Withaferin A and Celastrol Overwhelm Proteostasis
Abstract
1. Introduction
2. Proteomic Studies of WA Action
2.1. Direct Targets of WA and Effects on Protein Expression
2.2. Protein Unfolding Induced by WA
3. Proteomic Studies with CEL—Identification of Direct Targets of CEL
4. The Cell Context Generates Targeting Specificity
5. The Primary Mechanism of Action of WA and CEL Appears to Be the Induction of Proteotoxic Stress
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Kumar, S.; Mathew, S.O.; Aharwal, R.P.; Tulli, H.S.; Mohan, C.D.; Sethi, G.; Ahn, K.-S.; Sandhu, S.S.; Webber, K.; Bishayee, A. Withaferin A: A pleiotropic anticancer agent from the Indian medicinal plant Withania somnifera (L.) Dunal. Pharmaceuticals 2023, 16, 160. [Google Scholar] [CrossRef] [PubMed]
- Sultana, T.; Okla, M.K.; Ahmed, M.; Akhtar, N.; Al-Hashimi, A.; Abdelgawad, H.; Haq, I.-U. Withaferin A: From Ancient Remedy to Potential Drug Candidate. Molecules 2021, 26, 7696. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Sharma, P.; Dudhe, R.; Singh, S. Biological activities of Withania somnifera. Ann. Biol. Res. 2010, 1, 56–63. [Google Scholar]
- Yang, E.S.; Choi, M.J.; Kim, J.H.; Choi, K.S.; Kwon, T.K. Withaferin A enhances radiation-induced apoptosis in Caki cells through induction of reactive oxygen species, Bcl-2 downregulation and Akt inhibition. Chem.-Biol. Interact. 2011, 190, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Bale, S.; Pulivendala, G.; Godugu, C. Withaferin A attenuates bleomycin-induced scleroderma by targeting FoxO3a and NF-κβ signaling: Connecting fibrosis and inflammation. Biofactors 2018, 44, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Tewari, D.; Chander, V.; Dhyani, A.; Sahu, S.; Gupta, P.; Patni, P.; Kalick, L.S.; Bishayee, A. Withania somnifera (L.) Dunal: Phytochemistry, structure-activity relationship, and anticancer potential. Phytomedicine 2022, 98, 153949. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, H.; Kaul, A.; Li, K.; Priyandoko, D.; Kaul, S.C.; Wadhwa, R. Effect of Ashwagandha Withanolides on muscle cell differentiation. Biomolecules 2021, 11, 1454. [Google Scholar] [CrossRef]
- Mandlik, D.S.; Namdeo, A.G. Pharmacological evaluation of Ashwagandha highlighting its healthcare claims, safety, and toxicity aspects. J. Diet. Suppl. 2020, 18, 183–226. [Google Scholar] [CrossRef]
- Bungau, S.; Vesa, C.M.; Abid, A.; Behl, T.; Tit, D.M.; Purza, A.L.; Pasca, B.; Todan, L.M.; Endres, L. Withaferin A—A Promising Phytochemical Compound with Multiple Results in Dermatological Diseases. Molecules 2021, 26, 2407. [Google Scholar] [CrossRef]
- Grogan, P.T.; Sleder, K.D.; Samadi, A.K.; Zhang, H.; Timmermann, B.N.; Cohen, M.S. Cytotoxicity of withaferin A in glio-blastomas involves induction of an oxidative stress-mediated heat shock response while altering Akt/mTOR and MAPK signaling pathways. Investig. New Drugs 2013, 31, 545–557. [Google Scholar] [CrossRef]
- Antony, M.L.; Lee, J.; Hahm, E.-R.; Kim, S.-H.; Marcus, A.I.; Kumari, V.; Ji, X.; Yang, Z.; Vowell, C.L.; Wipf, P.; et al. Growth Arrest by the Antitumor Steroidal Lactone Withaferin A in Human Breast Cancer Cells Is Associated with Down-regulation and Covalent Binding at Cysteine 303 of β-Tubulin. J. Biol. Chem. 2014, 289, 1852–1865. [Google Scholar] [CrossRef] [PubMed]
- Stan, S.D.; Hahm, E.-R.; Warin, R.; Singh, S.V. Withaferin A Causes FOXO3a- and Bim-Dependent Apoptosis and Inhibits Growth of Human Breast Cancer Cells In vivo. Cancer Res. 2008, 68, 7661–7669. [Google Scholar] [CrossRef] [PubMed]
- Hahm, E.-R.; Moura, M.B.; Kelley, E.E.; Van Houten, B.; Shiva, S.; Singh, S.V. Withaferin A-Induced Apoptosis in Human Breast Cancer Cells Is Mediated by Reactive Oxygen Species. PLoS ONE 2011, 6, e23354. [Google Scholar] [CrossRef] [PubMed]
- Stan, S.D.; Zeng, Y.; Singh, S.V. Ayurvedic Medicine Constituent Withaferin A Causes G2 and M Phase Cell Cycle Arrest in Human Breast Cancer Cells. Nutr. Cancer 2008, 60 (Suppl. 1), 51–60. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Hahm, E.-R.; Singh, S.V. Withaferin A inhibits activation of signal transducer and activator of transcription 3 in human breast cancer cells. Carcinogenesis 2010, 31, 1991–1998. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.; De, S.; Mukherjee, S.; Das, S.; Ghosh, A.N.; Sengupta, S. Withaferin A induced impaired autophagy and unfolded protein response in human breast cancer cell-lines MCF-7 and MDA-MB-231. Toxicol. In Vitro 2017, 44, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Hahm, E.-R.; Lee, J.; Huang, Y.; Singh, S.V. Withaferin a suppresses estrogen receptor-α expression in human breast cancer cells. Mol. Carcinog. 2011, 50, 614–624. [Google Scholar] [CrossRef]
- Nagalingam, A.; Kuppusamy, P.; Singh, S.V.; Sharma, D.; Saxena, N.K. Mechanistic Elucidation of the Antitumor Properties of Withaferin A in Breast Cancer. Cancer Res. 2014, 74, 2617–2629. [Google Scholar] [CrossRef]
- Muniraj, N.; Siddharth, S.; Nagalingam, A.; Walker, A.; Woo, J.; Gyorffy, B.; Gabrielson, E.; Saxena, N.K.; Sharma, D. Witha-ferin A inhibits lysosomal activity to block autophagic flux and induces apoptosis via energetic impairment in breast cancer cells. Carcinogenesis 2019, 40, 1110–1120. [Google Scholar]
- Choi, B.Y.; Kim, B.-W. Withaferin-A Inhibits Colon Cancer Cell Growth by Blocking STAT3 Transcriptional Activity. J. Cancer Prev. 2015, 20, 185–192. [Google Scholar] [CrossRef]
- Suman, S.; Das, T.P.; Ankem, M.K.; Damodaran, C. Targeting Notch Signaling in Colorectal Cancer. Curr. Colorectal Cancer Rep. 2014, 10, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Roy, K.S.; Chakrabarti, T.; Mukhopadhyay, S.; Roychoudhury, S. Withaferin A modulates the Spindle Assembly Checkpoint by degradation of Mad2–Cdc20 complex in colorectal cancer cell lines. Biochem. Pharmacol. 2014, 91, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Samadi, A.K.; Tong, X.; Mukerji, R.; Zhang, H.; Timmermann, B.N.; Cohen, M.S. Withaferin A, a Cytotoxic Steroid from Vassobia breviflora, Induces Apoptosis in Human Head and Neck Squamous Cell Carcinoma. J. Nat. Prod. 2010, 73, 1476–1481. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tan, Y.; Liu, S.; Yin, H.; Duan, J.; Fan, L.; Zhao, X.; Jiang, B. Implications of Withaferin A for the metastatic potential and drug resistance in hepatocellular carcinoma cells via Nrf2-mediated EMT and ferroptosis. Toxicol. Mech. Methods 2022, 33, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, L.; Liang, T.; Tian, X.-D.; Liu, Y.; Zhang, T. Withaferin A induces mitochondrial-dependent apoptosis in non-small cell lung cancer cells via generation of reactive oxygen species. J. BUON Off. J. Balk. Union Oncol. 2017, 22, 244–250. [Google Scholar]
