A Longitudinal Study of Individual Radiation Responses in Pediatric Patients Treated with Proton and Photon Radiotherapy, and Interventional Cardiology: Rationale and Research Protocol of the HARMONIC Project
Abstract
1. Introduction
- -
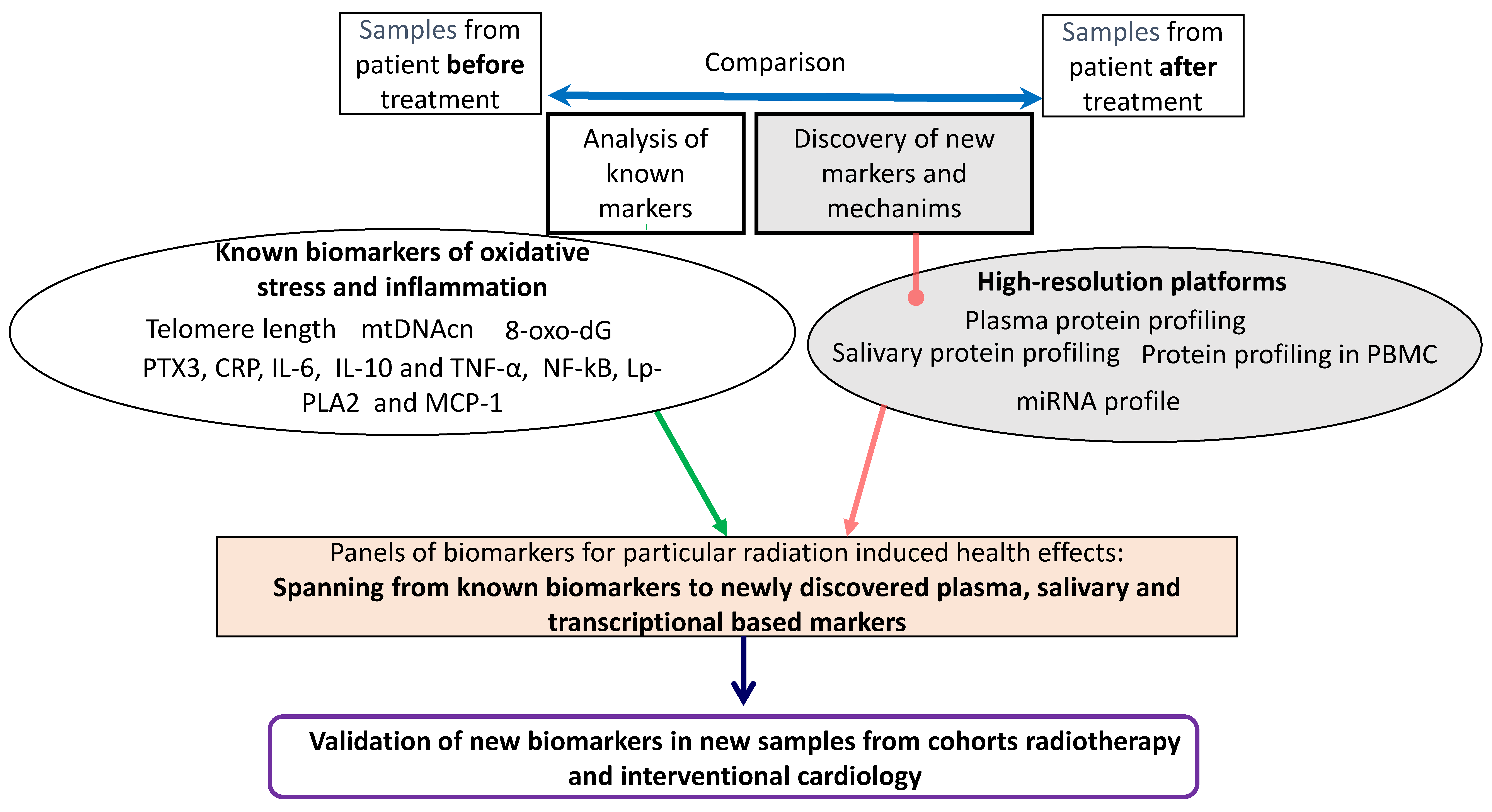
- identify radiation-induced biochemical responses in blood and saliva from pediatric patients exposed to medical IR;
- -
- evaluate dose–response relationships for different radiation qualities and delivery techniques with regards to specific biochemical responses;
- -
- search for pre-existing biomarkers of radiation sensitivity and health effects that may be useful for molecular epidemiological studies to identify patients with a potential higher risk of radiation-induced adverse health effects.
