Basic Pathogenic Mechanisms and Epigenetic Players Promoted by Extracellular Vesicles in Vascular Damage
Abstract
1. Introduction
2. EVs Biology and Function
3. The Epigenetic Role of EVs in Cardiovascular Damage
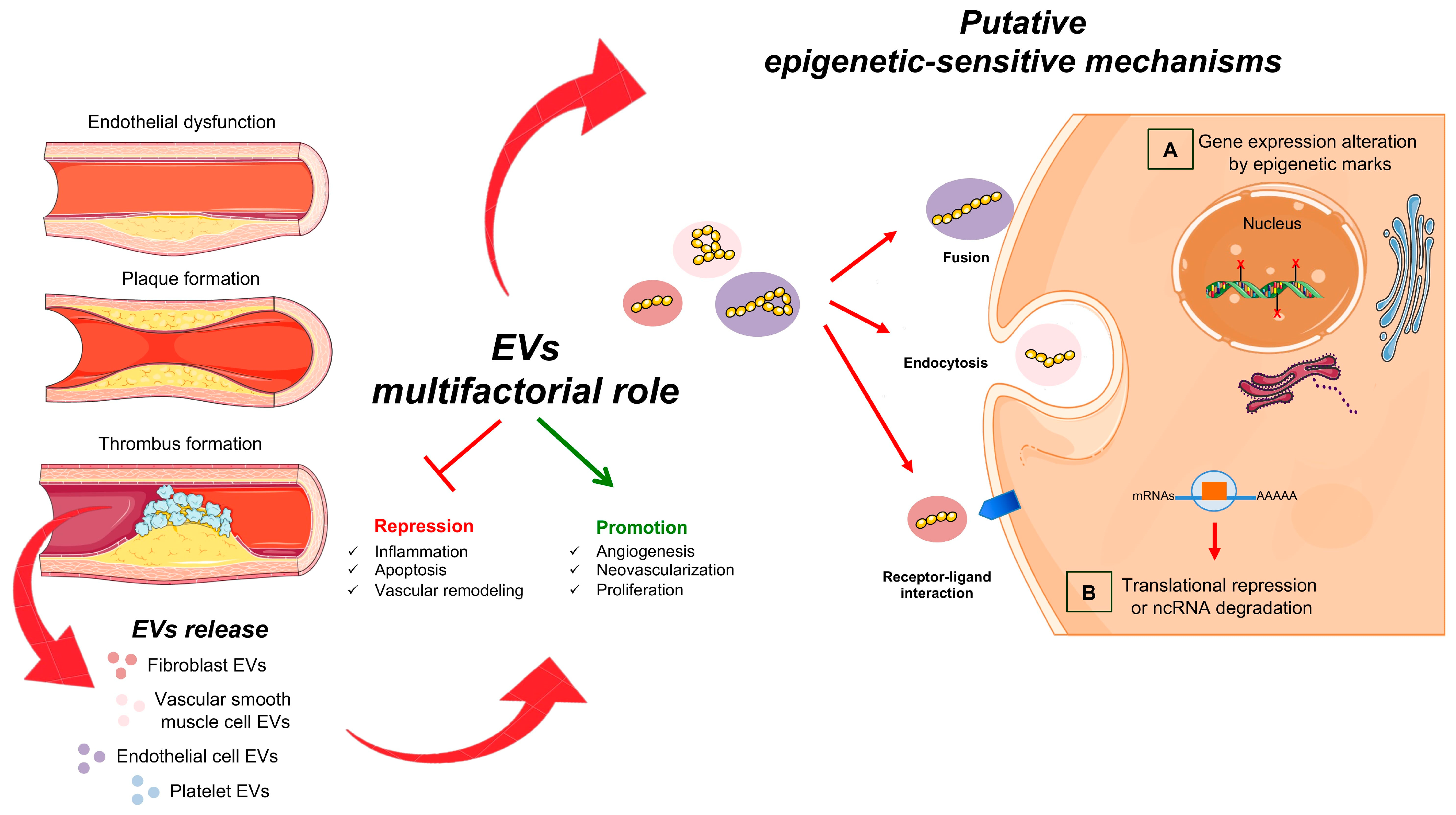
3.1. EVs and Peripheral Endothelial Dysfunction
3.2. EVs in Inflammation and Calcification
3.3. EVs and CHD
4. Basic Mechanisms and Epigenetic Role of EVs in Tumor Vascularization
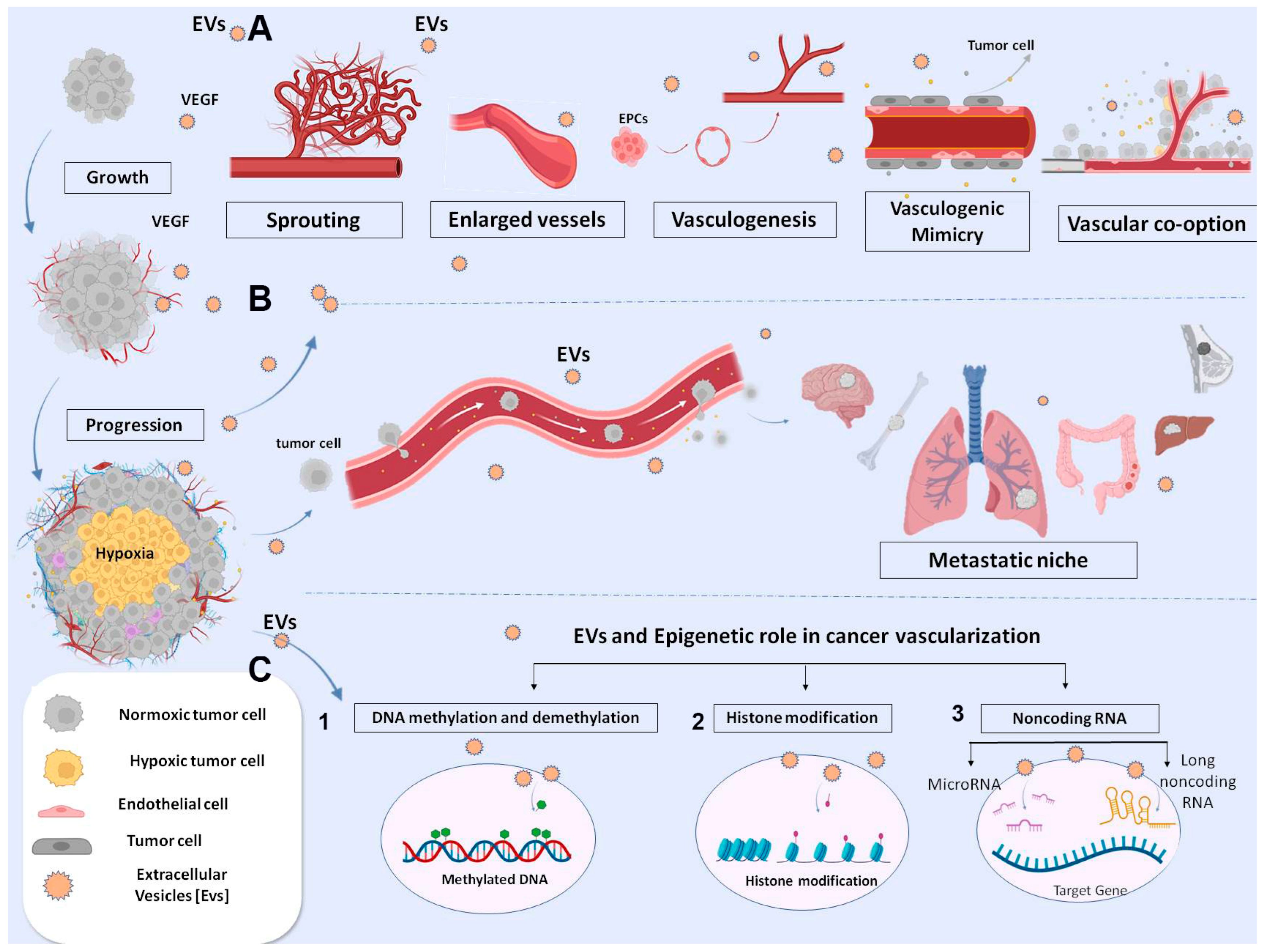
4.1. EVs in Neoangiogenesis
4.2. EVs and Vascular Alterations in Tumor Progression
4.3. EVs in Metastatic Niche: And Other Types of Vessels
5. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Folkman, J.; Shing, Y. Angiogenesis. J. Biol. Chem. 1992, 267, 10931–10934. [Google Scholar] [CrossRef]
- Napoli, C.; Crudele, V.; Soricelli, A.; Al-Omran, M.; Vitale, N.; Infante, T.; Mancini, F.P. Primary prevention of atherosclerosis: A clinical challenge for the reversal of epigenetic mechanisms? Circulation 2012, 125, 2363–2373. [Google Scholar] [CrossRef]
- Napoli, C.; D’Armiento, F.P.; Mancini, F.P.; Postiglione, A.; Witztum, J.L.; Palumbo, G.; Palinski, W. Fatty streak formation occurs in human fetal aortas and is greatly enhanced by maternal hypercholesterolemia. Intimal accumulation of low density lipoprotein and its oxidation precede monocyte recruitment into early atherosclerotic lesions. J. Clin. Investig. 1997, 100, 2680–2690. [Google Scholar] [CrossRef]
- Napoli, C.; Glass, C.K.; Witztum, J.L.; Deutsch, R.; D’Armiento, F.P.; Palinski, W. Influence of maternal hypercholesterolaemia during pregnancy on progression of early atherosclerotic lesions in childhood: Fate of early lesions in children (FELIC) study. Lancet 1999, 354, 1234–1241. [Google Scholar] [CrossRef]
- Stojanović, S.D.; Fiedler, J.; Bauersachs, J.; Thum, T.; Sedding, D.G. Senescence-induced inflammation: An important player and key therapeutic target in atherosclerosis. Eur. Heart J. 2020, 41, 2983–2996. [Google Scholar] [CrossRef] [PubMed]
- Napoli, C.; Lerman, L.O.; de Nigris, F.; Gossl, M.; Balestrieri, M.L.; Lerman, A. Rethinking primary prevention of atherosclerosis-related diseases. Circulation 2006, 114, 2517–2527. [Google Scholar] [CrossRef] [PubMed]
- Martin-Padura, I.; de Nigris, F.; Migliaccio, E.; Mansueto, G.; Minardi, S.; Rienzo, M.; Lerman, L.O.; Stendardo, M.; Giorgio, M.; De Rosa, G.; et al. p66Shc deletion confers vascular protection in advanced atherosclerosis in hypercholesterolemic apolipoprotein E knockout mice. Endothelium 2008, 15, 276–287. [Google Scholar] [CrossRef] [PubMed]
- Gistera, A.; Hansson, G.K. The immunology of atherosclerosis. Nat. Rev. Nephrol. 2017, 13, 368–380. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Libby, P. Inflammation in atherosclerosis. Nature 2002, 420, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; D’Asti, E.; Magnus, N.; Al-Nedawi, K.; Meehan, B.; Rak, J. Microvesicles as mediators of intercellular communication in cancer--the emerging science of cellular ‘debris’. Semin. Immunopathol. 2011, 33, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Sluijter, J.P.; Verhage, V.; Deddens, J.C.; van den Akker, F.; Doevendans, P.A. Microvesicles and exosomes for intracardiac communication. Cardiovasc. Res. 2014, 102, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Kugeratski, F.G.; Santi, A.; Zanivan, S. Extracellular vesicles as central regulators of blood vessel function in cancer. Sci. Signal. 2022, 15, eaaz4742. [Google Scholar] [CrossRef]
- Mathieu, M.; Martin-Jaular, L.; Lavieu, G.; Théry, C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat. Cell Biol. 2019, 21, 9–17. [Google Scholar] [CrossRef]
- Palinski, W.; Monti, M.; Camerlingo, R.; Iacobucci, I.; Bocella, S.; Pinto, F.; Iannuzzi, C.; Mansueto, G.; Pignatiello, S.; Fazioli, F.; et al. Lysosome purinergic receptor P2X4 regulates neoangiogenesis induced by microvesicles from sarcoma patients. Cell Death Dis. 2021, 12, 797. [Google Scholar] [CrossRef]
- Benincasa, G.; Mansueto, G.; Napoli, C. Fluid-based assays and precision medicine of cardiovascular diseases: The ‘hope’ for Pandora’s box? J. Clin. Pathol. 2019, 72, 785–799. [Google Scholar] [CrossRef] [PubMed]
- Sarno, F.; Benincasa, G.; List, M.; Barabasi, A.L.; Baumbach, J.; Ciardiello, F.; Filetti, S.; Glass, K.; Loscalzo, J.; Marchese, C.; et al. Clinical epigenetics settings for cancer and cardiovascular diseases: Real-life applications of network medicine at the bedside. Clin. Epigenet. 2021, 13, 66. [Google Scholar] [CrossRef]
- Barile, L.; Vassalli, G. Exosomes: Therapy delivery tools and biomarkers of diseases. Pharmacol. Ther. 2017, 174, 63–78. [Google Scholar] [CrossRef]
- Gurunathan, S.; Kang, M.H.; Qasim, M.; Khan, K.; Kim, J.H. Biogenesis, membrane trafficking, functions, and next generation nanotherapeutics medicine of mxtracellular vesicles. Int. J. Nanomed. 2021, 16, 3357–3383. [Google Scholar] [CrossRef]
- Rai, A.; Fang, H.; Fatmous, M.; Claridge, B.; Poh, Q.H.; Simpson, R.J.; Greening, D.W. A Protocol for isolation, purification, characterization, and functional dissection of exosomes. Methods Mol. Biol. 2021, 2261, 105–149. [Google Scholar]
- Barile, L.; Cervio, E.; Lionetti, V.; Milano, G.; Ciullo, A.; Biemmi, V.; Bolis, S.; Altomare, C.; Matteucci, M.; Di Silvestre, D.; et al. Cardioprotection by cardiac progenitor cell-secreted exosomes: Role of pregnan-cy-associated plasma protein-A. Cardiovasc. Res. 2018, 114, 992–1005. [Google Scholar] [CrossRef]
- Zhang, P.; Liang, T.; Chen, Y.; Wang, X.; Wu, T.; Xie, Z.; Luo, J.; Yu, Y.; Yu, H. Circulating Exosomal miRNAs as Novel Biomarkers for Stable Coronary Artery Disease. Biomed Res. Int. 2020, 2020, 3593962. [Google Scholar] [CrossRef]
- Han, Q.F.; Li, W.J.; Hu, K.S.; Gao, J.; Zhai, W.L.; Yang, J.H.; Zhang, S.J. Exosome biogenesis: Machinery, regulation, and therapeutic implications in cancer. Mol. Cancer 2022, 21, 207. [Google Scholar] [CrossRef]
- Arcucci, V.; Stacker, S.A.; Achen, M.G. Control of Gene Expression by Exosome-Derived Non-Coding RNAs in Cancer Angiogenesis and Lymphangiogenesis. Biomolecules 2021, 11, 249. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.A.; Ludwig, R.G.; Garcia-Martin, R.; Brandão, B.B.; Kahn, C.R. Extracellular miRNAs: From biomarkers to mediators of physiology and disease. Cell Metab. 2019, 30, 656–673. [Google Scholar] [CrossRef]
- Bjørge, I.M.; Kim, S.Y.; Mano, J.F.; Kalioni, S.B.; Chrzanowski, W. Extracellular vesicles, exosomes and shedding vesicles in regenerative medicine—A new paradigm for tissue repair. Biomater. Sci. 2017, 6, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Burrello, J.; Monticone, S.; Gai, C.; Gomez, Y.; Kholia, S.; Camussi, G. Stem cell-derived extracellular vsicles and immune-Modulation. Front. Cell Dev. Biol. 2016, 4, 83. [Google Scholar] [CrossRef]
- Aguiar Koga, B.A.; Fernandes, L.A.; Fratini, P.; Sogayar, M.C.; Carreira, A.C.O. Role of MSC-derived small extracellular vesicles in tissue repair and regeneration. Front. Cell Dev. Biol. 2023, 10, 1047094. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Qin, H.; Yan, Y.; Wu, W.; Gong, S.; Wang, L.; Jiang, R.; Zhao, Q.; Sun, Y.; Wang, Q.; et al. Exosomal circular RNAs: Biogenesis, effect, and application in cardiovascular diseases. Front. Cell Dev. Biol. 2022, 10, 948256. [Google Scholar] [CrossRef]
- Polanco, J.C.; Li, C.; Durisic, N.; Sullivan, R.; Götz, J. Exosomes taken up by neurons hijack the endosomal pathway to spread to interconnected neurons. Acta Neuropathol. Commun. 2018, 6, 10. [Google Scholar] [CrossRef]
- Yang, H.; Wang, J.; Huang, G. Small extracellular vesicles in metabolic remodeling of tumor cells: Cargos and translational application. Front. Pharmacol. 2022, 13, 1009952. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, C.; D’Alessio, A.; Tamagnone, L. Message in a bottle: Endothelial cell regulation by extracellular vesicles. Cancers 2022, 14, 1969. [Google Scholar] [CrossRef] [PubMed]
