Diagnosis of Familial Hypercholesterolemia in Children and Young Adults
Abstract
:1. Introduction
2. Results
2.1. The Main Characteristics of the Study Sample
2.2. Results of Molecular Genetic Analyses of Children with FH
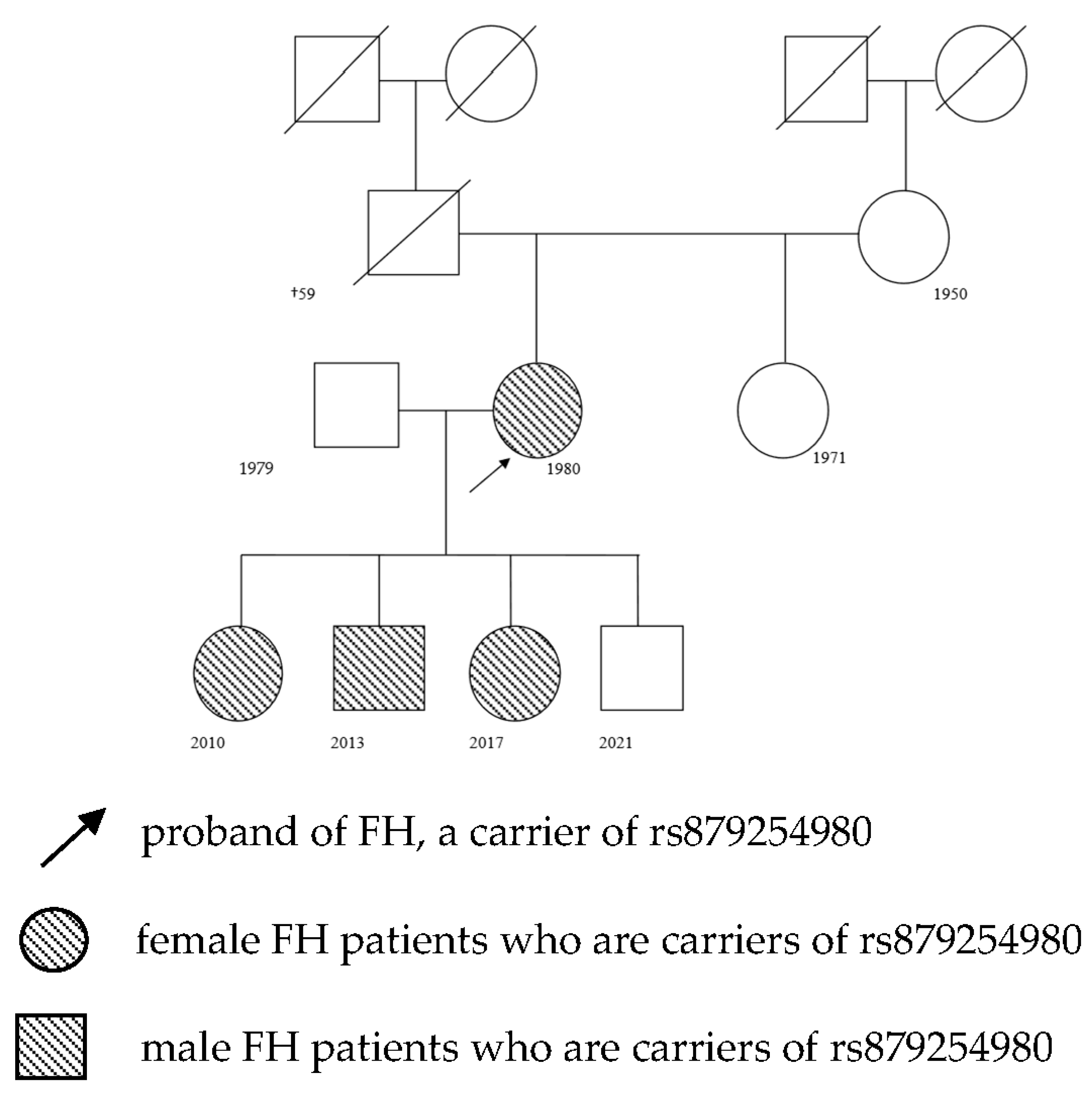
2.3. A Clinical Case
2.4. Results of the Molecular Genetic Testing of Young Adults with FH
2.5. Treatment of Patients under the Age of 18 with FH
3. Discussion
4. Materials and Methods
4.1. The Study Sample
4.2. Molecular Genetic Analysis
4.3. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CHD | coronary heart disease |
| CVD | cardiovascular disease |
| DLCN | Dutch Lipid Clinic Network |
| FH | familial hypercholesterolemia |
| HDL-C | high-density lipoprotein cholesterol |
| heFH | heterozygous familial hypercholesterolemia |
| hoFH | homozygous familial hypercholesterolemia form |
| LDL-C | low-density lipoprotein cholesterol |
| Me | median |
| MLPA | multiplex ligation-dependent probe amplification |
| TC | total cholesterol |
| TG | triglyceride |
References
- Ezhov, M.V.; Bazhan, S.S.; Ershova, A.I.; Meshkov, A.N.; Sokolov, A.A.; Kukharchuk, V.V.; Gurevich, V.S.; Voevoda, M.I.; Sergienko, I.V.; Shakhtshneider, E.V.; et al. Clinical guidelines for familial hypercholesterolemia. Ateroscleroz 2019, 15, 58–98. (In Russian) [Google Scholar]
- Di Taranto, M.D.; Fortunato, G. Genetic heterogeneity of Familial hypercholesterolemia: Repercussions for molecular diagnosis. Int. J. Mol. Sci. 2023, 24, 3224. [Google Scholar] [CrossRef] [PubMed]
- Akioyamen, L.E.; Genest, J.; Shan, S.D.; Reel, R.L.; Albaum, J.M.; Chu, A.; Tu, J.V. Estimating the prevalence of heterozygous familial hypercholesterolaemia: A systematic review and meta-analysis. BMJ Open 2017, 7, e016461. [Google Scholar] [CrossRef] [PubMed]
- Beheshti, S.O.; Madsen, C.M.; Varbo, A.; Nordestgaard, B.G. Worldwide prevalence of Familial hypercholesterolemia: Meta-analyses of 11 million subjects. J. Am. Coll. Cardiol. 2020, 75, 2553–2566. [Google Scholar] [CrossRef] [PubMed]