2. Experimental Design
3. Material and Equipment
3.1. Study Population
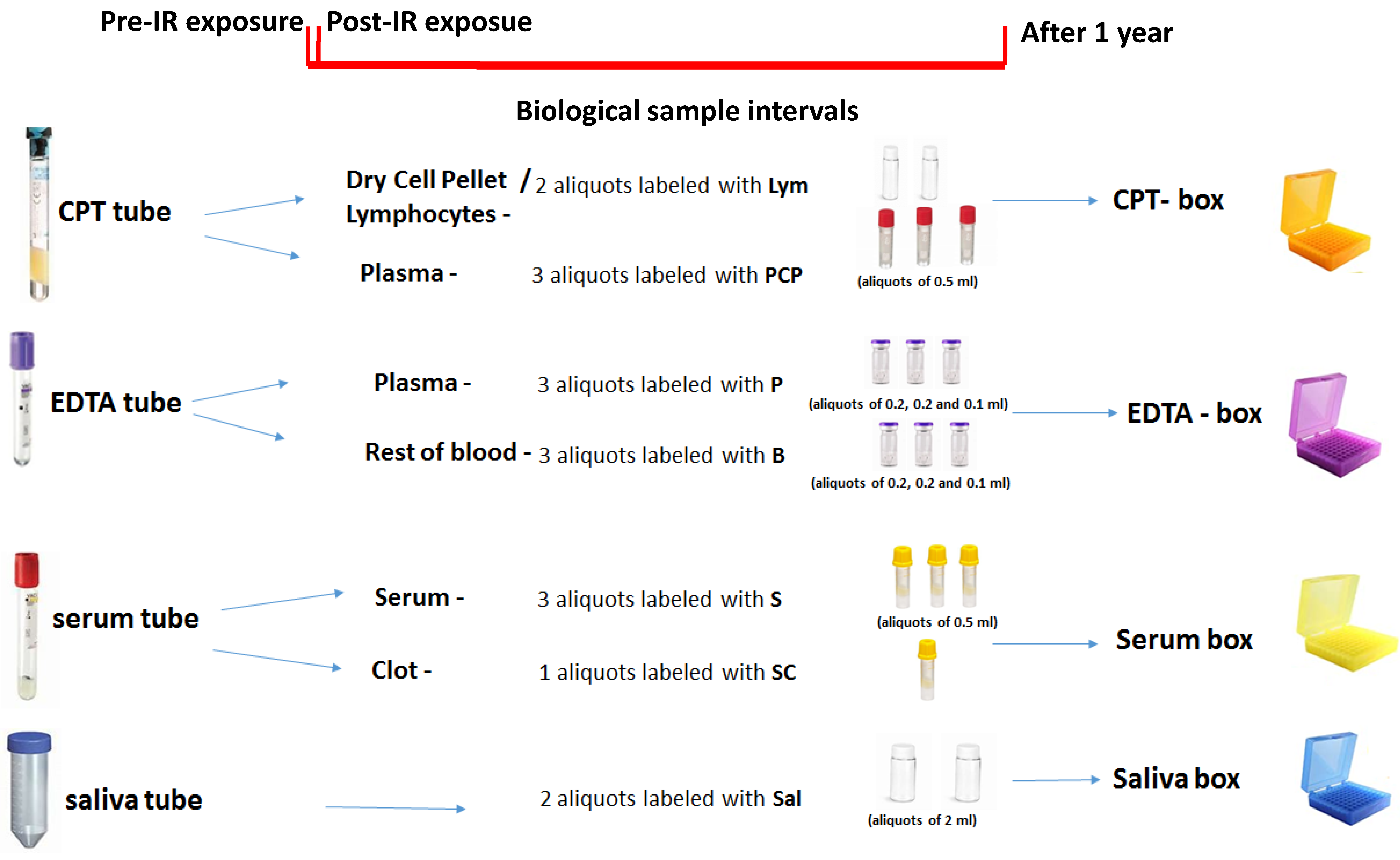
3.2. Biological Sample Collection
- -
- one BD vacutainer® CPT™ tube for the isolation of lymphocytes (~4 mL);
- -
- one vacutainer tube containing EDTA K2 (~4 mL);
- -
- one clot activator serum separation tube (~4 mL).
