The Effects of Momordica charantia on Type 2 Diabetes Mellitus and Alzheimer’s Disease
Abstract
1. Introduction
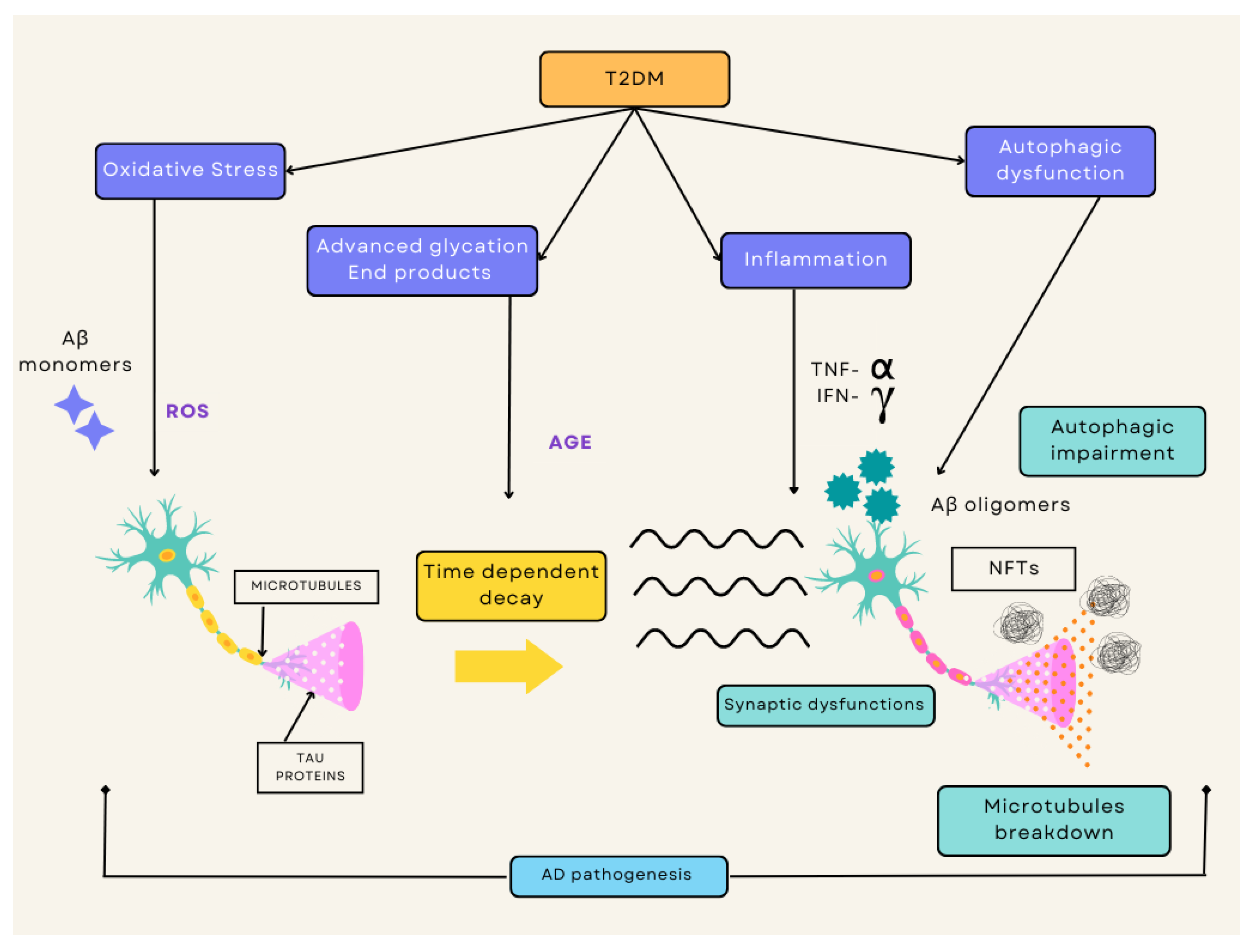
2. Pathophysiology of T2DM and AD
2.1. Type 2 Diabetes Mellitus
2.2. Alzheimer’s Disease
3. Common Link between T2DM and AD
3.1. Insulin Resistance
3.2. Chronic Inflammation
3.3. Oxidative Stress
4. The Potential of Plant-Based Medicine
4.1. Impact of Plant-Based Medicine
4.2. Utilization of Bitter Melon as Plant-Based Medicine
5. The Profile of Bitter Melon
5.1. Bitter Melon Description
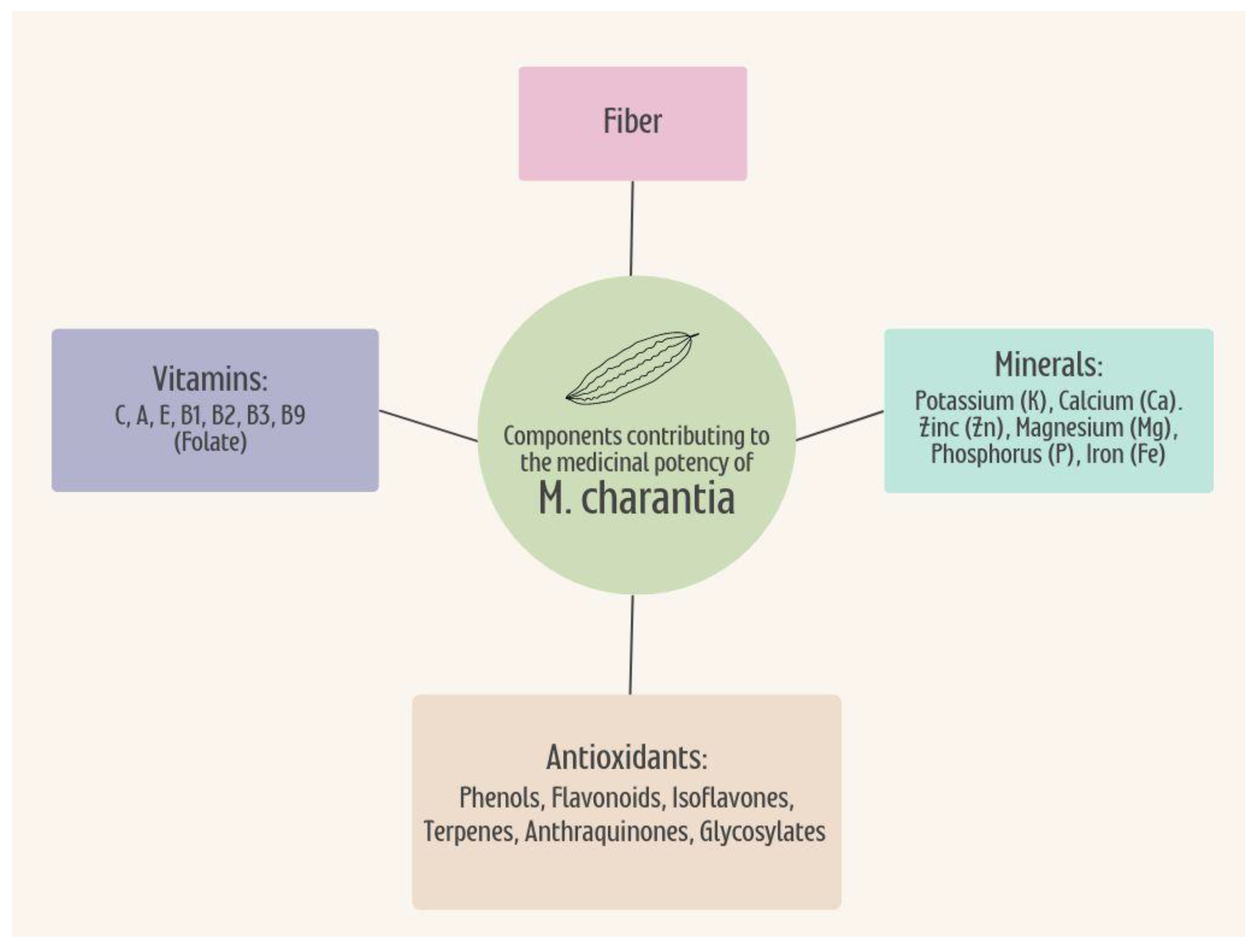
5.2. Nutrient Profile
5.3. Phytochemistry
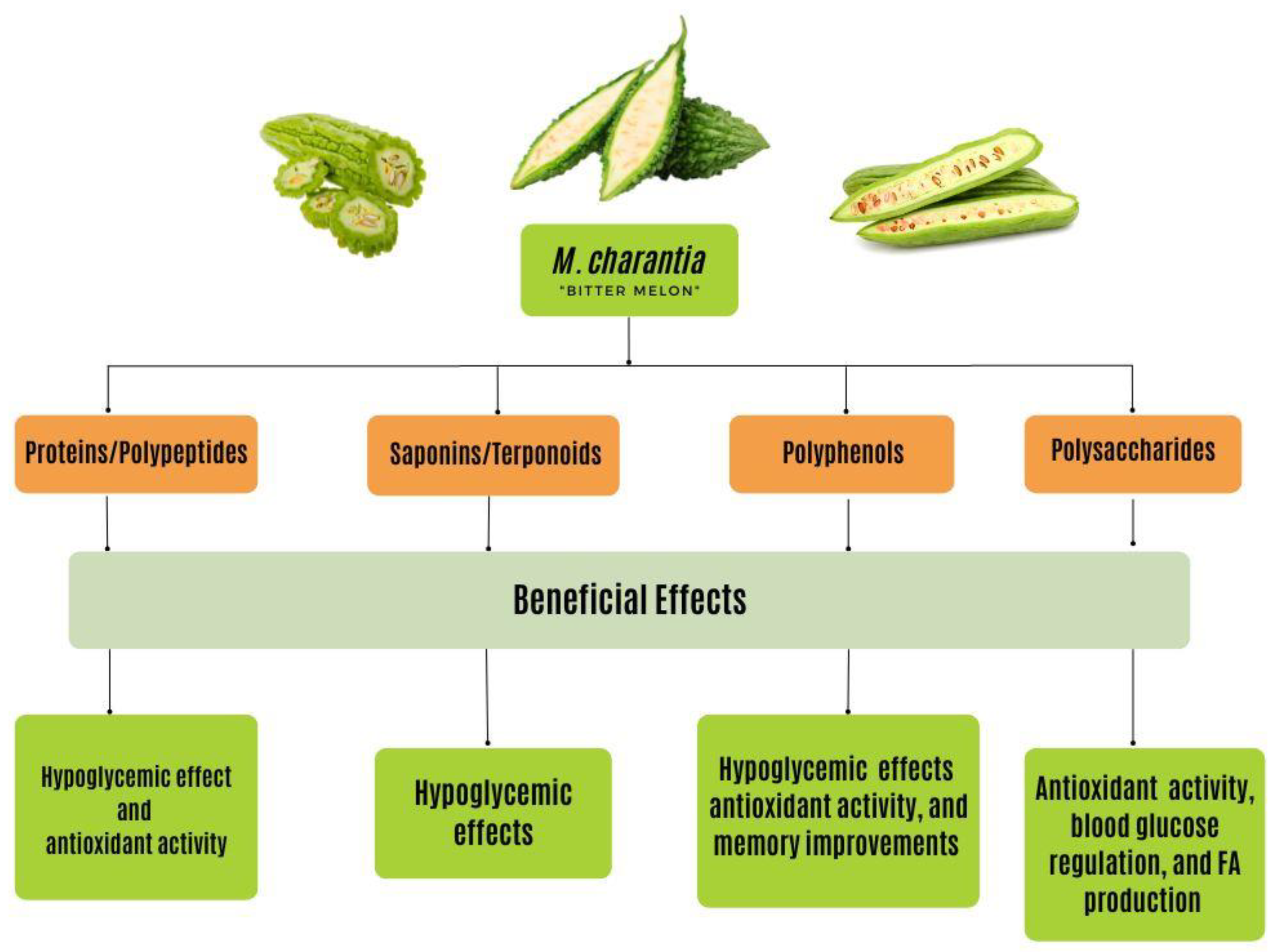
6. The Bioactive Compounds of Bitter Melon
6.1. Polysaccharides
6.2. Proteins and Peptides
6.2.1. Polypeptide-P
6.2.2. Peroxidase
6.3. Saponins and Terpenoids
Charantin
6.4. Flavonoids and Phenolic Compounds
6.4.1. Quercetin
6.4.2. Rutin
6.4.3. Kaempferol
6.4.4. Isorhamnetin
6.5. Other Components
7. Incorporating Bitter Melon into the Diet
7.1. Defining Medical Nutrition Therapy
7.2. Impact of Bitter Melon as Medical Nutrition Therapy
7.3. Incorporating Bitter Melon into the Diet
8. Summary
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Marras, C.; Beck, J.C.; Bower, J.H.; Roberts, E.; Ritz, B.; Ross, G.W.; Abbott, R.D.; Savica, R.; Van Den Eeden, S.K.; Willis, A.W.; et al. Prevalence of parkinson’s disease across North America. NPJ Parkinson’s Dis. 2018, 4, 21. [Google Scholar] [CrossRef]
