Tissue-Specific Regulation of CFTR Gene Expression
Abstract
1. Introduction
2. Clinical Issues
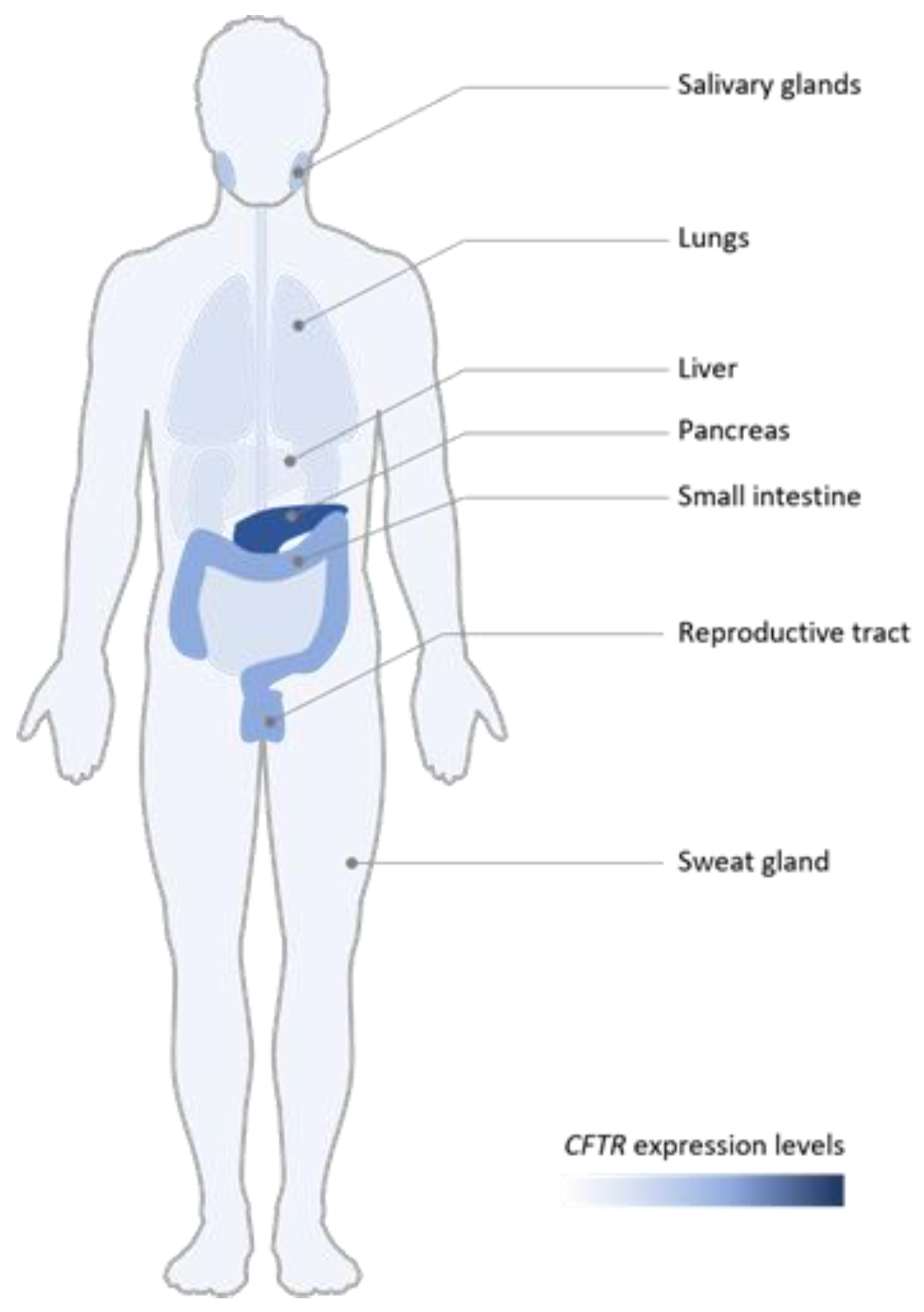
3. Tissue Expression
4. Characteristics of the CFTR Locus
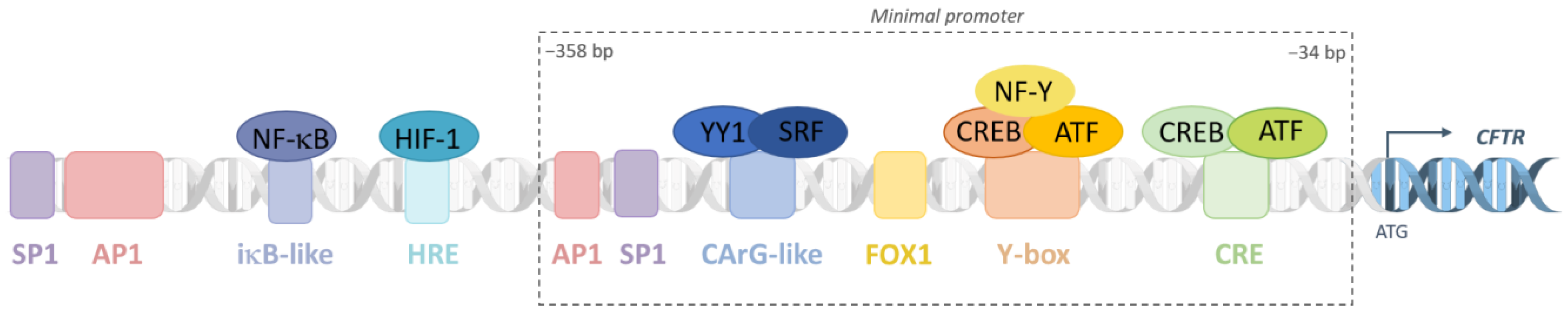
4.1. Promoter as a Housekeeping Gene
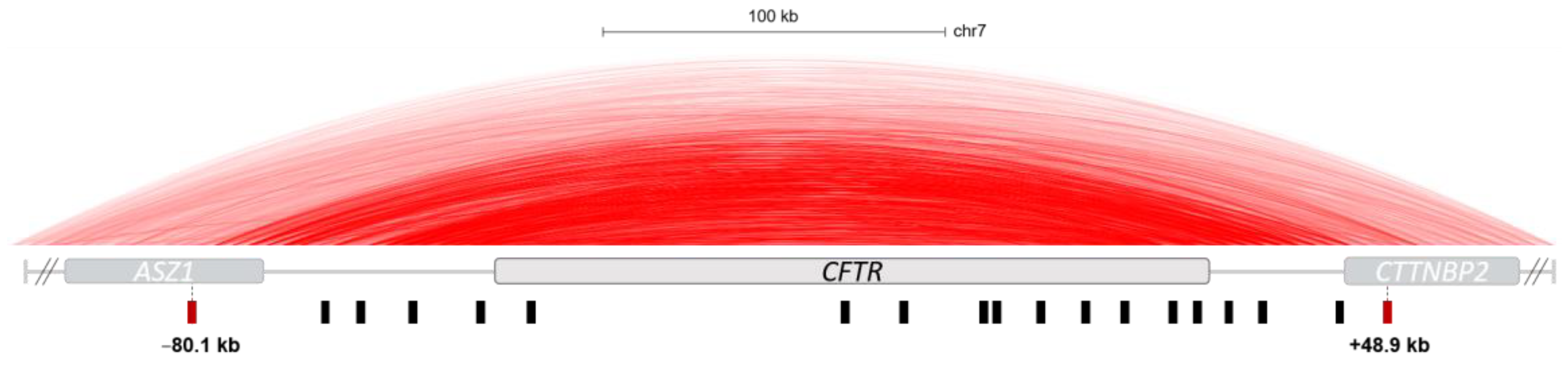
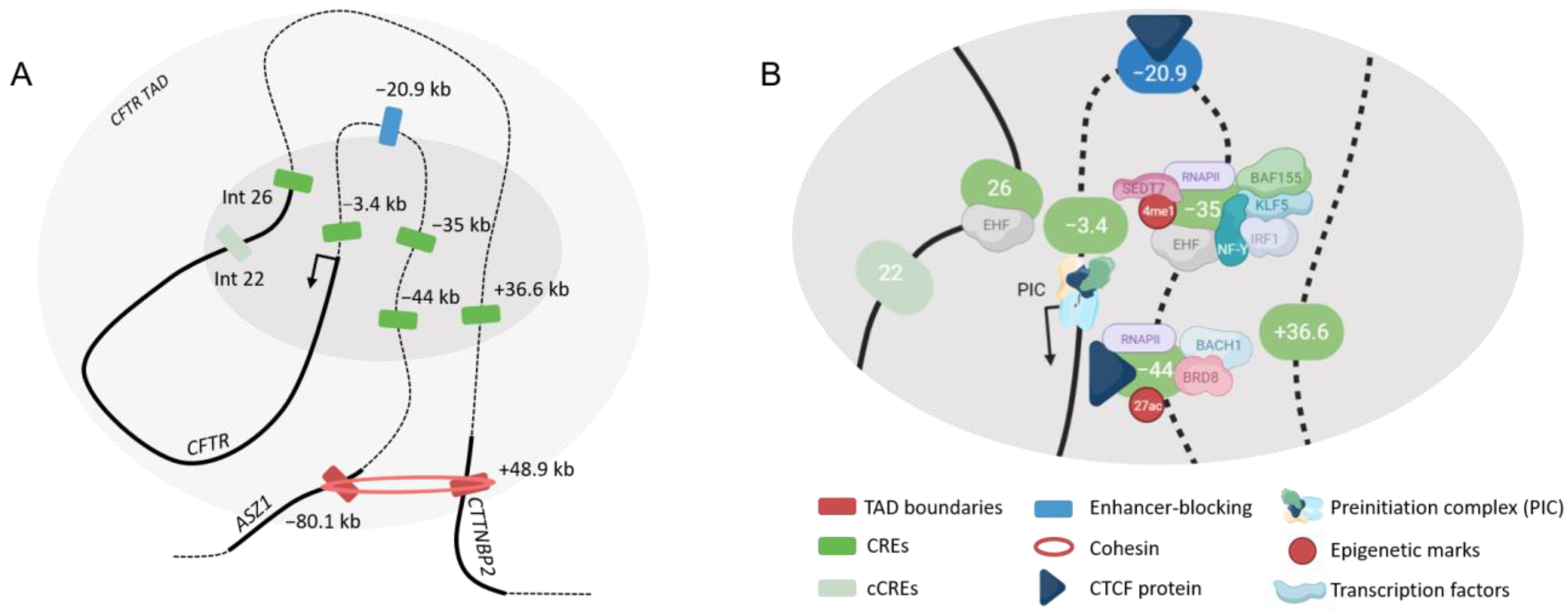
4.2. Cis-Regulation of the CFTR Gene
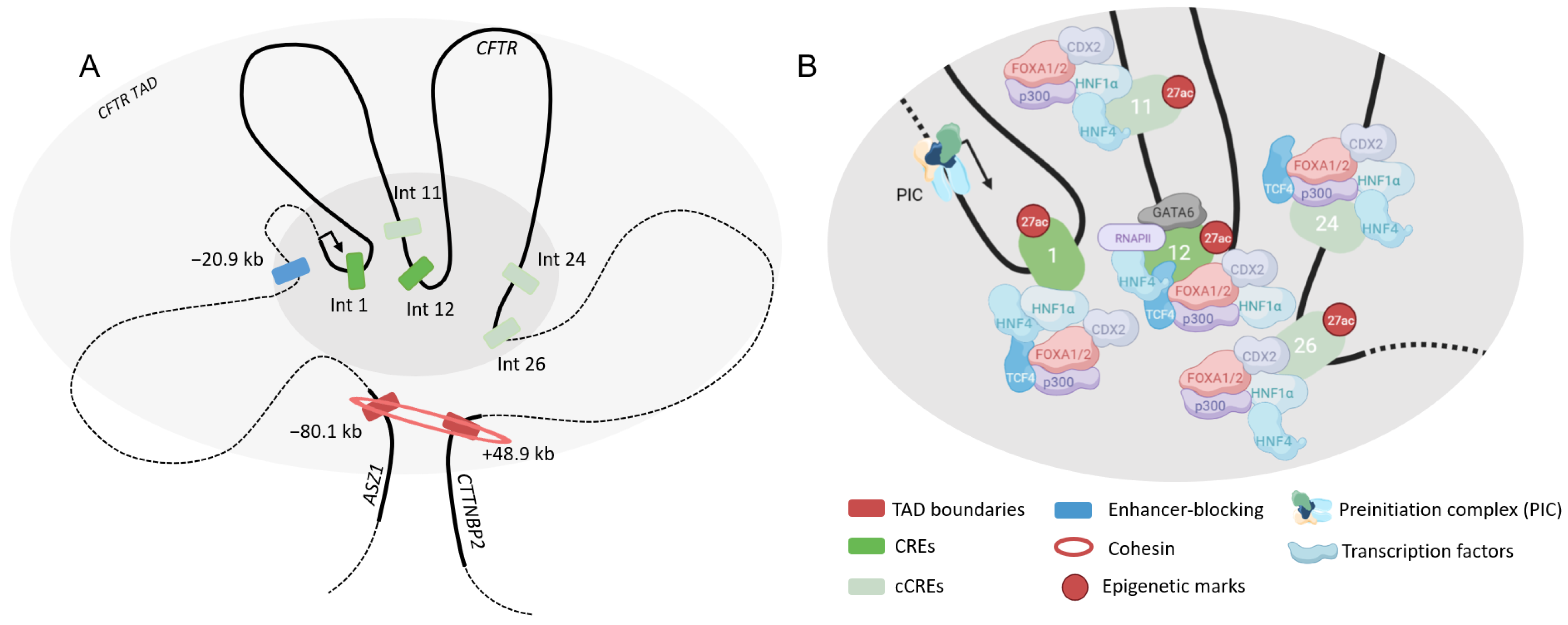
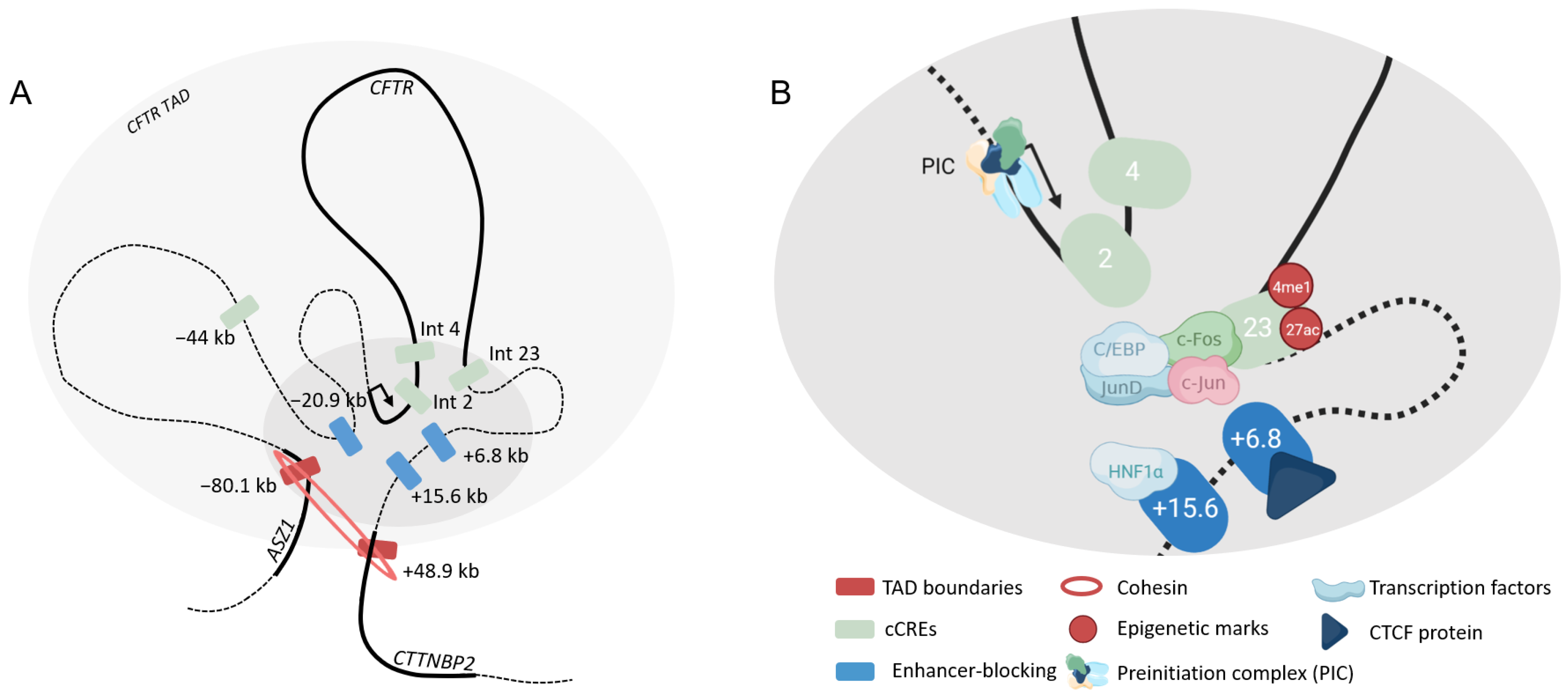
4.2.1. Airway
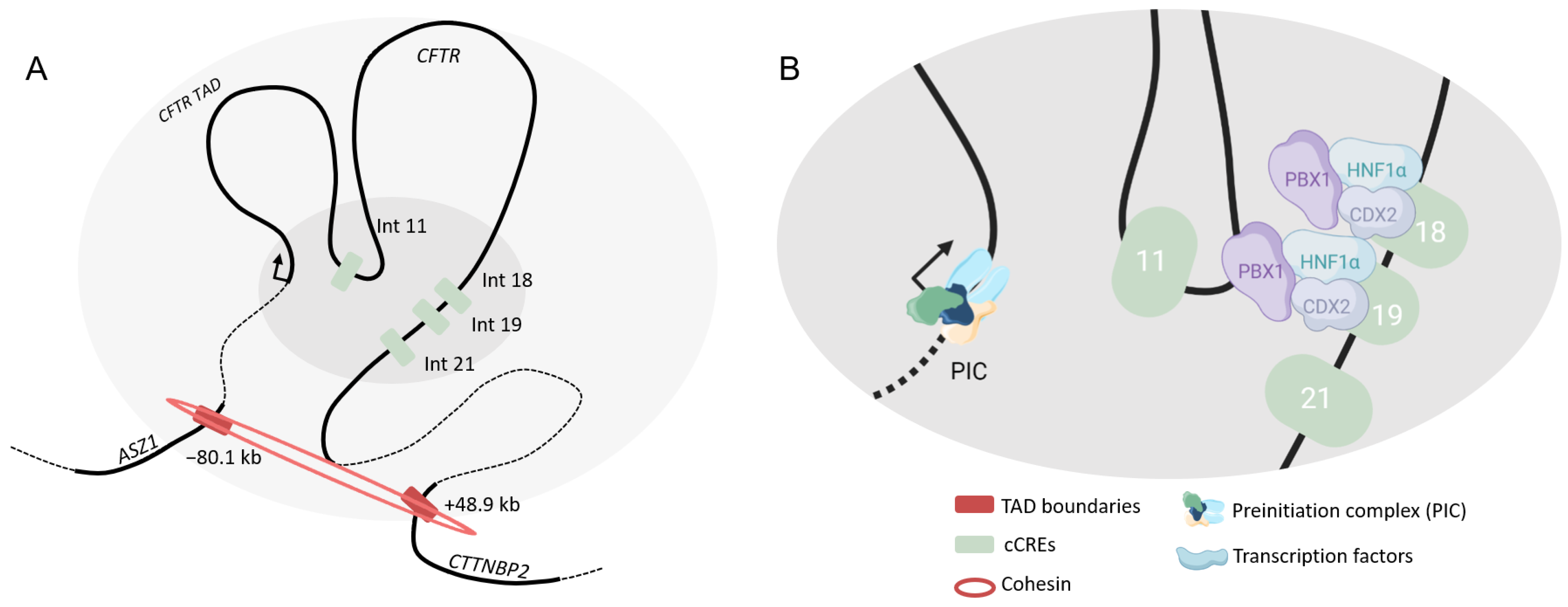
4.2.2. Intestine
4.2.3. Epididymis
4.2.4. Pancreas
4.3. Impact of CFTR Cis-Regulatory Variants?
4.4. microRNA and lncRNA
4.5. Modifier Genes
4.5.1. Pulmonary
4.5.2. Intestines
4.5.3. Epididymis
4.5.4. Pancreas
4.6. Complex Alleles
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Andersen, D.H. Cystic Fibrosis of the Pancreas and Its Relation to Celiac Disease: A Clinical and Pathologic Study. Am. J. Dis. Child. 1938, 56, 344. [Google Scholar] [CrossRef]
- Fanconi, G.; Uehlinger, E.; Knauer, C. Das Coeliakiesyndrom Bei Angeborenerzysticher Pankreasfibromatose Und Bronchiektasien. Wien. Med. Wochenschr. 1936, 86, 753–756. [Google Scholar]
- Andersen, D.H.; Hodges, R. Celiac Syndrome: V. Genetics of Cystic Fibrosis of the Pancreas with a Consideration of Etiology. Am. J. Dis. Child. 1946, 72, 62–80. [Google Scholar] [CrossRef] [PubMed]
- May, C.D. Fibrosis of the Pancreas in Infants and Children. J. Pediatr. 1949, 34, 663–687. [Google Scholar] [CrossRef]
- Kerem, B.-S.; Rommens, J.M.; Buchanan, J.A.; Markiewicz, D.; Cox, T.K.; Chakravarti, A.; Buchwald, M.; Tsui, L.-C. Identification of the Cystic Fibrosis Gene: Genetic Analysis. Science 1989, 245, 1073–1080. [Google Scholar] [CrossRef]
- Riordan, J.R.; Rommens, J.M.; Kerem, B.-S.; Alon, N.; Rozmahel, R.; Grzelczak, Z.; Zielenski, J.; Lok, S.; Plavsic, N.; Chou, J.-L.; et al. Identification of the Cystic Fibrosis Gene: Cloning and Characterization of Complementary DNA. Science 1989, 245, 1066–1073. [Google Scholar] [CrossRef]
- Rommens, J.M.; Iannuzzi, M.C.; Kerem, B.; Drumm, M.L.; Melmer, G.; Dean, M.; Rozmahel, R.; Cole, J.L.; Kennedy, D.; Hidaka, N. Identification of the Cystic Fibrosis Gene: Chromosome Walking and Jumping. Science 1989, 245, 1059–1065. [Google Scholar] [CrossRef]
- Becq, F. CFTR et mucoviscidose, une histoire cinquantenaire. Med. Sci. 2021, 37, 654–659. [Google Scholar] [CrossRef]
- De Boeck, K.; Amaral, M.D. Progress in Therapies for Cystic Fibrosis. Lancet Respir. Med. 2016, 4, 662–674. [Google Scholar] [CrossRef]
- Castellani, C.; Assael, B.M. Cystic Fibrosis: A Clinical View. Cell. Mol. Life Sci. 2017, 74, 129–140. [Google Scholar] [CrossRef]
- Bombieri, C.; Claustres, M.; De Boeck, K.; Derichs, N.; Dodge, J.; Girodon, E.; Sermet, I.; Schwarz, M.; Tzetis, M.; Wilschanski, M.; et al. Recommendations for the Classification of Diseases as CFTR-Related Disorders. J. Cyst. Fibros. 2011, 10 (Suppl. S2), S86–S102. [Google Scholar] [CrossRef]
- Bieth, E.; Hamdi, S.M.; Mieusset, R. Genetics of the Congenital Absence of the Vas Deferens. Hum. Genet. 2021, 140, 59–76. [Google Scholar] [CrossRef]