- Vallejo-Vaz, A.J.; Ray, K.K. Epidemiology of familial hypercholesterolaemia: Community and clinical. Atherosclerosis 2018, 277, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Mszar, R.; Buscher, S.; Taylor, H.L.; Rice-DeFosse, M.T.; McCann, D. Familial Hypercholesterolemia and the Founder Effect Among Franco-Americans: A Brief History and Call to Action. CJC Open 2020, 2, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Meshkov, A.N.; Ershova, A.I.; Shalnova, S.A.; Alieva, A.S.; Bazhan, S.S.; Barbarash, O.L.; Bogdanov, D.Y.; Viktorova, I.A.; Grinshtein, Y.I.; Duplyakov, D.V. Cross-sectional Study to Estimate the Prevalence of Familial Hypercholesterolemia in Selected Regions of the Russian Federation: Relevance, Design of the Study and Initial Characteristics of the Participants. Ration. Pharmacother. Cardiol. 2020, 16, 24–32. [Google Scholar] [CrossRef]
- Wiegman, A.; Gidding, S.S.; Watts, G.F.; Chapman, M.J.; Ginsberg, H.N.; Cuchel, M.; Ose, L.; Averna, M.; Boileau, C.; Borén, J. Familial hyper cholesterolaemia in children and adolescents: Gaining decades of life by optimizing detection and treatment. Eur. Heart J. 2015, 36, 2425–2437. [Google Scholar] [CrossRef]
- Wilemon, K.A.; Patel, J.; Aguilar-Salinas, C.; Ahmed, C.D.; Alkhnifsawi, M.; Almahmeed, W.; Alonso, R.; Al-Rasadi, K.; Badimon, L.; Bernal, L.M.; et al. Reducing the Clinical and Public Health Burden of Familial Hypercholesterolemia: A Global Call to Action. JAMA Cardiol. 2020, 5, 217–229. [Google Scholar] [CrossRef]
- Galimova, L.F.; Sadykova, D.I.; Slastnikova, E.S.; Usova, N.E. Diagnosis of familial hypercholesterolemia in children: Cascade screening from theory to practice. Cardiovasc. Ther. Prev. 2020, 19, 2348. [Google Scholar] [CrossRef]
- Leontyeva, I.V. Modern strategy of diagnosis and treatment of children with heterozygous familial hypercholesterolemia. Russ. Bull. Perinatol. Pediatr. 2020, 65, 27–40. [Google Scholar] [CrossRef]
- Alonso, R.; Perez de Isla, L.; Muñiz-Grijalvo, O.; Mata, P. Barriers to early diagnosis and treatment of familial hypercholesterolemia: Current perspectives on improving patient care. Vasc. Health Risk Manag. 2020, 16, 11–25. [Google Scholar] [CrossRef]
- US Preventive Services Task Force; Barry, M.J.; Nicholson, W.K.; Silverstein, M.; Chelmow, D.; Coker, T.R.; Davis, E.M.; Donahue, K.E.; Jaén, C.R.; Li, L.; et al. Screening for Lipid Disorders in Children and Adolescents: US Preventive Services Task Force Recommendation Statement. JAMA 2023, 330, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Clinical Recommendations. Lipid Metabolism Disorders 2023, 74. Available online: https://cr.minzdrav.gov.ru/recomend/752_1 (accessed on 9 October 2023).
