Hyperbaric Oxygen Therapy Reduces Oxidative Stress and Inflammation, and Increases Growth Factors Favouring the Healing Process of Diabetic Wounds
Abstract
1. Introduction
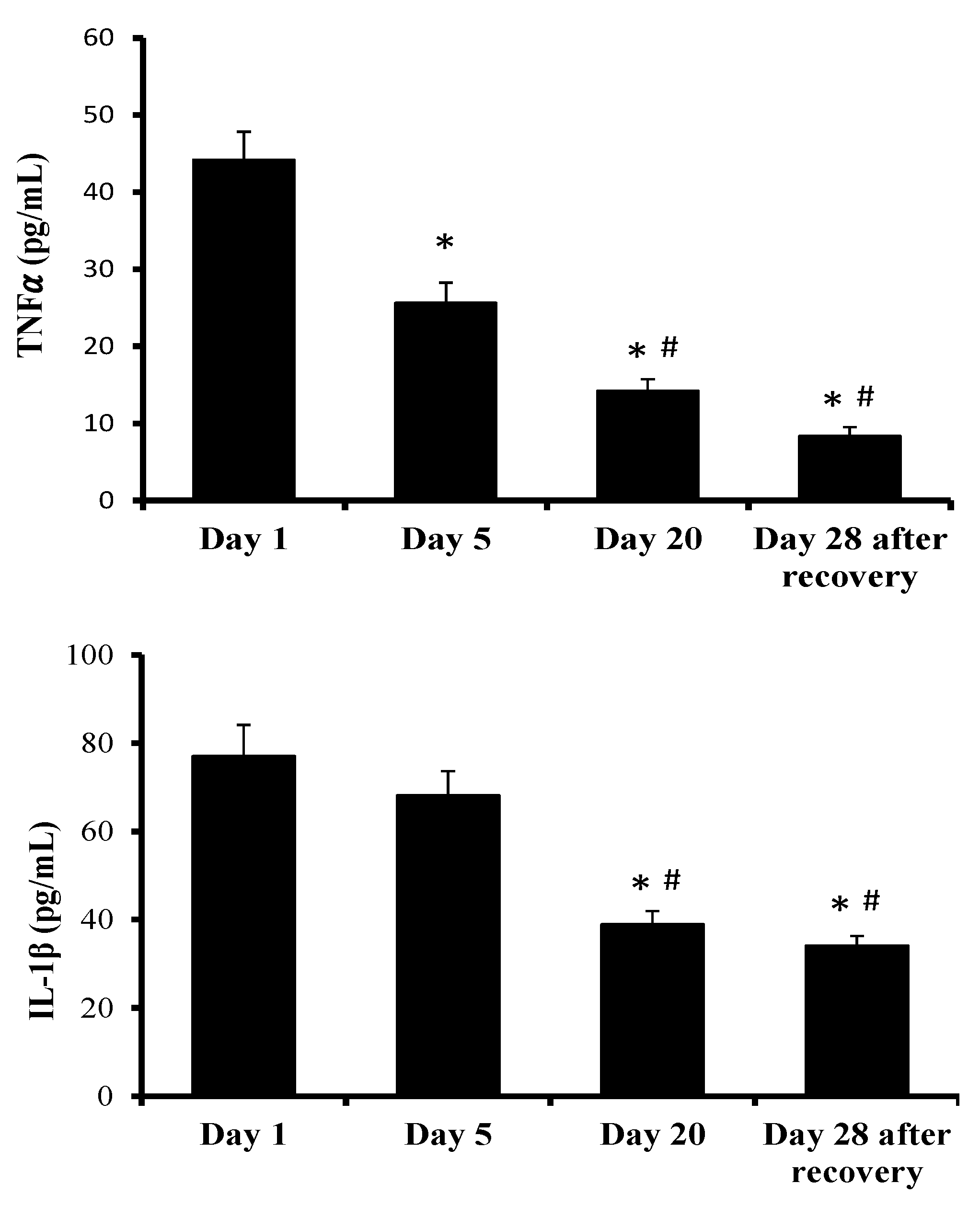
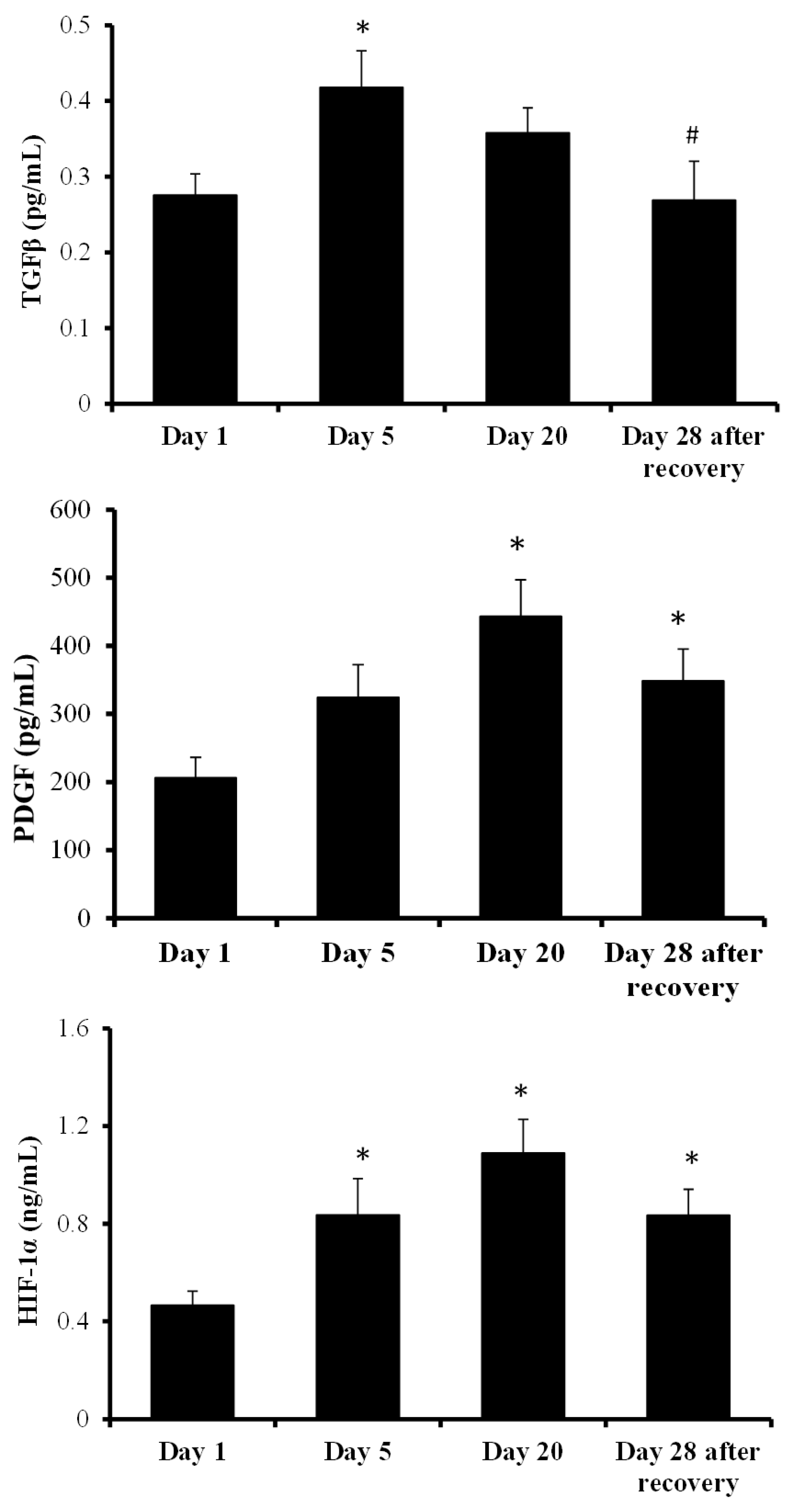
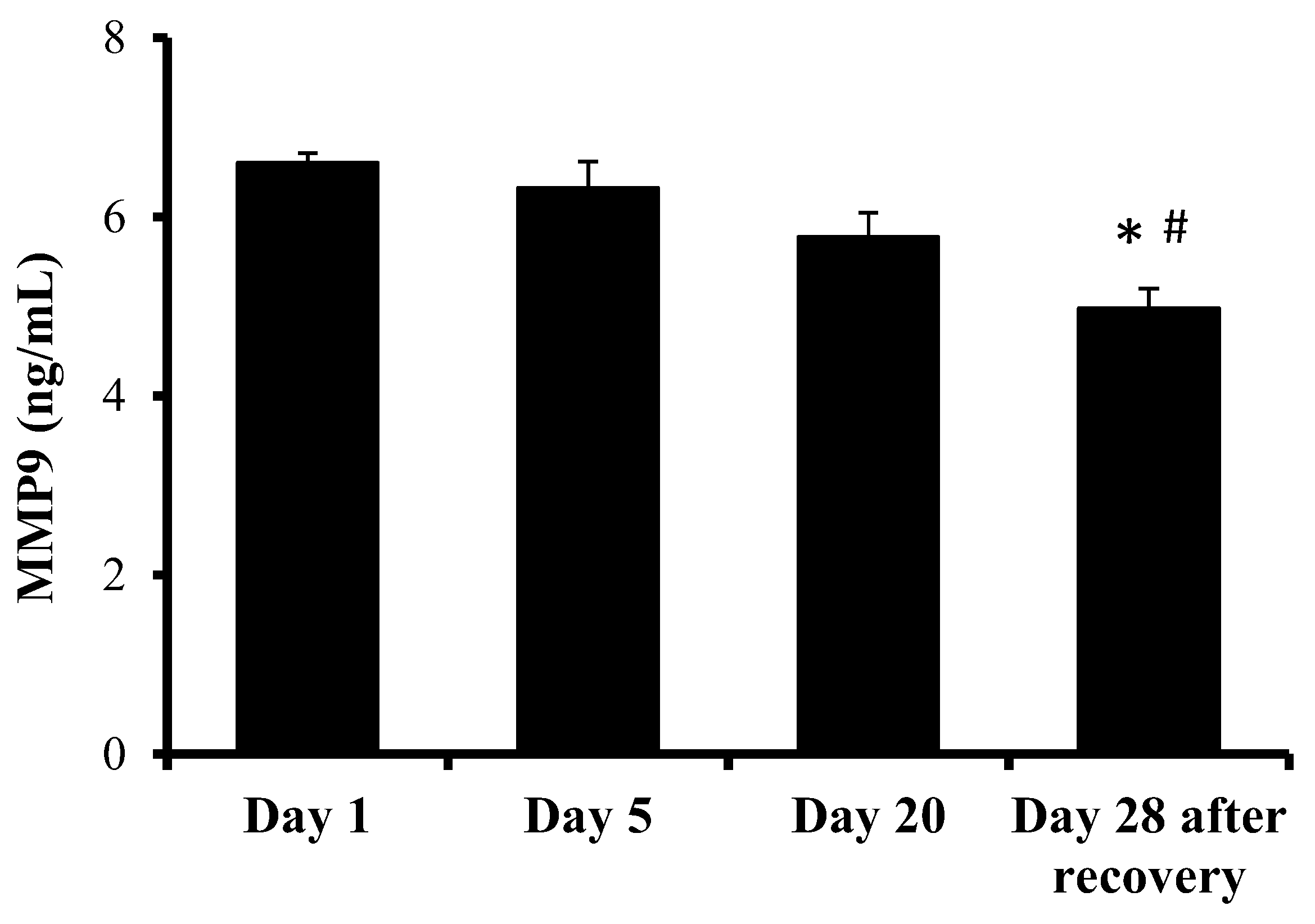
2. Results
3. Discussion
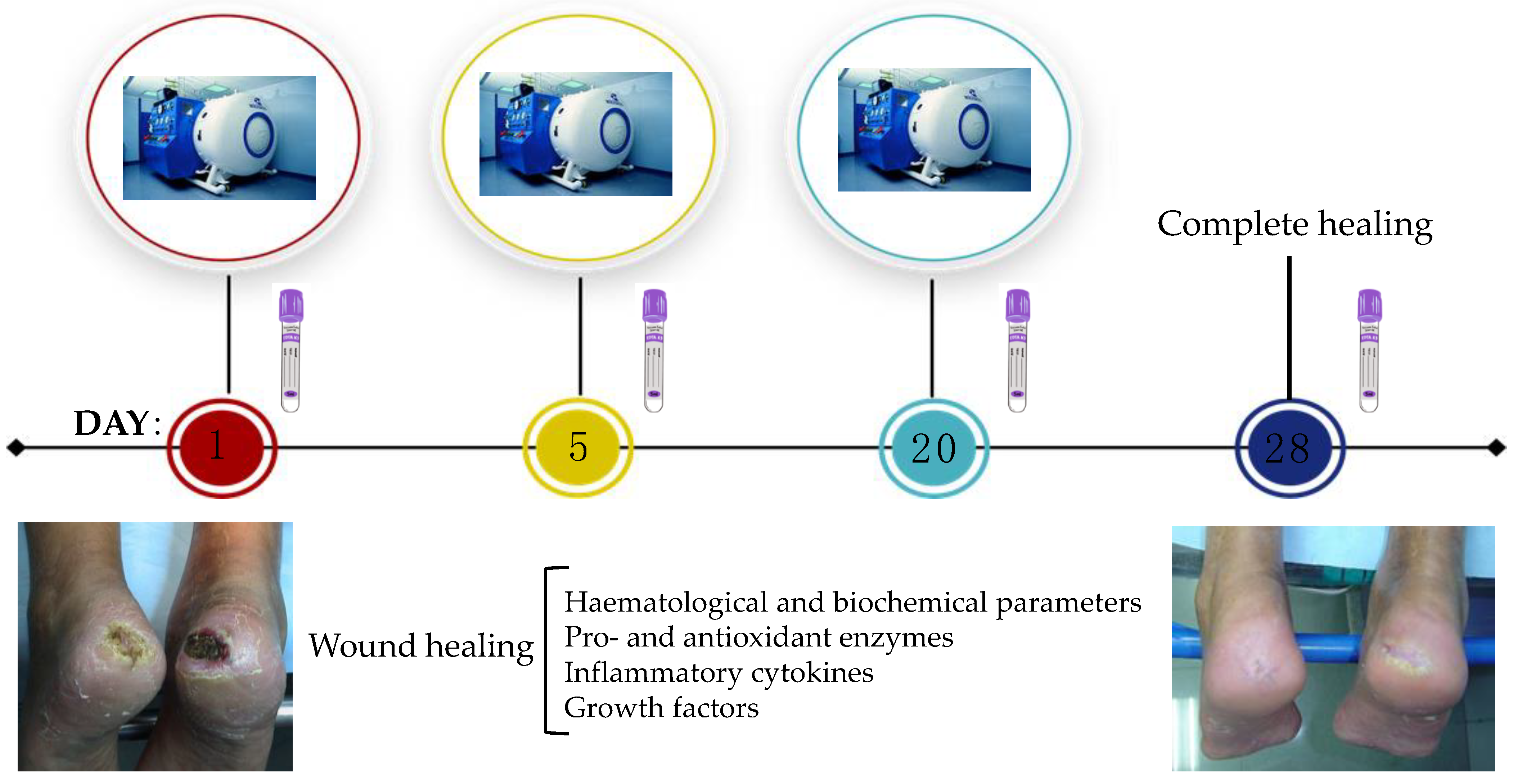
4. Materials and Methods
4.1. Patient Characteristics
4.2. Experimental Procedure
4.3. Western Blot Analysis
4.4. MDA Levels
4.5. Protein Carbonyl Derivates
4.6. ELISA Assays
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sureda, A.; Batle, J.M.; Martorell, M.; Capó, X.; Tejada, S.; Tur, J.A.; Pons, A. Antioxidant Response of Chronic Wounds to Hyperbaric Oxygen Therapy. PLoS ONE 2016, 11, e0163371. [Google Scholar] [CrossRef]
- Thackham, J.A.; McElwain, D.L.; Long, R.J. The use of hyperbaric oxygen therapy to treat chronic wounds: A review. Wound Repair Regen. 2008, 16, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Lindenmann, J.; Smolle, C.; Kamolz, L.P.; Smolle-Juettner, F.M.; Graier, W.F. Survey of Molecular Mechanisms of Hyperbaric Oxygen in Tissue Repair. Int. J. Mol. Sci. 2021, 22, 11754. [Google Scholar] [CrossRef]
- Edwards, M.L. Hyperbaric oxygen therapy. Part 1: History and principles. J. Vet. Emerg. Crit. Care 2010, 20, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Morrison, A.; Madden, C.; Messmer, J. Management of Chronic Wounds. Prim. Care 2022, 49, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Frykberg, R.G. Topical Wound Oxygen Therapy in the Treatment of Chronic Diabetic Foot Ulcers. Medicina 2021, 57, 917. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, D.; Marroni, A.; Kot, J. Tenth European Consensus Conference on Hyperbaric Medicine: Recommendations for accepted and non-accepted clinical indications and practice of hyperbaric oxygen treatment. Diving Hyperb. Med. 2017, 47, 24–32. [Google Scholar] [CrossRef]
- Smolle, C.; Lindenmann, J.; Kamolz, L.; Smolle-Juettner, F.M. The History and Development of Hyperbaric Oxygenation (HBO) in Thermal Burn Injury. Medicina 2021, 57, 49. [Google Scholar] [CrossRef]
