Can Baseline IL-6 Levels Predict Long COVID in Subjects Hospitalized for SARS-CoV-2 Disease?
Abstract
1. Introduction
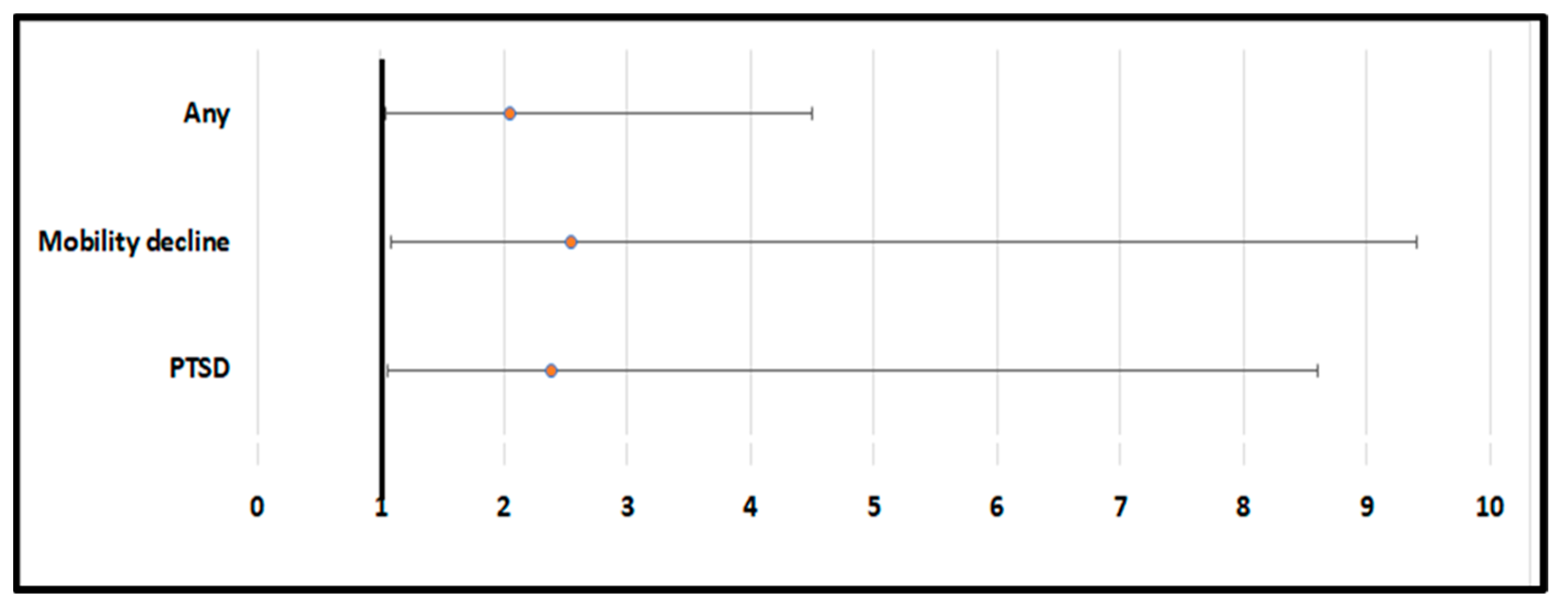
2. Results
3. Discussion
4. Materials and Methods
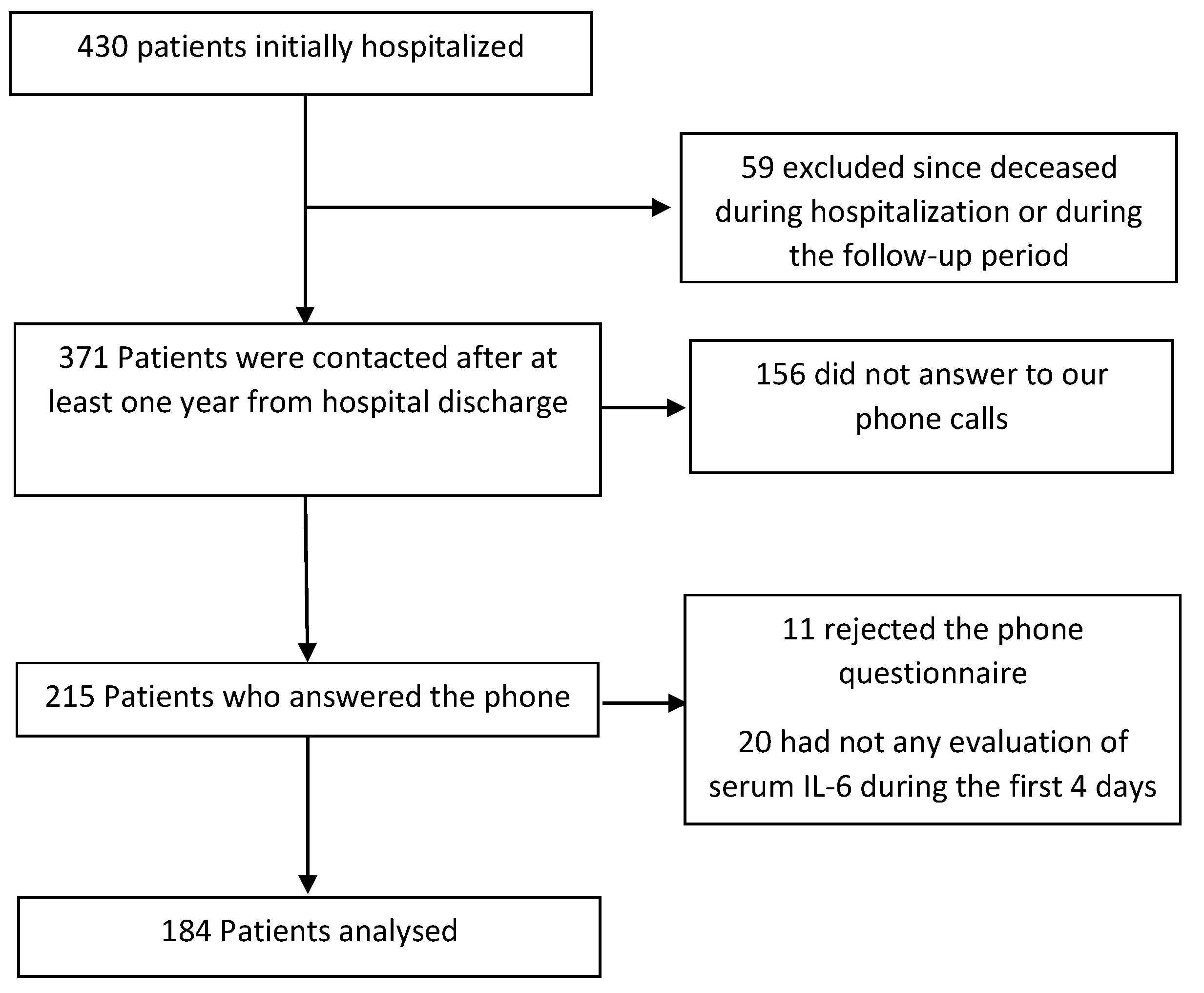
4.1. Study Population
4.2. Exposure: Serum IL-6 Levels
4.3. Outcomes: Long COVID
4.4. Covariates and Parameters
4.5. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, C.; Haupert, S.R.; Zimmermann, L.; Shi, X.; Fritsche, L.G.; Mukherjee, B. Global Prevalence of Post COVID-19 Condition or Long COVID: A Meta-Analysis and Systematic Review. J. Infect. Dis. 2022, 226, 1593–1607. [Google Scholar] [CrossRef]
- Natarajan, A.; Shetty, A.; Delanerolle, G.; Zeng, Y.; Zhang, Y.; Raymont, V.; Rathod, S.; Halabi, S.; Elliot, K.; Phiri, P. A systematic review and meta-analysis of Long COVID symptoms. medRxiv 2022. [Google Scholar] [CrossRef]
- Han, Q.; Zheng, B.; Daines, L.; Sheikh, A. Long-Term sequelae of COVID-19: A systematic review and meta-analysis of one-year follow-up studies on post-COVID symptoms. Pathogens 2022, 11, 269. [Google Scholar] [CrossRef]
- Logue, J.K.; Franko, N.M.; McCulloch, D.J.; McDonald, D.; Magedson, A.; Wolf, C.R.; Chu, H.Y. Sequelae in adults at 6 months after COVID-19 infection. JAMA Netw. Open 2021, 4, e210830. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Chi, Y.; Ge, Y.; Wu, B.; Zhang, W.; Wu, T.; Wen, T.; Liu, J.; Guo, X.; Huang, C.; Jiao, Y. Serum cytokine and chemokine profile in relation to the severity of coronavirus disease 2019 in China. J. Infect. Dis. 2020, 222, 746–754. [Google Scholar] [CrossRef]
- Cihakova, D.; Streiff, M.B.; Menez, S.P.; Chen, T.K.; Gilotra, N.A.; Michos, E.D.; Marr, K.A.; Karaba, A.H.; Robinson, M.L.; Blair, P.W. High-value laboratory testing for hospitalized COVID-19 patients: A review. Future Virol. 2021, 16, 691–705. [Google Scholar] [CrossRef]
- Gao, Y.; Li, T.; Han, M.; Li, X.; Wu, D.; Xu, Y.; Zhu, Y.; Liu, Y.; Wang, X.; Wang, L. Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19. J. Med. Virol. 2020, 92, 791–796. [Google Scholar] [CrossRef]
