Non-Alcoholic Fatty Liver Disease and Coronary Artery Disease: A Bidirectional Association Based on Endothelial Dysfunction
Abstract
1. Introduction
2. Epidemiology and the Consensus of NAFLD Terminology
3. Metabolic-Associated Fatty Liver Disease (MAFLD) and Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD)
4. NAFLD and Coronary Artery Disease
5. Shared Risk Factors of CAD and NAFLD
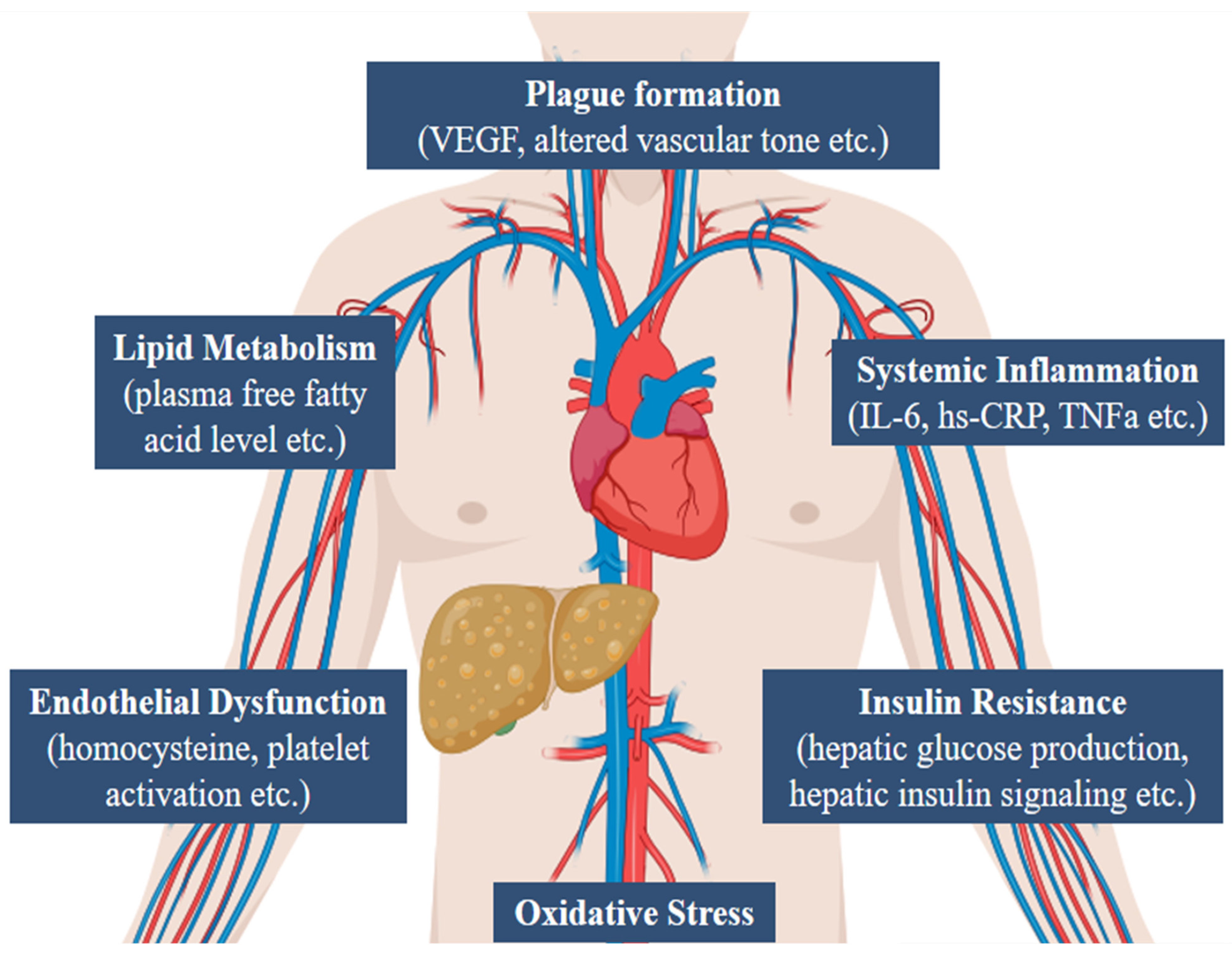
6. Pathophysiological Mechanisms and Association of CAD and NAFLD
7. The Role of the Endothelium in Atherosclerosis
8. Endothelial Dysfunction and NAFLD
9. Systemic Inflammation and NAFLD
10. Plaque Formation/Instability and NAFLD
11. Platelet-Activating Factor as the Connecting Link between NAFLD and CAD
12. Gut Microbiota and NAFLD
13. Lipid Metabolism Dysregulation and NAFLD
14. Insulin Resistance and NAFLD
15. Studies Associating CAD and NAFLD
16. Genetic Associations between NAFLD and CAD
17. Clinical Implications
18. Optimizing Medical Options
19. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ludwig, J.; Viggiano, T.R.; McGill, D.B.; Oh, B.J. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin. Proc. 1980, 55, 434–438. [Google Scholar] [PubMed]
- Federico, A.; Dallio, M.; Masarone, M.; Persico, M.; Loguercio, C. The epidemiology of non-alcoholic fatty liver disease and its connection with cardiovascular disease: Role of endothelial dysfunction. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4731–4741. [Google Scholar] [PubMed]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The diagnosis and management of non-alcoholic fatty liver disease: Practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012, 55, 2005–2023. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zou, B.; Yeo, Y.H.; Feng, Y.; Xie, X.; Lee, D.H.; Fujii, H.; Wu, Y.; Kam, L.Y.; Ji, F.; et al. Prevalence, incidence, and outcome of non-alcoholic fatty liver disease in Asia, 1999–2019: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2019, 4, 389–398. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Stepanova, M.; Afendy, M.; Fang, Y.; Younossi, Y.; Mir, H.; Srishord, M. Changes in the prevalence of the most common causes of chronic liver diseases in the United States from 1988 to 2008. Clin Gastroenterol. Hepatol. 2011, 9, 524–530.e521, quiz e560. [Google Scholar] [CrossRef]
- Grundy, S.M.; Hansen, B.; Smith, S.C., Jr.; Cleeman, J.I.; Kahn, R.A.; American Heart, A.; National Heart, L.; Blood, I.; American Diabetes, A. Clinical management of metabolic syndrome: Report of the American Heart Association/National Heart, Lung, and Blood Institute/American Diabetes Association conference on scientific issues related to management. Arterioscler. Thromb. Vasc. Biol. 2004, 24, e19–e24. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef]
- Marchesini, G.; Bugianesi, E.; Forlani, G.; Cerrelli, F.; Lenzi, M.; Manini, R.; Natale, S.; Vanni, E.; Villanova, N.; Melchionda, N.; et al. Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology 2003, 37, 917–923. [Google Scholar] [CrossRef]
- Hu, X.Y.; Li, Y.; Li, L.Q.; Zheng, Y.; Lv, J.H.; Huang, S.C.; Zhang, W.; Liu, L.; Zhao, L.; Liu, Z.; et al. Risk factors and biomarkers of non-alcoholic fatty liver disease: An observational cross-sectional population survey. BMJ Open 2018, 8, e019974. [Google Scholar] [CrossRef]
- Ogresta, D.; Mrzljak, A.; Cigrovski Berkovic, M.; Bilic-Curcic, I.; Stojsavljevic-Shapeski, S.; Virovic-Jukic, L. Coagulation and Endothelial Dysfunction Associated with NAFLD: Current Status and Therapeutic Implications. J. Clin. Transl. Hepatol. 2022, 10, 339–355. [Google Scholar] [CrossRef] [PubMed]
- Ismaiel, A.; Dumitrascu, D.L. Cardiovascular Risk in Fatty Liver Disease: The Liver-Heart Axis-Literature Review. Front. Med. 2019, 6, 202. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Marchesini, G.; Pinto-Cortez, H.; Petta, S. Epidemiology of Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis: Implications for Liver Transplantation. Transplantation 2019, 103, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Blissett, D.; Blissett, R.; Henry, L.; Stepanova, M.; Younossi, Y.; Racila, A.; Hunt, S.; Beckerman, R. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatology 2016, 64, 1577–1586. [Google Scholar] [CrossRef]
- Weintraub, W.S. The Economic Burden of Illness. JAMA Netw. Open 2023, 6, e232663. [Google Scholar] [CrossRef]
- Wang, D.; Xu, Y.; Zhu, Z.; Li, Y.; Li, X.; Li, Y.; Shen, H.; Wu, W.; Liu, Y.; Han, C. Changes in the global, regional, and national burdens of NAFLD from 1990 to 2019: A systematic analysis of the global burden of disease study 2019. Front. Nutr. 2022, 9, 1047129. [Google Scholar] [CrossRef]
- Simon, T.G.; Roelstraete, B.; Khalili, H.; Hagstrom, H.; Ludvigsson, J.F. Mortality in biopsy-confirmed nonalcoholic fatty liver disease: Results from a nationwide cohort. Gut 2021, 70, 1375–1382. [Google Scholar] [CrossRef]