3.3. Biological Measures
4. Detailed Procedure
4.1. 8-Hydroxy-2′-deoxyguanosine (8-oxo-dG) and Markers of Inflammation
4.2. Analysis of Telomere Length (TL) and mtDNA Copy Number (mtDNA-CN)
4.3. miRNA Profiling Analysis
4.4. Plasma Protein Profiling
4.5. Reverse-Phase Protein Arrays (RPPAs)
4.6. Saliva Protein Analysis
4.7. Radiation Doses Data
4.8. Integrative Analysis of Biological Function and Networks
4.9. Sample Size and Plan for Statistical Analysis
5. Expected Results
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| mtDNAcn | mtDNA copy number |
| 8-Oxo-dG | 8-hydroxy-2′-deoxyguanosine |
| IL-6 | interleukin-6 |
| IL-10 | interleukin-10 |
| TNF-α | tumor necrosis factor-α |
| MCP-1 | monocyte chemoattractant protein-1 |
| NF-kB | nuclear factor kappaB |
| PBMC | peripheral blood mononuclear cell |
| PTX3 | pentraxin 3 |
| PBMC | peripheral blood mononuclear cells |
| miRNA | microRNA |
| Lym | cell pellet/lymphocytes |
| PCP | plasma from CPT |
| P | plasma from EDTA |
| B | rest of blood |
| S | serum |
| SC | blood clot |
| Sal | saliva |
References
- Delaney, G.; Jacob, S.; Featherstone, C.; Barton, M. The role of radiotherapy in cancer treatment: Estimating optimal utilization from a review of evidence-based clinical guidelines. Cancer 2005, 104, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Brower, C.; Rehani, M.M. Radiation risk issues in recurrent imaging. Br. J. Radiol. 2021, 94, 20210389. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.D.; Frush, D.P.; Han, B.K.; Abbott, B.G.; Armstrong, A.K.; DeKemp, R.A.; Glatz, A.C.; Greenberg, S.B.; Herbert, A.S.; Justino, H.; et al. Radiation Safety in Children with Congenital and Acquired Heart Disease: A Scientific Position Statement on Multimodality Dose Optimization from the Image Gently Alliance. JACC Cardiovasc. Imaging 2017, 10, 797–818. [Google Scholar] [CrossRef] [PubMed]
- Andreassi, M.G.; Picano, E. Reduction of radiation to children: Our responsibility to change. Circulation 2014, 130, 135–137. [Google Scholar] [CrossRef]
- Hauptmann, M.; Daniels, R.D.; Cardis, E.; Cullings, H.M.; Kendall, G.; Laurier, D.; Linet, M.S.; Little, M.P.; Lubin, J.H.; Preston, D.L.; et al. Epidemiological Studies of Low-Dose Ionizing Radiation and Cancer: Summary Bias Assessment and Meta-Analysis. J. Natl. Cancer Inst. Monogr. 2020, 2020, 188–200. [Google Scholar] [CrossRef]
- Pettorini, B.L.; Park, Y.S.; Caldarelli, M.; Massimi, L.; Tamburrini, G.; Di Rocco, C. Radiation-induced brain tumours after central nervous system irradiation in childhood: A review. Childs Nerv. Syst. 2008, 24, 793–805. [Google Scholar] [CrossRef]
- Zahnreich, S.; Schmidberger, H. Childhood Cancer: Occurrence, Treatment and Risk of Second Primary Malignancies. Cancers 2021, 13, 2607. [Google Scholar] [CrossRef]
- Kreuzer, M.; Bouffler, S. Guest editorial: Non-cancer effects of ionizing radiation—Clinical implications, epidemiological and mechanistic evidence and research gaps. Environ. Int. 2021, 149, 106286. [Google Scholar] [CrossRef]
- El-Fayech, C.; Haddy, N.; Allodji, R.S.; Veres, C.; Diop, F.; Kahlouche, A.; Llanas, D.; Jackson, A.; Rubino, C.; Guibout, C.; et al. Cerebrovascular Diseases in Childhood Cancer Survivors: Role of the Radiation Dose to Willis Circle Arteries. Int. J. Radiat. Oncol. Biol. Phys. 2017, 97, 278–286. [Google Scholar] [CrossRef]
- Haddy, N.; Mousannif, A.; Tukenova, M.; Guibout, C.; Grill, J.; Dhermain, F.; Pacquement, H.; Oberlin, O.; El-Fayech, C.; Rubino, C.; et al. Relationship between the brain radiation dose for the treatment of childhood cancer and the risk of long-term cerebrovascular mortality. Brain 2011, 134, 1362–1372. [Google Scholar] [CrossRef]
