Polyphenols as the Main Compounds Influencing the Antioxidant Effect of Honey—A Review
Abstract
1. Introduction
2. Chemical Composition of Honey
3. Methods of Review
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DIRECTIVE2014/63/EU of the European Parliament and of the Council of 15 May 2014 Amending Council Directive 2001/110/EC Relating to Honey, Official Journal of the European Union L 164/1. 2014.
- Laaroussi, H.; Bouddine, T.; Bakour, M.; Ousaaid, D.; Lyoussi, B. Physicochemical Properties, Mineral Content, Antioxidant Activities, and Microbiological Quality of Bupleurum spinosum Gouan Honey from the Middle Atlas in Morocco. J. Food Qual. 2020, 2020, 7609454. [Google Scholar] [CrossRef]
- Bobis, O.; Moise, A.R.; Ballesteros, I.; Reyes, E.S.; Durán, S.S.; Sánchez-Sánchez, J.; Cruz-Quintana, S.; Giampieri, F.; Battino, M.; Alvarez-Suarez, J.M. Eucalyptus Honey: Quality Parameters, Chemical Composition and Health-Promoting Properties. Food Chem. 2020, 325, 126870. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, S.; Jurendic, T.; Sieber, R.; Gallmann, P. Honey for Nutrition and Health: A Review. J. Apic. Res. 2013, 27, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Boussaid, A.; Chouaibi, M.; Rezig, L.; Hellal, R.; Donsì, F.; Ferrari, G.; Hamdi, S. Physicochemical and Bioactive Properties of Six Honey Samples from Various Floral Origins from Tunisia. Arab. J. Chem. 2018, 11, 265–274. [Google Scholar] [CrossRef]
- Cheung, Y.; Meenu, M.; Yu, X.; Xu, B. Phenolic Acids and Flavonoids Profiles of Commercial Honey from Different Floral Sources and Geographic Sources. Int. J. Food Prop. 2019, 22, 290–308. [Google Scholar] [CrossRef]
- Erban, T.; Shcherbachenko, E.; Talacko, P.; Harant, K. The Unique Protein Composition of Honey Revealed by Comprehensive Proteomic Analysis: Allergens, Venom-like Proteins, Antibacterial Properties, Royal Jelly Proteins, Serine Proteases, and Their Inhibitors. J. Nat. Prod. 2019, 82, 1217–1226. [Google Scholar] [CrossRef]
- Marić, A.; Jovanov, P.; Sakać, M.; Novaković, A.; Hadnadev, M.; Pezo, L.; Mandić, A.; Milićewić, N.; Durović, A.; Gadžurić, S. A comprehensive study of parameters correlated with honey health benefits. RSC Adv. 2021, 11, 12434. [Google Scholar] [CrossRef]
- Pauliuc, D.; Ciursă, P.; Ropciuc, S.; Dranca, F.; Oroian, M. Physicochemical Parameters Prediction and Authentication of Different Monofloral Honeys Based on FTIR Spectra. J. Food Compos. Anal. 2021, 102, 104021. [Google Scholar] [CrossRef]
- Sakać, M.B.; Jovanov, P.T.; Marić, A.Z.; Pezo, L.L.; Kevrešan, Ž.S.; Novaković, A.R.; Nedeljković, N.M. Physicochemical Properties and Mineral Content of Honey Samples from Vojvodina (Republic of Serbia). Food Chem. 2019, 276, 15–21. [Google Scholar] [CrossRef]
- Oddo, L.P.; Piro, R.; Bruneau, É.; Guyot-Declerck, C.; Ivanov, T.; Piskulová, J.; Flamini, C.; Lheritier, J.; Morlot, M.; Russmann, H. Main European unifloral honeys: Descriptive sheets. Apidologie 2004, 1, 38–81. [Google Scholar] [CrossRef]
- Bogdanov, S. Physical Properties of Honey. Book of Honey; Chapter 4 Bee Product Science. 2009. Available online: www.bee-hexagon.net (accessed on 20 July 2024).
- Crăciun, M.E.; Pârvulescu, O.C.; Donise, A.C.; Dobre, T.; Stanciu, D.R. Characterization and classification of Romanian acacia honey based on its physicochemical parameters and chemometrics. Sci. Rep. 2020, 10, 20690. [Google Scholar] [CrossRef] [PubMed]
- Manzanares, A.B.; García, Z.H.; Galdón, B.R.; Rodríguez, E.R.; Romero, C.D. Physicochemical characteristics of minor monofloral honeys from Tenerife, Spain. LWT—Food Sci. Technol. 2014, 55, 572–578. [Google Scholar] [CrossRef]
- Gündoğdu, E.; Çakmakç, S.; Şat, İ.G. An Overview of Honey: Its Composition, Nutritional and Functional Properties. J. Food Sci. Eng. 2019, 9, 10–14. [Google Scholar] [CrossRef]
- El Sohaimy, S.A.; Masry, S.H.D.; Shehata, M.G. Physicochemical characteristics of honey from different origins. Ann. Agric. Sci. 2015, 60, 279–287. [Google Scholar] [CrossRef]
- Iglesias, M.T.; Martian-Alvarez, P.J.; Polo, M.C.; Lorenzo, C.; Gonzalez, M.; Pueyo, E.N. Changes in the free amino acid contents of honeys during storage at ambient temperature. J. Agric. Food Chem. 2006, 54, 9099–9104. [Google Scholar] [CrossRef]
- Rybak-Chmielewska, H. Changes in the carbohydrate composition of honey undergoing during storage. J. Apic. Sci. 2007, 51, 39–48. [Google Scholar]