- Leung, L.; Birtwhistle, R.; Kotecha, J.; Hannah, S.; Cuthbertson, S. Anti-diabetic and hypoglycaemic effects of Momordica charantia (bitter melon): A mini review. Br. J. Nutr. 2009, 102, 1703–1708. [Google Scholar] [CrossRef]
- Joseph, B.; Jini, D. Antidiabetic effects of Momordica charantia (bitter melon) and its medicinal potency. Asian Pac. J. Trop. Dis. 2013, 3, 93–102. [Google Scholar] [CrossRef]
- Forbes, J.M.; Cooper, M.E. Mechanisms of diabetic complications. Physiol. Rev. 2013, 93, 137–188. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. What Is Alzheimer’s Disease? Centers for Disease Control and Prevention. 2020. Available online: https://www.cdc.gov/aging/aginginfo/alzheimers.htm (accessed on 16 January 2023).
- WebMD. (n.d.). WebMD Diabetes Center: Types, Causes, Symptoms, Tests, and Treatments. WebMD. Available online: https://www.webmd.com/diabetes/peripheral-neuropathy-risk-factors-symptoms#1 (accessed on 16 January 2023).
- Akter, K.; Lanza, E.A.; Martin, S.A.; Myronyuk, N.; Rua, M.; Raffa, R.B. Diabetes mellitus and Alzheimer’s disease: Shared pathology and treatment? Br. J. Clin. Pharmacol. 2011, 71, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Mayo Foundation for Medical Education and Research. Alzheimer’s Disease. Mayo Clinic. 2022. Available online: https://www.mayoclinic.org/diseases-conditions/alzheimers-disease/symptomscauses/syc-20350447 (accessed on 5 November 2022).
- U.S. Department of Health and Human Services. (n.d.). Alzheimer’s Disease Fact Sheet. National Institute on Aging. Available online: https://www.nia.nih.gov/health/alzheimers-disease-fact-sheet (accessed on 16 January 2023).
