Exploring Mast Cell–CD8 T Cell Interactions in Inflammatory Skin Diseases
Abstract
1. Introduction
2. Mast Cells and CD8 T Cells in Tissue Homeostasis and Disease
2.1. Mast Cells
2.2. CD8 T Cells
| Characteristic | TRM Cells | Recirculating Cells | Ref. | |
|---|---|---|---|---|
| Trafficking property | Skin-resident | Skin-transient | [10] | |
| Percentage (%) of CD8 T cells in healthy human skin | Epidermis | 25% | <5% | [51] |
| Dermis | 13% | <5% | ||
| Surface markers in skin | CD103+/−CD69+CD49a+/− | CD69− | [58] | |
| Requirement for persistency in skin | TGF-β, IL-15/IL-7 | TGF-β, IL-15 | [56,74,75] | |
| Effector function | CD103+ > CD103− | Limited | [51,76] | |
| Proliferative capacity | CD103− > CD103+ | TCM > TEM | ||
| Antiviral protection | Critical for secondary infection | Required for optimal response | [77] | |
2.3. Modalities of Mast Cell-CD8 T Cell Interplay in the Skin
3. Modalities of Mast Cell–CD8 T Cell Interactions in the Skin: Cell–Cell Contact
3.1. MHC-TCR Mediated Mast Cell–CD8 T Cell Interactions
3.2. Costimulatory Molecule-Mediated Mast Cell–CD8 T Cell Interactions
3.2.1. CD28-CD80/CD86 Ligation
3.2.2. CD28-CD80/86 Ligation in Psoriasis
3.2.3. CD28-CD80/CD86 Ligation in Mast-Cell-Mediated CD8 T Cell Activation
3.2.4. OX40-OX40L
3.2.5. OX40-OX40 Ligation in Atopic Dermatitis
3.3. Soluble Mediators in Mast Cell–CD8 T Cell Interactions
Mediators Signalling in Vitiligo
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | alopecia areata |
| AD | atopic dermatitis |
| APC | antigen-presenting cells |
| AJ | adherens junctions |
| BMMC | bone-marrow-derived mast cells |
| CLA | cutaneous lymphocyte-associated antigen |
| CCL | CC chemokine ligand |
| CCR | CC chemokine receptor |
| CHS | cutaneous contact hypersensitivity |
| CTLA | cutaneous lymphocyte-associated antigen |
| CRC | colorectal cancer |
| CXCR | C-X-C chemokine receptor |
| CXCL | C-X-C chemokine ligand |
| CLT | cytotoxic lymphocyte-associated antigen-4 |
| DAMP | damage/danger-associated molecular pattern |
| DC | dendritic cells |
| EASI | Eczema Area and Severity Index |
| FcεRI | high-affinity IgE Fc receptor |
| GBD | Global Burden of Disease |
| Grb2 | growth factor receptor-bound protein 2 |
| GWAS | genome-wide association studies |
| IgICAM-1 | immunoglobulinintracellular adhesion receptor 1 |
| IS | immunological synapse |
| IL | interleukin |
| LAMP-1 | lysosomal-associated membrane protein-1 |
| LCMV | lymphocytic choriomeningitis virus |
| LFA-1 | lymphocyte function-associated antigen-1 |
| LTB4 | leukotriene B4 |
| MAPK | mitogen-activated protein kinase |
| MC | mast cell |
| MHC | major histocompatibility complex |
| NF-κB | nuclear factor-κB |
| NK | natural killer |
| OVA | ovalbumin |
| PGD2 | prostaglandin D2 |
| PI3K | phosphatidylinositol-3-kinase |
| PKB/Akt | protein kinase B |
| pMHC | peptide-MHC |
| PUVA | psoralen and ultraviolet light A |
| RAF | rapidly accelerated fibrosarcoma |
| RAS | rat sarcoma |
| S1PR1 | sphingosine-1-phosphate receptor 1 |
| SCF | stem cell factor |
| VEGF | vascular endothelial growth factor |
| TGF-β | transforming growth factor-β |
| TLR3 | toll-like receptor 3 |
| TNF | tumour necrosis factor |
| TNFR | tumour necrosis factor receptor |
| TNRSF | TNF receptor superfamilies |
| TNSF | TNF superfamilies |
| TCM | central memory T cells |
| TEM | effector memory T cells |
| TRM | resident memory T cells |
References
- Elias, P.M. The skin barrier as an innate immune element. Semin. Immunopathol. 2007, 29, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.V.; Soulika, A. The dynamics of the skin’s immune system. Int. J. Mol. Sci. 2019, 20, 1811. [Google Scholar] [CrossRef] [PubMed]
- Flohr, C.; Hay, R. Putting the burden of skin diseases on the global map. Br. J. Dermatol. 2021, 184, 189–190. [Google Scholar] [CrossRef]
- Karimkhani, C.; Dellavalle, R.P.; Coffeng, L.E.; Flohr, C.; Hay, R.J.; Langan, S.M.; Nsoesie, E.O.; Ferrari, A.J.; Erskine, H.E.; Silverberg, J.I.; et al. Global skin disease morbidity and mortality: An update from the global burden of disease study 2013. JAMA Dermatol. 2017, 153, 406–412. [Google Scholar] [CrossRef]
- Gudjonsson, J.E.; Kabashima, K.; Eyerich, K. Mechanisms of skin autoimmunity: Cellular and soluble immune components of the skin. J. Allergy Clin. Immunol. 2020, 146, 8–16. [Google Scholar] [CrossRef]
- Hay, R.; Augustin, M.; Griffiths, C.; Sterry, W.; the Board of the International League of Dermatological Societies and the Grand Challenges Consultation Groups. The global challenge for skin health. Br. J. Dermatol. 2015, 172, 1469–1472. [Google Scholar] [CrossRef]
- Boniface, K.; Jacquemin, C.; Darrigade, A.-S.; Dessarthe, B.; Martins, C.; Boukhedouni, N.; Vernisse, C.; Grasseau, A.; Thiolat, D.; Rambert, J.; et al. Vitiligo Skin Is Imprinted with Resident Memory CD8 T Cells Expressing CXCR3. J. Investig. Dermatol. 2017, 138, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Lili, Y.; Yi, W.; Ji, Y.; Yue, S.; Weimin, S.; Ming, L. Global Activation of CD8+ Cytotoxic T Lymphocytes Correlates with an Impairment in Regulatory T Cells in Patients with Generalized Vitiligo. PLoS ONE 2012, 7, e37513. [Google Scholar] [CrossRef]
- Toothaker, J.M.; Olaloye, O.; McCourt, B.T.; McCourt, C.C.; Silva, T.N.; Case, R.M.; Liu, P.; Yimlamai, D.; Tseng, G.; Konnikova, L. Immune landscape of human placental villi using single-cell analysis. Development 2022, 149, dev200013. [Google Scholar] [CrossRef]
- Ho, A.W.; Kupper, T.S. T cells and the skin: From protective immunity to inflammatory skin disorders. Nat. Rev. Immunol. 2019, 19, 490–502. [Google Scholar] [CrossRef]
- Krystel-Whittemore, M.; Dileepan, K.N.; Wood, J.G. Mast Cell: A Multi-Functional Master Cell. Front. Immunol. 2016, 6, 620. [Google Scholar] [CrossRef] [PubMed]