- Bonté, F.; Desmoulière, A. Le miel: Origine et composition. Actual. Pharm. 2013, 531, 18–21. [Google Scholar] [CrossRef]
- Janiszewska, K.; Aniołowska, M.; Nowakowski, P. Free amino acids content of honeys from Poland. Pol. J. Food Nutr. Sci. 2012, 62, 85–89. [Google Scholar] [CrossRef]
- Hermosi’n, I.; Chicón, R.M.; Dolores Cabezudo, M. Free amino acid composition and botanical origin of honey. Food Chem. 2002, 83, 263–268. [Google Scholar] [CrossRef]
- Pita-Calvo, C.; Vázquez, M. Differences between honeydew and blossom honeys: A review. Trends Food Sci. Technol. 2017, 59, 79–87. [Google Scholar] [CrossRef]
- Won, S.; Lee, D.; Ko, S.H.; Kim, J.; Rhee, H. Honey major protein characterization and its application to adulteration detection. Food Res. Int. 2008, 41, 952–956. [Google Scholar] [CrossRef]
- Sak-Bosnar, M.; Sakac, N. Direct potentiometric determination of diastase activity in honey. Food Chem. 2012, 135, 827–831. [Google Scholar] [CrossRef] [PubMed]
- Villacrés-Granda, I.; Coello, D.; Proaño, A.; Ballesteros, I.; Roubik, D.W.; Jijón, G.; Granda-Albuja, G.; Granda-Albuja, S.; Abreu-Naranjo, R.; Maza, F.; et al. Honey quality parameters, chemical composition and antimicrobial activity in twelve Ecuadorian stingless bees (Apidae: Apinae: Meliponini) tested against multiresistant human pathogens. LWT—Food Sci. Technol. 2021, 140, 110737. [Google Scholar] [CrossRef]
- Won, S.A.; Li, C.; Kim, J.; Rhee, H. Immunological characterization of honey major protein and its application. Food Chem. 2009, 113, 1334–1338. [Google Scholar] [CrossRef]
- Da Silva, P.M.; Gauche, C.; Gonzaga, L.V.; Oliveira Costa, A.C.; Fett, R. Honey: Chemical composition, stability and authenticity. Food Chem. 2016, 196, 309–323. [Google Scholar] [CrossRef]
- Wang, J.; Li, Q.X. Chemical Composition, Characterization, and Differentiation of Honey Botanical and Geographical Origins. In Advances in Food Nutrition Research; Taylor, S.L., Ed.; Academic Press: Cambridge, MA, USA, 2011; Volume 62, pp. 89–137. [Google Scholar] [CrossRef]
- Madejczyk, M.; Baralkiewicz, D. Characterization of Polish rape and honeydew honey according to their mineral contents using ICP-MS and F-AAS/AES. Anal. Chim. Acta 2008, 617, 11–17. [Google Scholar] [CrossRef]
- Alqarni, A.S.; Owayss, A.A.; Mahmoud, A.A. Mineral content and physical properties of local and imported honeys in Saudi Arabia. J. Saudi Chem. Soc. 2012, 5, 618–625. [Google Scholar] [CrossRef]
- Moreira, R.F.A.; Maria, C.A.B.; Pietroluongo, M.; Trugo, L.C. Chemical changes in the non-volatile fraction of Brazilian honeys during storage under tropical conditions. Food Chem. 2007, 104, 1236–1241. [Google Scholar] [CrossRef]
- Zammit Young, G.-W.; Blundell, R. A review on the phytochemical composition and health applications of honey. Heliyon 2023, 9, e12507. [Google Scholar] [CrossRef]
- Karabagias, I.K.; Badeka, A.; Kontakos, S.; Karabournioti, S.; Kontominas, M.G. Characterisation and classification of Greek pine honeys according to their geographical origin based on volatiles, physicochemical parameters and chemometrics. Food Chem. 2014, 146, 548–557. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Doberšek, U.; Jamnik, M.; Golob, T. Evaluation of the phenolic content, antioxidant activity and colour of Slovenian honey. Food Chem. 2007, 105, 822–828. [Google Scholar] [CrossRef]
- Mato, I.S.; Huidobro, J.F.; Simal-Lozano, J.S.; Sancho, M.T. Rapid determination of nonaromatic organic acids in honey by capillary zone electrophoresis with direct ultraviolet detection. J. Agric. Food Chem. 2006, 54, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.; Antunes, M.; Faleiro, M. Honey as a Complementary Medicine. Integr. Med. Insights 2017, 12, 1–15. [Google Scholar] [CrossRef]
- Combarros-Fuertes, P.; Estevinho, L.M.; Dias, L.G.; Castro, J.M.; Tomás-Barberán, F.A.; Tornadijo, M.E.; Fresno-Baro, J.M. Bioactive Components and Antioxidant and Antibacterial Activities of Different Varieties of Honey: A Screening Prior to Clinical Application. J. Agric. Food Chem. 2019, 67, 688–698. [Google Scholar] [CrossRef]
- Bucekova, M.; Jardekova, L.; Juricova, V.; Bugarova, V.; Di Marco, G.; Gismondi, A.; Leonardi, D.; Farkasovska, J.; Godocikova, J.; Laho, M.; et al. Antibacterial Activity of Different Blossom Honeys: New Findings. Molecules 2019, 24, 1573. [Google Scholar] [CrossRef]