- Zhang, D.-L.; Sun, Y.-J.; Ma, M.-L.; Wang, Y.; Lin, H.; Li, R.-R.; Liang, Z.-L.; Gao, Y.; Yang, Z.; He, D.-F.; et al. Gq Activity- and β-Arrestin-1 Scaffolding-Mediated ADGRG2/CFTR Coupling Are Required for Male Fertility. eLife 2018, 7, e33432. [Google Scholar] [CrossRef]
- Xu, W.M.; Shi, Q.X.; Chen, W.Y.; Zhou, C.X.; Ni, Y.; Rowlands, D.K.; Yi Liu, G.; Zhu, H.; Ma, Z.G.; Wang, X.F.; et al. Cystic Fibrosis Transmembrane Conductance Regulator Is Vital to Sperm Fertilizing Capacity and Male Fertility. Proc. Natl. Acad. Sci. USA 2007, 104, 9816–9821. [Google Scholar] [CrossRef]
- Angyal, D.; Bijvelds, M.J.C.; Bruno, M.J.; Peppelenbosch, M.P.; de Jonge, H.R. Bicarbonate Transport in Cystic Fibrosis and Pancreatitis. Cells 2021, 11, 54. [Google Scholar] [CrossRef]
- Harris, A.; Chalkley, G.; Goodman, S.; Coleman, L. Expression of the Cystic Fibrosis Gene in Human Development. Development 1991, 113, 305–310. [Google Scholar] [CrossRef]
- Montoro, D.T.; Haber, A.L.; Biton, M.; Vinarsky, V.; Lin, B.; Birket, S.E.; Yuan, F.; Chen, S.; Leung, H.M.; Villoria, J.; et al. A Revised Airway Epithelial Hierarchy Includes CFTR-Expressing Ionocytes. Nature 2018, 560, 319–324. [Google Scholar] [CrossRef]
- Plasschaert, L.W.; Žilionis, R.; Choo-Wing, R.; Savova, V.; Knehr, J.; Roma, G.; Klein, A.M.; Jaffe, A.B. A Single-Cell Atlas of the Airway Epithelium Reveals the CFTR-Rich Pulmonary Ionocyte. Nature 2018, 560, 377–381. [Google Scholar] [CrossRef]
- Okuda, K.; Dang, H.; Kobayashi, Y.; Carraro, G.; Nakano, S.; Chen, G.; Kato, T.; Asakura, T.; Gilmore, R.C.; Morton, L.C.; et al. Secretory Cells Dominate Airway CFTR Expression and Function in Human Airway Superficial Epithelia. Am. J. Respir. Crit. Care Med. 2021, 203, 1275–1289. [Google Scholar] [CrossRef]
- Yoshimura, K.; Nakamura, H.; Trapnell, B.C.; Dalemans, W.; Pavirani, A.; Lecocq, J.P.; Crystal, R.G. The Cystic Fibrosis Gene Has a “Housekeeping”-Type Promoter and Is Expressed at Low Levels in Cells of Epithelial Origin. J. Biol. Chem. 1991, 266, 9140–9144. [Google Scholar] [CrossRef]
- Chou, J.L.; Rozmahel, R.; Tsui, L.C. Characterization of the Promoter Region of the Cystic Fibrosis Transmembrane Conductance Regulator Gene. J. Biol. Chem. 1991, 266, 24471–24476. [Google Scholar] [CrossRef] [PubMed]
- Nuthall, H.N.; Moulin, D.S.; Huxley, C.; Harris, A. Analysis of DNase-I-Hypersensitive Sites at the 3’ End of the Cystic Fibrosis Transmembrane Conductance Regulator Gene (CFTR). Biochem. J. 1999, 341, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Brouillard, F.; Bouthier, M.; Leclerc, T.; Clement, A.; Baudouin-Legros, M.; Edelman, A. NF-ΚB Mediates Up-Regulation of CFTR Gene Expression in Calu-3 Cells by Interleukin-1β. J. Biol. Chem. 2001, 276, 9486–9491. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Kuhlicke, J.; Jäckel, K.; Eltzschig, H.K.; Singh, A.; Sjöblom, M.; Riederer, B.; Weinhold, C.; Seidler, U.; Colgan, S.P.; et al. Hypoxia Inducible Factor-1 (HIF-1)-Mediated Repression of Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) in the Intestinal Epithelium. FASEB J. 2009, 23, 204–213. [Google Scholar] [CrossRef] [PubMed]
