Synthesis and Antibacterial Activity of Mono- and Bi-Cationic Pyridinium 1,2,4-Oxadiazoles and Triazoles
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
2.2. Antibacterial Activity
3. Material and Methods
3.1. Materials
3.2. Methods
3.3. Synthesis and Characterization
3.3.1. General Procedure for the Synthesis of Compounds 11c and 16c
- N-Methyl-4-(5-pentadecafluoroheptyl-1,2,4-oxadiazol-3-yl)pyridinium tetrafluoroborate (11c)
- N-Methyl-4-(3-pentadecafluoroheptyl-1,2,4-oxadiazol-5-yl)pyridinium tetrafluoroborate (16c)
3.3.2. General Procedure for the Synthesis of Compounds 11d and 16d
- N-Methyl-4-(5-pentadecafluoroheptyl-1,2,4-oxadiazol-3-yl)pyridinium hexafluorophosphate (11d)
- N-Methyl-4-(3-pentadecafluoroheptyl-1,2,4-oxadiazol-5-yl)pyridinium hexafluorophosphate (16d)
3.3.3. General Procedure for the Synthesis of Compounds 11e and 16e
- N-Methyl-4-(5-pentadecafluoroheptyl-1,2,4-oxadiazol-3-yl)pyridinium bis((trifluoromethyl)sulfonyl)amide (11e)
- N-Methyl-4-(3-pentadecafluoroheptyl-1,2,4-oxadiazol-5-yl)pyridinium bis((trifluoromethyl)sulfonyl)amide (16e)
3.3.4. Synthesis of 1-Methyl-4-(5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl)pyridin-1-ium trifluoromethanesulfonate (13)
3.3.5. Synthesis of 4,4’-(1,2,4-Oxadiazole-3,5-diyl)bis(1-heptylpyridin-1-ium) iodide (26)
3.4. Antimicrobial Activity According to Minimum Inhibitory Concentrations (MICs) Determination
3.5. Biofilm Inhibition Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cox, G.; Wright, G.D. Intrinsic antibiotic resistance: Mechanisms, origins, challenges and solutions. Int. J. Med. Microbiol. IJMM 2013, 303, 287–292. [Google Scholar] [CrossRef]
- Banin, E.; Hughes, D.; Kuipers, O.P. Editorial: Bacterial pathogens, antibiotics and antibiotic resistance. FEMS Microbiol. Rev. 2017, 41, 450–452. [Google Scholar] [CrossRef] [PubMed]