- Chen, X.; Zhao, B.; Qu, Y.; Chen, Y.; Xiong, J.; Feng, Y.; Men, D.; Huang, Q.; Liu, Y.; Yang, B. Detectable serum SARS-CoV-2 viral load (RNAaemia) is closely correlated with drastically elevated interleukin 6 (IL-6) level in critically ill COVID-19 patients. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Kappelmann, N.; Dantzer, R.; Khandaker, G.M. Interleukin-6 as potential mediator of long-term neuropsychiatric symptoms of COVID-19. Psychoneuroendocrinology 2021, 131, 105295. [Google Scholar] [CrossRef]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. Immunotherapeutic implications of IL-6 blockade for cytokine storm. Immunotherapy 2016, 8, 959–970. [Google Scholar] [CrossRef]
- Hunter, C.A.; Jones, S.A. IL-6 as a keystone cytokine in health and disease. Nat. Immunol. 2015, 16, 448–457. [Google Scholar] [CrossRef]
- Pathan, N.; Hemingway, C.A.; Alizadeh, A.A.; Stephens, A.C.; Boldrick, J.C.; Oragui, E.E.; McCabe, C.; Welch, S.B.; Whitney, A.; O’Gara, P. Role of interleukin 6 in myocardial dysfunction of meningococcal septic shock. Lancet 2004, 363, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Hay, K.A.; Hanafi, L.-A.; Li, D.; Gust, J.; Liles, W.C.; Wurfel, M.M.; López, J.A.; Chen, J.; Chung, D.; Harju-Baker, S. Kinetics and biomarkers of severe cytokine release syndrome after CD19 chimeric antigen receptor–modified T-cell therapy. Blood J. Am. Soc. Hematol. 2017, 130, 2295–2306. [Google Scholar] [CrossRef]
- Wolf, J.; Rose-John, S.; Garbers, C. Interleukin-6 and its receptors: A highly regulated and dynamic system. Cytokine 2014, 70, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Schumertl, T.; Lokau, J.; Rose-John, S.; Garbers, C. Function and proteolytic generation of the soluble interleukin-6 receptor in health and disease. Biochim. Et Biophys. Acta Mol. Cell Res. 2022, 1869, 119143. [Google Scholar] [CrossRef]
- Hibi, M.; Murakami, M.; Saito, M.; Hirano, T.; Taga, T.; Kishimoto, T. Molecular cloning and expression of an IL-6 signal transducer, gp130. Cell 1990, 63, 1149–1157. [Google Scholar] [CrossRef]
- Proal, A.D.; VanElzakker, M.B. Long COVID or post-acute sequelae of COVID-19 (PASC): An overview of biological factors that may contribute to persistent symptoms. Front. Microbiol. 2021, 12, 698169. [Google Scholar] [CrossRef]