- Arab, J.P.; Dirchwolf, M.; Alvares-da-Silva, M.R.; Barrera, F.; Benitez, C.; Castellanos-Fernandez, M.; Castro-Narro, G.; Chavez-Tapia, N.; Chiodi, D.; Cotrim, H.; et al. Latin American Association for the study of the liver (ALEH) practice guidance for the diagnosis and treatment of non-alcoholic fatty liver disease. Ann. Hepatol. 2020, 19, 674–690. [Google Scholar] [CrossRef]
- Liu, H.; Lu, H.Y. Nonalcoholic fatty liver disease and cardiovascular disease. World J. Gastroenterol. 2014, 20, 8407–8415. [Google Scholar] [CrossRef]
- Ong, J.P.; Pitts, A.; Younossi, Z.M. Increased overall mortality and liver-related mortality in non-alcoholic fatty liver disease. J. Hepatol. 2008, 49, 608–612. [Google Scholar] [CrossRef]
- Eslam, M.; Sanyal, A.J.; George, J.; International Consensus, P. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology 2020, 158, 1999–2014.e1991. [Google Scholar] [CrossRef] [PubMed]
- Diaz, L.A.; Arab, J.P.; Louvet, A.; Bataller, R.; Arrese, M. The intersection between alcohol-related liver disease and nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 2023, 20, 764–783. [Google Scholar] [CrossRef] [PubMed]
- Vilar-Gomez, E.; Calzadilla-Bertot, L.; Wai-Sun Wong, V.; Castellanos, M.; Aller-de la Fuente, R.; Metwally, M.; Eslam, M.; Gonzalez-Fabian, L.; Alvarez-Quinones Sanz, M.; Conde-Martin, A.F.; et al. Fibrosis Severity as a Determinant of Cause-Specific Mortality in Patients With Advanced Nonalcoholic Fatty Liver Disease: A Multi-National Cohort Study. Gastroenterology 2018, 155, 443–457.e417. [Google Scholar] [CrossRef] [PubMed]
- Portincasa, P. NAFLD, MAFLD, and beyond: One or several acronyms for better comprehension and patient care. Intern. Emerg. Med. 2023, 18, 993–1006. [Google Scholar] [CrossRef]
- Hsu, C.L.; Loomba, R. From NAFLD to MASLD: Implications of the new nomenclature for preclinical and clinical research. Nat. Metab. 2024, 6, 600–602. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Paik, J.M.; Stepanova, M.; Ong, J.; Alqahtani, S.; Henry, L. Clinical profiles and mortality rates are similar for metabolic dysfunction-associated steatotic liver disease and non-alcoholic fatty liver disease. J. Hepatol. 2024, 80, 694–701. [Google Scholar] [CrossRef]
- Gaggini, M.; Morelli, M.; Buzzigoli, E.; DeFronzo, R.A.; Bugianesi, E.; Gastaldelli, A. Non-alcoholic fatty liver disease (NAFLD) and its connection with insulin resistance, dyslipidemia, atherosclerosis and coronary heart disease. Nutrients 2013, 5, 1544–1560. [Google Scholar] [CrossRef]
- Nseir, W.; Shalata, A.; Marmor, A.; Assy, N. Mechanisms linking nonalcoholic fatty liver disease with coronary artery disease. Dig. Dis. Sci. 2011, 56, 3439–3449. [Google Scholar] [CrossRef]
- Nagral, A.; Bangar, M.; Menezes, S.; Bhatia, S.; Butt, N.; Ghosh, J.; Manchanayake, J.H.; Mahtab, M.A.; Singh, S.P. Gender Differences in Nonalcoholic Fatty Liver Disease. Euroasian J. Hepatogastroenterol. 2022, 12, S19–S25. [Google Scholar] [CrossRef]
- Zelber-Sagi, S.; Nitzan-Kaluski, D.; Goldsmith, R.; Webb, M.; Zvibel, I.; Goldiner, I.; Blendis, L.; Halpern, Z.; Oren, R. Role of leisure-time physical activity in nonalcoholic fatty liver disease: A population-based study. Hepatology 2008, 48, 1791–1798. [Google Scholar] [CrossRef]
- Nakamuta, M.; Fujino, T.; Yada, R.; Yada, M.; Yasutake, K.; Yoshimoto, T.; Harada, N.; Higuchi, N.; Kato, M.; Kohjima, M.; et al. Impact of cholesterol metabolism and the LXRalpha-SREBP-1c pathway on nonalcoholic fatty liver disease. Int. J. Mol. Med. 2009, 23, 603–608. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cazac, G.D.; Lacatusu, C.M.; Mihai, C.; Grigorescu, E.D.; Onofriescu, A.; Mihai, B.M. New Insights into Non-Alcoholic Fatty Liver Disease and Coronary Artery Disease: The Liver-Heart Axis. Life 2022, 12, 1189. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.F.; Wang, Y.W.; Lin, C.C.; Wang, Y.J.; Ding, Y.Z.; Liou, T.L.; Huang, S.S.; Lu, T.M.; Chan, W.L.; Lin, S.J.; et al. The association of the steatosis severity in fatty liver disease with coronary plaque pattern in general population. Liver Int. 2021, 41, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Jebari-Benslaiman, S.; Galicia-Garcia, U.; Larrea-Sebal, A.; Olaetxea, J.R.; Alloza, I.; Vandenbroeck, K.; Benito-Vicente, A.; Martin, C. Pathophysiology of Atherosclerosis. Int. J. Mol. Sci. 2022, 23, 3346. [Google Scholar] [CrossRef]
- Vanhoutte, P.M. Endothelial dysfunction: The first step toward coronary arteriosclerosis. Circ. J. 2009, 73, 595–601. [Google Scholar] [CrossRef]
- Jose, N.; P, K.V.; Kulirankal, K.G. Study of Endothelial Dysfunction in Patients With Non-alcoholic Fatty Liver Disease. Cureus 2021, 13, e20515. [Google Scholar] [CrossRef]
- Francque, S.M.; van der Graaff, D.; Kwanten, W.J. Non-alcoholic fatty liver disease and cardiovascular risk: Pathophysiological mechanisms and implications. J. Hepatol. 2016, 65, 425–443. [Google Scholar] [CrossRef]
- Garbuzenko, D.V. Pathophysiological mechanisms of cardiovascular disorders in non-alcoholic fatty liver disease. Gastroenterol. Hepatol. Bed Bench 2022, 15, 194–203. [Google Scholar] [CrossRef]
