Metabolomics and Transcriptomics Analyses Reveals the Molecular Regulatory Mechanisms of Walnut (Juglans regia L.) Embryos in Response to Shade Treatment
Abstract
1. Introduction
2. Results
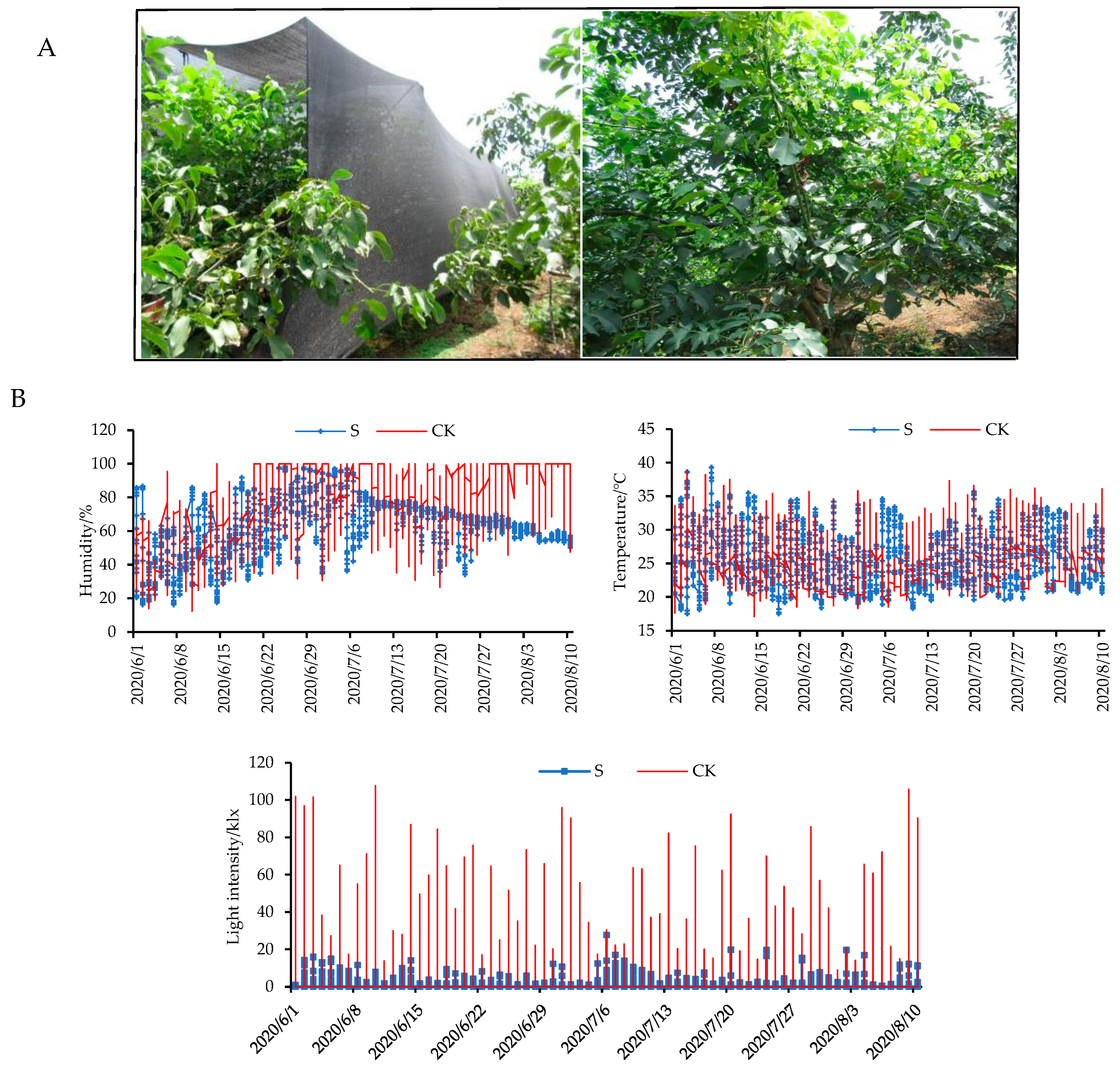
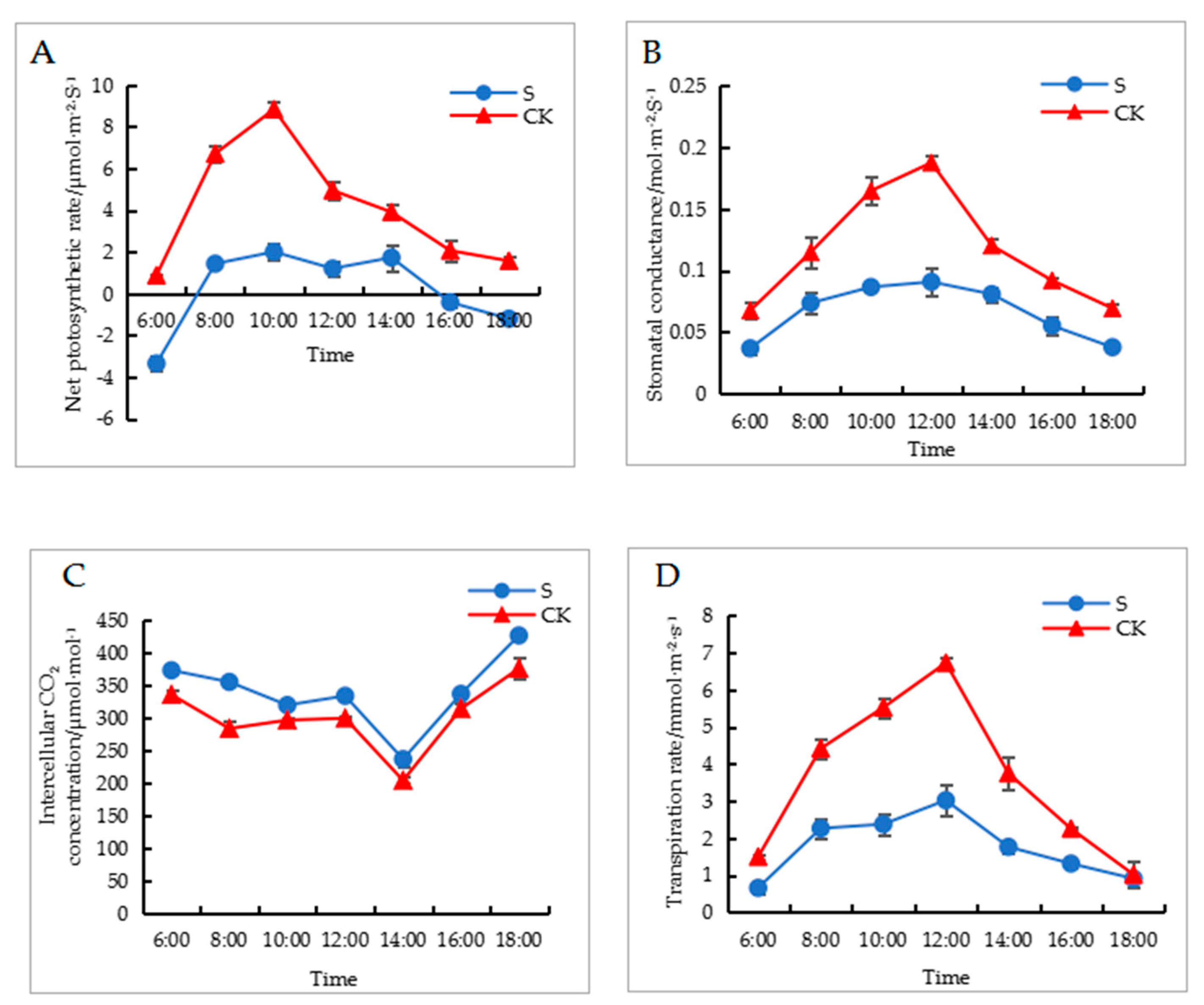
2.1. Effects of Shade Treatment on Environmental and Photosynthetic Parameters
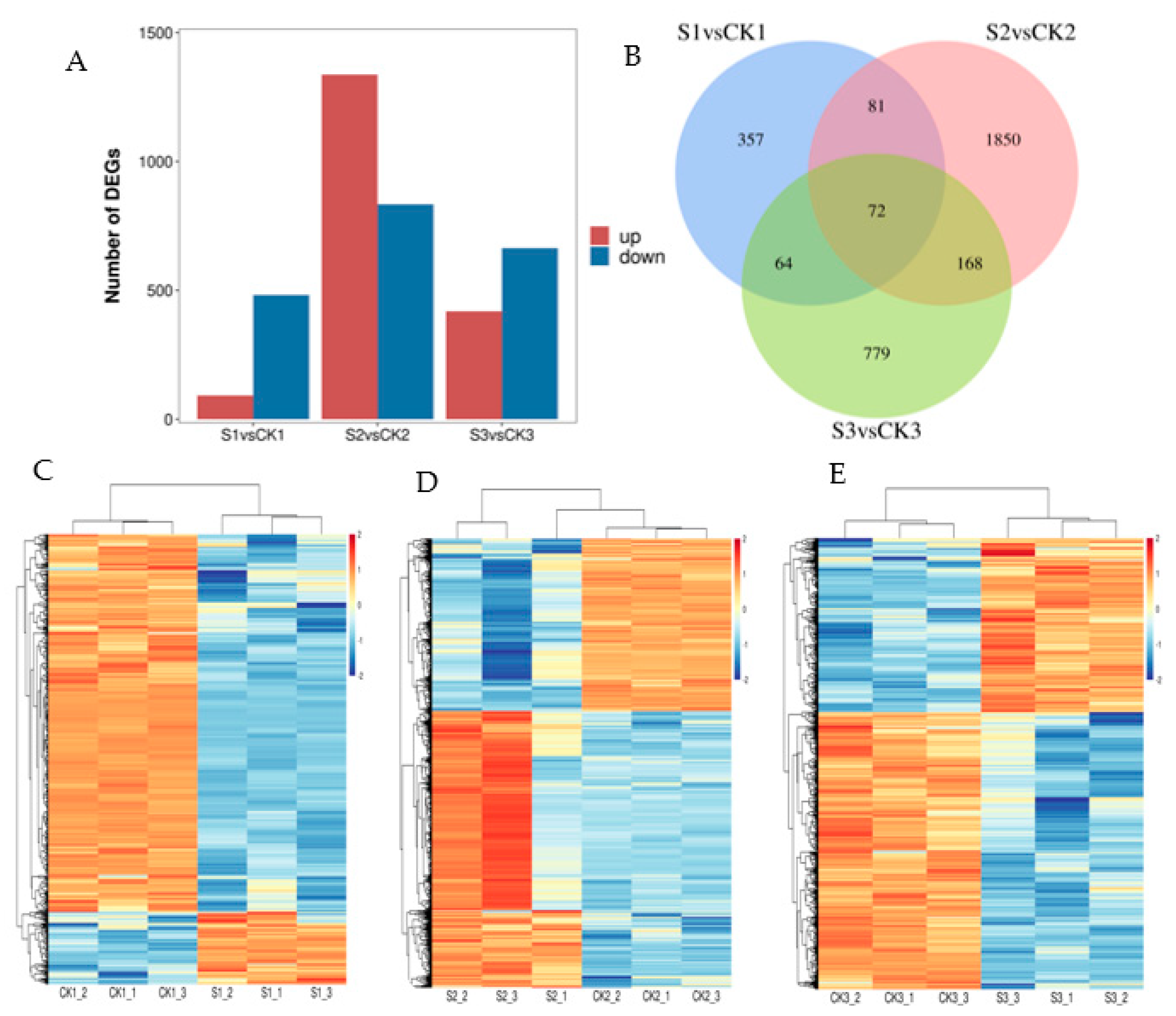
2.2. Analysis of Differentially Expressed Genes (DEGs) in Response to Shade Treatment
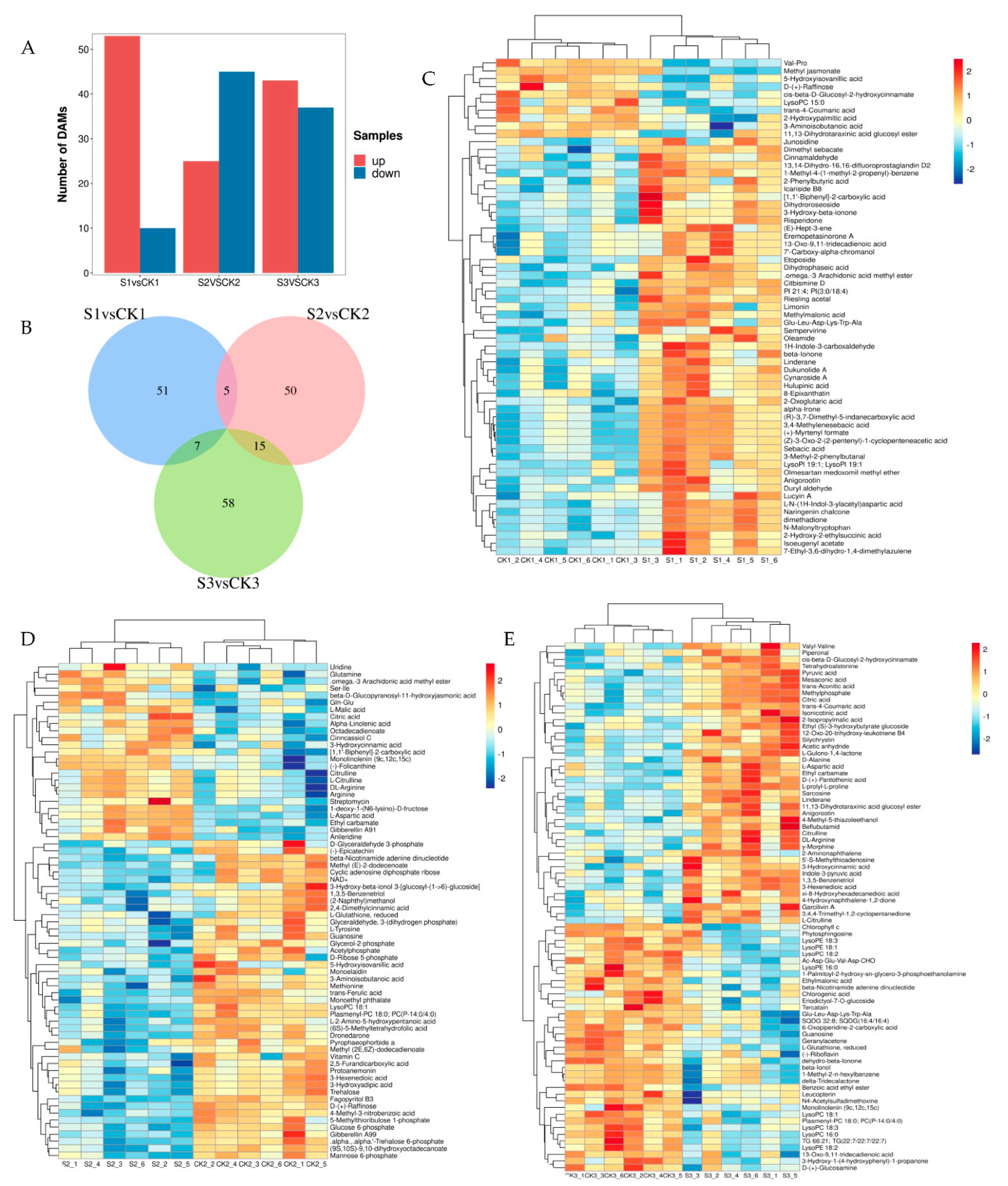
2.3. Analysis of Different Accumulated Metabolites (DAMs) in Response to Shade Treatment
2.4. Principal Component Analysis (PCA)
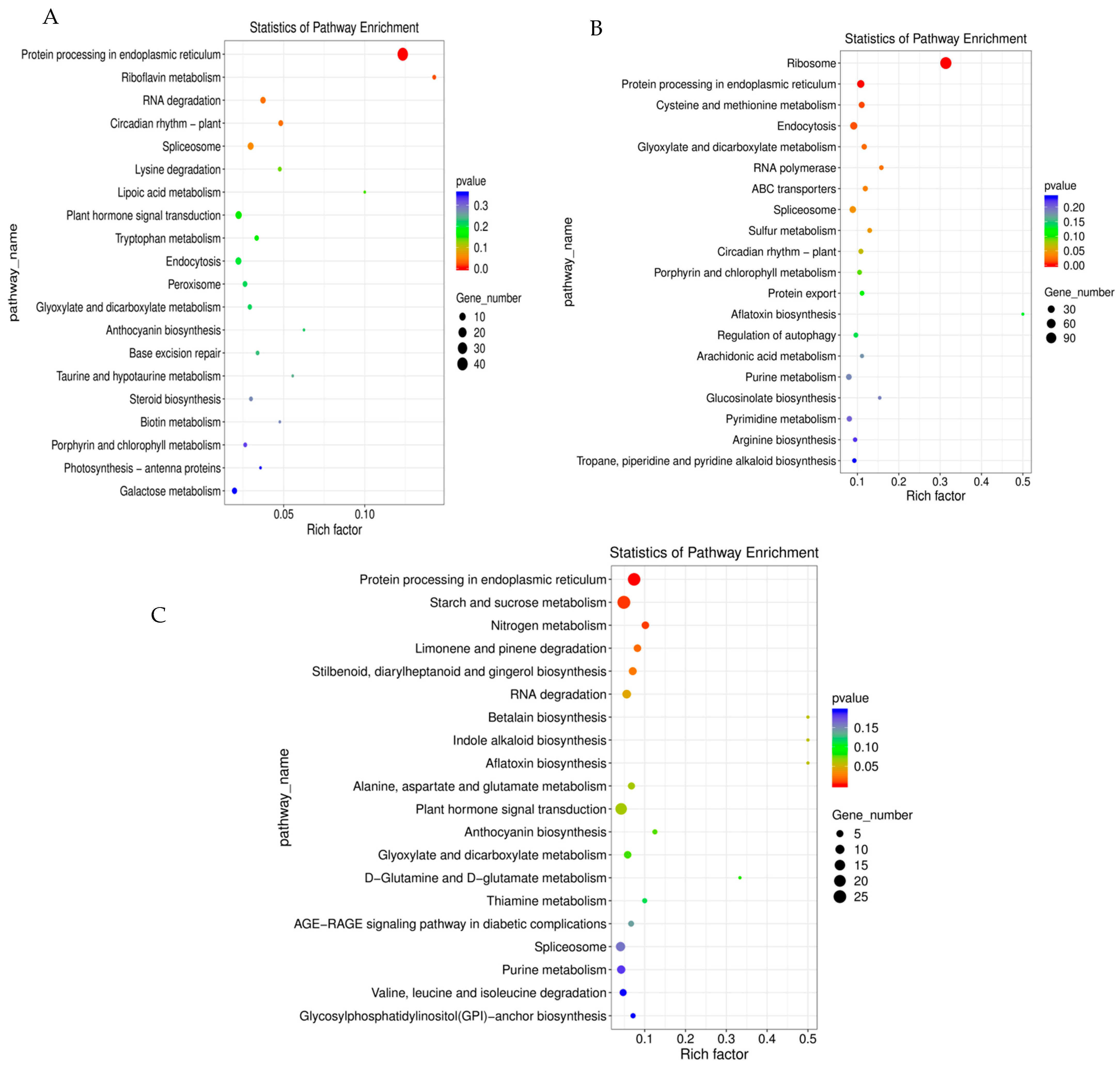
2.5. Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analysis of Candidate Pathways in Response to Shade Treatment
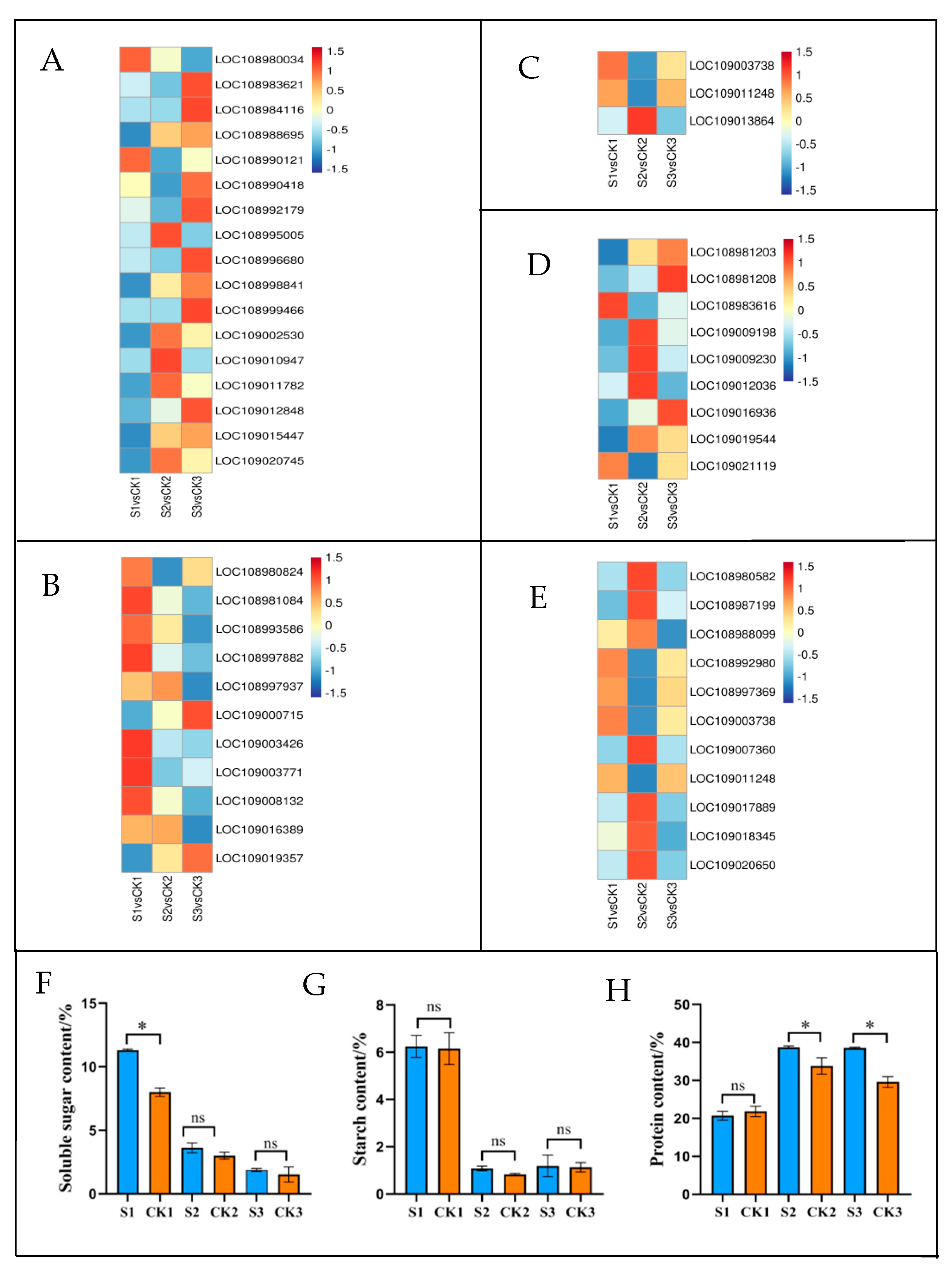
2.6. Differences in Metabolic Pathways under Varying Shade-Treatment Durations
2.6.1. Lipid Biosynthesis in Response to Shade Treatment
2.6.2. Plant Hormone Signal Transduction in Response to Shade Treatment
2.6.3. Starch and Sucrose Metabolism Processes in Response to Shade Treatment
2.6.4. Glycolysis/Gluconeogenesis in Response to Shade Treatment
2.6.5. Glutathione Metabolism in Response to Shade Treatment
2.6.6. Amino Acid Biosynthesis in Response to Shade Treatment
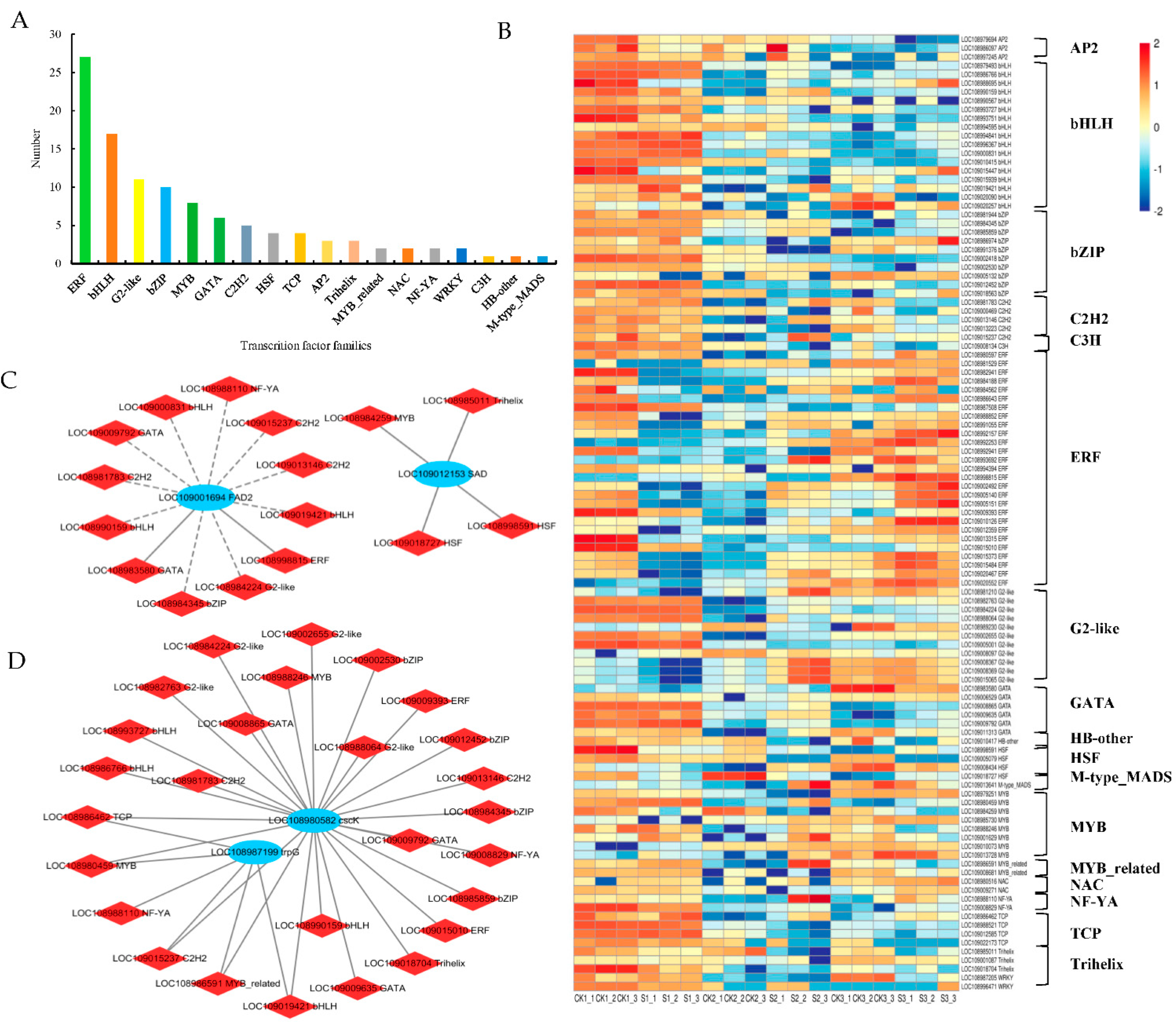
2.7. Analysis of Transcription Factors (TFs)
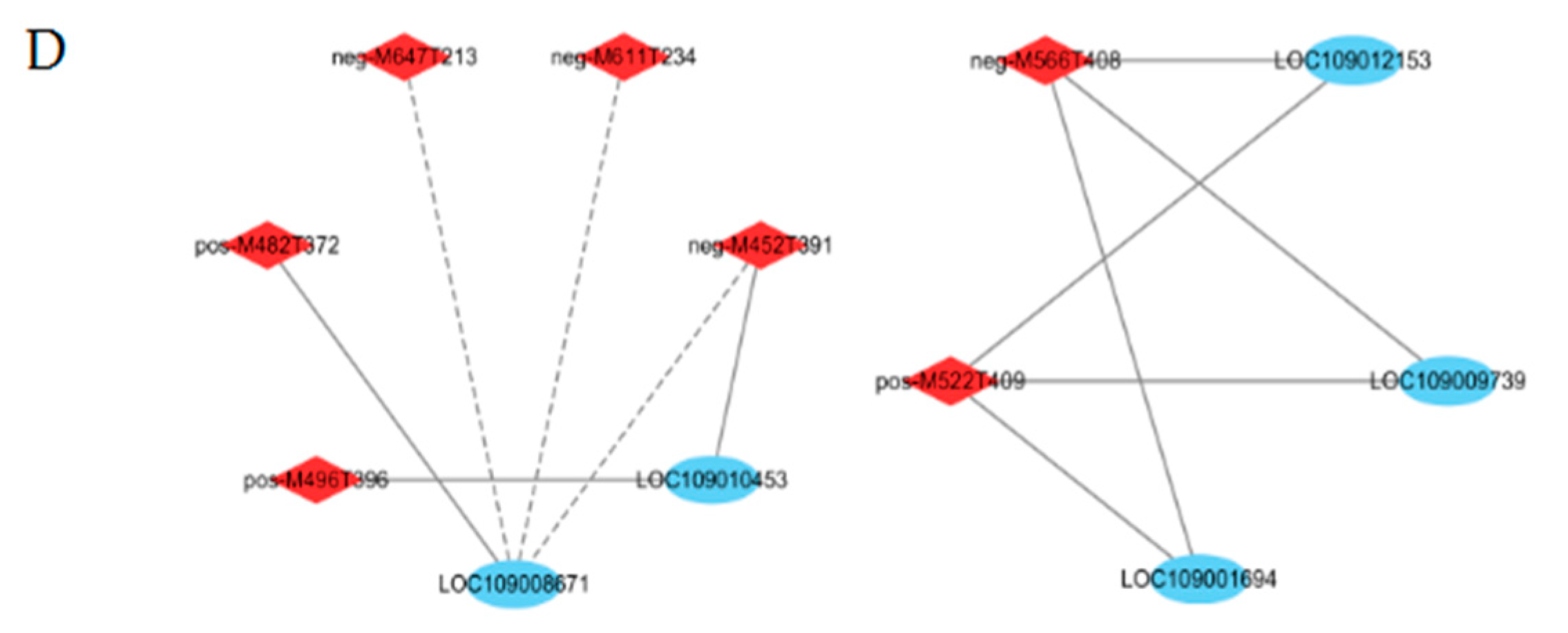
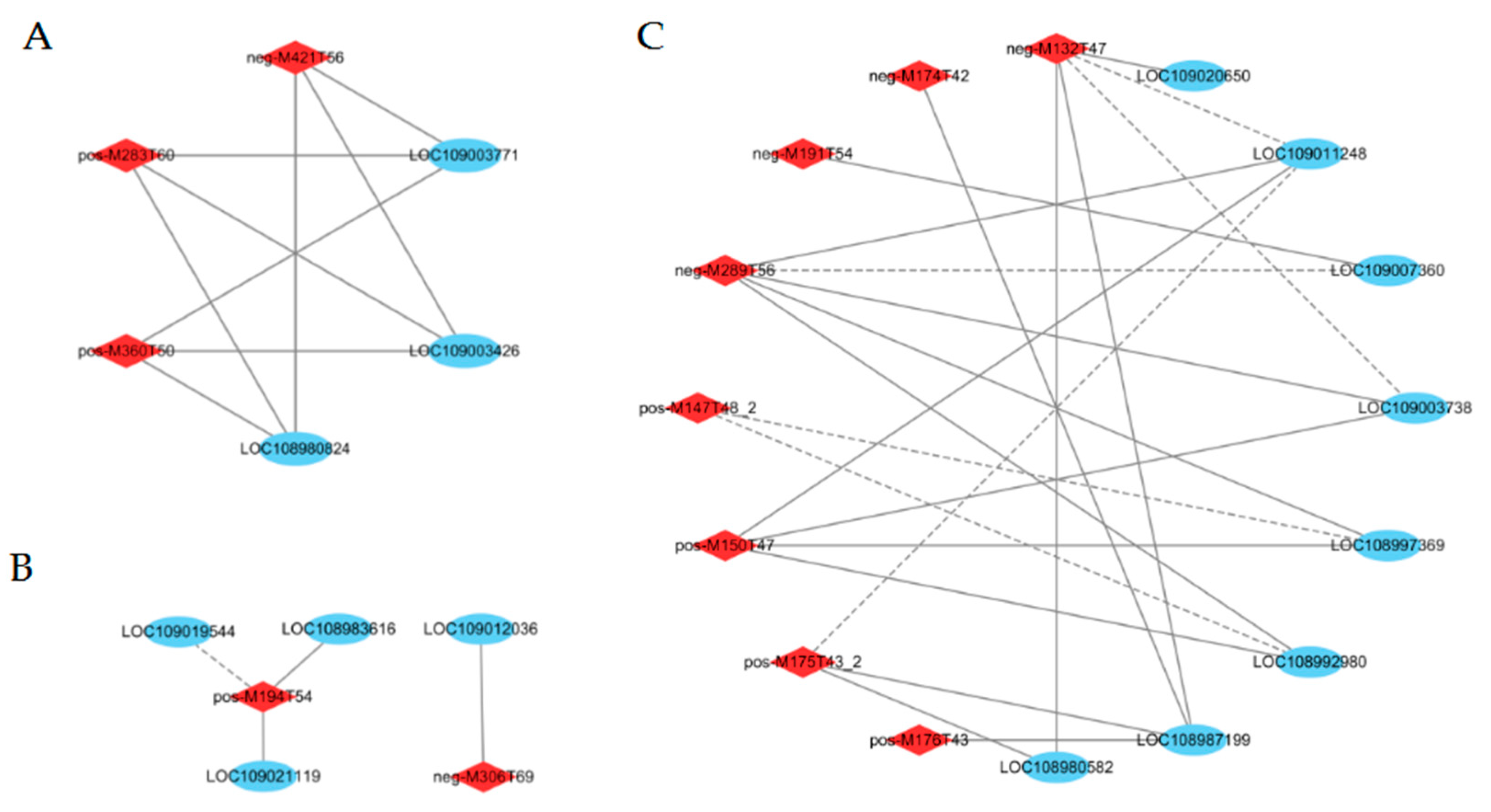
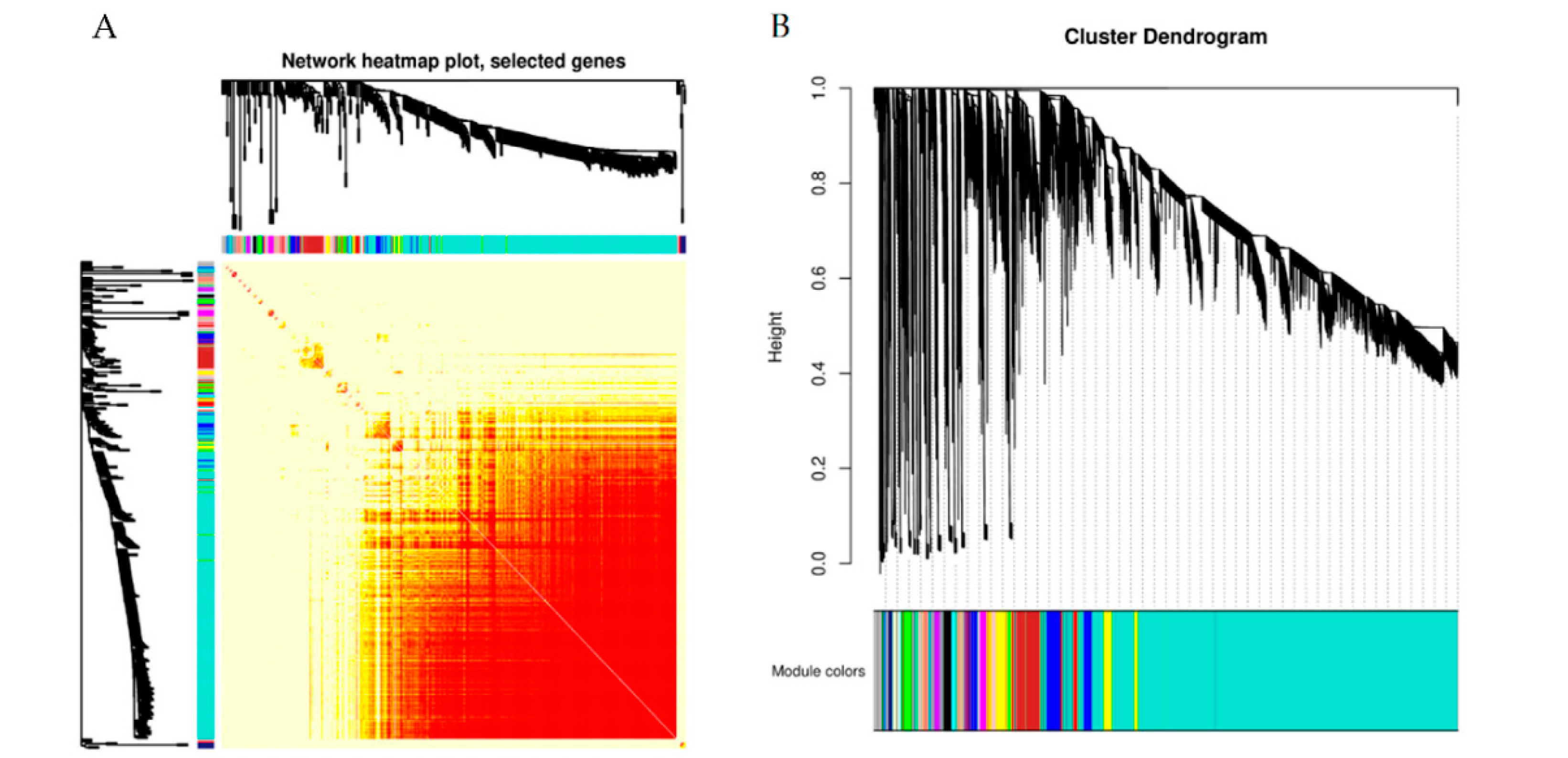
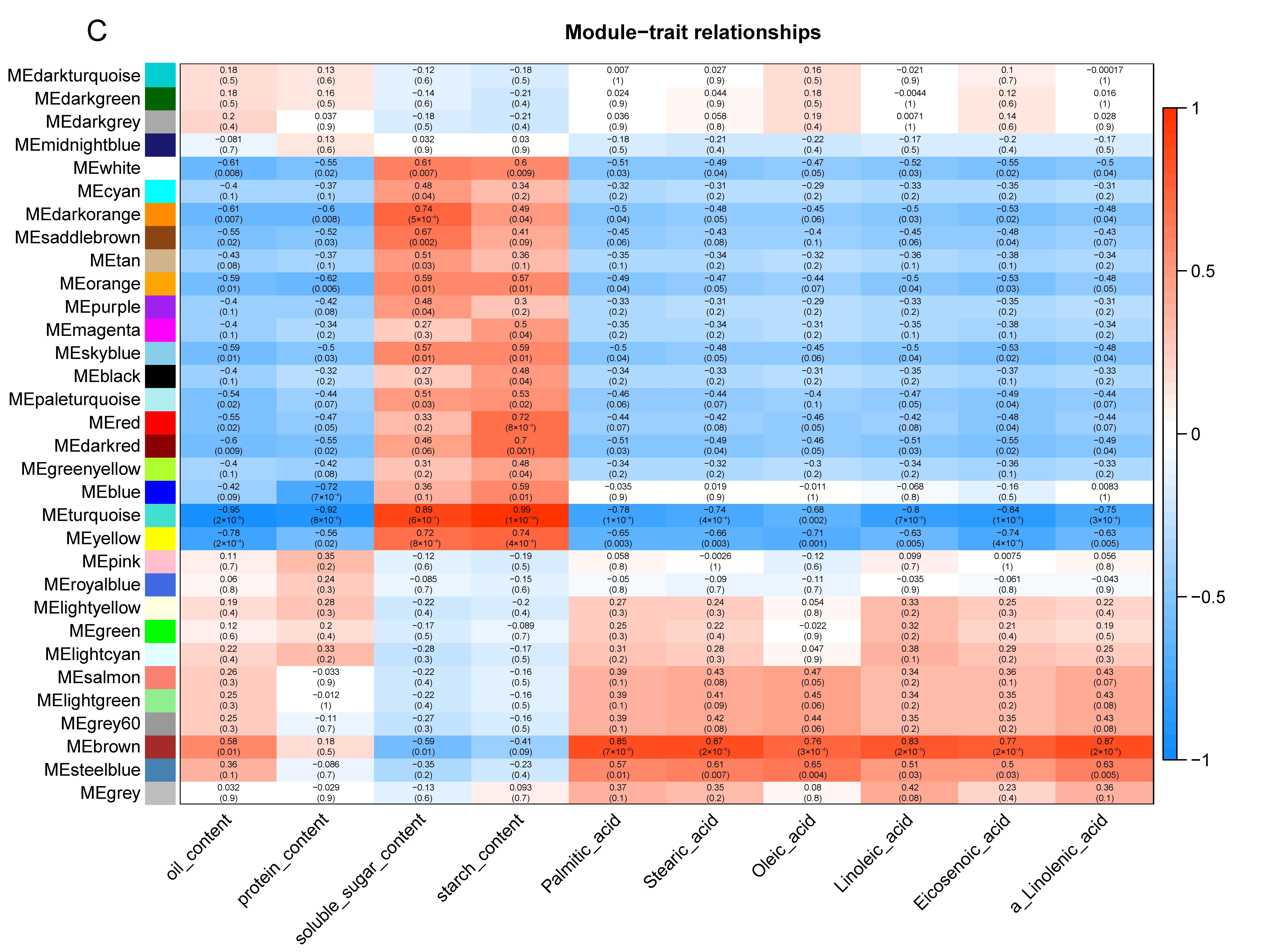
2.8. Weighted Gene Co-Expression Network Analysis (WGCNA)
2.9. Expression Validation of RNA-Seq Data by Real-Time PCR (qRT-PCR)
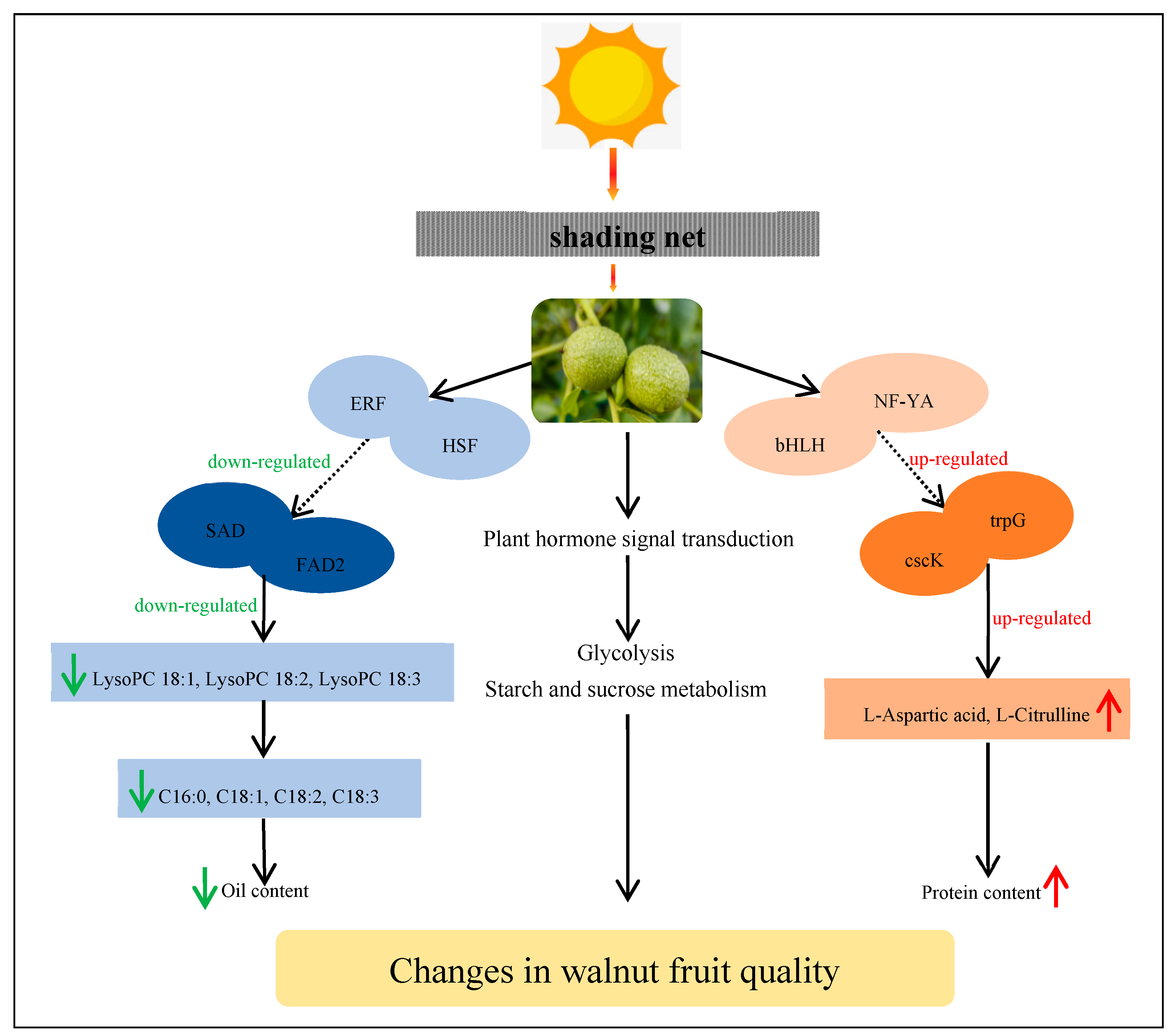
3. Discussion
3.1. Oil Accumulation in Control and Shaded Groups
3.2. Impact of Shade Treatment on Lipid Biosynthesis
3.3. Impact of Shade Treatment on Amino Acid Biosynthesis
3.4. Impact of Shade Treatment on TFs
3.5. Integrated Analysis of Transcriptome and Metabolome Levels
4. Materials and Methods
4.1. Shade Treatment Used in a Walnut Plantation
4.2. Determination of Physiological Indices during the Walnut Fruit-Development Process
4.3. Measurement of Photosynthetic Parameters
4.4. RNA Extraction and Library Construction
4.5. Bioinformatic Analysis of RNA-Seq Data and Co-Expression Network Construction
4.6. Quantitative Analysis
4.7. Metabolite Extraction and LC-MS Analysis
4.8. Metabolomics Data Processing