- Sharma, A.; Sharma, S.; Chaudhary, P.; Dobhal, M.; Sharma, M. Selective cytotoxicity of non-small cell lung cancer cells by the Withaferin A-fortified root extract of Ashwagandha involves differential cell-cycle arrest and apoptosis. Phytopharmacology 2011, 1, 54–70. [Google Scholar]
- Oh, J.H.; Kwon, T.K. Withaferin A inhibits tumor necrosis factor-induced expression of cell adhesion molecules by inactivation of Akt and NF-kappaB in human pulmonary epithelial cells. Int. Immunopharmacol. 2009, 9, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Sheng, Z.-Y.; Chen, Y.; Bai, C. Effect of Withaferin A on A549 Cellular Proliferation and Apoptosis in Non-small Cell Lung Cancer. Asian Pac. J. Cancer Prev. 2014, 15, 1711–1714. [Google Scholar] [CrossRef]
- Oh, J.H.; Lee, T.-J.; Kim, S.H.; Choi, Y.H.; Lee, S.H.; Lee, J.M.; Kim, Y.-H.; Park, J.-W.; Kwon, T.K. Induction of apoptosis by withaferin A in human leukemia U937 cells through down-regulation of Akt phosphorylation. Apoptosis 2008, 13, 1494–1504. [Google Scholar] [CrossRef]
- Okamoto, S.; Tsujioka, T.; Suemori, S.; Kida, J.; Kondo, T.; Tohyama, Y.; Tohyama, K. Withaferin A suppresses the growth of myelodysplasia and leukemia cell lines by inhibiting cell cycle progression. Cancer Sci. 2016, 107, 1302–1314. [Google Scholar] [CrossRef]
- Mandal, C.; Dutta, A.; Mallick, A.; Chandra, S.; Misra, L.; Sangwan, R.S.; Mandal, C. Withaferin A induces apoptosis by activating p38 mitogen-activated protein kinase signaling cascade in leukemic cells of lymphoid and myeloid origin through mitochondrial death cascade. Apoptosis 2008, 13, 1450–1464. [Google Scholar] [CrossRef] [PubMed]
- McKenna, M.; Gachuki, B.; Alhakeem, S.; Oben, K.; Rangnekar, V.; Gupta, R.; Bondada, S. Anti-cancer activity of withaferin A in B-cell lymphoma. Cancer Biol. Ther. 2015, 16, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Shefrin, S.; Sari, A.N.; Kumar, V.; Zhang, H.; Meidinna, H.N.; Kaul, S.C.; Wadhwa, R.; Sundar, D. Comparative computational and experimental analyses of some natural small molecules to restore transcriptional activation function of p53 in cancer cells harbouring wild type and p53Ser46 mutant. Curr. Res. Struct. Biol. 2022, 4, 320–331. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Okuzaki, D.; Fukushima, K.; Mukai, S.; Ohno, S.; Ozaki, Y.; Yabuta, N.; Nojima, H. Withaferin A Induces Cell Death Selectively in Androgen-Independent Prostate Cancer Cells but Not in Normal Fibroblast Cells. PLoS ONE 2015, 10, e0134137. [Google Scholar] [CrossRef] [PubMed]
- Um, H.J.; Min, K.-J.; Kim, D.E.; Kwon, T.K. Withaferin A inhibits JAK/STAT3 signaling and induces apoptosis of human renal carcinoma Caki cells. Biochem. Biophys. Res. Commun. 2012, 427, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Mayola, E.; Gallerne, C.; Degli Esposti, D.; Martel, C.; Pervaiz, S.; Larue, L.; Debuire, B.; Lemoine, A.; Brenner, C.; Lemaire, C. Withaferin A induces apoptosis in human melanoma cells through generation of reactive oxygen species and down-regulation of Bcl-2. Apoptosis 2011, 16, 1014–1027. [Google Scholar] [CrossRef]
- Cascão, R.; Fonseca, J.E.; Moita, L.F. Celastrol: A Spectrum of Treatment Opportunities in Chronic Diseases. Front. Med. 2017, 4, 69. [Google Scholar] [CrossRef]
- Hou, W.; Liu, B.; Xu, H. Celastrol: Progresses in structure-modifications, structure-activity relationships, pharmacology and toxicology. Eur. J. Med. Chem. 2020, 189, 112081. [Google Scholar] [CrossRef]
- Wang, J.; Chu, Y.; Zhou, X. Inhibitory effect of Triperygium wilfordii polyglucoside on dipeptidyl peptidase I in vivo and in vitro. Biomed. Pharmacother. 2017, 96, 466–470. [Google Scholar] [CrossRef]
- Tao, X.; Younger, J.; Fan, F.Z.; Wang, B.; Lipsky, P.E. Benefit of an extract of Tripterygium Wilfordii Hook F in patients with rheumatoid arthritis: A double-blind, placebo-controlled study. Arthritis Rheum. 2002, 46, 1735–1743. [Google Scholar] [CrossRef]
- Zhou, Y.-Y.; Xia, X.; Peng, W.-K.; Wang, Q.-H.; Peng, J.-H.; Li, Y.; Wu, J.-X.; Zhang, J.-Y.; Zhao, Y.; Chen, X.-M.; et al. The Effectiveness and Safety of Tripterygium wilfordii Hook. F Extracts in Rheu-matoid Arthritis: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2018, 9, 356. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, P.E.; Tao, X.-L. A potential new treatment for rheumatoid arthritis:Thunder god vine. Semin. Arthritis Rheum. 1997, 26, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Li, J.; Lin, Y.; Song, Y.-N.; Zhang, M.; Ke, Y.; Ren, Y.; Deng, X.; Zhang, J.; Huang, F.; et al. Report of 12 cases of ankylosing spondylitis patients treated with Tripterygium wilfordii. Clin. Rheumatol. 2010, 29, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Ren, J.; Wang, G.; Gu, G.; Hu, D.; Ren, H.; Hong, Z.; Wu, X.; Liu, S.; Li, J. T2 enhances in situ level of Foxp3+ regulatory cells and modulates inflammatory cytokines in Crohn’s disease. Int. Immunopharmacol. 2013, 18, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Wu, X.; Liao, N.; Wang, G.; Fan, C.; Liu, S.; Ren, H.; Zhao, Y.; Li, J. Prevention of postoperative recurrence of Crohn’s disease: Tripterygium wilfordii polyglycoside versus mesalazine. J. Int. Med. Res. 2013, 41, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Jin, H.-Z.; Shu, D.; Li, F.; He, C.-X.; Qiao, J.; Yu, X.-L.; Zhang, Y.; He, Y.-B.; Liu, T.-J. Efficacy and Safety of Tripterygium wilfordii Hook F Versus Acitretin in Moderate to Severe Psoriasis Vulgaris. Chin. Med. J. 2015, 128, 443–449. [Google Scholar] [CrossRef]
- Lv, M.; Deng, J.; Tang, N.; Zeng, Y.; Lu, C. Efficacy and Safety of Tripterygium Wilfordii Hook F on Psoriasis Vulgaris: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Evid.-Based Complement. Altern. Med. 2018, 2018, 2623085. [Google Scholar] [CrossRef]
- Wang, Z.; Yu, C.; Zhou, L.-N.; Chen, X. Effects of Tripterygium wilfordii Induction Therapy to IgA Nephropathy Patients with Heavy Proteinuria. Biol. Pharm. Bull. 2017, 40, 1833–1838. [Google Scholar] [CrossRef]
