Comprehensive Strategy for Identifying Extracellular Vesicle Surface Proteins as Biomarkers for Non-Alcoholic Fatty Liver Disease
Abstract
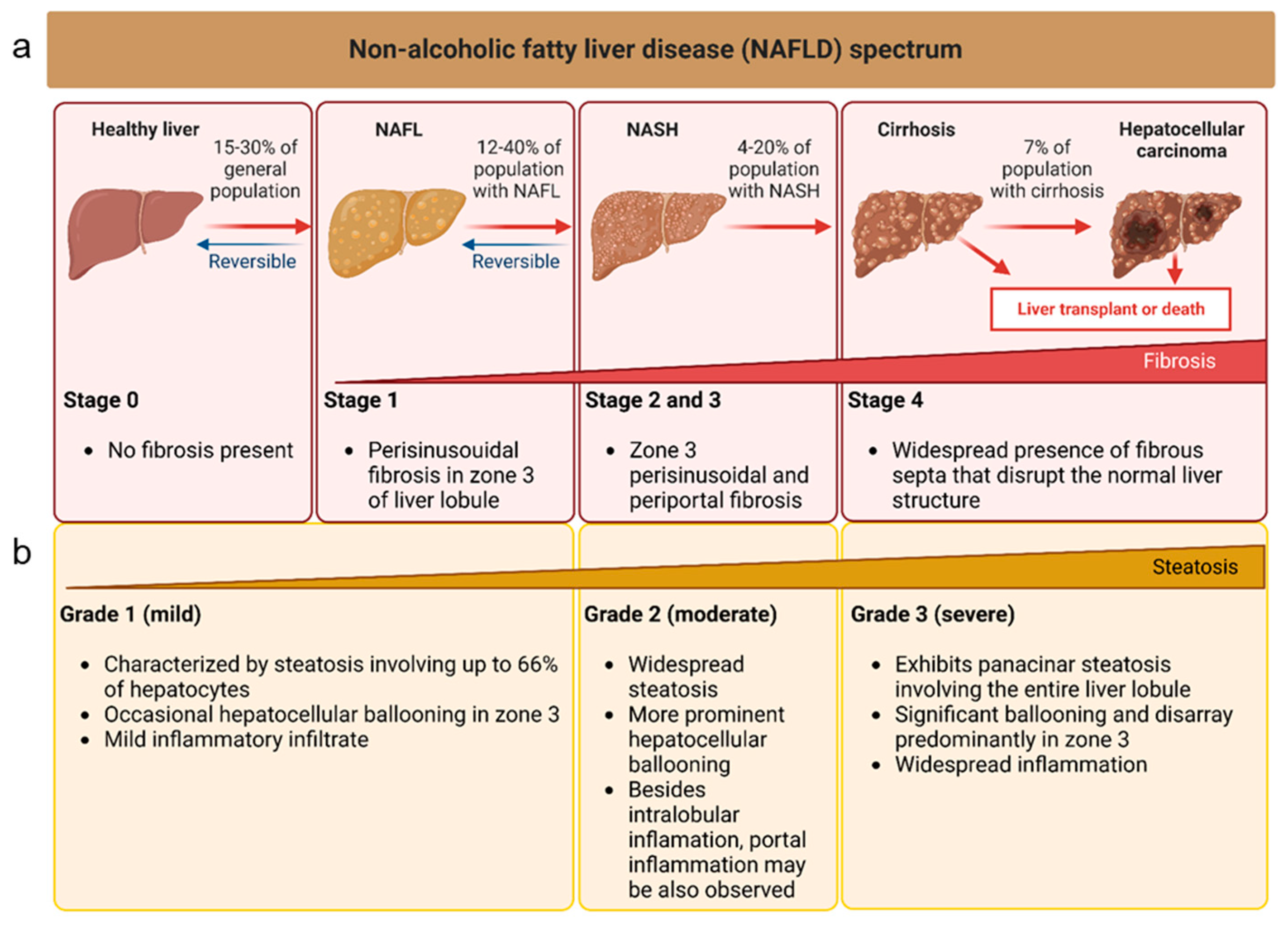
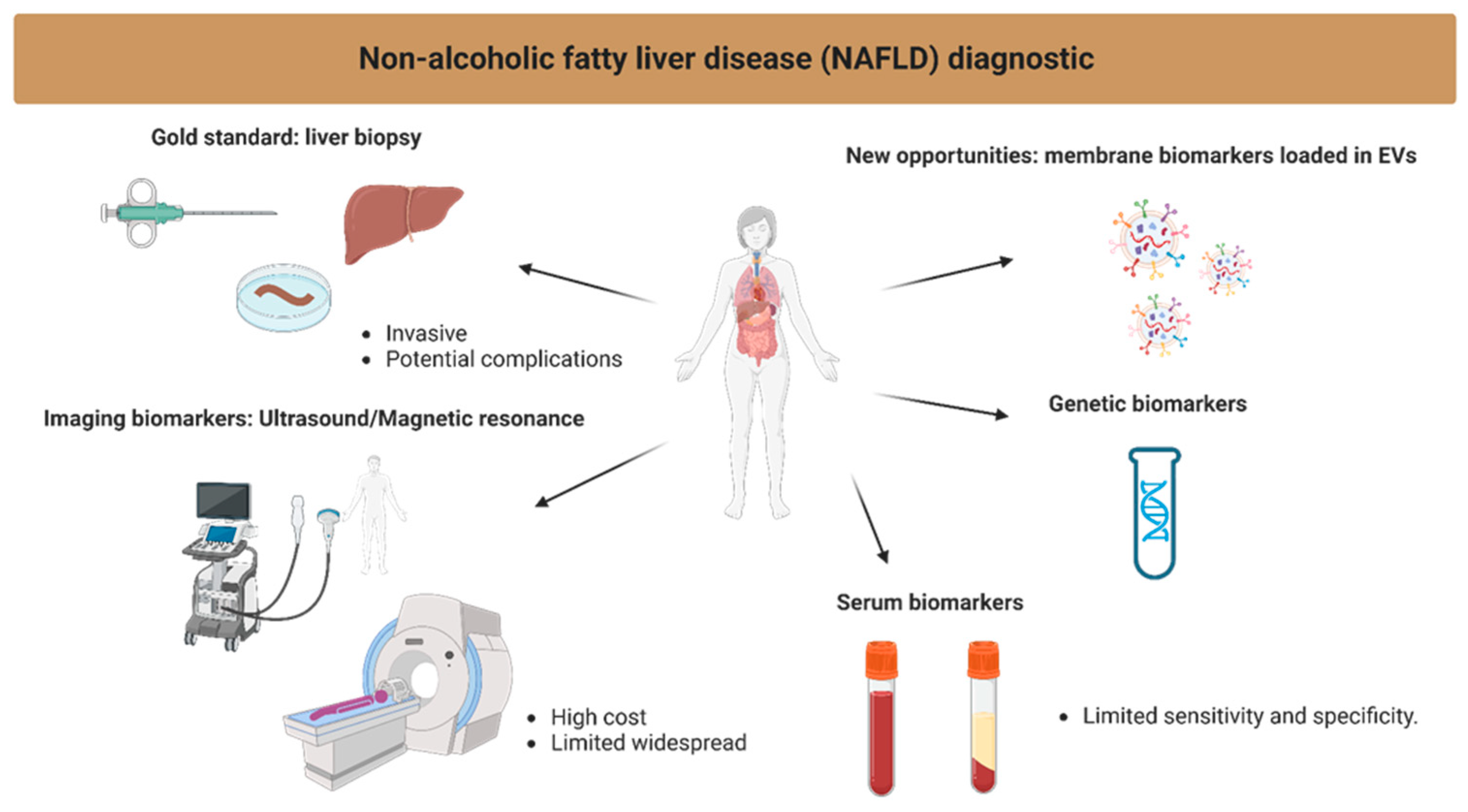
:1. Introduction
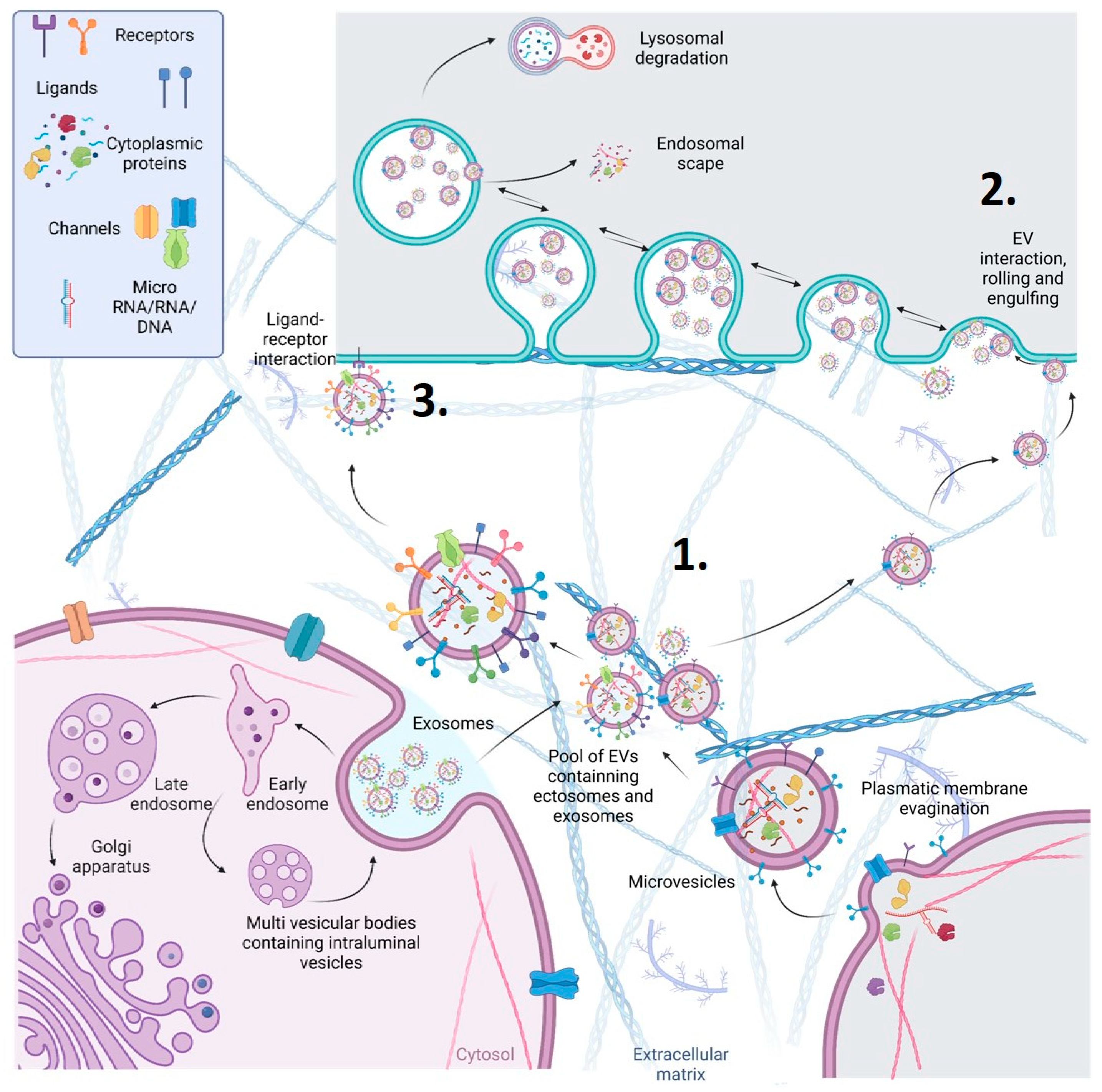
2. Novel Proposed EV Protein Biomarkers
3. Metabolism Related Proteins
4. Inflammation and Fibrosis
5. NOTCH Pathway
6. Wnt/β-Catenin Pathway
7. Plasma/Serum Secreted Proteins in NAFLD
8. Inter-Tissue Crosstalk
9. Perspective
Author Contributions
Funding
Conflicts of Interest
References
- Loomba, R.; Friedman, S.L.; Shulman, G.I. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell 2021, 184, 2537–2564. [Google Scholar] [CrossRef]
- Masoodi, M.; Gastaldelli, A.; Hyotylainen, T.; Arretxe, E.; Alonso, C.; Gaggini, M.; Brosnan, J.; Anstee, Q.M.; Millet, O.; Ortiz, P.; et al. Metabolomics and lipidomics in NAFLD: Biomarkers and non-invasive diagnostic tests. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 835–856. [Google Scholar] [CrossRef]
- Kechagias, S.; Ekstedt, M.; Simonsson, C.; Nasr, P. Non-invasive diagnosis and staging of non-alcoholic fatty liver disease. Hormones 2022, 21, 349–368. [Google Scholar] [CrossRef]
- Brunt, E.M.; Janney, C.G.; Di Bisceglie, A.M.; Neuschwander-Tetri, B.A.; Bacon, B.R. Nonalcoholic steatohepatitis: A proposal for grading and staging the histological lesions. Am. J. Gastroenterol. 1999, 94, 2467–2474. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Franzén, L.E.; Ekstedt, M.; Kechagias, S.; Bodin, L. Semiquantitative evaluation overestimates the degree of steatosis in liver biopsies: A comparison to stereological point counting. Mod. Pathol. 2005, 18, 912–916. [Google Scholar] [CrossRef]
- Anstee, Q.M.; Castera, L.; Loomba, R. Impact of non-invasive biomarkers on hepatology practice: Past, present and future. J. Hepatol. 2022, 76, 1362–1378. [Google Scholar] [CrossRef] [PubMed]
- Hernandez Roman, J.; Siddiqui, M.S. The role of noninvasive biomarkers in diagnosis and risk stratification in nonalcoholic fatty liver disease. Endocrinol. Diabetes Metab. 2020, 3, e00127. [Google Scholar] [CrossRef]
- Byrne, C.D.; Targher, G. NAFLD: A multisystem disease. J. Hepatol. 2015, 62, S47–S64. [Google Scholar] [CrossRef]
- Park, H.; Yoon, E.L.; Kim, M.; Lee, J.; Cho, S.; Jun, D.W.; Nah, E.H. Reappraisal of fibrosis-4 index and non-alcoholic fatty liver disease fibrosis score for advanced fibrosis in average-risk population. Front. Med. 2022, 9, 1024836. [Google Scholar] [CrossRef]
- Sun, W.; Cui, H.; Li, N.; Wei, Y.; Lai, S.; Yang, Y.; Yin, X.; Chen, D.F. Comparison of FIB-4 index, NAFLD fibrosis score and BARD score for prediction of advanced fibrosis in adult patients with non-alcoholic fatty liver disease: A meta-analysis study. Hepatol. Res. 2016, 46, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Atic, A.I.; Thiele, M.; Munk, A.; Dalgaard, L.T. Circulating miRNAs associated with nonalcoholic fatty liver disease. Am. J. Physiol. Cell Physiol. 2023, 324, C588–C602. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Lin, Y.; Yu, C. Circulating. miRNAs. as. Novel. Diagnostic. Biomarkers. in. Nonalcoholic. Fatty. Liver. Disease.: A. Systematic. Review. and. Meta.-Analysis. Can. J. Gastroenterol. Hepatol. 2019, 2019, 2096161. [Google Scholar] [CrossRef]
- van Niel, G.; Carter, D.R.F.; Clayton, A.; Lambert, D.W.; Raposo, G.; Vader, P. Challenges and directions in studying cell-cell communication by extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2022, 23, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Zhu, H.; Wang, H. Extracellular Vesicles in Non-alcoholic Fatty Liver Disease and Alcoholic Liver Disease. Front. Physiol. 2021, 12, 707429. [Google Scholar] [CrossRef]
- Newman, L.A.; Muller, K.; Rowland, A. Circulating cell-specific extracellular vesicles as biomarkers for the diagnosis and monitoring of chronic liver diseases. Cell. Mol. Life. Sci. 2022, 79, 232. [Google Scholar] [CrossRef]
- Garcia-Martinez, I.; Alen, R.; Rada, P.; Valverde, A.M. Insights Into Extracellular Vesicles as Biomarker of NAFLD Pathogenesis. Front. Med. 2020, 7, 395. [Google Scholar] [CrossRef]
- Baek, R.; Jorgensen, M.M. Multiplexed Phenotyping of Small Extracellular Vesicles Using Protein Microarray (EV Array). Methods Mol. Biol. 2017, 1545, 117–127. [Google Scholar] [CrossRef]
- Breitwieser, K.; Koch, L.F.; Tertel, T.; Proestler, E.; Burgers, L.D.; Lipps, C.; Adjaye, J.; Furst, R.; Giebel, B.; Saul, M.J. Detailed Characterization of Small Extracellular Vesicles from Different Cell Types Based on Tetraspanin Composition by ExoView R100 Platform. Int. J. Mol. Sci. 2022, 23, 8544. [Google Scholar] [CrossRef]
- Botha, J.; Pugsley, H.R.; Handberg, A. Conventional, High-Resolution and Imaging Flow Cytometry: Benchmarking Performance in Characterisation of Extracellular Vesicles. Biomedicines. 2021, 9, 124. [Google Scholar] [CrossRef]