4.9. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Crews, C.; Hough, P.; Godward, J.; Brereton, P.; Lees, M.; Guiet, S.; Winkelmann, W. Study of the Main Constituents of Some Authentic Walnut Oils. J. Agric. Food Chem. 2005, 53, 4853–4860. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Li, J.; Hu, F.B.; Salas-Salvadó, J.; Tobias, D.K. Effects of walnut consumption on blood lipids and other car-diovascular disease risk factors: An updated meta-analysis and systematic review of controlled trials. Am. J. Clin. Nutr. 2018, 108, 174–187. [Google Scholar] [CrossRef] [PubMed]
- Zibaeenezhad, M.J.; Farhadi, P.; Attar, A.; Mosleh, A.; Amirmoezi, F.; Azimi, A. Effects of walnut oil on lipid profiles in hyper-lipidemic type 2 diabetic patients: A randomized, double-blind, placebo-controlled trial. Nutr. Diabetes 2017, 7, 259. [Google Scholar] [CrossRef] [PubMed]
- Hama, J.R.; Fitzsimmons-Thoss, V. Determination of unsaturated fatty acids composition in walnut (Juglans regia L.) oil using NMR spectroscopy. Food Anal. Methods 2022, 15, 1226–1236. [Google Scholar] [CrossRef]
- Li, M.; Liu, Y.; Sun, C. Research advance about nutrients and medicinal value of walnut. J. Chin. Cereals Oils Assoc. 2009, 24, 166–170. [Google Scholar]
- Domenech, M.; Serramir, M.; Roth, I.; Freitassimoes, T.M.; Vallspedret, C.; Cofan, M.; Lopez, A.; Salavila, A.; Calvo, C.; Rajaram, S.; et al. Effect of a walnut diet on office and 24-h ambulatory blood pressure in elderly individuals. Hypertension 2019, 73, 1049–1057. [Google Scholar] [CrossRef]
- Bates, P.D.; Stymne, S.; Ohlrogge, J.B. Biochemical pathways in seed oil synthesis. Curr. Opin. Plant Biol. 2013, 16, 358–364. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, G.; Liu, X.; Yu, Z.; Peng, S. Integrated analysis of seed microRNA and mRNA transcriptome reveals important functional genes and microRNA-targets in the process of walnut (Juglans regia) seed oil accumulation. Int. J. Mol. Sci. 2020, 21, 9093. [Google Scholar] [CrossRef]
- Huang, R.; Zhou, Y.; Zhang, J.; Ji, F.; Jin, F.; Fan, W.; Pei, D. Transcriptome analysis of walnut (Juglans regia L.) embryos reveals key developmental stages and genes involved in lipid biosynthesis and polyunsaturated fatty acid metabolism. J. Agric. Food Chem. 2021, 69, 377–396. [Google Scholar] [CrossRef]
- Liang, M.; Zhang, X.; Dong, Q.; Li, H.; Guo, S.; Luan, H.; Jia, P.; Yang, M.; Qi, G. Metabolomics and Transcriptomics Provide Insights into Lipid Biosynthesis in the Embryos of Walnut (Juglans regia L.). Plants 2023, 12, 538. [Google Scholar] [CrossRef]