- Choi, B.-S.; Sapkota, K.; Kim, S.; Lee, H.J.; Choi, H.-S.; Kim, S.-J. Antioxidant Activity and Protective Effects of Tripterygium regelii Extract on Hydrogen Peroxide-Induced Injury in Human Dopaminergic Cells, SH-SY5Y. Neurochem. Res. 2010, 35, 1269–1280. [Google Scholar] [CrossRef]
- Qian, S.Z. Tripterygium Wilfordii, a chinese herb effective in male fertility regulation. Contraception 1987, 36, 335–345. [Google Scholar] [CrossRef]
- Ji, S.-M.; Wang, Q.-W.; Chen, J.-S.; Sha, G.-Z.; Liu, Z.-H.; Li, L.-S. Clinical Trial of Tripterygium Wilfordii Hook F. in Human Kidney Transplantation in China. Transplant. Proc. 2006, 38, 1274–1279. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Luo, Q.; Alitongbieke, G.; Chong, S.; Xu, C.; Xie, L.; Chen, X.; Zhang, D.; Zhou, Y.; Wang, Z.; et al. Celastrol-Induced Nur77 Interaction with TRAF2 Alleviates Inflammation by Promoting Mitochondrial Ubiquitination and Autophagy. Mol. Cell 2017, 66, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Trott, A.; West, J.D.; Klaić, L.; Westerheide, S.D.; Silverman, R.B.; Morimoto, R.I.; Morano, K.A. Activation of Heat Shock and Antioxidant Responses by the Natural Product Celastrol: Transcriptional Signatures of a Thiol-targeted Molecule. Mol. Biol. Cell 2008, 19, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Kannaiyan, R.; Shanmugam, M.K.; Sethi, G. Molecular targets of celastrol derived from Thunder of God Vine: Potential role in the treatment of inflammatory disorders and cancer. Cancer Lett. 2011, 303, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, D.; Sharma, A.; Tuli, H.S.; Sak, K.; Mukherjee, T.; Bishayee, A. Molecular targets of celastrol in cancer: Recent trends and advancements. Crit. Rev. Oncol./Hematol. 2018, 128, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Cleren, C.; Calingasan, N.Y.; Chen, J.; Beal, M.F. Celastrol protects against MPTP- and 3-nitropropionic acid-induced neuro-toxicity. J. Neurochem. 2005, 94, 995–1004. [Google Scholar] [CrossRef]
- Xu, C.; Wang, X.; Gu, C.; Zhang, H.; Zhang, R.; Dong, X.; Liu, C.; Hu, X.; Ji, X.; Huang, S.; et al. Celastrol ameliorates Cd-induced neuronal apoptosis by targeting NOX2-derived ROS-dependent PP5-JNK signaling pathway. J. Neurochem. 2017, 141, 48–62. [Google Scholar] [CrossRef]
- Allison, A.C.; Cacabelos, R.; Lombardi, V.R.M.; Álvarez, X.A.; Vigo, C. Central Nervous System Effects of Celastrol, a Potent Antioxidant and Antiinflammatory Agent. CNS Drug Rev. 2006, 6, 45–62. [Google Scholar] [CrossRef]
- Yang, C.; Swallows, C.L.; Zhang, C.; Lu, J.; Xiao, H.; Brady, R.O.; Zhuang, Z. Celastrol increases glucocerebrosidase activity in Gaucher disease by modulating molecular chaperones. Proc. Natl. Acad. Sci. USA 2014, 111, 249–254. [Google Scholar] [CrossRef]
- Li, X.; Wu, N.; Zou, L.; Jia, D. Protective effect of celastrol on myocardial ischemia–reperfusion injury. Anatol. J. Cardiol. 2017, 18, 384–390. [Google Scholar] [CrossRef]
- Hu, H.; Straub, A.; Tian, Z.; Bassler, N.; Cheng, J.; Peter, K. Celastrol, a Triterpene Extracted From Tripterygium wilfordii Hook F, Inhibits Platelet Activation. J. Cardiovasc. Pharmacol. 2009, 54, 240–245. [Google Scholar] [CrossRef]
- Gu, L.; Bai, W.; Li, S.; Zhang, Y.; Han, Y.; Gu, Y.; Meng, G.; Xie, L.; Wang, J.; Xiao, Y.; et al. Celastrol Prevents Atherosclerosis via Inhibiting LOX-1 and Oxidative Stress. PLoS ONE 2013, 8, e65477. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Yu, X.; Zuo, K.; Zhang, X.; Cao, C.; Xu, J.; Wang, S.; Tang, T.; Ye, M.; Pei, E.; et al. Tripterine Treatment Improves Endothelial Progenitor Cell Function via Integrin-Linked Kinase. Cell. Physiol. Biochem. 2015, 37, 1089–1103. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.H.; Cheng, Y.; Chen, W.P.; Zhong, H.M.; Wang, X.H. Celastrol, an inhibitor of heat shock protein 90β potently sup-presses the expression of matrix metalloproteinases, inducible nitric oxide synthase and cyclooxygenase-2 in primary human osteoarthritic chondrocytes. Eur. J. Pharmacol. 2013, 708, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kim, K.; Lee, H.; Han, S.; Lee, Y.-S.; Choe, J.; Kim, Y.-M.; Hahn, J.-H.; Ro, J.Y.; Jeoung, D. Celastrol binds to ERK and inhibits FcεRI signaling to exert an anti-allergic effect. Eur. J. Pharmacol. 2009, 612, 131–142. [Google Scholar] [CrossRef]
- Kim, D.Y.; Park, J.W.; Jeoung, D.; Ro, J.Y. Celastrol suppresses allergen-induced airway inflammation in a mouse allergic asthma model. Eur. J. Pharmacol. 2009, 612, 98–105. [Google Scholar] [CrossRef]
- Liu, J.; Lee, J.; Hernandez, M.A.S.; Mazitschek, R.; Ozcan, U. Treatment of Obesity with Celastrol. Cell 2015, 161, 999–1011. [Google Scholar] [CrossRef]
- Ma, X.; Xu, L.; Alberobello, A.T.; Gavrilova, O.; Bagattin, A.; Skarulis, M.; Liu, J.; Finkel, T.; Mueller, E. Celastrol Protects against Obesity and Metabolic Dysfunction through Activation of a HSF1-PGC1α Transcriptional Axis. Cell Metab. 2015, 22, 695–708. [Google Scholar] [CrossRef]
- Wang, C.; Shi, C.; Yang, X.; Yang, M.; Sun, H.; Wang, C. Celastrol suppresses obesity process via increasing antioxidant capacity and improving lipid metabolism. Eur. J. Pharmacol. 2015, 744, 52–58. [Google Scholar] [CrossRef]
- Moujir, L.; Gutiérrez-Navarro, A.M.; González, A.G.; Ravelo, A.G.; Luis, J.G. The relationship between structure and antimi-crobial activity in quinones from the Celastraceae. Biochem. Syst. Ecol. 1990, 18, 25–28. [Google Scholar] [CrossRef]
- Luo, D.; Wang, H.; Tian, X.; Shao, H.; Liu, J. Antifungal properties of pristimerin and celastrol isolated from Celastrus hypoleucus. Pest Manag. Sci. 2004, 61, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Paris, D.; Ganey, N.J.; Laporte, V.; Patel, N.S.; Beaulieu-Abdelahad, D.; Bachmeier, C.; March, A.; Ait-Ghezala, G.; Mullan, M.J. Reduction of β-amyloid pathology by celastrol in a transgenic mouse model of Alzheimer’s disease. J. Neuroinflamm. 2010, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Verschueren, P.; Lensen, F.; Lerut, E.; Claes, K.; De Vos, R.; Van Damme, B.; Westhovens, R. Benefit of anti-TNFalpha treatment for nephrotic syndrome in a patient with juvenile inflammatory bowel disease associated spondyloarthropathy complicated with amyloidosis and glomerulonephritis. Ann. Rheum. Dis. 2003, 62, 368–369. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, Y.-Y.; Huang, X.-Y.; Sun, Y.-N.; Jia, Y.-F.; Li, D. Beneficial effect of tripterine on systemic lupus erythematosus induced by active chromatin in BALB/c mice. Eur. J. Pharmacol. 2005, 512, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Yi, Z.; Zhang, J.; Lu, B.; Sung, B.; Qu, W.; Aggarwal, B.B.; Liu, M. Celastrol Suppresses Angiogenesis-Mediated Tumor Growth through Inhibition of AKT/Mammalian Target of Rapamycin Pathway. Cancer Res. 2010, 70, 1951–1959. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Zhang, Q.; Luo, P.; Gu, L.; Shen, S.; Tang, H.; Zhang, Y.; Lyu, M.; Shi, Q.; Yang, C.; et al. Neuroprotective Effects of Celastrol in Neurodegenerative Diseases-Unscramble Its Major Mechanisms of Action and Targets. Aging Dis. 2022, 13, 815–836. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, J.; Yang, C. Celastrol, a TFEB (transcription factor EB) agonist, is a promising drug candidate for Alzheimer disease. Autophagy 2022, 18, 1740–1742. [Google Scholar] [CrossRef]
