A Putative Receptor for Ferritin in Mollusks: Characterization of the Insulin-like Growth Factor Type 1 Receptor
Abstract
1. Introduction
2. Results
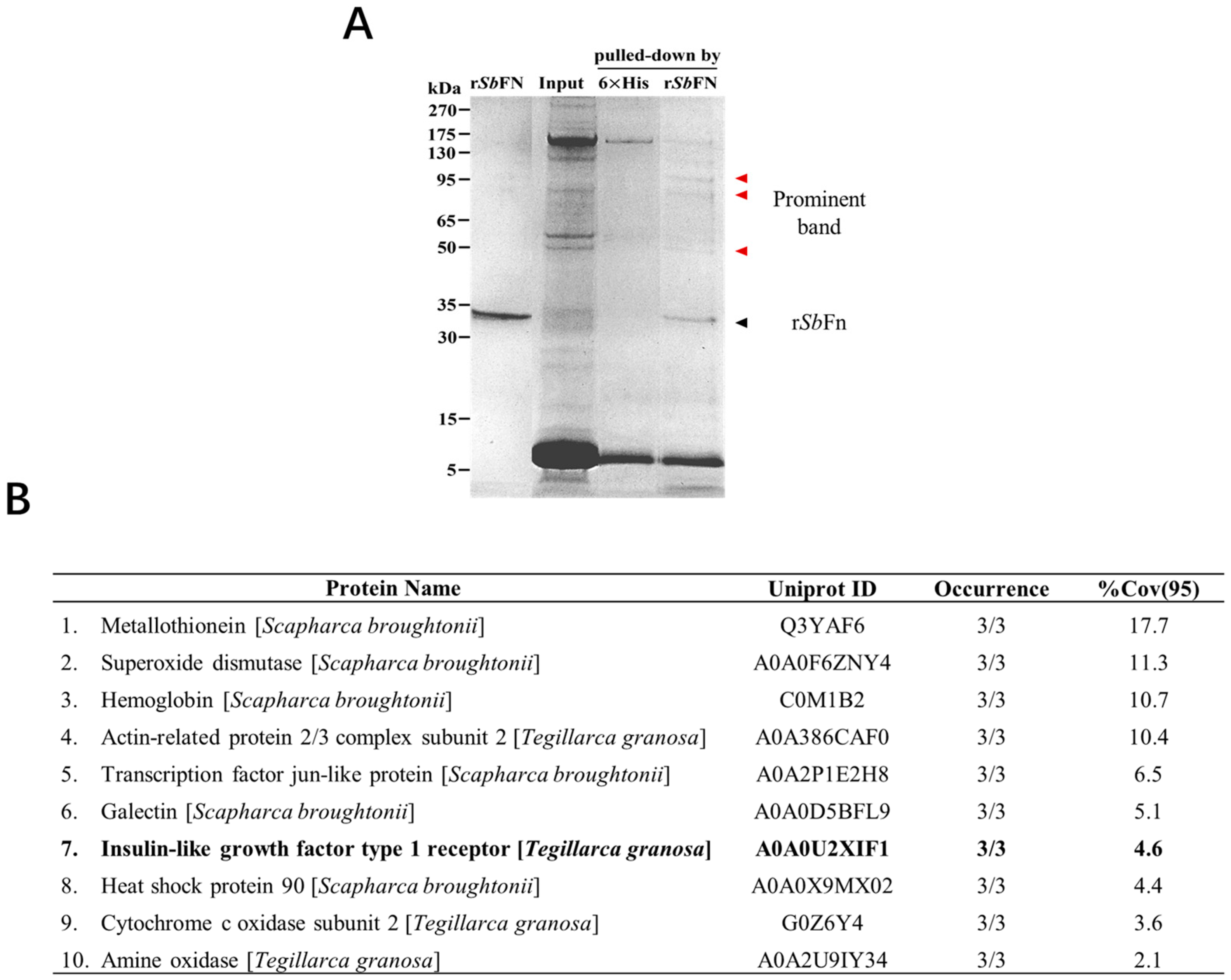
2.1. Identification of Potential Membrane Anchors for SbFn
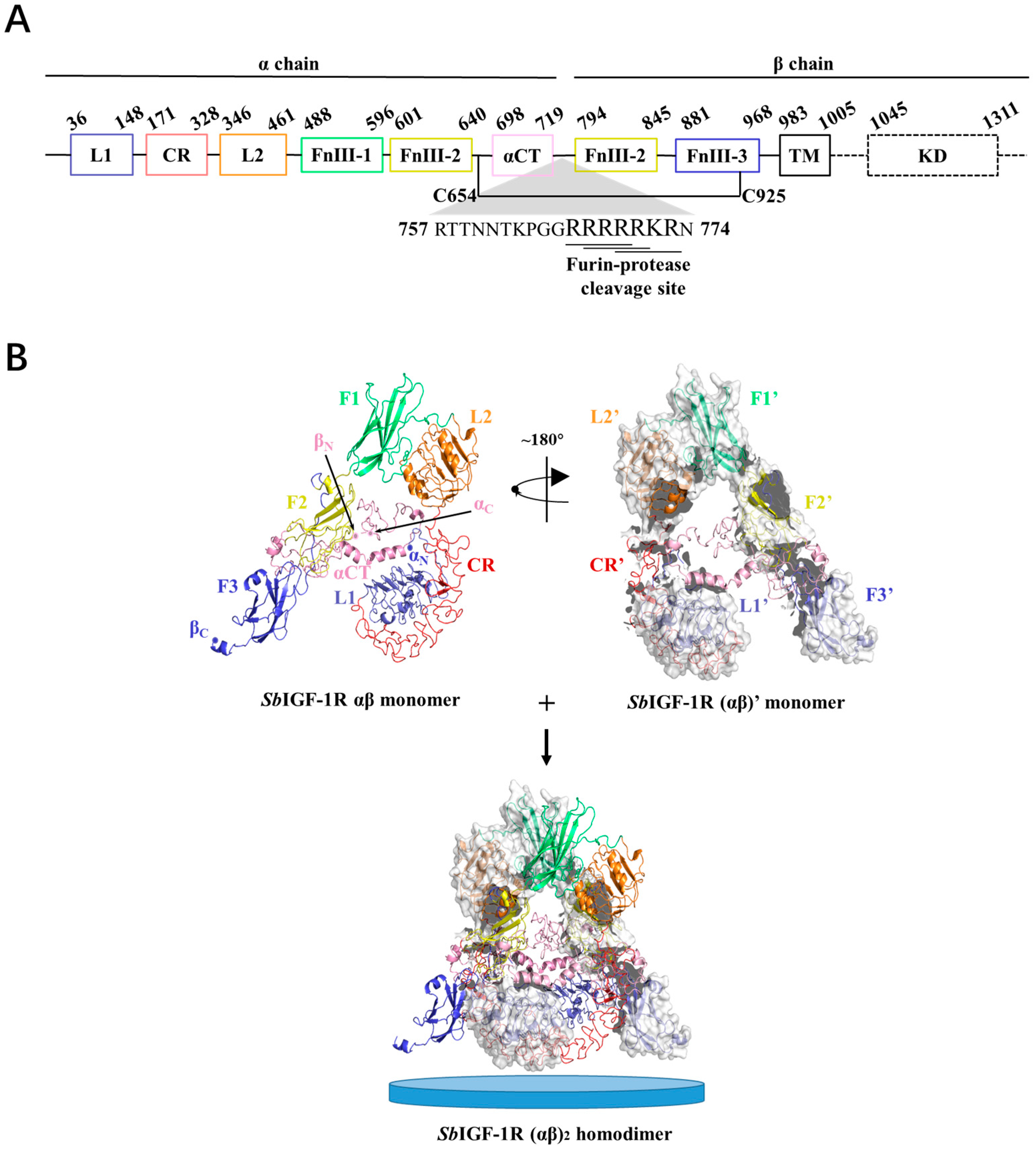
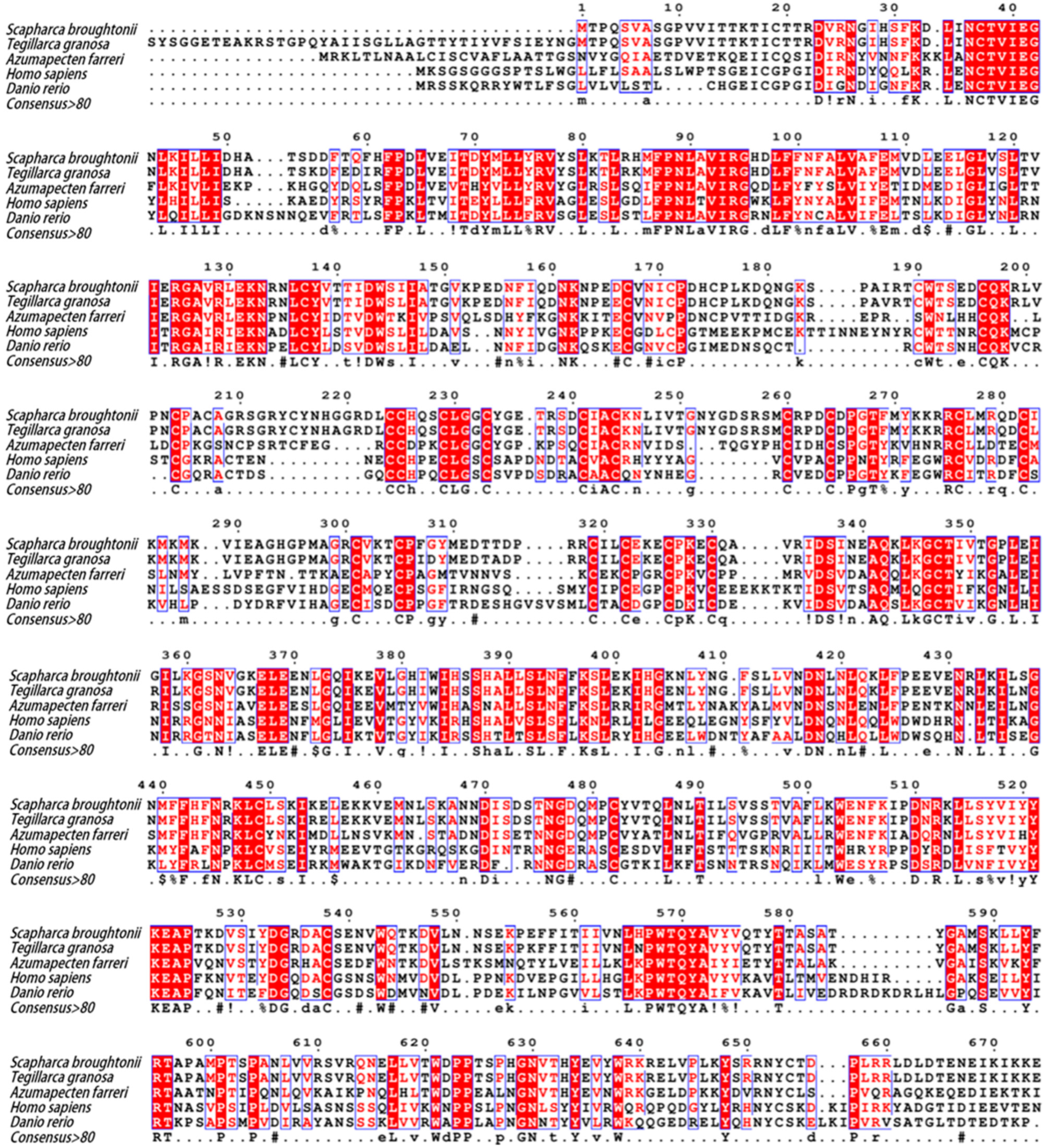
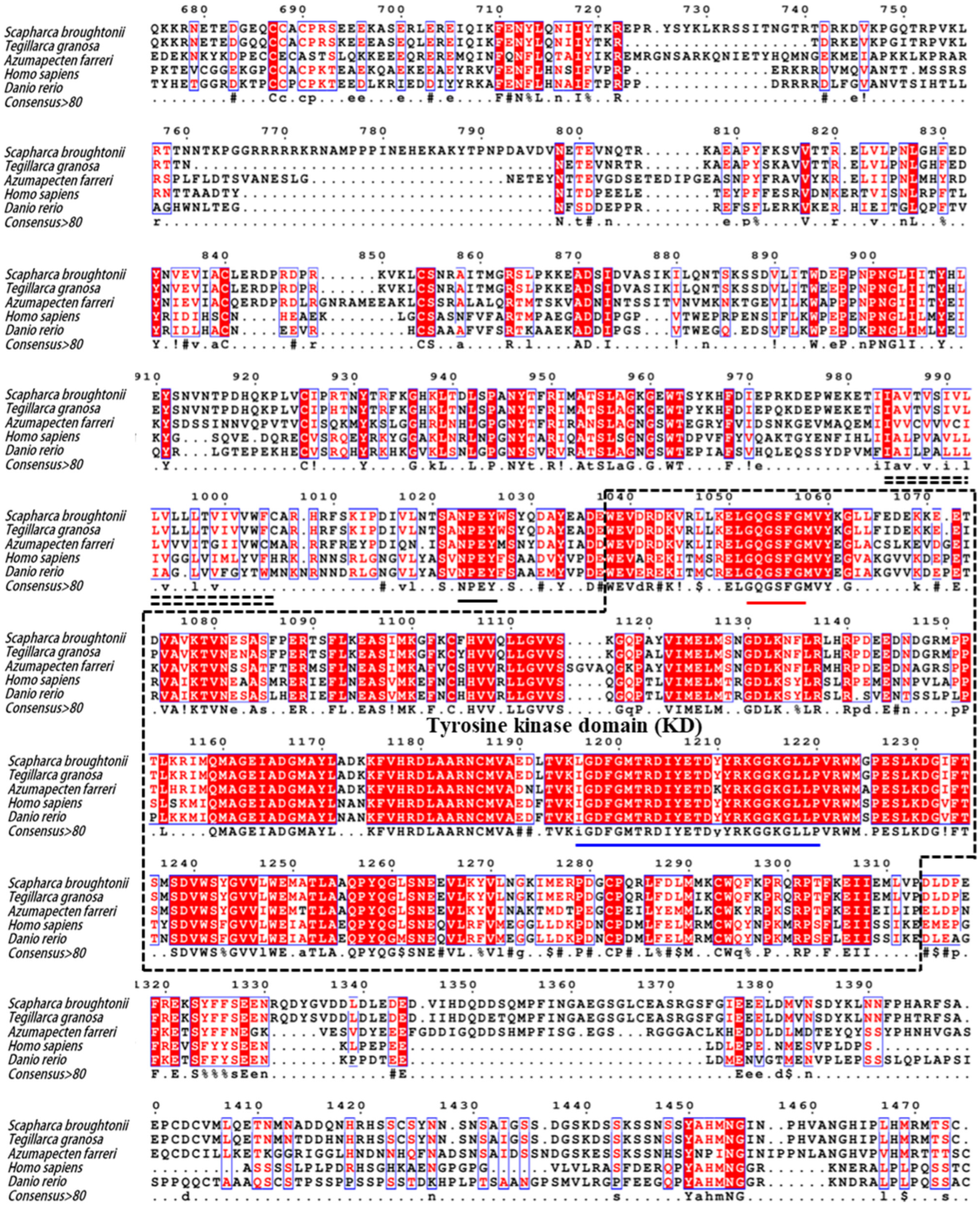
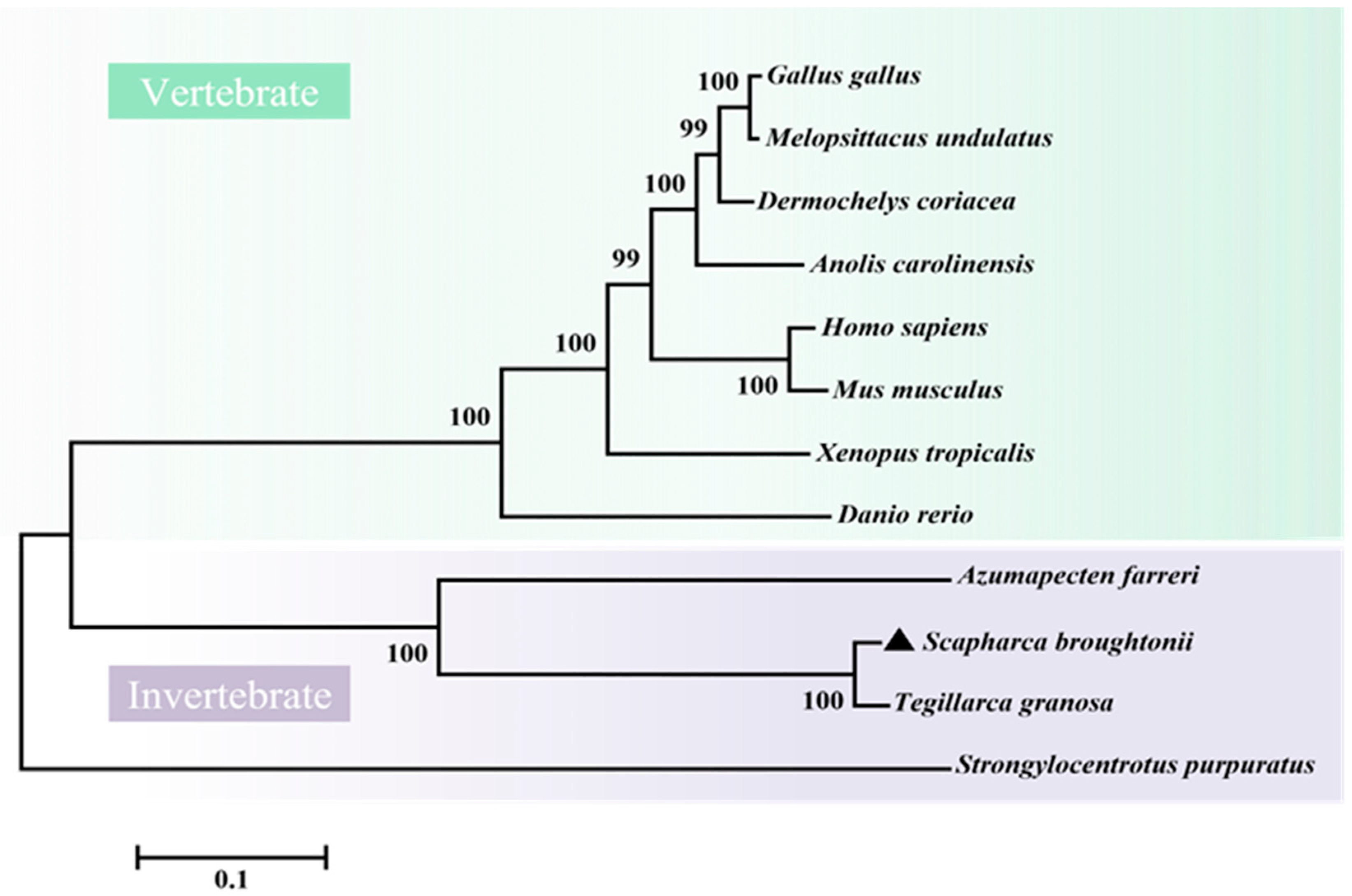
2.2. Molecular Characterization of SbIGF-1R
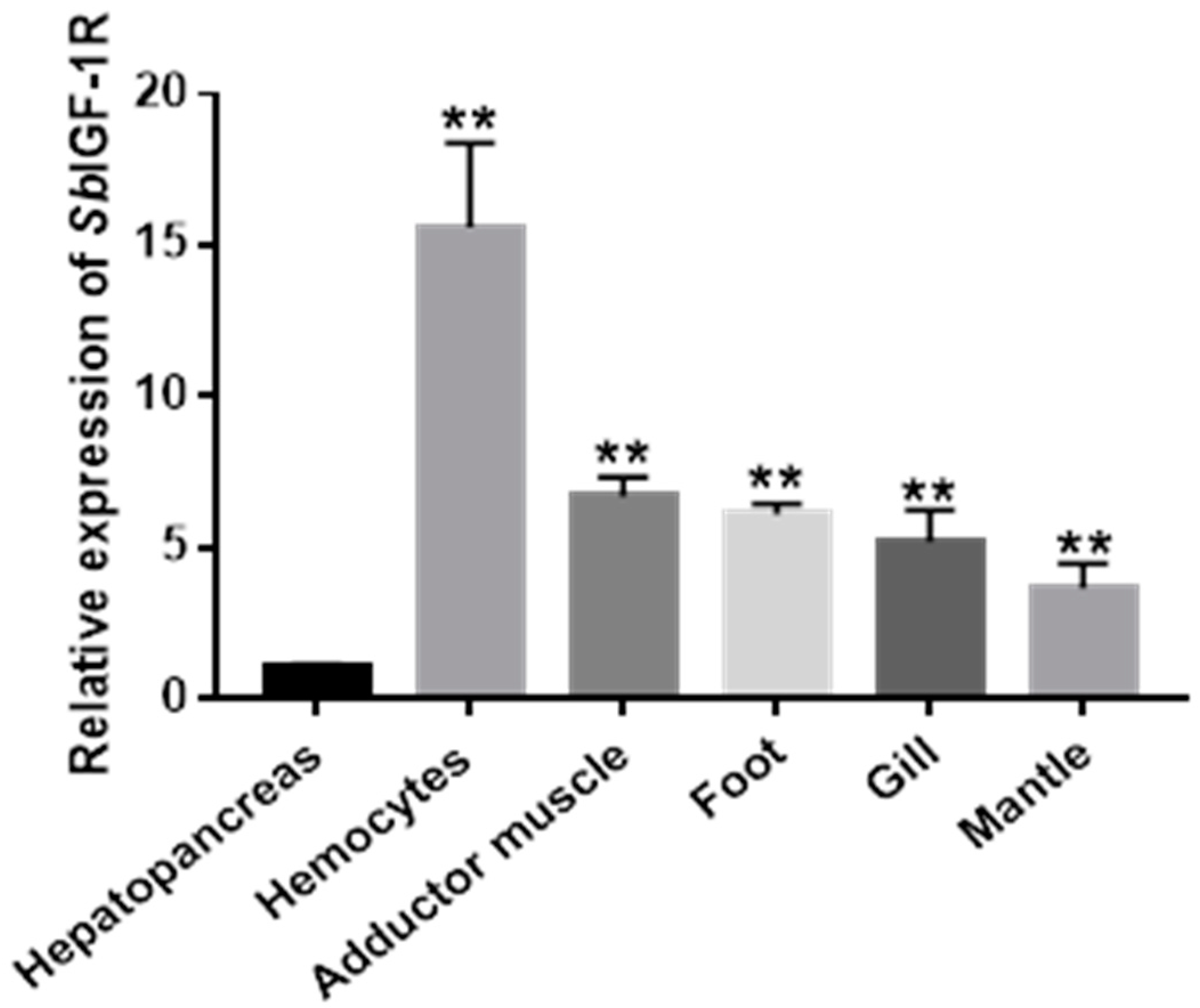
2.3. The mRNA Expression Pattern of SbIGF-1R in Various Tissues
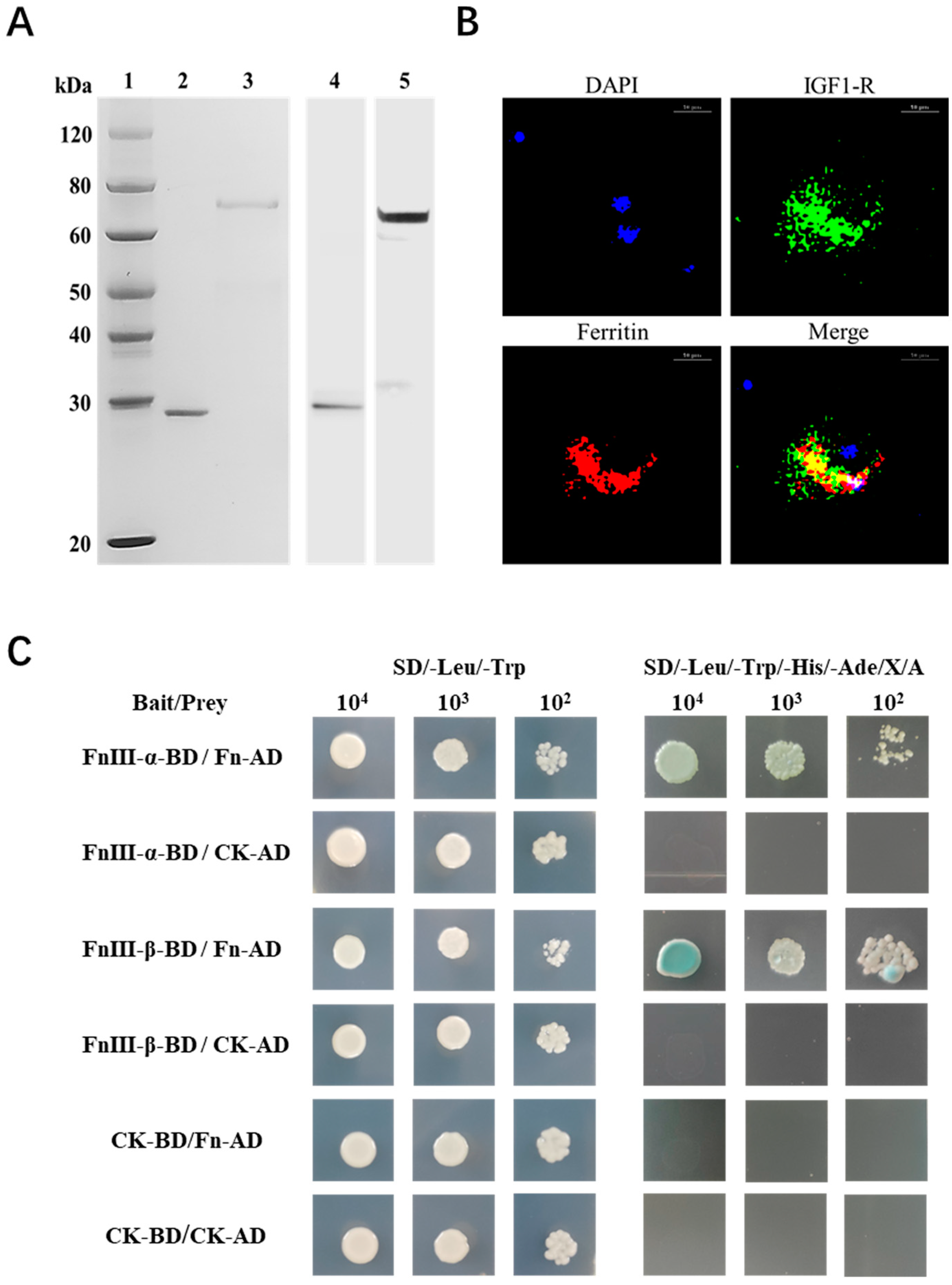
2.4. The Interaction Mode between SbIGF-1R and SbFn
3. Discussion
4. Materials and Methods
4.1. Animals and Sample Collection
4.2. His-Fusion Proteins and His Pull-Down Assays
4.3. Clone and Bioinformatic Analyses of SbIGF-1R
4.4. qPCR Assay of SbIGF-1R Expression
4.5. Preparation of Polyclonal Antibody
4.6. Western Blotting
4.7. Co-Locaalization Assay
4.8. Yeast Two-Hybrid Assay
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lieu, P.T.; Heiskala, M.; Peterson, P.A.; Yang, Y. The roles of iron in health and disease. Mol. Asp. Med. 2001, 22, 1–87. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T.; Nemeth, E. Iron homeostasis in host defence and inflammation. Nat. Rev. Immunol. 2015, 15, 500–510. [Google Scholar] [CrossRef]
- Schaible, U.E.; Kaufmann, S.H. Iron and microbial infection. Nat. Rev. Microbiol. 2004, 2, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Cairo, G.; Tacchini, L.; Pogliaghi, G.; Anzon, E.; Tomasi, A.; Bernelli-Zazzera, A. Induction of Ferritin Synthesis by Oxidative Stress: Transcriptional and post-transcriptional regulation by expansion of the “free” iron pool. J. Biol. Chem. 1995, 270, 700–703. [Google Scholar] [CrossRef]