- De Wolde, S.D.; Hulskes, R.H.; Weenink, R.P.; Hollmann, M.W.; Van Hulst, R.A. The Effects of Hyperbaric Oxygenation on Oxidative Stress, Inflammation and Angiogenesis. Biomolecules 2021, 11, 1210. [Google Scholar] [CrossRef]
- Kadkhoda, J.; Tarighatnia, A.; Barar, J.; Aghanejad, A.; Davaran, S. Recent advances and trends in nanoparticles based photothermal and photodynamic therapy. Photodiagnosis Photodyn. Ther. 2022, 37, 20. [Google Scholar] [CrossRef]
- Moulik, P.K.; Mtonga, R.; Gill, G.V. Amputation and mortality in new-onset diabetic foot ulcers stratified by etiology. Diabetes Care 2003, 26, 491–494. [Google Scholar] [CrossRef]
- Johnston, B.R.; Ha, A.Y.; Brea, B.; Liu, P.Y. The Mechanism of Hyperbaric Oxygen Therapy in the Treatment of Chronic Wounds and Diabetic Foot Ulcers. RI Med. J. 2013, 99, 26–29. [Google Scholar]
- Boulton, A.J.; Vileikyte, L.; Ragnarson-Tennvall, G.; Apelqvist, J. The global burden of diabetic foot disease. Lancet 2005, 366, 1719–1724. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-Soares, M.; Boyko, E.J.; Jeffcoate, W.; Mills, J.L.; Russell, D.; Morbach, S.; Game, F. Diabetic foot ulcer classifications: A critical review. Diabetes Metab. Res. Rev. 2020, 36, e3272. [Google Scholar] [CrossRef]
- Pendsey, S.P. Understanding diabetic foot. Int. J. Diabetes Dev. Ctries. 2010, 30, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Spampinato, S.F.; Caruso, G.I.; De Pasquale, R.; Sortino, M.A.; Merlo, S. The Treatment of Impaired Wound Healing in Diabetes: Looking among Old Drugs. Pharmaceuticals 2020, 13, 60. [Google Scholar] [CrossRef] [PubMed]
- Peschen, M.; Lahaye, T.; Hennig, B.; Weyl, A.; Simon, J.C.; Vanscheidt, W. Expression of the adhesion molecules ICAM-1, VCAM-1, LFA-1 and VLA-4 in the skin is modulated in progressing stages of chronic venous insufficiency. Acta Derm. Venereol. 1999, 79, 27–32. [Google Scholar]
- Tejada, S.; Batle, J.M.; Ferrer, M.D.; Busquets-Cortés, C.; Monserrat-Mesquida, M.; Nabavi, S.M.; Del Mar Bibiloni, M.; Pons, A.; Sureda, A. Therapeutic Effects of Hyperbaric Oxygen in the Process of Wound Healing. Curr. Pharm. Des. 2019, 25, 1682–1693. [Google Scholar] [CrossRef]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef]
