The Novel-Natural-Killer-Cell-Related Gene Signature Predicts the Prognosis and Immune Status of Patients with Hepatocellular Carcinoma
Abstract
1. Introduction
2. Results
2.1. ScRNA-seq Analysis Identified NK-Cell-Related Genes
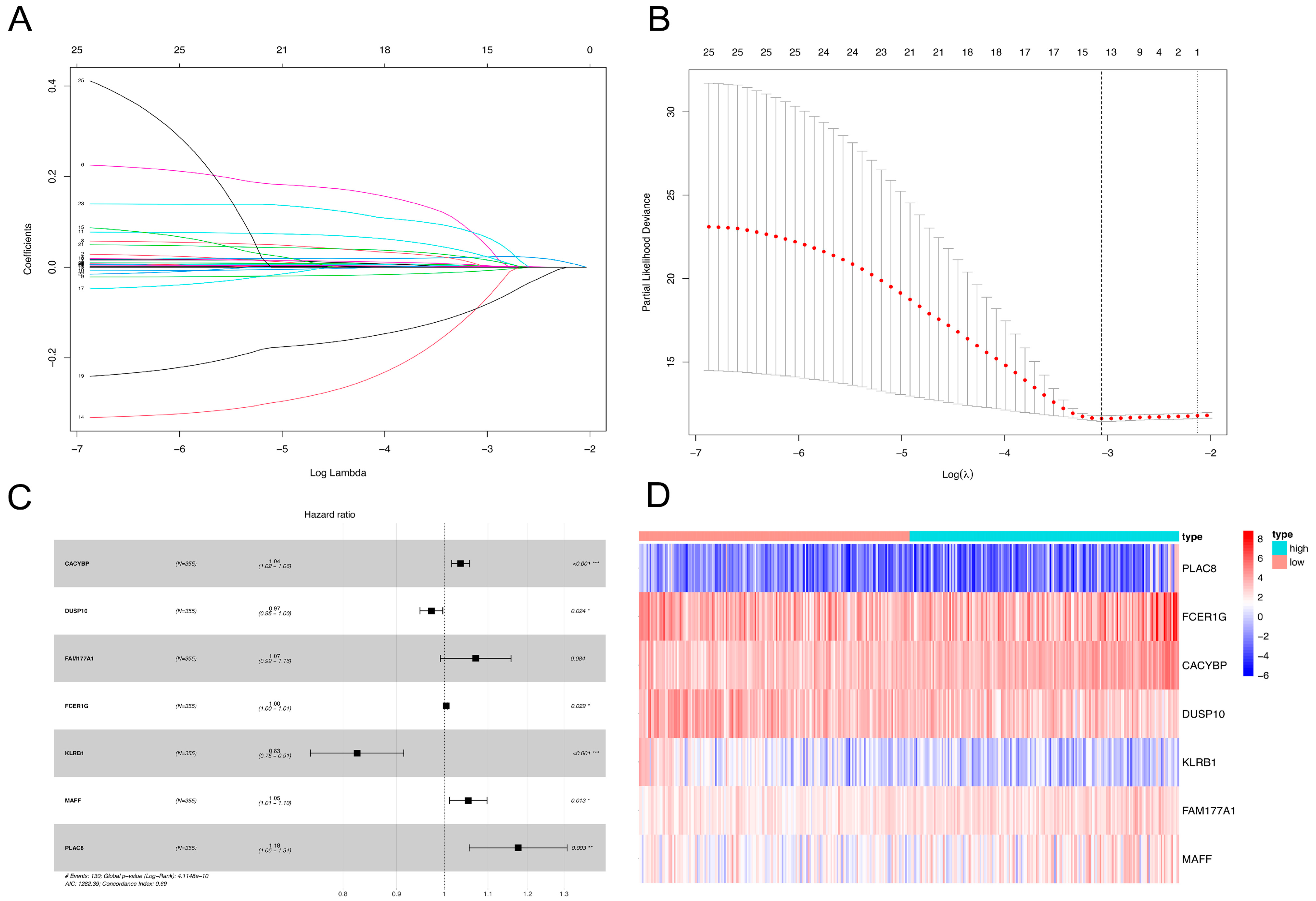
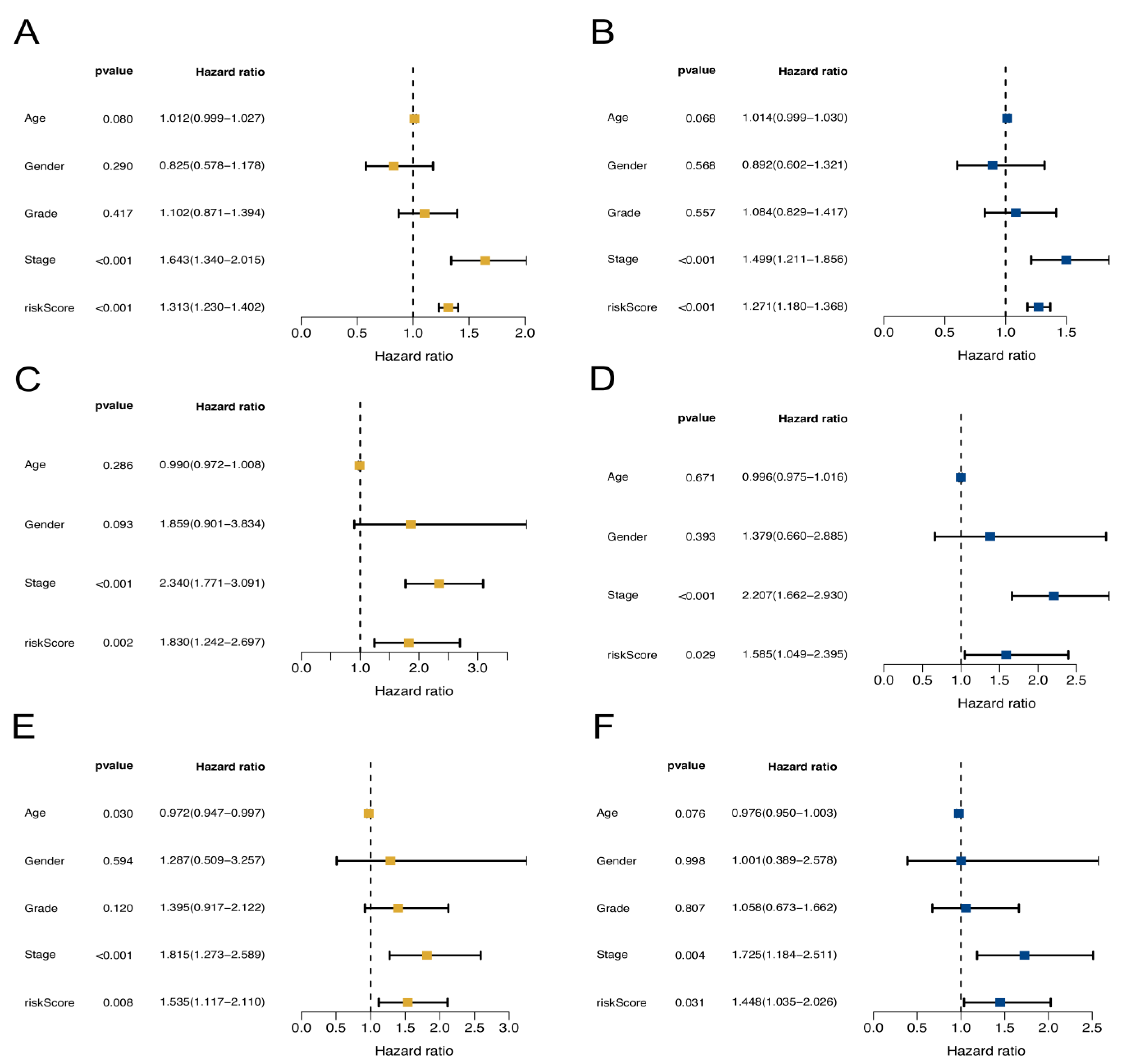
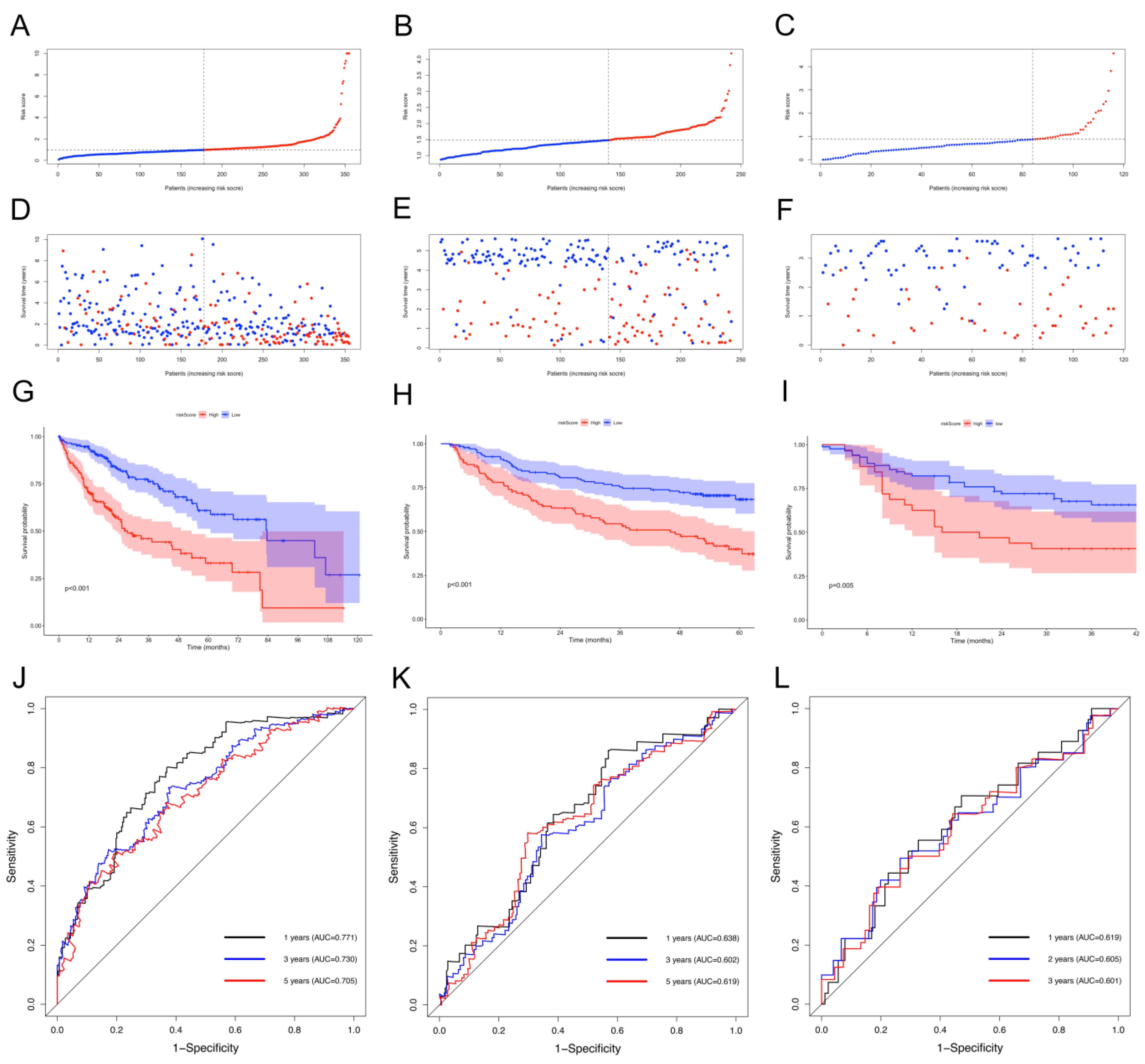
2.2. NKRGS Risk Model Predicted the Prognoses of Patients with HCC
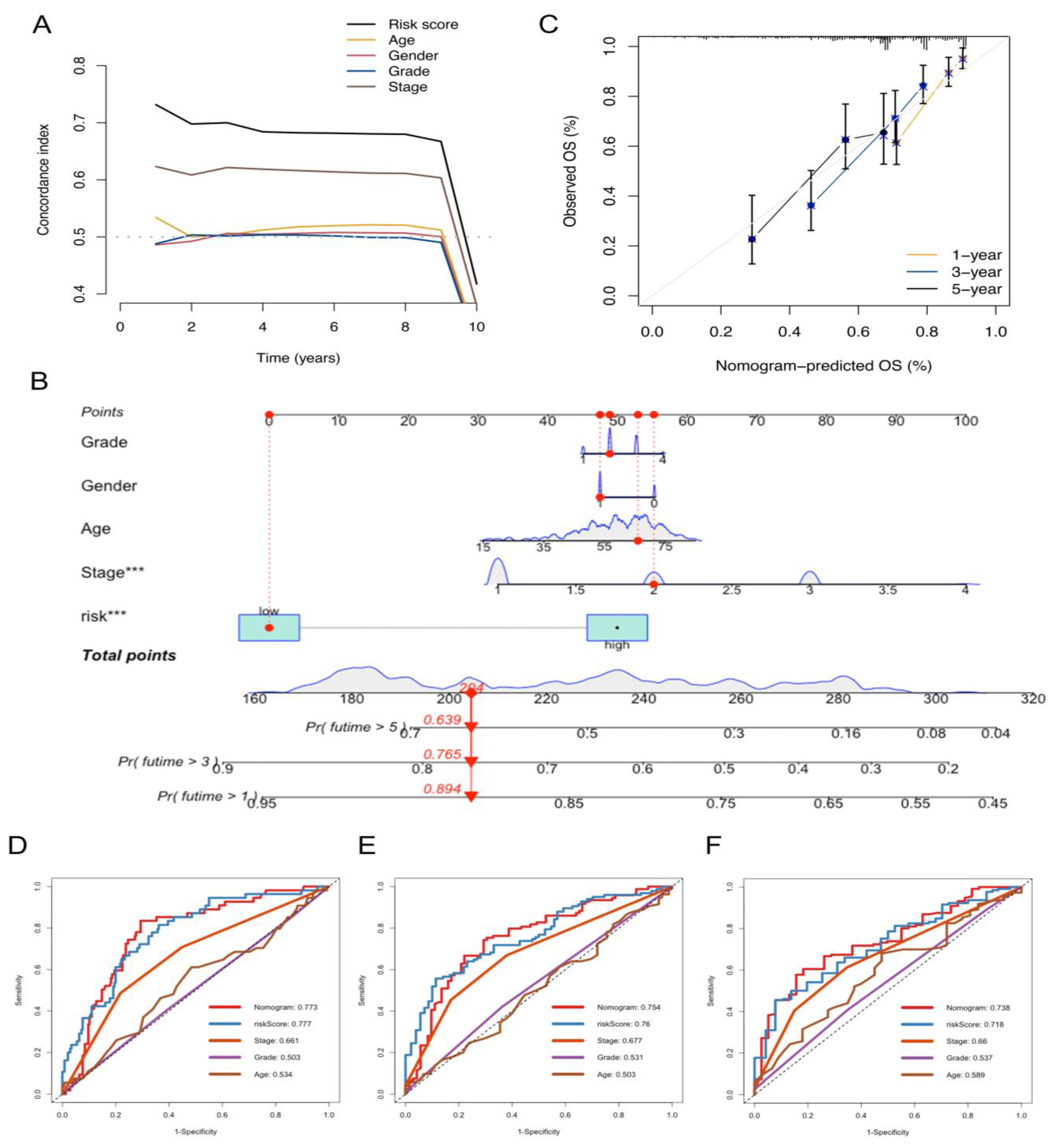
2.3. Development and Validation of the NKRGS-Based Nomogram
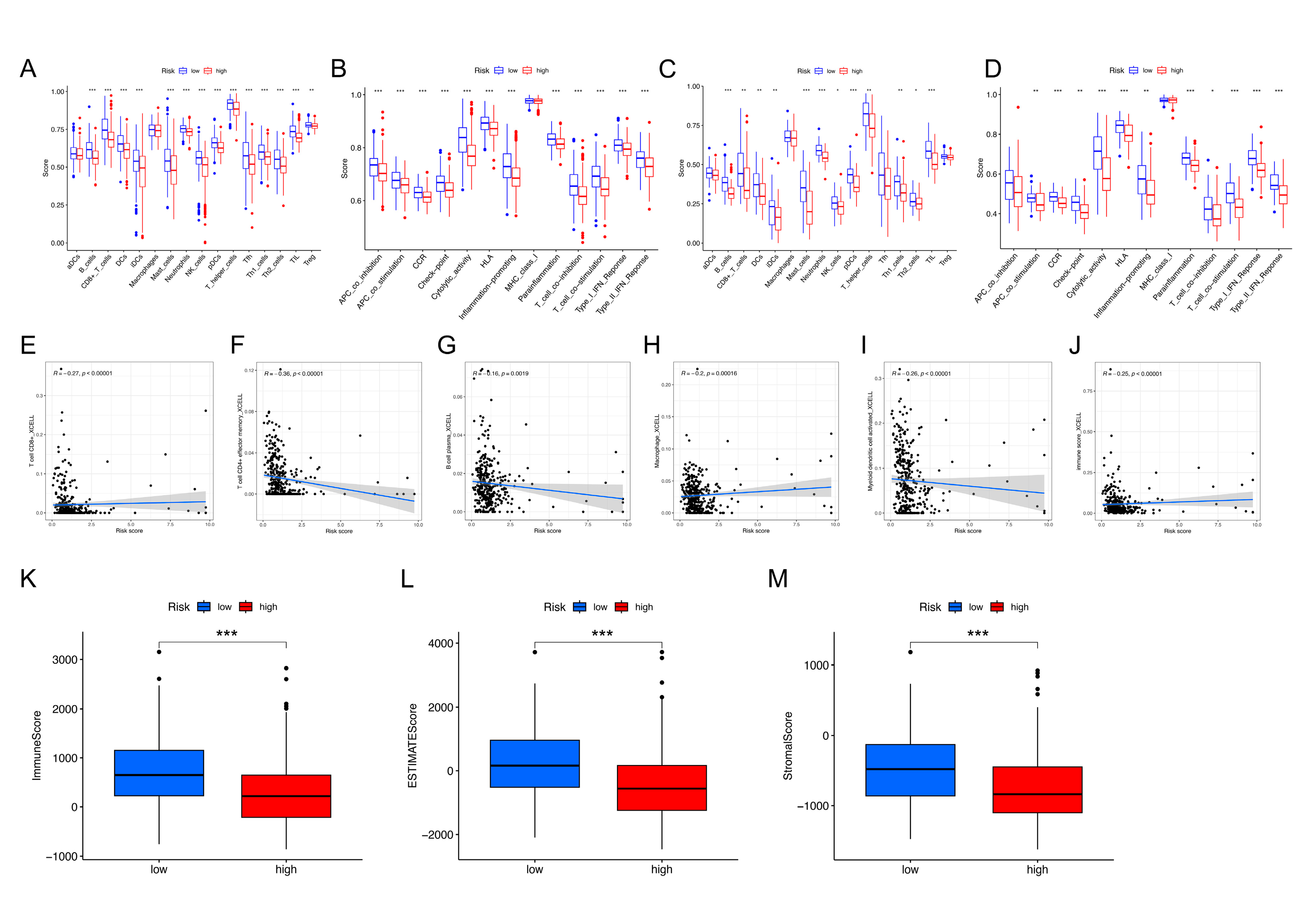
2.4. Comparison of Immune Infiltration Profiles between the NKRGS Risk Groups
2.5. NKRGS-Related Differentially Expressed Genes Correlated with Tumor Immunity and Metabolism
3. Discussion
4. Materials and Methods
4.1. Data Collection and Patient Selection
4.2. Identification of NK-Cell-Related Genes
4.3. Development and Validation of the NKRGS
4.4. Construction of a NKRGS-Based Nomogram
4.5. Immune Cell Infiltration Analysis
4.6. Functional Enrichment Analysis
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Ercolani, G.; Grazi, G.L.; Ravaioli, M.; Del Gaudio, M.; Gardini, A.; Cescon, M. Liver resection for hepatocellular carcinoma on cirrhosis: Univariate and multivariate analysis of risk factors for intrahepatic recurrence. Ann. Surg. 2003, 237, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.C.; Chow, P.K.; Allen, J.C.; Siddiqui, F.J.; Chan, E.S.; Tan, S.B. Systematic review of outcomes of liver resection for early hepatocellular carcinoma within the Milan criteria. Br. J. Surg. 2012, 99, 1622–1629. