.W.; Hogan, B.L.M. Type 2 Alveolar Cells Are Stem Cells in Adult Lung. J. Clin. Investig. 2013, 123, 3025–3036. [Google Scholar] [CrossRef]
- Jansing, N.L.; McClendon, J.; Henson, P.M.; Tuder, R.M.; Hyde, D.M.; Zemans, R.L. Unbiased Quantitation of Alveolar Type II to Alveolar Type i Cell Transdifferentiation during Repair after Lung Injury in Mice. Am. J. Respir. Cell Mol. Biol. 2017, 57, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Xu, C.; Jiang, L.; Zhu, X. Pulmonary Endogenous Progenitor Stem Cell Subpopulation: Physiology, Pathogenesis, and Progress. J. Intensive Med. 2023, 3, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Rock, J.R.; Hogan, B.L.M. Epithelial Progenitor Cells in Lung Development, Maintenance, Repair, and Disease. Annu. Rev. Cell Dev. Biol. 2011, 27, 493–512. [Google Scholar] [CrossRef]
- Matute-Bello, G.; Frevert, C.W.; Martin, T.R. Animal Models of Acute Lung Injury. Am. J. Physiol.—Lung Cell. Mol. Physiol. 2008, 295, L379–L399. [Google Scholar] [CrossRef] [PubMed]
- Matute-Bello, G.; Downey, G.; Moore, B.B.; Groshong, S.D.; Matthay, M.A.; Slutsky, A.S.; Kuebler, W.M. An Official American Thoracic Society Workshop Report: Features and Measurements of Experimental Acute Lung Injury in Animals. Am. J. Respir. Cell Mol. Biol. 2011, 44, 725–738. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhu, J.; Jiang, Y.; Xu, C.; Lv, Q.; Yu, D.; Shi, K.; Ruan, Z.; Wang, Y. SU5416 Attenuated Lipopolysaccharide-Induced Acute Lung Injury in Mice by Modulating Properties of Vascular Endothelial Cells. Drug Des. Dev. Ther. 2019, 13, 1763–1772. [Google Scholar] [CrossRef]
- Mccubrey, J.A.; Steelman, L.S.; Bertrand, F.E.; Davis, N.M.; Sokolosky, M.; Abrams, S.L.; Montalto, G.; D’Assoro, A.B.; Libra, M.; Nicoletti, F.; et al. GSK-3 as Potential Target for Therapeutic Intervention in Cancer. Oncotarget 2014, 5, 2881–2911. [Google Scholar] [CrossRef]
- Viatour, P.; Dejardin, E.; Warnier, M.; Lair, F.; Claudio, E.; Bureau, F.; Marine, J.C.; Merville, M.P.; Maurer, U.; Green, D.; et al. GSK3-Mediated BCL-3 Phosphorylation Modulates Its Degradation and Its Oncogenicity. Mol. Cell 2004, 16, 35–45. [Google Scholar] [CrossRef]
- Shaulian, E.; Karin, M. AP-1 in Cell Proliferation and Survival. Oncogene 2001, 20, 2390–2400. [Google Scholar] [CrossRef]
- MacRedmond, R.; Singhera, G.K.; Dorscheid, D.R. Erythropoietin Inhibits Respiratory Epithelial Cell Apoptosis in a Model of Acute Lung Injury. Eur. Respir. J. 2009, 33, 1403–1414. [Google Scholar] [CrossRef]