- Giannitrapani, L.; Augello, G.; Mirarchi, L.; Amodeo, S.; Veronese, N.; Sasso, B.L.; Giglio, R.V.; Licata, A.; Barbagallo, M.; Ciaccio, M. Outcome predictors in SARS-CoV-2 disease (COVID-19): The prominent role of IL-6 levels and an IL-6 gene polymorphism in a western Sicilian population. J. Infect. 2022, 85, 174–211. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.; Wong, P.; Klein, J.; Castro, T.B.; Silva, J.; Sundaram, M.; Ellingson, M.K.; Mao, T.; Oh, J.E.; Israelow, B. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature 2020, 584, 463–469. [Google Scholar] [CrossRef]
- Ruenjaiman, V.; Sodsai, P.; Kueanjinda, P.; Bunrasmee, W.; Klinchanhom, S.; Reantragoon, R.; Tunvirachaisakul, C.; Manothummetha, K.; Mejun, N.; Liengswangwong, K. Impact of SARS-CoV-2 infection on the profiles and responses of innate immune cells after recovery. J. Microbiol. Immunol. Infect. 2022, 55, 993–1004. [Google Scholar] [CrossRef]
- Visvabharathy, L.; Orban, Z.S.; Koralnik, I.J. Case report: Treatment of long COVID with a SARS-CoV-2 antiviral and IL-6 blockade in a patient with rheumatoid arthritis and SARS-CoV-2 antigen persistence. Front. Med. 2022, 9, 1003103. [Google Scholar] [CrossRef]
- Tay, M.Z.; Poh, C.M.; Rénia, L.; MacAry, P.A.; Ng, L.F. The trinity of COVID-19: Immunity, inflammation and intervention. Nat. Rev. Immunol. 2020, 20, 363–374. [Google Scholar] [CrossRef]
- Frere, J.J.; Serafini, R.A.; Pryce, K.D.; Zazhytska, M.; Oishi, K.; Golynker, I.; Panis, M.; Zimering, J.; Horiuchi, S.; Hoagland, D.A. SARS-CoV-2 infection in hamsters and humans results in lasting and unique systemic perturbations post recovery. Sci. Transl. Med. 2022, 14, eabq3059. [Google Scholar] [CrossRef]
- Silva Andrade, B.; Siqueira, S.; de Assis Soares, W.R.; de Souza Rangel, F.; Santos, N.O.; dos Santos Freitas, A.; Ribeiro da Silveira, P.; Tiwari, S.; Alzahrani, K.J.; Góes-Neto, A. Long-COVID and post-COVID health complications: An up-to-date review on clinical conditions and their possible molecular mechanisms. Viruses 2021, 13, 700. [Google Scholar] [CrossRef]
- Kirwan, R.; McCullough, D.; Butler, T.; Perez de Heredia, F.; Davies, I.G.; Stewart, C. Sarcopenia during COVID-19 lockdown restrictions: Long-term health effects of short-term muscle loss. GeroScience 2020, 42, 1547–1578. [Google Scholar] [CrossRef]