- Stoekenbroek, R.M.; Santema, T.B.; Legemate, D.A.; Ubbink, D.T.; van den Brink, A.; Koelemay, M.J. Hyperbaric oxygen for the treatment of diabetic foot ulcers: A systematic review. Eur. J. Vasc. Endovasc. Surg. 2014, 47, 647–655. [Google Scholar] [CrossRef]
- Mcintosh, A.; Chiverton, R.; Clarkson, S.; Feder, G.; Foster, A.; Gadsby, R.; Home, P.D.; Hutchinson, A.; O’Connor, M.; Peters, J. Prevention and Management of Foot Problems in Type 2 Diabetes: Clinical Guidelines and Evidence; University of Sheffield: Sheffield, UK, 2003. [Google Scholar]
- Yip, W.L. Influence of oxygen on wound healing. Int. Wound J. 2015, 12, 620–624. [Google Scholar] [CrossRef] [PubMed]
- Hopf, H.W.; Hunt, T.K.; West, J.M.; Blomquist, P.; Goodson, W.H., 3rd; Jensen, J.A.; Jonsson, K.; Paty, P.B.; Rabkin, J.M.; Upton, R.A.; et al. Wound tissue oxygen tension predicts the risk of wound infection in surgical patients. Arch. Surg. 1997, 132, 997–1004. [Google Scholar] [CrossRef]
- Memar, M.Y.; Ghotaslou, R.; Samiei, M.; Adibkia, K. Antimicrobial use of reactive oxygen therapy: Current insights. Infect. Drug Resist. 2018, 11, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Yıldırım, A.O.; Eryılmaz, M.; Kaldırım, U.; Eyi, Y.E.; Tuncer, S.K.; Eroğlu, M.; Durusu, M.; Topal, T.; Kurt, B.; Dilmen, S.; et al. Effectiveness of hyperbaric oxygen and ozone applications in tissue healing in generated soft tissue trauma model in rats: An experimental study. Ulus Travma Acil Cerrahi Derg 2014, 20, 167–175. [Google Scholar] [CrossRef]
- Sheikh, A.Y.; Rollins, M.D.; Hopf, H.W.; Hunt, T.K. Hyperoxia improves microvascular perfusion in a murine wound model. Wound Repair Regen. 2005, 13, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Sunkari, V.G.; Lind, F.; Botusan, I.R.; Kashif, A.; Liu, Z.J.; Ylä-Herttuala, S.; Brismar, K.; Velazquez, O.; Catrina, S.B. Hyperbaric oxygen therapy activates hypoxia-inducible factor 1 (HIF-1), which contributes to improved wound healing in diabetic mice. Wound Repair Regen. 2015, 23, 98–103. [Google Scholar] [CrossRef]
- Zhao, L.L.; Davidson, J.D.; Wee, S.C.; Roth, S.I.; Mustoe, T.A. Effect of hyperbaric oxygen and growth factors on rabbit ear ischemic ulcers. Arch. Surg. 1994, 129, 1043–1049. [Google Scholar] [CrossRef]