- Holmes, A.H.; Moore, L.S.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Munita, J.M.; Arias, C.A. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016, 4, vmbf-0016-2015. [Google Scholar] [CrossRef] [PubMed]
- WHO. Strategic Priorities on Antimicrobial Resistance. Available online: https://www.who.int/publications/i/item/9789240041387 (accessed on 20 December 2023).
- Pulingam, T.; Parumasivam, T.; Gazzali, A.M.; Sulaiman, A.M.; Chee, J.Y.; Lakshmanan, M.; Chin, C.F.; Sudesh, K. Antimicrobial resistance: Prevalence, economic burden, mechanisms of resistance and strategies to overcome. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2022, 170, 106103. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Antimicrobial Resistance and Use Surveillance System (GLASS) Report. 2022. Available online: https://www.who.int/publications/i/item/9789240062702 (accessed on 20 December 2023).
- Hays, J.P.; Ruiz-Alvarez, M.J.; Roson-Calero, N.; Amin, R.; Murugaiyan, J.; van Dongen, M.B.M. Perspectives on the Ethics of Antibiotic Overuse and on the Implementation of (New) Antibiotics. Infect. Dis. Ther. 2022, 11, 1315–1326. [Google Scholar] [CrossRef] [PubMed]
- Bush, K.; Courvalin, P.; Dantas, G.; Davies, J.; Eisenstein, B.; Huovinen, P.; Jacoby, G.A.; Kishony, R.; Kreiswirth, B.N.; Kutter, E.; et al. Tackling antibiotic resistance. Nat. Rev. Microbiol. 2011, 9, 894–896. [Google Scholar] [CrossRef]
- Hulscher, M.E.; van der Meer, J.W.; Grol, R.P. Antibiotic use: How to improve it? Int. J. Med. Microbiol. IJMM 2010, 300, 351–356. [Google Scholar] [CrossRef]