- Pacana, T.; Cazanave, S.; Verdianelli, A.; Patel, V.; Min, H.K.; Mirshahi, F.; Quinlivan, E.; Sanyal, A.J. Dysregulated Hepatic Methionine Metabolism Drives Homocysteine Elevation in Diet-Induced Nonalcoholic Fatty Liver Disease. PLoS ONE 2015, 10, e0136822. [Google Scholar] [CrossRef]
- Bruyndonckx, L.; Hoymans, V.Y.; Van Craenenbroeck, A.H.; Vissers, D.K.; Vrints, C.J.; Ramet, J.; Conraads, V.M. Assessment of endothelial dysfunction in childhood obesity and clinical use. Oxid. Med. Cell Longev. 2013, 2013, 174782. [Google Scholar] [CrossRef]
- Chiang, C.H.; Huang, P.H.; Chung, F.P.; Chen, Z.Y.; Leu, H.B.; Huang, C.C.; Wu, T.C.; Chen, J.W.; Lin, S.J. Decreased circulating endothelial progenitor cell levels and function in patients with nonalcoholic fatty liver disease. PLoS ONE 2012, 7, e31799. [Google Scholar] [CrossRef] [PubMed]
- Pastori, D.; Loffredo, L.; Perri, L.; Baratta, F.; Scardella, L.; Polimeni, L.; Pani, A.; Brancorsini, M.; Albanese, F.; Catasca, E.; et al. Relation of nonalcoholic fatty liver disease and Framingham Risk Score to flow-mediated dilation in patients with cardiometabolic risk factors. Am. J. Cardiol. 2015, 115, 1402–1406. [Google Scholar] [CrossRef] [PubMed]
- Sapmaz, F.; Uzman, M.; Basyigit, S.; Ozkan, S.; Yavuz, B.; Yeniova, A.; Kefeli, A.; Asilturk, Z.; Nazligul, Y. Steatosis Grade is the Most Important Risk Factor for Development of Endothelial Dysfunction in NAFLD. Medicine 2016, 95, e3280. [Google Scholar] [CrossRef] [PubMed]
- Theofilis, P.; Vordoni, A.; Nakas, N.; Kalaitzidis, R.G. Endothelial Dysfunction in Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis. Life 2022, 12, 718. [Google Scholar] [CrossRef]
- Sirbu, O.; Floria, M.; Dascalita, P.; Sorodoc, V.; Sorodoc, L. Non-alcoholic fatty liver disease-From the cardiologist perspective. Anatol. J. Cardiol. 2016, 16, 534–541. [Google Scholar] [CrossRef]
- Villanova, N.; Moscatiello, S.; Ramilli, S.; Bugianesi, E.; Magalotti, D.; Vanni, E.; Zoli, M.; Marchesini, G. Endothelial dysfunction and cardiovascular risk profile in nonalcoholic fatty liver disease. Hepatology 2005, 42, 473–480. [Google Scholar] [CrossRef]
- Yin, H.; Shi, A.; Wu, J. Platelet-Activating Factor Promotes the Development of Non-Alcoholic Fatty Liver Disease. Diabetes Metab. Syndr. Obes. 2022, 15, 2003–2030. [Google Scholar] [CrossRef]
- Lordan, R.; Tsoupras, A.; Zabetakis, I.; Demopoulos, C.A. Forty Years Since the Structural Elucidation of Platelet-Activating Factor (PAF): Historical, Current, and Future Research Perspectives. Molecules 2019, 24, 4414. [Google Scholar] [CrossRef]
- Zhou, W.; McCollum, M.O.; Levine, B.A.; Olson, M.S. Inflammation and platelet-activating factor production during hepatic ischemia/reperfusion. Hepatology 1992, 16, 1236–1240. [Google Scholar] [CrossRef]
- Yang, Y.; Nemoto, E.M.; Harvey, S.A.; Subbotin, V.M.; Gandhi, C.R. Increased hepatic platelet activating factor (PAF) and PAF receptors in carbon tetrachloride induced liver cirrhosis. Gut 2004, 53, 877–883. [Google Scholar] [CrossRef]
- Grypioti, A.D.; Theocharis, S.E.; Papadimas, G.K.; Demopoulos, C.A.; Papadopoulou-Daifoti, Z.; Basayiannis, A.C.; Mykoniatis, M.G. Platelet-activating factor (PAF) involvement in acetaminophen-induced liver toxicity and regeneration. Arch. Toxicol. 2005, 79, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Pei, K.; Gui, T.; Kan, D.; Feng, H.; Jin, Y.; Yang, Y.; Zhang, Q.; Du, Z.; Gai, Z.; Wu, J.; et al. An Overview of Lipid Metabolism and Nonalcoholic Fatty Liver Disease. Biomed. Res. Int. 2020, 2020, 4020249. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.V.; Asselbergs, F.W.; Palmer, T.M.; Drenos, F.; Lanktree, M.B.; Nelson, C.P.; Dale, C.E.; Padmanabhan, S.; Finan, C.; Swerdlow, D.I.; et al. Mendelian randomization of blood lipids for coronary heart disease. Eur. Heart J. 2015, 36, 539–550. [Google Scholar] [CrossRef] [PubMed]
- Vilar, C.P.; Cotrim, H.P.; Florentino, G.S.; Bragagnoli, G.; Schwingel, P.A.; Barreto, C.P. Nonalcoholic fatty liver disease in patients with coronary disease from a Brazil northeast area. Arq. Gastroenterol. 2015, 52, 111–116. [Google Scholar] [CrossRef]
- Ampuero, J.; Gallego-Duran, R.; Romero-Gomez, M. Association of NAFLD with subclinical atherosclerosis and coronary-artery disease: Meta-analysis. Rev. Esp. Enferm. Dig. 2015, 107, 10–16. [Google Scholar]
- Mantovani, A.; Csermely, A.; Petracca, G.; Beatrice, G.; Corey, K.E.; Simon, T.G.; Byrne, C.D.; Targher, G. Non-alcoholic fatty liver disease and risk of fatal and non-fatal cardiovascular events: An updated systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2021, 6, 903–913. [Google Scholar] [CrossRef]
- Wild, S.H.; Walker, J.J.; Morling, J.R.; McAllister, D.A.; Colhoun, H.M.; Farran, B.; McGurnaghan, S.; McCrimmon, R.; Read, S.H.; Sattar, N.; et al. Cardiovascular Disease, Cancer, and Mortality Among People With Type 2 Diabetes and Alcoholic or Nonalcoholic Fatty Liver Disease Hospital Admission. Diabetes Care 2018, 41, 341–347. [Google Scholar] [CrossRef]