- Santema, K.T.B.; Stoekenbroek, R.M.; Koelemay, M.J.W.; Reekers, J.A.; van Dortmont, L.M.C.; Oomen, A.; Smeets, L.; Wever, J.J.; Legemate, D.A.; Ubbink, D.T. Hyperbaric Oxygen Therapy in the Treatment of Ischemic Lower- Extremity Ulcers in Patients With Diabetes: Results of the DAMO(2)CLES Multicenter Randomized Clinical Trial. Diabetes Care 2018, 41, 112–119. [Google Scholar] [CrossRef]
- Fedorko, L.; Bowen, J.M.; Jones, W.; Oreopoulos, G.; Goeree, R.; Hopkins, R.B.; O’Reilly, D.J. Hyperbaric Oxygen Therapy Does Not Reduce Indications for Amputation in Patients With Diabetes With Nonhealing Ulcers of the Lower Limb: A Prospective, Double-Blind, Randomized Controlled Clinical Trial. Diabetes Care 2016, 39, 392–399. [Google Scholar] [CrossRef]
- Heyboer, M., 3rd; Sharma, D.; Santiago, W.; McCulloch, N. Hyperbaric Oxygen Therapy: Side Effects Defined and Quantified. Adv. Wound Care 2017, 6, 210–224. [Google Scholar] [CrossRef]
- Plafki, C.; Peters, P.; Almeling, M.; Welslau, W.; Busch, R. Complications and side effects of hyperbaric oxygen therapy. Aviat. Space Environ. Med. 2000, 71, 119–124. [Google Scholar]
- Barnes, R.C. Point: Hyperbaric oxygen is beneficial for diabetic foot wounds. Clin. Infect. Dis. 2006, 43, 188–192. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Jude, E.B. The role of matrix metalloproteinases in wound healing. J. Am. Podiatr. Med. Assoc. 2002, 92, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Rathur, H.M.; Boulton, A.J. The diabetic foot. Clin. Dermatol. 2007, 25, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Schlattner, U.; Möckli, N.; Speer, O.; Werner, S.; Wallimann, T. Creatine kinase and creatine transporter in normal, wounded, and diseased skin. J. Investig. Dermatol. 2002, 118, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Caux, F.; Chosidow, O.; Philippon, C.; Roujeau, J.C.; Revuz, J. Increased serum and blister fluid levels of creatine kinase in patients with toxic epidermal necrolysis. Br. J. Dermatol. 1994, 130, 337–341. [Google Scholar] [CrossRef]
- Ahrenholz, D.H.; Schubert, W.; Solem, L.D. Creatine kinase as a prognostic indicator in electrical injury. Surgery 1988, 104, 741–747. [Google Scholar]
