Dietary Exposure to Particles of Polytetrafluoroethylene (PTFE) and Polymethylmethacrylate (PMMA) Induces Different Responses in Periwinkles Littorina brevicula
Abstract
1. Introduction
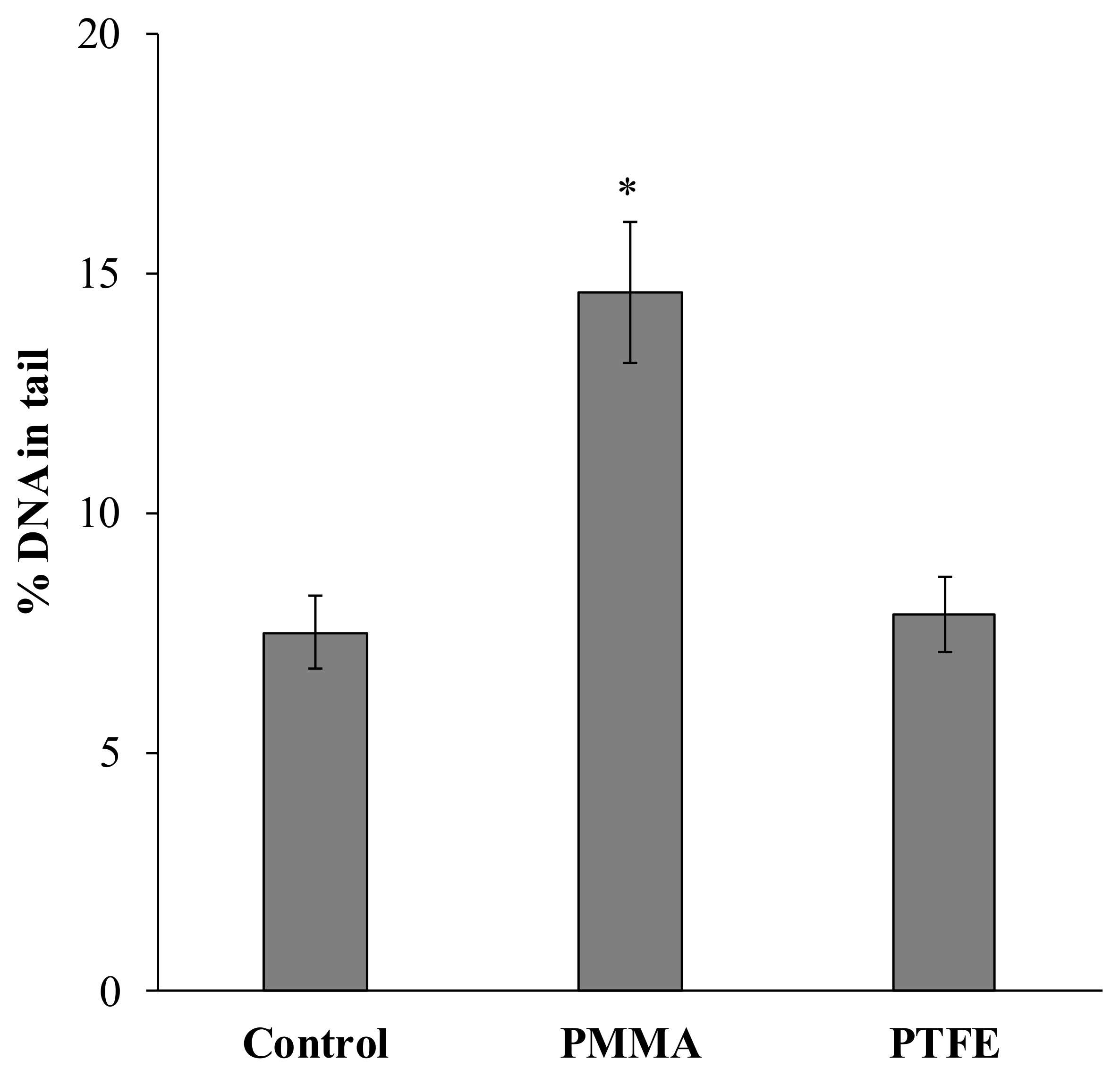
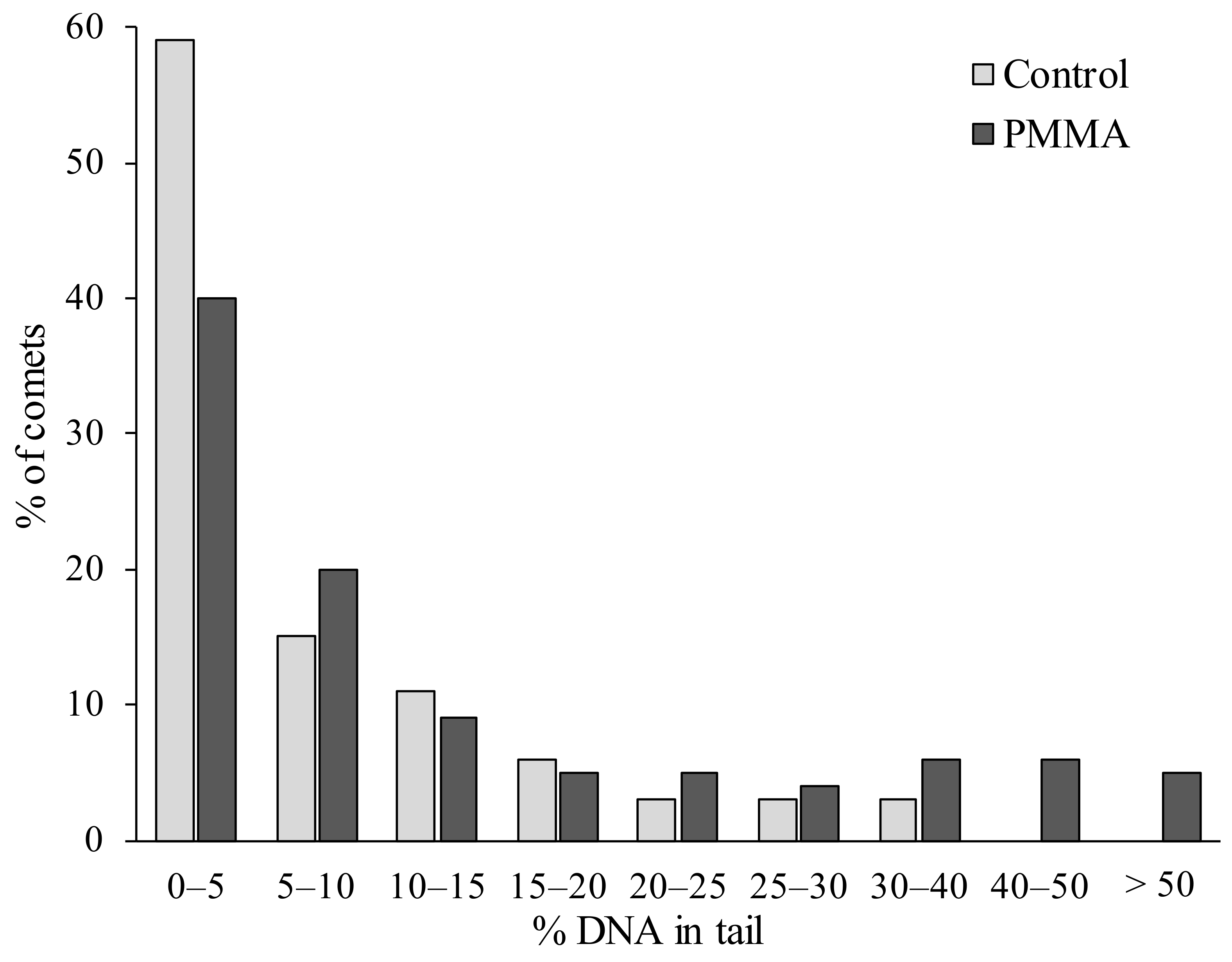
2. Results and Discussion
3. Material and Methods
3.1. Description of the Experiment
3.2. Comet Assay
3.3. MDA Concentration
3.4. Integral Antioxidant Activity
3.5. Carbonyl Concentration
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stefatos, A.; Charalampakis, M.; Papatheodorou, G.; Ferentinos, G. Marine debris on the seafloor of the mediterranean sea: Examples from two enclosed gulfs in Western Greece. Mar. Pollut. Bull. 1999, 38, 389–393. [Google Scholar] [CrossRef]
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.; McGonigle, D.; Russell, A.E. Lost at sea: Where is all the plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.K.; Walters, A.; Gonçalves, L. Macroplastics at sea around Antarctica. Mar. Environ. Res. 2010, 70, 250–252. [Google Scholar] [CrossRef] [PubMed]
- Isobe, A.; Uchiyama-Matsumoto, K.; Uchida, K.; Tokai, T. Microplastics in the Southern Ocean. Mar. Pollut. Bull. 2017, 114, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Murray, F.; Cowie, P.R. Plastic contamination in the decapod crustacean Nephrops norvegicus (Linnaeus, 1758). Mar. Pollut. Bull. 2011, 62, 1207–1217. [Google Scholar] [CrossRef]
- Van Cauwenberghe, L.; Janssen, C.R. Microplastics in bivalves cultured for human consumption. Environ. Pollut. 2014, 193, 65–70. [Google Scholar] [CrossRef]
- Anbumani, S.; Kakkar, P. Ecotoxicological effects of microplastics on biota: A review. Environ. Sci. Pollut. Res. 2018, 25, 14373–14396. [Google Scholar] [CrossRef]
- Bom, F.C.; Sá, F. Concentration of microplastics in bivalves of the environment: A systematic review. Environ. Monit. Assess. 2021, 193, 846. [Google Scholar] [CrossRef]
- Boerger, C.M.; Lattin, G.L.; Moore, S.L.; Moore, C.J. Plastic ingestion by planktivorous fishes in the North Pacific Central Gyre. Mar. Pollut. Bull. 2010, 60, 2275–2278. [Google Scholar] [CrossRef]
