The Effect of Respiratory Viral Infections on Breakthrough Hemolysis in Patients with Paroxysmal Nocturnal Hemoglobinuria
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
2.2. Infection Parameters
2.3. Hemolytic Parameters
3. Discussion
4. Materials and Methods
Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hillmen, P.; Lewis, S.; Bessler, M.; Luzzatto, L.; Dacie, J.V. Natural History of Paroxysmal Nocturnal Hemoglobinuria. N. Engl. J. Med. 1995, 333, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Takeda, J.; Miyata, T.; Kawagoe, K.; Iida, Y.; Endo, Y.; Fujita, T.; Takahashi, M.; Kitani, T.; Kinoshita, T. Deficiency of the GPI anchor caused by a somatic mutation of the PIG-A gene in paroxysmal nocturnal hemoglobinuria. Cell 1993, 73, 703–711. [Google Scholar] [CrossRef]
- Boccuni, P.; Del Vecchio, L.; Di Noto, R.; Rotoli, B. Glycosyl phosphatidylinositol (GPI)-anchored molecules and the pathogenesis of paroxysmal nocturnal hemoglobinuria. Crit. Rev. Oncol. 2000, 33, 25–43. [Google Scholar] [CrossRef] [PubMed]
- Notaro, R.; Sica, M. C3-mediated extravascular hemolysis in PNH on eculizumab: Mechanism and clinical implications. Semin. Hematol. 2018, 55, 130–135. [Google Scholar] [CrossRef]
- Brodsky, R.A. Paroxysmal nocturnal hemoglobinuria. Blood 2014, 124, 2804–2811. [Google Scholar] [CrossRef]
- Kinoshita, T.; Medof, M.E.; Silber, R.; Nussenzweig, V. Distribution of decay-accelerating factor in the peripheral blood of normal individuals and patients with paroxysmal nocturnal hemoglobinuria. J. Exp. Med. 1985, 162, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.J.; Norfolk, D.R.; Swirsky, D.M.; Hillmen, P. Lymphocyte subset analysis and glycosylphosphatidylinositol phenotype in patients with paroxysmal nocturnal hemoglobinuria. Blood 1998, 92, 1799–1806. [Google Scholar] [CrossRef]