- Liu, Z.; Hu, L.; Zhang, T.; Xu, H.; Li, H.; Yang, Z.; Zhou, M.; Smith, H.S.; Li, J.; Ran, J.; et al. PKCβ increases ROS levels leading to vascular endothelial injury in diabetic foot ulcers. Am. J. Transl. Res. 2020, 12, 6409–6421. [Google Scholar]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 2017, 14, 89–96. [Google Scholar] [CrossRef]
- Schäfer, M.; Werner, S. Oxidative stress in normal and impaired wound repair. Pharmacol. Res. 2008, 58, 165–171. [Google Scholar] [CrossRef]
- Roy, S.; Khanna, S.; Nallu, K.; Hunt, T.K.; Sen, C.K. Dermal wound healing is subject to redox control. Mol. Ther. 2006, 13, 211–220. [Google Scholar] [CrossRef]
- Muzumdar, S.; Hiebert, H.; Haertel, E.; Ben-Yehuda Greenwald, M.; Bloch, W.; Werner, S.; Schäfer, M. Nrf2-Mediated Expansion of Pilosebaceous Cells Accelerates Cutaneous Wound Healing. Am. J. Pathol. 2019, 189, 568–579. [Google Scholar] [CrossRef] [PubMed]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Jones, J.I.; Wolter, W.R.; Pérez, R.L.; Schroeder, V.A.; Champion, M.M.; Hesek, D.; Lee, M.; Suckow, M.A.; Mobashery, S.; et al. Hyperbaric oxygen therapy accelerates wound healing in diabetic mice by decreasing active matrix metalloproteinase-9. Wound Repair Regen. 2020, 28, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Hille, R.; Nishino, T. Flavoprotein structure and mechanism. 4. Xanthine oxidase and xanthine dehydrogenase. FASEB J. 1995, 9, 995–1003. [Google Scholar] [CrossRef]
- Berry, C.E.; Hare, J.M. Xanthine oxidoreductase and cardiovascular disease: Molecular mechanisms and pathophysiological implications. J. Physiol. 2004, 555, 589–606. [Google Scholar] [CrossRef]
- Farquharson, C.A.; Butler, R.; Hill, A.; Belch, J.J.; Struthers, A.D. Allopurinol improves endothelial dysfunction in chronic heart failure. Circulation 2002, 106, 221–226. [Google Scholar] [CrossRef]
- Verma, R.; Chopra, A.; Giardina, C.; Sabbisetti, V.; Smyth, J.A.; Hightower, L.E.; Perdrizet, G.A. Hyperbaric oxygen therapy (HBOT) suppresses biomarkers of cell stress and kidney injury in diabetic mice. Cell Stress Chaperones 2015, 20, 495–505. [Google Scholar] [CrossRef]
- Martin, P.; Nunan, R. Cellular and molecular mechanisms of repair in acute and chronic wound healing. Br. J. Dermatol. 2015, 173, 370–378. [Google Scholar] [CrossRef]