- Dellisanti, W.; Leung, M.M.L.; Lam, K.W.K.; Wang, Y.; Hu, M.; Lo, H.S.; Fang, J.K.H. A short review on the recent method development for extraction and identification of microplastics in mussels and fish, two major groups of seafood. Mar. Pollut. Bull. 2023, 186, 114221. [Google Scholar] [CrossRef]
- Prokic, M.; Radovanovi´c, T.B.; Gavri´c, J.P.; Faggio, C. Ecotoxicological effects of microplastics: Examination of biomarkers, current state and future perspectives. TrAC Trends Anal. Chem. 2019, 111, 37–46. [Google Scholar] [CrossRef]
- Agathokleous, E.; Iavicoli, I.; Barceló, D.; Calabrese, E.J. Micro/nanoplastics effects on organisms: A review focusing on ‘dose’. J. Hazard. Mater. 2021, 417, 126084. [Google Scholar] [CrossRef] [PubMed]
- Berlino, M.; Mangano, M.; De Vittor, C.; Sarà, G. Effects of microplastics on the functional traits of aquatic benthic organisms: A global-scale meta-analysis. Environ. Pollut. 2021, 285, 117174. [Google Scholar] [CrossRef] [PubMed]
- Roda, J.F.B.; Lauer, M.M.; Risso, W.E.; Bueno Dos Reis Martinez, C. Microplastics and copper effects on the neotropical teleost Prochilodus lineatus: Is there any interaction? Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2020, 242, 110659. [Google Scholar] [CrossRef]
- D’Costa, A.H. Microplastics in decapod crustaceans: Accumulation, toxicity and impacts, a review. Sci. Total. Environ. 2022, 832, 154963. [Google Scholar] [CrossRef]
- Gonzalez-Soto, N.; Hatfield, J.; Katsumiti, A.; Duroudier, N.; Lacave, J.M.; Bilbao, E.; Orbea, A.; Navarro, E.; Cajaraville, M.P. Impacts of dietary exposure to different sized polystyrene microplastics alone and with sorbed benzo[a]pyrene on biomarkers and whole organism responses in mussels Mytilus galloprovincialis. Sci. Total. Environ. 2019, 684, 548–566. [Google Scholar] [CrossRef]
- Détrée, C.; Gallardo-Escárate, C. Single and repetitive microplastics exposures induce immune system modulation and homeostasis alteration in the edible mussel Mytilus galloprovincialis. Fish Shellfish Immunol. 2018, 83, 52–60. [Google Scholar] [CrossRef]
- Guzzetti, E.; Sureda, A.; Tejada, S.; Faggio, C. Microplastic in marine organism: Environmental and toxicological effects. Environ. Toxicol. Pharmacol. 2018, 64, 164–171. [Google Scholar] [CrossRef]