- Zhu, C.; Yang, J.; Zhu, Y.; Li, J.; Chi, H.; Tian, C.; Meng, Y.; Liu, Y.; Wang, J.; Lin, N. Celastrol alleviates comorbid obesity and depression by directly binding amygdala HnRNPA1 in a mouse model. Clin. Transl. Med. 2021, 11, e394. [Google Scholar] [CrossRef]
- Luo, P.; Zhang, Q.; Zhong, T.-Y.; Chen, J.-Y.; Zhang, J.-Z.; Tian, Y.; Zheng, L.-H.; Yang, F.; Dai, L.-Y.; Zou, C.; et al. Celastrol mitigates inflammation in sepsis by inhibiting the PKM2-dependent Warburg effect. Mil. Med. Res. 2022, 9, 22. [Google Scholar] [CrossRef]
- Luo, P.; Liu, D.; Zhang, Q.; Yang, F.; Wong, Y.-K.; Xia, F.; Zhang, J.; Chen, J.; Tian, Y.; Yang, C.; et al. Celastrol induces ferroptosis in activated HSCs to ameliorate hepatic fibrosis via targeting peroxiredoxins and HO-1. Acta Pharm. Sin. B 2021, 12, 2300–2314. [Google Scholar] [CrossRef]
- Li, H.-Y.; Zhang, J.; Sun, L.-L.; Li, B.-H.; Gao, H.-L.; Xie, T.; Zhang, N.; Ye, Z.-M. Celastrol induces apoptosis and autophagy via the ROS/JNK signaling pathway in human osteosarcoma cells: An in vitro and in vivo study. Cell Death Dis. 2015, 6, e1604. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-W.; Jang, K.S.B.; Choi, H.J.; Jo, A.; Cheong, J.-H.; Chun, K.-H. Celastrol inhibits gastric cancer growth by induction of apoptosis and autophagy. BMB Rep. 2014, 47, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Sha, M.; Ye, J.; Zhang, L.X.; Luan, Z.Y.; Chen, Y.B.; Huang, J.X. Celastrol induces apoptosis of gastric cancer cells by miR-21 inhibiting PI3K/Akt-NF-kappaB signaling pathway. Pharmacology 2014, 93, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Sha, M.; Ye, J.; Luan, Z.Y.; Guo, T.; Wang, B.; Huang, J.X. Celastrol induces cell cycle arrest by MicroRNA-21-mTOR-mediated inhibition p27 protein degradation in gastric cancer. Cancer Cell Int. 2015, 15, 101. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Huang, X.; Wang, H.; Yang, H. Celastrol Induces Autophagy by Targeting AR/miR-101 in Prostate Cancer Cells. PLoS ONE 2015, 10, e0140745. [Google Scholar] [CrossRef]
- Hieronymus, H.; Lamb, J.; Ross, K.N.; Peng, X.P.; Clement, C.; Rodina, A.; Nieto, M.; Du, J.; Stegmaier, K.; Raj, S.M.; et al. Gene expression signature-based chemical genomic prediction identifies a novel class of HSP90 pathway modulators. Cancer Cell 2006, 10, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Murthy, S.; Sarkar, F.H.; Sheng, S.; Reddy, G.P.-V.; Dou, Q.P. Calpain-mediated androgen receptor breakdown in apoptotic prostate cancer cells. J. Cell. Physiol. 2008, 217, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Hamza, A.; Cao, X.; Wang, B.; Yu, S.; Zhan, C.G.; Sun, D. A novel Hsp90 inhibitor to disrupt Hsp90/Cdc37 complex against pancreatic cancer cells. Mol. Cancer Ther. 2008, 7, 162–170. [Google Scholar] [CrossRef]
- Sreeramulu, S.; Gande, S.L.; Gobel, M.; Schwalbe, H. Molecular mechanism of inhibition of the human protein complex Hsp90-Cdc37, a kinome chaperone- cochaperone, by triterpene celastrol. Angew. Chem. Int. Ed. Engl. 2009, 48, 5853–5855. [Google Scholar] [CrossRef]
- Chadli, A.; Felts, S.J.; Wang, Q.; Sullivan, W.P.; Botuyan, M.V.; Fauq, A.; Ramirez-Alvarado, M.; Mer, G. Celastrol Inhibits Hsp90 Chaperoning of Steroid Receptors by Inducing Fibrillization of the Co-chaperone p23. J. Biol. Chem. 2010, 285, 4224–4231. [Google Scholar] [CrossRef]
- Zanphorlin, L.M.; Alves, F.R.; Ramos, C.H. The effect of celastrol, a triterpene with antitumorigenic activity, on conformational and functional aspects of the human 90kDa heat shock protein Hsp90alpha, a chaperone implicated in the stabilization of the tumor phenotype. Biochim. Biophys. Acta 2014, 1840, 3145–3152. [Google Scholar] [CrossRef] [PubMed]
- Mou, H.; Zheng, Y.; Zhao, P.; Bao, H.; Fang, W.; Xu, N. Celastrol induces apoptosis in non-small-cell lung cancer A549 cells through activation of mitochondria and Fas/FasL-mediated pathways. Toxicol. In Vitro 2011, 25, 1027–1032. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Zhang, X.; Zhao, M.; Wang, Y.; Cheng, X.; Wang, D.; Xu, Y.; Du, Z.; Yu, X. Celastrol targets mitochondrial respiratory chain complex I to induce reactive oxygen species-dependent cytotoxicity in tumor cells. BMC Cancer 2011, 11, 170. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zhou, X.; Fu, C.; Wang, Q.; Nie, T.; Zou, F.; Guo, R.; Liu, H.; Zhang, B.; Dai, M. Celastrol induces apoptosis of human osteosarcoma cells via the mitochondrial apoptotic pathway. Oncol. Rep. 2015, 34, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Li, P.-P.; He, W.; Yuan, P.-F.; Song, S.-S.; Lu, J.-T.; Wei, W. Celastrol Induces Mitochondria-Mediated Apoptosis in Hepatocellular Carcinoma Bel-7402 Cells. Am. J. Chin. Med. 2015, 43, 137–148. [Google Scholar] [CrossRef]
- Shrivastava, S.; Jeengar, M.K.; Reddy, V.S.; Reddy, G.B.; Naidu, V. Anticancer effect of celastrol on human triple negative breast cancer: Possible involvement of oxidative stress, mitochondrial dysfunction, apoptosis and PI3K/Akt pathways. Exp. Mol. Pathol. 2015, 98, 313–327. [Google Scholar] [CrossRef]
- Lee, J.H.; Won, Y.S.; Park, K.H.; Lee, M.K.; Tachibana, H.; Yamada, K.; Seo, K.I. Celastrol inhibits growth and induces apoptotic cell death in melanoma cells via the activation ROS-dependent mitochondrial pathway and the suppression of PI3K/AKT si-gnaling. Apoptosis 2012, 17, 1275–1286. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, J.O.; Lee, S.K.; Kim, N.; You, G.Y.; Moon, J.W.; Sha, J.; Kim, S.J.; Park, S.H.; Kim, H.S. Celastrol suppresses breast cancer MCF-7 cell viability via the AMP-activated protein kinase (AMPK)-induced p53–polo like kinase 2 (PLK-2) pathway. Cell Signal. 2013, 25, 805–813. [Google Scholar] [CrossRef]