- ENCODE Project Consortium. The ENCODE (ENCyclopedia of DNA Elements) Project. Science 2004, 306, 636–640. [Google Scholar] [CrossRef]
- Gasperini, M.; Tome, J.M.; Shendure, J. Towards a Comprehensive Catalogue of Validated and Target-Linked Human Enhancers. Nat. Rev. Genet. 2020, 21, 292–310. [Google Scholar] [CrossRef]
- Yang, R.; Kerschner, J.L.; Gosalia, N.; Neems, D.; Gorsic, L.K.; Safi, A.; Crawford, G.E.; Kosak, S.T.; Leir, S.-H.; Harris, A. Differential Contribution of Cis-Regulatory Elements to Higher Order Chromatin Structure and Expression of the CFTR Locus. Nucleic Acids Res. 2016, 44, 3082–3094. [Google Scholar] [CrossRef]
- Smith, E.M.; Lajoie, B.R.; Jain, G.; Dekker, J. Invariant TAD Boundaries Constrain Cell-Type-Specific Looping Interactions between Promoters and Distal Elements around the CFTR Locus. Am. J. Hum. Genet. 2016, 98, 185–201. [Google Scholar] [CrossRef]
- Blackledge, N.P.; Carter, E.J.; Evans, J.R.; Lawson, V.; Rowntree, R.K.; Harris, A. CTCF Mediates Insulator Function at the CFTR Locus. Biochem. J. 2007, 408, 267–275. [Google Scholar] [CrossRef]
- Rowntree, R.; Harris, A. DNA Polymorphisms in Potential Regulatory Elements of the CFTR Gene Alter Transcription Factor Binding. Hum. Genet. 2002, 111, 66–74. [Google Scholar] [CrossRef]
- Zhang, Z.; Ott, C.J.; Lewandowska, M.A.; Leir, S.-H.; Harris, A. Molecular Mechanisms Controlling CFTR Gene Expression in the Airway. J. Cell. Mol. Med. 2012, 16, 1321–1330. [Google Scholar] [CrossRef]
- Zhang, Z.; Leir, S.-H.; Harris, A. Oxidative Stress Regulates CFTR Gene Expression in Human Airway Epithelial Cells through a Distal Antioxidant Response Element. Am. J. Respir. Cell. Mol. Biol. 2015, 52, 387–396. [Google Scholar] [CrossRef]
- NandyMazumdar, M.; Yin, S.; Paranjapye, A.; Kerschner, J.L.; Swahn, H.; Ge, A.; Leir, S.-H.; Harris, A. Looping of Upstream Cis-Regulatory Elements Is Required for CFTR Expression in Human Airway Epithelial Cells. Nucleic Acids Res. 2020, 48, 3513–3524. [Google Scholar] [CrossRef]
- Harrison, L.J.; Bose, D. Enhancer RNAs Step Forward: New Insights into Enhancer Function. Development 2022, 149, dev200398. [Google Scholar] [CrossRef]
- Yigit, E.; Bischof, J.M.; Zhang, Z.; Ott, C.J.; Kerschner, J.L.; Leir, S.-H.; Buitrago-Delgado, E.; Zhang, Q.; Wang, J.-P.Z.; Widom, J.; et al. Nucleosome Mapping across the CFTR Locus Identifies Novel Regulatory Factors. Nucleic Acids Res. 2013, 41, 2857–2868. [Google Scholar] [CrossRef]
- Zhang, Z.; Leir, S.-H.; Harris, A. Immune Mediators Regulate CFTR Expression through a Bifunctional Airway-Selective Enhancer. Mol. Cell. Biol. 2013, 33, 2843–2853. [Google Scholar] [CrossRef]
- Mutolo, M.J.; Leir, S.-H.; Fossum, S.L.; Browne, J.A.; Harris, A. A Transcription Factor Network Represses CFTR Gene Expression in Airway Epithelial Cells. Biochem. J. 2018, 475, 1323–1334. [Google Scholar] [CrossRef]
- NandyMazumdar, M.; Paranjapye, A.; Browne, J.; Yin, S.; Leir, S.-H.; Harris, A. BACH1, the Master Regulator of Oxidative Stress, has a Dual Effect on CFTR Expression. Biochem. J. 2021, 478, 3741–3756. [Google Scholar] [CrossRef]