- Casula, M.; Gazzotti, M.; Capra, M.E.; Olmastroni, E.; Galimberti, F.; Catapano, A.L.; Pederiva, C.; Anesi, A.; Arca, M.; Auricchio, R.; et al. Refinement of the diagnostic approach for the identification of children and adolescents affected by familial hypercholesterolemia: Evidence from the LIPIGEN study. Atherosclerosis 2023, 385, 117231. [Google Scholar] [CrossRef] [PubMed]
- Groselj, U.; Kovac, J.; Sustar, U.; Mlinaric, M.; Fras, Z.; Podkrajsek, K.T.; Battelino, T. Universal screening for familial hypercholesterolemia in children: The Slovenian model and literature review. Atherosclerosis 2018, 277, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, A.M.; Alves, A.C.; Aguiar, P.; Bourbon, M. Pediatric Investigators of the Portuguese Familial Hypercholesterolemia Study. Cardiovascular risk assessment of dyslipidemic children: Analysis of biomarkers to identify monogenic dyslipidemia. J. Lipid Res. 2014, 55, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Eissa, M.A.; Mihalopoulos, N.L.; Holubkov, R.; Dai, S.; Labarthe, D.R. Changes in Fasting Lipids during Puberty. J. Pediatr. 2016, 170, 199–205. [Google Scholar] [CrossRef]
- Shakhtshneider, E.; Ivanoshchuk, D.; Timoshchenko, O.; Orlov, P.; Semaev, S.; Valeev, E.; Goonko, A.; Ladygina, N.; Voevoda, M. Analysis of Rare Variants in Genes Related to Lipid Metabolism in Patients with Familial Hypercholesterolemia in Western Siberia (Russia). J. Pers. Med. 2021, 11, 1232. [Google Scholar] [CrossRef]
- Capra, M.E.; Pederiva, C.; Viggiano, C.; De Santis, R.; Banderali, G.; Biasucci, G. Nutritional Approach to Prevention and Treatment of Cardiovascular Disease in Childhood. Nutrients 2021, 13, 2359. [Google Scholar] [CrossRef]
- Obarzanek, E.; Kimm, S.Y.S.; Lauer, R.M.; Stevens, V.J.; Friedman, L.A.; Dorgan, J.F.; Greenlick, M.R.; Kwiterovich, P.O.; Franklin, F.A.; Barton, B.A.; et al. Long-Term Safety and Efficacy of a Cholesterol-Lowering Diet in Children with Elevated Low-Density Lipoprotein Cholesterol: Seven-Year Results of the Dietary Intervention Study in Children (DISC). Pediatrics 2001, 107, 256–264. [Google Scholar] [CrossRef]
- Rask-Nissilä, L.; Jokinen, E.; Terho, P.; Tammi, A.; Hakanen, M.; Rönnemaa, T.; Viikari, J.; Seppänen, R.; Välimäki, I.; Helenius, H.; et al. Effects of diet on the neurologic development of children at 5 years of age: The STRIP project. J. Pediatr. 2002, 140, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Pederiva, C.; Capra, M.; Viggiano, C.; Rovelli, V.; Banderali, G.; Biasucci, G. Early Prevention of Atherosclerosis: Detection and Management of Hypercholesterolaemia in Children and Adolescents. Life 2021, 11, 345. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Borjabad, C.; Narveud, I.; Christensen, J.J.; Ulven, S.M.; Malo, A.I.; Ibarretxe, D.; Girona, J.; Torvik, K.; Bogsrud, M.P.; Retterstøl, K.; et al. Dietary intake and lipid levels in Norwegian and Spanish children with familial hypercholesterolemia. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 1299–1307. [Google Scholar] [CrossRef] [PubMed]