- Pyrzynska, K.; Biesaga, M. Analysis of phenolic acids and flavonoids in honey. Trends Anal. Chem. 2009, 28, 893–902. [Google Scholar] [CrossRef]
- Kędzierska-Matysek, M.; Stryjecka, M.; Teter, A.; Skałecki, P.; Domaradzki, P.; Florek, M. Relationships between the Content of Phenolic Compounds and the Antioxidant Activity of Polish Honey Varieties as a Tool for Botanical Discrimination. Molecules 2021, 26, 1810. [Google Scholar] [CrossRef]
- Cianciosi, D.; Forbes-Hernández, T.Y.; Afrin, S.; Gasparrini, M.; Reboredo-Rodriguez, P.; Manna, P.P.; Zhang, J.; Bravo Lamas, L.; Martínez Flórez, S.; Agudo Toyos, P.; et al. Phenolic Compounds in Honey and Their Associated Health Benefits: A Review. Molecules 2018, 23, 2322. [Google Scholar] [CrossRef]
- Hernanz Vila, M.D.; Jara Palacios, M.J.; Santos Morcillo, J.L.; Gómez Pajuelo, A.; Heredia Mira, F.J.; Terrab Benjelloun, A. The profile of phenolic compounds by HPLC-MS in Spanish oak (Quercus) honeydew honey and their relationships with color and antioxidant activity. LWT—Food Sci. Technol. 2023, 180, 114724. [Google Scholar] [CrossRef]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Bamunuarachchi, N.I.; Tabassum, N.; Kim, Y.-M. Caffeic acid and its derivatives: Antimicrobial drugs toward microbial pathogens. J. Agric. Food Chem. 2021, 69, 2979–3004. [Google Scholar] [CrossRef] [PubMed]
- Ranneh, Y.; Faisal Ali, F.; Zarei, M.; Akim, A.M.; Hamid, H.A.; Khazaai, H. Malaysian stingless bee and Tualang honeys: A comparative characterization of total antioxidant capacity and phenolic profile using liquid chromatography-mass spectrometry. LWT—Food Sci. Technol. 2018, 89, 1–9. [Google Scholar] [CrossRef]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. Proc. Phytochem. Soc. Eur. 2018, 18, 241–272. [Google Scholar] [CrossRef]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef]
- Cushnie, T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Nešović, M.; Gašić, U.; Tosti, T.; Horvacki, N.; Šikoparija, B.; Nedić, N.; Blagojević, S.; Ignjatović, L.; Tešić, Ž. Polyphenol profile of buckwheat honey, nectar and pollen. R. Soc. Open Sci. 2020, 7, 201576. [Google Scholar] [CrossRef]
- Tanleque-Alberto, F.; Juan-Borrás, M.; Escriche, I. Antioxidant characteristics of honey from Mozambique based on specific flavonoids and phenolic acid compounds. J. Food Compos. Anal. 2020, 86, 103377. [Google Scholar] [CrossRef]
- Yayinie, M.; Atlabachew, M.; Tesfaye, A.; Hilluf, W.; Reta, C.; Alemneh, T. Polyphenols, flavonoids, and antioxidant content of honey coupled with chemometric method: Geographical origin classification from Amhara region, Ethiopia. Int. J. Food Prop. 2022, 25, 76–92. [Google Scholar] [CrossRef]
- Lv, C.; Yang, J.; Liu, R.; Lu, Q.; Ding, Y.; Zhang, J.; Deng, J. A comparative study on the adsorption and desorption characteristics of flavonoids from honey by six resins. Food Chem. 2018, 268, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Gašić, U.; Kečkeš, S.; Dabić, D.; Trifković, J.; Milojković-Opsenica, D.; Natić, M.; Tešić, Ž. Phenolic profile and antioxidant activity of Serbian polyfloral honeys. Food Chem. 2014, 145, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Iurlina, M.O.; Saiz, A.I.; Fritz, R.; Manrique, G.D. Major flavonoids of Argentinean honeys. Optimisation of the extraction method and analysis of their content in relationship to the geographical source of honeys. Food Chem. 2009, 115, 1141–1149. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Polak, T.; Kropf, U.; Korošec, M.; Golob, T. LC-DAD-ESI/MS analysis of flavonoids and abscisic acid with chemometric approach for the classification of Slovenian honey. Food Chem. 2011, 127, 296–302. [Google Scholar] [CrossRef]
- Formosa, J.P. Chemical Profiling of Honey Produced in the Maltese Islands. Master’s Thesis, University of Malta, Msida, Malta, 2017. [Google Scholar]
- Griep, M.A.; Blood, S.; Larson, M.A.; Koepsell, S.A.; Hinrichs, S.H. Myricetin inhibits Escherichia coli DnaB helicase but not primase. Bioorg. Med. Chem. 2007, 15, 7203–7208. [Google Scholar] [CrossRef]
- Escriche, I.; Kadar, M.; Juan-Borras, M.; Domenech, E. Using flavonoids compounds and headspace voiltale profile for botanical authentication of lemon and orange honeys. Food Res. Int. 2011, 44, 1504–1513. [Google Scholar] [CrossRef]
- Estevinho, L.; Pereira, A.P.; Moreira, L.; Dias, L.G.; Pereira, E. Antioxidant and antimirobial effects of phenolic compounds extracts of Northeast Portugal honey. Food Chem. Toxicol. 2008, 46, 3773–3779. [Google Scholar] [CrossRef]