- Napoli, C.; Coscioni, E.; de Nigris, F.; Donatelli, F. Emergent expansion of clinical epigenetics in patients with cardiovascular diseases. Curr. Opin. Cardiol. 2021, 36, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Kelly, T.K.; Jones, P.A. Epigenetics in cancer. Carcinogenesis 2010, 31, 27–36. [Google Scholar] [CrossRef]
- Rosa-Garrido, M.; Chapski, D.J.; Vondriska, T.M. Epigenomes in cardiovascular disease. Circ. Res. 2018, 122, 1586–1607. [Google Scholar] [CrossRef] [PubMed]
- Koene, R.J.; Prizment, A.E.; Blaes, A.; Konety, S.H. Shared risk factors in cardiovascular disease and cancer. Circulation 2016, 133, 1104–1114. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Buring, J.E.; Badimon, L.; Hansson, G.K.; Deanfield, J.; Bittencourt, M.S.; Tokgözoğlu, L.; Lewis, E.F. Atherosclerosis. Nat. Rev. Dis. Prim. 2019, 5, 56. [Google Scholar] [CrossRef]
- Peng, M.; Sun, R.; Hong, Y.; Wang, J.; Xie, Y.; Zhang, X.; Li, J.; Guo, H.; Xu, P.; Li, Y.; et al. Extracellular vesicles carrying proinflammatory factors may spread atherosclerosis to remote locations. Cell. Mol. Life Sci. 2022, 79, 430. [Google Scholar] [CrossRef]
- Costantino, S.; Libby, P.; Kishore, R.; Tardif, J.C.; El-Osta, A.; Paneni, F. Epigenetics and precision medicine in cardiovascular patients: From basic concepts to the clinical arena. Eur. Heart J. 2018, 39, 4150–4158. [Google Scholar] [CrossRef]
- Shu, Z.; Tan, J.; Miao, Y.; Zhang, Q. The role of microvesicles containing microRNAs in vascular endothelial dysfunction. J. Cell Mol. Med. 2019, 23, 7933–7945. [Google Scholar] [CrossRef]
- Oggero, S.; de Gaetano, M.; Marcone, S.; Fitzsimons, S.; Pinto, A.L.; Ikramova, D.; Barry, M.; Burke, D.; Montero-Melendez, T.; Cooper, D.; et al. Extracellular vesicles from monocyte/platelet aggregates modulate human atherosclerotic plaque reactivity. J. Extracell. Vesicles 2021, 10, 12084. [Google Scholar] [CrossRef]
- Lee, D.Y.; Yang, T.L.; Huang, Y.H.; Lee, C.I.; Chen, L.J.; Shih, Y.T.; Wei, S.Y.; Wang, W.L.; Wu, C.C.; Chiu, J.J. Induction of microRNA-10a using retinoic acid receptor-α and retinoid x receptor-α agonists inhibits atherosclerotic lesion formation. Atherosclerosis 2018, 271, 36–44. [Google Scholar] [CrossRef]
- Ismail, N.; Wang, Y.; Dakhlallah, D.; Moldovan, L.; Agarwal, K.; Batte, K.; Shah, P.; Wisler, J.; Eubank, T.D.; Tridandapani, S.; et al. Macrophage microvesicles induce macrophage differentiation and miR-223 transfer. Blood 2013, 121, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, G.; Meng, C.; Guo, X.; Cheruku, P.S.; Shi, L.; Xu, H.; Li, H.; Wang, G.; Evans, A.R.; Safe, S.; et al. A novel regulator of macrophage activation: miR-223 in obesity-associated adipose tissue inflammation. Circulation 2012, 125, 2892–2903. [Google Scholar] [CrossRef] [PubMed]
- Hall, I.F.; Climent, M.; Quintavalle, M.; Farina, F.M.; Schorn, T.; Zani, S.; Carullo, P.; Kunderfranco, P.; Civilini, E.; Condorelli, G.; et al. Circ_Lrp6, a Circular RNA Enriched in Vascular Smooth Muscle Cells, Acts as a Sponge Regulating miRNA-145 Function. Circ. Res. 2019, 124, 498–510. [Google Scholar] [CrossRef]
- Kuravi, S.J.; Harrison, P.; Rainger, G.E.; Nash, G.B. Ability of Platelet-Derived Extracellular Vesicles to Promote Neutrophil- Endothelial Cell Interactions. Inflammation 2019, 42, 290–305. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Aurora, A.B.; Johnson, B.A.; Qi, X.; McAnally, J.; Hill, J.A.; Richardson, J.A.; Bassel-Duby, R.; Olson, E.N. The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev. Cell 2008, 15, 261–271. [Google Scholar] [CrossRef]
- Eyyupkoca, F.; Ercan, K.; Kiziltunc, E.; Ugurlu, I.B.; Kocak, A.; Eyerci, N. Determination of microRNAs associated with adverse left ventricular remodeling after myocardial infarction. Mol. Cell. Biochem. 2022, 30, 55–63. [Google Scholar] [CrossRef]
- Liu, L.; Koike, H.; Ono, T.; Hayashi, S.; Kudo, F.; Kaneda, A.; Kagechika, H.; Manabe, I.; Nakashima, T.; Oishi, Y. Identification of a KLF5-dependent program and drug development for skeletal muscle atrophy. Proc. Natl. Acad. Sci. USA 2021, 118, e2102895118. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Zhang, X.; Jiang, L.; Li, Y.; Zheng, Q. Tumor endothelial cell-derived extracellular vesicles contribute to tumor microenvironment remodeling. Cell Commun. Signal. 2022, 20, 97. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Sun, B.; Gupta, A.; Rempel, H.; Pulliam, L. Monocyte exosomes induce adhesion molecules and cytokines via activation of NF-κB in endothelial cells. FASEB J. 2016, 30, 3097–3106. [Google Scholar] [CrossRef]
- Jansen, F.; Yang, X.; Proebsting, S.; Hoelscher, M.; Przybilla, D.; Baumann, K.; Schmitz, T.; Dolf, A.; Endl, E.; Franklin, B.S.; et al. MicroRNA expression in circulating microvesicles predicts cardiovascular events in patients with coronary artery disease. J. Am. Heart Assoc. 2014, 3, e001249. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.A.; Karunakaran, D.; Geoffrion, M.; Cheng, H.; Tandoc, K.; Perisic Matic, L.; Hedin, U.; Maegdefessel, L.; Fish, J.E.; Rayner, K.J. Extracellular vesicles secreted by atherogenic macrophages transfer MicroRNA to inhibit cell migration. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Wang, X.; Zhao, M.; Cai, T.; Liu, P.; Li, J.; Willard, B.; Zu, L.; Zhou, E.; Li, Y.; et al. Macrophage foam cell-derived extracellular vesicles promote vascular smooth muscle cell migration and adhesion. J. Am. Heart Assoc. 2016, 5, e004099. [Google Scholar] [CrossRef] [PubMed]
- Shan, K.; Jiang, Q.; Wang, X.Q.; Wang, Y.N.; Yang, H.; Yao, M.D.; Liu, C.; Li, X.M.; Yao, J.; Liu, B.; et al. Role of long non-coding RNA-RNCR3 in atherosclerosis-related vascular dysfunction. Cell Death Dis. 2016, 7, e2248. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Xue, C.; Auerbach, B.J.; Fan, J.; Bashore, A.C.; Cui, J.; Yang, D.Y.; Trignano, S.B.; Liu, W.; Shi, J.; et al. Single-cell genomics reveals a novel cell state during smooth muscle cell phenotypic witching and potential therapeutic targets for atherosclerosis in mouse and human. Circulation 2020, 142, 2060–2075. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, Z.; Li, L.; Yan, J.; Shao, C.; Bao, Z.; Jing, L.; Pang, Q.; Geng, Y.; Zhang, L. RAGE/galectin-3 yields intraplaque calcification transformation via sortilin. Acta Diabetol. 2019, 56, 457–472. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Y.; Luo, P.; Gao, Y.; Yang, J.; Lao, K.H.; Wang, G.; Cockerill, G.; Hu, Y.; Xu, Q.; et al. XBP1 splicing triggers miR-150 transfer from smooth muscle cells to endothelial cells via extracellular vesicles. Sci. Rep. 2016, 6, 28627. [Google Scholar] [CrossRef]
- Bian, Y.; Cai, W.; Lu, H.; Tang, S.; Yang, K.; Tan, Y. miR-150-5p affects AS plaque with ASMC proliferation and migration by STAT. Open Med. 2021, 1, 1642–1652. [Google Scholar] [CrossRef]
- Schiano, C.; Balbi, C.; Burrello, J.; Ruocco, A.; Infante, T.; Fiorito, C.; Panella, S.; Barile, L.; Mauro, C.; Vassalli, G.; et al. De novo DNA methylation induced by circulating extracellular vesicles from acute coronary syndrome patients. Atherosclerosis 2022, 354, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Zhou, E.; Zou, Y.; Mao, C.; Li, D.; Wang, C.; Zhang, Z. MicroRNA-221 inhibits the transition of endothelial progenitor cells to mesenchymal cells via the PTEN/FoxO3a signaling pathway. Adv. Clin. Exp. Med. 2021, 30, 1263–1270. [Google Scholar] [CrossRef]
- Schiattarella, G.G.; Altamirano, F.; Kim, S.Y.; Tong, D.; Ferdous, A.; Piristine, H.; Dasgupta, S.; Wang, X.; French, K.M.; Villalobos, E.; et al. Xbp1s-FoxO1 axis governs lipid accumulation and contractile performance in heart failure with preserved ejection fraction. Nat. Commun. 2021, 12, 1684. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, K.; Shi, M.; Chen, P.I.; Hennigs, J.K.; Zhao, Z.; Wang, M.; Li, C.G.; Saito, T.; Taylor, S.; Sa, S.; et al. Smooth muscle contact drives endothelial regeneration by BMPR2-Notch1-mediated metabolic and epigenetic changes. Circ. Res. 2019, 124, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Armache, A.; Yang, S.; Martínez de Paz, A.; Robbins, L.E.; Durmaz, C.; Cheong, J.Q.; Ravishankar, A.; Daman, A.W.; Ahimovic, D.J.; Klevorn, T.; et al. Histone H3.3 phosphorylation amplifies stimulation-induced transcription. Nature 2020, 583, 852–857. [Google Scholar] [CrossRef]