- Mohammed, S.A.; Yaqub, A.G.; Sanda, K.A.; Nicholas, A.O.; Arastus, W.; Muhammad, M. Review on diabetes, synthetic drugs and glycemic effects of medicinal plants. J. Med. Plants Res. 2013, 7, 2628–2637. [Google Scholar]
- Ma, T.; Tan, M.-S.; Yu, J.-T.; Tan, L. Resveratrol as a therapeutic agent for Alzheimer’s disease. BioMed Res. Int. 2014, 2014, 350516. [Google Scholar] [CrossRef]
- Walker, J.; Harrison, F. Shared neuropathological characteristics of obesity, type 2 diabetes and Alzheimer’s disease: Impacts on cognitive decline. Nutrients 2015, 7, 7332–7357. [Google Scholar] [CrossRef] [PubMed]
- De la Monte, S.M.; Wands, J.R. Alzheimer’s disease is type 3 diabetes—Evidence reviewed. J. Diabetes Sci. Technol. 2008, 2, 1101–1113. [Google Scholar] [CrossRef]
- Venkata Raman, B.; Vamsikrishna, A.; Rao, B.; Pudha, M.; Saradhi, M.V.; Rao, B. Plants with antidiabetic activities and their medicinal values. Int. Res. J. Pharm. 2012, 3, 11–15. [Google Scholar]
- Gothai, S.; Ganesan, P.; Park, S.-Y.; Fakurazi, S.; Choi, D.-K.; Arulselvan, P. Natural phyto-bioactive compounds for the treatment of type 2 diabetes: Inflammation as a target. Nutrients 2016, 8, 461. [Google Scholar] [CrossRef] [PubMed]
- Cefalu, W.; Ye, J.; Wang, Z. Efficacy of dietary supplementation with botanicals on carbohydrate metabolism in humans. Endocr. Metab. Immune Disord.-Drug Targets 2008, 8, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Cousens, G.; Rainoshek, D. There Is a Cure for Diabetes: The Tree of Life 21-Day+ Program; North Atlantic Books: Berkeley, CA, USA, 2008. [Google Scholar]
- McAnany, B.; Martirosyan, D. The effects of bioactive compounds on Alzheimer’s disease and mild cognitive impairment. Funct. Foods Health Dis. 2016, 6, 329. [Google Scholar] [CrossRef]
- Mayo Foundation for Medical Education and Research. Type 2 Diabetes; Mayo Clinic: Scottsdale, AZ, USA, 2021; Available online: https://www.mayoclinic.org/diseases-conditions/type-2-diabetes/symptoms-causes/syc-20351193 (accessed on 5 November 2022).
- Mahler, R.J.; Adler, M.L. Type 2 diabetes mellitus: Update on diagnosis, pathophysiology, and treatment. J. Clin. Endocrinol. Metab. 1999, 84, 1165–1171. [Google Scholar] [CrossRef]
- Sen, S.; Chakraborty, R.; De, B. Pancreatic hormones and control of blood glucose: A glance. In Diabetes Mellitus in 21st Century; Springer: Singapore, 2016; pp. 1–12. [Google Scholar] [CrossRef]
- Gelding, S.V.; Coldham, N.; Niththyananthan, R.; Anyaoku, V.; Johnston, D.G. Insulin resistance with respect to lipolysis in non-diabetic relatives of European patients with type 2 diabetes. Diabet. Med. 1995, 12, 66–73. [Google Scholar] [CrossRef]
- Charles, M.A.; Eschwe’ge, E.; Thibult, N.; Claude, J.-R.; Warnet, J.-M.; Rosselin, G.E.; Girard, J.; Balkau, B. The role of non-esterified fatty acids in the deterioration of glucose tolerance in Caucasian subjects: Results of the Paris Prospective Study. Diabetologia 1997, 40, 1101–1106. [Google Scholar] [CrossRef]
- Kitamura, T. The role of FOXO1 in β-cell failure and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2013, 9, 615–623. [Google Scholar] [CrossRef]
- Fischbach, S. The role of TGF-β signaling in β-cell dysfunction and type 2 diabetes: A Review. J. Cytol. Histol. 2014, 5, 1000282. [Google Scholar] [CrossRef]
- Li, R.; Zhang, Y.; Rasool, S.; Geetha, T.; Babu, J.R. Effects and Underlying Mechanisms of Bioactive Compounds on Type 2 Diabetes Mellitus and Alzheimer’s Disease. Oxidative Med. Cell. Longev. 2019, 2019, 8165707. [Google Scholar] [CrossRef]
- Granzotto, A.; Zatta, P. Resveratrol and Alzheimer’s disease: Message in a bottle on red wine and cognition. Front. Aging Neurosci. 2014, 6, 95. [Google Scholar] [CrossRef]
- Desai, G.S.; Zheng, C.; Geetha, T.; Mathews, S.T.; White, B.D.; Huggins, K.W.; Zizza, C.A.; Broderick, T.L.; Babu, J.R. The pancreas-brain axis: Insight into disrupted mechanisms associating type 2 diabetes and Alzheimer’s disease. J. Alzheimer’s Dis. 2014, 42, 347–356. [Google Scholar] [CrossRef]
- Querfurth, H.W.; LaFerla, F.M. Alzheimer’s disease. New Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef]
- Bharadwaj, P.R.; Dubey, A.K.; Masters, C.L.; Martins, R.N.; Macreadie, I.G. AB aggregation and possible implications in Alzheimer’s disease pathogenesis. J. Cell. Mol. Med. 2009, 13, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Finder, V.H.; Glockshuber, R. Amyloid-β aggregation. Neurodegener. Dis. 2007, 4, 13–27. [Google Scholar] [CrossRef] [PubMed]
- De la Monte, S.M.; Wands, J.R. Review of insulin and insulin-like growth factor expression, signaling, and malfunction in the central nervous system: Relevance to Alzheimer’s disease. J. Alzheimers Dis. 2005, 7, 45–61. [Google Scholar] [CrossRef] [PubMed]
- Steen, E.; Terry, B.M.; Rivera, E.J.; Cannon, J.L.; Neely, T.R.; Tavares, R.; Xu, X.J.; Wands, J.R.; de la Monte, S.M. Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer’s disease—Is this type 3 diabetes? J. Alzheimers Dis. 2005, 7, 63–80. [Google Scholar] [CrossRef] [PubMed]
- De la Monte, S.M.; Wands, J.R. Molecular indices of oxidative stress and mitochondrial dysfunction occur early and often progress with severity of Alzheimer’s disease. J. Alzheimers Dis. 2006, 9, 167–181. [Google Scholar] [CrossRef]
- Moreira, P.I.; Santos, M.S.; Seiça, R.; Oliveira, C.R. Brain mitochondrial dysfunction as a link between Alzheimer’s disease and diabetes. J. Neurol. Sci. 2007, 257, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, S. The brain insulin signal transduction system and sporadic (type II) Alzheimer disease: An update. J. Neural Transm. 2002, 109, 341–360. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A. The calpains in aging and aging-related diseases. Ageing Res. Rev. 2003, 2, 407–418. [Google Scholar] [CrossRef]
- Rivera, E.J.; Goldin, A.; Fulmer, N.; Tavares, R.; Wands, J.R.; de la Monte, S.M. Insulin and insulin-like growth factor expression and function deteriorate with progression of Alzheimer’s disease: Link to brain reductions in acetylcholine. J. Alzheimers Dis. 2005, 8, 247–268. [Google Scholar] [CrossRef]