- Browne, J.A.; NandyMazumdar, M.; Paranjapye, A.; Leir, S.-H.; Harris, A. The Bromodomain Containing 8 (BRD8) Transcriptional Network in Human Lung Epithelial Cells. Mol. Cell. Endocrinol. 2021, 524, 111169. [Google Scholar] [CrossRef]
- Moisan, S.; Berlivet, S.; Ka, C.; Le Gac, G.; Dostie, J.; Férec, C. Analysis of Long-Range Interactions in Primary Human Cells Identifies Cooperative CFTR Regulatory Elements. Nucleic Acids Res. 2016, 44, 2564–2576. [Google Scholar] [CrossRef]
- Ott, C.J.; Blackledge, N.P.; Kerschner, J.L.; Leir, S.-H.; Crawford, G.E.; Cotton, C.U.; Harris, A. Intronic Enhancers Coordinate Epithelial-Specific Looping of the Active CFTR Locus. Proc. Natl. Acad. Sci. USA 2009, 106, 19934–19939. [Google Scholar] [CrossRef] [PubMed]
- Ott, C.J.; Suszko, M.; Blackledge, N.P.; Wright, J.E.; Crawford, G.E.; Harris, A. A Complex Intronic Enhancer Regulates Expression of the CFTR Gene by Direct Interaction with the Promoter. J. Cell. Mol. Med. 2009, 13, 680–692. [Google Scholar] [CrossRef] [PubMed]
- Gosalia, N.; Neems, D.; Kerschner, J.L.; Kosak, S.T.; Harris, A. Architectural Proteins CTCF and Cohesin Have Distinct Roles in Modulating the Higher Order Structure and Expression of the CFTR Locus. Nucleic Acids Res. 2014, 42, 9612–9622. [Google Scholar] [CrossRef] [PubMed]
- Collobert, M.; Bocher, O.; Le Nabec, A.; Génin, E.; Férec, C.; Moisan, S. CFTR Cooperative Cis-Regulatory Elements in Intestinal Cells. Int. J. Mol. Sci. 2021, 22, 2599. [Google Scholar] [CrossRef] [PubMed]
- Van Mierlo, G.; Pushkarev, O.; Kribelbauer, J.F.; Deplancke, B. Chromatin Modules and Their Implication in Genomic Organization and Gene Regulation. Trends Genet. 2022, 39, 140–153. [Google Scholar] [CrossRef] [PubMed]
- Mouchel, N.; Henstra, S.A.; McCarthy, V.A.; Williams, S.H.; Phylactides, M.; Harris, A. HNF1alpha Is Involved in Tissue-Specific Regulation of CFTR Gene Expression. Biochem. J. 2004, 378, 909–918. [Google Scholar] [CrossRef]
- Paul, T.; Li, S.; Khurana, S.; Leleiko, N.S.; Walsh, M.J. The Epigenetic Signature of CFTR Expression Is Co-Ordinated via Chromatin Acetylation through a Complex Intronic Element. Biochem. J. 2007, 408, 317–326. [Google Scholar] [CrossRef]
- Yin, S.; NandyMazumdar, M.; Paranjapye, A.; Harris, A. Cross-Talk between Enhancers, Structural Elements and Activating Transcription Factors Maintains the 3D Architecture and Expression of the CFTR Gene. Genomics 2022, 114, 110350. [Google Scholar] [CrossRef]
- Rowntree, R.K.; Vassaux, G.; McDowell, T.L.; Howe, S.; McGuigan, A.; Phylactides, M.; Huxley, C.; Harris, A. An Element in Intron 1 of the CFTR Gene Augments Intestinal Expression in Vivo. Hum. Mol. Genet. 2001, 10, 1455–1464. [Google Scholar] [CrossRef]
- Blackledge, N.P.; Ott, C.J.; Gillen, A.E.; Harris, A. An Insulator Element 3’ to the CFTR Gene Binds CTCF and Reveals an Active Chromatin Hub in Primary Cells. Nucleic Acids Res. 2009, 37, 1086–1094. [Google Scholar] [CrossRef]
- Smith, D.J.; Nuthall, H.N.; Majetti, M.E.; Harris, A. Multiple Potential Intragenic Regulatory Elements in the CFTR Gene. Genomics 2000, 64, 90–96. [Google Scholar] [CrossRef]