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Reig, M.; Forner, A.; Rimola, J.; Ferrer-Fabrega, J.; Burrel, M.; Garcia-Criado, A. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J. Hepatol. 2022, 76, 681–693. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Ren, Z.; Xu, J.; Bai, Y.; Xu, A.; Cang, S.; Du, C. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): A randomised, open-label, phase 2–3 study. Lancet Oncol. 2021, 22, 977–990. [Google Scholar] [CrossRef]
- Hinshaw, D.C.; Shevde, L.A. The Tumor Microenvironment Innately Modulates Cancer Progression. Cancer Res. 2019, 79, 4557–4566. [Google Scholar] [CrossRef]
- Sharma, A.; Seow, J.J.W.; Dutertre, C.A.; Pai, R.; Bleriot, C.; Mishra, A. Onco-fetal Reprogramming of Endothelial Cells Drives Immunosuppressive Macrophages in Hepatocellular Carcinoma. Cell 2020, 183, 377–394.e21. [Google Scholar] [CrossRef]
- Sun, Y.; Wu, L.; Zhong, Y.; Zhou, K.; Hou, Y.; Wang, Z. Single-cell landscape of the ecosystem in early-relapse hepatocellular carcinoma. Cell 2021, 184, 404–421.e16. [Google Scholar] [CrossRef]
- Zheng, C.; Zheng, L.; Yoo, J.K.; Guo, H.; Zhang, Y.; Guo, X. Landscape of Infiltrating T Cells in Liver Cancer Revealed by Single-Cell Sequencing. Cell 2017, 169, 1342–1356.e16. [Google Scholar] [CrossRef]
- Chew, V.; Chen, J.; Lee, D.; Loh, E.; Lee, J.; Lim, K.H. Chemokine-driven lymphocyte infiltration: An early intratumoural event determining long-term survival in resectable hepatocellular carcinoma. Gut 2012, 61, 427–438. [Google Scholar] [CrossRef]