- Abboud, S.; Haile, D.J. A novel mammalian iron-regulated protein involved in intracellular iron metabolism. J. Biol. Chem. 2000, 275, 19906–19912. [Google Scholar] [CrossRef] [PubMed]
- Donovan, A.; Brownlie, A.; Zhou, Y.; Shepard, J.; Pratt, S.J.; Moynihan, J.; Paw, B.H.; Drejer, A.; Barut, B.; Zapata, A. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter. Nature 2000, 403, 776–781. [Google Scholar] [CrossRef]
- Harrison, P.M.; Arosio, P. The ferritins: Molecular properties, iron storage function and cellular regulation. Biochim. Biophys. Acta (BBA) Bioenerg. 1996, 1275, 161–203. [Google Scholar] [CrossRef]
- Li, L.; Fang, C.J.; Ryan, J.C.; Niemi, E.C.; Seaman, W.E. Binding and uptake of H-ferritin are mediated by human transferrin receptor-1. Proc. Natl. Acad. Sci. USA 2010, 107, 3505–3510. [Google Scholar] [CrossRef]
- Zhang, N.; Yu, X.; Xie, J.; Xu, H. New insights into the role of ferritin in iron homeostasis and neurodegenerative diseases. Mol. Neurobiol. 2021, 58, 2812–2823. [Google Scholar] [CrossRef]
- Li, J.Y.; Paragas, N.; Ned, R.M.; Qiu, A.; Viltard, M.; Leete, T.; Drexler, I.R.; Chen, X.; Sanna-Cherchi, S.; Mohammed, F. Scara5 Is a Ferritin Receptor Mediating Non-Transferrin Iron Delivery. Dev. Cell 2009, 16, 46. [Google Scholar] [CrossRef]
- Chen, T.T.; Li, L.; Chung, D.H.; Allen, C.D.; Torti, S.V.; Torti, F.M.; Cyster, J.G.; Chen, C.-Y.; Brodsky, F.M.; Niemi, E.C. TIM-2 is expressed on B cells and in liver and kidney and is a receptor for H-ferritin endocytosis. J. Exp. Med. 2005, 202, 955–965. [Google Scholar] [CrossRef]