- Zhao, S.; Zhang, X.; Su, Y.; Chen, Y.; Liu, Y.; Sun, M.; Qi, G. Transcriptome analysis reveals dynamic fat accumulation in the walnut kernel. Int. J. Genom. 2018, 2018, 8931651. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Zhang, D.; Ma, Z. Transcriptome analysis of genes involved in fatty acid and lipid biosynthesis in developing walnut (Juglans regia L.) seed kernels from Qinghai Plateau. Plants 2022, 11, 3207. [Google Scholar] [PubMed]
- Zhang, J.; Zhang, W.; Ji, F.; Qiu, J.; Song, X.; Bu, D.; Pan, G.; Ma, Q.; Chen, J.; Huang, R.; et al. A high-quality walnut genome assembly reveals extensive gene expression divergences after whole-genome duplication. Plant Biotechnol. J. 2020, 18, 1848–1850. [Google Scholar] [CrossRef] [PubMed]
- Hovav, R.; Faigenboimdoron, A.; Kadmon, N.; Hu, G.; Zhang, X.; Gallagher, J.P.; Wendel, J.F. A transcriptome profile for developing seed of polyploid cotton. Plant Genome 2015, 8, 1–15. [Google Scholar] [CrossRef]
- Dussert, S.; Guerin, C.; Andersson, M.; Joet, T.; Tranbarger, T.J.; Pizot, M.; Sarah, G.; Omore, A.; Durandgasselin, T.; Morcillo, F. Comparative transcriptome analysis of three oil palm fruit and seed tissues that differ in oil content and fatty acid composition. Plant Physiol. 2013, 162, 1337–1358. [Google Scholar] [CrossRef]
- Milić, D.; Pantelić, A.; Banović Đeri, B.; Samardžić, J.; Vidović, M. Contrasting Metabolisms in Green and White Leaf Sectors of Variegated Pelargonium zonale—An Integrative Transcriptomic and Metabolomic Study. Int. J. Mol. Sci. 2023, 24, 5288. [Google Scholar] [CrossRef]
- D’Oria, A.; Jing, L.; Arkoun, M.; Pluchon, S.; Pateyron, S.; Trouverie, J.; Etienne, P.; Diquélou, S.; Ourry, A. Transcriptomic, Metabolomic and Ionomic Analyses Reveal Early Modulation of Leaf Mineral Content in Brassica napus under Mild or Severe Drought. Int. J. Mol. Sci. 2022, 23, 781. [Google Scholar] [CrossRef]
- Orf, I.; Tenenboim, H.; Omranian, N.; Nikoloski, Z.; Fernie, A.R.; Lisec, J.; Brotman, Y.; Bromke, M.A. Transcriptomic and Metabolomic Analysis of a Pseudomonas-Resistant versus a Susceptible Arabidopsis Accession. Int. J. Mol. Sci. 2022, 23, 12087. [Google Scholar] [CrossRef]
- Liang, M.; Li, H.; Li, H.; Zhang, S.; Wang, Z.; Zhang, X.; Li, B.; Qi, G. Effects of shading treatments on quality and yield in ‘Lvling’ walnut. Non-Wood For. Res. 2017, 35, 174–178. [Google Scholar]
- Niu, X. Effects of Drought Stress on Fruit Quality of Thin-skinned Walnut. J. Shandong For. Sci. Technol. 2018, 48, 61–63. [Google Scholar]
- Gray, D.; Garrett, H. Nitrogen fertilization and aspects of fruit yield in a Missouri black walnut alley cropping practice. Ag-Roforestry Syst. 1998, 44, 333–344. [Google Scholar] [CrossRef]