- Nielsen, M.H.; Sabaratnam, R.; Pedersen, A.J.T.; Hojlund, K.; Handberg, A. Acute Exercise Increases Plasma Levels of Muscle-Derived Microvesicles Carrying Fatty Acid Transport Proteins. J. Clin. Endocrinol. Metab. 2019, 104, 4804–4814. [Google Scholar] [CrossRef] [PubMed]
- Motomura, W.; Yoshizaki, T.; Takahashi, N.; Kumei, S.; Mizukami, Y.; Jang, S.J.; Kohgo, Y. Analysis of vanin-1 upregulation and lipid accumulation in hepatocytes in response to a high-fat diet and free fatty acids. J. Clin. Biochem. Nutr. 2012, 51, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Povero, D.; Eguchi, A.; Niesman, I.R.; Andronikou, N.; de Mollerat du Jeu, X.; Mulya, A.; Berk, M.; Lazic, M.; Thapaliya, S.; Parola, M.; et al. Lipid-induced toxicity stimulates hepatocytes to release angiogenic microparticles that require Vanin-1 for uptake by endothelial cells. Sci. Signal. 2013, 6, ra88. [Google Scholar] [CrossRef]
- Hendrikx, T.; Porsch, F.; Kiss, M.G.; Rajcic, D.; Papac-Milicevic, N.; Hoebinger, C.; Goederle, L.; Hladik, A.; Shaw, L.E.; Horstmann, H.; et al. Soluble TREM2 levels reflect the recruitment and expansion of TREM2(+) macrophages that localize to fibrotic areas and limit NASH. J. Hepatol. 2022, 77, 1373–1385. [Google Scholar] [CrossRef]
- Hou, J.; Zhang, J.; Cui, P.; Zhou, Y.; Liu, C.; Wu, X.; Ji, Y.; Wang, S.; Cheng, B.; Ye, H.; et al. TREM2 sustains macrophage-hepatocyte metabolic coordination in nonalcoholic fatty liver disease and sepsis. J. Clin. Investig. 2021, 131, e135197. [Google Scholar] [CrossRef]
- Mallach, A.; Gobom, J.; Zetterberg, H.; Hardy, J.; Piers, T.M.; Wray, S.; Pocock, J.M. The influence of the R47H triggering receptor expressed on myeloid cells 2 variant on microglial exosome profiles. Brain Commun. 2021, 3, fcab009. [Google Scholar] [CrossRef] [PubMed]
- Corey, K.E.; Pitts, R.; Lai, M.; Loureiro, J.; Masia, R.; Osganian, S.A.; Gustafson, J.L.; Hutter, M.M.; Gee, D.W.; Meireles, O.R.; et al. ADAMTSL2 protein and a soluble biomarker signature identify at-risk non-alcoholic steatohepatitis and fibrosis in adults with NAFLD. J. Hepatol. 2022, 76, 25–33. [Google Scholar] [CrossRef]
- Povero, D.; Yamashita, H.; Ren, W.; Subramanian, M.G.; Myers, R.P.; Eguchi, A.; Simonetto, D.A.; Goodman, Z.D.; Harrison, S.A.; Sanyal, A.J.; et al. Characterization and Proteome of Circulating Extracellular Vesicles as Potential Biomarkers for NASH. Hepatol. Commun. 2020, 4, 1263–1278. [Google Scholar] [CrossRef]
- Garcia, N.A.; Gonzalez-King, H.; Grueso, E.; Sanchez, R.; Martinez-Romero, A.; Javega, B.; O’Connor, J.E.; Simons, P.J.; Handberg, A.; Sepulveda, P. Circulating exosomes deliver free fatty acids from the bloodstream to cardiac cells: Possible role of CD36. PLoS ONE 2019, 14, e0217546. [Google Scholar] [CrossRef]
- Rada, P.; Gonzalez-Rodriguez, A.; Garcia-Monzon, C.; Valverde, A.M. Understanding lipotoxicity in NAFLD pathogenesis: Is CD36 a key driver? Cell. Death. Dis. 2020, 11, 802. [Google Scholar] [CrossRef]
- Jung, J.W.; Kim, J.E.; Kim, E.; Lee, H.; Lee, H.; Shin, E.A.; Lee, J.W. Liver-originated small extracellular vesicles with TM4SF5 target brown adipose tissue for homeostatic glucose clearance. J. Extracell. Vesicles 2022, 11, e12262. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Um, H.; Park, J.; Jung, J.W.; Kim, J.E.; Lee, H.; Shin, E.A.; Pinanga, Y.; Lee, H.; Nam, S.H.; et al. TM4SF5-dependent crosstalk between hepatocytes and macrophages to reprogram the inflammatory environment. Cell Rep. 2021, 37, 110018. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Kim, E.; Lee, H.; Shin, E.A.; Lee, H.; Lee, J.W. Tetraspanin TM4SF5 in hepatocytes negatively modulates SLC27A transporters during acute fatty acid supply. Arch. Biochem. Biophys. 2021, 710, 109004. [Google Scholar] [CrossRef]
- Luo, F.; Oldoni, F.; Das, A. TM6SF2: A Novel Genetic Player in Nonalcoholic Fatty Liver and Cardiovascular Disease. Hepatol. Commun. 2022, 6, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.P.; Pietzner, M.; Wittemans, L.B.; Rolfe, E.L.; Kerrison, N.D.; Imamura, F.; Forouhi, N.G.; Fauman, E.; Allison, M.E.; Griffin, J.L.; et al. Insights into genetic variants associated with NASH-fibrosis from metabolite profiling. Hum. Mol. Genet. 2020, 29, 3451–3463. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Chiang, Y.P.; He, M.; Worgall, T.S.; Zhou, H.; Jiang, X.C. Liver sphingomyelin synthase 1 deficiency causes steatosis, steatohepatitis, fibrosis, and tumorigenesis: An effect of glucosylceramide accumulation. iScience 2021, 24, 103449. [Google Scholar] [CrossRef]
- Granja, S.C.; Longatto-Filho, A.; de Campos, P.B.; Oliveira, C.P.; Stefano, J.T.; Martins-Filho, S.N.; Chagas, A.L.; Herman, P.; D’Albuquerque, L.C.; Reis Alvares-da-Silva, M.; et al. Non-Alcoholic Fatty Liver Disease-Related Hepatocellular Carcinoma: Immunohistochemical Assessment of Markers of Cancer Cell Metabolism. Pathobiology 2022, 89, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Xia, T.; Du, Y.; Liu, J.; Xie, Y.; Zhang, Y.; Guan, F.; Wu, J.; Wang, X.; Shi, C. Exosomes from activated hepatic stellate cells contain GLUT1 and PKM2: A role for exosomes in metabolic switch of liver nonparenchymal cells. FASEB J. 2019, 33, 8530–8542. [Google Scholar] [CrossRef]