- Revill, P.; Moral, M.A.; Prous, J.R. Impaired insulin signaling and the pathogenesis of Alzheimer’s disease. Drugs Today 2006, 42, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Iwangoff, P.; Armbruster, R.; Enz, A.; Meier-Ruge, W. Glycolytic enzymes from human autoptic brain cortex: Normal aged and demented cases. Mech. Ageing Dev. 1980, 14, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Sims, N.R.; Bowen, D.M.; Smith, C.C.; Flack, R.H.; Davison, A.N.; Snowden, J.S.; Neary, D. Glucose metabolism and acetylcholine synthesis in relation to neuronal activity in Alzheimer’s disease. Lancet 1980, 315, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, S. Causes and consequences of disturbances of cerebral glucose metabolism in sporadic Alzheimer disease: Therapeutic implications. Adv. Exp. Med. Biol. 2004, 541, 135–152. [Google Scholar] [PubMed]
- Li, X.; Leng, S.; Song, D. Link between type 2 diabetes and Alzheimer’s disease: From epidemiology to mechanism and treatment. Clin. Interv. Aging 2015, 10, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Hölscher, C. Common pathological processes in alzheimer disease and type 2 diabetes: A Review. Brain Res. Rev. 2007, 56, 384–402. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Braak, E. Staging of alzheimer-related cortical destruction. Int. Psychogeriatr. 1997, 9, 257–261. [Google Scholar] [CrossRef]
- Phiel, C.J.; Wilson, C.A.; Lee, V.M.-Y.; Klein, P.S. GSK-3α regulates production of Alzheimer’s disease amyloid-β peptides. Nature 2003, 423, 435–439. [Google Scholar] [CrossRef]
- Bomfim, T.R.; Forny-Germano, L.; Sathler, L.B.; Brito-Moreira, J.; Houzel, J.-C.; Decker, H.; Silverman, M.A.; Kazi, H.; Melo, H.M.; McClean, P.L.; et al. An anti-diabetes agent protects the mouse brain from defective insulin signaling caused by Alzheimer’s disease–associated AΒ oligomers. J. Clin. Investig. 2012, 122, 1339–1353. [Google Scholar] [CrossRef]
- Kurochkin, I.V.; Goto, S. Alzheimer’s β-amyloid peptide specifically interacts with and is degraded by insulin degrading enzyme. FEBS Lett. 1994, 345, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Mirza, S.; Hossain, M.; Mathews, C.; Martinez, P.; Pino, P.; Gay, J.L.; Rentfro, A.; McCormick, J.B.; Fisher-Hoch, S.P. Type 2-diabetes is associated with elevated levels of TNF-alpha, IL-6 and adiponectin and low levels of leptin in a population of Mexican Americans: A cross-sectional study. Cytokine 2012, 57, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Spranger, J.; Kroke, A.; Möhlig, M.; Hoffmann, K.; Bergmann, M.M.; Ristow, M.; Boeing, H.; Pfeiffer, A.F.H. Inflammatory cytokines and the risk to develop type 2 diabetes. Diabetes 2003, 52, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Palmer, J.P.; Helqvist, S.; Spinas, G.A.; Mølvig, J.; Mandrup-Poulsen, T.; Andersen, H.U.; Nerup, J. Interaction of β-cell activity and IL-1 concentration and exposure time in isolated rat islets of Langerhans. Diabetes 1989, 38, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Banks, W. Blood-brain barrier transport of cytokines: A mechanism for neuropathology. Curr. Pharm. Des. 2005, 11, 973–984. [Google Scholar] [CrossRef]
- Taepavarapruk, P.; Song, C. Reductions of acetylcholine release and nerve growth factor expression are correlated with memory impairment induced by interleukin-1β administrations: Effects of omega-3 fatty acid EPA treatment. J. Neurochem. 2010, 112, 1054–1064. [Google Scholar] [CrossRef]
- Goh, S.-Y.; Cooper, M.E. The role of advanced glycation end products in progression and complications of diabetes. J. Clin. Endocrinol. Metab. 2008, 93, 1143–1152. [Google Scholar] [CrossRef]
- Abate, G.; Marziano, M.; Rungratanawanich, W.; Memo, M.; Uberti, D. Nutrition and age-ing: Focusing on Alzheimer’s disease. Oxidative Med. Cell. Longev. 2017, 2017, 7039816. [Google Scholar] [CrossRef]
- Chaney, M.O.; Stine, W.B.; Kokjohn, T.A.; Kuo, Y.-M.; Esh, C.; Rahman, A.; Luehrs, D.C.; Schmidt, A.M.; Stern, D.; Yan, S.D.; et al. Rage and amyloid beta interactions: Atomic force microscopy and Molecular Modeling. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2005, 1741, 199–205. [Google Scholar] [CrossRef]
- Deane, R.; Zlokovic, B. Role of the blood-brain barrier in the pathogenesis of alzheimers disease. Curr. Alzheimer Res. 2007, 4, 191–197. [Google Scholar] [CrossRef]
- Matrone, C.; Djelloul, M.; Taglialatela, G.; Perrone, L. Inflammatory risk factors and pathologies promoting Alzheimer’s disease progression: Is RAGE the key? Histol. Histopathol. 2015, 30, 125–139. [Google Scholar]
- Reddy, V.P.; Zhu, X.; Perry, G.; Smith, M.A. Oxidative stress in diabetes and Alzheimer’s disease. J. Alzheimer’s Dis. 2009, 16, 763–774. [Google Scholar] [CrossRef]
- Nita, M.; Grzybowski, A. The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxidative Med. Cell. Longev. 2016, 2016, 3164734. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, W.; Ijaz, B.; Shabbiri, K.; Ahmed, F.; Rehman, S. Oxidative toxicity in diabetes and Alzheimer’s disease: Mechanisms behind ROS/RNS generation. J. Biomed. Sci. 2017, 24, 76. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M. Nutraceutical and Functional Food Components; Academic Press: Cambridge, MA, USA, 2017. [Google Scholar]
- Huang, T.-C.; Lu, K.-T.; Wo, Y.-Y.P.; Wu, Y.-J.; Yang, Y.-L. Resveratrol protects rats from AΒ-induced neurotoxicity by the reduction of inos expression and lipid peroxidation. PLoS ONE 2011, 6, e0029102. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.F.; Hassan, N.A.; El Bassossy, H.M.; Fahmy, A. Quercetin protects against diabetes-induced exaggerated vasoconstriction in rats: Effect on low grade inflammation. PLoS ONE 2013, 8, e0063784. [Google Scholar] [CrossRef]
- Jia, S.; Shen, M.; Zhang, F.; Xie, J. Recent Advances in Momordica charantia: Functional Components and Biological Activities. Int. J. Mol. Sci. 2017, 18, 2555. [Google Scholar] [CrossRef]
- Grover, J.K.; Yadav, S.; Vats, V. Medicinal plants of India with anti-diabetic potential. J. Ethnopharmacol. 2002, 81, 81–100. [Google Scholar] [CrossRef]
- Teoh, S.L.; Latiff, A.A.; Das, S. The effect of topical extract of Momordica charantia (bitter gourd) on wound healing in nondiabetic rats and in rats with diabetes induced by streptozotocin. Clin. Exp. Dermatol. 2009, 34, 815–822. [Google Scholar] [CrossRef]
- Grover, J.K.; Yadav, S.P. Pharmacological actions and potential uses of Momordica charantia: A review. J. Ethnopharmacol. 2004, 93, 123–132. [Google Scholar] [CrossRef]