- Kabashima, K.; Honda, T.; Ginhoux, F.; Egawa, G. The immunological anatomy of the skin. Nat. Rev. Immunol. 2018, 19, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Möbus, L.; Weidinger, S.; Emmert, H. Epigenetic factors involved in the pathophysiology of inflammatory skin diseases. J. Allergy Clin. Immunol. 2020, 145, 1049–1060. [Google Scholar] [CrossRef]
- Zhernakova, A.; van Diemen, C.C.; Wijmenga, C. Detecting shared pathogenesis from the shared genetics of immune-related diseases. Nat. Rev. Genet. 2009, 10, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Gentek, R.; Ghigo, C.; Hoeffel, G.; Bulle, M.J.; Msallam, R.; Gautier, G.; Launay, P.; Chen, J.; Ginhoux, F.; Bajénoff, M. Hemogenic Endothelial Fate Mapping Reveals Dual Developmental Origin of Mast Cells. Immunity 2018, 48, 1160–1171.e5. [Google Scholar] [CrossRef] [PubMed]
- Derakhshan, T.; Boyce, J.A.; Dwyer, D.F. Defining mast cell differentiation and heterogeneity through single-cell transcriptomics analysis. J. Allergy Clin. Immunol. 2022, 150, 739–747. [Google Scholar] [CrossRef]
- Zhang, Z.; Ernst, P.B.; Kiyono, H.; Kurashima, Y. Utilizing mast cells in a positive manner to overcome inflammatory and allergic diseases. Front. Immunol. 2022, 13, 937120. [Google Scholar] [CrossRef]
- Wernersson, S.; Pejler, G. Mast cell secretory granules: Armed for battle. Nat. Rev. Immunol. 2014, 14, 478–494. [Google Scholar] [CrossRef]
- McHale, C.; Mohammed, Z.; Gomez, G. Human Skin-Derived Mast Cells Spontaneously Secrete Several Angiogenesis-Related Factors. Front. Immunol. 2019, 10, 1445. [Google Scholar] [CrossRef]
- Das, D.; Akhtar, S.; Kurra, S.; Gupta, S.; Sharma, A. Emerging role of immune cell network in autoimmune skin disorders: An update on pemphigus, vitiligo and psoriasis. Cytokine Growth Factor Rev. 2019, 45, 35–44. [Google Scholar] [CrossRef]
- Frossi, B.; Mion, F.; Tripodo, C.; Colombo, M.P.; Pucillo, C.E. Rheostatic Functions of Mast Cells in the Control of Innate and Adaptive Immune Responses. Trends Immunol. 2017, 38, 648–656. [Google Scholar] [CrossRef] [PubMed]
- Nagata, Y.; Suzuki, R. FcεRI: A Master Regulator of Mast Cell Functions. Cells 2022, 11, 622. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 2012, 18, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Babina, M. The pseudo-allergic/neurogenic route of mast cell activation via MRGPRX2: Discovery, functional programs, regulation, relevance to disease, and relation with allergic stimulation. Itch 2020, 5, e32. [Google Scholar] [CrossRef]
- McNeil, B.D.; Pundir, P.; Meeker, S.; Han, L.; Undem, B.J.; Kulka, M.; Dong, X. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature 2014, 519, 237–241. [Google Scholar] [CrossRef]
- Mukai, K.; Tsai, M.; Saito, H.; Galli, S.J. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol. Rev. 2018, 282, 121–150. [Google Scholar] [CrossRef]
- Lundequist, A.; Pejler, G. Biological implications of preformed mast cell mediators. Cell. Mol. Life Sci. 2010, 68, 965–975. [Google Scholar] [CrossRef]
- Navi, D.; Saegusa, J.; Liu, F.-T. Mast Cells and Immunological Skin Diseases. Clin. Rev. Allergy Immunol. 2007, 33, 144–155. [Google Scholar] [CrossRef]
- Galli, S.J.; Grimbaldeston, M.A.; Tsai, M. Immunomodulatory mast cells: Negative, as well as positive, regulators of immunity. Nat. Rev. Immunol. 2008, 8, 478–486. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, G. Mast Cell and Autoimmune Diseases. Mediat. Inflamm. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- da Silva, E.Z.M.; Jamur, M.; Oliver, C. Mast cell function: A new vision of an old cell. J. Histochem. Cytochem. 2014, 62, 698–738. [Google Scholar] [CrossRef] [PubMed]
- Harvima, I.T.; Nilson, G.; Suttle, M.-M.; Naukkarinen, A. Is there a role for mast cells in psoriasis? Arch. Dermatol. Res. 2008, 300, 461–478. [Google Scholar] [CrossRef] [PubMed]
- Bertolini, M.; Zilio, F.; Rossi, A.; Kleditzsch, P.; Emelianov, V.E.; Gilhar, A.; Keren, A.; Meyer, K.C.; Wang, E.; Funk, W.; et al. Abnormal Interactions between Perifollicular Mast Cells and CD8+ T-Cells May Contribute to the Pathogenesis of Alopecia Areata. PLoS ONE 2014, 9, e94260. [Google Scholar] [CrossRef] [PubMed]
- Keith, Y.H.; Honda, T.; Ono, S.; Lee, B.; Shibuya, R.; Hanakawa, S.; Ishida, Y.; Nakamizo, S.; Kabashima, K. Infiltration and local differentiation of bone marrow-derived integrinβ7-positive mast cell progenitors in atopic dermatitis-like skin. J. Allergy Clin. Immunol. 2022, 151, 159–171.e8. [Google Scholar] [CrossRef] [PubMed]
- Keith, Y.H.; Honda, T.; Ono, S.; Lee, B.; Hanakawa, S.; Ishida, Y.; Nakamizo, S.; Kabashima, K. Circulating mast cell progenitors migrate to atopic dermatitis-like skin then adapt locally. bioRxiv 2022. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, Y.; Lin, J.; Li, X.; Yang, B.; Zhou, J. Immune Cell Infiltration Analysis Demonstrates Excessive Mast Cell Activation in Psoriasis. Front. Immunol. 2021, 12, 773280. [Google Scholar] [CrossRef]
- Wang, Z.; Babina, M. MRGPRX2 signals its importance in cutaneous mast cell biology: Does MRGPRX2 connect mast cells and atopic dermatitis? Exp. Dermatol. 2020, 29, 1104–1111. [Google Scholar] [CrossRef]
- van der Zee, H.; de Ruiter, L.; Boer, J.; Broecke, D.V.D.; Hollander, J.D.; Laman, J.; Prens, E. Alterations in leucocyte subsets and histomorphology in normal-appearing perilesional skin and early and chronic hidradenitis suppurativa lesions. Br. J. Dermatol. 2011, 166, 98–106. [Google Scholar] [CrossRef]
- Freire, P.C.; Muñoz, C. Stingl GIgE autoreactivity in bullous pemphigoid: Eosinophils and mast cells as major targets of pathogenic immune reactants. Br. J. Dermatol. 2017, 177, 1644–1653. [Google Scholar] [CrossRef]
- Toyry, S.; Fräki, J.; Tammi, R. Mast cell density in psoriatic skin. Eff. PUVA Corticosteroid Ther. Arch. Dermatol. Res. 1988, 280, 282–285. [Google Scholar] [CrossRef]
- Jiang, W.Y.; Chattedee, A.D.; Raychaudhuri, S.P.; Raychaudhuri, S.K.; Farber, E.M. Mast cell density and IL-8 expression in nonlesional and lesional psoriatic skin. Int. J. Dermatol. 2001, 40, 699–703. [Google Scholar] [CrossRef] [PubMed]