- Florencio, L.L.; Fernández-de-Las-Peñas, C. Long COVID: Systemic inflammation and obesity as therapeutic targets. Lancet Respir. Med. 2022, 10, 726–727. [Google Scholar] [CrossRef]
- Barbagallo, M.; Citarrella, R.; Dominguez, L.; Giannitrapani, L.; Licata, A.; Mansueto, P.; Soresi, M.; Veronese, N. COMEPA (COVID-19 Medicina Policlinico Palermo): A study in hospitalized patients. Geriatr. Care 2021, 7, 9895. [Google Scholar]
- Stavem, K.; Ghanima, W.; Olsen, M.K.; Gilboe, H.M.; Einvik, G. Prevalence and determinants of fatigue after COVID-19 in non-hospitalized subjects: A population-based study. Int. J. Environ. Res. Public Health 2021, 18, 2030. [Google Scholar] [CrossRef]
- Blevins, C.A.; Weathers, F.W.; Davis, M.T.; Witte, T.K.; Domino, J.L. The posttraumatic stress disorder checklist for DSM-5 (PCL-5): Development and initial psychometric evaluation. J. Trauma. Stress 2015, 28, 489–498. [Google Scholar] [CrossRef]
- Bjelland, I.; Dahl, A.A.; Haug, T.T.; Neckelmann, D. The validity of the Hospital Anxiety and Depression Scale: An updated literature review. J. Psychosom. Res. 2002, 52, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Covelli, H.D.; Nessan, V.J.; Tuttle, W., 3rd. Oxygen derived variables in acute respiratory failure. Crit. Care Med. 1983, 11, 646–649. [Google Scholar] [CrossRef] [PubMed]
- Linn, B.S.; Linn, M.W.; Gurel, L. Cumulative illness rating scale. J. Am. Geriatr. Soc. 1968, 16, 622–626. [Google Scholar] [CrossRef] [PubMed]
| Parameter | IL-6 Normal (n = 58) | IL-6 Elevated (n = 123) | p-Value |
|---|---|---|---|
| Demographics | |||
| Age (mean, SD) | 60.0 (15.2) | 63.1 (12.6) | 0.18 |
| Female sex (%) | 48.3 | 47.2 | 0.89 |
| Actual/previous smokers (%) | 12.1 | 17.9 | 0.11 |
| Laboratory measures | |||
| PaO2/FiO2 ratio | 314 (79) | 324 (84) | 0.50 |
| White blood cells | 8318 (3194) | 7987 (5027) | 0.65 |
| Serum CRP levels (mg/dL) | 9.82 (2.65–25.04) | 56.0 (7.03–67.77) | <0.0001 |
| Hemoglobin (g/dL) | 13.9 (1.6) | 13.4 (1.8) | 0.12 |
| Creatinine clearance (mL/min) | 84 (25) | 81 (27) | 0.48 |
| Medical conditions | |||
| Cardiac diseases (%) | 15.2 | 12.1 | 0.31 |
| Vascular diseases (%) | 57.6 | 53.8 | 0.80 |
| Hematological diseases (%) | 3.0 | 19.8 | 0.11 |