- Wu, M.; Mei, F.; Liu, W.; Jiang, J. Comprehensive characterization of tumor infiltrating natural killer cells and clinical significance in hepatocellular carcinoma based on gene expression profiles. Biomed. Pharmacother. 2020, 121, 109637. [Google Scholar] [CrossRef] [PubMed]
- Crouse, J.; Xu, H.C.; Lang, P.A.; Oxenius, A. NK cells regulating T cell responses: Mechanisms and outcome. Trends Immunol. 2015, 36, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Mocikat, R.; Braumuller, H.; Gumy, A.; Egeter, O.; Ziegler, H.; Reusch, U. Natural killer cells activated by MHC class I(low) targets prime dendritic cells to induce protective CD8 T cell responses. Immunity 2003, 19, 561–569. [Google Scholar] [CrossRef]
- Pinyol, R.; Montal, R.; Bassaganyas, L.; Sia, D.; Takayama, T.; Chau, G.Y. Molecular predictors of prevention of recurrence in HCC with sorafenib as adjuvant treatment and prognostic factors in the phase 3 STORM trial. Gut 2019, 68, 1065–1075. [Google Scholar] [CrossRef] [PubMed]
- Sajid, M.; Liu, L.; Sun, C. The Dynamic Role of NK Cells in Liver Cancers: Role in HCC and HBV Associated HCC and Its Therapeutic Implications. Front. Immunol. 2022, 13, 887186. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, K.; Shahmoradgoli, M.; Martinez, E.; Vegesna, R.; Kim, H.; Torres-Garcia, W. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat. Commun. 2013, 4, 2612. [Google Scholar] [CrossRef]