- Bai, L.; Xie, T.; Hu, Q.; Deng, C.; Zheng, R.; Chen, W. Genome-wide comparison of ferritin family from Archaea, Bacteria, Eukarya, and Viruses: Its distribution, characteristic motif, and phylogenetic relationship. Sci. Nat. 2015, 102, 1–10. [Google Scholar] [CrossRef]
- Nichol, H.; Law, J.H.; Winzerling, J.J. Iron metabolism in insects. Annu. Rev. Entomol. 2002, 47, 535–559. [Google Scholar] [CrossRef]
- Tang, X.; Zhou, B. Ferritin is the key to dietary iron absorption and tissue iron detoxification in Drosophila melanogaster. FASEB J. 2013, 27, 288–298. [Google Scholar] [CrossRef]
- Winzerling, J.J.; Nez, P.; Porath, J.; Law, J.H. Rapid and efficient isolation of transferrin and ferritin from Manduca sexta. Insect Biochem. Mol. Biol. 1995, 25, 217–224. [Google Scholar] [CrossRef]
- Ong, S.T.; Ho, J.Z.S.; Ho, B.; Ding, J.L. Iron-withholding strategy in innate immunity. Immunobiology 2006, 211, 295–314. [Google Scholar] [CrossRef] [PubMed]
- Xin, L.; Huang, B.; Zhang, H.; Li, C.; Bai, C.; Wang, C. OsHV-1 infection leads to mollusc tissue lesion and iron redistribution, revealing a strategy of iron limitation against pathogen. Metallomics 2019, 11, 822–832. [Google Scholar] [CrossRef]
- Huang, B.; Zhang, X.; Liu, Q.; Bai, C.; Li, C.; Wang, C.; Xin, L. Iron Regulatory Protein 1 Inhibits Ferritin Translation Responding to OsHV-1 Infection in Ark Clams, Scapharca broughtonii. Cells 2022, 11, 982. [Google Scholar] [CrossRef] [PubMed]
- Dunkov, B.; Georgieva, T. Insect iron binding proteins: Insights from the genomes. Insect Biochem. Mol. Biol. 2006, 36, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Zhou, B. Iron homeostasis in insects: Insights from Drosophila studies. IUBMB Life 2013, 65, 863–872. [Google Scholar] [CrossRef]