- Mao, X.; Auer, D.L.; Buchalla, W.; Hiller, K.A.; Maisch, T.; Hellwig, E.; Al-Ahmad, A.; Cieplik, F. Cetylpyridinium Chloride: Mechanism of Action, Antimicrobial Efficacy in Biofilms, and Potential Risks of Resistance. Antimicrob. Agents Chemother. 2020, 64, e00576-20. [Google Scholar] [CrossRef]
- Quisno, R.; Foter, M.J. Cetyl pyridinium chloride; germicidal properties. J. Bacteriol. 1946, 52, 111–117. [Google Scholar] [CrossRef]
- Gilbert, P.; Moore, L.E. Cationic antiseptics: Diversity of action under a common epithet. J. Appl. Microbiol. 2005, 99, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Sun, G. Hydrophobicity and antimicrobial activities of quaternary pyridinium salts. J. Appl. Microbiol. 2008, 104, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Jennings, M.C.; Minbiole, K.P.; Wuest, W.M. Quaternary Ammonium Compounds: An Antimicrobial Mainstay and Platform for Innovation to Address Bacterial Resistance. ACS Infect. Dis. 2015, 1, 288–303. [Google Scholar] [CrossRef] [PubMed]
- Marinescu, M.; Popa, C.-V. Pyridine Compounds with Antimicrobial and Antiviral Activities. Int. J. Mol. Sci. 2022, 23, 5659. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P.; Al-taae, A. Antimicrobial activity of some alkyltrimethylammonium bromides. Lett. Appl. Microbiol. 1985, 1, 101–104. [Google Scholar] [CrossRef]
- Dan, W.; Gao, J.; Qi, X.; Wang, J.; Dai, J. Antibacterial quaternary ammonium agents: Chemical diversity and biological mechanism. Eur. J. Med. Chem. 2022, 243, 114765. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhou, S.; Zhang, X.; Zeng, S.; Xu, Y. Quaternary Ammonium Salts: Insights into Synthesis and New Directions in Antibacterial Applications. Bioconjugate Chem. 2023, 34, 302–325. [Google Scholar] [CrossRef]