- Castellano-Castillo, D.; Moreno-Indias, I.; Sanchez-Alcoholado, L.; Ramos-Molina, B.; Alcaide-Torres, J.; Morcillo, S.; Ocaña-Wilhelmi, L.; Tinahones, F.; Queipo-Ortuño, M.I.; Cardona, F. Altered adipose tissue DNA methylation status in metabolic syndrome: Relationships between global DNA methylation and specific methylation at adipogenic, lipid metabolism and inflammatory candidate genes and metabolic variables. J. Clin. Med. 2019, 13, 87. [Google Scholar] [CrossRef]
- Infante, T.; Forte, E.; Schiano, C.; Punzo, B.; Cademartiri, F.; Cavaliere, C.; Salvatore, M.; Napoli, C. Evidence of association of circulating epigenetic-sensitive biomarkers with suspected coronary heart disease evaluated by Cardiac Computed Tomography. PLoS ONE 2019, 14, e0210909. [Google Scholar] [CrossRef] [PubMed]
- Infante, T.; Del Viscovo, L.; De Rimini, M.L.; Padula, S.; Caso, P.; Napoli, C. Network Medicine: A clinical approach for precision medicine and personalized therapy in coronary Heart Disease. J. Atheroscler. Thromb. 2020, 27, 279–302. [Google Scholar] [CrossRef]
- Yuan, Y.; Liu, X.; Hao, S.; He, Q.; Shen, Z. Plasma levels of miR-143 and miR-145 are associated with coronary in-stent restenosis within 1 year of follow-up after drug-eluting stent implantation. Ann. Transl. Med. 2020, 8, 756. [Google Scholar] [CrossRef]
- Shi, C.; Alvarez-Olmedo, D.; Zhang, Y.; Pattar, B.S.B.; O’Brien, E.R. The Heat shock protein 27 immune complex enhances exosomal cholesterol efflux. Biomedicines 2020, 8, 290. [Google Scholar] [CrossRef]
- Mathiesen, A.; Hamilton, T.; Carter, N.; Brown, M.; McPheat, W.; Dobrian, A. Endothelial extracellular vesicles: From keepers of health to messengers of disease. Int. J. Mol. Sci. 2021, 22, 4640. [Google Scholar] [CrossRef] [PubMed]
- Balbi, C.; Burrello, J.; Bolis, S.; Lazzarini, E.; Biemmi, V.; Pianezzi, E.; Burrello, A.; Caporali, E.; Grazioli, L.G.; Martinetti, G.; et al. Circulating extracellular vesicles are endowed with enhanced procoagulant activity in SARS-CoV-2 infection. EBioMedicine 2021, 67, 103369. [Google Scholar] [CrossRef] [PubMed]
- Balbi, C.; Bolis, S.; Vassalli, G.; Barile, L. Flow cytometric analysis of extracellular vesicles from cell-conditioned media. J. Vis. Exp. 2019, 12, 144. [Google Scholar]
- Amabile, N.; Rautou, P.E.; Tedgui, A.; Boulanger, C.M. Microparticles: Key protagonists in cardiovascular disorders. Semin. Thromb. Hemost. 2010, 36, 907–916. [Google Scholar] [CrossRef]
- Mause, S.F.; Weber, C. Microparticles: Protagonists of a novel communication network for intercellular information exchange. Circ. Res 2010, 107, 1047–1057. [Google Scholar] [CrossRef]
- Zhang, Q.; Chen, L.; Huang, L.; Cheng, H.; Wang, L.; Xu, L.; Hu, D.; He, C.; Fu, C.; Wei, Q. CD44 promotes angiogenesis in myocardial infarction through regulating plasma exosome uptake and further enhancing FGFR2 signaling transduction. Mol. Med. 2022, 328, 145–147. [Google Scholar] [CrossRef]
- Wang, T.; Li, T.; Niu, X.; Hu, L.; Cheng, J.; Guo, D.; Ren, H.; Zhao, R.; Ji, Z.; Liu, P.; et al. ADSC-derived exosomes attenuate myocardial infarction injury by promoting miR-205-mediated cardiac angiogenesis. Biol. Direct. 2023, 18, 6. [Google Scholar] [CrossRef] [PubMed]
- Xuan, Y.; Chen, C.; Wen, Z.; Wang, D.W. The roles of cardiac fibroblasts and endothelial cells in myocarditis. Front. Cardiovasc. Med. 2022, 9, 882027. [Google Scholar] [CrossRef]
- Su, X.L.; Wang, S.H.; Komal, S.; Cui, L.G.; Ni, R.C.; Zhang, L.R.; Han, S.N. The caspase-1 inhibitor VX765 upregulates connexin 43 expression and improves cell-cell communication after myocardial infarction via suppressing the IL-1β/p38 MAPK pathway. Acta Pharmacol. Sin. 2022, 43, 2289–2301. [Google Scholar] [CrossRef]
- Bang, C.; Batkai, S.; Dangwal, S.; Gupta, S.K.; Foinquinos, A.; Holzmann, A.; Just, A.; Remke, J.; Zimmer, K.; Zeug, A.; et al. Cardiac fibroblast-derived microRNA passenger strand-enriched exosomes mediate cardiomyocyte hypertrophy. J. Clin. Investig. 2014, 124, 2136–2146. [Google Scholar] [CrossRef]
- Di, J.; Yang, M.; Zhou, H.; Li, M.; Zhao, J. MicroRNA-21-containing microvesicles from tubular epithelial cells promote cardiomyocyte hypertrophy. Ren. Fail. 2021, 1, 391–400. [Google Scholar] [CrossRef]
- Li, Q.; Yao, Y.; Shi, S.; Zhou, M.; Zhou, Y.; Wang, M.; Chiu, J.J.; Huang, Z.; Zhang, W.; Liu, M.; et al. Inhibition of miR-21 alleviated cardiac perivascular fibrosis via repressing EndMT in T1DM. J. Cell. Mol. Med. 2020, 1, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Fleissner, F.; Jazbutyte, V.; Fiedler, J.; Gupta, S.K.; Yin, X.; Xu, Q.; Galuppo, P.; Kneitz, S.; Mayr, M.; Ertl, G.; et al. Short communication: Asymmetric dimethylarginine impairs angiogenic progenitor cell function in patients with coronary artery disease through a microRNA-21-dependent mechanism. Circ. Res. 2010, 107, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.C.; Yamada, K.A.; Patel, A.Y.; Topkara, V.K.; George, I.; Cheema, F.H.; Ewald, G.A.; Mann, D.L.; Nerbonne, J.M. Deep RNA sequencing reveals dynamic regulation of myocardial noncoding RNAs in failing human heart and remodelling with mechanical circulatory support. Circulation 2014, 129, 1009–1021. [Google Scholar] [CrossRef]
- Yang, H.; Chen, J. Bone marrow mesenchymal stem cell-derived exosomes carrying long noncoding RNA ZFAS1 alleviate oxidative stress and inflammation in ischemic stroke by inhibiting microRNA-15a-5p. Metab. Brain Dis. 2022, 37, 2545–2557. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, X.; Li, H.; Li, Y.; Cheng, D.; Tang, Y.; Sang, H. Serum extracellular vesicles containing MIAT induces atrial fibrosis, inflammation and oxidative stress to promote atrial remodeling and atrial fibrillation via blockade of miR-485-5p-mediated CXCL10 inhibition. Clin. Transl. Med. 2021, 11, e482. [Google Scholar] [CrossRef]
- Kenneweg, F.; Bang, C.; Xiao, K.; Boulanger, C.M.; Loyer, X.; Mazlan, S.; Schroen, B.; Hermans-Beijnsberger, S.; Foinquinos, A.; Hirt, M.N.; et al. Long noncoding RNA-enriched vesicles secreted by hypoxic cardiomyocytes drive cardiac fibrosis. Mol. Ther. Nucleic Acids 2019, 18, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Zeyidan, J.; Chengyu, L.; Xinyu, Y.; Lingfei, S.; Emeli, C.; Lingying, Z.; Ji, L.; Guoping, L. Extracellular non-coding RNAs in cardiovascular diseases. Pharmaceutics 2023, 15, 155. [Google Scholar]
- Shao, C.; Yang, F.; Miao, S.; Liu, W.; Wang, C.; Shu, Y.; Shen, H. Role of hypoxia-induced exosomes in tumor biology. Mol. Cancer 2018, 17, 120. [Google Scholar] [CrossRef]
- Adnani, L.; Spinelli, C.; Tawil, N.; Rak, J. Role of extracellular vesicles in cancer-specific interactions between tumour cells and the vasculature. Semin. Cancer Biol. 2022, 87, 196–213. [Google Scholar] [CrossRef]
- Feng, Q.; Zhang, C.; Lum, D.; Druso, J.E.; Blank, B.; Wilson, K.F.; Welm, A.; Antonyak, M.A.; Cerione, R.A. A class of extracellular vesicles from breast cancer cells activates VEGF receptors and tumour angiogenesis. Nat. Commun. 2017, 8, 14450. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Li, J.; Liu, S.; Wang, T.; Ianni, A.; Bober, E.; Braun, T.; Xiang, R.; Yue, S. Exosomal tetraspanins mediate cancer metastasis by altering host microenvironment. Oncotarget 2017, 8, 62803–62815. [Google Scholar] [CrossRef]
- Brzozowa-Zasada, M. The role of Notch ligand, Delta-like ligand 4 (DLL4), in cancer angiogenesis—Implications for therapy. Eur. Surg. 2021, 53, 274–280. [Google Scholar] [CrossRef]