- Schubert, C.; Christophers, E. Mast cells and macrophages in early relapsing psoriasis. Arch. Dermatol. Res. 1985, 277, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Sabat, R.; Wolk, K.; Loyal, L.; Döcke, W.-D.; Ghoreschi, K. T cell pathology in skin inflammation. Semin. Immunopathol. 2019, 41, 359–377. [Google Scholar] [CrossRef] [PubMed]
- Osborn, J.F.; Hobbs, S.J.; Mooster, J.L.; Khan, T.N.; Kilgore, A.M.; Harbour, J.C.; Nolz, J.C. Central memory CD8+ T cells become CD69+ tissue-residents during viral skin infection independent of CD62L-mediated lymph node surveillance. PLoS Pathog. 2019, 15, e1007633. [Google Scholar] [CrossRef]
- Richmond, J.M.; Strassner, J.P.; Rashighi, M.; Agarwal, P.; Garg, M.; Essien, K.I.; Pell, L.S.; Harris, J.E. Resident Memory and Recirculating Memory T Cells Cooperate to Maintain Disease in a Mouse Model of Vitiligo. J. Investig. Dermatol. 2019, 139, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Szabo, P.A.; Miron, M.; Farber, D.L. Location, location, location: Tissue resident memory T cells in mice and humans. Sci. Immunol. 2019, 4, aas9673. [Google Scholar] [CrossRef]
- Klicznik, M.M.; Morawski, P.A.; Höllbacher, B.; Varkhande, S.R.; Motley, S.J.; Kuri-Cervantes, L.; Goodwin, E.; Rosenblum, M.D.; Long, S.A.; Bratchl, G.; et al. Human CD4+ CD103+ cutaneous resident memory T cells are found in the circulation of healthy individuals. Sci. Immunol. 2019, 4, eaav8995. [Google Scholar] [CrossRef]
- Watanabe, R. Protective and pathogenic roles of resident memory T cells in human skin disorders. J. Dermatol. Sci. 2019, 95, 2–7. [Google Scholar] [CrossRef]
- Chen, L.; Shen, Z. Tissue-resident memory T cells and their biological characteristics in the recurrence of inflammatory skin disorders. Cell. Mol. Immunol. 2019, 17, 64–75. [Google Scholar] [CrossRef]
- Mueller, S.; Zaid, A.; Carbone, F.R. Tissue-Resident T Cells: Dynamic Players in Skin Immunity. Front. Immunol. 2014, 5, 332. [Google Scholar] [CrossRef]
- Gehad, A.; Elco, C.P.; Schlapbach, C.; Huang, V.; Kupper, T.S.; Watanabe, R.; Scott, L.L.; Matos, T.R.; A Clark, R.; E Teague, J.; et al. Human skin is protected by four functionally and phenotypically discrete populations of resident and recirculating memory T cells. Sci. Transl. Med. 2015, 7, 279ra39. [Google Scholar] [CrossRef]
- Behr, F.M.; Parga-Vidal, L.; Kragten, N.A.M.; Van Dam, T.J.P.; Wesselink, T.H.; Sheridan, B.S.; Arens, R.; Van Lier, R.A.W.; Stark, R.; Van Gisbergen, K.P.J.M. Tissue-resident memory CD8+ T cells shape local and systemic secondary T cell responses. Nat. Immunol. 2020, 21, 1070–1081. [Google Scholar] [CrossRef] [PubMed]
- Hijnen, D.; Knol, E.F.; Gent, Y.Y.; Giovannone, B.; Bejin, S.J.P.; Kupper, T.S.; Bruijnzeel-Koomen, C.A.F.M.; Clark, R.A. CD8+ T cells in the lesional skin of atopic dermatitis and psoriasis patients are an important source of IFN-γ, IL-13, IL-17, and IL-22. J. Investig. Dermatol. 2013, 133, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Cheuk, S.; Schlums, H.; Sérézal, I.G.; Martini, E.; Chiang, S.C.; Marquardt, N.; Gibbs, A.; Detlofsson, E.; Introini, A.; Forkel, M.; et al. CD49a Expression Defines Tissue-Resident CD8+ T Cells Poised for Cytotoxic Function in Human Skin. Immunity 2017, 46, 287–300. [Google Scholar] [CrossRef]
- Hirai, T.; Whitley, S.K.; Kaplan, D.H. Migration and Function of Memory CD8+ T Cells in Skin. J. Investig. Dermatol. 2019, 140, 748–755. [Google Scholar] [CrossRef]
- Hirai, T.; Zenke, Y.; Yang, Y.; Bartholin, L.; Beura, L.K.; Masopust, D.; Kaplan, D.H. Keratinocyte-Mediated Activation of the Cytokine TGF-β Maintains Skin Recirculating Memory CD8+ T Cells. Immunity 2019, 50, 1249–1261.e5. [Google Scholar] [CrossRef]
- Di Meglio, P.; Villanova, F.; Navarini, A.A.; Mylonas, A.; Tosi, I.; Nestle, F.O.; Conrad, C. Targeting CD8+ T cells prevents psoriasis development. J. Allergy Clin. Immunol. 2016, 138, 274–276.e6. [Google Scholar] [CrossRef]
- Emmanuel, T.; Mistegård, J.; Bregnhøj, A.; Johansen, C.; Iversen, L. Tissue-Resident Memory T Cells in Skin Diseases: A Systematic Review. Int. J. Mol. Sci. 2021, 22, 9004. [Google Scholar] [CrossRef]
- Boorn, J.G.V.D.; Konijnenberg, D.; Dellemijn, T.A.; van der Veen, J.W.; Bos, J.D.; Melief, C.J.; Vyth-Dreese, F.A.; Luiten, R.M. Autoimmune Destruction of Skin Melanocytes by Perilesional T Cells from Vitiligo Patients. J. Investig. Dermatol. 2009, 129, 2220–2232. [Google Scholar] [CrossRef]
- Gunderson, A.J.; Mohammed, J.; Horvath, F.J.; Podolsky, M.A.; Anderson, C.R.; Glick, A.B. CD8(+) T cells mediate RAS-induced psoriasis-like skin inflammation through IFN-γ. J. Investig. Dermatol. 2013, 133, 955–963. [Google Scholar] [CrossRef]
- Killeen, M.E.; Ferris, L.; Kupetsky, E.A.; Falo, L.; Mathers, A.R. Signaling through Purinergic Receptors for ATP Induces Human Cutaneous Innate and Adaptive Th17 Responses: Implications in the Pathogenesis of Psoriasis. J. Immunol. 2013, 190, 4324–4336. [Google Scholar] [CrossRef]
- da Silva, H.B.; Peng, C.; Wang, H.; Wanhainen, K.M.; Ma, C.; Lopez, S.; Khoruts, A.; Zhang, N.; Jameson, S.C. Sensing of ATP via the purinergic receptor P2RX7 promotes CD8+ Trm cell generation by enhancing their sensitivity to the cytokine TGF-β. Immunity 2020, 53, 158–171. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, H.B.; Beura, L.K.; Wang, H.; Hanse, E.A.; Gore, R.; Scott, M.C.; Walsh, D.A.; Block, K.E.; Fonseca, R.; Yan, Y.; et al. The purinergic receptor P2RX7 directs metabolic fitness of long-lived memory CD8+ T cells. Nature 2018, 559, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Stark, R.; Wesselink, T.H.; Behr, F.M.; Kragten, N.A.M.; Arens, R.; Koch-Nolte, F.; van Gisbergen, K.P.J.M.; van Lier, R.A.W. TRM maintenance is regulated by tissue damage via P2RX7. Sci. Immunol. 2018, 3, eaau1022. [Google Scholar] [CrossRef] [PubMed]