- Paniak, T.J.; Jennings, M.C.; Shanahan, P.C.; Joyce, M.D.; Santiago, C.N.; Wuest, W.M.; Minbiole, K.P.C. The antimicrobial activity of mono-, bis-, tris-, and tetracationic amphiphiles derived from simple polyamine platforms. Bioorganic Med. Chem. Lett. 2014, 24, 5824–5828. [Google Scholar] [CrossRef]
- Taleb, K.; Mohamed-Benkada, M.; Benhamed, N.; Saidi-Besbes, S.; Grohens, Y.; Derdour, A. Benzene ring containing cationic gemini surfactants: Synthesis, surface properties and antibacterial activity. J. Mol. Liq. 2017, 241, 81–90. [Google Scholar] [CrossRef]
- Md Moshikur, R.; Chowdhury, M.R.; Moniruzzaman, M.; Goto, M. Biocompatible ionic liquids and their applications in pharmaceutics. Green Chem. 2020, 22, 8116–8139. [Google Scholar] [CrossRef]
- Fang, Z.; Zheng, X.; Li, L.; Qi, J.; Wu, W.; Lu, Y. Ionic Liquids: Emerging Antimicrobial Agents. Pharm. Res. 2022, 39, 2391–2404. [Google Scholar] [CrossRef]
- Costa, F.M.S.; Saraiva, M.L.M.F.S.; Passos, M.L.C. Ionic liquids and organic salts with antimicrobial activity as a strategy against resistant microorganisms. J. Mol. Liq. 2022, 368, 120750. [Google Scholar] [CrossRef]
- Vereshchagin, A.N.; Frolov, N.A. Quaternary Ammonium Compounds (QACs) and Ionic Liquids (ILs) as Biocides: From Simple Antiseptics to Tunable Antimicrobials. Int. J. Mol. Sci. 2021, 22, 6793. [Google Scholar] [CrossRef] [PubMed]
- Avellone, G.; Bongiorno, D.; Buscemi, S.; Ceraulo, L.; Indelicato, S.; Pace, A.; Pibiri, I.; Vivona, N. Characterization of Isomeric 1,2,4-Oxadiazolyl-N-Methylpyridinium Salts by Electrospray Ionization Tandem Mass Spectrometry. Eur. J. Mass Spectrom. 2007, 13, 199–205. [Google Scholar] [CrossRef]
- Pibiri, I.; Pace, A.; Buscemi, S.; Causin, V.; Rastrelli, F.; Saielli, G. Oxadiazolyl-pyridines and perfluoroalkyl-carboxylic acids as building blocks for protic ionic liquids: Crossing the thin line between ionic and hydrogen bonded materials. Phys. Chem. Chem. Phys. PCCP 2012, 14, 14306–14314. [Google Scholar] [CrossRef] [PubMed]