- Sheldon, H.; Heikamp, E.; Turley, H.; Dragovic, R.; Thomas, P.; Oon, C.E.; Leek, R.; Edelmann, M.; Kessler, B.; Sainson, R.C.; et al. New mechanism for Notch signaling to endothelium at a distance by Delta-like 4 incorporation into exosomes. Blood 2010, 116, 2385–2394. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Tomuleasa, C.; Monroig, P.; Cucuianu, A.; Berindan-Neagoe, I.; Calin, G.A. Exosomes as divine messengers: Are they the Hermes of modern molecular oncology? Cell Death Differ. 2015, 22, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lu, J.; Chen, L.; Bian, H.; Hu, J.; Li, D.; Xia, C.; Xu, H. Tumor-Derived EV-Encapsulated miR-181b-5p induces angiogenesis to foster tumorigenesis and metastasis of ESCC. Mol. Ther. Nucleic Acids 2020, 20, 421–437. [Google Scholar] [CrossRef]
- Tadokoro, H.; Umezu, T.; Ohyashiki, K.; Hirano, T.; Ohyashiki, J.H. Exosomes derived from hypoxic leukemia cells enhance tube formation in endothelial cells. J. Biol. Chem. 2013, 288, 34343–34351. [Google Scholar] [CrossRef]
- Chen, X.; Yang, F.; Zhang, T.; Wang, W.; Xi, W.; Li, Y.; Zhang, D.; Huo, Y.; Zhang, J.; Yang, A.; et al. MiR-9 promotes tumorigenesis and angiogenesis and is activated by MYC and OCT4 in human glioma. J. Exp. Clin. Cancer Res. 2019, 38, 99. [Google Scholar] [CrossRef]
- Kosaka, N.; Iguchi, H.; Hagiwara, K.; Yoshioka, Y.; Takeshita, F.; Ochiya, T. Neutral sphingomyelinase 2 (nSMase2)-dependent exosomal transfer of angiogenic microRNAs regulate cancer cell metastasis. J. Biol. Chem. 2013, 288, 10849–10859. [Google Scholar] [CrossRef]
- Camps, C.; Buffa, F.M.; Colella, S.; Moore, J.; Sotiriou, C.; Sheldon, H.; Harris, A.L.; Gleadle, J.M.; Ragoussis, J. hsa-miR-210 Is induced by hypoxia and is an independent prognostic factor in breast cancer. Clin. Cancer Res. 2008, 14, 1340–1348. [Google Scholar] [CrossRef]
- Li, B.; Hong, J.; Hong, M.; Wang, Y.; Yu, T.; Zang, S.; Wu, Q. piRNA-823 delivered by multiple myeloma-derived extracellular vesicles promoted tumorigenesis through re-educating endothelial cells in the tumor environment. Oncogene 2019, 38, 5227–5238. [Google Scholar] [CrossRef]
- Gao, H.; Wang, X.; Ma, H.; Lin, S.; Zhang, D.; Wu, W.; Liao, Z.; Chen, M.; Ye, H.; Li, Q.; et al. LncRNA CCAT2, involving miR-34a/TGF-β1/Smad4 signaling, regulate hepatic stellate cells proliferation. Sci. Rep. 2022, 12, 21199. [Google Scholar] [CrossRef] [PubMed]
- de Nigris, F.; Mancini, F.P.; Schiano, C.; Infante, T.; Zullo, A.; Minucci, P.B.; Al-Omran, M.; Giordano, A.; Napoli, C. Osteosarcoma cells induce endothelial cell proliferation during neo-angiogenesis. J. Cell. Physiol. 2013, 228, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, Q.; Yang, D.; Xie, F.; Wang, Z. The role of long non-coding RNAs in angiogenesis and anti-angiogenic therapy resistance in cancer. Mol. Ther. Nucleic Acids 2022, 28, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Du, P.; Cui, P.; Qin, Y.; Hu, C.; Wu, J.; Zhou, Z.; Zhang, W.; Qin, L.; Huang, G. LncRNA PVT1 promotes angiogenesis via activating the STAT3/VEGFA axis in gastric cancer. Oncogene 2018, 37, 4094–4109. [Google Scholar] [CrossRef] [PubMed]
- Castellano, J.J.; Marrades, R.M.; Molins, L.; Viñolas, N.; Moises, J.; Canals, J.; Han, B.; Li, Y.; Martinez, D.; Monzó, M.; et al. Extracellular vesicle lincRNA-p21 expression in tumor-draining pulmonary vein defines prognosis in NSCLC and modulates endothelial cell behavior. Cancers 2020, 12, 734. [Google Scholar] [CrossRef]
- Zang, X.; Gu, J.; Zhang, J.; Shi, H.; Hou, S.; Xu, X.; Yan, C.; Yu, Z.; Fei, M.; Hui, Q.; et al. Exosome-transmitted lncRNA UFC1 promotes non-small-cell lung cancer progression by EZH2-mediated epigenetic silencing of PTEN expression. Cell Death Dis. 2020, 11, 215. [Google Scholar] [CrossRef]
- Hou, Y.; Jia, H.; Cao, Y.; Zhang, S.; Zhang, X.; Wei, P.; Jun, X.; Wen, D.; Bao, W. LncRNA EPIC1 promotes tumor angiogenesis via activating the Ang2/Tie2 axis in non-small cell lung cancer. Life Sci. 2021, 267, 118933. [Google Scholar] [CrossRef]
- Leisegang, M.S.; Fork, C.; Josipovic, I.; Richter, F.M.; Preussner, J.; Hu, J.; Miller, M.J.; Epah, J.; Hofmann, P.; Günther, S.; et al. Long Noncoding RNA MANTIS facilitates endothelial angiogenic function. Circulation 2017, 136, 65–79. [Google Scholar] [CrossRef]
- Qiu, J.J.; Lin, X.J.; Tang, X.Y.; Zheng, T.T.; Lin, Y.Y.; Hua, K.Q. Sci Exosomal metastasis-associated lung adenocarcinoma transcript 1 promotes angiogenesis and predicts poor prognosis in epithelial ovarian cancer. Int. J. Biol. 2018, 14, 1960–1973. [Google Scholar] [CrossRef]
- Poulet, C.; Njock, M.S.; Moermans, C.; Louis, E.; Louis, R.; Malaise, M.; Guiot. Exosomal long non-coding RNAs in lung diseases. Int. J. Mol. Sci. 2020, 21, 3580. [Google Scholar] [CrossRef]
- Cheng, C.; Zhang, Z.; Cheng, F.; Shao, Z. Exosomal NA RAMP2-AS1 derived from chondrosarcoma cells promotes angiogenesis through miR-2355-5p/VEGFR2 Axis. OncoTargets Ther. 2020, 13, 3291–3301. [Google Scholar] [CrossRef]
- Guo, Z.; Wang, X.; Yang, Y.; Chen, W.; Zhang, K.; Teng, B.; Huang, C.; Zhao, Q.; Qiu, Z. Hypoxic tumor-derived exosomal long noncoding RNA UCA1 promotes angiogenesis via miR-96-5p/AMOTL2 in pancreatic cancer. Mol. Ther. Nucleic Acids 2020, 22, 179–195. [Google Scholar] [CrossRef] [PubMed]
- Dai, G.; Yang, Y.; Liu, S.; Liu, H. Hypoxic breast cancer cell-derived exosomal SNHG1 promotes breast cancer growth and angiogenesis via regulating miR-216b-5p/JAK2 Axis. Cancer Manag. Res. 2022, 14, 123–133. [Google Scholar] [CrossRef]
- Lang, H.L.; Hu, G.W.; Zhang, B.; Kuang, W.; Chen, Y.; Wu, L.; Xu, G.H. Glioma cells enhance angiogenesis and inhibit endothelial cell apoptosis through the release of exosomes that contain long non-coding RNA CCAT2. Oncol. Rep. 2017, 38, 785–798. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.L.; Shu, Y.G.; Tao, M.Y. LncRNA CCAT2 promotes angiogenesis in glioma through activation of VEGFA signalling by sponging miR-424. Mol. Cell. Biochem. 2020, 468, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Li, J.; Fan, F.; Zhou, P. LINC00707 regulates miR-382-5p/VEGFA pathway to enhance cervical cancer progression. J. Immunol. Res. 2021, 29, 5524632. [Google Scholar] [CrossRef]
- Gao, T.; Ji, Y. Long Noncoding RNA LINC00707 accelerates tumorigenesis and progression of bladder cancer via targeting miR-145/CDCA3 regulatory loop. Urol. Int. 2021, 105, 891–905. [Google Scholar] [CrossRef] [PubMed]
- Schillaci, O.; Fontana, S.; Monteleone, F.; Taverna, S.; Di Bella, M.A.; Di Vizio, D.; Alessandro, R. Exosomes from metastatic cancer cells transfer amoeboid phenotype to non-metastatic cells and increase endothelial permeability: Their emerging role in tumor heterogeneity. Sci. Rep. 2017, 7, 4711. [Google Scholar] [CrossRef]
- Holzner, S.; Senfter, D.; Stadler, S.; Staribacher, A.; Nguyen, C.H.; Gaggl, A.; Geleff, S.; Huttary, N.; Krieger, S.; Jager, W.; et al. Colorectal cancer cell-derived microRNA200 modulates the resistance of adjacent blood endothelial barriers in vitro. Oncol. Rep. 2016, 36, 3065–3071. [Google Scholar] [CrossRef]
- Zeng, Z.; Li, Y.; Pan, Y.; Lan, X.; Song, F.; Sun, J.; Zhou, K.; Liu, X.; Ren, X.; Wang, F.; et al. Cancer-derived exosomal miR-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat. Commun. 2018, 9, 5395. [Google Scholar] [CrossRef]
- Zhou, W.; Fong, M.Y.; Min, Y.; Somlo, G.; Liu, L.; Palomares, M.R.; Yu, Y.; Chow, A.; O’Connor, S.T.; Chin, A.R.; et al. Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer Cell 2014, 25, 501–515. [Google Scholar] [CrossRef] [PubMed]
- Di Modica, M.; Regondi, V.; Sandri, M.; Iorio, M.V.; Zanetti, A.; Tagliabue, E.; Casalini, P.; Triulzi, T. Breast cancer-secreted miR-939 downregulates VE-cadherin and destroys the barrier function of endothelial monolayers. Cancer Lett. 2017, 384, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, S.; Du, K.; Zheng, N.; Liu, Y.; Chen, H.; Xie, G.; Ma, Y.; Zhou, Y.; Zheng, Y.; et al. Gastric cancer–secreted exosomal X26nt increases angiogenesis and vascular permeability by targeting VE-cadherin. Cancer Sci. 2020, 112, 1839–1852. [Google Scholar] [CrossRef]
- Dou, R.; Liu, K.; Yang, C.; Zheng, J.; Shi, D.; Lin, X.; Wei, C.; Zhang, C.; Fang, Y.; Huang, S.; et al. EMT-cancer cells-derived exosomal miR-27b-3p promotes circulating tumour cells-mediated metastasis by modulating vascular permeability in colorectal cancer. Clin. Transl. Med. 2021, 11, e595. [Google Scholar] [CrossRef] [PubMed]
