Effects of High Irradiance and Low Water Temperature on Photoinhibition and Repair of Photosystems in Marimo (Aegagropila linnaei) in Lake Akan, Japan
Abstract
1. Introduction
2. Results
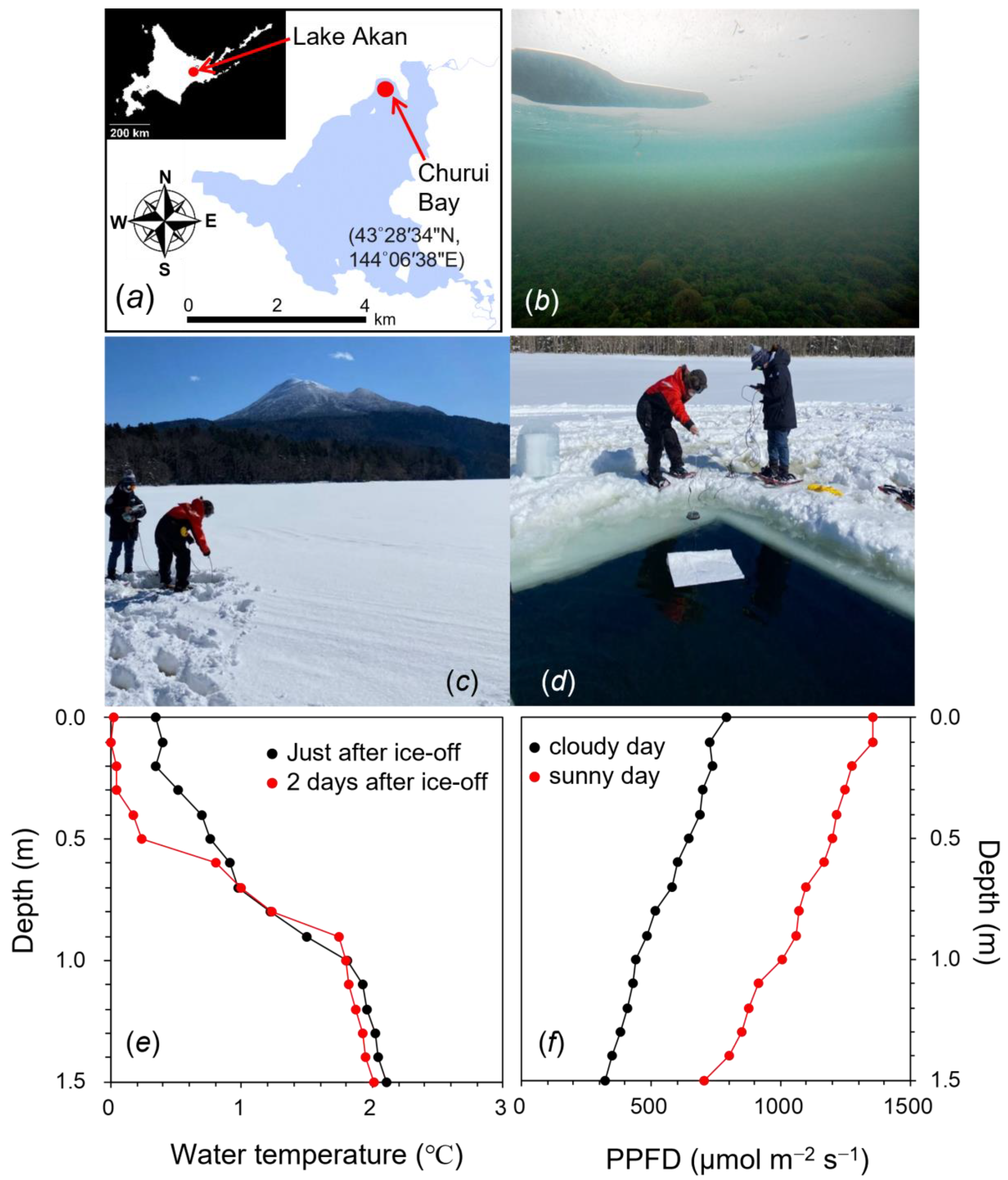