- Devalet, B.; Mullier, F.; Chatelain, B.; Dogné, J.-M.; Chatelain, C. Pathophysiology, diagnosis, and treatment of paroxysmal nocturnal hemoglobinuria: A review. Eur. J. Haematol. 2015, 95, 190–198. [Google Scholar] [CrossRef]
- Hillmen, P.; Hall, C.; Marsh, J.C.; Elebute, M.; Bombara, M.P.; Petro, B.E.; Cullen, M.J.; Richards, S.J.; Rollins, S.A.; Mojcik, C.F.; et al. Effect of eculizumab on hemolysis and transfusion requirements in patients with paroxysmal nocturnal hemoglobinuria. N. Engl. J. Med. 2004, 350, 552–559. [Google Scholar] [CrossRef]
- Sakai, K. Even mild hemolysis in paroxysmal nocturnal hemoglobinuria could severely compromise the quality of life due to long-term sustained intolerant fatigue. Leuk. Res. Rep. 2020, 14, 100224. [Google Scholar] [CrossRef]
- Socié, G.; Schrezenmeier, H.; Muus, P.; Lisukov, I.; Roeth, A.; Kulasekararaj, A.; Lee, J.W.; Araten, D.; Hill, A.; Brodsky, R.; et al. Changing prognosis in paroxysmal nocturnal haemoglobinuria disease subcategories: An analysis of the International PNH Registry. Intern. Med. J. 2016, 46, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.; Ellis, S.; Konia, T.H.; Brassard, A.; Tartar, D. Extensive purpura as presenting sign of parvovirus B19 infection in a patient with paroxysmal nocturnal hemoglobinuria. JAAD Case Rep. 2019, 5, 798–800. [Google Scholar] [CrossRef] [PubMed]
- Fattizzo, B.; Pasquale, R.; Bellani, V.; Barcellini, W.; Kulasekararaj, A.G. Complement Mediated Hemolytic Anemias in the COVID-19 Era: Case Series and Review of the Literature. Front. Immunol. 2021, 12, 791429. [Google Scholar] [CrossRef] [PubMed]
- Kulasekararaj, A.G.; Lazana, I.; Large, J.; Posadas, K.; Eagleton, H.; Villajin, J.L.; Zuckerman, M.; Gandhi, S.; Marsh, J.C.W. Terminal complement inhibition dampens the inflammation during COVID-19. Br. J. Haematol. 2020, 190, e141–e143. [Google Scholar] [CrossRef] [PubMed]