- Combres, J.; Pallas, B.; Rouan, L.; Mialet-Serra, I.; Caliman, J.; Braconnier, S.; Soulié, J.; Dingkuhn, M. Simulation of inflorescence dynamics in oil palm and estimation of environment-sensitive phenological phases: A model based analysis. Funct. Plant Biol. FPB. 2013, 40, 263–279. [Google Scholar] [CrossRef] [PubMed]
- Asghar, M.A.; Du, J.; Jiang, H.; Li, Y.; Sun, X.; Shang, J.; Liu, J.; Liu, W.; Imran, S.; Iqbal, N.; et al. Shade pretreatment enhanced drought resistance of soybean. Environ. Exp. Bot. 2019, 171, 103952. [Google Scholar] [CrossRef]
- Li, Y.; Wang, W.; Ma, Q. The Osmotic Adjustment and Photosynthesis of a Wheat Cultivar Hanfeng 9703 with High Yield, Drought Resistance under Drought Stress. Acta Agron. Sin. 2003, 29, 759–764. [Google Scholar]
- Powles, S.B. Phtoinhibition of photosynthesis induced by visible light. Annu. Rev. Plant Physiol. 1984, 35, 15–50. [Google Scholar] [CrossRef]
- Meng, Z.; Liu, Y.; Dong, W.; Li, T. Effect of calcium on photosynthesis of tomato under low light intensity. In Proceedings of the Third Conference on Horticulture Science and Technology, Beijing, China, 24–25 November 2012; pp. 309–313. [Google Scholar]
- Sofo, A.; Dichio, B.; Montanaro, G.; Xiloyannis, C. Photosynthetic performance and light response of two olive cultivars under different water and light regimes. Photosynthetica 2009, 47, 602–608. [Google Scholar] [CrossRef]
- Thakur, M.; Bhatt, V.; Kumar, R. Effect of shade level and mulch type on growth, yield and essential oil composition of damask rose (Rosa damascena Mill.) under mid hill conditions of Western Himalayas. PLoS ONE 2019, 14, e0214672. [Google Scholar] [CrossRef]
- Hamadziripi, E.T.; Theron, K.I.; Muller, M.; Steyn, W.J. Apple compositional and peel color differences resulting from canopy microclimate affect consumer preference for eating quality and appearance. HortScience 2014, 49, 384–392. [Google Scholar] [CrossRef]
- Musacchi, S.; Serra, S. Apple fruit quality: Overview on pre-harvest factors. Sci. Hortic. 2017, 234, 409–430. [Google Scholar] [CrossRef]
- Valladares, F.; Niinemets, Ü. Shade Tolerance, a Key Plant Feature of Complex Nature and Consequences. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 237–257. [Google Scholar] [CrossRef]
- Moradi, S.; Zamani, Z.; Moghadam, M.F.; Saba, M.K.; Inglese, P.; Liguori, G. Combination effects of preharvest tree net-shading and postharvest fruit treatments with salicylic acid or hot water on attributes of pomegranate fruit. Sci. Hortic. 2022, 304, 111257. [Google Scholar] [CrossRef]