- Darby, S.C.; Ewertz, M.; McGale, P.; Bennet, A.M.; Blom-Goldman, U.; Bronnum, D.; Correa, C.; Cutter, D.; Gagliardi, G.; Gigante, B.; et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N. Engl. J. Med. 2013, 368, 987–998. [Google Scholar] [CrossRef]
- Haddy, N.; Diallo, S.; El-Fayech, C.; Schwartz, B.; Pein, F.; Hawkins, M.; Veres, C.; Oberlin, O.; Guibout, C.; Pacquement, H.; et al. Cardiac Diseases Following Childhood Cancer Treatment: Cohort Study. Circulation 2016, 133, 31–38. [Google Scholar] [CrossRef]
- Cohen, S.; Liu, A.; Gurvitz, M.; Guo, L.; Therrien, J.; Laprise, C.; Kaufman, J.S.; Abrahamowicz, M.; Marelli, A.J. Exposure to Low-Dose Ionizing Radiation from Cardiac Procedures and Malignancy Risk in Adults with Congenital Heart Disease. Circulation 2018, 137, 1334–1345. [Google Scholar] [CrossRef]
- Abalo, K.D.; Malekzadeh-Milani, S.; Hascoet, S.; Dreuil, S.; Feuillet, T.; Cohen, S.; Dauphin, C.; Filippo, S.D.; Douchin, S.; Godart, F.; et al. Exposure to low-dose ionising radiation from cardiac catheterisation and risk of cancer: The COCCINELLE study cohort profile. BMJ. Open 2021, 11, e048576. [Google Scholar] [CrossRef]
- Lang, N.N.; Walker, N.L. Adult Congenital Heart Disease and Radiation Exposure: The Malignant Price of Cardiac Care. Circulation 2018, 137, 1346–1348. [Google Scholar] [CrossRef]
- Foffa, I.; Cresci, M.; Andreassi, M.G. Health risk and biological effects of cardiac ionising imaging: From epidemiology to genes. Int. J. Environ. Res. Public Health 2009, 6, 1882–1893. [Google Scholar] [CrossRef]
- Pernot, E.; Hall, J.; Baatout, S.; Benotmane, M.A.; Blanchardon, E.; Bouffler, S.; El Saghire, H.; Gomolka, M.; Guertler, A.; Harms-Ringdahl, M.; et al. Ionizing radiation biomarkers for potential use in epidemiological studies. Mutat. Res. 2012, 751, 258–286. [Google Scholar] [CrossRef]
- Averbeck, D.; Candeias, S.; Chandna, S.; Foray, N.; Friedl, A.A.; Haghdoost, S.; Jeggo, P.A.; Lumniczky, K.; Paris, F.; Quintens, R.; et al. Establishing mechanisms affecting the individual response to ionizing radiation. Int. J. Radiat. Biol. 2020, 96, 297–323. [Google Scholar] [CrossRef]
- Haghdoost, S.; Czene, S.; Naslund, I.; Skog, S.; Harms-Ringdahl, M. Extracellular 8-oxo-dG as a sensitive parameter for oxidative stress in vivo and in vitro. Free Radic. Res. 2005, 39, 153–162. [Google Scholar] [CrossRef]
- Haghdoost, S.; Svoboda, P.; Naslund, I.; Harms-Ringdahl, M.; Tilikides, A.; Skog, S. Can 8-oxo-dG be used as a predictor for individual radiosensitivity? Int. J. Radiat. Oncol. Biol. Phys. 2001, 50, 405–410. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Fiotakis, C. 8-hydroxy-2′-deoxyguanosine (8-OHdG): A critical biomarker of oxidative stress and carcinogenesis. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2009, 27, 120–139. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimian, T.; Le Gallic, C.; Stefani, J.; Dublineau, I.; Yentrapalli, R.; Harms-Ringdahl, M.; Haghdoost, S. Chronic Gamma-Irradiation Induces a Dose-Rate-Dependent Pro-inflammatory Response and Associated Loss of Function in Human Umbilical Vein Endothelial Cells. Radiat. Res. 2015, 183, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Halle, M.; Gabrielsen, A.; Paulsson-Berne, G.; Gahm, C.; Agardh, H.E.; Farnebo, F.; Tornvall, P. Sustained inflammation due to nuclear factor-kappa B activation in irradiated human arteries. J. Am. Coll. Cardiol. 2010, 55, 1227–1236. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Zhou, Z.; Wei, S.; Liu, Z.; Pooley, K.A.; Dunning, A.M.; Svenson, U.; Roos, G.; Hosgood, H.D.; Shen, M.; et al. Shortened telomere length is associated with increased risk of cancer: A meta-analysis. PLoS ONE 2011, 6, e20466. [Google Scholar] [CrossRef]