- Revel, M.; Lagarde, F.; Perrein-Ettajani, H.; Bruneau, M.; Akcha, F.; Sussarellu, R.; Rouxel, J.; Costil, K.; Decottignies, P.; Cognie, B. Tissue-specific biomarker responses in the blue mussel Mytilus spp. exposed to a mixture of microplastics at environmentally relevant concentrations. Front. Environ. Sci. 2019, 7, 33. [Google Scholar] [CrossRef]
- Wang, J.; Liu, X.; Li, Y.; Powell, T.; Wang, X.; Wang, G.; Zhang, P. Microplastics as contaminants in the soil environment: A mini-review. Sci. Total. Environ. 2019, 691, 848–857. [Google Scholar] [CrossRef]
- Doyle, D.; Gammell, M.; Frias, J.; Griffin, G.; Nash, R. Low levels of microplastics recorded from the common periwinkle, Littorina littorea on the west coast of Ireland. Mar. Pollut. Bull. 2019, 149, 110645. [Google Scholar] [CrossRef]
- Gutow, L.; Eckerlebe, A.; Giménez, L.; Saborowski, R. Experimental Evaluation of Seaweeds as a Vector for Microplastics into Marine Food Webs. Environ. Sci. Technol. 2016, 50, 915–923. [Google Scholar] [CrossRef] [PubMed]
- Gutow, L.; Bartl, K.; Saborowski, R.; Beermann, J. Gastropod pedal mucus retains microplastics and promotes the uptake of particles by marine periwinkles. Environ. Pollut. 2019, 246, 688–696. [Google Scholar] [CrossRef] [PubMed]
- Odintsov, V.S.; Karpenko, A.A.; Karpenko, M.A. Degradation of micro-nano-sized polytetrafluoroethylene and acrylic fluorinated copolymer particles in the periwinkle digestive tract. Environ. Sci. Pollut. Res. 2023, 30, 25972–25980. [Google Scholar] [CrossRef]
- Albertini, R.J. The lower alkyl methacrylates: Genotoxic profile of non-carcinogenic compounds. Regul. Toxicol. Pharmacol. 2017, 84, 77–93. [Google Scholar] [CrossRef] [PubMed]
- Venâncio, C.; Melnic, I.; Tamayo-Belda, M.; Oliveira, M.; Martins, M.A.; Lopes, I. Polymethylmethacrylate nanoplastics can cause developmental malformations in early life stages of Xenopus laevis. Sci. Total Environ. 2022, 806, 150491. [Google Scholar] [CrossRef] [PubMed]
- Henry, B.J.; Carlin, J.P.; Hammerschmidt, J.A.; Buck, R.C.; Buxton, L.W.; Fiedler, H.; Seed, J.; Hernandez, O. A critical review of the application of polymer of low concern and regulatory criteria to fluoropolymers. Integr. Environ. Assess. Manag. 2018, 14, 316–334. [Google Scholar] [CrossRef]