- Bhavsar, T.M.; Cantor, J.O.; Patel, S.N.; Lau-Cam, C.A. Attenuating Effect of Taurine on Lipopolysaccharide-Induced Acute Lung Injury in Hamsters. Pharmacol. Res. 2009, 60, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Gurrieri, C.; Piazza, F.; Gnoato, M.; Montini, B.; Biasutto, L.; Gattazzo, C.; Brunetta, E.; Cabrelle, A.; Cinetto, F.; Niero, R.; et al. 3-(2,4-Dichlorophenyl)-4-(1-Methyl-1 H -Indol-3-Yl)-1 H -Pyrrole-2,5-Dione (SB216763), a Glycogen Synthase Kinase-3 Inhibitor, Displays Therapeutic Properties in a Mouse Model of Pulmonary Inflammation and Fibrosis. J. Pharmacol. Exp. Ther. 2010, 332, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, H.; Zeng, M.; He, W.; Li, M.; Huang, X.; Deng, D.Y.B.; Wu, J. Bone Marrow Mesenchymal Stem Cells Protect Alveolar Macrophages from Lipopolysaccharide-induced Apoptosis Partially by Inhibiting the Wnt/Β-catenin Pathway. Cell Biol. Int. 2015, 39, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shi, J.; Li, P.; Guo, X.; Wang, T.; Liu, A. Genipin Attenuates Hyperoxia-Induced Lung Injury and Pulmonary Hypertension via Targeting Glycogen Synthase Kinase-3 β in Neonatal Rats. Nutrition 2019, 57, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Cuzzocrea, S.; Genovese, T.; Mazzon, E.; Esposito, E.; Muià, C.; Abdelrahman, M.; Di Paola, R.; Bramanti, P.; Thiemermann, C. Glycogen Synthase Kinase-3β Inhibition Attenuates the Development of Bleomycin-Induced Lung Injury. Int. J. Immunopathol. Pharmacol. 2007, 20, 619–630. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, P.; Cui, A.; Ye, D.-Y.; Xiang, M.; Chu, Y. Discovery and Anti-Inflammatory Evaluation of Benzothiazepinones (BTZs) as Novel Non-ATP Competitive Inhibitors of Glycogen Synthase Kinase-3β (GSK-3β). Bioorg. Med. Chem. 2018, 26, 5479–5493. [Google Scholar] [CrossRef]
- Li, C.; Zhang, S.; Lu, Y.; Zhang, Y.; Wang, E.; Cui, Z. The Roles of Notch3 on the Cell Proliferation and Apoptosis Induced by CHIR99021 in NSCLC Cell Lines: A Functional Link between Wnt and Notch Signaling Pathways. PLoS ONE 2013, 8, e84659. [Google Scholar] [CrossRef]
- Alvarez-Palomo, B.; Sanchez-Lopez, L.I.; Moodley, Y.; Edel, M.J.; Serrano-Mollar, A. Induced Pluripotent Stem Cell-Derived Lung Alveolar Epithelial Type II Cells Reduce Damage in Bleomycin-Induced Lung Fibrosis. Stem Cell Res. Ther. 2020, 11, 213. [Google Scholar] [CrossRef]