- Hussain, S.; Iqbal, N.; Rahman, T.; Ting, L.; Brestic, M.; Safdar, M.E.; Asghar, M.A.; Farooq, M.U.; Shafiq, I.; Ali, A. Changes in morphology, chlorophyll fluorescence performance and Rubisco activity of soybean in response to foliar application of ionic titanium under normal light and shade environment. Stoten 2019, 658, 626–637. [Google Scholar] [CrossRef] [PubMed]
- Baud, S. Seeds as oil factories. Plant Reprod. 2018, 31, 213–235. [Google Scholar] [CrossRef] [PubMed]
- Guchhait, R.B.; Polakis, S.E.; Dimroth, P.; Stoll, E.; Moss, J.; Lane, M.D. Acetyl coenzyme A carboxylase system of Escherichia coli:purification and properties of the biotin carboxylase, carboxyl transferase, and carboxyl carrier protein components. J. Biol. Chem. 1974, 249, 6633–6645. [Google Scholar] [CrossRef]
- Li, S.J.; Cronan, J.E. The genes encoding the two carboxyl-transferase subunits of Escherichia coli acetyl-CoA carboxylase. J. Biol. Chem. 1992, 267, 16841–16847. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, M.; Zhang, B.; Shrestha, P.; Petrie, J.R.; Green, A.G.; Singh, S.P. Genetic enhancement of palmitic acid accumulation in cotton seed oil through RNAi down-regulation of ghKAS2 encoding β-ketoacyl-ACP synthase II (KASII). Plant Biotechnol. J. 2017, 15, 132–143. [Google Scholar] [CrossRef]
- Aznarmoreno, J.A.; Venegascaleron, M.; Martinezforce, E.; Garces, R.; Salas, J.J. Acyl carrier proteins from sunflower (Helianthus nannuus L.) seeds and their influence on FatA and FatB acyl-ACP thioesterase activities. Planta 2016, 244, 479–490. [Google Scholar] [CrossRef]
- Baud, S.; Lepiniec, L. Regulation of de novo fatty acid synthesis in maturing oilseeds of Arabidopsis. Plant Physiol. Biochem. 2009, 47, 448–455. [Google Scholar] [CrossRef]
- Okuley, J.; Lightner, J.; Feldmann, K.; Yadav, N.; Lark, E.; Browse, J. Arabidopsis FAD2 gene encodes the enzyme that is essential for polyunsaturated lipid synthesis. Plant Cell 1994, 6, 147–158. [Google Scholar]
- Rajwade, A.V.; Kadoo, N.Y.; Borikar, S.P.; Harsulkar, A.; Ghorpade, P.B.; Gupta, V.S. Differential transcriptional activity of SAD, FAD2 and FAD3 desaturase genes in developing seeds of linseed contributes to varietal variation in α-linolenic acid content. Phytochemistry 2014, 98, 41–53. [Google Scholar] [CrossRef]