- Kenjeric, D.; Mandic, M.L.; Primorac, L.; Bubalo, D.; Perl, A. Flavonoid profile of Robinia honeys produced in Croatia. Food Chem. 2007, 102, 683–690. [Google Scholar] [CrossRef]
- Fatima, M.T.; Bhat, A.A.; Nisar, S.; Fakhro, K.A.; Akil, A.S.A.-S. The role of dietary antioxidants in type 2 diabetes and neurodegenerative disorders: An assessment of the benefit profile. Heliyon 2023, 9, e12698. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Katz, A.; Wang, Y.; Eck, P.; Kwon, O.; Lee, J.-H.; Chen, S.; Corpe, C.; Dutta, A.; Dutta, S.K.; et al. Vitamin C as an Antioxidant: Evaluation of Its Role in Disease Prevention. J. Am. Coll. Nutr. 2003, 22, 18–35. [Google Scholar] [CrossRef]
- Gęgotek, A.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Activity of Ascorbic Acid. Antioxidants 2022, 11, 1993. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, H.; Tsubota, T.; Kanki, K.; Shiota, G. All-trans retinoic acid ameliorates hepatic stellate cell activation via suppression of thioredoxin interacting protein expression. J. Cell. Physiol. 2018, 233, 607–616. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, A.; Ebissy, E.; Mohamed, R.; Ateya, A. Effects of antioxidant vitamins (A, D, E) and trace elements (Cu, Mn, Se, Zn) administration on gene expression, metabolic, antioxidants and immunological profiles during transition period in dromedary camels. BMC Vet. Res. 2024, 20, 101. [Google Scholar] [CrossRef] [PubMed]
- Velimirović, D.; Tošić, S.; Mitić, S.; Pavlović, A.; Rašić Mišić, I.; Stojanović, G. Mineral, phenolic content and antioxidant activity of selected honey samples consumed in Serbia. J. Apic. Res. 2023, 62, 850–862. [Google Scholar] [CrossRef]
- Kenjeric, D.; Mandic, M.L.; Primorac, L.; Cacic, F.A. Flavonoid pattern of sage (Salvia officinalis L.) unifloral honeys. Food Chem. 2008, 110, 187–192. [Google Scholar] [CrossRef]
- Kim, S.; Woo, E.-R.; Lee, D.G. Apigenin promotes antibacterial activity via regulation of nitric oxide and superoxide anion production. J. Basic Microbiol. 2020, 60, 862–872. [Google Scholar] [CrossRef]
- Kassim, M.; Achoui, M.; Mustafa, M.R.; Mohd, M.A.; Yusoff, K.M. Ellagic acid, phenolic acids and flavonoids in Malaysian honey extracts demonstrate in vitro antiflammatory activity. Nutr. Res. 2010, 30, 650–659. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, Y.; Zhang, Z.; Chen, M.; Zhang, D.; Tian, C.; Liu, M.; Jiang, G. The antibacterial activity and mechanism of action of Luteolin against Trueperella pyogenes. Infect. Drug Resist. 2020, 13, 1697–1711. [Google Scholar] [CrossRef]
- Silva, B.; Biluca, F.C.; Gonzaga, L.V.; Fett, R.; Monguilhott Dalmarco, E.; Caon, T.; Oliveira Costa, A.C. In vitro anti-inflammatory properties of honey flavonoids: A review. Food Res. Int. 2021, 141, 110086. [Google Scholar] [CrossRef]
- Candiracci, M.; Citterio, B.; Piatti, E. Antifungal activity of the honey flavonoid extract against Candida albicans. Food Chem. 2012, 131, 493–499. [Google Scholar] [CrossRef]
- Soobrattee, M.A.; Neergheen, V.S.; Luximon-Ramma, A.; Arouma, O.I.; Bahorun, T. Phenolics as potential antioxidant therapeutic agents: Mechanism and actions. Mutat. Res. 2005, 579, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Pluta, R.; Miziak, B.; Czuczwar, S.J. Apitherapy in Post-Ischemic Brain Neurodegeneration of Alzheimer’s Disease Proteinopathy: Focus on Honey and Its Flavonoids and Phenolic Acids. Molecules 2023, 28, 5624. [Google Scholar] [CrossRef] [PubMed]
- Tichonow, A.I.; Bondarenko, L.A.; Jarnych, T.G.; Szpyczak, O.S.; Kowal, W.M.; Skrypnik-Tichonow, R.I. Miód Naturalny w Medycynie i Farmacji (Pochodzenie, Właściwości, Zastosowanie, Preparaty Lecznicze); Gospodarstwo Pasieczne SĄDECKI BARTNIK: Stróże, Poland, 2017. [Google Scholar]
- Rosiak, E.; Jaworska, D. Probiotic and prebiotic properties of bee honey in terms of their quality and health safety. Żywn. Nauka Technol. Jakość 2019, 26, 36–48, (In Polish, Possibly Abstract in English). [Google Scholar]
- Hossen, M.S.; Ali, M.Y.; Jahurul, M.H.A.; Abdel-Daim, M.M.; Gan, S.H.; Khalil, M.I. Beneficial roles of honey polyphenols against some human degenerative diseases: A review. Pharmacol. Rep. 2017, 69, 1194–1205. [Google Scholar] [CrossRef]