- Raja, S.M.; Clubb, R.J.; Ortega-Cava, C.; Williams, S.H.; Bailey, T.A.; Duan, L.; Zhao, X.; Reddi, A.L.; Nyong, A.M.; Natarajan, A.; et al. Anticancer activity of Celastrol in combination with ErbB2-targeted therapeutics for treatment of ErbB2-overexpressing breast cancers. Cancer Biol. Ther. 2011, 11, 263–276. [Google Scholar] [CrossRef]
- Jang, S.Y.; Jang, S.W.; Ko, J. Celastrol inhibits the growth of estrogen positive human breast cancer cells through modulation of estrogen receptor alpha. Cancer Lett. 2011, 300, 57–65. [Google Scholar] [CrossRef]
- Nagase, M.; Oto, J.; Sugiyama, S.; Yube, K.; Takaishi, Y.; Sakato, N. Apoptosis Induction in HL-60 Cells and Inhibition of Topoisomerase II by Triterpene Celastrol. Biosci. Biotechnol. Biochem. 2003, 67, 1883–1887. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Ruan, X.; Zhang, J.; Zhao, Q. Celastrol Induces Cell Apoptosis and Inhibits the Expression of the AML1-ETO/C-KIT Oncoprotein in t(8;21) Leukemia. Molecules 2016, 21, 574. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, P.; Li, F.; Shanmugam, M.K.; Kannaiyan, R.; Goh, J.N.; Wong, K.F.; Wang, W.; Khin, E.; Tergaonkar, V.; Kumar, A.P.; et al. Celastrol suppresses growth and induces apoptosis of human hepatocellular carcinoma through the modulation of STAT3/JAK2 signaling cascade in vitro and in vivo. Cancer Prev. Res. 2012, 5, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Uttarkar, S.; Dassé, E.; Coulibaly, A.; Steinmann, S.; Jakobs, A.; Schomburg, C.; Trentmann, A.; Jose, J.; Schlenke, P.; Berdel, W.E.; et al. Targeting acute myeloid leukemia with a small molecule inhibitor of the Myb/p300 interaction. Blood 2016, 127, 1173–1182. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fribley, A.M.; Miller, J.R.; Brownell, A.L.; Garshott, D.M.; Zeng, Q.; Reist, T.E.; Narula, N.; Cai, P.; Xi, Y.; Callaghan, M.U.; et al. Celastrol induces unfolded protein response-dependent cell death in head and neck cancer. Exp. Cell Res. 2015, 330, 412–422. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Feng, L.; Zhang, D.; Fan, C.; Ma, C.; Yang, W.; Meng, Y.; Wu, W.; Guan, S.; Jiang, B.; Yang, M.; et al. ER stress-mediated apoptosis induced by celastrol in cancer cells and important role of glycogen synthase kinase-3beta in the signal network. Cell Death Dis. 2013, 4, e715. [Google Scholar] [CrossRef] [PubMed]
- Boridy, S.; Le, P.U.; Petrecca, K.; Maysinger, D. Celastrol targets proteostasis and acts synergistically with a heat-shock protein 90 inhibitor to kill human glioblastoma cells. Cell Death Dis. 2014, 5, e1216. [Google Scholar] [CrossRef]
- Ji, N.; Li, J.; Wei, Z.; Kong, F.; Jin, H.; Chen, X.; Li, Y.; Deng, Y. Effect of Celastrol on Growth Inhibition of Prostate Cancer Cells through the Regulation of hERG Channel In Vitro. BioMed Res. Int. 2015, 2015, 308475. [Google Scholar] [CrossRef]
- Shao, L.; Zhou, Z.; Cai, Y.; Castro, P.; Dakhov, O.; Shi, P.; Bai, Y.; Ji, H.; Shen, W.; Wang, J. Celastrol suppresses tumor cell growth through targeting an AR-ERG-NF-kappaB pathway in TMPRSS2/ERG fusion gene expressing prostate cancer. PLoS ONE 2013, 8, e58391. [Google Scholar] [CrossRef]
- Sha, M.; Ye, J.; Zhang, L.X.; Luan, Z.Y.; Chen, Y.B. Celastrol induces apoptosis of gastric cancer cells by miR-146a inhibition of NF-kappaB activity. Cancer Cell Int. 2013, 13, 50. [Google Scholar] [CrossRef]
- Kannaiyan, R.; Hay, H.S.; Rajendran, P.; Li, F.; Shanmugam, M.K.; Vali, S.; Abbasi, T.; Kapoor, S.; Sharma, A.; Kumar, A.P.; et al. Celastrol inhibits proliferation and induces chemosensitization through down-regulation of NF-kappaB and STAT3 regulated gene products in multiple myeloma cells. Br. J. Pharmacol. 2011, 164, 1506–1521. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.; Zhao, W.; Kong, X.; Li, H.; Ouyang, J. NF-Kappa B Modulation Is Involved in Celastrol Induced Human Multiple Myeloma Cell Apoptosis. PLoS ONE 2014, 9, e95846. [Google Scholar] [CrossRef] [PubMed]
- Tozawa, K.; Sagawa, M.; Kizaki, M. Quinone methide tripterine, celastrol, induces apoptosis in human myeloma cells via NF-kappaB pathway. Int. J. Oncol. 2011, 39, 1117–1122. [Google Scholar] [PubMed][Green Version]
- Yang, H.; Chen, D.; Cui, Q.C.; Yuan, X.; Dou, Q.P. Celastrol, a triterpene extracted from the Chinese “Thunder of God Vine”, is a potent proteasome inhibitor and suppresses human prostate cancer growth in nude mice. Cancer Res. 2006, 66, 4758–4765. [Google Scholar] [CrossRef]
- Dai, Y.; Desano, J.; Tang, W.; Meng, X.; Meng, Y.; Burstein, E.; Lawrence, T.S.; Xu, L. Natural proteasome inhibitor celastrol suppresses androgen-independent prostate cancer progression by modulating apoptotic proteins and NF-kappaB. PLoS ONE 2010, 5, e14153. [Google Scholar] [CrossRef]
- Wang, X.N.; Wu, Q.; Yang, X.; Zhang, L.S.; Wu, Y.P.; Lu, C. Effects of Celastrol on growth inhibition of U937 leukemia cells through the regulation of the Notch1/NF-kappaB signaling pathway in vitro. Chin. J. Cancer 2010, 29, 385–390. [Google Scholar] [CrossRef]
- Lee, J.H.; Koo, T.H.; Yoon, H.; Jung, H.S.; Jin, H.Z.; Lee, K.; Hong, Y.S.; Lee, J.J. Inhibition of NF-kappa B activation through targeting I kappa B kinase by celastrol, a quinone methide triterpenoid. Biochem. Pharmacol. 2006, 72, 1311–1321. [Google Scholar] [CrossRef]
- Chiang, K.C.; Tsui, K.H.; Chung, L.C.; Yeh, C.N.; Chen, W.T.; Chang, P.L.; Juang, H.H. Celastrol blocks interleukin-6 gene expression via downregulation of NF-kappaB in prostate carcinoma cells. PLoS ONE 2014, 9, e93151. [Google Scholar] [CrossRef]
- Vilaboa, N.; Boré, A.; Martin-Saavedra, F.; Bayford, M.; Winfield, N.; Firth-Clark, S.; Kirton, S.B.; Voellmy, R. New inhibitor targeting human transcription factor HSF1: Effects on the heat shock response and tumor cell survival. Nucleic Acids Res. 2017, 45, 5797–5817. [Google Scholar] [CrossRef]
- Vilaboa, N.; Lopez, J.A.; de Mesa, M.; Escudero-Duch, C.; Winfield, N.; Bayford, M.; Voellmy, R. Disruption of Proteostasis by Natural Products and Synthetic Compounds That Induce Pervasive Unfolding of Proteins: Therapeutic Implications. Pharmaceuticals 2023, 16, 616. [Google Scholar] [CrossRef]