- Bischof, J.M.; Gillen, A.E.; Song, L.; Gosalia, N.; London, D.; Furey, T.S.; Crawford, G.E.; Harris, A. A Genome-Wide Analysis of Open Chromatin in Human Epididymis Epithelial Cells Reveals Candidate Regulatory Elements for Genes Coordinating Epididymal Function. Biol. Reprod. 2013, 89, 104. [Google Scholar] [CrossRef]
- Kerschner, J.L.; Gosalia, N.; Leir, S.-H.; Harris, A. Chromatin Remodeling Mediated by the FOXA1/A2 Transcription Factors Activates CFTR Expression in Intestinal Epithelial Cells. Epigenetics 2014, 9, 557–565. [Google Scholar] [CrossRef]
- Browne, J.A.; Yang, R.; Eggener, S.E.; Leir, S.-H.; Harris, A. HNF1 Regulates Critical Processes in the Human Epididymis Epithelium. Mol. Cell. Endocrinol. 2016, 425, 94–102. [Google Scholar] [CrossRef]
- ENCODE Project Consortium. An Integrated Encyclopedia of DNA Elements in the Human Genome. Nature 2012, 489, 57–74. [Google Scholar] [CrossRef]
- McCarthy, V.A.; Ott, C.J.; Phylactides, M.; Harris, A. Interaction of Intestinal and Pancreatic Transcription Factors in the Regulation of CFTR Gene Expression. Biochim. Biophys. Acta 2009, 1789, 709–718. [Google Scholar] [CrossRef]
- Sancho, A.; Li, S.; Paul, T.; Zhang, F.; Aguilo, F.; Vashisht, A.; Balasubramaniyan, N.; Leleiko, N.S.; Suchy, F.J.; Wohlschlegel, J.A.; et al. CHD6 Regulates the Topological Arrangement of the CFTR Locus. Hum. Mol. Genet. 2015, 24, 2724–2732. [Google Scholar] [CrossRef]
- Phylactides, M.; Rowntree, R.; Nuthall, H.; Ussery, D.; Wheeler, A.; Harris, A. Evaluation of Potential Regulatory Elements Identified as DNase I Hypersensitive Sites in the CFTR Gene. Eur. J. Biochem. 2002, 269, 553–559. [Google Scholar] [CrossRef]
- Zaugg, J.B.; Sahlén, P.; Andersson, R.; Alberich-Jorda, M.; de Laat, W.; Deplancke, B.; Ferrer, J.; Mandrup, S.; Natoli, G.; Plewczynski, D.; et al. Current Challenges in Understanding the Role of Enhancers in Disease. Nat. Struct. Mol. Biol. 2022, 29, 1148–1158. [Google Scholar] [CrossRef]
- Vecchio-Pagán, B.; Blackman, S.M.; Lee, M.; Atalar, M.; Pellicore, M.J.; Pace, R.G.; Franca, A.L.; Raraigh, K.S.; Sharma, N.; Knowles, M.R.; et al. Deep Resequencing of CFTR in 762 F508del Homozygotes Reveals Clusters of Non-Coding Variants Associated with Cystic Fibrosis Disease Traits. Hum. Genome Var. 2016, 3, 16038. [Google Scholar] [CrossRef]
- Kerschner, J.L.; Ghosh, S.; Paranjapye, A.; Cosme, W.R.; Audrézet, M.-P.; Nakakuki, M.; Ishiguro, H.; Férec, C.; Rommens, J.; Harris, A. Screening for Regulatory Variants in 460 Kb Encompassing the CFTR Locus in Cystic Fibrosis Patients. J. Mol. Diagn. 2019, 21, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, S.; Karp, P.H.; Osterhaus, S.R.; Jiang, P.; Wohlford-Lenane, C.; Lennox, K.A.; Jacobi, A.M.; Praekh, K.; Rose, S.D.; Behlke, M.A.; et al. Post-Transcriptional Regulation of Cystic Fibrosis Transmembrane Conductance Regulator Expression and Function by MicroRNAs. Am. J. Respir. Cell. Mol. Biol. 2013, 49, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Gillen, A.E.; Gosalia, N.; Leir, S.-H.; Harris, A. MicroRNA Regulation of Expression of the Cystic Fibrosis Transmembrane Conductance Regulator Gene. Biochem. J. 2011, 438, 25–32. [Google Scholar] [CrossRef]