- Lan, X.B.; Papatheodoridis, G.; Teng, Y.X.; Zhong, J.H. The upward trend in the immunotherapy utilization for hepatobiliary cancers. Hepatobiliary Surg. Nutr. 2021, 10, 692–695. [Google Scholar] [CrossRef] [PubMed]
- Pfister, D.; Nunez, N.G.; Pinyol, R.; Govaere, O.; Pinter, M.; Szydlowska, M. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature 2021, 592, 450–456. [Google Scholar] [CrossRef]
- Shimasaki, N.; Jain, A.; Campana, D. NK cells for cancer immunotherapy. Nat. Rev. Drug. Discov. 2020, 19, 200–218. [Google Scholar] [CrossRef]
- Chen, X.; Han, G.; Zhai, H.; Zhang, F.; Wang, J.; Li, X. Expression and clinical significance of CacyBP/SIP in pancreatic cancer. Pancreatology 2008, 8, 470–477. [Google Scholar] [CrossRef]
- Wang, N.; Ma, Q.; Wang, Y.; Ma, G.; Zhai, H. CacyBP/SIP expression is involved in the clinical progression of breast cancer. World J. Surg. 2010, 34, 2545–2552. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Martinez, M.; Stamatakis, K.; Fresno, M. The Dual-Specificity Phosphatase 10 (DUSP10): Its Role in Cancer, Inflammation, and Immunity. Int. J. Mol. Sci. 2019, 20, 1626. [Google Scholar] [CrossRef]
- He, G.; Zhang, L.; Li, Q.; Yang, L. miR-92a/DUSP10/JNK signalling axis promotes human pancreatic cancer cells proliferation. Biomed. Pharmacother. 2014, 68, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Png, C.W.; Weerasooriya, M.; Guo, J.; James, S.J.; Poh, H.M.; Osato, M. DUSP10 regulates intestinal epithelial cell growth and colorectal tumorigenesis. Oncogene 2016, 35, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Chen, W.; Pan, X.; Wang, H.; Sun, Y.; Qian, C. FCER1G