- Choi, Y.H.; Kim, E.Y.; Nam, T.J. Involvement of insulin-like growth factor in intraspecific variation in growth of Pacific oyster Crassostrea gigas during winter. Fish. Sci. 2018, 84, 1017–1024. [Google Scholar] [CrossRef]
- Chandhini, S.; Trumboo, B.; Jose, S.; Varghese, T.; Rajesh, M.; Kumar, V.R. Insulin-like growth factor signalling and its significance as a biomarker in fish and shellfish research. Fish Physiol. Biochem. 2021, 47, 1011–1031. [Google Scholar] [CrossRef]
- Choi, Y.H.; Nam, T.J. Toxicity of cryoprotective agents and signaling of insulin-like growth factor in hen clam (Mactra chinensis) embryos. Cryo. Lett. 2015, 36, 158–164. [Google Scholar]
- Choi, Y.H.; Nam, T.J. Influence of the toxicity of cryoprotective agents on the involvement of insulin-like growth factor-I receptor in surf clam (Spisula sachalinensis) larvae. Cryo. Lett. 2014, 35, 537–543. [Google Scholar]
- Feng, L.; Li, X.; Yu, Q.; Ning, X.; Dou, J.; Zou, J.; Zhang, L.; Wang, S.; Hu, X.; Bao, Z. A scallop IGF binding protein gene: Molecular characterization and association of variants with growth traits. PLoS ONE 2014, 9, e89039. [Google Scholar] [CrossRef] [PubMed]
- Gricourt, L.; Bonnec, G.; Boujard, D.; Mathieu, M.; Kellner, K. Insulin-like system and growth regulation in the Pacific oyster Crassostrea gigas: hrIGF-1 effect on protein synthesis of mantle edge cells and expression of an homologous insulin receptor-related receptor. Gen. Comp. Endocrinol. 