- Capra, M.E.; Pederiva, C.; Viggiano, C.; Fabrizi, E.; Banderali, G.; Biasucci, G. Nutritional Treatment in a Cohort of Pediatric Patients with Familial Hypercholesterolaemia: Effect on Lipid Profile. Nutrients 2022, 14, 2817. [Google Scholar] [CrossRef] [PubMed]
- Watts, G.F.; Gidding, S.S.; Hegele, R.A.; Raal, F.J.; Sturm, A.C.; Jones, L.K.; Sarkies, M.N.; Al-Rasadi, K.; Blom, D.J.; Daccord, M.; et al. International Atherosclerosis Society guidance for implementing best practice in the care of familial hypercholesterolaemia. Nat. Rev. Cardiol. 2023, 20, 845–869. [Google Scholar] [CrossRef]
- Langslet, G.; Johansen, A.K.; Bogsrud, M.P.; Narverud, I.; Risstad, H.; Retterstøl, K.; Holven, K.B. Thirty percent of children and young adults with familial hypercholesterolemia treated with statins have adherence issues. Am. J. Prev. Cardiol. 2021, 6, 100180. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D.W. Purification of nucleic acids by extraction with phenol: Chloroform. CSH Protoc. 2006, 1, pdb.prot4455. [Google Scholar] [CrossRef]
- Chen, S.; Francioli, L.C.; Goodrich, J.K.; Collins, R.L.; Wang, Q.; Alföldi, J.; Watts, N.A.; Vittal, C.; Gauthier, L.D.; Poterba, T.; et al. A genome-wide mutational constraint map quantified from variation in 76,156 human genomes. bioRxiv 2022. [Google Scholar] [CrossRef]
| Parameter | Age Groups | p | |
|---|---|---|---|
| <18 Years | 18–44 Years | ||
| Number of subjects | 17 | 43 | - |
| Men, n (%) | 5 (29) | 15 (35) | 0.047 |
| Age, years, Me [25th; 75th] | 10 [7.5; 14] | 35 [28; 39] | <0.001 |
| Family history of CVDs, n (%) | 16 (94) | 38 (88) | <0.001 |
| Body-mass index, kg/m2 | 19.3 [16.4; 23.2] | 23.7 [21.6; 28.0] | 0.008 |
| Tendon xanthomas, n (%) | 2 (12) | 10 (23) | <0.001 |
| Arterial atherosclerosis, n (%) | - | 12 (28) | <0.001 |
| Maximum level of TC the patient, mmol/L, Me [25th; 75th] | 8.6 [6.6; 10.69] | 9.4 [8.0; 10.96] | 0.199 |
| TC, mmol/L, Me [25th; 75th] | 8.05 [6.43; 9.98] | 8.24 [6.8; 10.2] | 0.525 |
| LDL-C, mmol/L, Me [25th; 75th] | 5.85 [4.15; 7.55] | 5.73 [4.84; 7.31] | 0.717 |
| HDL-C, mmol/L, Me [25th; 75th] | 1.44 [1.17; 1.66] | 1.39 [1.07; 1.91] | 0.925 |
| TG, mmol/L, Me [25th; 75th] | 0.68 [0.55; 0.91] | 1.08 [0.82; 1.93] | 0.005 |
| Lipoprotein a, mg/dL, Me [25th; 75th] | 16.8 [14.1; 31.2] | 23.7 [20.3; 65.1] | 0.245 |
| Patient ID | Position Number in the Reference Sequence | Gene | Position on Chromosome (GRCh38) | Nucleotide Substitution | Amino Acid Replacement | Minor Allele Frequency (MAF) According to the GnomAD Database | Clinical Significance According to the ClinVar Database |
|---|---|---|---|---|---|---|---|
| P82 | rs121908038 | LDLR | 19:11113293 | c.1202T>A | p.Leu401His | ND | Likely pathogenic |
| P60 | rs879254566 | LDLR | 19:11105440 | c.534TT>G | p.Asp178Glu | ND | Pathogenic/likely pathogenic |
| P55 | rs879254721 | LDLR | 19:11107496 | c.922G>A | p.Glu308Lys | ND | Pathogenic |
| P4, P6, P7 | rs879254980 | LDLR | 19:11116179 | c.1672G>T | p.Glu558Ter | ND | Pathogenic |
| P36, P37 | rs879255191 | LDLR | 19:11128090 | c.2389+5G>A | - | ND | Conflicting interpretations of pathogenic/likely pathogenic |
| P73 | rs570942190 | LDLR | 19:11113337 | c.1246C>T | p.Arg416Trp | T = 0.000007 | Not reported in ClinVar |