positively relates to macrophage infiltration in clear cell renal cell carcinoma and contributes to unfavorable prognosis by regulating tumor immunity. BMC Cancer 2022, 22, 140. [Google Scholar] [CrossRef]
- Kuster, H.; Thompson, H.; Kinet, J.P. Characterization and expression of the gene for the human Fc receptor gamma subunit. Definition of a new gene family. J. Biol. Chem. 1990, 265, 6448–6452. [Google Scholar] [CrossRef]
- Zhang, X.; Cai, J.; Song, F.; Yang, Z. Prognostic and immunological role of FCER1G in pan-cancer. Pathol. Res. Pract. 2022, 240, 154174. [Google Scholar] [CrossRef]
- Yang, R.; Chen, Z.; Liang, L.; Ao, S.; Zhang, J.; Chang, Z. Fc Fragment of IgE Receptor Ig (FCER1G) acts as a key gene involved in cancer immune infiltration and tumour microenvironment. Immunology 2023, 168, 302–319. [Google Scholar] [CrossRef] [PubMed]
- Gentles, A.J.; Newman, A.M.; Liu, C.L.; Bratman, S.V.; Feng, W.; Kim, D. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 2015, 21, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Du, J.; Liu, C.; Zeng, H.; Chen, Y.; Liu, L. A Pan-Cancer Analysis of CD161, a Potential New Immune Checkpoint. Front. Immunol. 2021, 12, 688215. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Lu, T.; Huang, L.; Wang, Z.; Yan, Z.; Guan, Y. Circular RNA cia-MAF drives self-renewal and metastasis of liver tumor-initiating cells via transcription factor MAFF. J. Clin. Investig. 2021, 131, e148020. [Google Scholar] [CrossRef]