- Choi, D.H.; Lee, S.J.; Kang, C.D.; Park, M.O.; Choi, D.W.; Kim, T.S.; Lee, W.; Cho, B.R.; Kim, Y.H.; Lee, B.K.; et al. Nonalcoholic fatty liver disease is associated with coronary artery disease in Koreans. World J. Gastroenterol. 2013, 19, 6453–6457. [Google Scholar] [CrossRef]
- Kim, D.; Choi, S.Y.; Park, E.H.; Lee, W.; Kang, J.H.; Kim, W.; Kim, Y.J.; Yoon, J.H.; Jeong, S.H.; Lee, D.H.; et al. Nonalcoholic fatty liver disease is associated with coronary artery calcification. Hepatology 2012, 56, 605–613. [Google Scholar] [CrossRef]
- Soderberg, C.; Stal, P.; Askling, J.; Glaumann, H.; Lindberg, G.; Marmur, J.; Hultcrantz, R. Decreased survival of subjects with elevated liver function tests during a 28-year follow-up. Hepatology 2010, 51, 595–602. [Google Scholar] [CrossRef]
- Mahfood Haddad, T.; Hamdeh, S.; Kanmanthareddy, A.; Alla, V.M. Nonalcoholic fatty liver disease and the risk of clinical cardiovascular events: A systematic review and meta-analysis. Diabetes Metab. Syndr. 2017, 11 (Suppl. S1), S209–S216. [Google Scholar] [CrossRef] [PubMed]
- Meyersohn, N.M.; Mayrhofer, T.; Corey, K.E.; Bittner, D.O.; Staziaki, P.V.; Szilveszter, B.; Hallett, T.; Lu, M.T.; Puchner, S.B.; Simon, T.G.; et al. Association of Hepatic Steatosis With Major Adverse Cardiovascular Events, Independent of Coronary Artery Disease. Clin. Gastroenterol. Hepatol. 2021, 19, 1480–1488.e14. [Google Scholar] [CrossRef] [PubMed]
- Duell, P.B.; Welty, F.K.; Miller, M.; Chait, A.; Hammond, G.; Ahmad, Z.; Cohen, D.E.; Horton, J.D.; Pressman, G.S.; Toth, P.P.; et al. Nonalcoholic Fatty Liver Disease and Cardiovascular Risk: A Scientific Statement From the American Heart Association. Arterioscler. Thromb. Vasc. Biol. 2022, 42, e168–e185. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Henry, L. Epidemiology of non-alcoholic fatty liver disease and hepatocellular carcinoma. JHEP Rep. 2021, 3, 100305. [Google Scholar] [CrossRef]
- Chen, C.C.; Hsu, W.C.; Wu, H.M.; Wang, J.Y.; Yang, P.Y.; Lin, I.C. Association between the Severity of Nonalcoholic Fatty Liver Disease and the Risk of Coronary Artery Calcification. Medicina 2021, 57, 807. [Google Scholar] [CrossRef]
- Li, X.; Xia, M.; Ma, H.; Hofman, A.; Hu, Y.; Yan, H.; He, W.; Lin, H.; Jeekel, J.; Zhao, N.; et al. Liver fat content is associated with increased carotid atherosclerosis in a Chinese middle-aged and elderly population: The Shanghai Changfeng study. Atherosclerosis 2012, 224, 480–485. [Google Scholar] [CrossRef]
- Sinn, D.H.; Kang, D.; Chang, Y.; Ryu, S.; Gu, S.; Kim, H.; Seong, D.; Cho, S.J.; Yi, B.K.; Park, H.D.; et al. Non-alcoholic fatty liver disease and progression of coronary artery calcium score: A retrospective cohort study. Gut 2017, 66, 323–329. [Google Scholar] [CrossRef]
- Thevenot, T.; Vendeville, S.; Weil, D.; Akkouche, L.; Calame, P.; Canivet, C.M.; Vanlemmens, C.; Richou, C.; Cervoni, J.P.; Seronde, M.F.; et al. Systematic screening for advanced liver fibrosis in patients with coronary artery disease: The CORONASH study. PLoS ONE 2022, 17, e0266965. [Google Scholar] [CrossRef]
- Fiorentino, T.V.; Succurro, E.; Sciacqua, A.; Andreozzi, F.; Perticone, F.; Sesti, G. Non-alcoholic fatty liver disease is associated with cardiovascular disease in subjects with different glucose tolerance. Diabetes Metab. Res. Rev. 2020, 36, e3333. [Google Scholar] [CrossRef]
- Langroudi, T.F.; Haybar, H.; Parsa, S.A.; Mahjoorian, M.; Khaheshi, I.; Naderian, M. The severity of coronary artery disease was not associated with non-alcoholic fatty liver disease in a series of 264 non-diabetic patients who underwent coronary angiography. Rom. J. Intern. Med. 2018, 56, 167–172. [Google Scholar] [CrossRef]
- Ishiba, H.; Sumida, Y.; Kataoka, S.; Kuroda, M.; Akabame, S.; Tomiyasu, K.; Tanaka, M.; Arai, M.; Taketani, H.; Seko, Y.; et al. Association of coronary artery calcification with liver fibrosis in Japanese patients with non-alcoholic fatty liver disease. Hepatol. Res. 2016, 46, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Osawa, K.; Miyoshi, T.; Yamauchi, K.; Koyama, Y.; Nakamura, K.; Sato, S.; Kanazawa, S.; Ito, H. Nonalcoholic Hepatic Steatosis Is a Strong Predictor of High-Risk Coronary-Artery Plaques as Determined by Multidetector CT. PLoS ONE 2015, 10, e0131138. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.W.; Wong, G.L.; Yeung, J.C.; Fung, C.Y.; Chan, J.K.; Chang, Z.H.; Kwan, C.T.; Lam, H.W.; Limquiaco, J.; Chim, A.M.; et al. Long-term clinical outcomes after fatty liver screening in patients undergoing coronary angiogram: A prospective cohort study. Hepatology 2016, 63, 754–763. [Google Scholar] [CrossRef] [PubMed]
- Idilman, I.S.; Akata, D.; Hazirolan, T.; Doganay Erdogan, B.; Aytemir, K.; Karcaaltincaba, M. Nonalcoholic fatty liver disease is associated with significant coronary artery disease in type 2 diabetic patients: A computed tomography angiography study. J. Diabetes 2015, 7, 279–286. [Google Scholar] [CrossRef]
- Puchner, S.B.; Lu, M.T.; Mayrhofer, T.; Liu, T.; Pursnani, A.; Ghoshhajra, B.B.; Truong, Q.A.; Wiviott, S.D.; Fleg, J.L.; Hoffmann, U.; et al. High-risk coronary plaque at coronary CT angiography is associated with nonalcoholic fatty liver disease, independent of coronary plaque and stenosis burden: Results from the ROMICAT II trial. Radiology 2015, 274, 693–701. [Google Scholar] [CrossRef]