- Shearer, A.M.; Wang, Y.; Fletcher, E.K.; Rana, R.; Michael, E.S.; Nguyen, N.; Abdelmalek, M.F.; Covic, L.; Kuliopulos, A. PAR2 promotes impaired glucose uptake and insulin resistance in NAFLD through GLUT2 and Akt interference. Hepatology 2022, 76, 1778–1793. [Google Scholar] [CrossRef]
- Roncal-Jimenez, C.A.; Lanaspa, M.A.; Rivard, C.J.; Nakagawa, T.; Sanchez-Lozada, L.G.; Jalal, D.; Andres-Hernando, A.; Tanabe, K.; Madero, M.; Li, N.; et al. Sucrose induces fatty liver and pancreatic inflammation in male breeder rats independent of excess energy intake. Metabolism 2011, 60, 1259–1270. [Google Scholar] [CrossRef]
- Garcia, N.A.; Moncayo-Arlandi, J.; Sepulveda, P.; Diez-Juan, A. Cardiomyocyte exosomes regulate glycolytic flux in endothelium by direct transfer of GLUT transporters and glycolytic enzymes. Cardiovasc. Res. 2016, 109, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Karim, S.; Liaskou, E.; Fear, J.; Garg, A.; Reynolds, G.; Claridge, L.; Adams, D.H.; Newsome, P.N.; Lalor, P.F. Dysregulated hepatic expression of glucose transporters in chronic disease: Contribution of semicarbazide-sensitive amine oxidase to hepatic glucose uptake. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 307, G1180–G1190. [Google Scholar] [CrossRef] [PubMed]
- de Gracia Hahn, D.; Duret, A.; Mann, J.P. An AGTR1 Variant Worsens Nonalcoholic Fatty Liver Disease and the Metabolic Syndrome. Am. J. Gastroenterol. 2019, 114, 556–559. [Google Scholar] [CrossRef]
- Eshraghian, A.; Iravani, S.; Azimzadeh, P. The Association between Angiotensin II Type 1 Receptor Gene A1166C Polymorphism and Non-alcoholic Fatty Liver Disease and Its Severity. Middle East J. Dig. Dis. 2018, 10, 96–104. [Google Scholar] [CrossRef] [PubMed]
- van der Graaff, D.; Chotkoe, S.; De Winter, B.; De Man, J.; Casteleyn, C.; Timmermans, J.P.; Pintelon, I.; Vonghia, L.; Kwanten, W.J.; Francque, S. Vasoconstrictor antagonism improves functional and structural vascular alterations and liver damage in rats with early NAFLD. JHEP Rep. 2022, 4, 100412. [Google Scholar] [CrossRef]
- Han, M.; Pioronska, W.; Wang, S.; Nwosu, Z.C.; Sticht, C.; Wang, S.; Gao, Y.; Ebert, M.P.; Dooley, S.; Meyer, C. Hepatocyte caveolin-1 modulates metabolic gene profiles and functions in non-alcoholic fatty liver disease. Cell Death.Dis. 2020, 11, 104. [Google Scholar] [CrossRef]
- Logozzi, M.; De Milito, A.; Lugini, L.; Borghi, M.; Calabro, L.; Spada, M.; Perdicchio, M.; Marino, M.L.; Federici, C.; Iessi, E.; et al. High levels of exosomes expressing CD63 and caveolin-1 in plasma of melanoma patients. PLoS ONE 2009, 4, e5219. [Google Scholar] [CrossRef]
- Ibrahim, S.H.; Hirsova, P.; Tomita, K.; Bronk, S.F.; Werneburg, N.W.; Harrison, S.A.; Goodfellow, V.S.; Malhi, H.; Gores, G.J. Mixed lineage kinase 3 mediates release of C-X-C motif ligand 10-bearing chemotactic extracellular vesicles from lipotoxic hepatocytes. Hepatology 2016, 63, 731–744. [Google Scholar] [CrossRef]
- Javeed, N.; Her, T.K.; Brown, M.R.; Vanderboom, P.; Rakshit, K.; Egan, A.M.; Vella, A.; Lanza, I.; Matveyenko, A.V. Pro-inflammatory beta cell small extracellular vesicles induce beta cell failure through activation of the CXCL10/CXCR3 axis in diabetes. Cell Rep. 2021, 36, 109613. [Google Scholar] [CrossRef]
- Nair, B.; Nath, L.R. Inevitable role of TGF-beta1 in progression of nonalcoholic fatty liver disease. J. Recept. Signal Transduct. Res. 2020, 40, 195–200. [Google Scholar] [CrossRef]
- Shelke, G.V.; Yin, Y.; Jang, S.C.; Lasser, C.; Wennmalm, S.; Hoffmann, H.J.; Li, L.; Gho, Y.S.; Nilsson, J.A.; Lotvall, J. Endosomal signalling via exosome surface TGFbeta-1. J. Extracell. Vesicles 2019, 8, 1650458. [Google Scholar] [CrossRef]
- Rodrigues-Junior, D.M.; Tsirigoti, C.; Lim, S.K.; Heldin, C.H.; Moustakas, A. Extracellular Vesicles and Transforming Growth Factor beta Signaling in Cancer. Front. Cell. Dev. Biol. 2022, 10, 849938. [Google Scholar] [CrossRef] [PubMed]
- Wallace, S.J.; Tacke, F.; Schwabe, R.F.; Henderson, N.C. Understanding the cellular interactome of non-alcoholic fatty liver disease. JHEP Rep. 2022, 4, 100524. [Google Scholar] [CrossRef]
- Yang, L.; Roh, Y.S.; Song, J.; Zhang, B.; Liu, C.; Loomba, R.; Seki, E. Transforming growth factor beta signaling in hepatocytes participates in steatohepatitis through regulation of cell death and lipid metabolism in mice. Hepatology 2014, 59, 483–495. [Google Scholar] [CrossRef]
- Albadawy, R.; Agwa, S.H.A.; Khairy, E.; Saad, M.; El Touchy, N.; Othman, M.; Matboli, M. Clinical Significance of HSPD1/MMP14/ITGB1/miR-6881-5P/Lnc-SPARCL1-1:2 RNA Panel in NAFLD/NASH Diagnosis: Egyptian Pilot Study. Biomedicines 2021, 9, 1248. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Furuta, K.; Lucien, F.; Gutierrez Sanchez, L.H.; Hirsova, P.; Krishnan, A.; Kabashima, A.; Pavelko, K.D.; Madden, B.; Alhuwaish, H.; et al. Integrin beta(1)-enriched extracellular vesicles mediate monocyte adhesion and promote liver inflammation in murine NASH. J. Hepatol. 2019, 71, 1193–1205. [Google Scholar] [CrossRef] [PubMed]