- Amaral, J.S.; Casal, S.; Pereira, J.A.; Seabra, R.M.; Oliveira, B.P.P. Determination of sterol and fatty acid compositions, oxidative stability, and nutritional value of six walnut (Juglans regia L.) cultivars grown in Portugal. J. Agric. Food Chem. 2003, 51, 7698–7702. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Manchester, L.C.; Tan, D.X. Melatonin in walnuts: Influence on levels of melatonin and total antioxidant capacity of blood. Nutrition 2005, 21, 920–924. [Google Scholar] [CrossRef]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.; Gong, M.; Li, H.; Zhang, S.; Wang, Z.; Zhang, X.; Guo, S.; Qi, G. Effects of Different Shading Treatments on the Growth and Development of “Lvling” Juglans regia L. J. Northwest For. Univ. 2017, 32, 120–124. [Google Scholar]
- Dar, A.A.; Choudhury, A.R.; Kancharla, P.K.; Arumugam, N. The FAD2 gene in Plants: Occurrence, Regulation, and Role. Front. Plant Sci. 2017, 8, 1789. [Google Scholar] [CrossRef] [PubMed]
- Kachroo, A.; Shanklin, J.; Whittle, E.; Lapchyk, L.; Hildebrand, D.; Kachroo, P. The Arabidopsis stearoyl-acyl carrier protein-desaturase family and the contribution of leaf isoforms to oleic acid synthesis. Plant Mol. Biol. 2007, 63, 257–271. [Google Scholar] [CrossRef]
- Yuan, S.; Wu, X.; Liu, Z.-H.; Luo, H.-B.; Huang, R.-Z. Abiotic Stresses and Phytohormones Regulate Expression of FAD2 Gene in Arabidopsis thaliana. J. Integr. Agric. 2012, 12, 62–72. [Google Scholar] [CrossRef]
- Huang, C.; Huang, Y.; Wu, J.; Huang, R.; Luan, Y.; Zhang, S.; Wang, Z.; Zhang, Q.; Huang, J. SAD and FAD Genes Regulate the Ratio of Unsaturated Fatty Acid Components in Carya cathayensis. Acta Hortic. Sin. 2018, 45, 250–260. [Google Scholar]
- Bellaloui, N.; Smith, J.R.; Gillen, A.M.; Fisher, D.K.; Mengistu, A. Effect of Shade on Seed Protein, Oil, Fatty Acids, and Minerals in Soybean Lines Varying in Seed Germinability in the Early Soybean Production System. Am. J. Plant Sci. 2012, 3, 84–95. [Google Scholar] [CrossRef]
- Hao, B.; Zhao, Z.; Cai, Y.; Wang, L.; Li, M.; Rui, Y. Effects of different environmental conditions on composition of walnut oil. Fresen. Environ. Bull. 2021, 30, 3130–3133. [Google Scholar]
- Hou, G.; Ablett, G.R.; Pauls, K.P.; Rajcan, I. Environmental effects on fatty acid levels in soybean seed oil. J. Am. Oil Chem. Soc. 2006, 83, 759–763. [Google Scholar] [CrossRef]
- Ronde, J.A.D.; Mescht, A.V.D.; Steyn, H.S.F. Proline accumulation in response to drought and heat stress in cotton. Afr. Crop. Sci. J. 2000, 8, 85–91. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Rahman, M.A.; Rathinasabapathi, B.; Babar, A. UPLC-HRMS-based untargeted metabolic profiling reveals changes in chickpea (Cicer arietinum) metabolome following long-term drought stress. Plant Cell Environ. 