- Barrientos, S.; Stojadinovic, O.; Golinko, M.S.; Brem, H.; Tomic-Canic, M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008, 16, 585–601. [Google Scholar] [CrossRef]
- Beidler, S.K.; Douillet, C.D.; Berndt, D.F.; Keagy, B.A.; Rich, P.B.; Marston, W.A. Inflammatory cytokine levels in chronic venous insufficiency ulcer tissue before and after compression therapy. J. Vasc. Surg. 2009, 49, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Szade, A.; Grochot-Przeczek, A.; Florczyk, U.; Jozkowicz, A.; Dulak, J. Cellular and molecular mechanisms of inflammation-induced angiogenesis. IUBMB Life 2015, 67, 145–159. [Google Scholar] [CrossRef]
- Niu, C.C.; Lin, S.S.; Yuan, L.J.; Lu, M.L.; Ueng, S.W.N.; Yang, C.Y.; Tsai, T.T.; Lai, P.L. Upregulation of miR-107 expression following hyperbaric oxygen treatment suppresses HMGB1/RAGE signaling in degenerated human nucleus pulposus cells. Arthritis Res. Ther. 2019, 21, 019–1830. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Lee, M.H.; Phillips, S.A.; Stacey, M.C. Growth factors for treating chronic venous leg ulcers: A systematic review and meta-analysis. Wound Repair Regen. 2022, 30, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Rey, S.; Semenza, G.L. Hypoxia-inducible factor-1-dependent mechanisms of vascularization and vascular remodelling. Cardiovasc. Res. 2010, 86, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Palazon, A.; Goldrath, A.W.; Nizet, V.; Johnson, R.S. HIF transcription factors, inflammation, and immunity. Immunity 2014, 41, 518–528. [Google Scholar] [CrossRef]
- Manalo, D.J.; Rowan, A.; Lavoie, T.; Natarajan, L.; Kelly, B.D.; Ye, S.Q.; Garcia, J.G.; Semenza, G.L. Transcriptional regulation of vascular endothelial cell responses to hypoxia by HIF-1. Blood 2005, 105, 659–669. [Google Scholar] [CrossRef]
- Fallah, A.; Sadeghinia, A.; Kahroba, H.; Samadi, A.; Heidari, H.R.; Bradaran, B.; Zeinali, S.; Molavi, O. Therapeutic targeting of angiogenesis molecular pathways in angiogenesis-dependent diseases. Biomed. Pharmacother. 2019, 110, 775–785. [Google Scholar] [CrossRef]