- Miura, K.; Yang, L.; van Rooijen, N.; Brenner, D.A.; Ohnishi, H.; Seki, E. Toll-like receptor 2 and palmitic acid cooperatively contribute to the development of nonalcoholic steatohepatitis through inflammasome activation in mice. Hepatology 2013, 57, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Sharifnia, T.; Antoun, J.; Verriere, T.G.; Suarez, G.; Wattacheril, J.; Wilson, K.T.; Peek, R.M., Jr.; Abumrad, N.N.; Flynn, C.R. Hepatic TLR4 signaling in obese NAFLD. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G270–G278. [Google Scholar] [CrossRef]
- Sun, B.L.; Sun, X.; Kempf, C.L.; Song, J.H.; Casanova, N.G.; Camp, S.M.; Reyes Hernon, V.; Fallon, M.; Bime, C.; Martin, D.R.; et al. Involvement of eNAMPT/TLR4 inflammatory signaling in progression of non-alcoholic fatty liver disease, steatohepatitis, and fibrosis. FASEB J. 2023, 37, e22825. [Google Scholar] [CrossRef]
- Zhang, Y.; Meng, J.; Zhang, L.; Ramkrishnan, S.; Roy, S. Extracellular Vesicles with Exosome-like Features Transfer TLRs between Dendritic Cells. Immunohorizons 2019, 3, 186–193. [Google Scholar] [CrossRef]
- Rossato, M.; Di Vincenzo, A.; Pagano, C.; El Hadi, H.; Vettor, R. The P2X7 Receptor and NLRP3 Axis in Non-Alcoholic Fatty Liver Disease: A Brief Review. Cells 2020, 9, 1047. [Google Scholar] [CrossRef]
- Mederacke, I.; Filliol, A.; Affo, S.; Nair, A.; Hernandez, C.; Sun, Q.; Hamberger, F.; Brundu, F.; Chen, Y.; Ravichandra, A.; et al. The purinergic P2Y14 receptor links hepatocyte death to hepatic stellate cell activation and fibrogenesis in the liver. Sci. Transl. Med. 2022, 14, eabe5795. [Google Scholar] [CrossRef]
- Boujedidi, H.; Robert, O.; Bignon, A.; Cassard-Doulcier, A.M.; Renoud, M.L.; Gary-Gouy, H.; Hemon, P.; Tharinger, H.; Prevot, S.; Bachelerie, F.; et al. CXCR4 dysfunction in non-alcoholic steatohepatitis in mice and patients. Clin. Sci. 2015, 128, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Lu, Y.; Xu, Y.; Wang, J.; Zhang, C.; Du, Y.; Wang, L.; Li, L.; Wang, B.; Shen, J.; et al. Horizontal transfer of exosomal CXCR4 promotes murine hepatocarcinoma cell migration, invasion and lymphangiogenesis. Gene 2018, 676, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, S.; Li, Y.; Qian, X.; Luan, J.; Lv, X. Emerging Importance of Chemokine Receptor CXCR4 and Its Ligand in Liver Disease. Front. Cell Dev. Biol. 2021, 9, 716842. [Google Scholar] [CrossRef] [PubMed]
- Lambrecht, J.; Verhulst, S.; Mannaerts, I.; Sowa, J.P.; Best, J.; Canbay, A.; Reynaert, H.; van Grunsven, L.A. A PDGFRbeta-based score predicts significant liver fibrosis in patients with chronic alcohol abuse, NAFLD and viral liver disease. EBioMedicine 2019, 43, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Geng, T.; Sutter, A.; Harland, M.D.; Law, B.A.; Ross, J.S.; Lewin, D.; Palanisamy, A.; Russo, S.B.; Chavin, K.D.; Cowart, L.A. SphK1 mediates hepatic inflammation in a mouse model of NASH induced by high saturated fat feeding and initiates proinflammatory signaling in hepatocytes. J. Lipid Res. 2015, 56, 2359–2371. [Google Scholar] [CrossRef]
- Rigogliuso, S.; Donati, C.; Cassara, D.; Taverna, S.; Salamone, M.; Bruni, P.; Vittorelli, M.L. An active form of sphingosine kinase-1 is released in the extracellular medium as component of membrane vesicles shed by two human tumor cell lines. J. Oncol. 2010, 2010, 509329. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.S.; Kang, L.; Zheng, J.; Grueter, C.; Bracy, D.P.; James, F.D.; Pozzi, A.; Wasserman, D.H. Integrin alpha1-null mice exhibit improved fatty liver when fed a high fat diet despite severe hepatic insulin resistance. J. Biol. Chem. 2015, 290, 6546–6557. [Google Scholar] [CrossRef]
- Guo, X.; Khosraviani, N.; Raju, S.; Singh, J.; Farahani, N.Z.; Abramian, M.; Torres, V.J.; Howe, K.L.; Fish, J.E.; Kapus, A.; et al. Endothelial ACKR1 is induced by neutrophil contact and down-regulated by secretion in extracellular vesicles. Front. Immunol. 2023, 14, 1181016. [Google Scholar] [CrossRef]
- Nasiri-Ansari, N.; Androutsakos, T.; Flessa, C.M.; Kyrou, I.; Siasos, G.; Randeva, H.S.; Kassi, E.; Papavassiliou, A.G. Endothelial Cell Dysfunction and Nonalcoholic Fatty Liver Disease (NAFLD): A Concise Review. Cells 2022, 11, 2511. [Google Scholar] [CrossRef]
- Ramachandran, P.; Dobie, R.; Wilson-Kanamori, J.R.; Dora, E.F.; Henderson, B.E.P.; Luu, N.T.; Portman, J.R.; Matchett, K.P.; Brice, M.; Marwick, J.A.; et al. Resolving the fibrotic niche of human liver cirrhosis at single-cell level. Nature 2019, 575, 512–518. [Google Scholar] [CrossRef]
- Katayama, A.; Nakatsuka, A.; Eguchi, J.; Murakami, K.; Teshigawara, S.; Kanzaki, M.; Nunoue, T.; Hida, K.; Wada, N.; Yasunaka, T.; et al. Beneficial impact of Gpnmb and its significance as a biomarker in nonalcoholic steatohepatitis. Sci. Rep. 2015, 5, 16920. [Google Scholar] [CrossRef]