- Santagata, S.; Xu, Y.-M.; Wijeratne, E.M.K.; Kontnik, R.; Rooney, C.; Perley, C.C.; Kwon, H.; Clardy, J.; Kesari, S.; Whitesell, L.; et al. Using the Heat-Shock Response To Discover Anticancer Compounds that Target Protein Homeostasis. ACS Chem. Biol. 2011, 7, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, W.; Wang, M.; Zhang, X.; Zhang, H.; Tong, X.; Xiao, Y. Competitive profiling of celastrol targets in human cervical cancer HeLa cells via quantitative chemical proteomics. Mol. Biosyst. 2016, 13, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Narayan, M.; Seeley, K.W.; Jinwal, U.K. Identification and quantitative analysis of cellular proteins affected by treatment with withaferin a using a SILAC-based proteomics approach. J. Ethnopharmacol. 2015, 175, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Dom, M.; Offner, F.; Berghe, W.V.; Van Ostade, X. Proteomic characterization of Withaferin A-targeted protein networks for the treatment of monoclonal myeloma gammopathies. J. Proteom. 2018, 179, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Westerheide, S.D.; Bosman, J.D.; Mbadugha, B.N.; Kawahara, T.L.; Matsumoto, G.; Kim, S.; Gu, W.; Devlin, J.P.; Silverman, R.B.; Morimoto, R.I. Celastrols as Inducers of the Heat Shock Response and Cytoprotection. J. Biol. Chem. 2004, 279, 56053–56060. [Google Scholar] [CrossRef] [PubMed]
- Dom, M.; Berghe, W.V.; Van Ostade, X. Broad-spectrum antitumor properties of Withaferin A: A proteomic perspective. RSC Med. Chem. 2019, 11, 30–50. [Google Scholar] [CrossRef]
- Berghe, W.V.; Sabbe, L.; Kaileh, M.; Haegeman, G.; Heyninck, K. Molecular insight in the multifunctional activities of Withaferin A. Biochem. Pharmacol. 2012, 84, 1282–1291. [Google Scholar] [CrossRef]
- Vyas, A.R.; Singh, S.V. Molecular Targets and Mechanisms of Cancer Prevention and Treatment by Withaferin A, A Naturally Occurring Steroidal Lactone. AAPS J. 2013, 16, 1–10. [Google Scholar] [CrossRef]
- Tonelli, C.; Chio, I.I.C.; Tuveson, D.A. Transcriptional Regulation by Nrf2. Antioxid. Redox Signal. 2018, 29, 1727–1745. [Google Scholar] [CrossRef]
- Grover, A.; Shandilya, A.; Bisaria, V.S.; Sundar, D. Probing the anticancer mechanism of prospective herbal drug Withaferin A on mammals: A case study on human and bovine proteasomes. BMC Genom. 2010, 11, S15. [Google Scholar] [CrossRef]
- Kaileh, M.; Berghe, W.V.; Heyerick, A.; Horion, J.; Piette, J.; Libert, C.; De Keukeleire, D.; Essawi, T.; Haegeman, G. Withaferin A Strongly Elicits IκB Kinase β Hyperphosphorylation Concomitant with Potent Inhibition of Its Kinase Activity. J. Biol. Chem. 2007, 282, 4253–4264. [Google Scholar] [CrossRef] [PubMed]
- Heyninck, K.; Lahtela-kakkonen, M.; Van Der Veken, P.; Haegeman, G.; Vanden, W. Withaferin A inhibits NF-kappaB activation by targeting cysteine 179 in IKKβ. Biochem. Pharmacol. 2014, 91, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2008, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhou, J.; Zhu, Y.; Wong, Y.K.; Liu, D.; Gao, P.; Lin, Q.; Zhang, J.; Chen, X.; Wang, J. Quantitative chemical proteomics reveals anti-cancer targets of Celastrol in HCT116 human colon cancer cells. Phytomedicine 2022, 101, 154096. [Google Scholar] [CrossRef]
- Hong, Z.; Cao, J.; Liu, D.; Liu, M.; Chen, M.; Zeng, F.; Qin, Z.; Wang, J.; Tao, T. Celastrol targeting Nedd4 reduces Nrf2-mediated oxidative stress in astrocytes after ischemic stroke. J. Pharm. Anal. 2023, 13, 156–169. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.D.; Luo, P.; Gu, L.; Zhang, Q.; Gao, P.; Zhu, Y.; Chen, X.; Guo, Q.; Zhang, J.; Ma, N.; et al. Celastrol exerts a neuroprotective effect by directly binding to HMGB1 protein in cerebral ischemia-reperfusion. J. Neuroinflamm. 2021, 18, 174. [Google Scholar] [CrossRef]
- Kumar, R.; Nayak, D.; Somasekharan, S.P. SILAC-based quantitative MS approach reveals Withaferin A regulated proteins in prostate cancer. J. Proteom. 2021, 247, 104334. [Google Scholar] [CrossRef]
- Boccadoro, M.; Morgan, G.; Cavenagh, J. Preclinical evaluation of the proteasome inhibitor bortezomib in cancer therapy. Cancer Cell Int. 2005, 5, 18. [Google Scholar] [CrossRef]
- Gandolfi, S.; Laubach, J.P.; Hideshima, T.; Chauhan, D.; Anderson, K.C.; Richardson, P.G. The proteasome and proteasome inhibitors in multiple myeloma. Cancer Metastasis Rev. 2017, 36, 561–584. [Google Scholar] [CrossRef]
- Galehdar, Z.; Swan, P.; Fuerth, B.; Callaghan, S.M.; Park, D.S.; Cregan, S.P. Neuronal Apoptosis Induced by Endoplasmic Reticulum Stress Is Regulated by ATF4–CHOP-Mediated Induction of the Bcl-2 Homology 3-Only Member PUMA. J. Neurosci. 2010, 30, 16938–16948. [Google Scholar] [CrossRef]
- Zheng, G.-F.; Cai, Z.; Meng, X.-K.; Zhang, Y.; Zhu, W.; Pang, X.-Y.; Dou, L. Unfolded protein response mediated JNK/AP-1 signal transduction, a target for ovarian cancer treatment. Int. J. Clin. Exp. Pathol. 2015, 8, 6505–6511. [Google Scholar] [PubMed]
- Obeng, E.A.; Carlson, L.M.; Gutman, D.M.; Harrington, W.J., Jr.; Lee, K.P.; Boise, L.H. Proteasome Inhibitors Induce a Terminal Unfolded Protein Response in Multiple Myeloma Cells. Blood 2006, 107, 4907–4916. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.; Chao, A.; Tsai, C.-L.; Chuang, W.-C.; Huang, W.-P.; Chen, G.-C.; Lin, C.-Y.; Wang, T.-H.; Wang, H.-S.; Lai, C.-H. Bortezomib enhances cancer cell death by blocking the autophagic flux through stimulating ERK phosphorylation. Cell Death Dis. 2014, 5, e1510. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.J.; Park, E.J.; Min, K.J.; Park, J.-W.; Kwon, T.K. Endoplasmic reticulum stress mediates withaferin A-induced apoptosis in human renal carcinoma cells. Toxicol. In Vitro 2011, 25, 692–698. [Google Scholar] [CrossRef]
- Khan, S.; Rammeloo, A.W.; Heikkila, J.J. Withaferin A Induces Proteasome Inhibition, Endoplasmic Reticulum Stress, the Heat Shock Response and Acquisition of Thermotolerance. PLoS ONE 2012, 7, e50547. [Google Scholar] [CrossRef]
- Ren, B.; Liu, H.; Gao, H.; Liu, S.; Zhang, Z.; Fribley, A.M.; Callaghan, M.U.; Xu, Z.; Zeng, Q.; Li, Y. Celastrol induces apoptosis in hepatocellular carcinoma cells via targeting ER-stress/UPR. Oncotarget 2017, 8, 93039–93050. [Google Scholar] [CrossRef]

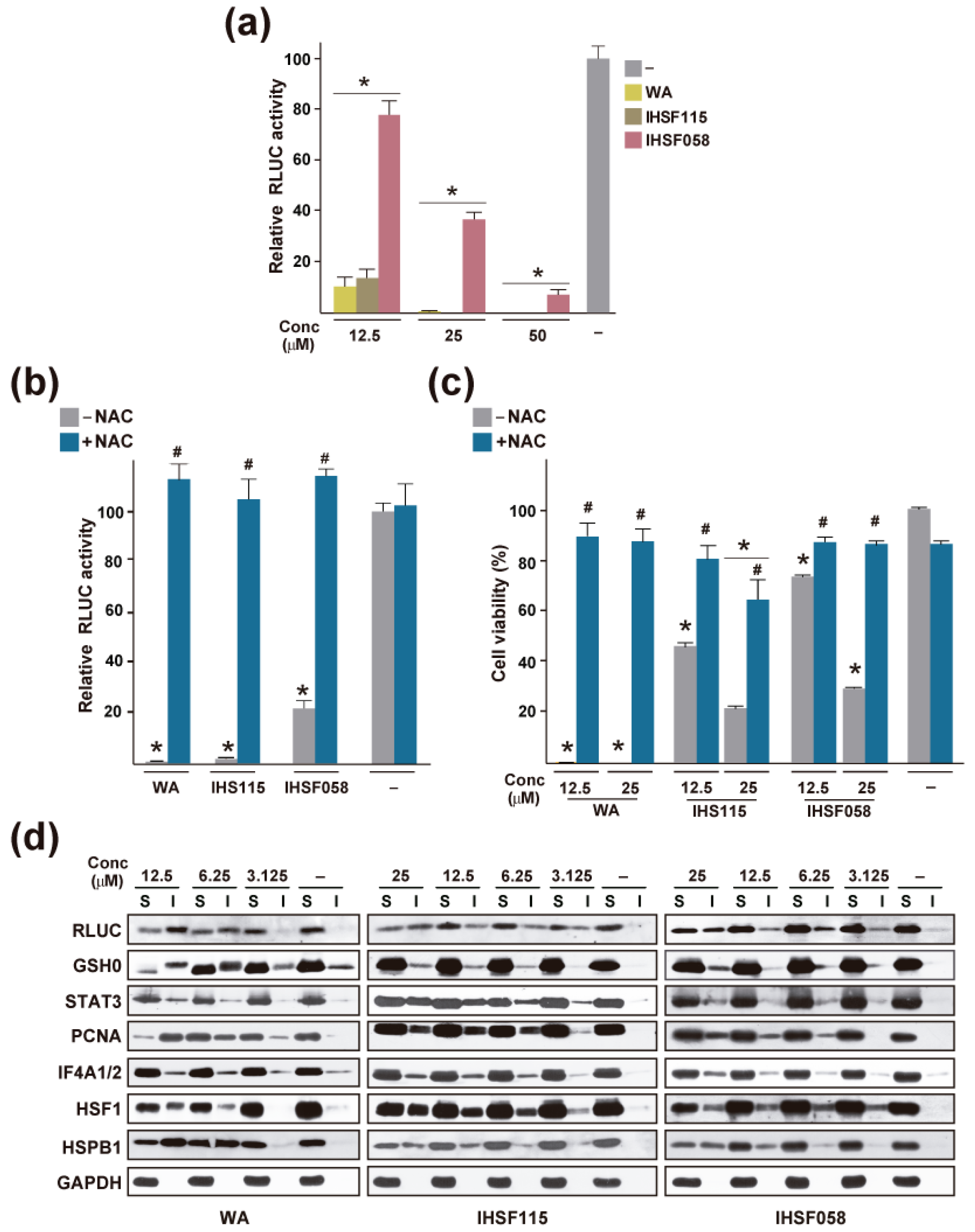
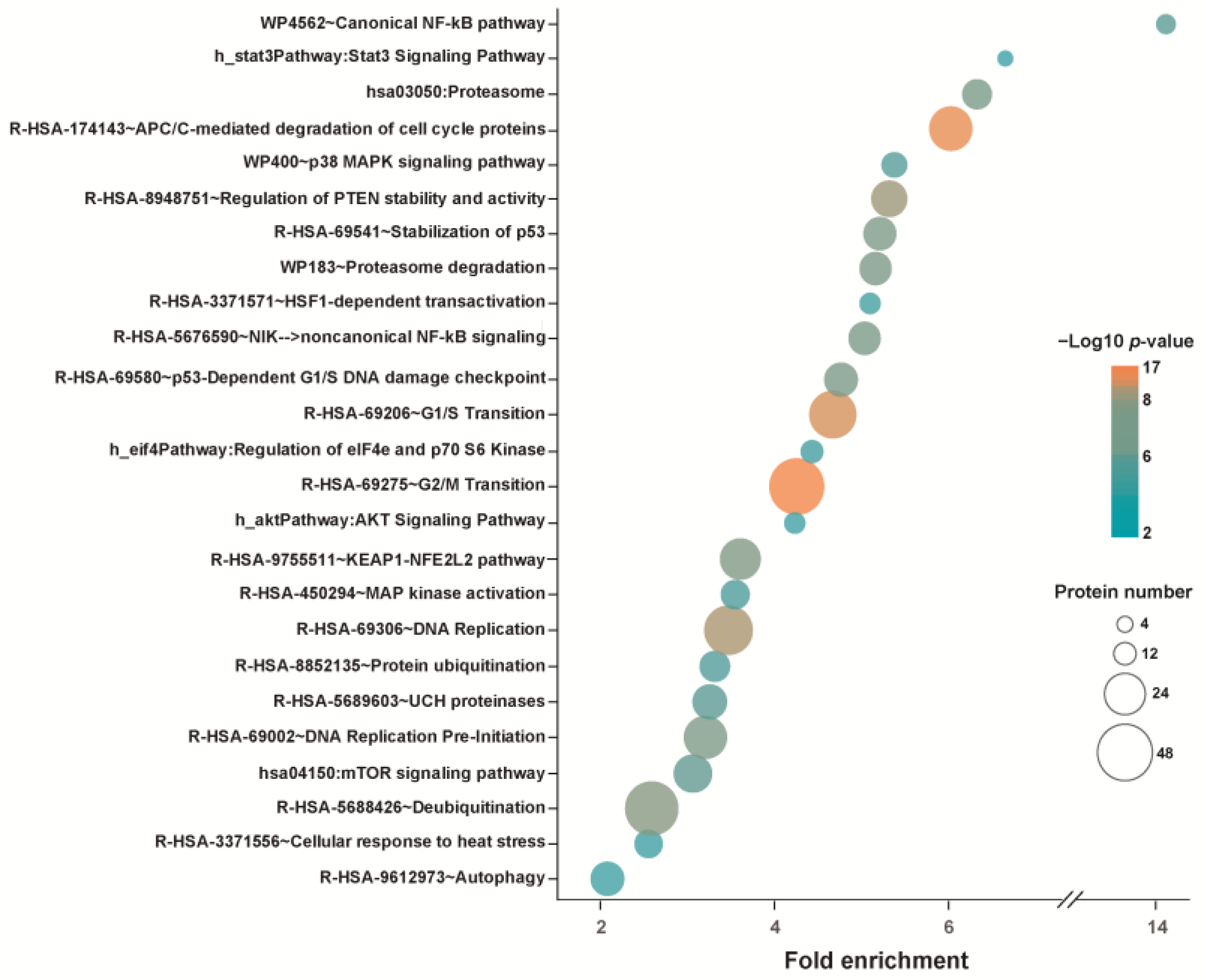



| Cancer Cell Line | Direct or Indirect Protein or Pathway Targets (Upregulated, Downregulated, or Post-Translationally Modified Proteins) | Refs. |
|---|---|---|
| U87, U251, GL26 | Akt, mTOR, p70S6K, AMPKα, tuberin | [10] |
| MCF7, SUM159, SK-BR-3 | WA binds to Cys303 of β-tubulin, α- and β-tubulin | [11] |