- Bueno-Costa, F.M.; Zambiazi, R.C.; Bohmer, B.W.; Chaves, F.C.; da Silva, W.P.; Zanusso, J.T.; Dutra, I. Antibacterial and antioxidant activity of honeys from the state of Rio Grande do Sul, Brazil. LWT—Food Sci. Technol. 2016, 65, 333–340. [Google Scholar] [CrossRef]
- Terzo, S.; Mulè, F.; Amato, A. Honey and obesity-related dysfunctions: A summary on health benefits. J. Nutr. Biochem. 2020, 82, 108401. [Google Scholar] [CrossRef]
- Lori, G.; Cecchi, L.; Mulinacci, N.; Melani, F.; Caselli, A.; Cirri, P.; Paoli, P. Honey extracts inhibit PTP1B, upregulate insulin receptor expression, and enhance glucose uptake in human HepG2 cells. Biomed. Pharmacother. 2019, 113, 108752. [Google Scholar] [CrossRef]
- Manna, P.; Jain, S.K. Obesity, oxidative stress, adipose tissue dysfunction, and the associated health risks: Causes and therapeutic strategies. Metab. Syndr. Relat. Disord. 2015, 13, 423–444. [Google Scholar] [CrossRef]
- Bobiş, O.; Dezmirean, D.S.; Moise, A.R. Honey and diabetes: The importance of natural simple sugars in diet for preventing and treating different type of diabetes. Oxid. Med. Cell. Longev. 2018, 2018, 4757893. [Google Scholar] [CrossRef]
- Meo, S.A.; Al-Asiri, S.A.; Mahesar, A.L.; Ansari, M.J. Role of honey in modern medicine. Saudi J. Biol. Sci. 2017, 24, 975–978. [Google Scholar] [CrossRef]
- Schramm, D.D.; Karim, M.; Schrader, H.R.; Holt, R.R.; Cardetti, R.; Keen, C.L. Honey with high levels of antioxidants can provide protection to healthy human subjects. J. Agric. Food. Chem. 2003, 51, 1732–1735. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Sulaiman, S.A.; Baig, A.A.; Ibrahim, M.; Liaqat, S.; Fatima, S.; Othman, N.H. Honey as a potential natural antioxidant medicine: An insight into its molecular mechanisms of action. Oxid. Med. Cell. Longev. 2018, 2018, 8367846. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.; Amaral, J.S.; Oliveira, M.B.P.P.; Mafra, I. A Comprehensive review on the main honey authentication issues: Production and origin. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1072–1100. [Google Scholar] [CrossRef]
- Samarghandian, S.; Farkhondeh, T.; Samini, F. Honey and Health: A Review of Recent Clinical Research. Pharmacogn. Res. 2017, 9, 121–127. [Google Scholar]
- Ramsay, E.I.; Rao, S.; Madathil, L.; Hegde, S.K.; Baliga-Rao, M.P.; George, T.; Baliga, M.S. Honey in oral health and care: A mini review. J. Oral Biosci. 2019, 61, 32–36. [Google Scholar] [CrossRef]
- Kaur, S.; Mirza, A.; Singh, J. Recent advances of honey in modern medicines: A review. J. Pharmacogn. Phytochem. 2017, 6, 2063–2067. [Google Scholar]
- Dżugan, M.; Tomczyk, M.; Sowa, P.; Grabek-Lejko, D. Antioxidant activity as biomarker of honey variety. Molecules 2018, 23, 2069. [Google Scholar] [CrossRef]
- Küçük, M.; Kolayli, S.; Karaoğlu, S.; Ulusoy, E.; Baltaci, C.; Candan, F. Biological activities and chemical composition of three honeys of different types from Anatolia. Food Chem. 2007, 100, 526–534. [Google Scholar] [CrossRef]
- Becerril-Sánchez, A.L.; Quintero-Salazar, B.; Dublán-García, O.; Escalona-Buendía, H.B. Phenolic Compounds in Honey and Their Relationship with Antioxidant Activity, Botanical Origin, and Color. Antioxidants 2021, 10, 1700. [Google Scholar] [CrossRef]
- Ranneh, Y.; Akim, A.M.; Hamid, H.A.; Khazaai, H.; Fadel, A.; Zakaria, Z.A.; Albujja, M.; Bakar, M.F.A. Honey and Its Nutritional and Anti-Inflammatory Value. BMC Complement. Med. Ther. 2021, 21, 30. [Google Scholar] [CrossRef]
- Chua, L.S.; Ismail, N.I.M. Anti-inflammatory and anti-microbial activities of selected honey samples. Asian J. Agric. Res. 2015, 9, 293–304. [Google Scholar] [CrossRef]
- Cho, H.; Yun, C.-W.; Park, W.-K.; Kong, J.-Y.; Kim, K.S.; Park, Y.; Lee, S.; Kim, B.-K. Modulation of the activity of pro-inflammatory enzymes, COX-2 and iNOS, by chrysin derivatives. Pharmacol. Res. 2004, 49, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Riaz, T.; Akram, M.; Laila, U.; Khalil, M.T.; Zainab, R.; Iftikhar, M.; Ozdemir, F.A.; Sołowski, G.; Alinia-Ahandani, E.; Altable, M.; et al. A review of pharmacology and medicinal properties of honey. IAIM 2023, 10, 37–46. [Google Scholar]
- Bt Hj Idrus, R.; Sainik, N.Q.A.V.; Nordin, A.; Saim, A.B.; Sulaiman, N. Cardioprotective Effects of Honey and Its Constituent: An Evidence-Based Review of Laboratory Studies and Clinical Trials. Int. J. Environ. Res. Public Health 2020, 21, 3613. [Google Scholar] [CrossRef]