- Angeloni, N.L.; McMahon, K.M.; Swaminathan, S.; Plebanek, M.P.; Osman, I.; Volpert, O.V.; Thaxton, C.S. Pathways for Modulating Exosome Lipids Identified By High-Density Lipoprotein-Like Nanoparticle Binding to Scavenger Receptor Type B-1. Sci. Rep. 2016, 6, 22915. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.S.; Cao, Z.; Lis, R.; Nolan, D.J.; Guo, P.; Simons, M.; Penfold, M.E.; Shido, K.; Rabbany, S.Y.; Rafii, S. Divergent angiocrine signals from vascular niche balance liver regeneration and fibrosis. Nature 2014, 505, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Hirsova, P.; Weng, P.; Salim, W.; Bronk, S.F.; Griffith, T.S.; Ibrahim, S.H.; Gores, G.J. TRAIL Deletion Prevents Liver, but Not Adipose Tissue, Inflammation during Murine Diet-Induced Obesity. Hepatol. Commun. 2017, 1, 648–662. [Google Scholar] [CrossRef]
- Bacil, G.P.; Romualdo, G.R.; Piagge, P.; Cardoso, D.R.; Vinken, M.; Cogliati, B.; Barbisan, L.F. Unraveling Hepatic Metabolomic Profiles and Morphological Outcomes in a Hybrid Model of NASH in Different Mouse Strains. Antioxidants 2023, 12, 290. [Google Scholar] [CrossRef]
- Osada-Oka, M.; Shiota, M.; Izumi, Y.; Nishiyama, M.; Tanaka, M.; Yamaguchi, T.; Sakurai, E.; Miura, K.; Iwao, H. Macrophage-derived exosomes induce inflammatory factors in endothelial cells under hypertensive conditions. Hypertens. Res. 2017, 40, 353–360. [Google Scholar] [CrossRef]
- Gonzalez-King, H.; Garcia, N.A.; Ontoria-Oviedo, I.; Ciria, M.; Montero, J.A.; Sepulveda, P. Hypoxia Inducible Factor-1alpha Potentiates Jagged 1-Mediated Angiogenesis by Mesenchymal Stem Cell-Derived Exosomes. Stem Cells 2017, 35, 1747–1759. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-King, H.; Tejedor, S.; Ciria, M.; Gil-Barrachina, M.; Soriano-Navarro, M.; Sanchez-Sanchez, R.; Sepulveda, P.; Garcia, N.A. Non-classical Notch signaling by MDA-MB-231 breast cancer cell-derived small extracellular vesicles promotes malignancy in poorly invasive MCF-7 cells. Cancer Gene Ther. 2022, 29, 1056–1069. [Google Scholar] [CrossRef]
- Gridley, T. Human Genetics. Notch, stroke and dementia. Nature 1996, 383, 673. [Google Scholar] [CrossRef]
- Olsauskas-Kuprys, R.; Zlobin, A.; Osipo, C. Gamma secretase inhibitors of Notch signaling. Onco Targets Ther. 2013, 6, 943–955. [Google Scholar] [CrossRef]
- Sassoli, C.; Pini, A.; Mazzanti, B.; Quercioli, F.; Nistri, S.; Saccardi, R.; Zecchi-Orlandini, S.; Bani, D.; Formigli, L. Mesenchymal stromal cells affect cardiomyocyte growth through juxtacrine Notch-1/Jagged-1 signaling and paracrine mechanisms: Clues for cardiac regeneration. J. Mol. Cell. Cardiol. 2011, 51, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, L. The Role of Notch Signaling Pathway in Non-Alcoholic Fatty Liver Disease. Front. Mol. Biosci. 2021, 8, 792667. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Baker, S.S.; Shahein, A.; Choudhury, S.; Liu, W.; Bhatia, T.; Baker, R.D.; Lee, T. Upregulation of non-canonical Wnt ligands and oxidative glucose metabolism in NASH induced by methionine-choline deficient diet. Trends. Cell. Mol. Biol. 2018, 13, 47–56. [Google Scholar] [CrossRef]
- HM, A.E.; Veidal, S.S.; Feigh, M.; Hallenborg, P.; Puglia, M.; Pers, T.H.; Vrang, N.; Jelsing, J.; Kornum, B.R.; Blagoev, B.; et al. Multi-omics characterization of a diet-induced obese model of non-alcoholic steatohepatitis. Sci. Rep. 2020, 10, 1148. [Google Scholar] [CrossRef]
- Xiong, X.; Kuang, H.; Ansari, S.; Liu, T.; Gong, J.; Wang, S.; Zhao, X.Y.; Ji, Y.; Li, C.; Guo, L.; et al. Landscape of Intercellular Crosstalk in Healthy and NASH Liver Revealed by Single-Cell Secretome Gene Analysis. Mol. Cell 2019, 75, 644–660.e5. [Google Scholar] [CrossRef]
- Scavo, M.P.; Depalo, N.; Rizzi, F.; Carrieri, L.; Serino, G.; Franco, I.; Bonfiglio, C.; Pesole, P.L.; Cozzolongo, R.; Gianuzzi, V.; et al. Exosomal FZD-7 Expression Is Modulated by Different Lifestyle Interventions in Patients with NAFLD. Nutrients 2022, 14, 1133. [Google Scholar] [CrossRef]
- Saponara, E.; Penno, C.; Orsini, V.; Wang, Z.Y.; Fischer, A.; Aebi, A.; Matadamas-Guzman, M.L.; Brun, V.; Fischer, B.; Brousseau, M.; et al. Loss of Hepatic Leucine-Rich Repeat-Containing G-Protein Coupled Receptors 4 and 5 Promotes Nonalcoholic Fatty Liver Disease. Am. J. Pathol. 2023, 193, 161–181. [Google Scholar] [CrossRef]
- Shree Harini, K.; Ezhilarasan, D. Wnt/beta-catenin signaling and its modulators in nonalcoholic fatty liver diseases. Hepatobiliary Pancreat. Dis. Int. 2023, 22, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Chairoungdua, A.; Smith, D.L.; Pochard, P.; Hull, M.; Caplan, M.J. Exosome release of beta-catenin: A novel mechanism that antagonizes Wnt signaling. J. Cell. Biol. 2010, 190, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Dovrat, S.; Caspi, M.; Zilberberg, A.; Lahav, L.; Firsow, A.; Gur, H.; Rosin-Arbesfeld, R. 14-3-3 and beta-catenin are secreted on extracellular vesicles to activate the oncogenic Wnt pathway. Mol. Oncol. 2014, 8, 894–911. [Google Scholar] [CrossRef] [PubMed]