- Vecoli, C.; Borghini, A.; Foffa, I.; Ait-Ali, L.; Picano, E.; Andreassi, M.G. Leukocyte telomere shortening in grown-up patients with congenital heart disease. Int. J. Cardiol. 2016, 204, 17–22. [Google Scholar] [CrossRef]
- Borghini, A.; Vecoli, C.; Piccaluga, E.; Guagliumi, G.; Picano, E.; Andreassi, M.G. Increased mitochondrial DNA4977-bp deletion in catheterization laboratory workers with long-term low-dose exposure to ionizing radiation. Eur. J. Prev. Cardiol. 2019, 26, 976–984. [Google Scholar] [CrossRef]
- Hu, L.; Yao, X.; Shen, Y. Altered mitochondrial DNA copy number contributes to human cancer risk: Evidence from an updated meta-analysis. Sci. Rep. 2016, 6, 35859. [Google Scholar] [CrossRef]
- Kam, W.W.; Banati, R.B. Effects of ionizing radiation on mitochondria. Free Radic. Biol. Med. 2013, 65, 607–619. [Google Scholar] [CrossRef]
- Campa, D.; Barrdahl, M.; Santoro, A.; Severi, G.; Baglietto, L.; Omichessan, H.; Tumino, R.; Bueno-de-Mesquita, H.B.A.; Peeters, P.H.; Weiderpass, E.; et al. Mitochondrial DNA copy number variation, leukocyte telomere length, and breast cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Breast Cancer Res. 2018, 20, 29. [Google Scholar] [CrossRef]
- Assarsson, E.; Lundberg, M.; Holmquist, G.; Björkesten, J.; Thorsen, S.B.; Ekman, D.; Eriksson, A.; Rennel Dickens, E.; Ohlsson, S.; Edfeldt, G.; et al. Homogenous 96-plex pea immunoassay exhibiting high sensitivity, specificity, and excellent scalability. PLoS ONE 2014, 9, e95192. [Google Scholar] [CrossRef]
- Troncale, S.; Barbet, A.; Coulibaly, L.; Henry, E.; He, B.; Barillot, E.; Dubois, T.; Hupé, P.; de Koning, L. NormaCurve: A SuperCurve-based method that simultaneously quantifies and normalizes reverse phase protein array data. PLoS ONE 2012, 7, e38686. [Google Scholar] [CrossRef]
- Borghini, A.; Vecoli, C.; Mercuri, A.; Carpeggiani, C.; Piccaluga, E.; Guagliumi, G.; Picano, E.; Andreassi, M.G. Low-Dose Exposure to Ionizing Radiation Deregulates the Brain-Specific MicroRNA-134 in Interventional Cardiologists. Circulation 2017, 136, 2516–2518. [Google Scholar] [CrossRef]
- Harbron, R.W.; Thierry-Chef, I.; Pearce, M.S.; Bernier, M.O.; Dreuil, S.; Rage, E.; Andreassi, M.G.; Picano, E.; Dreger, S.; Zeeb, H.; et al. The HARMONIC project: Study design for the assessment of radiation doses and associated cancer risks following cardiac fluoroscopy in childhood. J. Radiol. Prot. 2020, 40, 1074. [Google Scholar]
- De Saint-Hubert, M.; Verbeek, N.; Baumer, C.; Esser, J.; Wulff, J.; Nabha, R.; Van Hoey, O.; Dabin, J.; Stuckmann, F.; Vasi, F.; et al. Validation of a Monte Carlo Framework for Out-of-Field Dose Calculations in Proton Therapy. Front. Oncol. 2022, 12, 882489. [Google Scholar] [CrossRef]
- De Saint-Hubert, M.; Suesselbeck, F.; Vasi, F.; Stuckmann, F.; Rodriguez, M.; Dabin, J.; Timmermann, B.; Thierry-Chef, I.; Schneider, U.; Brualla, L. Experimental Validation of an Analytical Program and a Monte Carlo Simulation for the Computation of the Far Out-of-Field Dose in External Beam Photon Therapy Applied to Pediatric Patients. Front. Oncol. 2022, 12, 882506. [Google Scholar] [CrossRef]