- Simonetti, O.; Lucarini, G.; Cirioni, O.; Zizzi, A.; Orlando, F.; Provinciali, M.; Di Primio, R.; Giacometti, A.; Offidani, A. Delayed wound healing in aged skin rat models after thermal injury is associated with an increased MMP-9, K6 and CD44 expression. Burns 2013, 39, 776–787. [Google Scholar] [CrossRef]
- Vaalamo, M.; Weckroth, M.; Puolakkainen, P.; Kere, J.; Saarinen, P.; Lauharanta, J.; Saarialho-Kere, U.K. Patterns of matrix metalloproteinase and TIMP-1 expression in chronic and normally healing human cutaneous wounds. Br. J. Dermatol. 1996, 135, 52–59. [Google Scholar] [CrossRef]
- Chang, M.; Nguyen, T.T. Strategy for Treatment of Infected Diabetic Foot Ulcers. Acc. Chem. Res. 2021, 54, 1080–1093. [Google Scholar] [CrossRef]
- Silva, F.S.; de Souza, K.S.C.; Galdino, O.A.; de Moraes, M.V.; Ishikawa, U.; Medeiros, M.A.; Lima, J.; de Paula Medeiros, K.C.; da Silva Farias, N.B.; de Araújo Júnior, R.F.; et al. Hyperbaric oxygen therapy mitigates left ventricular remodeling, upregulates MMP-2 and VEGF, and inhibits the induction of MMP-9, TGF-β1, and TNF-α in streptozotocin-induced diabetic rat heart. Life Sci. 2022, 295, 12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Gould, L.J. Hyperbaric oxygen reduces matrix metalloproteinases in ischemic wounds through a redox-dependent mechanism. J. Investig. Dermatol. 2014, 134, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Monserrat-Mesquida, M.; Quetglas-Llabrés, M.; Capó, X.; Bouzas, C.; Mateos, D.; Pons, A.; Tur, J.A.; Sureda, A. Metabolic Syndrome is Associated with Oxidative Stress and Proinflammatory State. Antioxidants 2020, 9, 236. [Google Scholar] [CrossRef] [PubMed]
| Day 1 | Day 5 | Day 20 | Day 28 | |
|---|---|---|---|---|
| Erythrocytes (106 cells/µL) | 4.16 ± 0.08 | 4.12 ± 0.07 | 4.13 ± 0.08 | 4.21 ± 0.10 |
| Haematocrit (%) | 38.8 ± 0.6 | 38.1 ± 0.6 | 38.4 ± 0.5 | 39.1 ± 0.7 |
| Haemoglobin (g/L) | 12.8 ± 0.9 | 12.6 ± 0.9 | 12.7 ± 1.0 | 12.8 ± 0.8 |
| Leukocytes (103 cells/µL) | 5.97 ± 0.18 | 6.18 ± 0.26 | 6.24 ± 0.23 | 5.89 ± 0.28 |
| CPK (U/L) | 256 ± 26 | 178 ± 17 * | 124 ± 10 *,# | 122 ± 13 *,# |
| LDH (U/L) | 370 ± 15 | 352 ± 17 | 340 ± 19 | 335 ± 19 |
| AST (U/L) | 28.9 ± 2.2 | 26.7 ± 2.5 | 22.7 ± 1.1* | 22.1 ± 1.3 * |
| ALT (U/L) | 29.6 ± 4.5 | 28.9 ± 4.6 | 23.0 ± 2.5 | 22.9 ± 2.8 |