- Wong, V.W.; Wong, G.L.; Yip, G.W.; Lo, A.O.; Limquiaco, J.; Chu, W.C.; Chim, A.M.; Yu, C.M.; Yu, J.; Chan, F.K.; et al. Coronary artery disease and cardiovascular outcomes in patients with non-alcoholic fatty liver disease. Gut 2011, 60, 1721–1727. [Google Scholar] [CrossRef]
- Sun, L.; Lu, S.Z. Association between non-alcoholic fatty liver disease and coronary artery disease severity. Chin. Med. J. (Engl.) 2011, 124, 867–872. [Google Scholar]
- Acikel, M.; Sunay, S.; Koplay, M.; Gundogdu, F.; Karakelleoglu, S. Evaluation of ultrasonographic fatty liver and severity of coronary atherosclerosis, and obesity in patients undergoing coronary angiography. Anadolu Kardiyol. Derg. 2009, 9, 273–279. [Google Scholar]
- Carter, J.; Heseltine, T.D.; Meah, M.N.; Tzolos, E.; Kwiecinski, J.; Doris, M.; McElhinney, P.; Moss, A.J.; Adamson, P.D.; Hunter, A.; et al. Hepatosteatosis and Atherosclerotic Plaque at Coronary CT Angiography. Radiol. Cardiothorac. Imaging 2022, 4, e210260. [Google Scholar] [CrossRef]
- Ichikawa, K.; Miyoshi, T.; Osawa, K.; Miki, T.; Toda, H.; Ejiri, K.; Yoshida, M.; Nakamura, K.; Morita, H.; Ito, H. Incremental prognostic value of non-alcoholic fatty liver disease over coronary computed tomography angiography findings in patients with suspected coronary artery disease. Eur. J. Prev. Cardiol. 2022, 28, 2059–2066. [Google Scholar] [CrossRef]
- Ichikawa, K.; Miyoshi, T.; Osawa, K.; Miki, T.; Toda, H.; Ejiri, K.; Yoshida, M.; Nanba, Y.; Yoshida, M.; Nakamura, K.; et al. Prognostic value of non-alcoholic fatty liver disease for predicting cardiovascular events in patients with diabetes mellitus with suspected coronary artery disease: A prospective cohort study. Cardiovasc. Diabetol. 2021, 20, 8. [Google Scholar] [CrossRef] [PubMed]
- Saraya, S.; Saraya, M.; Mahmoud, M.; Galal, M.; Soliman, H.H.; Raafat, M. The associations between coronary artery disease, and non-alcoholic fatty liver disease by computed tomography. Egypt. Heart J. 2021, 73, 96. [Google Scholar] [CrossRef] [PubMed]
- Bae, Y.S.; Ko, Y.S.; Yun, J.M.; Eo, A.Y.; Kim, H. Association and Prediction of Subclinical Atherosclerosis by Nonalcoholic Fatty Liver Disease in Asymptomatic Patients. Can. J. Gastroenterol. Hepatol. 2020, 2020, 8820445. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.K.; Allison, M.A.; Criqui, M.H.; Denenberg, J.O.; Wright, C.M. The association between liver fat and systemic calcified atherosclerosis. J. Vasc. Surg. 2020, 71, 204–211.e204. [Google Scholar] [CrossRef] [PubMed]
- Oni, E.; Budoff, M.J.; Zeb, I.; Li, D.; Veledar, E.; Polak, J.F.; Blankstein, R.; Wong, N.D.; Blaha, M.J.; Agatston, A.; et al. Nonalcoholic Fatty Liver Disease Is Associated With Arterial Distensibility and Carotid Intima-Media Thickness: (from the Multi-Ethnic Study of Atherosclerosis). Am. J. Cardiol. 2019, 124, 534–538. [Google Scholar] [CrossRef]
- Pais, R.; Redheuil, A.; Cluzel, P.; Ratziu, V.; Giral, P. Relationship Among Fatty Liver, Specific and Multiple-Site Atherosclerosis, and 10-Year Framingham Score. Hepatology 2019, 69, 1453–1463. [Google Scholar] [CrossRef]
- Lee, S.B.; Park, G.M.; Lee, J.Y.; Lee, B.U.; Park, J.H.; Kim, B.G.; Jung, S.W.; Jeong, I.D.; Bang, S.J.; Shin, J.W.; et al. Association between non-alcoholic fatty liver disease and subclinical coronary atherosclerosis: An observational cohort study. J. Hepatol. 2018, 68, 1018–1024. [Google Scholar] [CrossRef]
- Kim, J.; Lee, D.Y.; Park, S.E.; Park, C.Y.; Lee, W.Y.; Oh, K.W.; Park, S.W.; Rhee, E.J. Increased risk for development of coronary artery calcification in subjects with non-alcoholic fatty liver disease and systemic inflammation. PLoS ONE 2017, 12, e0180118. [Google Scholar] [CrossRef]
- Wu, R.; Hou, F.; Wang, X.; Zhou, Y.; Sun, K.; Wang, Y.; Liu, H.; Wu, J.; Zhao, R.; Hu, J. Nonalcoholic Fatty Liver Disease and Coronary Artery Calcification in a Northern Chinese Population: A Cross Sectional Study. Sci. Rep. 2017, 7, 9933. [Google Scholar] [CrossRef]
- Kim, B.J.; Kim, H.S.; Kang, J.G.; Kim, B.S.; Kang, J.H. Association of epicardial fat volume and nonalcoholic fatty liver disease with metabolic syndrome: From the CAESAR study. J. Clin. Lipidol. 2016, 10, 1423–1430.e1. [Google Scholar] [CrossRef]
- Park, H.E.; Kwak, M.S.; Kim, D.; Kim, M.K.; Cha, M.J.; Choi, S.Y. Nonalcoholic Fatty Liver Disease Is Associated With Coronary Artery Calcification Development: A Longitudinal Study. J. Clin. Endocrinol. Metab. 2016, 101, 3134–3143. [Google Scholar] [CrossRef] [PubMed]