- Mohan, A.; Quek, S.-Y.; Gutierrez-Maddox, N.; Gao, Y.; Shu, Q. Effect of Honey in Improving the Gut Microbial Balance. Food Qual. Saf. 2017, 1, 107–115. [Google Scholar] [CrossRef]
- Favarin, L.; Laureano-Melo, R.; Luchese, R.H. Survival of free and microencapsulated Bifidobacterium: Effect of honey addition. J. Microencapsul. 2015, 32, 329–335. [Google Scholar] [CrossRef]
- Catalkaya, G.; Venema, K.; Lucini, L.; Rocchetti, G.; Delmas, D.; Daglia, M.; De Filippis, A.; Xiao, H.; Quiles, J.L.; Xiao, J.; et al. Interaction of Dietary Polyphenols and Gut Microbiota: Microbial Metabolism of Polyphenols, Influence on the Gut Microbiota, and Implications on Host Health. Food Front. 2020, 1, 109–133. [Google Scholar] [CrossRef]
- Ray, S.K.; Mukherjee, S. Evolving Interplay Between Dietary Polyphenols and Gut Microbiota—An Emerging Importance in Healthcare. Front. Nutr. 2021, 8, 195. [Google Scholar] [CrossRef]
- de Souza, E.L.; de Albuquerque, T.M.R.; Dos Santos, A.S.; Massa, N.M.L.; de Brito Alves, J.L. Potential Interactions among Phenolic Compounds and Probiotics for Mutual Boosting of Their Health-Promoting Properties and Food Functionalities—A Review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1645–1659. [Google Scholar] [CrossRef]
- Jones, R. Honey and healing through the ages. J. ApiProduct ApiMedical Sci. 2009, 1, 2–5. [Google Scholar] [CrossRef]
- Magdas, T.M.; David, M.; Hategan, A.R.; Filip, G.A.; Magdas, D.A. Geographical Origin Authentication—A Mandatory Step in the Efficient Involvement of Honey in Medical Treatment. Foods 2024, 13, 532. [Google Scholar] [CrossRef]
- Stefanis, C.; Stavropoulou, E.; Giorgi, E.; Voidarou, C.; Constantinidis Vrioni, G.; Tsakris, A. Honey’s antioxidant and antimicrobial properties: A bibliometric study. Antioxidants 2023, 12, 414. [Google Scholar] [CrossRef]
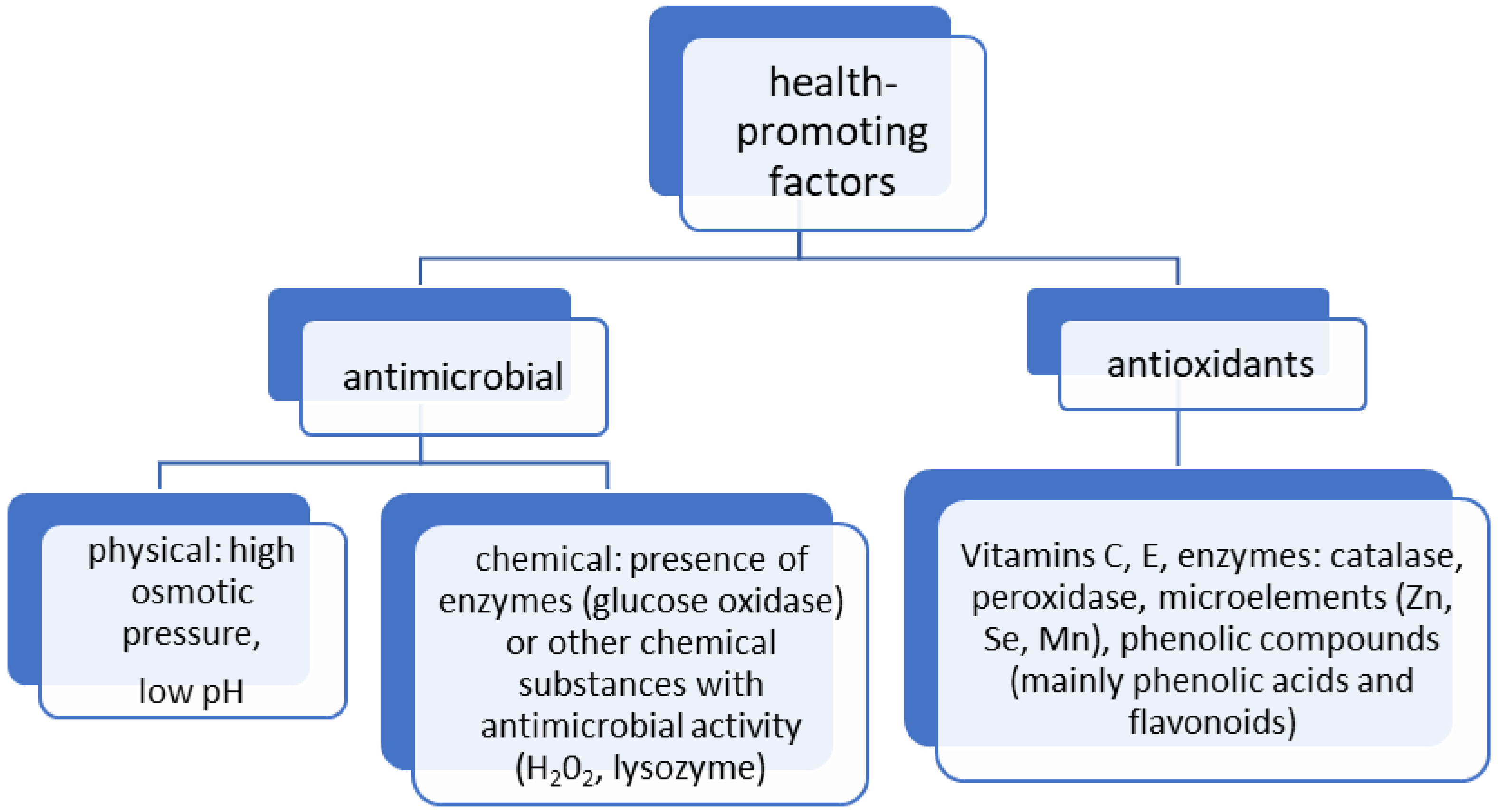
| Components | Average Content | Comments |
|---|---|---|
| Water | 17–20% | Some varieties (e.g., heather honey) may contain more—up to 23% [11,12,13,14,15]. |
| Carbohydrates, including: | 65–87% | Carbohydrates are the main component of honey. The sugar composition of honey depends on the type of honey, the time of harvesting, and the length of the storage period—unripe honey is characterized by a higher sucrose content. However, the most important factor affecting the content of saccharides is the origin of the honey. The fructose content in honey ranges from 30 to just over 40%. The glucose content is slightly lower and ranges, on average, from 19 to 31%. The ratio of fructose to glucose in most varieties of honey ranges from 1.2 to 1.7. The honeydew honey contain more oligosaccharides and dextrins, while the nectar honey varieties are dominated by mono- and disaccharides [11,12,13,14,15,16,17,18,19]. |
| monosaccharides (glucose, fructose) | 67–70% | |
disaccharides
| 0–15% | |
| oligosaccharides (reducing and non-reducing) | 0.5–10% | |
| dextrins | 1–10% | |
Nitrogen compounds, including:
| 0.25–3% | Nitrogen compounds appear in honey in small quantities, and these are simple proteins: albumins and globulins, enzymes, and free amino acids. Honey contains 175 mg of free amino acids per 100 g, of which the basic one is proline, constituting about 50–85% of all amino acids. The proline content is a measure of the adulteration of honey with sucrose. Honey enzymes are of both plant origin (from pollen and honeydew) and animal origin (bee glands). They play the role of effective biological catalysts in decomposition and synthesis reactions, e.g., invertase decomposes sucrose, and diastase participates in starch hydrolysis [20,21,22,23,24]. |
| 0.2% | |
| 0.2–1% | |
| Organic acids (gluconic, citric, lactic, malic, succinic, butyric, propionic, tartaric, oxalic, linoleic, linolenic). | 0.01–1.5% | Gluconic acid constitutes 70–90% of organic acids in honey. Organic acids determine the taste and the aroma of honey as well as its microbial properties. Just like the content of other components, the acid content is mainly determined by the botanical origin of honey [15,18,25,26]. |
| Inorganic acids (hydrochloric and phosphoric) | 0.03–0.1% | |
| Vitamins (A, C, D, K, B1, B2, PP, B6, B5 B9, H) [2,15,27,28]. | 0.04% | |