- Sahin, F.; Yilmaz, A.F.; Ozkan, M.C.; Gokmen, N.M.; Saydam, G. PNH is a debilitating, fatal but treatable disease: Same disease, different clinical presentations. Am. J. Blood. Res. 2015, 5, 30–33. [Google Scholar]
- Lee, J.W.; Jang, J.H.; Kim, J.S.; Yoon, S.S.; Lee, J.H.; Kim, Y.K.; Jo, D.Y.; Chung, J.; Sohn, S.K. Clinical signs and symptoms associated with increased risk for thrombosis in patients with paroxysmal nocturnal hemoglobinuria from a Korean Registry. Int. J. Hematol. 2013, 97, 749–757. [Google Scholar] [CrossRef]
- Rother, R.P.; Bell, L.; Hillmen, P.; Gladwin, M.T. The clinical sequelae of intravascular hemolysis and extracellular plasma hemoglobin: A novel mechanism of human disease. JAMA 2005, 293, 1653–1662. [Google Scholar] [CrossRef]
- Risitano, A.M.; Notaro, R.; Luzzatto, L.; Hill, A.; Kelly, R.; Hillmen, P. Paroxysmal nocturnal hemoglobinuria—Hemolysis before and after eculizumab. N. Engl. J. Med. 2010, 363, 2270–2272. [Google Scholar] [CrossRef]
- Abe, R.; Shichishima, T.; Ogawa, K.; Saitoh, Y.; Maruyama, Y. Neutropenia due to parvovirus B19 infections in patients with paroxysmal nocturnal hemoglobinuria: Blood transfusion and natural infection cases. Acta. Haematol. 2006, 116, 245–248. [Google Scholar] [CrossRef]
- Kurita, N.; Obara, N.; Fukuda, K.; Nishikii, H.; Sato, S.; Inagawa, S.; Kurokawa, T.; Owada, Y.; Ninomiya, H.; Chiba, S. Perisurgical induction of eculizumab in a patient with paroxysmal nocturnal hemoglobinuria: Its inhibition of surgery-triggered hemolysis and the consequence of subsequent discontinuation. Blood Coagul. Fibrinolysis 2013, 24, 658–662. [Google Scholar] [CrossRef]
- Nakayama, H.; Usuki, K.; Echizen, H.; Ogawa, R.; Orii, T. Eculizumab Dosing Intervals Longer than 17 Days May Be Associated with Greater Risk of Breakthrough Hemolysis in Patients with Paroxysmal Nocturnal Hemoglobinuria. Biol. Pharm. Bull. 2016, 39, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Notaro, R.; Luzzatto, L. Breakthrough Hemolysis in PNH with Proximal or Terminal Complement Inhibition. N. Engl. J. Med. 2022, 387, 160–166. [Google Scholar] [CrossRef] [PubMed]