- Ding, G.X.; Alaei, P.; Curran, B.; Flynn, R.; Gossman, M.; Mackie, T.R.; Miften, M.; Morin, R.; Xu, X.G.; Zhu, T.C. Image guidance doses delivered during radiotherapy: Quantification, management, and reduction: Report of the AAPM Therapy Physics Committee Task Group 180. Med. Phys. 2018, 45, e84–e99. [Google Scholar] [CrossRef]
- McFadden, S.L.; Hughes, C.M.; Mooney, R.B.; Winder, R.J. An analysis of radiation dose reduction in paediatric interventional cardiology by altering frame rate and use of the anti-scatter grid. J. Radiol. Prot. 2013, 33, 433–443. [Google Scholar] [CrossRef]
- Reisz, J.A.; Bansal, N.; Qian, J.; Zhao, W.; Furdui, C.M. Effects of ionizing radiation on biological molecules-mechanisms of damage and emerging methods of detection. Antioxid. Redox. Signal. 2014, 21, 260–292. [Google Scholar] [CrossRef]
- Hall, J.; Jeggo, P.A.; West, C.; Gomolka, M.; Quintens, R.; Badie, C.; Laurent, O.; Aerts, A.; Anastasov, N.; Azimzadeh, O.; et al. Ionizing radiation biomarkers in epidemiological studies—An update. Mutat. Res. Rev. Mutat. Res. 2017, 771, 59–84. [Google Scholar] [CrossRef]
- Pfaffe, T.; Cooper-White, J.; Beyerlein, P.; Kostner, K.; Punyadeera, C. Diagnostic potential of saliva: Current state and future applications. Clin. Chem. 2011, 57, 675–687. [Google Scholar] [CrossRef]
- Michalke, B.; Rossbach, B.; Goen, T.; Schaferhenrich, A.; Scherer, G. Saliva as a matrix for human biomonitoring in occupational and environmental medicine. Int. Arch. Occup. Environ. Health 2015, 88, 1–44. [Google Scholar] [CrossRef] [PubMed]
| Radiotherapy | Interventional Cardiology |
|---|---|
| Inclusion criteria | |
|
|
| Exclusion criteria | |
| |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andreassi, M.G.; Haddy, N.; Harms-Ringdahl, M.; Campolo, J.; Borghini, A.; Chevalier, F.; Schwenk, J.M.; Fresneau, B.; Bolle, S.; Fuentes, M.; et al. A Longitudinal Study of Individual Radiation Responses in Pediatric Patients Treated with Proton and Photon Radiotherapy, and Interventional Cardiology: Rationale and Research Protocol of the HARMONIC Project. Int. J. Mol. Sci. 2023, 24, 8416. https://doi.org/10.3390/ijms24098416
Andreassi MG, Haddy N, Harms-Ringdahl M, Campolo J, Borghini A, Chevalier F, Schwenk JM, Fresneau B, Bolle S, Fuentes M, et al. A Longitudinal Study of Individual Radiation Responses in Pediatric Patients Treated with Proton and Photon Radiotherapy, and Interventional Cardiology: Rationale and Research Protocol of the HARMONIC Project. International Journal of Molecular Sciences. 2023; 24(9):8416. https://doi.org/10.3390/ijms24098416
Chicago/Turabian StyleAndreassi, Maria Grazia, Nadia Haddy, Mats Harms-Ringdahl, Jonica Campolo, Andrea Borghini, François Chevalier, Jochen M. Schwenk, Brice Fresneau, Stephanie Bolle, Manuel Fuentes, and et al. 2023. "A Longitudinal Study of Individual Radiation Responses in Pediatric Patients Treated with Proton and Photon Radiotherapy, and Interventional Cardiology: Rationale and Research Protocol of the HARMONIC Project" International Journal of Molecular Sciences 24, no. 9: 8416. https://doi.org/10.3390/ijms24098416
APA StyleAndreassi, M. G., Haddy, N., Harms-Ringdahl, M., Campolo, J., Borghini, A., Chevalier, F., Schwenk, J. M., Fresneau, B., Bolle, S., Fuentes, M., & Haghdoost, S. (2023). A Longitudinal Study of Individual Radiation Responses in Pediatric Patients Treated with Proton and Photon Radiotherapy, and Interventional Cardiology: Rationale and Research Protocol of the HARMONIC Project. International Journal of Molecular Sciences, 24(9), 8416. https://doi.org/10.3390/ijms24098416