- Mellinger, J.L.; Pencina, K.M.; Massaro, J.M.; Hoffmann, U.; Seshadri, S.; Fox, C.S.; O’Donnell, C.J.; Speliotes, E.K. Hepatic steatosis and cardiovascular disease outcomes: An analysis of the Framingham Heart Study. J. Hepatol. 2015, 63, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Al Rifai, M.; Silverman, M.G.; Nasir, K.; Budoff, M.J.; Blankstein, R.; Szklo, M.; Katz, R.; Blumenthal, R.S.; Blaha, M.J. The association of nonalcoholic fatty liver disease, obesity, and metabolic syndrome, with systemic inflammation and subclinical atherosclerosis: The Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis 2015, 239, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Park, H.J.; Jeon, W.S.; Park, S.E.; Park, C.Y.; Lee, W.Y.; Oh, K.W.; Park, S.W.; Rhee, E.J. Higher association of coronary artery calcification with non-alcoholic fatty liver disease than with abdominal obesity in middle-aged Korean men: The Kangbuk Samsung Health Study. Cardiovasc. Diabetol. 2015, 14, 88. [Google Scholar] [CrossRef] [PubMed]
- VanWagner, L.B.; Ning, H.; Lewis, C.E.; Shay, C.M.; Wilkins, J.; Carr, J.J.; Terry, J.G.; Lloyd-Jones, D.M.; Jacobs, D.R., Jr.; Carnethon, M.R. Associations between nonalcoholic fatty liver disease and subclinical atherosclerosis in middle-aged adults: The Coronary Artery Risk Development in Young Adults Study. Atherosclerosis 2014, 235, 599–605. [Google Scholar] [CrossRef]
- Juarez-Rojas, J.G.; Medina-Urrutia, A.X.; Jorge-Galarza, E.; Gonzalez-Salazar, C.; Kimura-Hayama, E.; Cardoso-Saldana, G.; Posadas-Sanchez, R.; Martinez-Alvarado, R.; Posadas-Romero, C. Fatty liver increases the association of metabolic syndrome with diabetes and atherosclerosis. Diabetes Care 2013, 36, 1726–1728. [Google Scholar] [CrossRef]
- Sung, K.C.; Lim, Y.H.; Park, S.; Kang, S.M.; Park, J.B.; Kim, B.J.; Shin, J.H. Arterial stiffness, fatty liver and the presence of coronary artery calcium in a large population cohort. Cardiovasc. Diabetol. 2013, 12, 162. [Google Scholar] [CrossRef]
- Sung, K.C.; Wild, S.H.; Kwag, H.J.; Byrne, C.D. Fatty liver, insulin resistance, and features of metabolic syndrome: Relationships with coronary artery calcium in 10,153 people. Diabetes Care 2012, 35, 2359–2364. [Google Scholar] [CrossRef]
- Lin, Y.C.; Lo, H.M.; Chen, J.D. Sonographic fatty liver, overweight and ischemic heart disease. World J. Gastroenterol. 2005, 11, 4838–4842. [Google Scholar] [CrossRef]
- Li, X.L.; Sui, J.Q.; Lu, L.L.; Zhang, N.N.; Xu, X.; Dong, Q.Y.; Xin, Y.N.; Xuan, S.Y. Gene polymorphisms associated with non-alcoholic fatty liver disease and coronary artery disease: A concise review. Lipids Health Dis. 2016, 15, 53. [Google Scholar] [CrossRef]
- Ren, Z.; Simons, P.; Wesselius, A.; Stehouwer, C.D.A.; Brouwers, M. Relationship between NAFLD and coronary artery disease: A Mendelian randomization study. Hepatology 2023, 77, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Miao, Z.; Garske, K.M.; Pan, D.Z.; Koka, A.; Kaminska, D.; Mannisto, V.; Sinsheimer, J.S.; Pihlajamaki, J.; Pajukanta, P. Identification of 90 NAFLD GWAS loci and establishment of NAFLD PRS and causal role of NAFLD in coronary artery disease. HGG Adv. 2022, 3, 100056. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Liu, S.; Wang, M.; Wang, Y.; Xin, Y.; Xuan, S. Relationship between AGT rs2493132 polymorphism and the risk of coronary artery disease in patients with NAFLD in the Chinese Han population. J. Int. Med. Res. 2021, 49, 3000605211019263. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.J.; Peloso, G.M.; Yu, H.; Butterworth, A.S.; Wang, X.; Mahajan, A.; Saleheen, D.; Emdin, C.; Alam, D.; Alves, A.C.; et al. Exome-wide association study of plasma lipids in >300,000 individuals. Nat. Genet. 2017, 49, 1758–1766. [Google Scholar] [CrossRef]
- Tang, C.S.; Zhang, H.; Cheung, C.Y.; Xu, M.; Ho, J.C.; Zhou, W.; Cherny, S.S.; Zhang, Y.; Holmen, O.; Au, K.W.; et al. Exome-wide association analysis reveals novel coding sequence variants associated with lipid traits in Chinese. Nat. Commun. 2015, 6, 10206. [Google Scholar] [CrossRef]
- Rinella, M.E.; Neuschwander-Tetri, B.A.; Siddiqui, M.S.; Abdelmalek, M.F.; Caldwell, S.; Barb, D.; Kleiner, D.E.; Loomba, R. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology 2023, 77, 1797–1835. [Google Scholar] [CrossRef]
- Francque, S.; Vonghia, L. Pharmacological Treatment for Non-alcoholic Fatty Liver Disease. Adv. Ther. 2019, 36, 1052–1074. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Friedman, S.L.; McCullough, A.J.; Dimick-Santos, L. Challenges and opportunities in drug and biomarker development for nonalcoholic steatohepatitis: Findings and recommendations from an American Association for the Study of Liver Diseases-U.S. Food and Drug Administration Joint Workshop. Hepatology 2015, 61, 1392–1405. [Google Scholar] [CrossRef]
- Stahl, E.P.; Dhindsa, D.S.; Lee, S.K.; Sandesara, P.B.; Chalasani, N.P.; Sperling, L.S. Nonalcoholic Fatty Liver Disease and the Heart: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 948–963. [Google Scholar] [CrossRef]
- Neuschwander-Tetri, B.A.; Loomba, R.; Sanyal, A.J.; Lavine, J.E.; Van Natta, M.L.; Abdelmalek, M.F.; Chalasani, N.; Dasarathy, S.; Diehl, A.M.; Hameed, B.; et al. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): A multicentre, randomised, placebo-controlled trial. Lancet 2015, 385, 956–965. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Stepanova, M.; Nader, F.; Loomba, R.; Anstee, Q.M.; Ratziu, V.; Harrison, S.; Sanyal, A.J.; Schattenberg, J.M.; Barritt, A.S.; et al. Obeticholic Acid Impact on Quality of Life in Patients With Nonalcoholic Steatohepatitis: REGENERATE 18-Month Interim Analysis. Clin. Gastroenterol. Hepatol. 2022, 20, 2050–2058.e2012. [Google Scholar] [CrossRef] [PubMed]