| Respiratory diseases (%) | 15.2 | 11.0 | 0.15 |
| Eyes, ears, nose, throat, and larynx conditions (%) | 6.1 | 7.7 | 0.94 |
| Upper gastrointestinal conditions (%) | 10.1 | 11.0 | 0.33 |
| Lower gastrointestinal conditions (%) | 3.0 | 10.9 | 0.60 |
| Liver, pancreas, and biliary conditions (%) | 0.0 | 9.9 | 0.17 |
| Kidney conditions (%) | 3.0 | 4.4 | 0.85 |
| Genitourinary conditions (%) | 6.1 | 14.3 | 0.43 |
| Musculoskeletal and skin conditions (%) | 11.1 | 4.4 | 0.17 |
| Neurological conditions (%) | 12.1 | 7.7 | 0.15 |
| Endocrine (including sepsis and breast) conditions (%) | 27.3 | 39.6 | 0.26 |
| Psychiatric illnesses (including dementia) (%) | 3.0 | 8.7 | 0.68 |
| Pneumonia (%) | 89.7 | 96.7 | 0.048 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giannitrapani, L.; Mirarchi, L.; Amodeo, S.; Licata, A.; Soresi, M.; Cavaleri, F.; Casalicchio, S.; Ciulla, G.; Ciuppa, M.E.; Cervello, M.; et al. Can Baseline IL-6 Levels Predict Long COVID in Subjects Hospitalized for SARS-CoV-2 Disease? Int. J. Mol. Sci. 2023, 24, 1731. https://doi.org/10.3390/ijms24021731
Giannitrapani L, Mirarchi L, Amodeo S, Licata A, Soresi M, Cavaleri F, Casalicchio S, Ciulla G, Ciuppa ME, Cervello M, et al. Can Baseline IL-6 Levels Predict Long COVID in Subjects Hospitalized for SARS-CoV-2 Disease? International Journal of Molecular Sciences. 2023; 24(2):1731. https://doi.org/10.3390/ijms24021731
Chicago/Turabian StyleGiannitrapani, Lydia, Luigi Mirarchi, Simona Amodeo, Anna Licata, Maurizio Soresi, Francesco Cavaleri, Salvatore Casalicchio, Gregorio Ciulla, Maria Elena Ciuppa, Melchiorre Cervello, and et al. 2023. "Can Baseline IL-6 Levels Predict Long COVID in Subjects Hospitalized for SARS-CoV-2 Disease?" International Journal of Molecular Sciences 24, no. 2: 1731. https://doi.org/10.3390/ijms24021731
APA StyleGiannitrapani, L., Mirarchi, L., Amodeo, S., Licata, A., Soresi, M., Cavaleri, F., Casalicchio, S., Ciulla, G., Ciuppa, M. E., Cervello, M., Barbagallo, M., Veronese, N., & the COMEPA Group. (2023). Can Baseline IL-6 Levels Predict Long COVID in Subjects Hospitalized for SARS-CoV-2 Disease? International Journal of Molecular Sciences, 24(2), 1731. https://doi.org/10.3390/ijms24021731