- Prasad, V.; Jain, V.; Girish, D.; Dorle, A.K. Wound-healing property of Momordica charantia L. fruit powder. J. Hreb. Pharmacother. 2006, 6, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Raman, A.; Lau, C. Anti-diabetic properties and phytochemistry of Momordica charantia L. (Cucurbitaceae). Phytomedicine 1996, 2, 349–362. [Google Scholar] [CrossRef] [PubMed]
- Joseph, B.; Jini, D. Insight into the hypoglycaemic effect of traditional Indian herbs used in the treatment of diabetes. Res. J. Med. Plant 2011, 5, 352–376. [Google Scholar] [CrossRef]
- Joseph, B.; Jini, D. A medicinal potency of Capparis decidua–a harsh terrain plant. Res. J. Phytochem. 2011, 5, 1–13. [Google Scholar] [CrossRef]
- Ayodhya, S.; Kusum, S.; Anjali, S. Hypoglycaemic activity of different extracts of various herbal plants. IJRAP 2010, 1, 212–224. [Google Scholar]
- Malviya, N.; Jain, S.; Malviya, S. Anti-diabetic potential of medicinal plants. Acta Pol. Pharm. 2010, 67, 113–118. [Google Scholar]
- Patel, P.; Harde, P.; Pillai, J.; Darji, N.; Patel, B. Antidiabetic herbal drugs a review. Pharmacophore 2012, 3, 18–29. [Google Scholar]
- Kavishankar, G.B.; Lakshmidevi, N.; Murthy, S.M.; Prakash, H.S.; Niranjana, S.R. Diabetes and medicinal plants-A review. Int. J. Pharm. Biomed Sci. 2011, 2, 65–80. [Google Scholar]
- Hui, H.; Tang, G.; Liang, V.W.; Go, V.L.W. Hypoglycemic herbs and their action mechanisms. Chin. Med. 2009, 4, 11–14. [Google Scholar] [CrossRef]
- Bakare, R.I.; Magbagbeola, O.A.; Akinwande, A.I.; Okunowo, O.W. Nutritional and chemical evaluation of Momordica charantia. J. Med. Plant Res. 2010, 4, 2189–2193. [Google Scholar]
- Islam, S.; Jalaluddin, M.; Hettiarachchy, N.S. Bio-active compounds of bitter melon genotypes (Momordica charantia L.) in relation to their physiological functions. Funct. Foods Health Dis. 2011, 1, 61–74. [Google Scholar] [CrossRef]
- Snee, L.S.; Nerurkar, V.R.; Dooley, D.A.; Efird, J.T.; Shovic, A.C.; Nerurkar, P.V. Strategies to improve palatability and increase consumption intentions for Momordica charantia (bitter melon): A vegetable commonly used for diabetes management. Nutr. J. 2011, 10, 78. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.K.; Shahzadi, I.; Ahmad, I.; Ahmad, R.; Shahzad, K.; Ashraf, M. Viqar-un-Nisa. Nutritional analysis and antioxidant activity of bitter gourd (Momordica charantia) from Pakistan. Pharmacologyonline 2010, 1, 252–260. [Google Scholar]
- Budrat, P.; Shotipruk, A. Extraction of phenolic compounds from fruits of bitter melon (Momordica charantia) with subcritical water extraction and antioxidant activities of these extracts. Chiang Mai J. Sci. 2008, 35, 123–130. [Google Scholar]
- Chang, C.I.; Chen, C.R.; Liao, Y.W.; Cheng, H.L.; Chen, Y.C.; Chou, C.H. Cucurbitane-type triterpenoids from Momordica charantia. J. Nat. Prod. 2006, 71, 1327–1330. [Google Scholar] [CrossRef]
- Tayyab, F.; Lal, S.S.; Mishra, M.; Kumar, U. A review: Medicinal plants and its impact on diabetes. World J. Pharm Res. 2012, 1, 1019–1046. [Google Scholar]
- Puri, M.; Kaur, I.; Kanwar, R.K.; Gupta, R.C.; Chauhan, A.; Kanwar, J.R. Ribosome inactivating proteins (RIPs) from Momordica charantia for anti viral therapy. Curr. Mol. Med. 2009, 9, 1080–1094. [Google Scholar] [CrossRef]
- Fang, E.F.; Zhang, C.Z.Y.; Wong, J.H.; Shen, J.Y.; Li, C.H.; Ng, T.B. The MAP30 protein from bitter gourd (Momordica charantia) seeds promotes apoptosis in liver cancer cells in vitro and in vivo. Cancer Lett. 2012, 324, 66–74. [Google Scholar] [CrossRef]
- Pu, Z.; Lu, B.Y.; Liu, W.Y.; Jin, S.W. Characterization of the enzymatic mechanism of γ-momorcharin, a novel ribosome-inactivating protein with lower molecular weight of 11,500 purified from the seeds of bitter gourd (Momordica charantia). Biochem. Biophys. Res. Commun. 1996, 229, 287–294. [Google Scholar] [CrossRef]
- Meng, Y.; Liu, S.; Li, J.; Meng, Y.; Zhao, X. Preparation of an antitumor and antivirus agent: Chemical modification of α-MMC and MAP30 from Momordica charantia L. with covalent conjugation of polyethyelene glycol. Int. J. Nanomed 2012, 7, 3133. [Google Scholar]
- Leung, S.O.; Yeung, H.W.; Leung, K.N. The immunosuppressive activities of two abortifacient proteins isolated from the seeds of bitter melon (Momordica charantia). Immunopharmacology 1987, 13, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Jabeen, U.; Khanum, A. Isolation and characterization of potential food preservative peptide from Momordica charantia L. Arab. J. Chem. 2017, 10, S3982–S3989. [Google Scholar] [CrossRef]
- Fang, E.F.; Zhang, C.Z.Y.; Ng, T.B.; Wong, J.H.; Pan, W.L.; Ye, X.J.; Chan, Y.S.; Fong, W.P. Momordica charantia lectin, a type II ribosome inactivating protein, exhibits antitumor activity toward human nasopharyngeal carcinoma cells in vitro and in vivo. Cancer Prev. Res. 2012, 5, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.; Raychaudhuri, S.S. Medicinal uses and molecular identification of two Momordica charantia varieties—A review. Electron. J. Biol. 2010, 6, 43–51. [Google Scholar]
- Wang, B.L.; Zhang, W.; Zhao, J.; Wang, F.; Fan, L.; Hu, Z. Gene cloning and expression of a novel hypoglycaemic peptide from Momordica charantia. J. Sci. Food Agric. 2011, 91, 2443–2448. [Google Scholar] [CrossRef]
- Yuan, X.; Gu, X.; Tang, J. Purification and characterisation of a hypoglycemic peptide from Momordica charantia L. Var. abbreviata Ser. Food Chem. 2008, 111, 415–420. [Google Scholar] [CrossRef]
- Mahatmanto, T. Review seed biopharmaceutical cyclic peptides: From discovery to applications. Pept. Sci. 2015, 104, 804–814. [Google Scholar] [CrossRef]
- Zhang, B.; Xie, C.; Wei, Y.; Li, J.; Yang, X. Purification and characterisation of an antifungal protein, MCha-Pr, from the intercellular fluid of bitter gourd (Momordica charantia) leaves. Protein Expr. Purif. 2015, 107, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Altuhafi, A.; Altun, M.; Hadwan, M.H. The Correlation between Selenium-Dependent Glutathione Peroxidase Activity and Oxidant/Antioxidant Balance in Sera of Diabetic Patients with Nephropathy. Rep. Biochem. Mol. Biol. 2021, 10, 164–172. [Google Scholar] [CrossRef]
- Shi, J.; Arunasalam, K.; Yeung, D.; Kakuda, Y.; Mittal, G.; Jiang, Y.; Ercan, P.; El, S.N.; Kim, J.E.; Go, J.; et al. Saponins from edible legumes: Chemistry, processing, and health benefits. J. Med. Food 2004, 7, 67–78. [Google Scholar] [CrossRef]
- Vincken, J.P.; Heng, L.; de Groot, A.; Gruppen, H. Saponins, classification and occurrence in the plant kingdom. Phytochemistry 2007, 68, 275–297. [Google Scholar] [CrossRef]