- Lithner, D.; Larsson, A.; Dave, G. Environmental and health hazard ranking and assessment of plastic polymers based on chemical composition. Sci. Total Environ. 2011, 409, 3309–3324. [Google Scholar] [CrossRef]
- Yuan, M.; Huang, D.; Zhao, Y. Development of Synthesis and Application of High Molecular Weight Poly(Methyl Methacrylate). Polymers 2022, 14, 2632. [Google Scholar] [CrossRef]
- Thompson, R.C.; Johnson, L.E.; Hawkins, S.J. A method for spatial and temporal assessment of gastropod grazing intensity in the field: The use of radula scrapes on wax surfaces. J. Exp. Mar. Biol. Ecol. 1997, 218, 63–76. [Google Scholar] [CrossRef]
- Davies, M.S.; Beckwith, P. Role of mucus trails and trail-following in the behaviour and nutrition of the periwinkle Littorina littorea. Mar. Ecol. Prog. Ser. 1999, 179, 247–257. [Google Scholar] [CrossRef]
- Avio, C.G.; Gorbi, S.; Milan, M.; Benedetti, M.; Fattorini, D.; Errico, G.; Pauletto, M.; Bargelloni, L.; Regoli, F. Pollutants bioavailability and toxicological risk from microplastics to marine mussels. Environ. Pollut. 2015, 198, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Cole, M.; Galloway, T.S. Ingestion of nanoplastics and microplastics by pacific oyster larvae. Environ. Sci. Technol. 2015, 49, 14625–14632. [Google Scholar] [CrossRef] [PubMed]
- Bergami, E.; Bocci, E.; Vannuccini, M.L.; Monopoli, M.; Salvati, A.; Dawson, K.A.; Corsi, I. Nano-sized polystyrene affects feeding, behavior and physiology of brine shrimp Artemia franciscana larvae. Ecotoxicol. Environ. Saf. 2016, 123, 18–25. [Google Scholar] [CrossRef]
- Jeong, C.B.; Won, E.J.; Kang, H.M.; Lee, M.C.; Hwang, D.S.; Hwang, U.; Zhou, B.; Souissi, S.; Lee, S.J.; Lee, J.S. Microplastic size-dependent toxicity, oxidative stress induction, and p-JNK and p-p38 activation in the monogonont rotifer (Brachionus koreanus). Environ. Sci. Technol. 2016, 50, 8849–8857. [Google Scholar] [CrossRef]
- Paul-Pont, I.; Tallec, K.; Gonzalez-Fernandez, C.; Lambert, C.; Vincent, D.; Mazurais, D.; Zambonino-Infante, J.L.; Brotons, G.; Lagarde, F.; Fabioux, C. Constraints and priorities for conducting experimental exposures of marine organisms to microplastics. Front. Mar. Sci. 2018, 252, 1–22. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Gimenez, B.C.G. Microplastics in the marine environment: Current trends and future perspectives. Mar. Pollut. Bull. 2015, 97, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, C.; Morgana, S.; Bramini, M.; Rotini, A.; Manfra, L.; Migliore, L.; Piazza, V.; Garaventa, F.; Faimali, M. Ecotoxicological effects of polystyrene microbeads in a battery of marine organisms belonging to different trophic levels. Mar. Environ. Res. 2018, 141, 313–321. [Google Scholar] [CrossRef]
- Dovzhenko, N.V.; Chelomin, V.P.; Mazur, A.A.; Kukla, S.P.; Slobodskova, V.V.; Istomina, A.A.; Zhukovskaya, A.F. Oxidative Stress in Far Eastern Mussel Mytilus trossulus (Gould, 1850) Exposed to Combined Polystyrene Microspheres (µPSs) and CuO-Nanoparticles (CuO-NPs). J. Mar. Sci. Eng. 2022, 10, 707. [Google Scholar] [CrossRef]
- Chelomin, V.P.; Mazur, A.A.; Slobodskova, V.V.; Kukla, S.P.; Dovzhenko, N.V. Genotoxic Properties of Polystyrene (PS) Microspheres in the Filter-Feeder Mollusk Mytilus trossulus (Gould, 1850). J. Mar. Sci. Eng. 2022, 10, 273. [Google Scholar] [CrossRef]
- Masiá, P.; Ardura, A.; García-Vázquez, E. Virgin Polystyrene Microparticles Exposure Leads to Changes in Gills DNA and Physical Condition in the Mediterranean Mussel Mytilus Galloprovincialis. Animals 2021, 11, 2317. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Chen, N.; Yang, X.; Xia, Y.; Wu, D. Effects induced by polyethylene microplastics oral exposure on colon mucin release, inflammation, gut microflora composition and metabolism in mice. Ecotoxicol. Environ. Saf. 2021, 220, 112340. [Google Scholar] [CrossRef] [PubMed]
- Brandts, I.; Teles, M.; Tvarijonaviciute, A.; Pereira, M.L.; Martins, M.A.; Tort, L.; Oliveira, M. Effects of polymethylmethacrylate nanoplastics on Dicentrarchus labrax. Genomics 2018, 110, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Von Moos, N.; Burkhardt-Holm, P.; Köhler, A. Uptake and effects of microplastics on cells and tissue of the blue mussel Mytilus edulis L. after an experimental exposure. Environ. Sci. Technol. 2012, 46, 11327–11335. [Google Scholar] [CrossRef]
- Shukla, A.K.; Pragya, P.; Chowdhuri, K.D. A modified alkaline Comet assay for in vivo detection of oxidative DNA damage in Drosophila melanogaster. Mutat. Res. 2011, 726, 222–226. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Lin, S.; Turner, J.P.; Ke, P.C. Physical adsorption of charged plastic nanoparticles affects algal photosynthesis. J. Phys. Chem. 2010, 114, 16556–16561. [Google Scholar] [CrossRef]