2003, 134, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Canesi, L.; Ciacci, C.; Betti, M.; Malatesta, M.; Gazzanelli, G.; Gallo, G. Growth factors stimulate the activity of key glycolytic enzymes in isolated digestive gland cells from mussels (Mytilus galloprovincialis Lam.) through tyrosine kinase mediated signal transduction. Gen. Comp. Endocrinol. 1999, 116, 241–248. [Google Scholar] [CrossRef]
- Girnita, L.; Worrall, C.; Takahashi, S.I.; Seregard, S.; Girnita, A. Something old, something new and something borrowed: Emerging paradigm of insulin-like growth factor type 1 receptor (IGF-1R) signaling regulation. Cell. Mol. Life Sci. 2014, 71, 2403–2427. [Google Scholar] [CrossRef]
- Ullrich, A.; Schlessinger, J. Signal transduction by receptors with tyrosine kinase activity. Cell 1990, 61, 203–212. [Google Scholar] [CrossRef]
- LeRoith, D.; Holly, J.M.; Forbes, B. The insulin-like growth factors: Ligands, binding proteins and receptors. Mol. Metab. 2021, 52, 101245. [Google Scholar] [CrossRef]
- Martins, A.S.; Ordóñez, J.L.; Amaral, A.T.; Prins, F.; Floris, G.; Debiec-Rychter, M.; Hogendoorn, P.C.; de Alava, E. IGF1R signaling in Ewing sarcoma is shaped by clathrin-/caveolin-dependent endocytosis. PLoS ONE 2011, 6, e19846. [Google Scholar] [CrossRef]
- Wang, W.; Knovich, M.A.; Coffman, L.G.; Torti, F.M.; Torti, S.V. Serum ferritin: Past, present and future. Biochim. Biophys. Acta (BBA) Gen. Subj. 2010, 1800, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Hentze, M.W.; Muckenthaler, M.U.; Galy, B.; Camaschella, C. Two to tango: Regulation of Mammalian iron metabolism. Cell 2010, 142, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Truman-Rosentsvit, M.; Berenbaum, D.; Spektor, L.; Cohen, L.A.; Belizowsky-Moshe, S.; Lifshitz, L.; Ma, J.; Li, W.; Kesselman, E.; Abutbul-Ionita, I. Ferritin is secreted via 2 distinct nonclassical vesicular pathways. Blood J. Am. Soc. Hematol. 2018, 131, 342–352. [Google Scholar] [CrossRef]