- Yokota, Y.; Noda, T.; Okumura, Y.; Kobayashi, S.; Iwagami, Y.; Yamada, D.; Tomimaru, Y.; Akita, H.; Gotoh, K.; Takeda, Y.; et al. Serum exosomal miR-638 is a prognostic marker of HCC via downregulation of VE-cadherin and ZO-1 of endothelial cells. Cancer Sci. 2021, 112, 1275–1288. [Google Scholar] [CrossRef]
- Shuangshuang, M.; Sufei, Z.; Zhiliang, L.; Xinfeng, W.; Yan, W.; Guochao, Z.; Haiyan, X.; Jianbing, H.; Yuanyuan, L.; Chengming, L.; et al. Exosomal miR-375-3p breaks vascular barrier and promotes small cell lung cancer metastasis by targeting claudin-1. Transl. Lung Cancer 2021, 10, 3155–3172. [Google Scholar]
- Yokoi, A.; Yoshioka, Y.; Yamamoto, Y.; Ishikawa, M.; Ikeda, S.I.; Kato, T.; Kiyono, T.; Takeshita, F.; Kajiyama, H.; Kikkawa, F.; et al. Malignant extracellular vesicles carrying MMP1 mRNA facilitate peritoneal dissemination in ovarian cancer. Nat. Commun. 2017, 8, 14470. [Google Scholar] [CrossRef] [PubMed]
- Bobrie, A.; Krumeich, S.; Reyal, F.; Recchi, C.; Moita, L.F.; Seabra, M.C.; Ostrowski, M.; Thery, C. Rab27a supports exosome-dependent and -independent mechanisms that modify the tumor microenvironment and can promote tumor progression. Cancer Res. 2012, 72, 4920–4930. [Google Scholar] [CrossRef]
- Dongming, W.; Shihua, D.; Li, L.; Teng, L.; Ting, Z.; Jing, L.; Ye, Y.; Ying, X. TGF-β1-mediated exosomal lnc-MMP2-2 increases blood-brain barrier permeability via the miRNA-1207-5p/EPB41L5 axis to promote non-small cell lung cancer brain metastasis. Cell Death 2021, 20, 721. [Google Scholar]
- Gasparics, A.; Rosivall, L.; Krizbai, I.A.; Sebe, A. When the endothelium scores an own goal: Endothelial cells actively augment metastatic extravasation through endothelial-mesenchymal transition. Am. J. Physiol.-Heart Circ. Physiol. 2016, 310, H1055–H1063. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.G.; Lee, A.; Chang, W.; Lee, M.S.; Kim, J. Endothelial to mesenchymal transition represents a key link in the interaction between inflammation and endothelial dysfunction. Front. Immunol. 2018, 9, 294. [Google Scholar] [CrossRef] [PubMed]
- Akino, T.; Hida, K.; Hida, Y.; Tsuchiya, K.; Freedman, D.; Muraki, C.; Ohga, N.; Matsuda, K.; Akiyama, K.; Harabayashi, T.; et al. Cytogenetic abnormalities of tumor-associated endothelial cells in human malignant tumors. Am. J. Pathol. 2009, 175, 2657–2667. [Google Scholar] [CrossRef]
- Ohga, N.; Ishikawa, S.; Maishi, N.; Akiyama, K.; Hida, Y.; Kawamoto, T.; Sadamoto, Y.; Osawa, T.; Yamamoto, K.; Kondoh, M.; et al. Heterogeneity of tumor endothelial cells: Comparison between tumor endothelial cells isolated from high- and low-metastatic tumors. Am. J. Pathol. 2012, 180, 1294–1307. [Google Scholar] [CrossRef]
- Choi, S.H.; Kim, A.R.; Nam, J.K.; Kim, J.M.; Kim, J.Y.; Seo, H.R.; Lee, H.J.; Cho, J.; Lee, Y.J. Tumour vasculature development via endothelial-to-mesenchymal transition after radiotherapy controls CD44v6+cancer cell and macrophage polarization. Nat. Commun. 2018, 9, 5108. [Google Scholar] [CrossRef] [PubMed]
- Nie, L.; Lyros, O.; Medda, R.; Jovanovic, N.; Schmidt, J.L.; Otterson, M.F.; Johnson, C.P.; Behmaram, B.; Shaker, R.; Rafiee, P. Endothelial-mesenchymal transition in normal human esophageal endothelial cells cocultured with esophageal adenocarcinoma cells: Role of IL-1beta and TGF-beta. Am. J. Physiol.-Cell Physiol. 2014, 307, C859–C877. [Google Scholar] [CrossRef]
- Beerepoot, L.V.; Mehra, N.; Vermaat, J.S.; Zonnenberg, B.A.; Gebbink, M.F.; Voest, E.E. Increased levels of viable circulating endothelial cells are an indicator of progressive disease in cancer patients. Ann. Oncol. 2004, 15, 139–145. [Google Scholar] [CrossRef]
- Gopal, S.K.; Greening, D.W.; Hanssen, E.G.; Zhu, H.J.; Simpson, R.J.; Mathias, R.A. Oncogenic epithelial cell-derived exosomes containing Rac1 and PAK2 induce angiogenesis in recipient endothelial cells. Oncotarget 2016, 7, 19709–19722. [Google Scholar] [CrossRef]
- Dai, J.; Su, Y.; Zhong, S.; Cong, L.; Liu, B.; Yang, J.; Tao, Y.; He, Z.; Chen, C.; Jiang, Y. Exosomes: Key players in cancer and potential therapeutic strategy. Signal Transduct. Target. Ther. 2020, 5, 145. [Google Scholar] [CrossRef]
- Yeon, J.H.; Jeong, H.E.; Seo, H.; Cho, S.; Kim, K.; Na, D.; Chung, S.; Park, J.; Choi, N.; Kang, J.Y. Cancer-derived exosomes trigger endothelial to mesenchymal transition followed by the induction of cancer-associated fibroblasts. Acta Biomater. 2018, 76, 146–153. [Google Scholar] [CrossRef]
- Fang, J.H.; Zhang, Z.J.; Shang, L.R.; Luo, Y.W.; Lin, Y.F.; Yuan, Y.; Zhuang, S.M. Hepatoma cell-secreted exosomal microRNA-103 increases vascular permeability and promotes metastasis by targeting junction proteins. Hepatology 2018, 68, 1459–1475. [Google Scholar] [CrossRef] [PubMed]
- de Jong, O.G.; van Balkom, B.W.; Gremmels, H.; Verhaar, M.C. Exosomes from hypoxic endothelial cells have increased collagen crosslinking activity through up-regulation of lysyl oxidase-like. J. Cell. Mol. Med. 2016, 20, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Cuypers, A.; Khanh Truong, A.C.; Becker, L.M.; Saavedra-García, P.; Carmeliet, P. Tumor vessel co-option: The past & the future. Front. Oncol. 2022, 12, 965277. [Google Scholar]
- Neumann, P.; Jaé, N.; Knau, A.; Glaser, S.F.; Fouani, Y.; Rossbach, O.; Krüger, M.; John, D.; Bindereif, A.; Grote, P.; et al. The lncRNA GATA6-AS epigenetically regulates endothelial gene expression via interaction with LOXL. Nat. Commun. 2018, 9, 237. [Google Scholar] [CrossRef] [PubMed]
- Peinado, H.; Alečković, M.; Lavotshkin, S.; Matei, I.; Costa-Silva, B.; Moreno-Bueno, G.; Hergueta-Redondo, M.; Williams, C.; García-Santos, G.; Ghajar, C. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 2012, 18, 883–891. [Google Scholar] [CrossRef]
- Zeng, Y.; Yao, X.; Liu, X.; He, X.; Li, L.; Liu, X.; Yan, Z.; Wu, J.; Fu, B.M. Antiangiogenesis triggers exosomes release from endothelial cells to promote tumor vasculogenesis. J. Extracell. Vesicles 2019, 8, 1629865. [Google Scholar] [CrossRef]
- Maniotis, A.J.; Folberg, R.; Hess, A.; Seftor, E.A.; Gardner, L.M.; Pe’er, J.; Trent, J.M.; Meltzer, P.S.; Hendrix, M.J. Vascular channel formation by human melanoma cells in vivo and in vitro: Vasculogenic mimicry. Am. J. Pathol. 1999, 155, 739–752. [Google Scholar] [CrossRef]
- Yang, J.P.; Liao, Y.D.; Mai, D.M.; Xie, P.; Qiang, Y.Y.; Zheng, L.S.; Wang, M.Y.; Mei, Y.; Meng, D.F.; Xu, L.; et al. Tumor vasculogenic mimicry predicts poor prognosis in cancer patients: A meta-analysis. Angiogenesis 2016, 19, 191–200. [Google Scholar] [CrossRef]
- Xiang, T.; Lin, Y.X.; Ma, W.; Zhang, H.J.; Chen, K.M.; He, G.P.; Zhang, X.; Xu, M.; Feng, Q.S.; Chen, M.Y.; et al. Vasculogenic mimicry formation in EBV-associated epithelial malignancies. Nat. Commun. 2018, 9, 5009. [Google Scholar] [CrossRef]
- Qin, Y.; Zhao, W.; Cai, Z.; Wang, Q.; Gao, J.; Ci, H.; Feng, Z.; Ma, L. The Biomarker like the correlation between vasculogenic mimicry, vascular endothelial cadherin, sex-determining region on Y-Box transcription factor 17, and Cyclin D1 in oesophageal squamous cell carcinoma. J. Oncol. 2022, 29, 8915503. [Google Scholar]
- Luo, Q.; Wang, J.; Zhao, W.; Peng, Z.; Liu, X.; Li, B.; Zhang, H.; Shan, B.; Zhang, C.; Duan, C. Vasculogenic mimicry in carcinogenesis and clinical applications. J. Hematol. Oncol. 2020, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- Tiantian, Y.; Jing, W.; Yuchen, H.; Min, Z.; Jie, H. Long non-coding RNA HULC stimulates the epithelial–mesenchymal transition process and vasculogenic mimicry in human glioblastoma. Cancer Med. 2021, 10, 5270–5282. [Google Scholar]
- Li, Y.; Wu, Z.; Yuan, J.; Sun, L.; Lin, L.; Huang, N.; Bin, J.; Liao, Y.; Liao, W. Long non-coding RNA MALAT1 promotes gastric cancer tumorigenicity and metastasis by regulating vasculogenic mimicry and angiogenesis. Cancer Lett. 2017, 1, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Shen, Y.; Ren, H.; Wang, L.; Yang, J.; Wang, Y. Repression of linc01555 up-regulates angiomotin-p130 via the microRNA-122-5p/clic1 axis to impact vasculogenic mimicry-mediated chemotherapy resistance in small cell lung cancer. Cell Cycle 2023, 22, 255–268. [Google Scholar] [CrossRef]