- Gambardella, C.; Morgana, S.; Ferrando, S.; Bramini, M.; Piazza, V.; Costa, E.; Garaventa, F.; Faimali, M. Effects of polystyrene microbeads in marine planktonic crustaceans. Ecotoxicol. Environ. Saf. 2017, 145, 250–257. [Google Scholar] [CrossRef]
- Mazur, A.A.; Chelomin, V.P.; Zhuravel, E.V.; Kukla, S.P.; Slobodskova, V.V.; Dovzhenko, N.V. Genotoxicity of Polystyrene (PS) Microspheres in Short-Term Exposure to Gametes of the Sand Dollar Scaphechinus mirabilis (Agassiz, 1864) (Echinodermata, Echinoidea). J. Mar. Sci. Eng. 2021, 9, 1088. [Google Scholar] [CrossRef]
- Naftalovich, R.; Naftalovich, D.; Greenway, F.L. Polytetrafluoroethylene Ingestion as a Way to Increase Food Volume and Hence Satiety Without Increasing Calorie Content. J. Diabetes Sci. Technol. 2016, 10, 971–976. [Google Scholar] [CrossRef]
- Bakopoulou, A.; Papadopoulos, T.; Garefis, P. Molecular toxicology of substances released from resin-based dental restorative materials. Int. J. Mol. Sci. 2009, 10, 3861–3899. [Google Scholar] [CrossRef]
- Araújo, A.M.; Alves, G.R.; Avanço, G.T.; Parizi, J.L.; Nai, G.A. Assessment of methyl methacrylate genotoxicity by the micronucleus test. Braz. Oral Res. 2013, 27, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Krifka, S.; Spagnuolo, G.; Schmalz, G.; Schweikl, H. A review of adaptive mechanisms in cell responses towards oxidative stress caused by dental resin monomers. Biomaterials 2013, 34, 4555–4563. [Google Scholar] [CrossRef] [PubMed]
- Brauner, C.; Joveleviths, D.; Álvares-da-Silva, M.R. Hepatotoxicity by methyl methacrylate. Clin. Res. Hepatol. Gastroenterol. 2021, 45, 101513. [Google Scholar] [CrossRef] [PubMed]
- Trifuoggi, M.; Pagano, G.; Oral, R.; Pavičić-Hamer, D.; Burić, P.; Kovačić, I.; Siciliano, A.; Toscanesi, M.; Thomas, P.J.; Paduano, L.; et al. Microplastic-induced damage in early embryonal development of sea urchin Sphaerechinus granularis. Environ. Res. 2019, 179, 108815. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.J.; Oral, R.; Pagano, G.; Tez, S.; Toscanesi, M.; Ranieri, P.; Trifuoggi, M.; Lyons, D.M. Mild toxicity of polystyrene and polymethylmethacrylate microplastics in Paracentrotus lividus early life stages. Mar. Environ. Res. 2020, 161, 105132. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.R. Investigating oxidative DNA damage and its pepair using the comet assay. Mutat. Res. 2009, 681, 24–32. [Google Scholar] [CrossRef]
- Buege, J.A.; Aust, S.D. Microsomal lipid peroxidation. Methods Enzymol. 1978, 52, 302–310. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chelomin, V.P.; Slobodskova, V.V.; Kukla, S.P.; Mazur, A.A.; Dovzhenko, N.V.; Zhukovskaya, A.F.; Karpenko, A.A.; Karpenko, M.A.; Odintsov, V.S. Dietary Exposure to Particles of Polytetrafluoroethylene (PTFE) and Polymethylmethacrylate (PMMA) Induces Different Responses in Periwinkles Littorina brevicula. Int. J. Mol. Sci. 2023, 24, 8243. https://doi.org/10.3390/ijms24098243
Chelomin VP, Slobodskova VV, Kukla SP, Mazur AA, Dovzhenko NV, Zhukovskaya AF, Karpenko AA, Karpenko MA, Odintsov VS. Dietary Exposure to Particles of Polytetrafluoroethylene (PTFE) and Polymethylmethacrylate (PMMA) Induces Different Responses in Periwinkles Littorina brevicula. International Journal of Molecular Sciences. 2023; 24(9):8243. https://doi.org/10.3390/ijms24098243
Chicago/Turabian StyleChelomin, Victor Pavlovich, Valentina Vladimirovna Slobodskova, Sergey Petrovich Kukla, Andrey Alexandrovich Mazur, Nadezda Vladimirovna Dovzhenko, Avianna Fayazovna Zhukovskaya, Alexander Alexandrovich Karpenko, Maxim Alexandrovich Karpenko, and Vyacheslav Sergeevich Odintsov. 2023. "Dietary Exposure to Particles of Polytetrafluoroethylene (PTFE) and Polymethylmethacrylate (PMMA) Induces Different Responses in Periwinkles Littorina brevicula" International Journal of Molecular Sciences 24, no. 9: 8243. https://doi.org/10.3390/ijms24098243
APA StyleChelomin, V. P., Slobodskova, V. V., Kukla, S. P., Mazur, A. A., Dovzhenko, N. V., Zhukovskaya, A. F., Karpenko, A. A., Karpenko, M. A., & Odintsov, V. S. (2023). Dietary Exposure to Particles of Polytetrafluoroethylene (PTFE) and Polymethylmethacrylate (PMMA) Induces Different Responses in Periwinkles Littorina brevicula. International Journal of Molecular Sciences, 24(9), 8243. https://doi.org/10.3390/ijms24098243