- Kavran, J.M.; McCabe, J.M.; Byrne, P.O.; Connacher, M.K.; Wang, Z.; Ramek, A.; Sarabipour, S.; Shan, Y.; Shaw, D.E.; Hristova, K. How IGF-1 activates its receptor. Elife 2014, 3, e03772. [Google Scholar] [CrossRef]
- Koide, A.; Bailey, C.W.; Huang, X.; Koide, S. The fibronectin type III domain as a scaffold for novel binding proteins. J. Mol. Biol. 1998, 284, 1141–1151. [Google Scholar] [CrossRef]
- Yang, F.; West Jr, A.P.; Allendorph, G.P.; Choe, S.; Bjorkman, P.J. Neogenin interacts with hemojuvelin through its two membrane-proximal fibronectin type III domains. Biochemistry 2008, 47, 4237–4245. [Google Scholar] [CrossRef]
- Hecht, O.; Dingley, A.J.; Schwanter, A.; Özbek, S.; Rose-John, S.; Grötzinger, J. The solution structure of the membrane-proximal cytokine receptor domain of the human interleukin-6 receptor. Biol. Chem. 2006, 387, 1255–1259. [Google Scholar] [CrossRef] [PubMed]
- Miknis, Z.J.; Magracheva, E.; Li, W.; Zdanov, A.; Kotenko, S.V.; Wlodawer, A. Crystal structure of human interferon-λ1 in complex with its high-affinity receptor interferon-λR1. J. Mol. Biol. 2010, 404, 650–664. [Google Scholar] [CrossRef]
- Li, J.; Choi, E.; Yu, H.; Bai, X.-c. Structural basis of the activation of type 1 insulin-like growth factor receptor. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef]
- Ohno, H.; Stewart, J.; Fournier, M.C.; Bosshart, H.; Rhee, I.; Miyatake, S.; Saito, T.; Gallusser, A.; Kirchhausen, T.; Bonifacino, J.S. Interaction of tyrosine-based sorting signals with clathrin-associated proteins. Science 1995, 269, 1872–1875. [Google Scholar] [CrossRef]