- Wang, X.; Wei, L.; Cramer, J.M.; Leibowitz, B.J.; Judge, C.; Epperly, M.; Greenberger, J.; Wang, F.; Li, L.; Stelzner, M.G.; et al. Pharmacologically Blocking P53-Dependent Apoptosis Protects Intestinal Stem Cells and Mice from Radiation. Sci. Rep. 2015, 5, srep08566. [Google Scholar] [CrossRef]
- Pachenari, N.; Kiani, S.; Javan, M. Inhibition of Glycogen Synthase Kinase 3 Increased Subventricular Zone Stem Cells Proliferation. Biomed. Pharmacother. 2017, 93, 1074–1082. [Google Scholar] [CrossRef]
- Willert, K.; Nusse, R. Wnt Proteins. Cold Spring Harb. Perspect. Biol. 2012, 4, a007864. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, N.; Toba, H.; Miyoshi, K.; Sakamoto, S.; Matsumoto, D.; Takashima, M.; Aoyama, M.; Inoue, S.; Morimoto, M.; Nishino, T.; et al. Bronchioalveolar Stem Cells Derived from Mouse-Induced Pluripotent Stem Cells Promote Airway Epithelium Regeneration. Stem Cell Res. Ther. 2020, 11, 430. [Google Scholar] [CrossRef] [PubMed]
- Salwig, I.; Spitznagel, B.; Vazquez-Armendariz, A.I.; Khalooghi, K.; Guenther, S.; Herold, S.; Szibor, M.; Braun, T. Bronchioalveolar Stem Cells Are a Main Source for Regeneration of Distal Lung Epithelia In Vivo. EMBO J. 2019, 38, e102099. [Google Scholar] [CrossRef]
- Liu, K.; Tang, M.; Liu, Q.; Han, X.; Jin, H.; Zhu, H.; Li, Y.; He, L.; Ji, H.; Zhou, B. Bi-Directional Differentiation of Single Bronchioalveolar Stem Cells during Lung Repair. Cell Discov. 2020, 6, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Haviland, D.L.; Burns, A.R.; Zsigmond, E.; Wetsel, R.A. A Pure Population of Lung Alveolar Epithelial Type II Cells Derived from Human Embryonic Stem Cells. Proc. Natl. Acad. Sci. USA 2007, 104, 4449–4454. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.; Liu, Y. Function of Epithelial Stem Cell in the Repair of Alveolar Injury. Stem Cell Res. Ther. 2022, 13, 170. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Park, J.-E.; Tsagkogeorga, G.; Yanagita, M.; Koo, B.-K.; Han, N.; Lee, J.-H. Inflammatory Signals Induce AT2 Cell-Derived Damage-Associated Transient Progenitors That Mediate Alveolar Regeneration. Cell Stem Cell 2020, 27, 366–382.e7. [Google Scholar] [CrossRef]
- Katsura, H.; Kobayashi, Y.; Tata, P.R.; Hogan, B.L.M. IL-1 and TNFα Contribute to the Inflammatory Niche to Enhance Alveolar Regeneration. Stem Cell Rep. 2019, 12, 657–666. [Google Scholar] [CrossRef]
- Liberti, D.C.; Kremp, M.M.; Liberti, W.A.; Penkala, I.J.; Li, S.; Zhou, S.; Morrisey, E.E. Alveolar Epithelial Cell Fate Is Maintained in a Spatially Restricted Manner to Promote Lung Regeneration after Acute Injury. Cell Rep. 2021, 35, 109092. [Google Scholar] [CrossRef]