- Cai, Y.; Wu, J.; Li, Z.; Long, Q. Mathematical modelling of a brain tumour initiation and early development: A coupled model of glioblastoma growth, pre-existing vessel co-option, angiogenesis and blood perfusion. PLoS ONE 2016, 11, e0150296. [Google Scholar] [CrossRef] [PubMed]
- Krusche, B.; Ottone, C.; Clements, M.P.; Johnstone, E.R.; Goetsch, K.; Lieven, H.; Mota, S.G.; Singh, P.; Khadayate, S.; Ashraf, A.; et al. EphrinB2 drives perivascular invasion and proliferation of glioblastoma stem-like cells. eLife 2016, 5, e14845. [Google Scholar] [CrossRef] [PubMed]
| Number | NCT Number | Status | Conditions | Study Type | Population (Participants Number) | Outcome |
|---|---|---|---|---|---|---|
| 1 | NCT03660683 | Terminated | Diabetes Mellitus, Type 2 Cardiovascular Diseases | Interventional (Dapagliflozin 10 mg) | 15 | Evaluation of exosomes released from kidney podocyte after treatment, as indicator of kidney podocyte health, via urine assay |
| 2 | NCT04142138 | Completed | Prehypertension | Interventional (DASH) | 9 | Characterization of urine exosome protein abundance pattern during nutritional changes, shifting from a “westernized diet” to a DASH diet |
| 3 | NCT03478410 | Recruiting | AF | Interventional (Epicardial fat biopsy) | 35 | Quantification of epicardial fat-derived exosomes in patients with and without AF |
| 4 | NCT04356300 | Not yet recruiting | MODS after surgical repaired for ATAAD | Interventional (MSC-exosome administration intravenously to patients once a day for two weeks) | 60 | Intravenously treatment with MSC-derived exosomes immediately after ascending aortic replacement combined with open placement of triple branched stent graft |
| 5 | NCT05669144 | Recruiting | MI Myocardial Ischemia Myocardial Stunning | Interventional (Intracoronary and intramyocardial injection of exosomes) | 20 | Mitochondria and MSC-derived exosomes evaluation |
| 6 | NCT03034265 | Completed | Hypertension | Observational | 24 | Quantification of urinary exosomal sodium channels |
| 7 | NCT03384433 | Unknown status | Cerebrovascular Disorders | Interventional (Intravenous administration of allogenic MSC-generated exosomes transfected by miR-124 post-stroke) | 5 | Intravenous administration of allogenic MSC-generated exosomes transfected by miR-124 post-stroke Evaluation of disability improvement of patients with acute ischemic stroke after exosome administration |
| 8 | NCT03984006 | Completed | Autoimmune thyroid disease Heart Failure | Observational | 5 | Urinary exosomal NT-proBNP detection to find earlier predicting biomarkers for heart dysfunction |
| 9 | NCT02822131 | Completed | Hypertension | Interventional (High phosphate diet) | 10 | Urine exosome evaluation after high phosphate intake |
| 10 | NCT05490173 | Not yet recruiting | Premature Birth Extreme Prematurity Preterm Intraventricular Hemorrhage Hypoxia-Ischemia, Cerebral Neurodevelopmental Disorders | Interventional (MSC-derived exosomes intranasal administration) | 10 | EVs effect investigation through analysis of perinatal brain injury biomarkers (S-100, NSE, EPO) and mRNA |
| 11 | NCT05035134 | Recruiting | Intracerebral Hemorrhage Circulating Exosomes | Observational | 300 | Circulating exosomes evaluation by RNA and proteome sequencing |
| 12 | NCT04127591 | Unknown status | MI | Observational | 10 | Peripheral plasma exosome miRNA evaluation |
| 13 | NCT05326724 | Recruiting | Post-stroke Dementia Acupuncture | Interventional (Acupuncture) | 30 | Quantification of acupuncture-induced exosomes |
| 14 | NCT00331331 | Completed | Uveitis Vasculitis Ocular Inflammatory Disease | Observational | 147 | Vitreus exosome evaluation |
| 15 | NCT05243368 | Not yet recruiting | CCU | Interventional (MSC-derived exosome treatment) | 30 | Evaluation of microenvironment favoring tissue regeneration after MSC-derived exosome treatment |
| 16 | NCT03837470 | Completed | HFpEF | Interventional (Sodium Chloride; Furosemide administration) | 14 | Urine exosome evaluation after treatment |
| 17 | NCT05370105 | Recruiting | Stroke Rehabilitation | Observational | 100 | Characterization of blood EVs |
| 18 | NCT03264976 | Not yet recruiting | DR | Observational | 200 | miRNA sequencing of serum exosomes |
| 19 | NCT04334603 | Recruiting | HF | Interventional (Exercise training program) | 120 | Plasma exosomes concentration evaluation |
| 20 | NCT03227055 | Unknown status | Childhood Chronic Kidney Disease | Observational | 155 | Urine exosome miRNAs evaluation |
| 21 | NCT04184076 | Unknown status | Acute ischemic stroke | Interventional (Time-restricted feeding) | 40 | Characterization of plasma exosome markers (phospho-IRS1, phospho-Tau, Abeta1–42, phospho-IRS1) |
| 22 | NCT04641585 | Not yet recruiting | Brugada Syndrome 1 | Interventional (Brugada Syndrome diagnosti test) | 144 | Evaluation of exosome coding and noncoding RNAs |
| 23 | NCT05155358 | Recruiting | Heat Stroke Early Waking MODS Proteinosis Liver Injury Kidney Injury | Observational | 150 | Plasma exosome miRNAs evaluation |
| 24 | NCT04235023 | Unknown status | Sleep Apnea Inflammation Atherosclerosis | Interventional (apnea hypopnea index ≥ 15 per hour) | 60 | Plasma exosome evaluation |
| 25 | NCT03275363 | Unknown status | Neurocognitive Disorder Mild Cognitive Impairment Alzheimer Dementia Vascular Dementia Age-related Cognitive Decline | Observational | 500 | Blood microvesicles and exosome analysis |
| 26 | NCT04266639 | Completed | Stroke, Acute Ischemic Stroke Cerebrovascular Disorders Central Nervous System Diseases | Interventional (Remote ischemic conditioning) | 45 | Characterization of EVs surface markers and content after treatment |
| 27 | NCT05624203 | Not yet recruiting | Myocardial Reperfusion Injury Treatment Outcome Prognosis ST Elevation Myocardial Infarction | Interventional (Exercise training) | 100 | Blood microvesicles analysis |
| 28 | NCT04250493 | Recruiting | Multiple System Atrophy | Interventional (HOMA) | 124 | Evaluation of neural-derived plasma exosomes after HOMA |
| Number | NCT Number | Status | Conditions | Study Type | Population (Participants Number) | Outcome |
|---|---|---|---|---|---|---|
| 1 | NCT02702856 | Completed | Prostate Cancer | Observational Multi center | 2000 | Validation of the association between exosome urine gene signature with high Gleason grade/score (GS ≥ 7) |
| 2 | NCT04529915 | Active, not recruiting | Lung Cancer | Observational Multi center | 470 | Analysis of blood exosomes signature as an early marker of lung cancer |
| 3 | NCT04394572 | Completed | Colorectal Cancer | Observational | 80 | Quantification of serum exosomes using specific exosomes markers as CD63 and CD81 |
| 4 | NCT04939324 | Recruiting | Lung Cancer NSCL Cancer | Interventional exosome will be collected from tumor drain vein during surgery | 30 | Analysis of molecular profiling of exosome with a sample in tumor-draining vein in order to identify prognostic molecular characteristics associated with cancer recurrence after surgery |
| 5 | NCT02393703 | Recruiting | Pancreatic Cancer Benign Pancreatic Disease | Observational diagnostic | 111 | Differential gene expression in exosomes from cancer and healthy subjects to connect disease recurrence and outcomes with peculiar gene expression |
| 6 | NCT01294072 | Recruiting | Colon Cancer | Interventional Curcumin delivery | 35 | Curcumin delivery by using plant exosomes to target the drug to colon tumors and normal colon tissue |
| 7 | NCT01159288 | Completed | NSCL Cancer | Interventional cyclophosphamide (mCTX) followed by vaccinations with tumor antigen-loaded dendritic cell-derived exosomes (Dex). | 41 | Evaluation of tumor antigen-loaded in dendritic cell-derived exosomes |