- Bonifacino, J.S.; Traub, L.M. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 2003, 72, 395–447. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Keyel, P.A.; Hawryluk, M.J.; Agostinelli, N.R.; Watkins, S.C.; Traub, L.M. Disabled-2 exhibits the properties of a cargo-selective endocytic clathrin adaptor. EMBO J. 2002, 21, 4915–4926. [Google Scholar] [CrossRef]
- Morris, S.M.; Cooper, J.A. Disabled-2 colocalizes with the LDLR in clathrin-coated pits and interacts with AP-2. Traffic 2001, 2, 111–123. [Google Scholar] [CrossRef]
- Hobden, D.J. Aspects of iron metabolism in a freshwater mussel. Can. J. Zool. 1970, 48, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.P.; Hamza, I. Macrophages and iron metabolism. Immunity 2016, 44, 492–504. [Google Scholar] [CrossRef]
- Xin, L.; Huang, B.; Bai, C.; Wang, C. Validation of housekeeping genes for quantitative mRNA expression analysis in OsHV-1 infected ark clam, Scapharca broughtonii. J. Invertebr. Pathol. 2018, 155, 44–51. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
| Primer Name | Primer Sequence (5′-3′) |
|---|---|
| Gene clone primer | |
| SbFn-F | CTCCGAAACTCCGCCATATTCTCAC |
| SbFn-R | CATGGAATTTATTTAGAAAAAGGACTG |
| SbIGF-1R-F | ATGACACCACAAAGTGTGGCATCAG |
| SbIGF-1R-R | TCAGCATGATGTCATCCTCATGTGT |
| Real-time quantitative PCR primers | |
| qSbIGF-1R-F | GAAGATCACTGCCGAAGAAGGA |
| qSbIGF-1R-R | CGTGGAATGCATACAAGAGGTTTC |
| qSbRL15-F | AGACCAGACAAAGCCAGAAGAC |
| qSbRL15-R | GCTGAAGTAAGTCCACGCATT |
| Vector construction primers | (The underline means restriction enzyme site) |
| SbFn-BamH I | CGCGGATCCATGGCTCAAACACAACCAAG |