- Willemsen, M.; Linkutė, R.; Luiten, R.M.; Matos, T.R. Skin-resident memory T cells as a potential new therapeutic target in vitiligo and melanoma. Pigment. Cell Melanoma Res. 2019, 32, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Wijngaard, R.V.D.; Wankowicz-Kalinska, A.; Le Poole, C.; Tigges, B.; Westerhof, W.; Das, P. Local Immune Response in Skin of Generalized Vitiligo Patients. Lab. Investig. 2000, 80, 1299–1309. [Google Scholar] [CrossRef]
- Suárez-Fariñas, M.; Fuentes-Duculan, J.; A Lowes, M.; Krueger, J.G. Resolved Psoriasis Lesions Retain Expression of a Subset of Disease-Related Genes. J. Investig. Dermatol. 2011, 131, 391–400. [Google Scholar] [CrossRef]
- Funch, A.B.; Mraz, V.; Gadsbøll, A.S.Ø.; Jee, M.H.; Weber, J.F.; Ødum, N.; Woetmann, A.; Johansen, J.D.; Geisler, C.; Bonefeld, C.M. CD8+ tissue-resident memory T cells recruit neutrophils that are essential for flare-ups in contact dermatitis. Allergy 2022, 77, 513–524. [Google Scholar] [CrossRef]
- Puig, L.; Costanzo, A.; Muñoz-Elías, E.J.; Jazra, M.; Wegner, S.; Paul, C.F.; Conrad, C. The biological basis of disease recurrence in psoriasis: A historical perspective and current models. Br. J. Dermatol. 2021, 186, 773–781. [Google Scholar] [CrossRef]
- Wakim, L.M.; Waithman, J.; van Rooijen, N.; Heath, W.R.; Carbone, F.R. Dendritic Cell-Induced Memory T Cell Activation in Nonlymphoid Tissues. Science 2008, 319, 198–202. [Google Scholar] [CrossRef]
- Matos, T.R.; O’Malley, J.T.; Lowry, E.L.; Hamm, D.; Kirsh, I.R.; Robins, H.S.; Kupper, T.S.; Krueger, J.G.; Clark, R.A. Clinically resolved psoriatic lesions contain psoriasis-specific IL-17–producing αβ T cell clones. J. Clin. Investig. 2017, 127, 4031–4041. [Google Scholar] [CrossRef] [PubMed]
- Cheuk, S.; Wikén, M.; Blomqvist, L.; Nylén, S.; Talme, T.; Ståhle, M.; Eidsmo, L. Epidermal Th22 and Tc17 Cells Form a Localized Disease Memory in Clinically Healed Psoriasis. J. Immunol. 2014, 192, 3111–3120. [Google Scholar] [CrossRef] [PubMed]
- Iversen, L.; Eidsmo, L.; Austad, J.; de Rie, M.; Osmancevic, A.; Skov, L.; Talme, T.; Bachmann, I.; van de Kerkhof, P.; Stahle, M.; et al. Secukinumab treatment in new-onset psoriasis: Aiming to understand the potential for disease modification–rationale and design of the randomized, multicenter STEPI n study. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 1930–1939. [Google Scholar] [CrossRef] [PubMed]
- Adachi, T.; Kobayashi, T.; Sugihara, E.; Yamada, T.; Ikuta, K.; Pittaluga, S.; Saya, H.; Amagai, M.; Nagao, K. Hair follicle–derived IL-7 and IL-15 mediate skin-resident memory T cell homeostasis and lymphoma. Nat. Med. 2015, 21, 1272–1279. [Google Scholar] [CrossRef]
- Jarjour, N.N.; Wanhainen, K.M.; Peng, C.; Gavil, N.V.; Maurice, N.J.; da Silva, H.B.; Martinez, R.J.; Dalzell, T.S.; Huggins, M.A.; Masopust, D.; et al. Responsiveness to interleukin-15 therapy is shared between tissue-resident and circulating memory CD8 + T cell subsets. Proc. Natl. Acad. Sci. USA 2022, 119, e2209021119. [Google Scholar] [CrossRef] [PubMed]
- Behr, F.M.; Chuwonpad, A.; Stark, R.; Van Gisbergen, K.P.J.M. Armed and Ready: Transcriptional Regulation of Tissue-Resident Memory CD8 T Cells. Front. Immunol. 2018, 9, 1770. [Google Scholar] [CrossRef]
- Park, S.L.; Zaid, A.; Hor, J.L.; Christo, S.N.; Prier, J.E.; Davies, B.; Alexandre, Y.O.; Gregory, J.L.; Russell, T.A.; Gebhardt, T.; et al. Local proliferation maintains a stable pool of tissue-resident memory T cells after antiviral recall responses. Nat. Immunol. 2018, 19, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Russi, A.E.; Walker-Caulfield, M.E.; Guo, Y.; Lucchinetti, C.F.; Brown, M.A. Meningeal mast cell-T cell crosstalk regulates T cell encephalitogenicity. J. Autoimmun. 2016, 73, 100–110. [Google Scholar] [CrossRef]
- Friedman, M.; Kaliner, M. In situ degranulation of human nasal mucosal mast cells: Ultrastructural features and cell-cell associations. J. Allergy Clin. Immunol. 1985, 76, 70–82. [Google Scholar] [CrossRef]
- Stelekati, E.; Bahri, R.; D’Orlando, O.; Orinska, Z.; Mittrücker, H.-W.; Langenhaun, R.; Glatzel, M.; Bollinger, A.; Paus, R.; Bulfone-Paus, S. Mast Cell-Mediated Antigen Presentation Regulates CD8+ T Cell Effector Functions. Immunity 2009, 31, 665–676. [Google Scholar] [CrossRef]
- Ott, V.L.; Cambier, J.; Kappler, J.W.; Marrack, P.; Swanson, B.J. Mast cell–dependent migration of effector CD8+ T cells through production of leukotriene B4. Nat. Immunol. 2003, 4, 974–981. [Google Scholar] [CrossRef] [PubMed]
- McLachlan, J.B.; Hart, J.P.; Pizzo, S.V.; Shelburne, C.P.; Staats, H.; Gunn, M.; Abraham, S.N. Mast cell–derived tumor necrosis factor induces hypertrophy of draining lymph nodes during infection. Nat. Immunol. 2003, 4, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Podlech, J.; Ebert, S.; Becker, M.; Reddehase, M.; Stassen, M.; Lemmermann, N.A.W. Mast cells: Innate attractors recruiting protective CD8 T cells to sites of cytomegalovirus infection. Med. Microbiol. Immunol. 2015, 204, 327–334. [Google Scholar] [CrossRef]
- Nakae, S.; Suto, H.; Iikura, M.; Kakurai, M.; Sedgwick, J.D.; Tsai, M.; Galli, S.J. Mast Cells Enhance T Cell Activation: Importance of Mast Cell Costimulatory Molecules and Secreted TNF. J. Immunol. 2006, 176, 2238–2248. [Google Scholar] [CrossRef] [PubMed]
- Gregory, G.D.; Robbie-Ryan, M.; Secor, V.H.; Sabatino, J.J., Jr.; Brown, M.A. Mast cells are required for optimal autoreactive T cell responses in a murine model of multiple sclerosis. Eur. J. Immunol. 2005, 35, 3478–3486. [Google Scholar] [CrossRef]
- Hackler, Y.; Siebenhaar, F.; Löhning, M.; Maurer, M.; Muñoz, M. Mast Cells Modulate Antigen-Specific CD8+ T Cell Activation During LCMV Infection. Front. Immunol. 2021, 12, 688347. [Google Scholar] [CrossRef]
- Komi, D.E.A.; Grauwet, K. Role of Mast Cells in Regulation of T Cell Responses in Experimental and Clinical Settings. Clin. Rev. Allergy Immunol. 2017, 54, 432–445. [Google Scholar] [CrossRef]