- Moon, E.J.; Mello, S.S.; Li, C.G.; Chi, J.T.; Thakkar, K.; Kirkland, J.G. The HIF target MAFF promotes tumor invasion and metastasis through IL11 and STAT3 signaling. Nat. Commun. 2021, 12, 4308. [Google Scholar] [CrossRef]
- Chen, W.; Wu, J.; Wang, W.; Yu, L.; Xu, X. PLAC8 Overexpression Promotes Lung Cancer Cell Growth via Wnt/beta-Catenin Signaling. J. Immunol. Res. 2022, 2022, 8854196. [Google Scholar] [CrossRef]
- Kaistha, B.P.; Lorenz, H.; Schmidt, H.; Sipos, B.; Pawlak, M.; Gierke, B. PLAC8 Localizes to the Inner Plasma Membrane of Pancreatic Cancer Cells and Regulates Cell Growth and Disease Progression through Critical Cell-Cycle Regulatory Pathways. Cancer Res. 2016, 76, 96–107. [Google Scholar] [CrossRef]
- Cong, J.; Wang, X.; Zheng, X.; Wang, D.; Fu, B.; Sun, R. Dysfunction of Natural Killer Cells by FBP1-Induced Inhibition of Glycolysis during Lung Cancer Progression. Cell Metab. 2018, 28, 243–255.e5. [Google Scholar] [CrossRef]
- O’Brien, K.L.; Finlay, D.K. Immunometabolism and natural killer cell responses. Nat. Rev. Immunol. 2019, 19, 282–290. [Google Scholar] [CrossRef]
- Brand, A.; Singer, K.; Koehl, G.E.; Kolitzus, M.; Schoenhammer, G.; Thiel, A. LDHA-Associated Lactic Acid Production Blunts Tumor Immunosurveillance by T and NK Cells. Cell Metab. 2016, 24, 657–671. [Google Scholar] [CrossRef]
- Chang, C.H.; Qiu, J.; O’Sullivan, D.; Buck, M.D.; Noguchi, T.; Curtis, J.D. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell 2015, 162, 1229–1241. [Google Scholar] [CrossRef] [PubMed]