- Chen, J.C.; Chiu, M.H.; Nie, R.L.; Cordell, G.A.; Qiu, S.X. Cucurbitacins and cucurbitane glycosides: Structures and biological activities. Nat. Prod. Rep. 2005, 22, 386–399. [Google Scholar] [CrossRef] [PubMed]
- Murakami, T.; Emoto, A.; Matsuda, H.; Yoshikawa, M. Medicinal foodstuffs. XXI. Structures of new cucurbitane-type triterpene glycosides, goyaglycosides-a,-b,-c,-d,-e,-f,-g, and-h, and new oleanane-type triterpene saponins, goyasaponins I, II, and III, from the fresh fruit of Japanese Momordica charantia L. Chem. Pharm. Bull. 2001, 49, 54–63. [Google Scholar] [CrossRef]
- Harinantenaina, L.; Tanaka, M.; Takaoka, S.; Oda, M.; Mogami, O.; Uchida, M.; Asakawa, Y. Momordica charantia constituents and antidiabetic screening of the isolated major compounds. Chem. Pharm. Bull. 2006, 54, 1017–1021. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.J.; Ye, J.M.; Turner, N.; Hohnen-Behrens, C.; Ke, C.Q.; Tang, C.P.; Chen, T.; Weiss, H.-C.; Gesing, E.-R.; Rowland, A.; et al. Antidiabetic activities of triterpenoids isolated from bitter melon associated with activation of the AMPK pathway. Chem. Biol. 2008, 15, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Krawinkel, M.B.; Keding, G.B. Bitter gourd (Momordica charantia): A dietary approach to hyperglycemia. Nutr. Rev. 2006, 64, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Pitiphanpong, J.; Chitprasert, S.; Goto, M.; Jiratchariyakul, W.; Sasaki, M.; Shotipruk, A. New approach for extraction of charantin from Momordica charantia with pressurized liquid extraction. Sep. Purif. Technol. 2007, 52, 416–422. [Google Scholar] [CrossRef]
- Patel, S.; Patel, T.; Parmar, K.; Bhatt, Y.; Patel, Y.; Patel, N.M.D. Isolation, characterization and antimicrobial activity of charantin from Momordica charantia linn. Fruit. Int. J. Drug Dev. Res. 2010, 2, 629–634. [Google Scholar]
- Hollman, P.C.; de Vries, J.H.; van Leeuwen, S.D.; Mengelers, M.J.; Katan, M.B. Absorption of dietary quercetin glycosides and quercetin in healthy ileostomy volunteers. Am. J. Clin. Nutr. 1995, 62, 1276–1282. [Google Scholar] [CrossRef]
- Haddad, P.S.; Eid, H.M.; Nachar, A.; Thong, F.; Sweeney, G. The molecular basis of the antidiabetic action of quercetin in cultured skeletal muscle cells and hepatocytes. Pharmacogn. Mag. 2015, 11, 74–81. [Google Scholar] [CrossRef]
- Youl, E.; Bardy, G.; Magous, R.; Cros, G.; Sejalon, F.; Virsolvy, A.; Richard, S.; Quignard, J.F.; Gross, R.; Petit, P.; et al. Quercetin potentiates insulin secretion and protects INS-1 pancreatic β-cells against oxidative damage via the ERK1/2 pathway. Br. J. Pharmacol. 2010, 161, 799–814. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Li, Q.; Li, K.; Zhu, L.; Lin, X.; Lin, X.; Shen, Q.; Li, G.; Xie, X. Quercetin improves glucose and lipid metabolism of diabetic rats: Involvement of Akt signaling and SIRT1. J. Diabetes Res. 2017, 2017, 3417306. [Google Scholar] [CrossRef] [PubMed]
- Eid, H.M.; Haddad, P.S. The Antidiabetic Potential of Quercetin: Underlying Mechanisms. Curr. Med. Chem. 2017, 24, 355–364. [Google Scholar]
- Ergul, A. Endothelin-1 and diabetic complications: Focus on the vasculature. Pharmacol. Res. 2011, 63, 477–482. [Google Scholar] [CrossRef]
- Qu, L.; Liang, X.; Gu, B.; Liu, W. Quercetin alleviates high glucose-induced Schwann cell damage by autophagy. Neural Regen. Res. 2014, 9, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.-F.; Xie, Z.-X.; Qiao, Y.; Li, L.-R.; Cheng, X.-R.; Tang, X.; Shi, Y.-H.; Le, G.-W. Differential effects of quercetin on hippocampus-dependent learning and memory in mice fed with different diets related with oxidative stress. Physiol. Behav. 2015, 138, 325–331. [Google Scholar] [CrossRef]
- Zhu, X.; Cheng, Y.-Q.; Lu, Q.; Du, L.; Yin, X.-X.; Liu, Y.-W. Enhancement of glyoxalase 1, a polyfunctional defense enzyme, by quercetin in the brain in streptozotocin-induced diabetic rats. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2018, 391, 1237–1245. [Google Scholar] [CrossRef]
- Ansari, M.A.; Abdul, H.M.; Joshi, G.; Opii, W.O.; Butterfield, D.A. Protective effect of quercetin in primary neurons against AΒ(1–42): Relevance to Alzheimer’s disease. J. Nutr. Biochem. 2009, 20, 269–275. [Google Scholar] [CrossRef]
- Kong, Y.; Li, K.; Fu, T.; Wan, C.; Zhang, D.; Song, H.; Zhang, Y.; Liu, N.; Gan, Z.; Yuan, L. Quercetin ameliorates AΒ toxicity in drosophila AD model by modulating cell cycle-related protein expression. Oncotarget 2016, 7, 67716–67731. [Google Scholar] [CrossRef]
- Sabogal-Guáqueta, A.M.; Muñoz-Manco, J.I.; Ramírez-Pineda, J.R.; Lamprea-Rodriguez, M.; Osorio, E.; Cardona-Gómez, G.P. The flavonoid quercetin ameliorates Alzheimer’s disease pathology and protects cognitive and emotional function in aged triple transgenic Alzheimer’s disease model mice. Neuropharmacology 2015, 93, 134–145. [Google Scholar] [CrossRef]
- Wang, D.-M.; Li, S.-Q.; Wu, W.-L.; Zhu, X.-Y.; Wang, Y.; Yuan, H.-Y. Effects of long-term treatment with quercetin on cognition and mitochondrial function in a mouse model of Alzheimer’s disease. Neurochem. Res. 2014, 39, 1533–1543. [Google Scholar] [CrossRef]
- Vargas-Restrepo, F.; Sabogal-Guáqueta, A.M.; Cardona-Gómez, G.P. Quercetin ameliorates inflammation in CA1 hippocampal region in aged triple transgenic Alzheimer’s disease mice model. Biomédica 2017, 38, 69–76. [Google Scholar] [CrossRef]
- Yang, J.; Guo, J.; Yuan, J. In vitro antioxidant properties of Rutin. LWT-Food Sci. Technol. 2008, 41, 1060–1066. [Google Scholar] [CrossRef]
- Ghorbani, A. Mechanisms of antidiabetic effects of flavonoid Rutin. Biomed. Pharmacother. 2017, 96, 305–312. [Google Scholar] [CrossRef]
- Ahmed, O.M.; Moneim, A.A.; Yazid, I.A.; and Mahmoud, A.M. Antihyperglycemic, antihyperlipidemic and antioxidant effects and the probable mechanisms of action of Ruta graveolens infusion and rutin in nicotinamide-streptozotocin- induced diabetic rats. Diabetol. Croat. 2010, 39, 15–35. [Google Scholar]
- Wang, S.W.; Wang, Y.J.; Su, Y.J.; Zhou, W.W.; Yang, S.G.; Zhang, R.; Zhao, M.; Li, Y.N.; Zhang, Z.P.; Zhan, D.W.; et al. Rutin inhibits β-amyloid aggregation and cytotoxicity, attenuates oxidative stress, and decreases the production of nitric oxide and pro-inflammatory cytokines. Neurotoxicology 2012, 33, 482–490. [Google Scholar] [CrossRef]