- Pibiri, I.; Beneduci, A.; Carraro, M.; Causin, V.; Casella, G.; Corrente, G.A.; Chidichimo, G.; Pace, A.; Riccobono, A.; Saielli, G. Mesomorphic and electrooptical properties of viologens based on non-symmetric alkyl/polyfluoroalkyl functionalization and on an oxadiazolyl-extended bent core. J. Mater. Chem. C 2019, 7, 7974–7983. [Google Scholar] [CrossRef]
- Lo Celso, F.; Pibiri, I.; Triolo, A.; Triolo, R.; Pace, A.; Buscemi, S.; Vivona, N. Study on the thermotropic properties of highly fluorinated 1,2,4-oxadiazolylpyridinium salts and their perspective applications as ionic liquid crystals. J. Mater. Chem. 2007, 17, 1201–1208. [Google Scholar] [CrossRef]
- Weber, M.S.; Schulze, M.; Lazzara, G.; Palumbo Piccionello, A.; Pace, A.; Pibiri, I. Oxadiazolyl-Pyridinium as Cationic Scaffold for Fluorinated Ionic Liquid Crystals. Appl. Sci. 2021, 11, 10347. [Google Scholar] [CrossRef]
- Pibiri, I.; Pace, A.; Piccionello, A.P.; Pierro, P.; Buscemi, S. Synthesis and Characterization of a Series of Alkyl-oxadiazolylpyridinium Salts as Perspective Ionic Liquids. Heterocycles 2006, 68, 2653–2661. [Google Scholar] [CrossRef]
- Pibiri, I.; Pace, A.; Buscemi, S.; Vivona, N.; Malpezzi, L. Designing fluorous domains. Synthesis of a series of pyridinium salts bearing a perfluoroalkylated azole moiety. Heterocycles 2006, 68, 307–322. [Google Scholar]
- Mangione, M.R.; Palumbo Piccionello, A.; Marino, C.; Ortore, M.G.; Picone, P.; Vilasi, S.; Di Carlo, M.; Buscemi, S.; Bulone, D.; San Biagio, P.L. Photo-inhibition of Aβ fibrillation mediated by a newly designed fluorinated oxadiazole. RSC Adv. 2015, 5, 16540–16548. [Google Scholar] [CrossRef]