| P22 | rs5742904 | APOB | 2:21006288 | c.10580G>A | p.Arg3527Gln | T = 0.000275 | Pathogenic |
| P95 | rs145164937 | ABCG5 | 2:43832056 | c.293C>G | p.Ala98Gly | C = 0.002223 | Conflicting interpretations of pathogenicity/likely pathogenic |
| Subject ID | Position No. in Reference Sequence | Gene | Position on Chromosome (GRCh38) | Nucleotide Substitution | Amino Acid Substitution | Minor Allele Frequency (MAF) According to GnomAD | Clinical Significance According to ClinVar |
|---|---|---|---|---|---|---|---|
| P5 | rs879254980 | LDLR | 19:11116179 | c.1672G>T | p.Glu558Ter | ND | Pathogenic |
| P34, P53 | rs28942078 | LDLR | 19:11113376 | c.1285G>A | p.Val429Met | A = 0.000012 | Pathogenic |
| P38, P39 | rs879255191 | LDLR | 19:11128090 | c.2389+5G>A | - | ND | Conflicting interpretations of pathogenic/likely pathogenic |
| P50 | NM_000527.4:c.(67+1_68-1)_(1586+1_1587-1)del | LDLR | - | - | - | ND | Eliminated a region spanning exons 2 to 10 |
| P57, P58, P81, P103 | rs121908038 | LDLR | 19:11113293 | c.1202T>A | p.Leu401His | ND | Likely pathogenic |
| P65 | rs137853964 | LDLR | 19:11129602 | c.2479G>A | p.Val827Ile | A = 0.001006 | Likely pathogenic |
| P70 | rs570942190 | LDLR | 19:11113337 | c.1246C>T | p.Arg416Trp | T = 0.000024 | Not reported in ClinVar |
| P78 | rs875989907 | LDLR | 19:11106666 | c.796G>A | p.Asp266Asn | A = 0.000012 | Pathogenic |
| rs879254769 | LDLR | 19:11110765 | c.1054T>C | p.Cys352Ser | ND | Likely pathogenic | |
| P90 | rs755757866 | LDLR | 19:11110730 | c.1019G>T | p.Cys340Tyr | T = 0.000008 | Likely pathogenic |
| P76 | rs2147257524 | LDLR | 19:11116909 | c.1756T>C | p.Ser586Pro | ND | Likely pathogenic |
| P23 | rs773328511 | LDLR | 19:11106680 | c.810C>A | p.Cys270Ter | T = 0.000008 | Pathogenic |
| P25, P94 | rs5742904 | APOB | 2:21006288 | c.10580G>A | p.Arg3527Gln | T = 0.000275 | Pathogenic |
| P96 | rs145164937 | ABCG5 | 2:43832056 | c.293C>G | p.Ala98Gly | C = 0.002223 | Conflicting interpretations of pathogenicity/likely pathogenic |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timoshchenko, O.; Ivanoshchuk, D.; Semaev, S.; Orlov, P.; Zorina, V.; Shakhtshneider, E. Diagnosis of Familial Hypercholesterolemia in Children and Young Adults. Int. J. Mol. Sci. 2024, 25, 314. https://doi.org/10.3390/ijms25010314
Timoshchenko O, Ivanoshchuk D, Semaev S, Orlov P, Zorina V, Shakhtshneider E. Diagnosis of Familial Hypercholesterolemia in Children and Young Adults. International Journal of Molecular Sciences. 2024; 25(1):314. https://doi.org/10.3390/ijms25010314
Chicago/Turabian StyleTimoshchenko, Olga, Dinara Ivanoshchuk, Sergey Semaev, Pavel Orlov, Valentina Zorina, and Elena Shakhtshneider. 2024. "Diagnosis of Familial Hypercholesterolemia in Children and Young Adults" International Journal of Molecular Sciences 25, no. 1: 314. https://doi.org/10.3390/ijms25010314
APA StyleTimoshchenko, O., Ivanoshchuk, D., Semaev, S., Orlov, P., Zorina, V., & Shakhtshneider, E. (2024). Diagnosis of Familial Hypercholesterolemia in Children and Young Adults. International Journal of Molecular Sciences, 25(1), 314. https://doi.org/10.3390/ijms25010314