| SbFn-Xho I | CCGCTCGAGTATATGTAGATAAAAGCCTAA |
| SbIGF-1R-FNIII-BamH I | CGCGGATCCTATGTAACTCAACTCAACCTGACTA |
| SbIGF-1R-FNIII-Xho I | CCGCTCGAGTCTAAAAATGTTTATATGATGTCCATTCT |
| FnⅢ-α-BD-BamH I | CGCGGATCCGTTATGTAACTCAACTCAACCTGACTA |
| FnⅢ-α-BD-Not I | AAGGAAAAAAGCGGCCGCTTTACGCCAATACACTTCGTAATGA |
| FnⅢ-β-BD-BamH I | CGCGGATCCGTGCAGTCGATGTGAATGAAACTGAGG |
| FnⅢ-β-BD-Not I | AAGGAAAAAAGCGGCCGCAAAATGTTTATATGATGTCCATTCT |
| SbFn-AD-EcoR I | CCGGAATTCGCTCAAACACAACCAAGACAAAACT |
| SbFn-AD-BamH I | CGCGGATCCCACTGCTCATGGTTTCCTTGTCATAC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, B.; Liu, Q.; Bai, C.; Li, C.; Wang, C.; Xin, L. A Putative Receptor for Ferritin in Mollusks: Characterization of the Insulin-like Growth Factor Type 1 Receptor. Int. J. Mol. Sci. 2023, 24, 6175. https://doi.org/10.3390/ijms24076175
Huang B, Liu Q, Bai C, Li C, Wang C, Xin L. A Putative Receptor for Ferritin in Mollusks: Characterization of the Insulin-like Growth Factor Type 1 Receptor. International Journal of Molecular Sciences. 2023; 24(7):6175. https://doi.org/10.3390/ijms24076175
Chicago/Turabian StyleHuang, Bowen, Qin Liu, Changming Bai, Chen Li, Chongming Wang, and Lusheng Xin. 2023. "A Putative Receptor for Ferritin in Mollusks: Characterization of the Insulin-like Growth Factor Type 1 Receptor" International Journal of Molecular Sciences 24, no. 7: 6175. https://doi.org/10.3390/ijms24076175
APA StyleHuang, B., Liu, Q., Bai, C., Li, C., Wang, C., & Xin, L. (2023). A Putative Receptor for Ferritin in Mollusks: Characterization of the Insulin-like Growth Factor Type 1 Receptor. International Journal of Molecular Sciences, 24(7), 6175. https://doi.org/10.3390/ijms24076175