- Viart, V.; Bergougnoux, A.; Bonini, J.; Varilh, J.; Chiron, R.; Tabary, O.; Molinari, N.; Claustres, M.; Taulan-Cadars, M. Transcription Factors and MiRNAs That Regulate Fetal to Adult CFTR Expression Change Are New Targets for Cystic Fibrosis. Eur. Respir. J. 2015, 45, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Landgraf, P.; Rusu, M.; Sheridan, R.; Sewer, A.; Iovino, N.; Aravin, A.; Pfeffer, S.; Rice, A.; Kamphorst, A.O.; Landthaler, M.; et al. A Mammalian MicroRNA Expression Atlas Based on Small RNA Library Sequencing. Cell 2007, 129, 1401–1414. [Google Scholar] [CrossRef]
- Browne, J.A.; Leir, S.-H.; Eggener, S.E.; Harris, A. Region-Specific MicroRNA Signatures in the Human Epididymis. Asian J. 2018, 20, 539–544. [Google Scholar] [CrossRef]
- Glasgow, A.M.A.; De Santi, C.; Greene, C.M. Non-Coding RNA in Cystic Fibrosis. Biochem. Soc. Trans. 2018, 46, 619–630. [Google Scholar] [CrossRef]
- McKiernan, P.J.; Molloy, K.; Cryan, S.A.; McElvaney, N.G.; Greene, C.M. Long Noncoding RNA Are Aberrantly Expressed in Vivo in the Cystic Fibrosis Bronchial Epithelium. Int. J. Biochem. Cell. Biol. 2014, 52, 184–191. [Google Scholar] [CrossRef]
- Balloy, V.; Koshy, R.; Perra, L.; Corvol, H.; Chignard, M.; Guillot, L.; Scaria, V. Bronchial Epithelial Cells from Cystic Fibrosis Patients Express a Specific Long Non-Coding RNA Signature upon Pseudomonas Aeruginosa Infection. Front. Cell. Infect. Microbiol. 2017, 7, 218. [Google Scholar] [CrossRef]
- Saayman, S.M.; Ackley, A.; Burdach, J.; Clemson, M.; Gruenert, D.C.; Tachikawa, K.; Chivukula, P.; Weinberg, M.S.; Morris, K.V. Long Non-Coding RNA BGas Regulates the Cystic Fibrosis Transmembrane Conductance Regulator. Mol. Ther. 2016, 24, 1351–1357. [Google Scholar] [CrossRef]
- Corvol, H.; Blackman, S.M.; Boëlle, P.-Y.; Gallins, P.J.; Pace, R.G.; Stonebraker, J.R.; Accurso, F.J.; Clement, A.; Collaco, J.M.; Dang, H.; et al. Genome-Wide Association Meta-Analysis Identifies Five Modifier Loci of Lung Disease Severity in Cystic Fibrosis. Nat. Commun. 2015, 6, 8382. [Google Scholar] [CrossRef]
- Zhou, Y.-H.; Gallins, P.J.; Pace, R.G.; Dang, H.; Aksit, M.A.; Blue, E.E.; Buckingham, K.J.; Collaco, J.M.; Faino, A.V.; Gordon, W.W.; et al. Genetic Modifiers of Cystic Fibrosis Lung Disease Severity: Whole Genome Analysis of 7840 Patients. Am. J. Respir. Crit. Care Med. 2023, 207, 1324–1333. [Google Scholar] [CrossRef]
- Jonckheere, N.; Van Seuningen, I. Integrative Analysis of the Cancer Genome Atlas and Cancer Cell Lines Encyclopedia Large-Scale Genomic Databases: MUC4/MUC16/MUC20 Signature Is Associated with Poor Survival in Human Carcinomas. J. Transl. Med. 2018, 16, 259. [Google Scholar] [CrossRef]
- Dorfman, R.; Taylor, C.; Lin, F.; Sun, L.; Sandford, A.; Paré, P.; Berthiaume, Y.; Corey, M.; Durie, P.; Zielenski, J.; et al. Modulatory Effect of the SLC9A3 Gene on Susceptibility to Infections and Pulmonary Function in Children with Cystic Fibrosis. Pediatr. Pulmonol. 2011, 46, 385–392. [Google Scholar] [CrossRef]