2019, 42, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Showler, A.T. Effects of Water Deficit Stress, Shade, Weed Competition, and Kaolin Particle Film on Selected Foliar Free Amino Acid Accumulations in Cotton, Gossypium hirsutum (L.). J. Chem. Ecol. 2002, 28, 631–651. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.; Deng, Z.; Liu, J.; Li, Y.; Zhang, C.; Yao, S.; Zuo, H.; Shi, Y.; Yuan, S.; Qin, L.; et al. Effects of Preharvest Shading on Dynamic Changes in Metabolites, Gene Expression, and Enzyme Activity of Three Tea Types during Processing. J. Agric. Food Chem. 2022, 70, 14544–14558. [Google Scholar] [CrossRef]
- Muthuramalingam, P.; Jeyasri, R.; Bharathi, R.K.A.S.; Suba, V.; Pandian, S.T.K.; Ramesh, M. Global integrated omics expression analyses of abiotic stress signaling HSF transcription factor genes in Oryza sativa L.: An in silico approach. Genomics 2020, 112, 908–918. [Google Scholar] [CrossRef]
- Wang, S.; Yao, W.; Wei, H.; Jiang, T.; Zhou, B. Expression Patterns of ERF Genes Underlying Abiotic Stresses in Di-Haploid Populus simonii × P. nigra. Sci. World J. 2014, 2014, 745091. [Google Scholar]
- Li, H.; Sun, Q.; Zhao, S. Plant Physiological and Biochemical Principles and Experimental Techniques; Higher Education Press (HEP): Beijing, China, 2000; pp. 184–185. [Google Scholar]
- GB 5009.168-2016; National Food Safety Standard-Determination of Fatty Acid in Foods. ISO: Geneva, Switzerland, 2016.
- Wei, J.; Wu, C.; Jiang, Y. Sample preparation optimization for determination of soluble sugar in red jujube fruits by anthrone method. Food Sci. 2014, 35, 136–140. [Google Scholar]
- GB/T 5511-2008; Cereals and Pulses. Determination of the Nitrogen Content and Calculation of the Protein Content—Kjeldahl Method. ISO: Geneva, Switzerland, 2006.
- Thompson, O.; Meyenn, F.; Hewitt, Z.; Alexander, J.; Wood, A.; Weightman, R.; Gregory, S.; Krueger, F.; Andrews, S.; Barbaric, I.; et al. Low rates of mutation in clinical grade human pluripotent stem cells under different culture conditions. Nat. Commun. 2020, 23, 1528. [Google Scholar] [CrossRef]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef]
- Jin, J.; Tian, F.; Yang, D.; Meng, Y.; Kong, L.; Luo, J.; Gao, G. PlantTFDB 4.0: Toward a central hub for transcription factors and regulatory interactions in plants. Nucleic Acids Res. 2017, 45, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using Real-Time quantitative PCR and the 2−∆∆CT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fang, J.; Qi, X.; Lin, M.; Zhong, Y.; Sun, L.; Cui, W. Combined Analysis of the Fruit Metabolome and Transcriptome Reveals Candidate Genes Involved in Flavonoid Biosynthesis in Actinidia arguta. Int. J. Mol. Sci. 2018, 19, 1471. [Google Scholar] [CrossRef] [PubMed]