- Nakae, S.; Suto, H.; Kakurai, M.; Sedgwick, J.D.; Tsai, M.; Galli, S.J. Mast cells enhance T cell activation: Importance of mast cell-derived TNF. Proc. Natl. Acad. Sci. USA 2005, 102, 6467–6472. [Google Scholar] [CrossRef]
- Burkett, M.W.; Shafer-Weaver, K.A.; Strobl, S.; Baseler, M.; Malyguine, A. A Novel Flow Cytometric Assay for Evaluating Cell-Mediated Cytotoxicity. J. Immunother. 2005, 28, 396–402. [Google Scholar] [CrossRef]
- Mekori, Y.A.; Metcalfe, D. Mast cell–T cell interactions. J. Allergy Clin. Immunol. 1999, 104, 517–523. [Google Scholar] [CrossRef]
- McCoy-Simandle, K.; Hanna, S.J.; Cox, D. Exosomes and nanotubes: Control of immune cell communication. Int. J. Biochem. Cell Biol. 2015, 71, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Adil, M.S.; Narayanan, S.P.; Somanath, P.R. Cell-cell junctions: Structure and regulation in physiology and pathology. Tissue Barriers 2020, 9, 1848212. [Google Scholar] [CrossRef] [PubMed]
- Huppa, J.B.; Davis, M.M. T-cell-antigen recognition and the immunological synapse. Nat. Rev. Immunol. 2003, 3, 973–983. [Google Scholar] [CrossRef]
- Joulia, R.; Gaudenzio, N.; Rodrigues, M.; Lopez, J.; Blanchard, N.; Valitutti, S.; Espinosa, E. Mast cells form antibody-dependent degranulatory synapse for dedicated secretion and defence. Nat. Commun. 2015, 6, 6174. [Google Scholar] [CrossRef]
- Bulfone-Paus, S.; Bahri, R. Mast Cells as Regulators of T Cell Responses. Front. Immunol. 2015, 6, 394. [Google Scholar] [CrossRef]
- Garcia, E.; Ismail, S. Spatiotemporal Regulation of Signaling: Focus on T Cell Activation and the Immunological Synapse. Int. J. Mol. Sci. 2020, 21, 3283. [Google Scholar] [CrossRef]
- Schuijs, M.J.; Hammad, H.; Lambrecht, B. Professional and ‘amateur’antigen-presenting cells in type 2 immunity. Trends Immunol. 2019, 40, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Katsoulis-Dimitriou, K.; Kotrba, J.; Voss, M.; Dudeck, J.; Dudeck, A. Mast Cell Functions Linking Innate Sensing to Adaptive Immunity. Cells 2020, 9, 2538. [Google Scholar] [CrossRef] [PubMed]
- Lotfi-Emran, S.; Ward, B.R.; Le, Q.T.; Pozez, A.L.; Manjili, M.H.; Woodfolk, J.A.; Schwartz, L.B. Human mast cells present antigen to autologous CD4+ T cells. J. Allergy Clin. Immunol. 2018, 141, 311–321.e10. [Google Scholar] [CrossRef] [PubMed]
- Orinska, Z.; Bulanova, E.; Budagian, V.; Metz, M.; Maurer, M.; Bulfone-Paus, S. TLR3-induced activation of mast cells modulates CD8+ T-cell recruitment. Blood 2005, 106, 978–987. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, K.; Hida, S.; Weng, Y.; Saiura, A.; Sato, K.; Takayanagi, H.; Sakaguchi, S.; Yokochi, T.; Kodama, T.; Naitoh, M. Requirement of the IFN-α/β-induced CXCR3 chemokine signalling for CD8+ T cell activation. Genes Cells 2002, 7, 309–320. [Google Scholar] [CrossRef]
- Muntjewerff, E.M.; Meesters, L.D.; Bogaart, G.V.D.; Revelo, N.H. Reverse Signaling by MHC-I Molecules in Immune and Non-Immune Cell Types. Front. Immunol. 2020, 11, 605958. [Google Scholar] [CrossRef] [PubMed]
- Malbec, O.; Cassard, L.; Albanesi, M.; Jönsson, F.; Mancardi, D.; Chicanne, G.; Payrastre, B.; Dubreuil, P.; Vivier, E.; Daëron, M. Trans-inhibition of activation and proliferation signals by Fc receptors in mast cells and basophils. Sci. Signal. 2016, 9, 126. [Google Scholar] [CrossRef]
- Carroll-Portillo, A.; Cannon, J.L.; Riet, J.T.; Holmes, A.M.; Kawakami, Y.; Kawakami, T.; Cambi, A.; Lidke, D.S. Mast cells and dendritic cells form synapses that facilitate antigen transfer for T cell activation. J. Cell Biol. 2015, 210, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Maurer, M.; Kostka, S.L.; Siebenhaar, F.; Moelle, K.; Metz, M.; Knop, J.; Stebut, E. Skin mast cells control T cell-dependent host defense in Leishmania major infections. FASEB J. 2006, 20, 2460–2467. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Janmey, P.A.; Finkel, T.H. The receptor deformation model of TCR triggering. FASEB J. 2007, 22, 1002–1008. [Google Scholar] [CrossRef]
- Kaizuka, Y. Regulations of T Cell Activation by Membrane and Cytoskeleton. Membranes 2020, 10, 443. [Google Scholar] [CrossRef]
- Gaudenzio, N.; Espagnolle, N.; Mars, L.T.; Liblau, R.; Valitutti, S.; Espinosa, E. Cell-cell cooperation at the T helper cell/mast cell immunological synapse. Blood 2009, 114, 4979–4988. [Google Scholar] [CrossRef]
- Kumari, S.; Colin-York, H.; Irvine, D.J.; Fritzsche, M. Not All T Cell Synapses Are Built the Same Way. Trends Immunol. 2019, 40, 977–980. [Google Scholar] [CrossRef]
- Dustin, M.L.; Choudhuri, K. Signaling and Polarized Communication Across the T Cell Immunological Synapse. Annu. Rev. Cell Dev. Biol. 2016, 32, 303–325. [Google Scholar] [CrossRef]
- Harjunpää, H.; Asens, M.L.; Guenther, C.; Fagerholm, S.C. Cell Adhesion Molecules and Their Roles and Regulation in the Immune and Tumor Microenvironment. Front. Immunol. 2019, 10, 1078. [Google Scholar] [CrossRef]
- Kashiwakura, J.-I.; Yokoi, H.; Saito, H.; Okayama, Y. T Cell Proliferation by Direct Cross-Talk between OX40 Ligand on Human Mast Cells and OX40 on Human T Cells: Comparison of Gene Expression Profiles between Human Tonsillar and Lung-Cultured Mast Cells. J. Immunol. 2004, 173, 5247–5257. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.A.; Bevan, M.J. Effector and Memory CTL Differentiation. Annu. Rev. Immunol. 2007, 25, 171–192. [Google Scholar] [CrossRef] [PubMed]
- Rudd, C.E.; Taylor, A.; Schneider, H. CD28 and CTLA-4 coreceptor expression and signal transduction. Immunol. Rev. 2009, 229, 12–26. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Lin, Q.; Zhang, Z.; Zhang, L. Therapeutic strategies for the costimulatory molecule OX40 in T-cell-mediated immunity. Acta Pharm. Sin. B 2019, 10, 414–433. [Google Scholar] [CrossRef] [PubMed]
- Croft, M.; So, T.; Duan, W.; Soroosh, P. The significance of OX40 and OX40L to T-cell biology and immune disease. Immunol. Rev. 2009, 229, 173–191. [Google Scholar] [CrossRef]