| GGT (U/L) | 57.1 ± 5.0 | 53.2 ± 5.7 | 46.6 ± 4.9 | 40.7 ± 7.4 |
| Creatinine (mg/dL) | 1.07 ± 0.4 | 1.04 ± 0.04 | 1.00 ± 0.04 | 0.98 ± 0.03 |
| Day 1 | Day 5 | Day 20 | Day 28 | |
|---|---|---|---|---|
| CAT (%) | 1.80 ± 0.19 | 1.70 ± 0.21 | 1.66 ± 0.10 | 1.00 ± 0.09 *,#,& |
| EcSOD (%) | 1.65 ± 0.14 | 1.49 ± 0.13 | 1.30 ± 0.11 | 1.00 ± 0.18 *,# |
| MPO (%) | 5.15 ± 0.81 | 2.63 ± 0.24 * | 1.79 ± 0.33 *,# | 1.00 ± 0.10 *,# |
| XOX (%) | 3.13 ± 0.21 | 1.70 ± 0.09 * | 1.34 ± 0.09 * | 1.00 ± 0.07 *,# |
| Oxidative damage markers | ||||
| MDA (µM) | 0.11 ± 0.008 | 0.068 ± 0.002 * | 0.058 ± 0.002 * | 0.047 ± 0.002 *,# |
| Carbonyl index (%) | 175 ± 25 | 137 ± 22 | 116 ± 18 * | 100 ± 29 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capó, X.; Monserrat-Mesquida, M.; Quetglas-Llabrés, M.; Batle, J.M.; Tur, J.A.; Pons, A.; Sureda, A.; Tejada, S. Hyperbaric Oxygen Therapy Reduces Oxidative Stress and Inflammation, and Increases Growth Factors Favouring the Healing Process of Diabetic Wounds. Int. J. Mol. Sci. 2023, 24, 7040. https://doi.org/10.3390/ijms24087040
Capó X, Monserrat-Mesquida M, Quetglas-Llabrés M, Batle JM, Tur JA, Pons A, Sureda A, Tejada S. Hyperbaric Oxygen Therapy Reduces Oxidative Stress and Inflammation, and Increases Growth Factors Favouring the Healing Process of Diabetic Wounds. International Journal of Molecular Sciences. 2023; 24(8):7040. https://doi.org/10.3390/ijms24087040
Chicago/Turabian StyleCapó, Xavier, Margalida Monserrat-Mesquida, Magdalena Quetglas-Llabrés, Juan M. Batle, Josep A. Tur, Antoni Pons, Antoni Sureda, and Silvia Tejada. 2023. "Hyperbaric Oxygen Therapy Reduces Oxidative Stress and Inflammation, and Increases Growth Factors Favouring the Healing Process of Diabetic Wounds" International Journal of Molecular Sciences 24, no. 8: 7040. https://doi.org/10.3390/ijms24087040
APA StyleCapó, X., Monserrat-Mesquida, M., Quetglas-Llabrés, M., Batle, J. M., Tur, J. A., Pons, A., Sureda, A., & Tejada, S. (2023). Hyperbaric Oxygen Therapy Reduces Oxidative Stress and Inflammation, and Increases Growth Factors Favouring the Healing Process of Diabetic Wounds. International Journal of Molecular Sciences, 24(8), 7040. https://doi.org/10.3390/ijms24087040