| MDA-MB-231, MCF7 | FOXO3a, Bim | [12] |
| MDA-MB-231, MCF7 | Complex III, Bax, Bak | [13] |
| MDA-MB-231, MCF7 | Cdk1, Cdc25B/C | [14] |
| MDA-MB-231, MCF7 | STAT3 | [15] |
| MDA-MB-231, MCF7 | Proteasome, autophagy | [16] |
| MDA-MB-231, MCF7, T-47D | ER-α, p53 | [17] |
| MDA-MB-231, MCF7, T-47D, MDA-MB-68 | p90-ribosomal S6 kinase, extracellular signal-regulated kinase 1/2 | [18] |
| MDA-MB-231, MCF7, T-47D, MDA-MB-468 | Autophagy | [19] |
| HCT116 | STAT3 | [20] |
| HCT-116, SW-480, SW-620 | pS6K, p4E-BP1, Notch signaling | [21] |
| HCT116, SW480 | Mad2, Cdc20 | [22] |
| MDA-1986, JMAR, UM-SCC-2, JHU011 | Cell cycle alteration | [23] |
| HepG2, SNU449 | Keap1, Nrf2 | [24] |
| H358, H460 | ROS | [25] |
| H358, H460 | Oxidative stress, lipid peroxidation, increased GSSG/GSH | [26] |
| A549 | CAMs, Akt, NF-kB | [27] |
| A549 | PI3K/Akt pathway | [28] |
| U-937 | MMP, MAPK pathway | [29] |
| MDS92, MDS-L, HL-60, THP-1, Jurkat, Ramos | HMOX1, LC3A/B | [30] |
| MOLT-4, Jurkat, REH, K-562, HeLa, Saos-2, SP2/0 | p38-MAPK signaling, ATF-2, HSP27 | [31] |
| LY-10, LY-3, SudHL-6, Ramos, Raji, Mino, Jeko | Hsp90, NF-kB | [32] |
| HSC3, U-2 OS | mortalin-p53 interaction | [33] |
| PC-3, DU-145, LNCaP | c-Fos, HSPA6, Hsp70, c-FLIP(L) | [34] |
| Caki | STAT3 | [35] |
| Caki | ROS, Bcl-2, Akt | [4] |
| M14, Mel501, SK28, Lu1205 | Bcl-2, Bax | [36] |
| Cancer Cell Type | Direct or Indirect Protein or Pathway Targets (Upregulated, Downregulated, or Post-Translationally Modified Proteins) | Refs. |
|---|---|---|
| Osteosarcoma cells | Activation of reactive oxygen species (ROS)/c-Jun N-terminal kinases (JNK) signaling | [81] |
| Gastric cancer cells | Downregulation of miR-21 expression; reduced phosphorylation of Akt, mTOR, and S6K and increased phosphorylation of AMPK | [82,83,84] |
| Androgen-receptor-positive prostate cancer cells | The AR/miR-101 axis; degradation of androgen receptor via HSP90 inhibition of calpain activation | [85,86,87] |
| Pancreatic cancer cells | Disruption of the HSP90-CDC37 interaction by targeting CDC37 | [88,89] |
| HeLa (cervical cancer) cells and in vitro studies | Inhibition of p23 | [90] |
| In vitro | Interaction with the carboxy-terminal region of HSP90A | [91] |
| Non-small-cell lung carcinoma, liver cancer, osteosarcoma, and hepatocellular carcinoma cells | Inhibition of mitochondrial respiratory chain (MRC) complex I and, consequently, ROS accumulation | [92,93,94,95] |
| Triple-negative breast cancer and melanoma cells | Mitochondrial dysfunction and PI3K/Akt/mTOR pathway inhibition | [96,97] |
| MCF-7 (breast cancer) cells | AMPK-dependent cell death; increased ROS levels; increased AMPK and p53 phosphorylation | [98] |
| Breast cancer cells | Destabilization of the ErbB2 and estrogen receptors | [99,100] |
| HL-60 (leukemia) cells | Inhibition of topoisomerase II | [101] |
| AML cells | Mitochondrial instability, activation of caspases, and downregulation of AML1-ETO/C-KIT oncoprotein, thus inhibiting the Akt, STAT3, and Erk1/2 downstream pathways | [102] |
| Hepatocellular carcinoma cells | Inhibition of STAT3/Janus kinase 2 (JAK2) | [103] |
| AML cells | Inhibition of Myb | [104] |
| Oral squamous cell carcinoma cells | Induction of unfolded protein response-dependent cell death, endoplasmic reticulum (ER) stress, and PERK-eukaryotic initiation factor 2 (eIF2)–activating transcription factor (ATF4)-C/EBP homology protein (CHOP) signaling | [105] |
| HeLa (cervical cancer) cells | Reduction in GSK3β levels | [106] |
| Glioblastoma cells | Inhibition of autophagy, accumulation of polyubiquitinated proteins, induction of the heat shock response, potentiating the heat shock response induced by HSP90 inhibition | [107] |
| Prostate cancer cells | Targeting of AR, ERG, and NF-kB signaling pathways | [108,109] |
| Gastric cancer cells | Upregulation of miR-146a expression, suppressing the NF-Kb activity | [110] |
| Multiple myeloma and prostate cancer cells; breast cancer xenografts | Inhibition of NF-kB | [111,112,113,114,115,116,117] |
| Prostate cancer cells | Downregulation of IL-6 gene expression via NF-kB inhibition | [118] |
| Prostate cancer xenografts and cells | Inhibition of the proteasome system | [114,115] |
| Pathway | Direct WA Target or Downregulation by WA | Protein | UniProtKB Entry Name | Role/Function | Aggregation Observed |
|---|---|---|---|---|---|
| Oxidative stress (NRF2) | Target | Peroxiredoxin 1 | PRDX1_HUMAN | Dual functioning thiol-specific peroxidase and molecular chaperon | No |
| Target | Kelch-like ECH- associated protein 1 | KEAP1_HUMAN | Inhibitor of NRF2 | No | |
| Downregulation | Glutathione peroxidase 1 | GPX1_HUMAN | Reduces hydrogen peroxide to water | No | |
| Downregulation | Phospholipid hydroperoxide glutathione peroxidase | GPX4_HUMAN | Reduces phospholipid hydroperoxide | No | |
| Ubiquitin- proteasome system | Target | Proteasome subunit beta type-10 | PSB10_HUMAN | Proteasome subunit involved in antigen processing | No |
| Target | Proteasome subunit beta type-5 | PSB5_HUMAN | Chymotrypsin-like activity of the 20S proteasome | No | |
| Target | AAA+ chaperone p97 | TERA_HUMAN | Transitional endoplasmic reticulum ATPase: involved in the transport of ubiquitinated proteins and autophagy | No | |
| Target | Ubiquitin carboxyl- terminal hydrolase 14 | UBP14_HUMAN | Deubiquitinating enzyme | No | |
| Target | Ubiquitin carboxyl- terminal hydrolase isozyme L5 | UCHL5_HUMAN | Deubiquitinating enzyme | Yes | |
| Autophagy | Target | Histone deacetylase 6 | HDAC6_HUMAN | Deacetylation of lysine residues of histones and adapter protein | No |
| Target | Phospholipase A-2- activating protein | PLAP_HUMAN | Autophagy cofactor | Yes | |
| Target | WD repeat domain phosphoinositide interacting protein 2 | WIPI2_HUMAN | Formation of pre- autophagosome structures | Yes | |
| Target | SNARE-associated protein Snapin | SNAPN_HUMAN | Required for biogenesis of lysosome-related organelles | No | |
| Target Downregulation | Tubulin beta chain | TBB1_HUMAN | Major constituent of microtubules | No | |
| Downregulation | Annexin A4 | ANXA4_HUMAN | Membrane fusion | No | |
| Heat shock response | Target | Heat shock factor 1 | HSF1_HUMAN | Master regulator of the heat shock response | Yes |
| Target | Heat shock protein HSP 90-alpha | HS90A_HUMAN | Maturation and structural maintenance of proteins | No | |