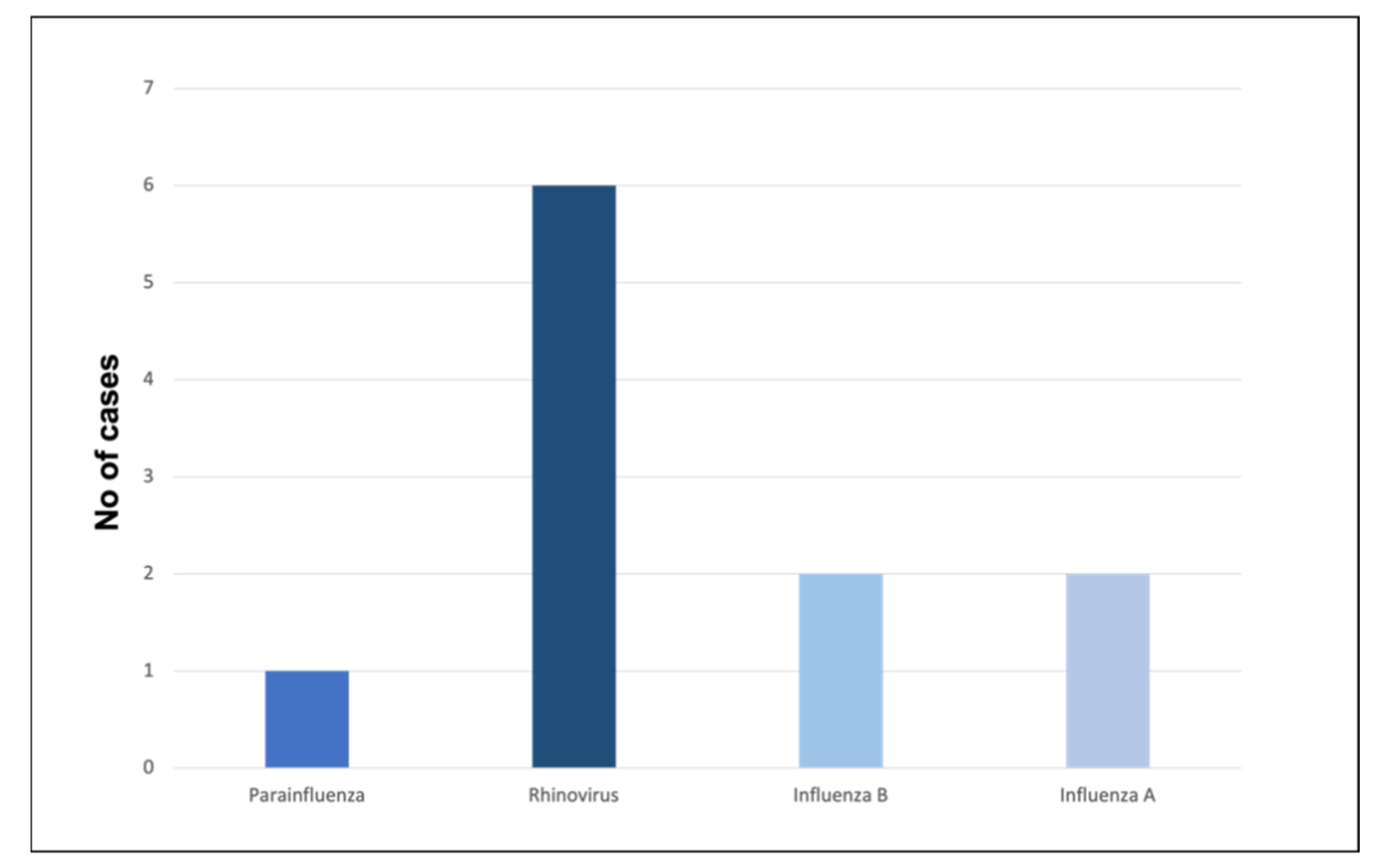
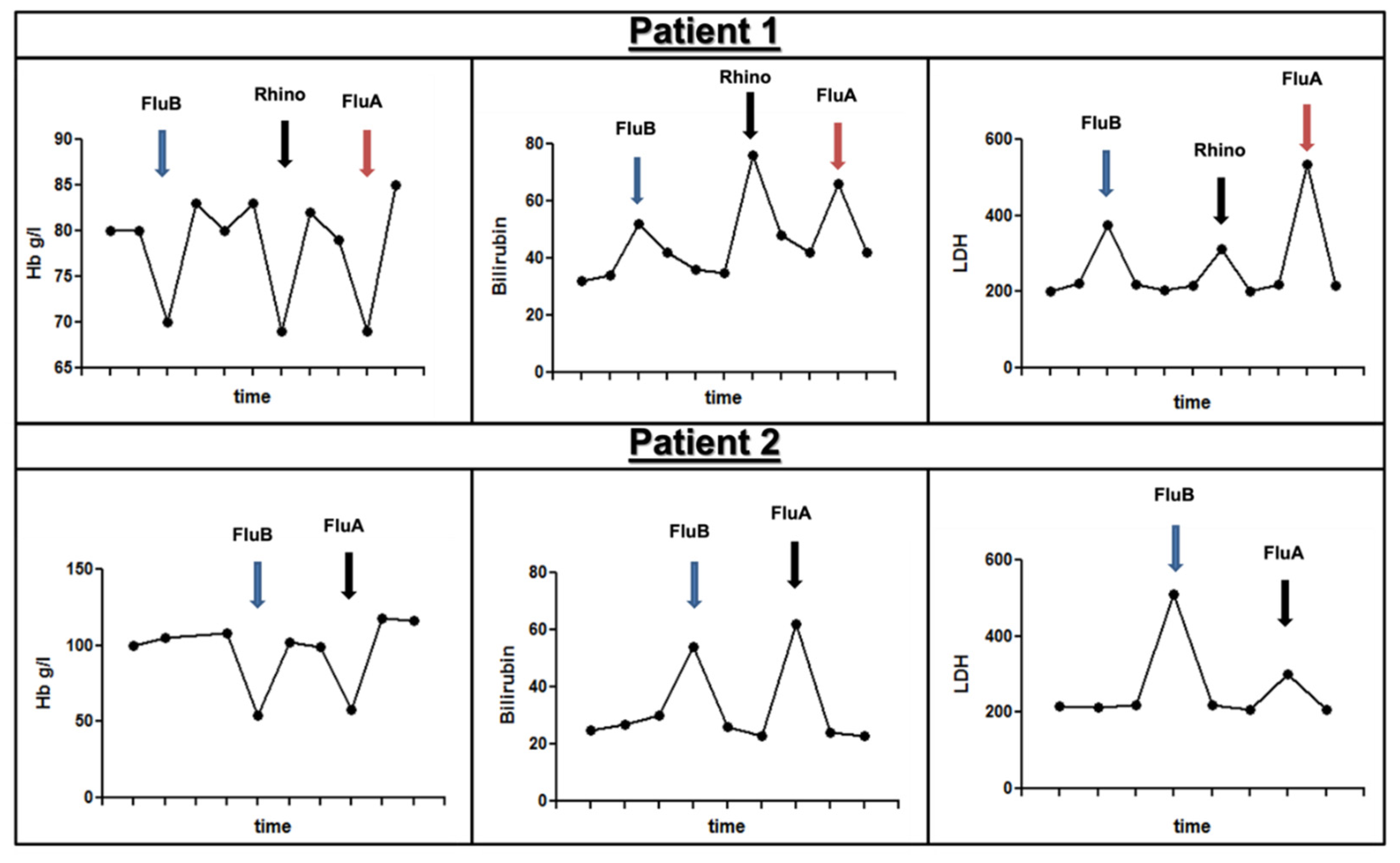
| Characteristics | NTS+ | NTS− | p-Value |
|---|---|---|---|
| Number of patients | 11 | 23 | |
| Age, years (median) | 49.5 | 51 | |
| Sex | |||
| Male | 7 | 6 | |
| Female | 4 | 17 | |
| Classification of PNH | |||
| Classical | 7 | 19 | |
| Associated with other BM disorder | 4 | 4 | |
| Indication for Eculizumab Rx | |||
| Hemolysis | 7 | 22 | |
| Thrombosis | 3 | 0 | |
| Other (pregnancy) | 1 | 1 | |
| Eculizumab duration of Rx months, mean (SEM) | 83.1 (16.78) | 52.7 (6.28) | 0.0007 |
| Antibiotic prophylaxis | |||
| Yes | 10 | 20 | |
| No | 1 | 3 | |
| On Cyclosporin | |||
| No | 11 | 21 | |
| Yes | 0 | 2 | |
| Transfusion Dependent | 0.07 | ||
| Yes | 5 | 4 | |
| No | 6 | 19 |
| Characteristics | NTS+ | NTS− | p-Value |
|---|---|---|---|
| CRP rise | |||
| Yes | 6 | 8 | |
| No | 5 | 15 | |
| Percentage increase in CRP (mean, SEM) | 34.5 (9.7) | 12.2 (3.4) | 0.04 |
| WCC rise | |||
| Yes | 8 | 6 | |
| No | 3 * | 17 | |
| Percentage increase in WCC (mean, SEM) | 33.8 (7.1) | 11.1 (4.7) | 0.01 |
| Neutrophil rise | 8 | 6 | |
| Percentage increase in neutrophil count (mean, SEM) | 42.8 (14.1) | 12.7 (5.5) | 0.04 |
| Neutrophil drop | 3 | 8 | |
| Percentage drop in neutrophil count (mean, SEM) | 39.07 (24.37) | 20.05 (3.06) | 0.22 |
| Hospitalization | |||
| Yes | 3 | 2 | |
| No | 8 | 21 | |
| Need for Antibiotics | |||
| Yes | 6 | 5 | |
| No | 5 | 18 | |
| Need for Oseltamivir | |||
| Yes | 4 | 0 | |
| No | 7 | 23 | |
| Need for GCSF | |||
| Yes | 1 | 0 | |
| No | 10 | 23 |
| Characteristic | NTS+ | NTS− | p-Value |
|---|---|---|---|
| LDH increase | |||
| Yes | 7 | 13 | |
| No | 4 | 10 | |
| Percentage increase in LDH (mean, SEM) | 87.9 (28.4) | 51.6 (14.5) | 0.2 |
| Bilirubin | |||