- Hevener, A.L.; He, W.; Barak, Y.; Le, J.; Bandyopadhyay, G.; Olson, P.; Wilkes, J.; Evans, R.M.; Olefsky, J. Muscle-specific PPARG deletion causes insulin resistance. Nat. Med. 2003, 9, 1491–1497. [Google Scholar] [CrossRef]
- Cai, E.P.; Lin, J.-K. Epigallocatechin gallate (EGCG) and Rutin suppress the glucotoxicity through activating IRS2 and AMPK signaling in rat pancreatic β cells. J. Agric. Food Chem. 2009, 57, 9817–9827. [Google Scholar] [CrossRef]
- Niture, N.T.; Ansari, A.A.; Naik, S.R. Anti-hyperglycemic activity of rutin in streptozotocin-induced diabetic rats: An effect mediated through cytokines, antioxidants and lipid biomarkers. Indian J. Exp. Biol. 2014, 52, 720–727. [Google Scholar]
- Habtemariam, S. Rutin as a natural therapy for Alzheimer’s disease: Insights into its mechanisms of action. Curr. Med. Chem. 2016, 23, 860–873. [Google Scholar] [CrossRef]
- Jiménez-Aliaga, K.; Bermejo-Bescós, P.; Benedí, J.; Martín-Aragón, S. Quercetin and rutin exhibit antiamyloidogenic and fibril-disaggregating effects in vitro and potent antioxidant activity in APPswe cells. Life Sci. 2011, 89, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Moghbelinejad, S.; Nassiri-Asl, M.; Farivar, T.N.; Abbasi, E.; Sheikhi, M.; Taghiloo, M.; Farsad, F.; Samimi, A.; Hajiali, F. Rutin activates the MAPK pathway and BDNF gene expression on beta-amyloid induced neurotoxicity in rats. Toxicol. Lett. 2014, 224, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Lee, J.M.; Lee, D.G.; Cho, S.; Yoon, Y.-H.; Cho, E.J.; Lee, S. The n-butanol fraction and rutin from Tartary buckwheat improve cognition and memory in an in vivo model of amyloid-β-induced Alzheimer’s disease. J. Med. Food 2015, 18, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Al-Ishaq, R.K.; Abotaleb, M.; Kubatka, P.; Kajo, K.; Büsselberg, D. Flavonoids and Their Anti-Diabetic Effects: Cellular Mechanisms and Effects to Improve Blood Sugar Levels. Biomolecules 2019, 9, 430. [Google Scholar] [CrossRef] [PubMed]
- Calderon-Montano, J.M.; Burgos-Moron, E.; Perez-Guerrero, C.; Lopez-Lazaro, M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Zhang, Z.; Ding, Y.; Dai, X.; Wang, J.; Li, Y. Epigallocatechin-3-gallate protects pro-inflammatory cytokine induced injuries in insulin-producing cells through the mitochondrial pathway. Eur. J. Pharmacol. 2011, 670, 311–316. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, D. Flavonol kaempferol improves chronic hyperglycemia-impaired pancreatic beta-cell viability and insulin secretory function. Eur. J. Pharmacol. 2011, 670, 325–332. [Google Scholar] [CrossRef]
- Alkhalidy, H.; Moore, W.; Wang, Y.; Luo, J.; McMillan, R.P.; Zhen, W.; Zhou, K.; Liu, D. The Flavonoid Kaempferol Ameliorates Streptozotocin-Induced Diabetes by Suppressing Hepatic Glucose Production. Molecules 2018, 23, 2338. [Google Scholar] [CrossRef]
- Sharma, D.; Gondaliya, P.; Tiwari, V.; Kalia, K. Kaempferol attenuates diabetic nephropathy by inhibiting RhoA/Rho-kinase mediated inflammatory signaling. Biomed. Pharmacother. 2019, 109, 1610–1619. [Google Scholar] [CrossRef]
- Vinayagam, R.; Xu, B. Antidiabetic properties of dietary flavonoids: A cellular mechanism review. Nutr. Metab. 2015, 12, 60. [Google Scholar] [CrossRef]
- Lee, Y.S.; Lee, S.; Lee, H.S.; Kim, B.K.; Ohuchi, K.; Shin, K.H. Inhibitory effects of isorhamnetin-3-O-beta-D-glucoside from Salicornia herbacea on rat lens aldose reductase and sorbitol accumulation in streptozotocin-induced diabetic rat tissues. Biol. Pharm. Bull. 2005, 28, 916–918. [Google Scholar] [CrossRef] [PubMed]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1043. [Google Scholar] [CrossRef]
- Zhang, F.; Lin, L.; Xie, J. A mini-review of chemical and biological properties of polysaccharides from Momordica charantia. Int. J. Biol. Macromol. 2016, 92, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.Y.; Yi, Y.; Zhang, L.F.; Zhang, R.F.; Zhang, Y.; Wei, Z.C.; Zhang, M.W. Immunomodulatory activity and partial characterization of polysaccharides from Momordica charantia. Molecules 2014, 19, 13432–13447. [Google Scholar] [CrossRef]
- Duan, Z.Z.; Zhou, X.L.; Li, Y.H.; Zhang, F.; Li, F.Y.; Su-Hua, Q. Protection of Momordica charantia polysaccharide against intracerebral hemorrhage-induced brain injury through JNK3 signaling pathway. J. Recept. Signal Transduct 2015, 35, 523–529. [Google Scholar] [CrossRef]
- Cai, Y.; Liu, M.; Wu, X.; Wang, Z.; Liang, C.; Yang, Y. Study on the antitumor and immune-stimulating activity of polysaccharide from Momordica charantia. Pharm. Clin. Res. 2010, 18, 131–134. [Google Scholar]
- Zhang, P.P.; Liu, J.F.; Wang, C.L.; Ye, Y.T.; Xie, J.H. Study on the antimicrobial activities of the extracts from Momordica charantia L. Nat. Prod. Res. 2008, 20, 721–724. [Google Scholar]
- Panda, B.C.; Mondal, S.; Devi, K.S.P.; Maiti, T.K.; Khatua, S.; Acharya, K.; Islam, S.S. Pectic polysaccharide from the green fruits of Momordica charantia (Karela): Structural characterization and study of immunoenhancing and antioxidant properties. Carbohydr. Res. 2015, 401, 24–31. [Google Scholar] [CrossRef]
- Dong, Y.; Xu, B.; Lu, Q.; Zha, Q. Studies on the Isolation, Purification and Composition of Momordica charantia L. Polysaccharide. Food Sci. 2005, 11, 023. [Google Scholar]
- Fan, T.; Hu, J.; Fu, L.; Zhang, L. Optimization of enzymolysis-ultrasonic assisted extraction of polysaccharides from Momordica charantia L. by response surface methodology. Carbohydr. Polym. 2015, 115, 701–706. [Google Scholar] [CrossRef]
- Tan, H.F.; Gan, C.Y. Polysaccharide with antioxidant, α-amylase inhibitory and ACE inhibitory activities from Momordica charantia. Int. J. Biol. Macromol. 2016, 85, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Shan, B.; Liao, C.H.; Xie, J.H.; Wen, P.W.; Shi, J.Y. Anti-diabetic properties of Momordica charantia L. polysaccharide in alloxan-induced diabetic mice. Int. J. Biol. Macromol. 2015, 81, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Barrett, M.L.; Udani, J.K. A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): A review of clinical studies on weight loss and glycemic control. Nutr. J. 2011, 10, 24. [Google Scholar] [CrossRef]
- Hypoglycemia (Low Blood Glucose). Hypoglycemia (Low Blood Glucose)|ADA. (n.d.). Available online: https://diabetes.org/healthy-living/medication-treatments/blood-glucose-testing-and-control/hypoglycemia (accessed on 15 October 2022).