- Piccionello, A.P.; Pace, A.; Buscemi, S. Rearrangements of 1,2,4-Oxadiazole: “One Ring to Rule Them All”. Chem. Heterocycl. Compd. 2017, 53, 936–947. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed]
- Scutt, J.N.W.; James, N.; Delaney, J.S. Herbicidal Compounds; W.I.P. Organization: Macatawa, MI, USA, 2020. [Google Scholar]
- Palumbo Piccionello, A.; Guarcello, A.; Calabrese, A.; Pibiri, I.; Pace, A.; Buscemi, S. Synthesis of fluorinated oxadiazoles with gelation and oxygen storage ability. Org. Biomol. Chem. 2012, 10, 3044–3052. [Google Scholar] [CrossRef]
- O’Toole, G.; Kaplan, H.B.; Kolter, R. Biofilm Formation as Microbial Development. Annu. Rev. Microbiol. 2000, 54, 49–79. [Google Scholar] [CrossRef]
- Stepanovic, S.; Vukovic, D.; Dakic, I.; Savic, B.; Svabic-Vlahovic, M. A modified microtiter-plate test for quantification of staphylococcal biofilm formation. J. Microbiol. Methods 2000, 40, 175–179. [Google Scholar] [CrossRef]
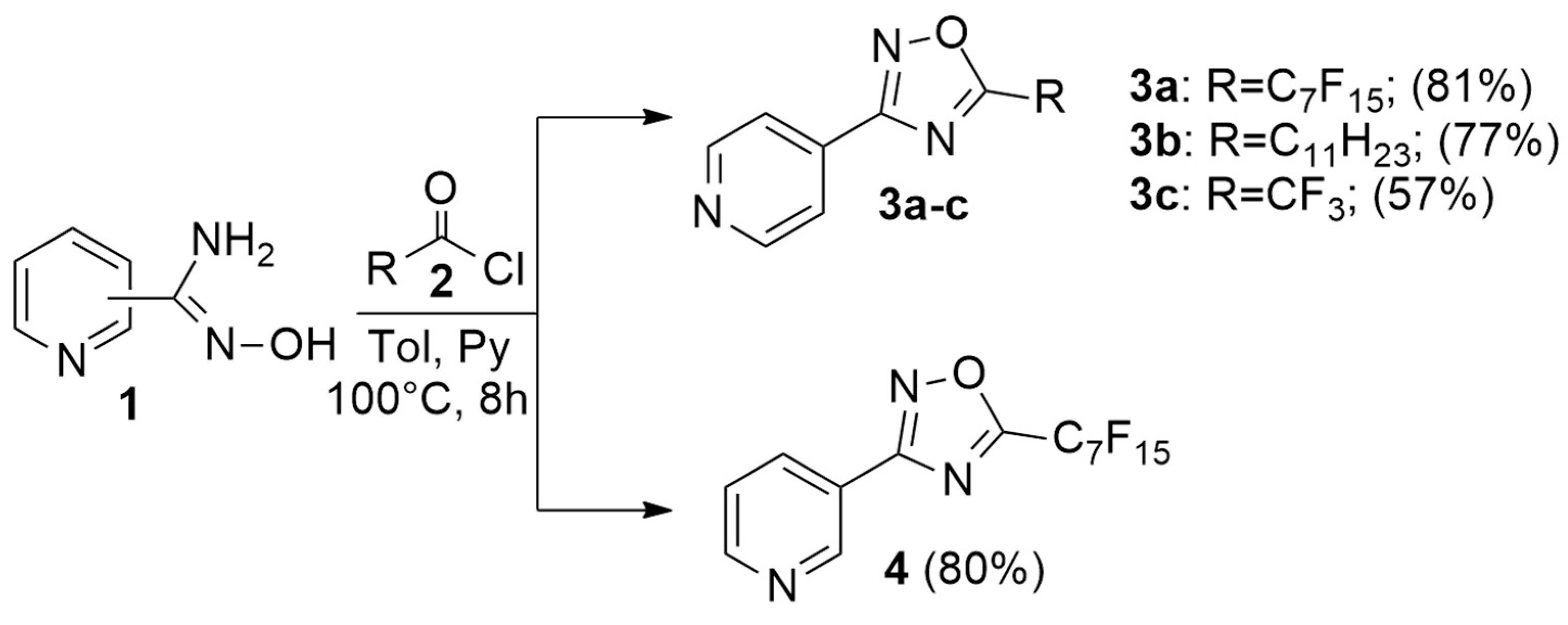
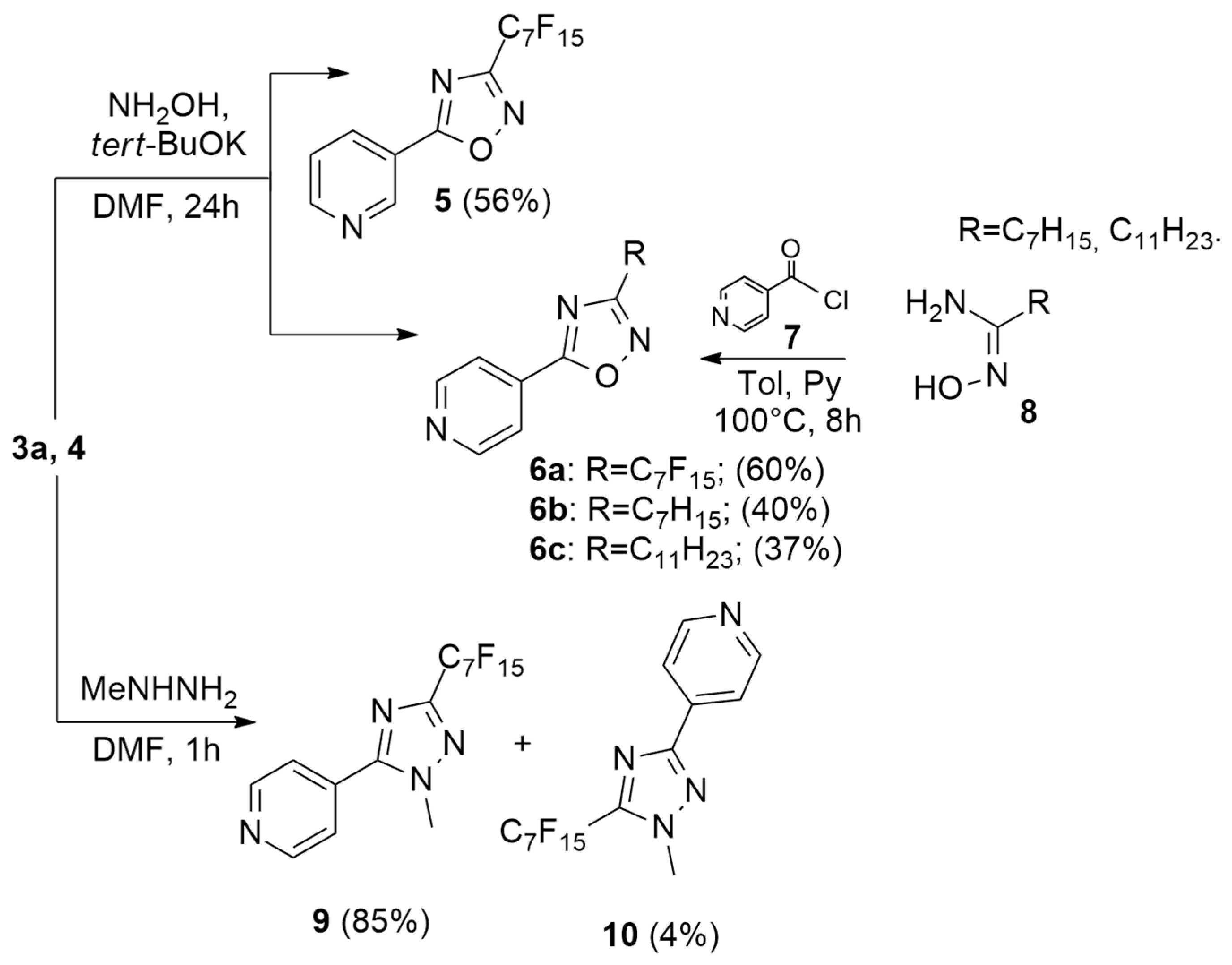
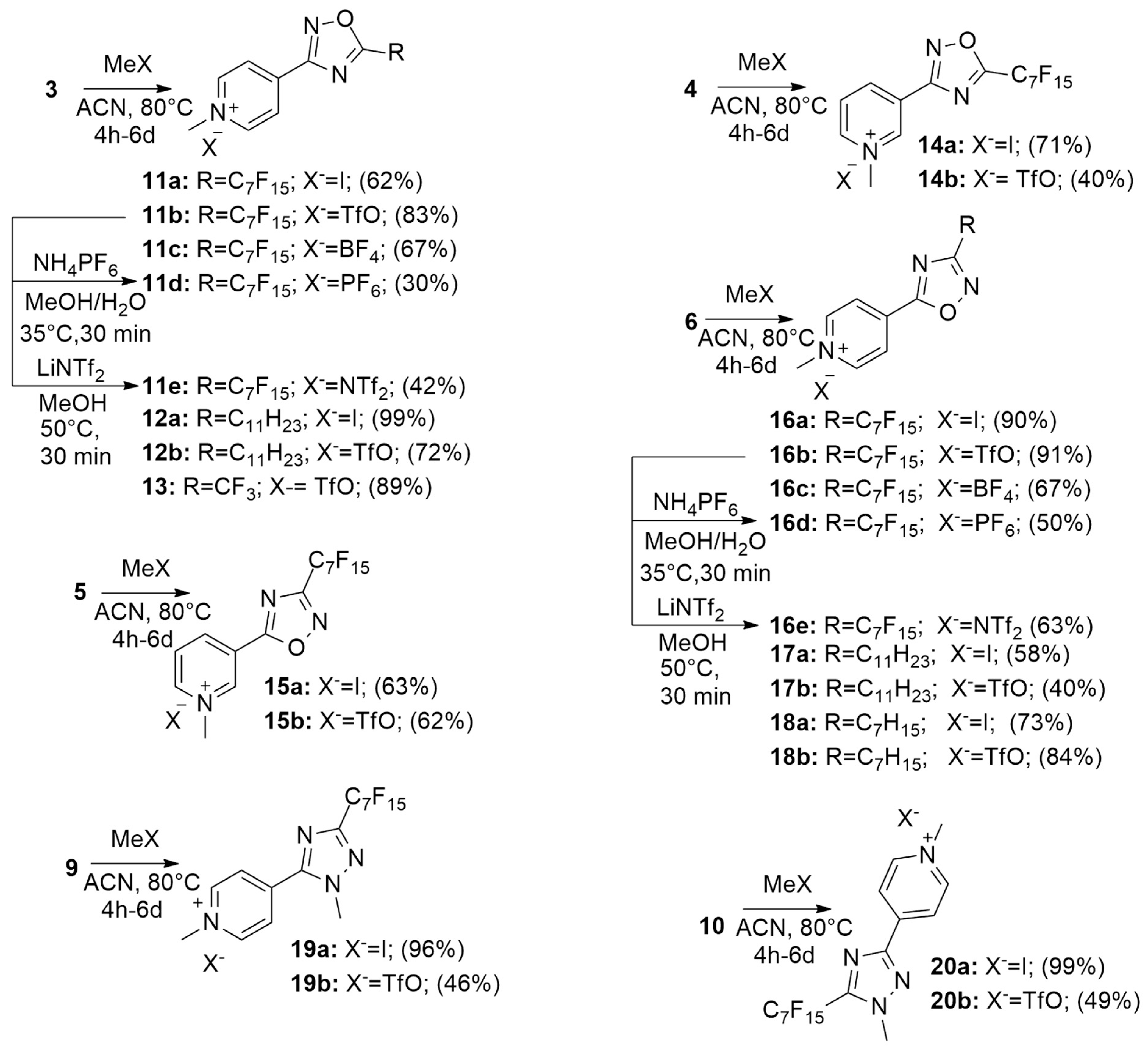
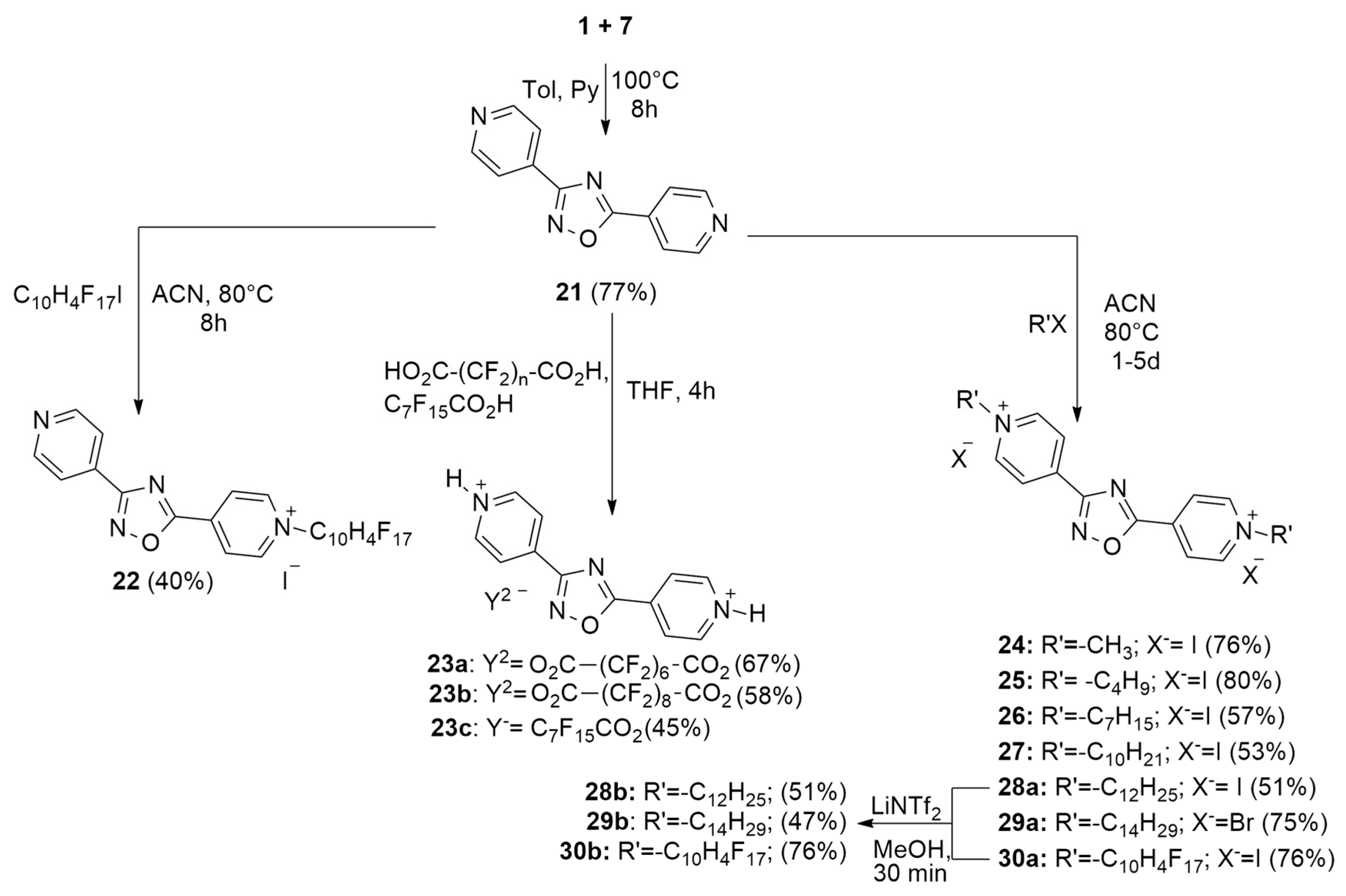
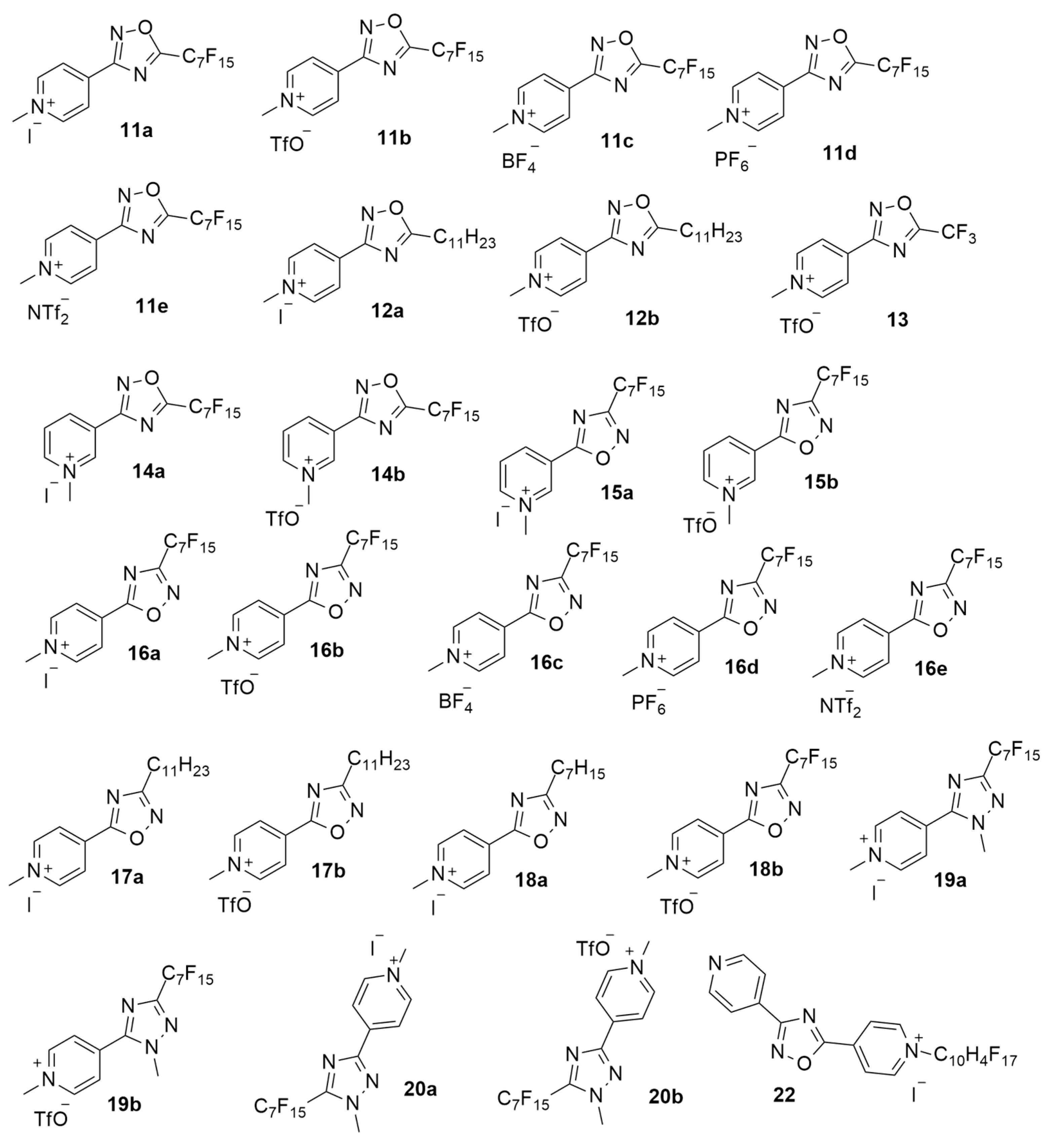
| Compound | R | X− | MIC (µg/mL) S. aureus a | MIC (µg/mL) E. coli b |
|---|---|---|---|---|
| 11a | C7F15 | I | >64 | >64 |
| 11b | C7F15 | TfO | 16 | >64 |
| 11c | C7F15 | BF4 | 16 | >64 |
| 11d | C7F15 | PF6 | 16 | >64 |
| 11e | C7F15 | NTf2 | 16 | >64 |
| 12a | C11H23 | I | 2 | >64 |
| 12b | C11H23 | TfO | 2 | >64 |
| 13 | CF3 | TfO | >64 | >64 |
| 14a | C7F15 | I | >64 | >64 |
| 14b | C7F15 | TfO | >64 | >64 |
| 15a | C7F15 | I | >64 | >64 |
| 15b | C7F15 | TfO | >64 | >64 |
| 16a | C7F15 | I | 8 | >64 |
| 16b | C7F15 | TfO | 8 | >64 |
| 16c | C7F15 | BF4 | 8 | >64 |
| 16d | C7F15 | PF6 | 8 | 64 |
| 16e | C7F15 | NTf2 | 16 | >64 |