- Rinella, M.E.; Dufour, J.F.; Anstee, Q.M.; Goodman, Z.; Younossi, Z.; Harrison, S.A.; Loomba, R.; Sanyal, A.J.; Bonacci, M.; Trylesinski, A.; et al. Non-invasive evaluation of response to obeticholic acid in patients with NASH: Results from the REGENERATE study. J. Hepatol. 2022, 76, 536–548. [Google Scholar] [CrossRef] [PubMed]
- Ratziu, V.; Harrison, S.A.; Francque, S.; Bedossa, P.; Lehert, P.; Serfaty, L.; Romero-Gomez, M.; Boursier, J.; Abdelmalek, M.; Caldwell, S.; et al. Elafibranor, an Agonist of the Peroxisome Proliferator-Activated Receptor-alpha and -delta, Induces Resolution of Nonalcoholic Steatohepatitis Without Fibrosis Worsening. Gastroenterology 2016, 150, 1147–1159.e1145. [Google Scholar] [CrossRef] [PubMed]
- Ratziu, V.; Harrison, S.A.; Hajji, Y.; Magnanensi, J.; Petit, S.; Majd, Z.; Delecroix, E.; Rosenquist, C.; Hum, D.; Staels, B.; et al. NIS2+(TM) as a screening tool to optimize patient selection in metabolic dysfunction-associated steatohepatitis clinical trials. J. Hepatol. 2024, 80, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Friedman, S.L.; Ratziu, V.; Harrison, S.A.; Abdelmalek, M.F.; Aithal, G.P.; Caballeria, J.; Francque, S.; Farrell, G.; Kowdley, K.V.; Craxi, A.; et al. A randomized, placebo-controlled trial of cenicriviroc for treatment of nonalcoholic steatohepatitis with fibrosis. Hepatology 2018, 67, 1754–1767. [Google Scholar] [CrossRef]
- Harrison, S.A.; Bedossa, P.; Guy, C.D.; Schattenberg, J.M.; Loomba, R.; Taub, R.; Labriola, D.; Moussa, S.E.; Neff, G.W.; Rinella, M.E.; et al. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N. Engl. J. Med. 2024, 390, 497–509. [Google Scholar] [CrossRef]
| First Author | Year of Publication | Number of Patients | NAFLD and CAD Diagnostic Method | Results |
|---|---|---|---|---|
| Thévenot [68] | 2022 | 189 | Non-invasive fibrosis tests, Fibroscan and coronary angiography | 5% of patients with documented CAD had NAFLD. |
| Hsu [33] | 2021 | 1502 | Abdominal ultrasonography and cardiac computed tomography | NAFLD severity is associated with coronary artery atherosclerosis burden, and steatosis severity correlated with the risk of presence of coronary plaques. |
| Fiorentino [69] | 2020 | 1254 | Abdominal ultrasonography and coronary angiography | NAFLD independently associated with an increased risk of having CVD in patients with glucose disturbances. |
| Langroudi [70] | 2018 | 264 | Abdominal ultrasonography and cardiac computed tomography | NAFLD may NOT be associated with atherosclerosis of coronary arteries and its severity in non-diabetic patients. |
| Sinn [67] | 2017 | 4731 | Abdominal ultrasonography and cardiac computed tomography | NAFLD was significantly associated with the development of CAD. |
| Ishiba [71] | 2016 | 698 | Abdominal and cardiac computed tomography | The progression of arteriosclerosis and that of liver fibrosis may be associated in NAFLD patients. |
| Osawa [72] | 2015 | 414 | Abdominal and cardiac computed tomography | Diagnosis of NAFLD may be important when assessing the risk of CAD. |
| Wong [73] | 2015 | 612 | Abdominal ultrasonography and coronary angiography | NAFLD is associated with CAD and need for coronary intervention, but not increased mortality or cardiovascular complications. |
| Idilman [74] | 2015 | 273 | Abdominal and cardiac computed tomography | NAFLD is associated with significant CAD in type 2 diabetic patients. |
| Puchner [75] | 2014 | 445 | Abdominal and cardiac computed tomography | NAFLD is associated with advanced high-risk coronary plaque. |
| Wong [76] | 2011 | 612 | Abdominal ultrasonography and coronary angiography | NAFLD is associated with CAD independently of other metabolic factors. |
| Sun [77] | 2011 | 542 | Abdominal and cardiac computed tomography | NAFLD is associated with high severity of CAD, requiring consciousness in patients with abdominal obesity. |
| Açikel [78] | 2009 | 355 | Abdominal ultrasonography and coronary angiography | NAFLD is associated with the presence of CAD and severity of coronary atherosclerosis. |
| First Author | Year of Publication | Number of Patients | NAFLD and CAD Diagnostic Method | Results |
|---|---|---|---|---|
| Carter [79] | 2022 | 1726 | Abdominal and cardiac computed tomography | Hepatosteatosis was associated with an increased prevalence of CAD. |
| Ichikawa [80] | 2022 | 1148 | Abdominal and cardiac computed tomography | Stable CAD and concurrent evaluation of NAFLD enables more accurate detection of patients at higher risk of MACEs. |
| Chen [65] | 2021 | 545 | Abdominal ultrasonography and cardiac computed tomography | NAFLD was associated with the presence of significant CAD. |
| Ichikawa [81] | 2021 | 529 | Abdominal and cardiac computed tomography | NAFLD in addition to CAD and risk factors, could be useful for identifying diabetic patients at risk of MACEs. |