| Target | Heat shock protein HSP 90-beta | HS90B_HUMAN | Maturation and structural maintenance of proteins | No | |
| Target | DnaJ homolog subfamily A member 2 | DNJA2_HUMAN | Co-chaperone of HSP70 that stimulates ATP hydrolysis | No | |
| Target | BAG family molecular chaperone regulator 2 | BAG2_HUMAN | Co-chaperone of HSP70 | No | |
| Target | ER membrane protein complex subunit 9 | EMC9_HUMAN | Chaperone complex that was relatively recently discovered | No | |
| Downregulation | Heat shock 70 kDa protein 13 | HSP13_HUMAN | Processing of cytosolic and secretory proteins | No | |
| Downregulation | DnaJ homolog subfamily C member 10 | DJC10_HUMAN | Disulfide reductase involved in the correct folding of proteins and degradation of misfolded proteins | No | |
| Downregulation | Peptidyl-prolyl cis-trans isomerase FKBP4 | FKBP4_HUMAN | Prolyl isomerase and co-chaperone activities | No | |
| Isomerase and disulfide reductase | Target | Peptidyl-prolyl cis-trans isomerase D | PPID_HUMAN | Co-chaperone of HSP90 complexes | Yes |
| Target | Peptidyl-prolyl isomerase domain and WD repeat-containing protein 1 | PPWD1_HUMAN | cis-trans isomerization of proline imidic peptide bonds | No | |
| Target | Peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 | PIN1_HUMAN | Phosphorylation-specific prolyl isomerase | No | |
| Downregulation | Peptidyl-prolyl cis-trans isomerase FKBP4 | FKBP4_HUMAN | Prolyl isomerase and co-chaperone activities | No | |
| Downregulation | DnaJ homolog subfamily C member 10 | DJC10_HUMAN | Disulfide reductase involved in the correct folding of proteins and degradation of misfolded proteins | No | |
| ER- associated protein degradation | Target | ER degradation enhancing alpha- mannosidase-like protein 3 | EDEM3_HUMAN | Catalyzes mannose trimming from Man8GlcNAc2 to Man7GlcNAc2 | No |
| Target | AAA+ chaperone p97 (VCP) | TERA_HUMAN | ATPase activity | No | |
| Target | Endoplasmic Reticulum-Golgi intermediate compartment protein 3 | ERGI3_HUMAN | Transport between endoplasmic reticulum and Golgi | No | |
| Target | Homocysteine-responsive endoplasmic reticulum-resident ubiquitin-like domain member 1 protein | HERP1_HUMAN | Involved in ubiquitin- dependent degradation of misfolded endoplasmic reticulum proteins | No | |
| Target | Transport and Golgi organization protein 1 homolog | TGO1_HUMAN | Involved in the export process exported from the endoplasmic reticulum | No | |
| Protein translation | Target | Eukaryotic translation initiation factor 5A-1 | IF5A1_HUMAN | mRNA-binding protein involved in the level of mRNA turnover, acting downstream of decapping | Yes |
| Target | Eukaryotic translation initiation factor 4B | IF4B_HUMAN | Required for the binding of mRNA to ribosomes | No | |
| Downregulation | Eukaryotic peptide chain release factor subunit 1 | ERF1_HUMAN | Director of termination of the nascent peptide synthesis | No | |
| Downregulation | Eukaryotic translation initiation factor 3 subunit M | EIF3M_HUMAN | Responsible for the initiation of protein synthesis | No | |
| Cytoskeleton functions | Target | Vimentin | VIME_HUMAN | An intermediate filament type III protein | No |
| Target | Annexin A2 | ANXA2_HUMAN | A multifunctional adapter protein that is part of the actin microfilament cytoskeleton | No | |
| Target | Tubulin beta | TUBB1_HUMAN | A major component of the microtubules | No | |
| Cell cycle | Target | Dual specificity protein kinase TTK | TTK_HUMAN | Kinase essential for chromosome alignment | Yes |
| Target | Nucleoporin Nup43 | NUP43_HUMAN | Involved in kinetochore microtubule attachment, mitotic progression, and chromosome segregation | Yes | |
| Target | Protein phosphatase 1B | PPM1B_HUMAN | Phosphatase | No | |
| Target | Serrate RNA effector molecule homolog | SRRT_HUMAN | Mediator between the cap- binding complex (CBC) and the primary microRNAs (miRNAs) processing machinery during cell proliferation | No | |
| Target | Wings apart-like protein homolog | WAPL_HUMAN | Regulator of sister chromatid cohesion in mitosis | Yes | |
| Target | Nuclear-interacting partner of ALK | NIPA_HUMAN | Controls entering mitotic phase | No | |
| Target | Mini-chromosome maintenance complex-binding protein | MCMBP_HUMAN | Associated component of the MCM complex that acts as a regulator of DNA replication | Yes | |
| Target | Serine/threonine- protein phosphatase 2A 55 kDa regulatory subunit B alpha isoform | 2ABA_HUMAN | Phosphatase. The B regulatory subunit modulates substrate selectivity and catalytic activity | No | |
| Protein kinase functions | Target | TTK protein kinase | TTK_HUMAN | Involved in chromosome alignment | Yes |
| Target | Serine/threonine- protein phosphatase 2A 65 kDa regulatory subunit A alpha isoform | 2AAA_HUMAN | Phosphatase. The B regulatory subunit modulates substrate selectivity and catalytic activity | No | |
| Target | Serine/threonine- protein phosphatase 2A 55 kDa regulatory subunit B alpha isoform | 2ABA_HUMAN | Phosphatase. The B regulatory subunit modulates substrate selectivity and catalytic activity | No | |
| NFkB pathway | Target | Coiled-coil domain-containing protein 22 | CCD22_HUMAN | Promoting IκB ubiquitination | Yes |
| Target | Activating signal co- integrator 1 complex subunit 2 | ASCC2_HUMAN | Transactivator of NFκB | No | |
| Target | ELKS/Rab6- interacting/CAST family member 1 | RB6I2_HUMAN | Regulatory subunit of IKK | No | |
| Target | COMM domain- containing protein 3 | COMD3_HUMAN | Downregulates activation of NFκB | Yes | |
| Target | Transcription factor p65 (RelA) | TF65_HUMAN | Transcription factor | Yes | |
| Target | Nuclear factor NFκB p105 subunit | NFκB1_HUMAN | DNA binding | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilaboa, N.; Voellmy, R. Withaferin A and Celastrol Overwhelm Proteostasis. Int. J. Mol. Sci. 2024, 25, 367. https://doi.org/10.3390/ijms25010367
Vilaboa N, Voellmy R. Withaferin A and Celastrol Overwhelm Proteostasis. International Journal of Molecular Sciences. 2024; 25(1):367. https://doi.org/10.3390/ijms25010367
Chicago/Turabian StyleVilaboa, Nuria, and Richard Voellmy. 2024. "Withaferin A and Celastrol Overwhelm Proteostasis" International Journal of Molecular Sciences 25, no. 1: 367. https://doi.org/10.3390/ijms25010367
APA StyleVilaboa, N., & Voellmy, R. (2024). Withaferin A and Celastrol Overwhelm Proteostasis. International Journal of Molecular Sciences, 25(1), 367. https://doi.org/10.3390/ijms25010367