- Kalra, H.; Gangoda, L.; Fonseka, P.; Chitti, S.V.; Liem, M.; Keerthikumar, S.; Samuel, M.; Boukouris, S.; Al Saffar, H.; Collins, C.; et al. Extracellular vesicles containing oncogenic mutant beta-catenin activate Wnt signalling pathway in the recipient cells. J. Extracell. Vesicles 2019, 8, 1690217. [Google Scholar] [CrossRef]
- Niu, L.; Geyer, P.E.; Wewer Albrechtsen, N.J.; Gluud, L.L.; Santos, A.; Doll, S.; Treit, P.V.; Holst, J.J.; Knop, F.K.; Vilsboll, T.; et al. Plasma proteome profiling discovers novel proteins associated with non-alcoholic fatty liver disease. Mol. Syst. Biol. 2019, 15, e8793. [Google Scholar] [CrossRef] [PubMed]
- Pitkanen, N.; Finkenstedt, A.; Lamina, C.; Juonala, M.; Kahonen, M.; Makela, K.M.; Dieplinger, B.; Viveiros, A.; Melmer, A.; Leitner, I.; et al. Afamin predicts the prevalence and incidence of nonalcoholic fatty liver disease. Clin. Chem. Lab. Med. 2022, 60, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hu, Y.; Deng, L. The Underlying Roles of Exosome-Associated PIGR in Fatty Acid Metabolism and Immune Signaling in Colorectal Cancer. J. Oncol. 2022, 2022, 4675683. [Google Scholar] [CrossRef]
- Tey, S.K.; Wong, S.W.K.; Chan, J.Y.T.; Mao, X.; Ng, T.H.; Yeung, C.L.S.; Leung, Z.; Fung, H.L.; Tang, A.H.N.; Wong, D.K.H.; et al. Patient pIgR-enriched extracellular vesicles drive cancer stemness, tumorigenesis and metastasis in hepatocellular carcinoma. J. Hepatol. 2022, 76, 883–895. [Google Scholar] [CrossRef]
- Veyel, D.; Wenger, K.; Broermann, A.; Bretschneider, T.; Luippold, A.H.; Krawczyk, B.; Rist, W.; Simon, E. Biomarker discovery for chronic liver diseases by multi-omics-a preclinical case study. Sci. Rep. 2020, 10, 1314. [Google Scholar] [CrossRef]
- Aarts, S.; Reiche, M.; den Toom, M.; Gijbels, M.; Beckers, L.; Gerdes, N.; Lutgens, E. Depletion of CD40 on CD11c(+) cells worsens the metabolic syndrome and ameliorates hepatic inflammation during NASH. Sci. Rep. 2019, 9, 14702. [Google Scholar] [CrossRef]
- Yuan, M.; Wu, B.; Zhang, L.; Wang, H.; Yang, Y. CD40L/CD40 Regulates Adipokines and Cytokines by H3K4me3 Modification in Epicardial Adipocytes. J. Cardiovasc. Pharmacol. 2021, 78, 228–234. [Google Scholar] [CrossRef]
- He, S.; Ryu, J.; Liu, J.; Luo, H.; Lv, Y.; Langlais, P.R.; Wen, J.; Dong, F.; Sun, Z.; Xia, W.; et al. LRG1 is an adipokine that mediates obesity-induced hepatosteatosis and insulin resistance. J. Clin. Investig. 2021, 131, e148545. [Google Scholar] [CrossRef]
- Zhong, M.E.; Chen, Y.; Xiao, Y.; Xu, L.; Zhang, G.; Lu, J.; Qiu, H.; Ge, W.; Wu, B. Serum extracellular vesicles contain SPARC and LRG1 as biomarkers of colon cancer and differ by tumour primary location. EBioMedicine 2019, 50, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Chi, C.; Fu, H.; Li, Y.H.; Zhang, G.Y.; Zeng, F.Y.; Ji, Q.X.; Shen, Q.R.; Wang, X.J.; Li, Z.C.; Zhou, C.C.; et al. Exerkine fibronectin type-III domain-containing protein 5/irisin-enriched extracellular vesicles delay vascular ageing by increasing SIRT6 stability. Eur. Heart. J. 2022, 43, 4579–4595. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.D.; Wu, L.L.; Qi, X.Y.; Wang, Y.Y.; Liao, Z.Z.; Liu, J.H.; Xiao, X.H. New insight of obesity-associated NAFLD: Dysregulated “crosstalk” between multi-organ and the liver? Genes Dis. 2023, 10, 799–812. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Yang, K.; Shen, Z.; Jia, K.; Liu, P.; Pan, M.; Sun, C. ER stress-induced adipocytes secrete-aldo-keto reductase 1B7-containing exosomes that cause nonalcoholic steatohepatitis in mice. Free. Radic. Biol. Med. 2021, 163, 220–233. [Google Scholar] [CrossRef] [PubMed]
- O’Farrell, M.; Duke, G.; Crowley, R.; Buckley, D.; Martins, E.B.; Bhattacharya, D.; Friedman, S.L.; Kemble, G. FASN inhibition targets multiple drivers of NASH by reducing steatosis, inflammation and fibrosis in preclinical models. Sci. Rep. 2022, 12, 15661. [Google Scholar] [CrossRef]
- Sano, S.; Izumi, Y.; Yamaguchi, T.; Yamazaki, T.; Tanaka, M.; Shiota, M.; Osada-Oka, M.; Nakamura, Y.; Wei, M.; Wanibuchi, H.; et al. Lipid synthesis is promoted by hypoxic adipocyte-derived exosomes in 3T3-L1 cells. Biochem. Biophys. Res. Commun. 2014, 445, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, C.M.; Shepherd, A.J.; Miller, V.M.; Jayachandran, M. Age- and sex-specific differences in blood-borne microvesicles from apparently healthy humans. Biol. Sex Differ. 2015, 6, 10. [Google Scholar] [CrossRef]
- Moreno-Vedia, J.; Girona, J.; Ibarretxe, D.; Masana, L.; Rodriguez-Calvo, R. Unveiling the Role of the Fatty Acid Binding Protein 4 in the Metabolic-Associated Fatty Liver Disease. Biomedicines 2022, 10, 197. [Google Scholar] [CrossRef]
- Phoonsawat, W.; Aoki-Yoshida, A.; Tsuruta, T.; Sonoyama, K. Adiponectin is partially associated with exosomes in mouse serum. Biochem. Biophys. Res. Commun. 2014, 448, 261–266. [Google Scholar] [CrossRef]