| 8 | NCT01668849 | Completed | Head and Neck Cancer Oral Mucositis | Interventional Chemoradiation Lortab, Fentanyl patch. | 60 | Investigate the ability of plant (grape) exosomes to prevent oral mucositis associated with chemoradiation |
| 9 | NCT04288141 | Recruiting | HER2-positive Breast Cancer | Observational diagnostic | 40 | Evaluation of peripheral blood exosomes containing HER2 and HER3 dimers to predict tumor positivity |
| 10 | NCT05587114 | Recruiting | Lung Cancer | Observational diagnostic | 150 | Exosome markers derived from peripheral blood and pulmonary venous blood from patients who underwent lung cancer surgery as early tumor markers |
| 11 | NCT03608631 | Recruiting | Metastatic Pancreatic Adenocarcinoma Pancreatic Ductal Adenocarcinoma | Interventional Drug: Mesenchymal Stromal Cells-derived Exosomes with KRAS G12D siRNA | 28 | Evaluation of Exosomes with KRAS G12D siRNA in blood |
| 12 | NCT04653740 | Recruiting | Advanced Breast Cancer | Interventional changes in the profile of tumor, associated with resistance to palbociclib at the individual level | 25 | Exosome characterization associated with palbociclib resistance |
| 13 | NCT03830619 | Completed | Lung Cancer | Observational diagnostic | 1000 | Serum exosomal lncRNAs characterization |
| 14 | NCT04155359 | Recruiting | Bladder Cancer | Observational diagnostic | 3000 | Evaluation of urine exosomes miR Sentinel™ BCa Test, a urine exosome-based diagnostic test, as an aid in diagnosing bladder cancer |
| 15 | NCT05427227 | Recruiting | Advanced or Late Stage Gastrointestinal Cancer | Observational diagnostic | 500 | Plasma derived exosomes to explore the efficacy and mechanism of anti-HER2, immunotherapy and anti-CLDN18.2 of gastrointestinal cancer |
| 16 | NCT04167722 | Recruiting | Prostate Cancer | Observational Robotic Radical Prostatectomy | 100 | Evaluation of exosomal small RNAs in lean vs. obese patients (array) |
| 17 | NCT04258735 | Recruiting | Metastatic Breast Cancer | Interventional First line of treatment | 300 | Peripheral blood exosome nucleic acid content evaluation after first line of treatment |
| 18 | NCT03109873 | Completed | Larynx, Lip, Oral Cavity, Pharynx Cancer | Interventional Drug: Metformin Hydrochloride | 9 | Characterization of exosome profile |
| 19 | NCT04629079 | Recruiting | Lung Cancer | Observational Diagnostic | 800 | Characterization of exosomal pre-microRNA early detection in hypoxia tumor |
| 20 | NCT04556916 | Recruiting | Prostate Cancer | Observational Diagnostic | 320 | Molecular characterization of circulating blood exosomes as diagnostic markers |
| 21 | NCT03895216 | Completed | Bone Metastases | Observational Diagnostic | 34 | Evaluation of miRNA content of circulating tumor exosomes after surgery of bone metastasis |
| 22 | NCT02147418 | Recruiting | Oropharyngeal Cancer | Observational Case control | 30 | Exosome testing as a screening modality for human papillomavirus-positive oropharyngeal squamous cell carcinoma |
| 23 | NCT04530890 | Recruiting | Breast Cancer Digestive Cancer Gynecologic Cancer | Interventional Diagnostic | 1000 | Evaluation of the exosomes diagnostic value |
| 24 | NCT03032913 | Completed | PDAC | Observational Diagnostic | 52 | Evaluation of onco-exosome quantification in the diagnosis of pancreatic cancer |
| 25 | NCT03488134 | Active, not recruiting | Thyroid Cancer | Observational Diagnostic | 74 | Predicting prognosis and recurrence of thyroid cancer via new biomarkers, urinary exosomal Thyroglobulin and Galectin-3 |
| 26 | NCT04453046 | Terminated | Squamous Cell Carcinoma of the Head and Neck | Interventional Drug: Hemopurifier, Pembrolizumab | 2 | Kinetics of exosome depletion and reactivation in individual patient during the treatment |
| 27 | NCT02662621 | Completed | Cancer | Interventional hormone therapy | 71 | Evaluation of HSP70-exosomes in blood and urine prognostic marker |
| 28 | NCT05424029 | Recruiting | NSCL Cancer | Observational Diagnostic | 200 | Extracellular vesicles and particles (EVP) number as biomarkers of recurrence in non-small cell lung cancer |
| 29 | NCT04960956 | Terminated | Prostate Cancer Urothelial Carcinoma | Observational Diagnostic | 13 | Evaluation of urine glycosylate exosomes |
| 30 | NCT04781062 | Active, not recruiting | Breast Cancer | Interventional | 367 | MiRNA exosome sequencing As signature of early diagnosis |
| 31 | NCT04100811 | Recruiting | Prostate Cancer | Observational Diagnostic | 4000 | SncRNAs urine exosome validation platform |
| 32 | NCT04720599 | Completed | Urologic Cancer | Observational Diagnostic | 120 | ExoDx Prostate (IntelliScore) in men presenting for initial prostate biopsy |
| 33 | NCT03811600 | Completed | Sleep Apnea Syndromes Obstructive Cancer | Observational Diagnostic | 90 | Evaluation of exosomal PD-1/PD-L1 expression |
| 34 | NCT03432806 | Recruiting | Colon Cancer Liver Tumors | Observational Diagnostic | 80 | Characterization of exosomal proteins as biomarkers to monitor the liver pre-metastatic niche |
| 35 | NCT04357717 | Terminated | Prostate Cancer | Observational Diagnostic early marker | 150 | Clinical performance of the ExoDx prostate cancer |
| 36 | NCT05559177 | Recruiting | Chimeric exosomal tumor vaccines | Interventional Chimeric exosomal tumor vaccines | 9 | Dose evaluation of chimeric exosome vaccine |
| 37 | NCT04852653 | Recruiting | Rectal Cancer, Liquid Biopsy | Observational | 40 | Evaluating extracellular vesicles obtained via liquid biopsy for neoadjuvant treatment response assessment in rectal cancer (RECC-EV) |
| 38 | NCT02977468 | Recruiting | Triple Negative Breast Cancer | Interventional Drug: Pembrolizumab | 15 | Evaluation of serum exosomes as marker in response to therapy |
| 39 | NCT03824275 | Active, not recruiting | Prostatic Neoplasms | Interventional Drug: 18F-DCFPyL PET/CT | 129 | Characterization of circulating exosomes |
| 40 | NCT03911999 | Completed | Prostate Cancer | Observational | 180 | Identification of potential exosomal microRNAs in urine as marker of diagnosis |
| 41 | NCT05572099 | Recruiting | Prostate Cancer | Observational | 750 | Evaluation of the ExoDx prostate test |
| 42 | NCT03711890 | Recruiting | Pancreatic Carcinoma Pancreatic Intraductal Papillary Mucinous Neoplasm | Interventional procedure: Optical Coherence Tomography | 75 | Evaluation of blood cancer-derived exosomes |
| 43 | NCT03031418 | Completed | Cancer of Prostate | Observational Validation diagnostic tool | 532 | Clinical Evaluation of the ‘ExoDx Prostate IntelliScore’ |
| 44 | NCT03317080 | Active, not recruiting | Lung Cancer | Observational Early diagnosis | 400 | Clinical evaluation of tumor exosome liquid biopsy |
| 45 | NCT05397548 | Recruiting | Gastric Cancer | Observational Early diagnosis | 700 | Evaluation of predictive value of circulating exosomal lncRNA-GC1 |
| 46 | NCT02862470 | Completed | Thyroid Cancer | Observational Early diagnosis | 22 | Evaluation of urine exosomes as biological markers |
| 47 | NCT03791073 | Recruiting | Oncology | Observational | 200 | Evaluation of interstitial tissue fluid as source of exosomes |
| 48 | NCT03235687 | Active, not recruiting | Cancer of the Prostate | Interventional Surgery | 1000 | Evaluation of the ExoDx Prostate (IntelliScore) |
| 49 | NCT02063464 | Completed | Ovarian Cancer Cancer of the Ovary Ovarian Neoplasms | Observational | 85 | Evaluation of blood exosomes as prognostic and outcome |
| 50 | NCT05375604 | Recruiting | Advanced HCC Gastric Cancer Metastatic to Liver Colorectal Cancer Metastatic to Liver | Interventional Drug: CDK-004 | 30 | Evaluation of CDK-004 as marker of cell-derived exosomes efficiency |
| 51 | NCT05101655 | Completed | Osteosarcoma Pulmonary Metastases | Observational Diagnosis | 60 | Development of microfluidic chip technology to capture exosome subgroups |
| 52 | NCT03108677 | Active, not recruiting | Lung Metastases Osteosarcoma | Observational Diagnosis | 90 | Identification of RNA profile from circulating exosomes |