- Zhong, L.; Strug, L.J. RoPE: A Robust Profile Likelihood Method for Differential Gene Expression Analysis. Genet. Epidemiol. 2023, 47, 379–393. [Google Scholar] [CrossRef]
- Mercier, J.; Calmel, C.; Mésinèle, J.; Sutanto, E.; Merabtene, F.; Longchampt, E.; Sage, E.; Kicic, A.; Boëlle, P.-Y.; Corvol, H.; et al. SLC6A14 Impacts Cystic Fibrosis Lung Disease Severity via MTOR and Epithelial Repair Modulation. Front. Mol. Biosci. 2022, 9, 850261. [Google Scholar] [CrossRef]
- Fossum, S.L.; Mutolo, M.J.; Tugores, A.; Ghosh, S.; Randell, S.H.; Jones, L.C.; Leir, S.-H.; Harris, A. Ets Homologous Factor (EHF) Has Critical Roles in Epithelial Dysfunction in Airway Disease. J. Biol. Chem. 2017, 292, 10938–10949. [Google Scholar] [CrossRef]
- Butnariu, L.I.; Țarcă, E.; Cojocaru, E.; Rusu, C.; Moisă, Ș.M.; Leon Constantin, M.-M.; Gorduza, E.V.; Trandafir, L.M. Genetic Modifying Factors of Cystic Fibrosis Phenotype: A Challenge for Modern Medicine. J. Clin. Med. 2021, 10, 5821. [Google Scholar] [CrossRef]
- Sun, L.; Rommens, J.M.; Corvol, H.; Li, W.; Li, X.; Chiang, T.A.; Lin, F.; Dorfman, R.; Busson, P.-F.; Parekh, R.V.; et al. Multiple Apical Plasma Membrane Constituents Are Associated with Susceptibility to Meconium Ileus in Individuals with Cystic Fibrosis. Nat. Genet. 2012, 44, 562–569. [Google Scholar] [CrossRef]
- Gong, J.; Wang, F.; Xiao, B.; Panjwani, N.; Lin, F.; Keenan, K.; Avolio, J.; Esmaeili, M.; Zhang, L.; He, G.; et al. Genetic Association and Transcriptome Integration Identify Contributing Genes and Tissues at Cystic Fibrosis Modifier Loci. PLoS Genet. 2019, 15, e1008007. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Lin, Y.-H.; Wu, Y.-N.; Chen, Y.-L.; Lin, Y.-C.; Cheng, C.-Y.; Chiang, H.-S. Loss of SLC9A3 Decreases CFTR Protein and Causes Obstructed Azoospermia in Mice. PLoS Genet. 2017, 13, e1006715. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.; Mavuduru, R.S.; Singh, S.K.; Prasad, R. Heterogeneous Spectrum of Mutations in CFTR Gene from Indian Patients with Congenital Absence of the Vas Deferens and Their Association with Cystic Fibrosis Genetic Modifiers. Mol. Hum. Reprod. 2014, 20, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.; Pierucci-Alves, F.; Schultz, B.D. Transforming Growth Factor-Β1 Impairs CFTR-Mediated Anion Secretion across Cultured Porcine Vas Deferens Epithelial Monolayer via the P38 MAPK Pathway. Am. J. Physiol.-Cell. Physiol. 2013, 305, C867–C876. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Trouvé, P.; Génin, E.; Férec, C. In Silico Search for Modifier Genes Associated with Pancreatic and Liver Disease in Cystic Fibrosis. PLoS ONE 2017, 12, e0173822. [Google Scholar] [CrossRef]
- Felderbauer, P.; Hoffmann, P.; Einwächter, H.; Bulut, K.; Ansorge, N.; Schmitz, F.; Schmidt, W.E. A Novel Mutation of the Calcium Sensing Receptor Gene Is Associated with Chronic Pancreatitis in a Family with Heterozygous SPINK1 Mutations. BMC Gastroenterol. 2003, 3, 34. [Google Scholar] [CrossRef]