- Botha, J.; Nielsen, M.H.; Christensen, M.H.; Vestergaard, H.; Handberg, A. Bariatric surgery reduces CD36-bearing microvesicles of endothelial and monocyte origin. Nutr. Metab. 2018, 15, 76. [Google Scholar] [CrossRef]
- Bansal, S.; Tokman, S.; Fleming, T.; Maine, G.N.; Sanborn, K.; Hachem, R.; Bharat, A.; Smith, M.A.; Bremner, R.M.; Mohanakumar, T. SARS-CoV-2 infection in lung transplant recipients induces circulating exosomes with SARS-CoV-2 spike protein S2. Clin. Transl. Med. 2021, 11, e576. [Google Scholar] [CrossRef]
- Peiseler, M.; Schwabe, R.; Hampe, J.; Kubes, P.; Heikenwalder, M.; Tacke, F. Immune mechanisms linking metabolic injury to inflammation and fibrosis in fatty liver disease-novel insights into cellular communication circuits. J. Hepatol. 2022, 77, 1136–1160. [Google Scholar] [CrossRef]
- Lee, Y.A.; Friedman, S.L. Inflammatory and fibrotic mechanisms in NAFLD-Implications for new treatment strategies. J. Int. Med. 2022, 291, 11–31. [Google Scholar] [CrossRef]
- Liu, J.; Xiao, Q.; Xiao, J.; Niu, C.; Li, Y.; Zhang, X.; Zhou, Z.; Shu, G.; Yin, G. Wnt/beta-catenin signalling: Function, biological mechanisms, and therapeutic opportunities. Signal Transduct. Target. Ther. 2022, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Goel, C.; Monga, S.P.; Nejak-Bowen, K. Role and Regulation of Wnt/beta-Catenin in Hepatic Perivenous Zonation and Physiological Homeostasis. Am. J. Pathol. 2022, 192, 4–17. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Martinez-Ramirez, A.S.; Borders, T.L.; Gao, F.; Sosa-Pineda, B. Metabolic and non-metabolic liver zonation is established non-synchronously and requires sinusoidal Wnts. Elife 2020, 9, e46206. [Google Scholar] [CrossRef]
- Gross, J.C.; Zelarayan, L.C. The Mingle-Mangle of Wnt Signaling and Extracellular Vesicles: Functional Implications for Heart Research. Front. Cardiovasc. Med. 2018, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Korf, H.; Vidal-Puig, A. An adipocentric perspective on the development and progression of non-alcoholic fatty liver disease. J. Hepatol. 2023, 78, 1048–1062. [Google Scholar] [CrossRef]
- Chakravarthy, M.V.; Siddiqui, M.S.; Forsgren, M.F.; Sanyal, A.J. Harnessing Muscle-Liver Crosstalk to Treat Nonalcoholic Steatohepatitis. Front. Endocrinol. 2020, 11, 592373. [Google Scholar] [CrossRef]

| Mechanism | Name | Description | Direct Corroboration | References |
|---|---|---|---|---|
| Novel proposed biomarkers | VANIN-1 | Released on EVs surface from lipotoxic hepatocytes | yes | [22,23] |
| TREM2 | Soluble TREM2 levels correlate with NAFLD to NASH progression | no | [24,25,26] | |
| ADAMTS2 | Soluble ADAMTS2 levels correlate with NAFLD to NASH progression | no | [27] | |
| IL13RA1 | upregulated levels in circulating EVs in NASH | yes | [28] | |
| IL27RA | upregulated levels in circulating EVs in NASH | yes | [28] | |
| ICAM2 | upregulated levels in circulating EVs in NASH | yes | [28] | |
| STK16 | upregulated levels in circulating EVs in NASH | yes | [28] | |
| Metabolism related proteins | CD36 | circulating levels of a soluble form of CD36 are abnormally elevated in NAFLD patients | yes | [29,30] |
| TM4SF5 | liver-derived EVs with TM4SF5 target brown adipose tissue for glucose clearance | yes | [31,32,33] | |
| TM6SF2 | TM6SF2 variants were related to hepatic triglyceride in NAFLD and NASH | no | [34,35] | |
| SLC27A5 | upregulated levels in circulating EVs in NASH | yes | [28,33] | |
| SGMS1 | NASH patients had higher liver GluCer synthase and higher plasma GluCer levels | no | [36] | |
| GLUT1 | increased liver GLUT1 levels correlate with a higher degree of steatosis in NASH | yes | [37,38] | |
| GLUT2 | Decreased liver levels in NAFLD | no | [39] | |
| GLUT5 | Increased liver levels in NAFLD induced by high fructose intake in rats | no | [40,41,42] | |
| GLUT4 | Altered liver levels in patients with chronic liver disease | no | [41,42] | |
| AGTR1 | gene variants of AGTR1 have been related to a predisposition to develop NAFLD | no | [43,44,45] | |
| CAV1 | hepatocytes CAV1 modulates metabolic gene profiles and function in NAFLD | no | [46,47] | |
| Inflamation/fibrosis | CXCL10 | hepatocyte lipotoxicity induces the release of CXCL10-bearing vesicles | no | [48,49] |
| TGFB1 | promotes HSC activation and extracellular matrix production in NAFLD | no | [50,51] | |
| TGFB2 | drives multiple types of fibrosis during NAFLD to NASH progression | no | [52,53,54] | |
| TGFBR2 | drives multiple types of fibrosis during NAFLD to NASH progression | no | [52,53,54] | |
| TGFBR3 | drives multiple types of fibrosis during NAFLD to NASH progression | no | [52,53,54] | |
| TGFBR1 | drives multiple types of fibrosis during NAFLD to NASH progression | no | [52,53,54] | |
| ITGB1 | ITGB1 is released in EVs from hepatocytes under lipotoxic stress | yes | [55,56] | |
| TLR2 | activate the inflammasome in Kupffer cells/macrophages in NASH development | no | [57] | |
| TLR4 | contributes to NAFLD severity and NASH/hepatic fibrosis | no | [58,59,60] | |
| P2X7R | related to NAFLD and its inflammatory and fibrotic evolution | no | [61] | |