- Harmon, C.; Robinson, M.W.; Hand, F.; Almuaili, D.; Mentor, K.; Houlihan, D.D. Lactate-Mediated Acidification of Tumor Microenvironment Induces Apoptosis of Liver-Resident NK Cells in Colorectal Liver Metastasis. Cancer Immunol. Res. 2019, 7, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.L.; Qiu, X.; Lin, J.Y.; Chen, X.Y.; Zhang, Y.M.; Hu, X.Y. Potential Role and Clinical Value of PPP2CA in Hepatocellular Carcinoma. J. Clin. Transl. Hepatol. 2021, 9, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Tibshirani, R. The lasso method for variable selection in the Cox model. Stat. Med. 1997, 16, 385–395. [Google Scholar] [CrossRef]
- Heagerty, P.J.; Lumley, T.; Pepe, M.S. Time-dependent ROC curves for censored survival data and a diagnostic marker. Biometrics 2000, 56, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Aran, D.; Hu, Z.; Butte, A.J. xCell: Digitally portraying the tissue cellular heterogeneity landscape. Genome Biol. 2017, 18, 220. [Google Scholar] [CrossRef]


| Variable | TCGA Cohort n = 355 | Internal Cohort n = 116 | GSE14520 Cohort n = 242 |
|---|---|---|---|
| Age, years | |||
| ≤60 | 169 | 101 | 196 |
| >60 | 186 | 25 | 46 |
| Sex | |||
| Male | 240 | 101 | 211 |
| Female | 115 | 15 | 31 |
| Grade | |||
| I–II | 222 | 59 | NA |
| III–IV | 128 | 57 | NA |
| Unknown | 5 | 0 | NA |
| Alpha-fetoprotein, ng/mL | |||
| ≤400 | 207 | 45 | NA |
| >400 | 62 | 71 | NA |
| Unknown | 86 | 0 | NA |
| TNM stage | |||
| I–II | 246 | NA | 174 |
| III–IV | 85 | NA | 51 |
| Unknown | 24 | NA | 17 |
| BCLC stage | |||
| 0–A | NA | 58 | 172 |