| 17a | C11H23 | I | 1 | 64 |
| 17b | C11H23 | TfO | 2 | >64 |
| 18a | C7H15 | I | >64 | >64 |
| 18b | C7H15 | TfO | >64 | >64 |
| 19a | C7F15 | I | 8 | >64 |
| 19b | C7F15 | TfO | 8 | >64 |
| 20a | C7F15 | I | 8 | >64 |
| 20b | C7F15 | TfO | 8 | >64 |
| 22 | C10H4F17 | I | 2 | >64 |
| CIP | - | - | ≤0.5 | - |
| LZD | - | - | 1 | - |
| LVX | - | - | - | ≤0.5 |
| TZP | - | - | - | ≤4/4 |
| Compound | R | X | MIC (µg/mL) S. aureus a | MIC (µg/mL) E. coli b |
|---|---|---|---|---|
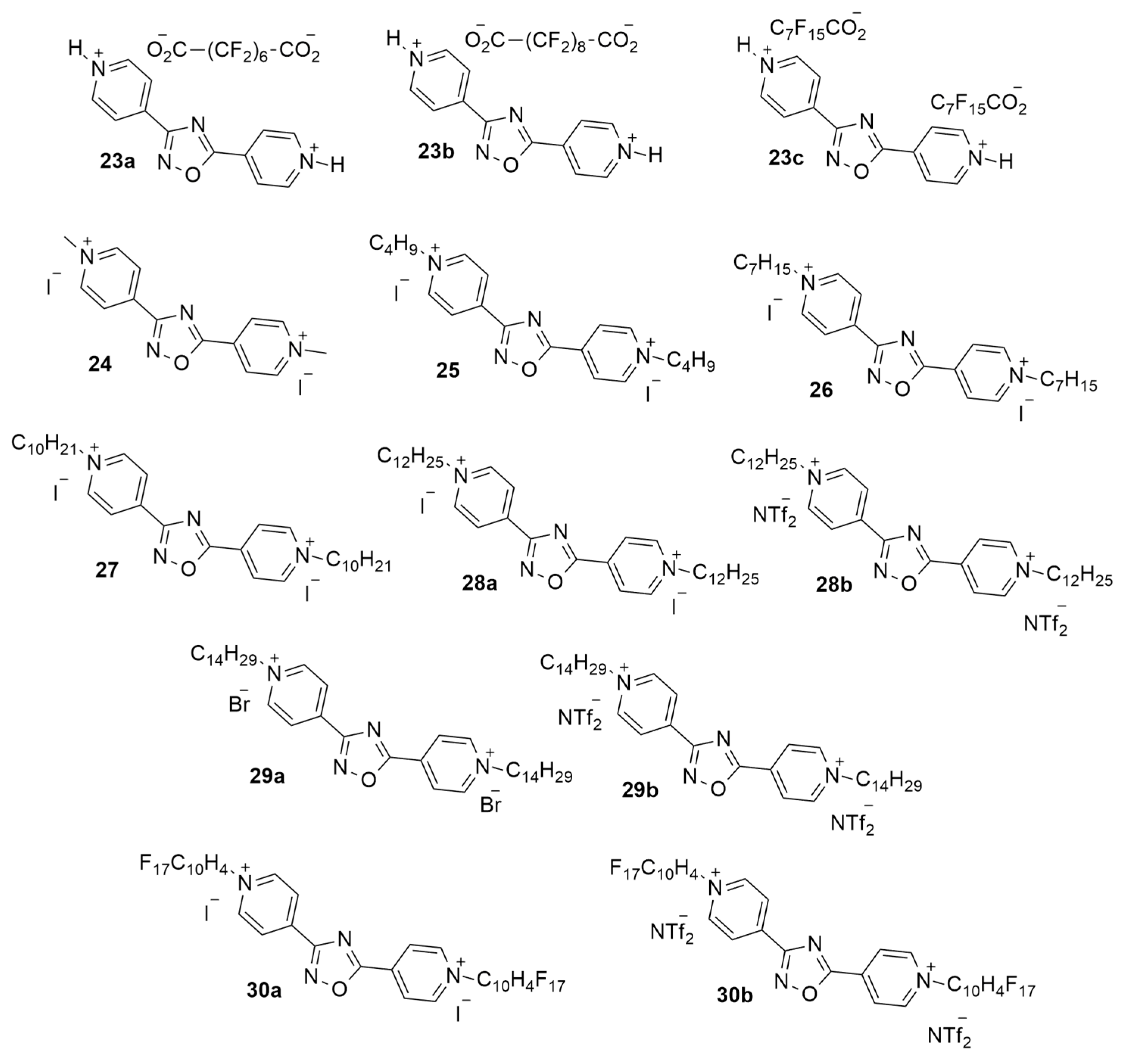
| 23a | H | O2C(CF2)6CO2 | >64 | >64 |
| 23b | H | O2C(CF2)8CO2 | >64 | >64 |
| 23c | H | C7F15CO2 | >64 | >64 |
| 24 | CH3 | I | >64 | >64 |
| 25 | C4H9 | I | >64 | >64 |
| 26 | C7H15 | I | >64 | >64 |
| 27 | C10H21 | I | 0.25 | 0.5 |
| 28a | C12H25 | I | 0.25 | 4 |
| 28b | C12H25 | NTf2 | 0.25 | 4 |
| 29a | C14H29 | Br | 2 | >64 |
| 29b | C14H29 | NTf2 | 4 | >64 |
| 30a | C10H4F17 | I | >64 | >64 |
| 30b | C10H4F17 | NTf2 | >64 | >64 |
| CIP | - | - | ≤0.5 | - |
| LZD | - | - | 1 | - |
| LVX | - | - | - | ≤0.5 |
| TZP | - | - | - | ≤4/4 |
| Strain | MIC 17a | MIC 17b | MIC 12b | MIC 12a | MIC 27 | MIC 28a | MIC CIP | MIC LZD |
|---|---|---|---|---|---|---|---|---|
| S. aureus 32081873 | 8 | 8 | 8 | 8 | 2 | 2 | >4 | 1 |
| S. aureus 32106008 | 2 | 8 | 2 | 2 | 1 | 2 | ≤0.5 | 1 |
| S. aureus 32116042 | 1 | 2 | 2 | 2 | 1 | 1 | ≤0.5 | 1 |
| S. epidermidis 32080691 | 4 | 4 | 4 | 8 | 1 | 1 | 4 | 2 |
| S. epidermidis 32078856 | 4 | 16 | 16 | 8 | 0.5 | 1 | 4 | 2 |
| S. haemolyticus 32105436 | 8 | 16 | 16 | 16 | 1 | 1 | >4 | 1 |
| S. haemolyticus 32076211 | 8 | 8 | 8 | 8 | 0.5 | 1 | >4 | 2 |
| Strain | MIC (µg/mL) 27 | MIC (µg/mL) LVX | MIC (µg/mL) TZP |
|---|---|---|---|
| K. pneumoniae 32084798 | 16 | >8 | 32/4 |
| K. pneumoniae 32084853 | 16 | >8 | >32/4 |