| P2Y14R | links hepatocyte death to hepatic stellate cell activation and fibrogenesis | no | [62] | |
| CXCR4 | functionally and mechanistically involved in the progression of liver fibrosis | no | [63,64,65] | |
| PDGFRA | increased liver expression levels in NASH patients | no | [36,66] | |
| PDGFRB | circulating levels of PDGFRB are progressively increased with increasing fibrosis stage | no | [36,66] | |
| SPHK1 | mediates hepatic inflammation in mice | yes | [67,68] | |
| ITGA1 | facilitates hepatic insulin action while promoting lipid accumulation in mice | no | [69] | |
| ACKR1 | related to leucocyte recruitment by cirrhotic endothelial cells | no | [70,71,72] | |
| GPNMB | increased serum levels in NASH | no | [73] | |
| SCARB1 | SCARB1 deficiency increased inflammatory dyslipidaemia and adipocytes hypertrophy | no | [74] | |
| FGFR1 | central player in the response to liver injury and fibrosis | no | [75] | |
| TNFSF10 | increased liver expression levels in NASH | no | [76] | |
| CD68 | Increased liver infiltration with CD68+ macrophages is related to liver fibrosis | yes | [77,78] | |
| NOTCH | NOTCH1 | directly involved in NAFLD development | no | [79,80,81,82,83,84] |
| NOTCH2 | directly involved in NAFLD development | no | [79,80,81,82,83,84] | |
| DLL1 | directly involved in NAFLD development | no | [79,80,81,82,83,84] | |
| DLL3 | directly involved in NAFLD development | no | [79,80,81,82,83,84] | |
| DLL4 | directly involved in NAFLD development | no | [79,80,81,82,83,84] | |
| JAG1 | directly involved in NAFLD development | no | [79,80,81,82,83,84] | |
| JAG2 | directly involved in NAFLD development | no | [79,80,81,82,83,84] | |
| WNT/β-catenin | WNT1 | related to hepatic glucose oxidation in NASH | no | [85] |
| WNT3a | related to hepatic glucose oxidation in NASH | no | [85] | |
| WNT5a | related to hepatic glucose oxidation in NASH | no | [85] | |
| WNT11 | related to hepatic glucose oxidation in NASH | no | [85] | |
| WNT2 | increased expression levels in liver pericentral endothelial cells in NASH | no | [86] | |
| RSPO3 | increased expression levels in liver pericentral endothelial cells in NASH | no | [86] | |
| WNT9b | altered liver expression levels in NASH | no | [87] | |
| WNT4 | altered liver expression levels in NASH | no | [87] | |
| FZD7 | modulated levels by lifestyle intervention in NAFLD patients | yes | [88] | |
| LGR4/5 | its activity promotes NAFLD | no | [89] | |
| LRP6 | Mutations in LRP6 are one of the major causes of NAFLD induction | no | [90] | |
| DKK1 | related to hyperlipidaemia in NAFLD | no | [90] | |
| β-catenin | efector of the pathway. It has been found in EVs | no | [91,92,93] | |
| Plasma/serum secreted proteins in NAFLD | AFM | desregulated plasma levels in NAFLD and NASH patientis | no | [94,95] |
| PIGR | desregulated plasma levels in NAFLD and NASH patientis | no | [94,96,97,98] | |
| FTCD | proposed indicative biomarker for NAFLD to NASH progression | no | [27] | |
| Inter-tissue crosstalk | CD40 | CD40 expressing CD11c+ dendritic cells contribute to liver inflammation in NASH | no | [99,100] |
| LRG1 | adipokine that mediates obesity-induced hepatosteatosis and insulin resistance | no | [101,102] | |
| FNDC5 | serum irisin levels are reduced in patients with obesity-related NAFLD | yes | [103,104] | |
| AKR1B7 | AKR1B7 in EVs derived from metabolic stressed adipocytes induce NASH in mice | yes | [105] | |
| FASN | drives de novo lipogenesis, inflammation and fibrogenic signalling in NAFLD | yes | [106,107] | |
| FABP4 | High levels of circulating FABP4 have been described in NAFLD patients | yes | [108,109,110] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia, N.A.; Mellergaard, M.; Gonzalez-King, H.; Salomon, C.; Handberg, A. Comprehensive Strategy for Identifying Extracellular Vesicle Surface Proteins as Biomarkers for Non-Alcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2023, 24, 13326. https://doi.org/10.3390/ijms241713326
Garcia NA, Mellergaard M, Gonzalez-King H, Salomon C, Handberg A. Comprehensive Strategy for Identifying Extracellular Vesicle Surface Proteins as Biomarkers for Non-Alcoholic Fatty Liver Disease. International Journal of Molecular Sciences. 2023; 24(17):13326. https://doi.org/10.3390/ijms241713326
Chicago/Turabian StyleGarcia, Nahuel Aquiles, Maiken Mellergaard, Hernan Gonzalez-King, Carlos Salomon, and Aase Handberg. 2023. "Comprehensive Strategy for Identifying Extracellular Vesicle Surface Proteins as Biomarkers for Non-Alcoholic Fatty Liver Disease" International Journal of Molecular Sciences 24, no. 17: 13326. https://doi.org/10.3390/ijms241713326
APA StyleGarcia, N. A., Mellergaard, M., Gonzalez-King, H., Salomon, C., & Handberg, A. (2023). Comprehensive Strategy for Identifying Extracellular Vesicle Surface Proteins as Biomarkers for Non-Alcoholic Fatty Liver Disease. International Journal of Molecular Sciences, 24(17), 13326. https://doi.org/10.3390/ijms241713326