| B | NA | 32 | 24 |
| C | NA | 26 | 29 |
| Unknown | NA | 0 | 17 |
| Vascular invasion | |||
| Micro | 86 | 50 | NA |
| Macro | 16 | 23 | NA |
| None | 199 | 43 | NA |
| Unknown | 54 | 0 | NA |
| HBV/HCV infection | |||
| Yes | NA | 99 | 218 |
| No | NA | 17 | 6 |
| Unknown | NA | 0 | 18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Huang, J.; Zhan, G.; Li, Y.; Fang, C.; Xiang, B. The Novel-Natural-Killer-Cell-Related Gene Signature Predicts the Prognosis and Immune Status of Patients with Hepatocellular Carcinoma. Int. J. Mol. Sci. 2023, 24, 9587. https://doi.org/10.3390/ijms24119587
Li M, Huang J, Zhan G, Li Y, Fang C, Xiang B. The Novel-Natural-Killer-Cell-Related Gene Signature Predicts the Prognosis and Immune Status of Patients with Hepatocellular Carcinoma. International Journal of Molecular Sciences. 2023; 24(11):9587. https://doi.org/10.3390/ijms24119587
Chicago/Turabian StyleLi, Minjun, Juntao Huang, Guohua Zhan, Yuankuan Li, Chunye Fang, and Bangde Xiang. 2023. "The Novel-Natural-Killer-Cell-Related Gene Signature Predicts the Prognosis and Immune Status of Patients with Hepatocellular Carcinoma" International Journal of Molecular Sciences 24, no. 11: 9587. https://doi.org/10.3390/ijms24119587
APA StyleLi, M., Huang, J., Zhan, G., Li, Y., Fang, C., & Xiang, B. (2023). The Novel-Natural-Killer-Cell-Related Gene Signature Predicts the Prognosis and Immune Status of Patients with Hepatocellular Carcinoma. International Journal of Molecular Sciences, 24(11), 9587. https://doi.org/10.3390/ijms24119587