- Chung, M.-I.; Bujnis, M.; Barkauskas, C.E.; Kobayashi, Y.; Hogan, B.L.M. Niche-Mediated BMP/SMAD Signaling Regulates Lung Alveolar Stem Cell Proliferation and Differentiation. Development 2018, 145, dev163014. [Google Scholar] [CrossRef]
- Finn, J.; Sottoriva, K.; Pajcini, K.V.; Kitajewski, J.K.; Chen, C.; Zhang, W.; Malik, A.B.; Liu, Y. Dlk1-Mediated Temporal Regulation of Notch Signaling Is Required for Differentiation of Alveolar Type II to Type I Cells during Repair. Cell Rep. 2019, 26, 2942–2954.e5. [Google Scholar] [CrossRef] [PubMed]
- Nabhan, A.N.; Brownfield, D.G.; Harbury, P.B.; Krasnow, M.A.; Desai, T.J. Single-Cell Wnt Signaling Niches Maintain Stemness of Alveolar Type 2 Cells. Science 2018, 359, 1118–1123. [Google Scholar] [CrossRef] [PubMed]
- LaCanna, R.; Liccardo, D.; Zhang, P.; Tragesser, L.; Wang, Y.; Cao, T.; Chapman, H.A.; Morrisey, E.E.; Shen, H.; Koch, W.J.; et al. Yap/Taz Regulate Alveolar Regeneration and Resolution of Lung Inflammation. J. Clin. Investig. 2019, 129, 2107–2122. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, Z.; Huang, H.; Li, J.; Wang, Z.; Yu, Y.; Zhang, C.; Li, J.; Dai, H.; Wang, F.; et al. Pulmonary Alveolar Type I Cell Population Consists of Two Distinct Subtypes That Differ in Cell Fate. Proc. Natl. Acad. Sci. USA 2018, 115, 2407–2412. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.J.; Cabral, L.J.; Stephens, R.J.; Freeman, G. Transformation of Alveolar Type 2 Cells to Type 1 Cells Following Exposure to NO2. Exp. Mol. Pathol. 1975, 22, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Mizuno, T.; Sridharan, A.; Du, Y.; Guo, M.; Tang, J.; Wikenheiser-Brokamp, K.A.; Perl, A.-K.T.; Funari, V.A.; Gokey, J.J.; et al. Single-Cell RNA Sequencing Identifies Diverse Roles of Epithelial Cells in Idiopathic Pulmonary Fibrosis. JCI Insight 2016, 1, e90558. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Deng, J.-S.; Huang, W.-C.; Jiang, W.-P.; Huang, G.-J. Attenuation of Lipopolysaccharide-Induced Acute Lung Injury by Hispolon in Mice, Through Regulating the TLR4/PI3K/Akt/MTOR and Keap1/Nrf2/HO-1 Pathways, and Suppressing Oxidative Stress-Mediated ER Stress-Induced Apoptosis and Autophagy. Nutrients 2020, 12, 1742. [Google Scholar] [CrossRef]
- Krynytska, I.; Marushchak, M.; Birchenko, I.; Dovgalyuk, A.; Tokarskyy, O. COVID-19-Associated Acute Respiratory Distress Syndrome versus Classical Acute Respiratory Distress Syndrome (a Narrative Review). Iran. J. Microbiol. 2021, 13, 737–747. [Google Scholar] [CrossRef]