| Mineral substances (about 47 elements: potassium, silicon, sodium, iron, copper, magnesium, manganese, sulfur, nickel, phosphorus, chlorine, cobalt, iodine, zinc, palladium, arsenic, strontium, aluminum, tungsten, lead, chromium, titanium, barium, molybdenum, vanadium, tin, silver) [2,10,29,30]. | Blossom honey contains 0.05–0.50% of minerals, nectar-honeydew honey—0.32–0.52%, and honeydew honey about 1% of minerals. | The mineral content is influenced by environmental factors, primarily the content of mineral substances in the soil where melliferous plants are grown [24,30,31,32,33]. |
| Aromatic substances (about 200): aldehydes and ketones (formic, acetic and isobutyric aldehydes as well as acetone and diacetyl), polyphenolic compounds, esters, higher aliphatic alcohols. Dyes—carotenoids (carotene and xanthophyll), flavonoids (and the compounds formed by their combination), chlorophyll and anthocyanins, and tannins [29,33,34]. | 0.07–0.1% | Dyes, ethereal oils, and other aromatic substances in honey are of the floral origin which determines their organoleptic properties. The honey flavor is connected primarily with aldehydes, ketones, esters, higher alcohols, and polyphenolic compounds [19,29,30,33]. The content of aromatic substances may decrease during heating and long storage [18,35]. |
| Component(s) | Mechanisms of Action | Pro-Health Effects | Source |
|---|---|---|---|
| Vitamins C, A, E | Among the antioxidant vitamins, vitamin C is found in the largest amounts in honey. Vitamin C reduces ROS levels and thus prevents the oxidation of lipids, proteins, and DNA, recovers vasodilation, and decreases nitrate tolerance. Vitamin A and β-carotenes improve oxidative stress and cognitive function and reduce toxic amyloid β by inhibiting amyloid β oligomerization and aggregation in a streptozotocin-induced Alzheimer’s disease mouse model. It was also reported that retinoic acid plays a key role in the inhibition of hepatic stellate cell activation (an effector of hepatocellular carcinoma) via suppressing the thioredoxin-interacting protein and reducing oxidative stress levels. Vitamin E reduces the markers of oxidative stress, increases glutathione peroxidase and superoxide dismutase, changes/improves the total antioxidant capacity and glycemic control, and delays the onset as well as the progression of type 2 diabetes. However, considering the content of these vitamins in honey, their contribution to the development of antioxidant and health-promoting properties of honey is insignificant. | Nutritional Properties Antioxidant Effect | [63,64,65,66] |
| Macro and microelements | Macro- and microelements are compounds necessary to ensure the proper development, growth, and other vital functions of the human body. They regulate a wide array of physiological mechanisms with substantial specificity and selectivity, as components of enzymes and other organic molecular complexes. In honey, trace elements such as copper, zinc, selenium and magnesium, which have antioxidant properties, have been detected. Selenium (Se) is a crucial component of the glutathione peroxidase enzyme, which may eliminate lipid hydroperoxides and hydrogen peroxide (H2O2). Copper (Cu), as well as zinc (Zn), are necessary building blocks for a number of enzymes, including those involved in oxidation–reduction reaction and Cu–Zn superoxide dismutase (SOD). Manganese (Mn) is an essential co-factor in enzymatic processes connected to the metabolic control of gene expression. Similar to vitamins, the antioxidant effect of honey’s macro- and microelements is of little importance. | Nutritional Properties Antioxidant Properties | [67] |
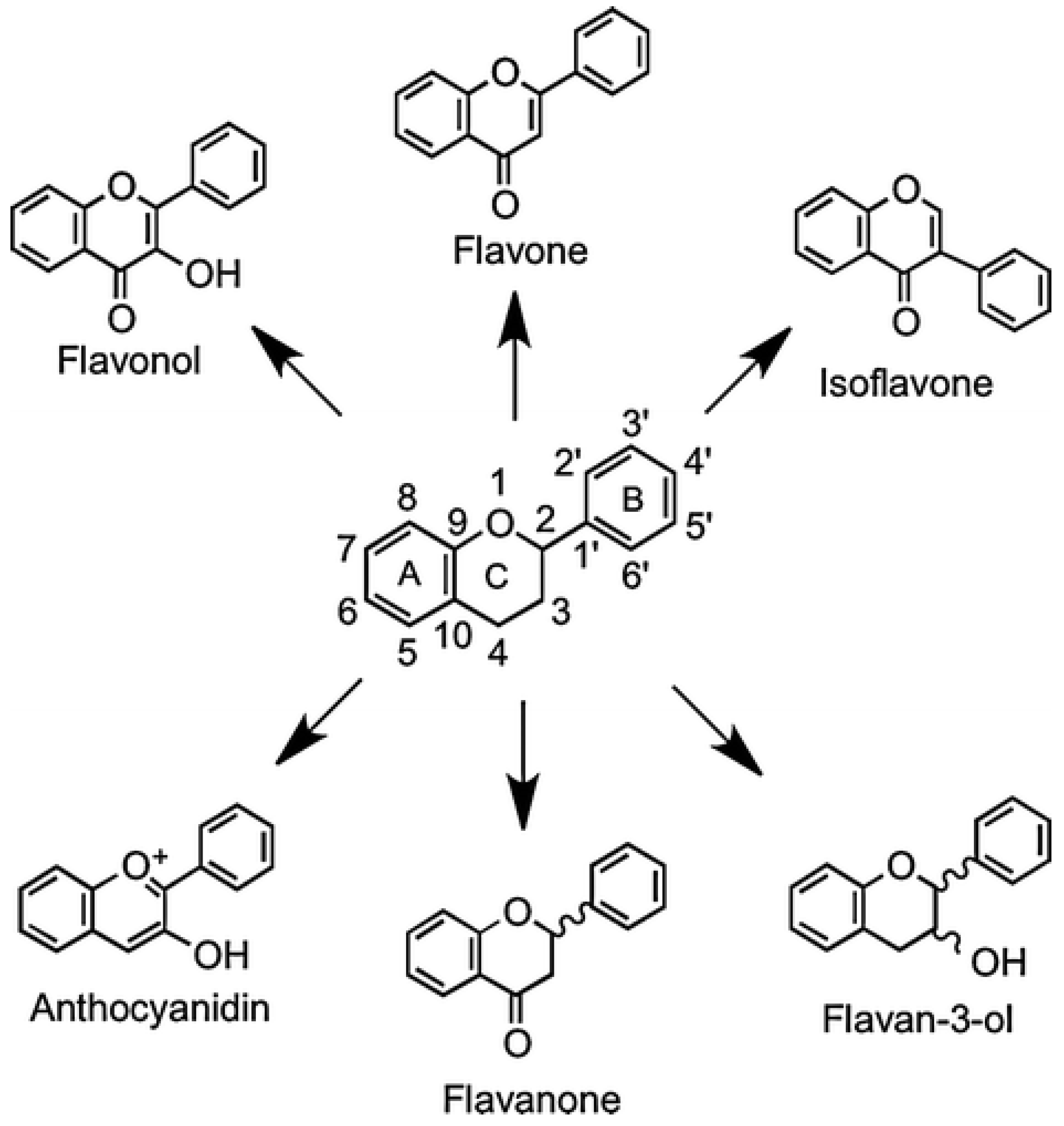
| Polyphenols (phenolic acids and flavonoids) | Polyphenols have a protective effect on a number of diseases, e.g., cardiovascular diseases, cancer, neurodegenerative diseases, diabetes, etc. Honey flavonoids exhibit antioxidant activity and also have the ability to inhibit some pro-inflammatory enzymes (cyclooxygenases COX, lipoxygenases LOX, inducible nitric oxide synthase iNOS) and mediators (nitric oxide, cytokines and chemokines). They reduce the oxidation of LDL and lipids and stimulate the maintenance of lipid parameters at the appropriate level, which prevents the clogging of arteries and prevents the effects of atherosclerotic changes in the blood vessels. Flavonoids counteract tumor development by inhibiting tumor-producing enzymes, blocking the activity of certain hormones, and interfering with the delivery of oxygen and other components necessary for tumor formation. These compounds are responsible, among other things, for reducing oxidative