| Meyersohn [62] | 2021 | 3756 | Abdominal and cardiac computed tomography | Hepatic steatosis is associated with MACEs independently of other cardiovascular risk factors or extent of coronary artery disease. Strategies to reduce steatosis might reduce risk of MACEs. |
| Saraya [82] | 2021 | 800 | Abdominal and cardiac computed tomography | NAFLD is associated with CAD. |
| Bae [83] | 2020 | 3693 | Abdominal ultrasonography and cardiac computed tomography | NAFLD was associated with CAD. |
| Koo [84] | 2020 | 719 | Abdominal and cardiac computed tomography | The association between NAFLD and arterial calcification is mainly mediated by conventional risk factors. |
| Oni [85] | 2019 | 4123 | Abdominal and cardiac computed tomography | NAFLD is associated with carotid atherosclerosis and CAD. |
| Pais [86] | 2019 | 2617 | Abdominal ultrasonography and cardiac computed tomography | Steatosis is associated with carotid and coronary, but not femoral atherosclerosis, and with cardiovascular mortality risk. |
| Sinn [67] | 2019 | 111,492 | Abdominal ultrasonography and cardiac computed tomography | NAFLD was associated with the development of CAD independent of cardiovascular and metabolic risk factors. |
| Lee [87] | 2018 | 5121 | Abdominal ultrasonography and cardiac computed tomography | NAFLD was associated with non-calcified plaque. |
| Kim [88] | 2017 | 1575 | Abdominal and cardiac computed tomography | NAFLD and systemic inflammation increases the risk of CAD over 4 years. |
| Wu [89] | 2017 | 2345 | Abdominal ultrasonography and cardiac computed tomography | NAFLD and CAD associations were significant in female but not in male. |
| Kim [90] | 2016 | 1472 | Abdominal ultrasonography and cardiac computed tomography | Epicardial fat volume and NAFLD are associated with the presence of metabolic syndrome. |
| Park [91] | 2016 | 1732 | Abdominal ultrasonography and cardiac computed tomography | NAFLD was associated with the early development of CAD, but not the progression. |
| Mellinger [92] | 2015 | 3529 | Abdominal and cardiac computed tomography | Association of hepatic steatosis with subclinical CVD. |
| Al Rifai [93] | 2015 | 3976 | Abdominal and cardiac computed tomography | NAFLD is associated with increased inflammation and CAD, independent of traditional risk factors, obesity, and metabolic syndrome. |
| Lee [94] | 2015 | 10,063 | Abdominal ultrasonography and cardiac computed tomography | NAFLD is associated with CAD. |
| VanWagner [95] | 2014 | 2424 | Abdominal and cardiac computed tomography | Obesity attenuates the relationship between NAFLD and subclinical atherosclerosis. |
| Juarez-Rojas [96] | 2013 | 765 | Abdominal and cardiac computed tomography | NAFLD increases the association of metabolic syndrome with type 2 diabetes and subclinical atherosclerosis. |
| Sung [97] | 2013 | 7371 | Abdominal ultrasonography and cardiac computed tomography | Both NAFLD and brachial-ankle pulse wave velocity are associated with CAD. |
| Kim [59] | 2012 | 4023 | Abdominal and cardiac computed tomography | NAFLD patients are at increased risk for CAD. |
| Sung [98] | 2012 | 3784 | Abdominal and cardiac computed tomography | NAFLD and insulin resistance are both associated with CAD. |
| Lin [99] | 2005 | 2088 | Abdominal ultrasonography and digital electrocardiography | Synergistic effect between NAFLD and overweight for developing ischemic heart disease. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ktenopoulos, N.; Sagris, M.; Gerogianni, M.; Pamporis, K.; Apostolos, A.; Balampanis, K.; Tsioufis, K.; Toutouzas, K.; Tousoulis, D. Non-Alcoholic Fatty Liver Disease and Coronary Artery Disease: A Bidirectional Association Based on Endothelial Dysfunction. Int. J. Mol. Sci. 2024, 25, 10595. https://doi.org/10.3390/ijms251910595
Ktenopoulos N, Sagris M, Gerogianni M, Pamporis K, Apostolos A, Balampanis K, Tsioufis K, Toutouzas K, Tousoulis D. Non-Alcoholic Fatty Liver Disease and Coronary Artery Disease: A Bidirectional Association Based on Endothelial Dysfunction. International Journal of Molecular Sciences. 2024; 25(19):10595. https://doi.org/10.3390/ijms251910595
Chicago/Turabian StyleKtenopoulos, Nikolaos, Marios Sagris, Maria Gerogianni, Konstantinos Pamporis, Anastasios Apostolos, Konstantinos Balampanis, Konstantinos Tsioufis, Konstantinos Toutouzas, and Dimitris Tousoulis. 2024. "Non-Alcoholic Fatty Liver Disease and Coronary Artery Disease: A Bidirectional Association Based on Endothelial Dysfunction" International Journal of Molecular Sciences 25, no. 19: 10595. https://doi.org/10.3390/ijms251910595
APA StyleKtenopoulos, N., Sagris, M., Gerogianni, M., Pamporis, K., Apostolos, A., Balampanis, K., Tsioufis, K., Toutouzas, K., & Tousoulis, D. (2024). Non-Alcoholic Fatty Liver Disease and Coronary Artery Disease: A Bidirectional Association Based on Endothelial Dysfunction. International Journal of Molecular Sciences, 25(19), 10595. https://doi.org/10.3390/ijms251910595