- Huang, C.; Chen, A.; Tu, Y.; Liu, Z. Expression of CD28/B7 in the Lesional Skin and Peripheral Lymphocytes in Psoriasis. Chin. J. Dermatol. 2003, 12. [Google Scholar]
- Elsner, J.S.; Carlsson, M.; Stougaard, J.K.; Nygaard, U.; Buchner, M.; Fölster-Holst, R.; Hvid, M.; Vestergaard, C.; Deleuran, M.; Deleuran, B. The OX40 Axis is Associated with Both Systemic and Local Involvement in Atopic Dermatitis. Acta Derm. Venereol. 2020, 100, adv00099. [Google Scholar] [CrossRef]
- Yoo, J.K.; Choo, Y.-K.; Kwak, D.H.; Lee, J.M.; Lim, C.-Y.; Lee, J.-H.; Park, M.-Y.; Kim, C.-H. Protective effects of agonistic anti-4-1BB antibody on the development of imiquimod-induced psoriasis-like dermatitis in mice. Immunol. Lett. 2016, 178, 131–139. [Google Scholar] [CrossRef]
- Boomer, J.S.; Green, J. An enigmatic tail of CD28 signaling. Cold Spring Harb. Perspect. Biol. 2010, 2, a002436. [Google Scholar] [CrossRef]
- Fuse, S.; Zhang, W.; Usherwood, E. Control of memory CD8+ T cell differentiation by CD80/CD86-CD28 costimulation and restoration by IL-2 during the recall response. J. Immunol. 2008, 180, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- Nandi, D.; Pathak, S.; Verma, T.; Singh, M.; Chattopadhyay, A.; Thakur, S.; Raghavan, A.; Vijayamahantesh, G.A. T cell costimulation, checkpoint inhibitors and anti-tumor therapy. J. Biosci. 2020, 45, 50. [Google Scholar] [CrossRef]
- Alegre, M.-L.; Frauwirth, K.A.; Thompson, C.B. T-cell regulation by CD28 and CTLA-4. Nat. Rev. Immunol. 2001, 1, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Jadav, G.; Doodipala, M. The Role of Cd28 Costimulatory Receptors in Signalling Pathways. Int. J. Health Policy Plan. 2022, 46, 54. [Google Scholar]
- Bromley, S.K.; Iaboni, A.; Davis, S.J.; Whitty, A.; Green, J.M.; Shaw, A.; Weiss, A.; Dustin, M. The immunological synapse and CD28-CD80 interactions. Nat. Immunol. 2001, 2, 1159–1166. [Google Scholar] [CrossRef]
- Zumerle, S.; Molon, B.; Viola, A. Membrane Rafts in T Cell Activation: A Spotlight on CD28 Costimulation. Front. Immunol. 2017, 8, 1467. [Google Scholar] [CrossRef]
- Halstead, E.S.; Mueller, Y.M.; Altman, J.D.; Katsikis, P.D. In vivo stimulation of CD137 broadens primary antiviral CD8+ T cell responses. Nat. Immunol. 2002, 3, 536–541. [Google Scholar] [CrossRef]
- Williams, M.A.; Tyznik, A.J.; Bevan, M.J. Interleukin-2 signals during priming are required for secondary expansion of CD8+ memory T cells. Nature 2006, 441, 890–893. [Google Scholar] [CrossRef]
- Mou, D.; Espinosa, J.; Lo, D.J.; Kirk, A.D. CD28 negative T cells: Is their loss our gain? Am. J. Transplant. 2014, 14, 2460–2466. [Google Scholar] [CrossRef]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schad, D.; Hassel, J.C.; et al. Improv. Surviv. Ipilimumab Patients Metastatic Melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef]
- Iannone, F.; Lapadula, G. The Inhibitor of Costimulation of T Cells: Abatacept. J. Rheumatol. 2012, 89, 100–102. [Google Scholar] [CrossRef] [PubMed]
- Parisi, R.; Iskandar, I.Y.K.; Kontopantelis, E.; Augustin, M.; Griffiths, C.E.M.; Ashcroft, D.M. National, regional, and worldwide epidemiology of psoriasis: Systematic analysis and modelling study. BMJ 2020, 369, m1590. [Google Scholar] [CrossRef] [PubMed]
- Rendon, A.; Schäkel, K. Psoriasis pathogenesis and treatment. Int. J. Mol. Sci. 2019, 20, 1475. [Google Scholar] [CrossRef]
- Kim, W.B.; Jerome, D.; Yeung, J. Diagnosis and management of psoriasis. Can. Fam. Phys. 2017, 63, 278–285. [Google Scholar]
- Van der Fits, L.; Mourits, S.; Voerman, J.S.A.; Kant, M.; Boon, L.; Laman, J.D.; Cornelissen, F.; Mus, A.-M.; Florencia, E.; Prens, E.P.; et al. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J. Immunol. 2009, 182, 5836–5845. [Google Scholar] [CrossRef]
- Pandey, R.; Al-Nuaimi, Y.; Mishra, R.K.; Spurgeon, S.K.; Goodfellow, M. Role of subnetworks mediated by TNF α, IL-23/IL-17 and IL-15 in a network involved in the pathogenesis of psoriasis. Sci. Rep. 2021, 11, 2204. [Google Scholar] [CrossRef]
- Kamata, M.; Tada, Y. Efficacy and Safety of Biologics for Psoriasis and Psoriatic Arthritis and Their Impact on Comorbidities: A Literature Review. Int. J. Mol. Sci. 2020, 21, 1690. [Google Scholar] [CrossRef]
- Rosman, Z.; Shoenfeld, Y.; Zandman-Goddard, G. Biologic therapy for autoimmune diseases: An update. BMC Med. 2013, 11, 88. [Google Scholar] [CrossRef]
- Brody, I. Mast Cell Degranulation in the Evolution of Acute Eruptive Guttate Psoriasis Vulgaris. J. Investig. Dermatol. 1984, 82, 460–464. [Google Scholar] [CrossRef]
- Abrams, J.R.; Lebwohl, M.G.; Guzzo, C.A.; Jegasothy, B.V.; Goldfarb, M.T.; Goffe, B.S.; Menter, A.; Lowe, N.J.; Krueger, G.; Brown, M.J.; et al. CTLA4Ig-mediated blockade of T-cell costimulation in patients with psoriasis vulgaris. J. Clin. Investig. 1999, 103, 1243–1252. [Google Scholar] [CrossRef]
- Harris, K.M.; Smilek, D.E.; Byron, M.; Lim, N.; Barry, W.T.; McNamara, J.; Garcet, S.; Konrad, R.J.; Stengelin, M.; Bathala, P.; et al. Effect of Costimulatory Blockade with Abatacept After Ustekinumab Withdrawal in Patients with Moderate to Severe Plaque Psoriasis: The PAUSE Randomized Clinical Trial. JAMA Dermatol. 2021, 157, 1306–1315. [Google Scholar] [CrossRef] [PubMed]
- Webb, G.J.; Hirschfield, G.M.; Lane, P.J.L. OX40, OX40L and Autoimmunity: A Comprehensive Review. Clin. Rev. Allergy Immunol. 2016, 50, 312–332. [Google Scholar] [CrossRef] [PubMed]
- Kotani, A.; Hori, T.; Fujita, T.; Kambe, N.; Matsumura, Y.; Ishikawa, T.; Miyachi, Y.; Nagai, K.; Tanaka, Y.; Uchiyama, T. Involvement of OX40 ligand+ mast cells in chronic GVHD after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2007, 39, 373–375. [Google Scholar] [CrossRef]