stress, decreasing apoptosis, necrosis, brain atrophy and behavioral and neurological deficits. Recently, there have been reports on the use of honey’s phenolic compounds for the treatment of Alzheimer’s disease. They influence, among other things, neurodegeneration associated with amyloid pathology and ischemia in proteinopathy. Honey polyphenols can also prevent numerous neurodegenerative diseases by protecting neurons from oxidative damage, enhancing neuronal function and regeneration, protecting neurons from Ab-induced neuronal injury and neurotoxicity, protecting hippocampal cells against nitric oxide-induced toxicity, and modulating neuronal and glial cell signaling pathways. For example, ferulic acid exerted neuroprotective effects in a mouse model of Parkinson’s disease by decreasing the levels of phospho-Akt, phospho-pyruvate dehydrogenase kinase-1, and phospho-Bad and increasing the caspase-3 levels. The mechanisms of action of particular polyphenols are different and depend mainly on the molecule structure, e.g., kaempferol, luteolin, chrysin, pinocembrin, and gallic acid induce apoptosis, while apigenin promotes interleukin 1b and tumor necrosis factor. Polyphenols also neutralize oxidative stress in various ways. Caffeic acid inhibits oxidative stress in iron-overloaded rats by reducing lipid peroxidation and increasing vitamin E levels in the plasma. Quercetin reduces oxidative stress by scavenging free radicals, chelating metal ions, and inhibiting xanthine oxidase and lipid peroxidation. Kaempferol reduces oxidative stress caused by glutamate in the mouse hippocampal cell line HT-22 by blocking ROS generation, and also blocks oxidative stress in granule cells during low-potassium-induced apoptosis. Through antioxidant pathways, honey’s polyphenols ameliorate cholesterol and cardiac enzyme levels. Polyphenols also exhibit bactericidal effects against both Gram-negative and Gram-positive bacteria. Their antibacterial mechanism is based on inhibiting bacterial biofilm formation or inactivating enzymes. In alkaline conditions (pH 7.0–8.0), polyphenols can display pro-oxidative properties, inhibiting microbial growth by accelerating hydroxyl radical formation and oxidative strand breakage in DNA. They could also support the production of considerable amounts of H2O2 via a non-enzymatic pathway. | Anti-Allergic Effect Antioxidant, Anti-Inflammatory, and Anty-hiperlipidemic Properties Application in Disorders of the Immune System Cardio- and NeuroProtective Effects Antibacterial and Antiviral Effect | [29,41,46,47,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86] |
| Enzymes (e.g., catalase, peroxidase) | They enable digestion by breaking down complex molecules such as sugars, fats, and carbohydrates into simpler elements. The enzyme glucose oxidase, most likely originating from the digestive tract of bees, is responsible for the bacteriostatic and bactericidal properties of honey. This enzyme belongs to the group of oxidoreductases and catalyzes the oxidation of glucose to glucono-δ-lactone. The by-product of this reaction is H2O2. Catalase is also included in the group of oxidoreductases. This enzyme comes from nectar, honeydew, or pollen. Catalase acts as a regulator of the H2O2 content, catalyzing the reaction of its decomposition into water and molecular oxygen, thus contributing to the reduction in the bactericidal and bacteriostatic properties of honey. | Antioxidant Effect | [87,88,89] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilczyńska, A.; Żak, N. Polyphenols as the Main Compounds Influencing the Antioxidant Effect of Honey—A Review. Int. J. Mol. Sci. 2024, 25, 10606. https://doi.org/10.3390/ijms251910606
Wilczyńska A, Żak N. Polyphenols as the Main Compounds Influencing the Antioxidant Effect of Honey—A Review. International Journal of Molecular Sciences. 2024; 25(19):10606. https://doi.org/10.3390/ijms251910606
Chicago/Turabian StyleWilczyńska, Aleksandra, and Natalia Żak. 2024. "Polyphenols as the Main Compounds Influencing the Antioxidant Effect of Honey—A Review" International Journal of Molecular Sciences 25, no. 19: 10606. https://doi.org/10.3390/ijms251910606
APA StyleWilczyńska, A., & Żak, N. (2024). Polyphenols as the Main Compounds Influencing the Antioxidant Effect of Honey—A Review. International Journal of Molecular Sciences, 25(19), 10606. https://doi.org/10.3390/ijms251910606