- Raish, M. Momordica charantia, polysaccharides ameliorate oxidative stress, hyperlipidemia, inflammation, and apoptosis during myocardial infarction by inhibiting the nf-kb signaling pathway. Int. J. Biol. Macromol. 2017, 97, 544–551. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Kang, J.; Zeng, B.; Tang, S.; Wang, M.; Han, X.; Zhou, C.; Tan, Z. Effects of Momordica charantia polysaccharide on in vitro ruminal fermentation and cellulolytic bacteria. Ital. J. Anim. Sci. 2017, 16, 226–233. [Google Scholar] [CrossRef]
- Kumar, D.S.; Sharathnath, V.K.; Yogeswaran, P.; Harani, A.; Sudhakar, K.; Sudha, P.; Banji, D. A medicinal potency of Momordica charantia. Int. J. Pharm Sci Rev Res. 2010, 1, 95–99. [Google Scholar]
- Hazarika, R.; Parida, P.; Neog, B.; Yadav, R.N.S. Binding energy calculation of GSK-3 protein of human against some anti- diabetic compounds of Momordica charantia linn (Bitter melon). Bioinformation 2012, 8, 251–254. [Google Scholar] [CrossRef]
- Leclerc, M.; Dudonné, S.; Calon, F. Can Natural Products Exert Neuroprotection without Crossing the Blood–Brain Barrier? Int. J. Mol. Sci. 2021, 22, 3356. [Google Scholar] [CrossRef]
- Shan, B.; Xie, J.H.; Zhu, J.H.; Peng, Y. Ethanol modified supercritical carbon dioxide extraction of flavonoids from Momordica charantia L. and its antioxidant activity. Food Bioprod. Process. 2012, 90, 579–587. [Google Scholar] [CrossRef]
- Tan, S.P.; Stathopoulos, C.; Parks, S.; Roach, P. An optimised aqueous extract of phenolic compounds from bitter melon with high antioxidant capacity. Antioxidants 2014, 3, 814–829. [Google Scholar] [CrossRef] [PubMed]
- Abotaleb, M.; Samuel, S.M.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Busselberg, D. Flavonoids in Cancer and Apoptosis. Cancers 2018, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Madala, N.E.; Piater, L.; Dubery, I.; Steenkamp, P. Distribution patterns of flavonoids from three momordica species by ultra-high performance liquid chromatography quadrupole time of flight mass spectrometry: A metabolomic profiling approach. Rev. Bras. De Farmacogn. 2016, 26, 507–513. [Google Scholar] [CrossRef]
- Chuang, C.Y.; Hsu, C.; Chao, C.Y.; Wein, Y.S.; Kuo, Y.H.; Huang, C.J. Fractionation and identification of 9c, 11t, 13t-conjugated linolenic acid as an activator of PPARα in bitter gourd (Momordica charantia L.). J. Biomed. Sci. 2006, 13, 763–772. [Google Scholar] [CrossRef]
- Lucas, E.A.; Dumancas, G.G.; Smith, B.J.; Clarke, S.L.; Arjmandi, B.H. Health benefits of bitter melon (Momordica charantia). Bioact. Foods Promot. Health 2010, 35, 525–549. [Google Scholar]
- Yuwai, K.E.; Rao, K.S.; Kaluwin, C.; Jones, G.P.; Rivett, D.E. Chemical composition of Momordica charantia L. fruits. J. Agric. Food Chem. 1991, 39, 1762–1763. [Google Scholar] [CrossRef]
- Kenny, O.; Smyth, T.J.; Hewage, C.M.; Brunton, N.P. Antioxidant properties and quantitative UPLC-MS analysis of phenolic compounds from extracts of fenugreek (Trigonella foenum-graecum) seeds and bitter melon (Momordica charantia) fruit. Food Chem. 2013, 141, 4295–4302. [Google Scholar] [CrossRef]
- Ke, L.J.; Lu, W.; Chang, J.L.; Yuan, F.Y.; Rao, P.F.; Zhou, J.W. Effects of heat drying process on amino acid content of Momordica charantia L. Amino Acids Biol. Resour. 2010, 32, 14–16. [Google Scholar]
- Han, C.; Zuo, J.; Wang, Q.; Xu, L.; Wang, Z.; Dong, H.; Gao, L. Effects of 1-MCP on postharvest physiology and quality of bitter melon (Momordica charantia L.). Sci. Hortic-Ansterdam. 2015, 182, 86–91. [Google Scholar] [CrossRef]
- Shi, L.; Du, X.; Guo, P.; Huang, L.; Qi, P.; Gong, Q. Ascorbic acid supplementation in type 2 diabetes mellitus: A protocol for systematic review and meta-analysis. Medicine 2020, 99, e23125. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L. Bitter melon (Momordica charantia). In Herbal Secrets of the Rainforest, 2nd ed.; Texas, A., Ed.; Sage Press: Thousand Oaks, CA, USA, 2002; pp. 1–100. [Google Scholar]

| Kingdom: | Plantae |
|---|---|
| Common Name: | Bitter melon, bitter gourd, karela. |
| Order: | Cucurbitales |
| Species: | M. charantia |
| Genus: | Momordica |
| Family: | Cucurbitaceous |
| Class: | Magnoliopsida |
| Division: | Magnoliophyta |
| Major Bioactive Compounds | Bioactive Functions | Mechanism of Action | Beneficial Effects | Distribution | Reference |
|---|---|---|---|---|---|
| Proteins/Peptides: (1) Polypeptide-P (2) Peroxidase | RNA N-glycosidase, polynucleotide adenosine glycosidase (PAG), DNase-like, phospholipase, superoxide dismutase, anti-tumor, immune suppression, antimicrobial | (1) Binds to INS receptor (2) ↓ oxidative stress; ↓ toxicity | (1) Hypoglycemic effect (2) Neutralization by the antioxidant activity | Seed | [3,64,84,85,86,87,88,89,90,91,92,93,94,95,96,97] |
| Saponins/Terpenoids: Charantin | Antioxidant, antidiabetic, hypoglycemic, anticancer, hypolipidemic, antiviral | AMP-activated protein kinase activity; ↓ blood lipid levels; ↓ blood glucose response | Hypoglycemic effects | Fruit, root, seed, stem, and leaves | [3,17,64,93,98,99,100,101,102,103,104,105,106] |
| Polyphenols: (1) Quercetin | Antioxidant, anti-inflammatory, antiapoptotic, and immune-enhancing | (1) ↓ Lipid peroxidation & ↓ Oxidative stress via ERK1/2 activation; ↑ activation of AMPK; ↓ activity of G6pase; ↓ TNF-α, CRP, NF- κB; ↓ Aß1-40, Aß1-42, and BACE1 | (1) ↓ Aß-42-induced apoptotic cell death and cell toxicity; ↑ GLUT4 translocation; ↓ hepatic glucose production; ↓ diabetes-induced HTN and vasoconstriction; ↑ Learning and memory function | Fruit, pericarp, and seed | [3,79,102,107,108,109,110,111,112,113,114,115,116,117,118,119,120] |
| (2) Rutin | (2) Free-radical scavenger activity; via PI3K, atypical protein kinase C and MAPK pathways; ↑ IRK activity, ↑ GLUT4 translocation; ↓ activation of MAPK pathway, ↑ BDNF gene expression | (2) ↓ formation of Aß fibrils and disaggregated Aß fibrils, ↓ neurotoxicity; ↑ glucose; ↓ bg; ↓ Aß-induced learning and memory deficits, ↓ Aß-induced neurotoxicity | [3,32,108,121,122,123,124,125,126,127,128,129,130,131] | ||
| (3) Kaempferol | (3) ↑ production and secretion of insulin from ß cells; ↑ glucose uptake via protein kinase C and PI3K pathway; regulation of AMPK activation; ↓ of hepatic gluconeogenesis; ↓ RhoA/Rho kinase-mediated pro-inflammatory signaling | (3) ↓ cellular apoptosis; enhance synthesis of glucose transporter proteins; ↓ HbA1c and fasting blood glucose; ↑ glucose metabolism; ↓ diabetic neuropathy | [132,133,134,135,136,137] | ||
| (4) Isorhamnetin | (4) Activation of JAK2/STAT pathway and promotion of GLUT4 translocation; Activation of NO/GC/cGMP pathway and cyclooxygenase pathway | (4) Hypoglycemic effect; ↓ oxidative stress, ↓ blood glucose levels; ↓ sorbitol aggregation | [138,139,140] | ||
| Polysaccharides | Antioxidant, antidiabetic, immune enhancing, neuroprotective, antitumor, antimicrobial, hypoglycemic, and anti-inflammatory | Inhibition of α-amylase, ACE, and NF-κB signaling pathway | ↓ oxidative stress; regulates blood glucose; ↑ volatile FA production | Various parts of the plant | [64,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richter, E.; Geetha, T.; Burnett, D.; Broderick, T.L.; Babu, J.R. The Effects of Momordica charantia on Type 2 Diabetes Mellitus and Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 4643. https://doi.org/10.3390/ijms24054643
Richter E, Geetha T, Burnett D, Broderick TL, Babu JR. The Effects of Momordica charantia on Type 2 Diabetes Mellitus and Alzheimer’s Disease. International Journal of Molecular Sciences. 2023; 24(5):4643. https://doi.org/10.3390/ijms24054643
Chicago/Turabian StyleRichter, Erika, Thangiah Geetha, Donna Burnett, Tom L. Broderick, and Jeganathan Ramesh Babu. 2023. "The Effects of Momordica charantia on Type 2 Diabetes Mellitus and Alzheimer’s Disease" International Journal of Molecular Sciences 24, no. 5: 4643. https://doi.org/10.3390/ijms24054643
APA StyleRichter, E., Geetha, T., Burnett, D., Broderick, T. L., & Babu, J. R. (2023). The Effects of Momordica charantia on Type 2 Diabetes Mellitus and Alzheimer’s Disease. International Journal of Molecular Sciences, 24(5), 4643. https://doi.org/10.3390/ijms24054643