- Szmola, R.; Sahin-Toth, M. Pancreatitis-Associated Chymotrypsinogen C (CTRC) Mutant Elicits Endoplasmic Reticulum Stress in Pancreatic Acinar Cells. Gut 2010, 59, 365–372. [Google Scholar] [CrossRef]
- Sofia, V.M.; Surace, C.; Terlizzi, V.; Da Sacco, L.; Alghisi, F.; Angiolillo, A.; Braggion, C.; Cirilli, N.; Colombo, C.; Di Lullo, A.; et al. Trans-Heterozygosity for Mutations Enhances the Risk of Recurrent/Chronic Pancreatitis in Patients with Cystic Fibrosis. Mol. Med. 2018, 24, 38. [Google Scholar] [CrossRef]
- Pereira, S.V.-N.; Ribeiro, J.D.; Ribeiro, A.F.; Bertuzzo, C.S.; Marson, F.A.L. Novel, Rare and Common Pathogenic Variants in the CFTR Gene Screened by High-Throughput Sequencing Technology and Predicted by in Silico Tools. Sci. Rep. 2019, 9, 6234. [Google Scholar] [CrossRef]
- Bergougnoux, A.; Deletang, K.; Varilh, J.; Houriez, F.; Altieri, J.-P.; Koenig, M.; Férec, C.; Mireille, C.; Bienvenu, T.; Audrezet, M.-P.; et al. Large Phenotypic Spectrum Associated with Two New Deep Intronic Variants on the CFTR Gene. Eur. Respir. J. 2018, 52, PA3422. [Google Scholar] [CrossRef]
- Lefferts, J.W.; Boersma, V.; Hagemeijer, M.C.; Hajo, K.; Beekman, J.M.; Splinter, E. Targeted Locus Amplification and Haplotyping. In Haplotyping: Methods and Protocols; Peters, B.A., Drmanac, R., Eds.; Methods in Molecular Biology; Springer US: New York, NY, USA, 2023; pp. 31–48. ISBN 978-1-07-162819-5. [Google Scholar]
- Keating, D.; Marigowda, G.; Burr, L.; Daines, C.; Mall, M.A.; McKone, E.F.; Ramsey, B.W.; Rowe, S.M.; Sass, L.A.; Tullis, E.; et al. VX-445-Tezacaftor-Ivacaftor in Patients with Cystic Fibrosis and One or Two Phe508del Alleles. N. Engl. J. Med. 2018, 379, 1612–1620. [Google Scholar] [CrossRef]
- Davies, J.C.; Moskowitz, S.M.; Brown, C.; Horsley, A.; Mall, M.A.; McKone, E.F.; Plant, B.J.; Prais, D.; Ramsey, B.W.; Taylor-Cousar, J.L.; et al. VX-659–Tezacaftor–Ivacaftor in Patients with Cystic Fibrosis and One or Two Phe508del Alleles. N. Engl. J. Med. 2018, 379, 1599–1611. [Google Scholar] [CrossRef]
- Trouvé, P.; Férec, C.; Génin, E. The Interplay between the Unfolded Protein Response, Inflammation and Infection in Cystic Fibrosis. Cells 2021, 10, 2980. [Google Scholar] [CrossRef]
- Chevalier, B.; Hinzpeter, A. The Influence of CFTR Complex Alleles on Precision Therapy of Cystic Fibrosis. J. Cyst. Fibros. 2020, 19, S15–S18. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blotas, C.; Férec, C.; Moisan, S. Tissue-Specific Regulation of CFTR Gene Expression. Int. J. Mol. Sci. 2023, 24, 10678. https://doi.org/10.3390/ijms241310678
Blotas C, Férec C, Moisan S. Tissue-Specific Regulation of CFTR Gene Expression. International Journal of Molecular Sciences. 2023; 24(13):10678. https://doi.org/10.3390/ijms241310678
Chicago/Turabian StyleBlotas, Clara, Claude Férec, and Stéphanie Moisan. 2023. "Tissue-Specific Regulation of CFTR Gene Expression" International Journal of Molecular Sciences 24, no. 13: 10678. https://doi.org/10.3390/ijms241310678
APA StyleBlotas, C., Férec, C., & Moisan, S. (2023). Tissue-Specific Regulation of CFTR Gene Expression. International Journal of Molecular Sciences, 24(13), 10678. https://doi.org/10.3390/ijms241310678