| 53 | NCT02439008 | Terminated | Carcinoma Hepatocellular Colorectal Neoplasms Melanoma Kidney Neoplasms | Interventional Radiotherapy | 28 | Quantification of secreted exosomes before and after radiotherapy |
| 54 | NCT05334849 | Completed | Advanced Gastric Carcinoma Immunotherapy | Observational | 80 | Identification of identified circulating exosomal lncRNA-GC1 as marker of efficacy of immunotherapy |
| 55 | NCT03250078 | Recruiting | Pancreatic Neoplasms | Observational | 100 | Evaluation of serum exosomes carrying gene mutation associate with high risk of hereditary gastric cancer |
| 56 | NCT03109847 | Completed | Thyroid Cancer | Interventional Drug: Metformin Hydrochloride Radioactive iodine | 13 | Characterization of serum and salivary exosome profile |
| 57 | NCT02921854 | Completed | NSCL Cancer | Interventional Immunotherapy | 60 | Detection of circulating markers of immunogenic cell death |
| 58 | NCT05192694 | Recruiting | Prostate Cancer | Interventional Procedure: Tomography | 40 | Extracellular vesicles (exosomes) isolated and markers of reactive stroma |
| 59 | NCT02507583 | Completed | Malignant Glioma Neoplasms | Interventional Drug: Exosomal IGF-1R/AS | 33 | Evaluation of blood IGF-1R/AS as dose escalation |
| 60 | NCT04948437 | Recruiting | Sarcoma | Observational Diagnosis | 103 | Urinary exosomal thyroglobulin, galectin-3, calprotectin A9, transketolase, keratin 19, angiopoietin-1, tissue inhibitor of metalloproteinas, keratin 8, calprotectin A8, annexin II. |
| 61 | NCT03800121 | Recruiting | Glioblastoma Multiforme | Observational Diagnosis | 30 | Evaluation of blood exosomes |
| 62 | NCT02071719 | Terminated | Renal Cell Cancer | Observational | 6 | Evaluation of serum and urine exosomes |
| 63 | NCT01550523 | Completed | Malignant Glioma of Brain | Interventional Drug: exosomal IGF-1R/AS ODN | 13 | Evaluation of exosomal IGF-1R/AS ODN-induced tumor cell |
| 64 | NCT02892734 | Terminated | HER2/Neu Negative, Recurrent Inflammatory Breast Carcinoma Breast Cancer Breast Carcinoma | Interventional Drug: Ipililumab Nivolumab | 3 | Evaluation of blood exosomes as response to therapy |
| 65 | NCT04053855 | Recruiting | Clear Cell Renal Cell Carcinoma | Observational Diagnosis | 100 | Evaluation of urinary exosomes |
| 66 | NCT05705583 | Recruiting | Renal Cell Carcinoma | Observational | 100 | Evaluation of circulating exosomes blood and urine as marker of response to immunotherapy |
| 67 | NCT02928432 | Completed | Prostate Cancer | Interventional Drug: Rednisone to dexamethasone change in mCRPC patients treated with abiraterone | 26 | Evaluation of blood exosomes as marker of therapy switch |
| 68 | NCT04499794 | Recruiting | Untreated Advanced NSCL cancer Patients FISH Identified ALK Fusion Positive or Negative | Interventional Drug: ULK inhibitor | 75 | Study of exosomal EML4-ALK fusion |
| 69 | NCT03228277 | Completed | NSCL Cancer | Interventional Drug: Olmutinib | 25 | Evaluation of DNA T790M mutation from bronchoalveolar lavage fluid EVs |
| 70 | NCT03334708 | Recruiting | Pancreatic Cancer, Pancreatic Diseases, Pancreatitis, Pancreatic Cyst | Observational Diagnosis | 700 | Evaluation of blood exosome profile |
| 71 | NCT03985696 | Recruiting | Lymphoma, B-cell, Aggressive Non-Hodgkin (B-NHL) | Interventional Drug: anti-CD20 and Nivolumab | 90 | Evaluation of exosomal CD20 and PD-L1 exosomes and resistance to immunotherapy |
| 72 | NCT04340245 | Active, not recruiting | Prostate Cancer | Observational Diagnosis | 60 | Evaluation of blood and urine exosomes to discriminate progressors from non-progressors |
| 73 | NCT05575622 | Recruiting | Hepatocellular Carcinoma | Observational Diagnosis | 200 | Clinical analysis of CTC and exosome combination and efficacy of immunotherapy in patients with hepatocellular carcinoma |
| 74 | NCT03854032 | Active, not recruiting | Lip, Oral Cavity Squamous Cell Carcinoma, Pharynx, Larynx, Squamous Cell Carcinoma | Interventional Drug: Nivolumab | 45 | Evaluation of blood exosome abundance and composition and treating patients with stage II-IV squamous cell cancer |
| 75 | NCT03236675 | Active, not recruiting | Carcinoma, Non-Small-Cell Lung | Observational Diagnosis | 60 | Evaluation of exosomal EML4-ALK fusion transcripts and T790M EGFR mutation |
| 76 | NCT01629498 | Recruiting | Recurrent Lung Non-Small Cell Carcinoma | Interventional Image-Guided | 100 | Evaluation of blood exosomes and treatment of regionally metastatic head and neck squamous cell cancer |
| 77 | NCT05110781 | Recruiting | Cutaneous Squamous Cell Carcinoma of the Head and Neck | Interventional Drug: Atezolizumab | 18 | Evaluation of plasma exosomal levels |
| 78 | NCT05441189 | Completed | Stage I–II PDAC | Observational Diagnosis | 70 | Evaluation of specific exosome signature and gene signature for predicting |
| 79 | NCT05166616 | Recruiting | Advanced NSCL Carcinoma | Interventional Drug: Minnelide and Osimertinib | 30 | Evaluation of exosomal pattern and treatment of advanced EGFR mutated non-small cell lung cancer |
| 80 | NCT03096340 | Terminated | Cancer | Observational Drug: IT-141 | 10 | Evaluation of exosomes presence and monotherapy in patients with advanced cancer |
| 81 | NCT00578240 | Active, not recruiting | Prostate Cancer | Observational Diagnosis | 5290 | Evaluation of exosome presence into peripheral blood, bone marrow, urine and other bodily fluids |
| 82 | NCT02051101 | Completed | Port-Wine Stain | Interventional | 19 | Characterization of exosome profile and pathogenic mechanisms of port wine stain |
| 83 | NCT02535247 | Terminated | Lymphoma T-Cell, Peripheral | Interventional Drug: MK-3475 | 17 | Circulating exosome analysis and study of MK-3475 R/R NK and T-cell non-Hodgkin Lymphoma |
| 84 | NCT03410030 | Completed | Pancreatic Adenocarcinoma Resectable and Metastases | Interventional Drug: AA NABPLAGEM | 27 | Evaluation of potential exosome pattern as biomarker and AA NABPLAGEM |
| 85 | NCT03083678 | Active, not recruiting | Chordoma | Interventional Drug: Afatinib | 43 | Evaluation of circulating exosomes and Afatinib in locally advanced and metastatic chordoma |
| 86 | NCT03217266 | Active, not recruiting | Soft Tissue Sarcoma | Interventional Drug: Navtemadlin | 46 | Evaluation of tumor genetic mutations in deoxyribonucleic acid ribonucleic acid isolated from exosomes and Navtemadlin and radiation therapy in treating patients with soft tissue sarcoma |
| 87 | NCT03537599 | Completed | Recurrent Acute Myeloid Leukemia with Myelodysplasia | Interventional Drug: Daratumumab | 4 | Evaluation of exosome pattern and Daratumumab and donor lymphocyte infusion in treating participants with relapsed acute myeloid leukemia after stem cell transplant |
| 88 | NCT04483219 | Recruiting | Metastatic Colorectal Adenocarcinoma | Interventional Drug: Tyrosine Kinase Inhibitor (TKI) + Anti-PD-1 Antibody | 53 | Evaluation of circulating exosome pattern in metastatic colorectal adenocarcinoma |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schiano, C.; Balbi, C.; de Nigris, F.; Napoli, C. Basic Pathogenic Mechanisms and Epigenetic Players Promoted by Extracellular Vesicles in Vascular Damage. Int. J. Mol. Sci. 2023, 24, 7509. https://doi.org/10.3390/ijms24087509
Schiano C, Balbi C, de Nigris F, Napoli C. Basic Pathogenic Mechanisms and Epigenetic Players Promoted by Extracellular Vesicles in Vascular Damage. International Journal of Molecular Sciences. 2023; 24(8):7509. https://doi.org/10.3390/ijms24087509
Chicago/Turabian StyleSchiano, Concetta, Carolina Balbi, Filomena de Nigris, and Claudio Napoli. 2023. "Basic Pathogenic Mechanisms and Epigenetic Players Promoted by Extracellular Vesicles in Vascular Damage" International Journal of Molecular Sciences 24, no. 8: 7509. https://doi.org/10.3390/ijms24087509
APA StyleSchiano, C., Balbi, C., de Nigris, F., & Napoli, C. (2023). Basic Pathogenic Mechanisms and Epigenetic Players Promoted by Extracellular Vesicles in Vascular Damage. International Journal of Molecular Sciences, 24(8), 7509. https://doi.org/10.3390/ijms24087509