| KEGG Pathway | Compounds | 60 DAP | 90 DAP | 120 DAP |
|---|---|---|---|---|
| Ratio (S1/CK1) | Ratio (S2/CK2) | Ratio (S3/CK3) | ||
| Starch and sucrose metabolism (ko00500) | alpha, alpha’-Trehalose 6-phosphate | 1 | 0.49 | 1 |
| Glucose 6-phosphate | 1 | 0.48 | 1 | |
| Trehalose | 1 | 0.46 | 1 | |
| Biosynthesis of amino acids (ko01230) | L-Aspartic acid | 1 | 9.28 | 2.22 |
| 2-Oxoglutaric acid | 2.04 | 1 | 1 | |
| L-Citrulline | 1 | 2.65 | 8.92 | |
| 2-Isopropylmalic acid | 1 | 1 | 3.19 | |
| L-Tyrosine | 1 | 0.48 | 1 | |
| Citric acid | 1 | 2.15 | 1 | |
| D-Ribose 5-phosphate | 1 | 0.37 | 1 | |
| Pyruvic acid | 1 | 1 | 4.6 | |
| Glutamine | 1 | 2.04 | 1 | |
| Methionine | 1 | 0.47 | 1 | |
| Arginine | 1 | 3.24 | 1 | |
| Citrulline | 1 | 2.46 | 5.61 | |
| (-)-Riboflavin | 1 | 1 | 0.49 | |
| Glutathione metabolism (ko00480) | L-Glutathione | 1 | 0.47 | 0.36 |
| Vitamin C | 1 | 0.47 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, M.; Dong, Q.; Zhang, X.; Liu, Y.; Li, H.; Guo, S.; Luan, H.; Jia, P.; Yang, M.; Qi, G. Metabolomics and Transcriptomics Analyses Reveals the Molecular Regulatory Mechanisms of Walnut (Juglans regia L.) Embryos in Response to Shade Treatment. Int. J. Mol. Sci. 2023, 24, 10871. https://doi.org/10.3390/ijms241310871
Liang M, Dong Q, Zhang X, Liu Y, Li H, Guo S, Luan H, Jia P, Yang M, Qi G. Metabolomics and Transcriptomics Analyses Reveals the Molecular Regulatory Mechanisms of Walnut (Juglans regia L.) Embryos in Response to Shade Treatment. International Journal of Molecular Sciences. 2023; 24(13):10871. https://doi.org/10.3390/ijms241310871
Chicago/Turabian StyleLiang, Manman, Qinglong Dong, Xuemei Zhang, Yang Liu, Han Li, Suping Guo, Haoan Luan, Peng Jia, Minsheng Yang, and Guohui Qi. 2023. "Metabolomics and Transcriptomics Analyses Reveals the Molecular Regulatory Mechanisms of Walnut (Juglans regia L.) Embryos in Response to Shade Treatment" International Journal of Molecular Sciences 24, no. 13: 10871. https://doi.org/10.3390/ijms241310871
APA StyleLiang, M., Dong, Q., Zhang, X., Liu, Y., Li, H., Guo, S., Luan, H., Jia, P., Yang, M., & Qi, G. (2023). Metabolomics and Transcriptomics Analyses Reveals the Molecular Regulatory Mechanisms of Walnut (Juglans regia L.) Embryos in Response to Shade Treatment. International Journal of Molecular Sciences, 24(13), 10871. https://doi.org/10.3390/ijms241310871