- Sayama, K.; Diehn, M.; Matsuda, K.; Lunderius, C.; Tsai, M.; Tam, S.-Y.; Botstein, D.; O Brown, P.; Galli, S.J. Transcriptional response of human mast cells stimulated via the FcεRI and identification of mast cells as a source of IL-11. BMC Immunol. 2002, 3, 5. [Google Scholar] [CrossRef]
- Redmond, W.; Ruby, C.E.; Weinberg, A.D. The Role of OX40-Mediated Co-stimulation in T-Cell Activation and Survival. Crit. Rev. Immunol. 2009, 29, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Bansal-Pakala, P.; Halteman, B.S.; Cheng, M.H.; Croft, M. Costimulation of CD8 T Cell Responses by OX40. J. Immunol. 2004, 172, 4821–4825. [Google Scholar] [CrossRef]
- Gramaglia, I.; Jember, A.; Pippig, S.D.; Weinberg, A.D.; Killeen, N.; Croft, M. The OX40 Costimulatory Receptor Determines the Development of CD4 Memory by Regulating Primary Clonal Expansion. J. Immunol. 2000, 165, 3043–3050. [Google Scholar] [CrossRef]
- Silverberg, J.I.; Barbarot, S.; Gadkari, A.; Simpson, E.L.; Weidinger, S.; Mina-Osorio, P.; Rossi, A.B.; Brignoli, L.; Saba, G.; Guillemin, I.; et al. Atopic dermatitis in the pediatric population: A cross-sectional, international epidemiologic study. Ann. Allergy Asthma Immunol. 2021, 126, 417–428. [Google Scholar] [CrossRef]
- Barbarot, S.; Auziere, S.; Gadkari, A.; Girolomoni, G.; Puig, L.; Simpson, E.L.; Margolis, D.J.; De Bruin-Weller, M.; Eckert, L. Epidemiology of atopic dermatitis in adults: Results from an international survey. Allergy 2018, 73, 1284–1293. [Google Scholar] [CrossRef]
- Schneider, L.; Hanifin, J.; Boguniewicz, M.; Eichenfield, L.F.; Spergel, J.M.; Dakovic, R.; Paller, A.S. Study of the Atopic March: Development of Atopic Comorbidities. Pediatr. Dermatol. 2016, 33, 388–398. [Google Scholar] [CrossRef]
- Paller, A.; Jaworski, J.C.; Simpson, E.L.; Boguniewicz, M.; Russell, J.J.; Block, J.K.; Tofte, S.; Dunn, J.D.; Feldman, S.R.; Clark, A.R.; et al. Major Comorbidities of Atopic Dermatitis: Beyond Allergic Disorders. Am. J. Clin. Dermatol. 2018, 19, 821–838. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, B.E.; Leung, D.Y.M. Pathophysiology of atopic dermatitis: Clinical implications. Allergy Asthma Proc. 2019, 40, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, T.; Ando, T.; Kimura, M.; Wilson, B.S.; Kawakami, Y. Mast cells in atopic dermatitis. Curr. Opin. Immunol. 2009, 21, 666–678. [Google Scholar] [CrossRef] [PubMed]
- Mashiko, S.; Bouguermouh, S.; Rubio, M.; Baba, N.; Bissonnette, R.; Sarfati, M. Human mast cells are major IL-22 producers in patients with psoriasis and atopic dermatitis. J. Allergy Clin. Immunol. 2015, 136, 351–359.e1. [Google Scholar] [CrossRef]
- Furue, M.; Furue, M. OX40L–OX40 signaling in atopic dermatitis. J. Clin. Med. 2021, 10, 2578. [Google Scholar] [CrossRef]
- Bieber, T. Atopic dermatitis: An expanding therapeutic pipeline for a complex disease. Nat. Rev. Drug Discov. 2021, 21, 21–40. [Google Scholar] [CrossRef]
- Nakagawa, H.; Iizuka, H.; Nemoto, O.; Shimabe, M.; Furukawa, Y.; Kikuta, N.; Ootaki, K. Safety, tolerability and efficacy of repeated intravenous infusions of KHK4083, a fully human anti-OX40 monoclonal antibody, in Japanese patients with moderate to severe atopic dermatitis. J. Dermatol. Sci. 2020, 99, 82–89. [Google Scholar] [CrossRef]
- Strassner, J.; Rashighi, M.; Refat, M.A.; Richmond, J.M.; Harris, J.E. Suction blistering the lesional skin of vitiligo patients reveals useful biomarkers of disease activity. J. Am. Acad. Dermatol. 2017, 76, 847–855.e5. [Google Scholar] [CrossRef]
- Kuo, P.T.; Zeng, Z.; Salim, N.; Mattarollo, S.; Wells, J.W.; Leggatt, G.R. The Role of CXCR3 and Its Chemokine Ligands in Skin Disease and Cancer. Front. Med. 2018, 5, 271. [Google Scholar] [CrossRef]
- Chong, S.; Lan, H.; Zeng, K.; Zhao, X. Serum Fractalkine (CX3CL1) Concentration Correlates with Clinical Severity in Pediatric Atopic Dermatitis Patients. Ann. Clin. Lab. Sci. 2016, 46, 168–173. [Google Scholar]
- Wang, X.X.; Wang, Q.Q.; Wu, J.Q.; Jiang, M.; Chen, L.; Zhang, C.F.; Xiang, L.H. Increased expression of CXCR 3 and its ligands in patients with vitiligo and CXCL 10 as a potential clinical marker for vitiligo. Br. J. Dermatol. 2016, 174, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Niño, J.L.G.; Pageon, S.V.; Tay, S.S.; Colakoglu, F.; Kempe, D.; Hywood, J.; Mazalo, J.K.; Cremasco, J.; A Govendir, M.; Dagley, L.F.; et al. Cytotoxic T cells swarm by homotypic chemokine signalling. eLife 2020, 9, 56554. [Google Scholar] [CrossRef] [PubMed]
- Griffith, J.W.; Sokol, C.L.; Luster, A.D. Chemokines and Chemokine Receptors: Positioning Cells for Host Defense and Immunity. Annu. Rev. Immunol. 2014, 32, 659–702. [Google Scholar] [CrossRef] [PubMed]
- McAlpine, S.M.; Issekutz, T.B.; Marshall, J.S. Virus stimulation of human mast cells results in the recruitment of CD56 + T cells by a mechanism dependent on CCR5 ligands. FASEB J. 2011, 26, 1280–1289. [Google Scholar] [CrossRef]
- Zaid, A.; Hor, J.L.; Christo, S.N.; Groom, J.R.; Heath, W.R.; Mackay, L.K.; Mueller, S.N. Chemokine receptor–dependent control of skin tissue–resident memory T cell formation. J. Immunol. 2017, 199, 2451–2459. [Google Scholar] [CrossRef]
- Alanio, C.; da Silva, R.B.; Michonneau, D.; Bousso, P.; Ingersoll, M.A.; Albert, M.L. CXCR3/CXCL10 axis shapes tissue distribution of memory phenotype CD8+ T cells in nonimmunized mice. J. Immunol. 2018, 200, 139–146. [Google Scholar] [CrossRef]
- Sung, J.H.; Zhang, H.; Moseman, E.A.; Alvarez, D.; Iannacone, M.; Henrickson, S.E.; de la Torre, J.C.; Groom, J.R.; Luster, A.D.; von Andrian, U.H. Chemokine Guidance of Central Memory T Cells Is Critical for Antiviral Recall Responses in Lymph Nodes. Cell 2012, 150, 1249–1263. [Google Scholar] [CrossRef]
- Clark, A.K.; Staniland, A.; Malcangio, M. Fractalkine/CX3CR1 signalling in chronic pain and inflammation. Curr. Pharm. Biotechnol. 2011, 12, 1707–1714. [Google Scholar] [CrossRef]