- Rana, A.K.; Rahmatkar, S.N.; Kumar, A.; Singh, D. Glycogen Synthase Kinase-3: A Putative Target to Combat Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Pandemic. Cytokine Growth Factor Rev. 2021, 58, 92–101. [Google Scholar] [CrossRef]
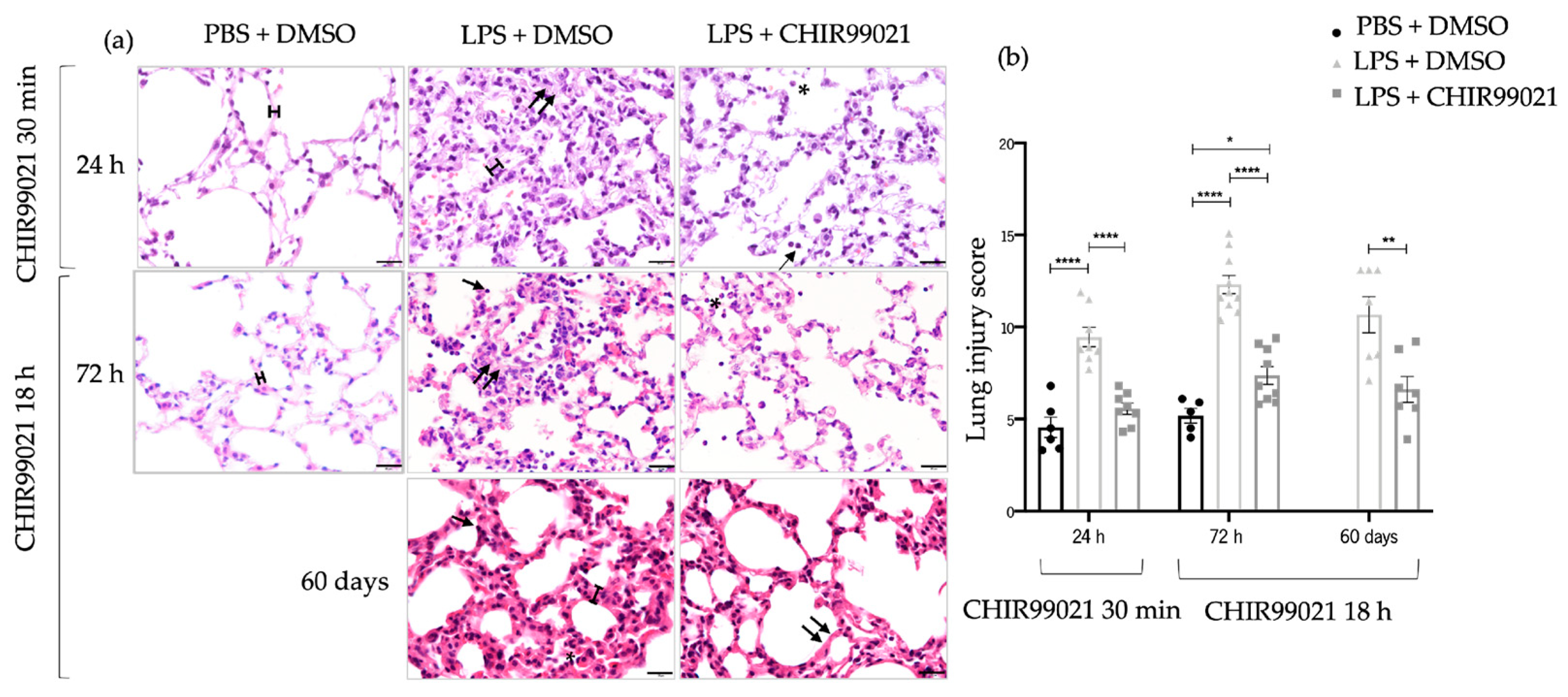
| Parameter | Score |
|---|---|
| Alveoli congestion | 0: Minimal damage; 1: Mild damage 2: Moderate damage; 3: Severe damage; 4: Maximal damage |
| Immune infiltration | 0: Minimal damage; 1: Mild damage 2: Moderate damage; 3: Severe damage; 4: Maximal damage |
| Number of macrophages | 0: <5; 1: 5–10; 2: 10–20; 3: 20–50; 4: >50 |
| Number of neutrophils | 0: <5; 1: 5–10; 2: 10–20; 3: 20–50; 4: >50 |
| Hyaline membranes | 0: Minimal damage; 1: Mild damage 2: Moderate damage; 3: Severe damage; 4: Maximal damage |
| Alveolar wall thickness | 0: equal to control; 1: 2x; 2: 2x–4x; 3: >4x |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, R.; Barbosa-Matos, C.; Borges-Pereira, C.; Carvalho, A.L.R.T.d.; Costa, S. Glycogen Synthase Kinase-3 Inhibition by CHIR99021 Promotes Alveolar Epithelial Cell Proliferation and Lung Regeneration in the Lipopolysaccharide-Induced Acute Lung Injury Mouse Model. Int. J. Mol. Sci. 2024, 25, 1279. https://doi.org/10.3390/ijms25021279
Fernandes R, Barbosa-Matos C, Borges-Pereira C, Carvalho ALRTd, Costa S. Glycogen Synthase Kinase-3 Inhibition by CHIR99021 Promotes Alveolar Epithelial Cell Proliferation and Lung Regeneration in the Lipopolysaccharide-Induced Acute Lung Injury Mouse Model. International Journal of Molecular Sciences. 2024; 25(2):1279. https://doi.org/10.3390/ijms25021279
Chicago/Turabian StyleFernandes, Raquel, Catarina Barbosa-Matos, Caroline Borges-Pereira, Ana Luísa Rodrigues Toste de Carvalho, and Sandra Costa. 2024. "Glycogen Synthase Kinase-3 Inhibition by CHIR99021 Promotes Alveolar Epithelial Cell Proliferation and Lung Regeneration in the Lipopolysaccharide-Induced Acute Lung Injury Mouse Model" International Journal of Molecular Sciences 25, no. 2: 1279. https://doi.org/10.3390/ijms25021279
APA StyleFernandes, R., Barbosa-Matos, C., Borges-Pereira, C., Carvalho, A. L. R. T. d., & Costa, S. (2024). Glycogen Synthase Kinase-3 Inhibition by CHIR99021 Promotes Alveolar Epithelial Cell Proliferation and Lung Regeneration in the Lipopolysaccharide-Induced Acute Lung Injury Mouse Model. International Journal of Molecular Sciences, 25(2), 1279. https://doi.org/10.3390/ijms25021279