| Yes | 6 | 2 | |
| No | 5 | 21 | |
| Percentage increase in bilirubin (mean, SEM) | 66.2 (13.7) | 22.6 (3.0) | 0.02 |
| Hemoglobin drop | |||
| Yes | 8 | 8 | |
| No | 3 | 15 | |
| Percentage drop in hemoglobin (mean, SEM) | 25.4 (4.5) | 4.7 (1.6) | 0.002 |
| Need for RBC transfusion | |||
| Yes | 3 | 0 | |
| No | 8 | 23 | |
| Extra eculizumab dose | |||
| Yes | 2 | 0 | |
| No | 9 | 23 | |
| Time from last eculizumab dose (days) at NPA testing (mean, SEM) | 11.2 (5.2) | 2.8 (1.2) | 0.53 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazana, I.; Apap Mangion, S.; Babiker, S.; Large, J.; Trikha, R.; Zuckerman, M.; Gandhi, S.; Kulasekararaj, A.G. The Effect of Respiratory Viral Infections on Breakthrough Hemolysis in Patients with Paroxysmal Nocturnal Hemoglobinuria. Int. J. Mol. Sci. 2023, 24, 9358. https://doi.org/10.3390/ijms24119358
Lazana I, Apap Mangion S, Babiker S, Large J, Trikha R, Zuckerman M, Gandhi S, Kulasekararaj AG. The Effect of Respiratory Viral Infections on Breakthrough Hemolysis in Patients with Paroxysmal Nocturnal Hemoglobinuria. International Journal of Molecular Sciences. 2023; 24(11):9358. https://doi.org/10.3390/ijms24119358
Chicago/Turabian StyleLazana, Ioanna, Sean Apap Mangion, Selma Babiker, Joanna Large, Roochi Trikha, Mark Zuckerman, Shreyans Gandhi, and Austin G. Kulasekararaj. 2023. "The Effect of Respiratory Viral Infections on Breakthrough Hemolysis in Patients with Paroxysmal Nocturnal Hemoglobinuria" International Journal of Molecular Sciences 24, no. 11: 9358. https://doi.org/10.3390/ijms24119358
APA StyleLazana, I., Apap Mangion, S., Babiker, S., Large, J., Trikha, R., Zuckerman, M., Gandhi, S., & Kulasekararaj, A. G. (2023). The Effect of Respiratory Viral Infections on Breakthrough Hemolysis in Patients with Paroxysmal Nocturnal Hemoglobinuria. International Journal of Molecular Sciences, 24(11), 9358. https://doi.org/10.3390/ijms24119358