- Imai, T.; Hieshima, K.; Haskell, C.; Baba, M.; Nagira, M.; Nishimura, M.; Kakizaki, M.; Takagi, S.; Nomiyama, H.; Schall, T.J.; et al. Identification and Molecular Characterization of Fractalkine Receptor CX3CR1, which Mediates Both Leukocyte Migration and Adhesion. Cell 1997, 91, 521–530. [Google Scholar] [CrossRef]
- Böttcher, J.P.; Beyer, M.; Meissner, F.; Abdullah, Z.; Sander, J.; Höchst, B.; Eickhoff, S.; Rieckmann, J.C.; Russo, C.; Bauer, T.; et al. Functional classification of memory CD8+ T cells by CX3CR1 expression. Nat. Commun. 2015, 6, 8306. [Google Scholar] [CrossRef]
- Gerlach, C.; Moseman, E.A.; Loughhead, S.M.; Alvarez, D.; Zwijnenburg, A.J.; Waanders, L.; Garg, R.; de la Torre, J.C.; von Andrian, U.H. The Chemokine Receptor CX3CR1 Defines Three Antigen-Experienced CD8 T Cell Subsets with Distinct Roles in Immune Surveillance and Homeostasis. Immunity 2016, 45, 1270–1284. [Google Scholar] [CrossRef]
- Staumont-Salle, D.; Fleury, S.; Lazzari, A.; Molendi-Coste, O.; Hornez, N.; Lavogiez, C.; Kanda, A.; Wartelle, J.; Fries, A.; Pennino, D.; et al. CX3CL1 (fractalkine) and its receptor CX3CR1 regulate atopic dermatitis by controlling effector T cell retention in inflamed skin. J. Exp. Med. 2014, 211, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Morimura, S.; Oka, T.; Sugaya, M.; Sato, S. CX3CR1 deficiency attenuates imiquimod-induced psoriasis-like skin inflammation with decreased M1 macrophages. J. Dermatol. Sci. 2016, 82, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Sugaya, M. Chemokines and skin diseases. Arch. Immunol. Therap. Exp. 2015, 63, 109–115. [Google Scholar] [CrossRef]
- Echigo, T.; Hasegawa, M.; Shimada, Y.; Takehara, K.; Sato, S. Expression of fractalkine and its receptor, CX3CR1, in atopic dermatitis: Possible contribution to skin inflammation. J. Allergy Clin. Immunol. 2004, 113, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, I.; Erreni, M.; van Brakel, M.; Debets, R.; Allavena, P. Enhanced recruitment of genetically modified CX3CR1-positive human T cells into Fractalkine/CX3CL1 expressing tumors: Importance of the chemokine gradient. J. Immunother. Cancer 2016, 4, 21. [Google Scholar] [CrossRef]
- Mackay, L.K.; Rahimpour, A.; Ma, J.Z.; Collins, N.; Stock, A.T.; Hafon, M.-L.; Vega-Ramos, J.; Lauzurica, P.; Mueller, S.N.; Stefanovic, T.; et al. The developmental pathway for CD103+CD8+ tissue-resident memory T cells of skin. Nat. Immunol. 2013, 14, 1294–1301. [Google Scholar] [CrossRef]
- Frisoli, M.L.; Essien, K.; Harris, J.E. Vitiligo: Mechanisms of Pathogenesis and Treatment. Annu. Rev. Immunol. 2020, 38, 621–648. [Google Scholar] [CrossRef]
- Boniface, K.; Seneschal, J.; Picardo, M.; Taïeb, A. Vitiligo: Focus on clinical aspects, immunopathogenesis, and therapy. Clin. Rev. Allergy Immunol. 2018, 54, 52–67. [Google Scholar] [CrossRef]
- Bergqvist, C.; Ezzedine, K. Vitiligo: A review. Dermatology 2020, 236, 571–592. [Google Scholar] [CrossRef]
- Richmond, J.M.; Strassner, J.; Essien, K.I.; Harris, J.E. T-cell positioning by chemokines in autoimmune skin diseases. Immunol. Rev. 2019, 289, 186–204. [Google Scholar] [CrossRef] [PubMed]
- Katayama, I.; Yang, L.; Takahashi, A.; Yang, F.; Wataya-Kaneda, M. The two faces of mast cells in vitiligo pathogenesis. Explor. Immunol. 2021, 1, 269–284. [Google Scholar] [CrossRef]
- Yoshida, M.; Takahashi, Y.; Inoue, S. Histamine Induces Melanogenesis and Morphologic Changes by Protein Kinase a Activation via H2 Receptors in Human Normal Melanocytes. J. Investig. Dermatol. 2000, 114, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.; Na, J.; Choi, J.; Park, K. Melasma: Updates and perspectives. Exp. Dermatol. 2018, 28, 704–708. [Google Scholar] [CrossRef] [PubMed]
- Rork, J.F.; Rashighi, M.; Harris, J.E. Understanding autoimmunity of vitiligo and alopecia areata. Curr. Opin. Pediatr. 2016, 28, 463–469. [Google Scholar] [CrossRef]
- Rashighi, M.; Agarwal, P.; Richmond, J.M.; Harris, T.H.; Dresser, K.; Su, M.-W.; Zhou, Y.; Deng, A.; Hunter, C.A.; Luster, A.D.; et al. CXCL10 Is Critical for the Progression and Maintenance of Depigmentation in a Mouse Model of Vitiligo. Sci. Transl. Med. 2014, 6, 223ra23. [Google Scholar] [CrossRef]
- Rottman, J.B.; Smith, T.L.; Ganley, K.G.; Kikuchi, T.; Krueger, J.G. Potential role of the chemokine receptors CXCR3, CCR4, and the integrin αEβ7 in the pathogenesis of psoriasis vulgaris. Lab. Investig. 2001, 81, 335–347. [Google Scholar] [CrossRef]
- Galli, S.J.; Tsai, M.; Marichal, T.; Tchougounova, E.; Reber, L.L.; Pejler, G. Approaches for Analyzing the Roles of Mast Cells and Their Proteases In Vivo. Adv. Imunnol. 2015, 126, 45–127. [Google Scholar] [CrossRef]
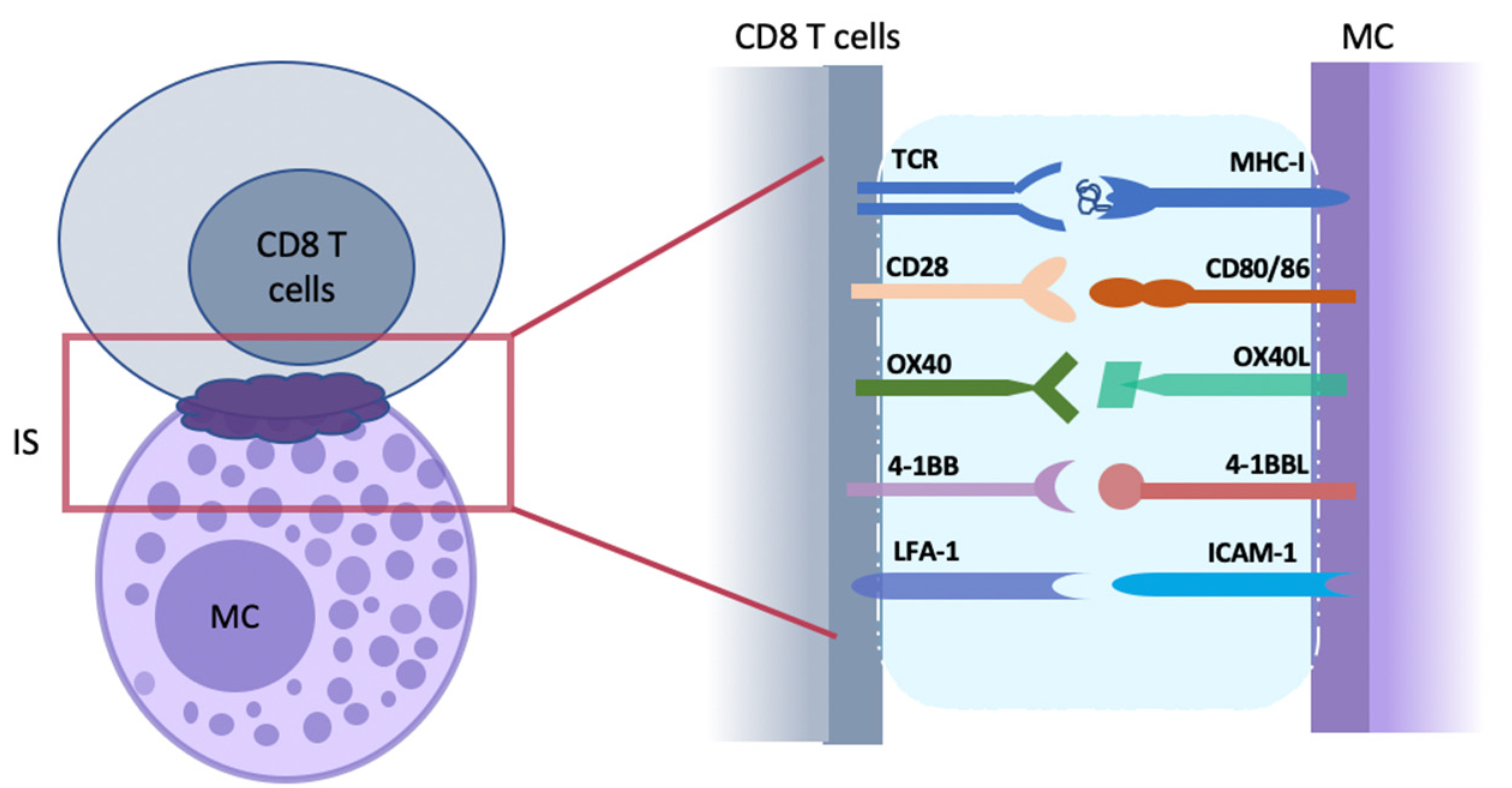
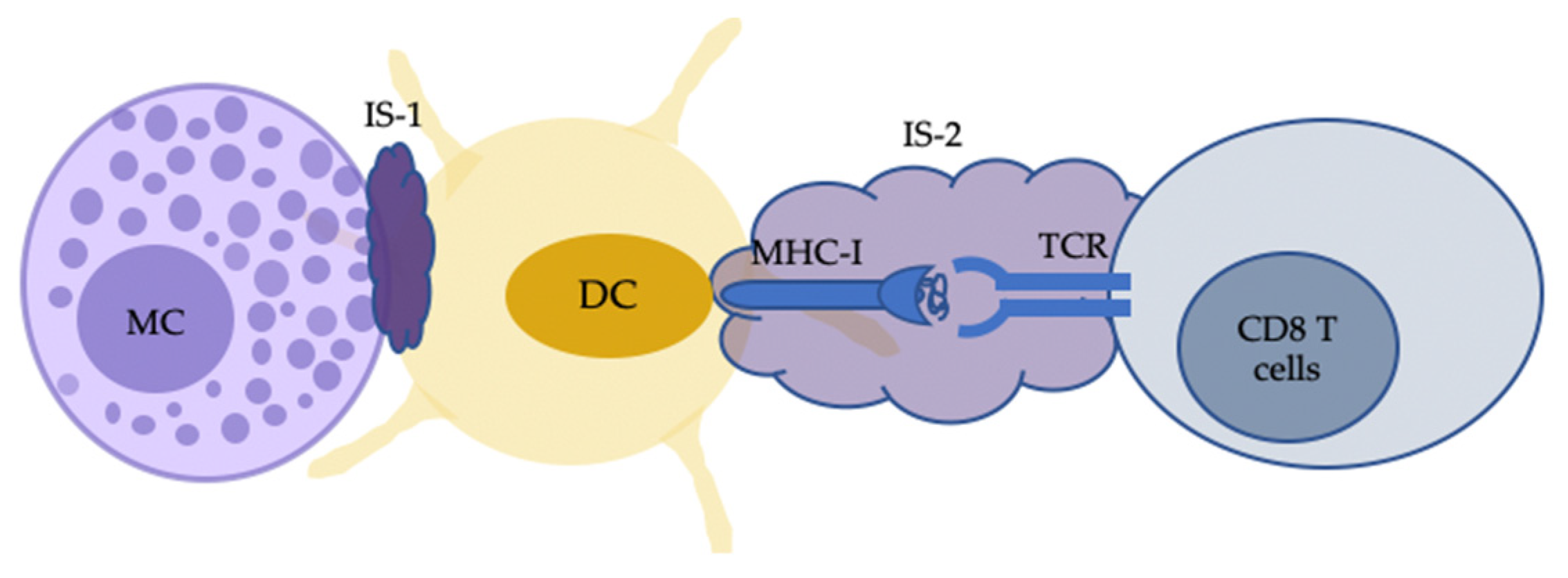
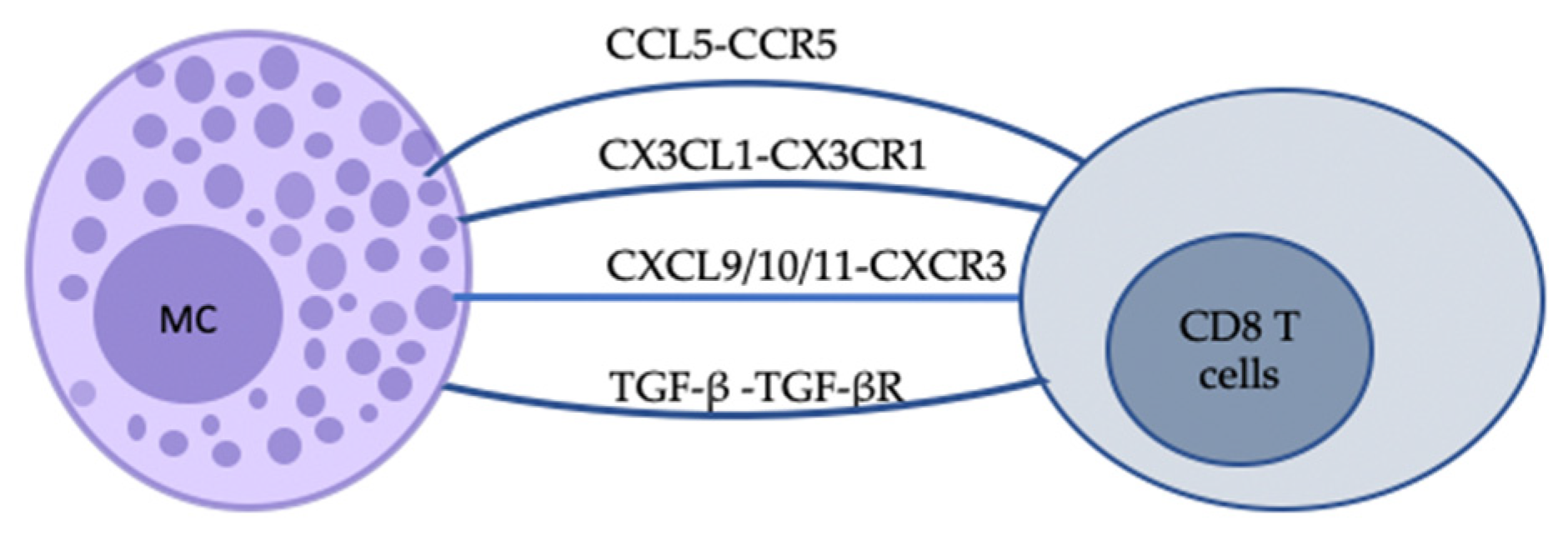
| CD8 T Cell Receptors | MC Ligands | Receptor Family | Disease | Ref. |
|---|---|---|---|---|
| CD28 | CD80 (B7-1); CD86 (B7-2) | IgG-CD28/B7 | psoriasis | [117] |
| OX40 (CD134) | OX40L (CD252) | TNF/TNFR | atopic dermatitis; alopecia areata | [33,118] |
| 4-1BB (CD137) | 4-1BBL | TNF/TNFR | psoriasis | [119] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Griffiths, C.E.M.; Bulfone-Paus, S. Exploring Mast Cell–CD8 T Cell Interactions in Inflammatory Skin Diseases. Int. J. Mol. Sci. 2023, 24, 1564. https://doi.org/10.3390/ijms24021564
Chen Y, Griffiths CEM, Bulfone-Paus S. Exploring Mast Cell–CD8 T Cell Interactions in Inflammatory Skin Diseases. International Journal of Molecular Sciences. 2023; 24(2):1564. https://doi.org/10.3390/ijms24021564
Chicago/Turabian StyleChen, Yiqiao, Christopher E. M. Griffiths, and Silvia Bulfone-Paus. 2023. "Exploring Mast Cell–CD8 T Cell Interactions in Inflammatory Skin Diseases" International Journal of Molecular Sciences 24, no. 2: 1564. https://doi.org/10.3390/ijms24021564
APA StyleChen, Y., Griffiths, C. E. M., & Bulfone-Paus, S. (2023). Exploring Mast Cell–CD8 T Cell Interactions in Inflammatory Skin Diseases. International Journal of Molecular Sciences, 24(2), 1564. https://doi.org/10.3390/ijms24021564