| K. pneumoniae 32086018 | 8 | >8 | >32/4 |
| K. pneumoniae 32105351 | 8 | >8 | >32/4 |
| K. pneumoniae 32108273 | 16 | >8 | 32/4 |
| A. baumannii 32111798 | 8 | >8 | >32/4 |
| P. aeruginosa 32107321 | 8 | 1 | 8/4 |
| Biofilm Inhibition (%) | |||
|---|---|---|---|
| Strain | 27 | 28a | 17a |
| S. aureus ATCC 25923 | 22.0 | 38.0 | 38.3 |
| S. haemolyticus 32076211 | 44.3 | 94.1 | 29.5 |
| E. coli ATCC 25922 | 48.9 | 48.8 | N.D. a |
| A. baumannii 32111798 | 97.4 | 39.5 | N.D. a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amata, S.; Calà, C.; Rizzo, C.; Pibiri, I.; Pizzo, M.; Buscemi, S.; Palumbo Piccionello, A. Synthesis and Antibacterial Activity of Mono- and Bi-Cationic Pyridinium 1,2,4-Oxadiazoles and Triazoles. Int. J. Mol. Sci. 2024, 25, 377. https://doi.org/10.3390/ijms25010377
Amata S, Calà C, Rizzo C, Pibiri I, Pizzo M, Buscemi S, Palumbo Piccionello A. Synthesis and Antibacterial Activity of Mono- and Bi-Cationic Pyridinium 1,2,4-Oxadiazoles and Triazoles. International Journal of Molecular Sciences. 2024; 25(1):377. https://doi.org/10.3390/ijms25010377
Chicago/Turabian StyleAmata, Sara, Cinzia Calà, Carla Rizzo, Ivana Pibiri, Mariangela Pizzo, Silvestre Buscemi, and Antonio Palumbo Piccionello. 2024. "Synthesis and Antibacterial Activity of Mono- and Bi-Cationic Pyridinium 1,2,4-Oxadiazoles and Triazoles" International Journal of Molecular Sciences 25, no. 1: 377. https://doi.org/10.3390/ijms25010377
APA StyleAmata, S., Calà, C., Rizzo, C., Pibiri, I., Pizzo, M., Buscemi, S., & Palumbo Piccionello, A. (2024). Synthesis and Antibacterial Activity of Mono- and Bi-Cationic Pyridinium 1,2,4-Oxadiazoles and Triazoles. International Journal of Molecular Sciences, 25(1), 377. https://doi.org/10.3390/ijms25010377