2.1. Environmental Conditions at the Sampling Site of Churui Bay in Lake Akan
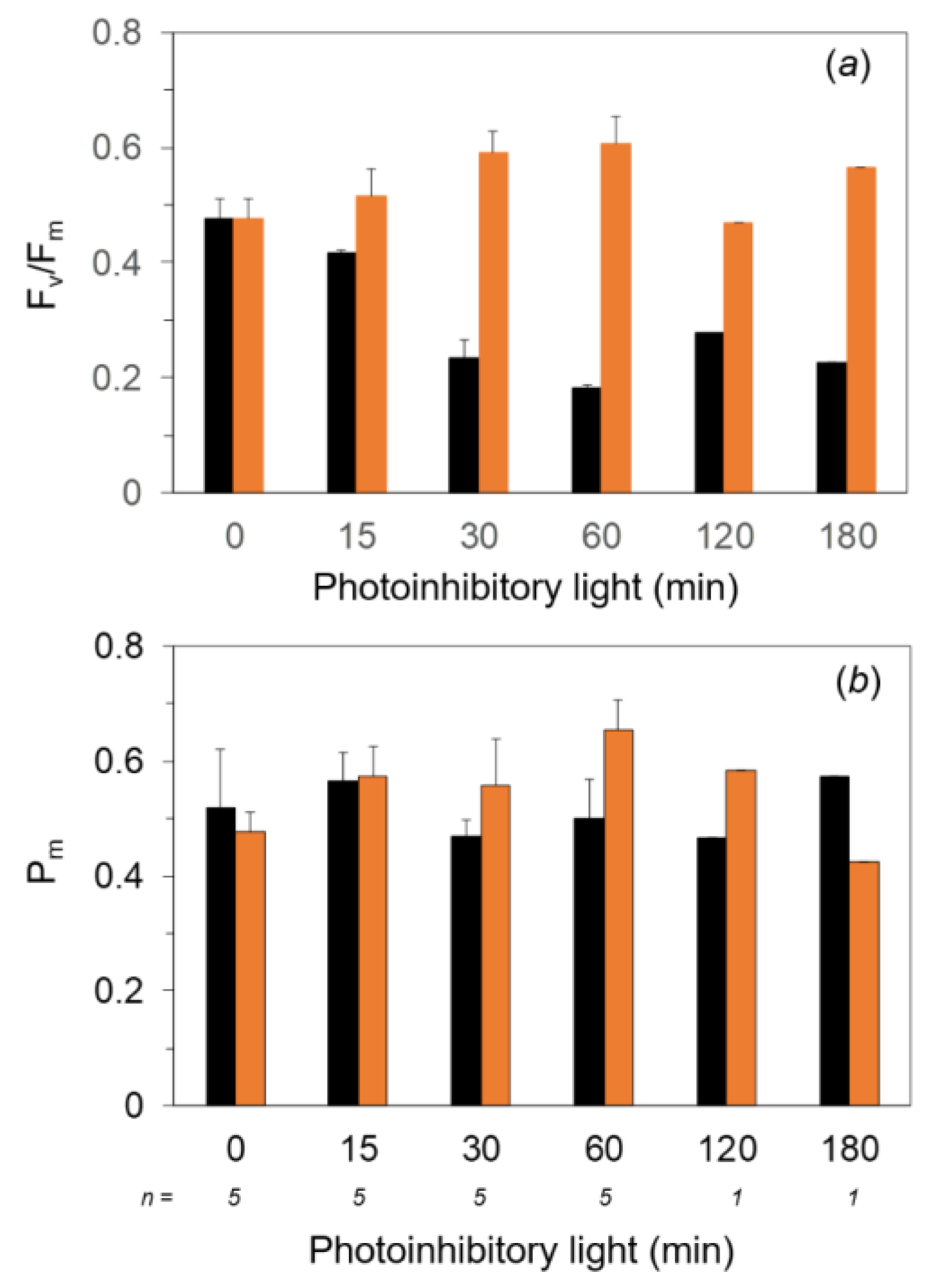
2.2. Measurements of Fv/Fm and Pm against the Exposure to Light-Chilling Treatments
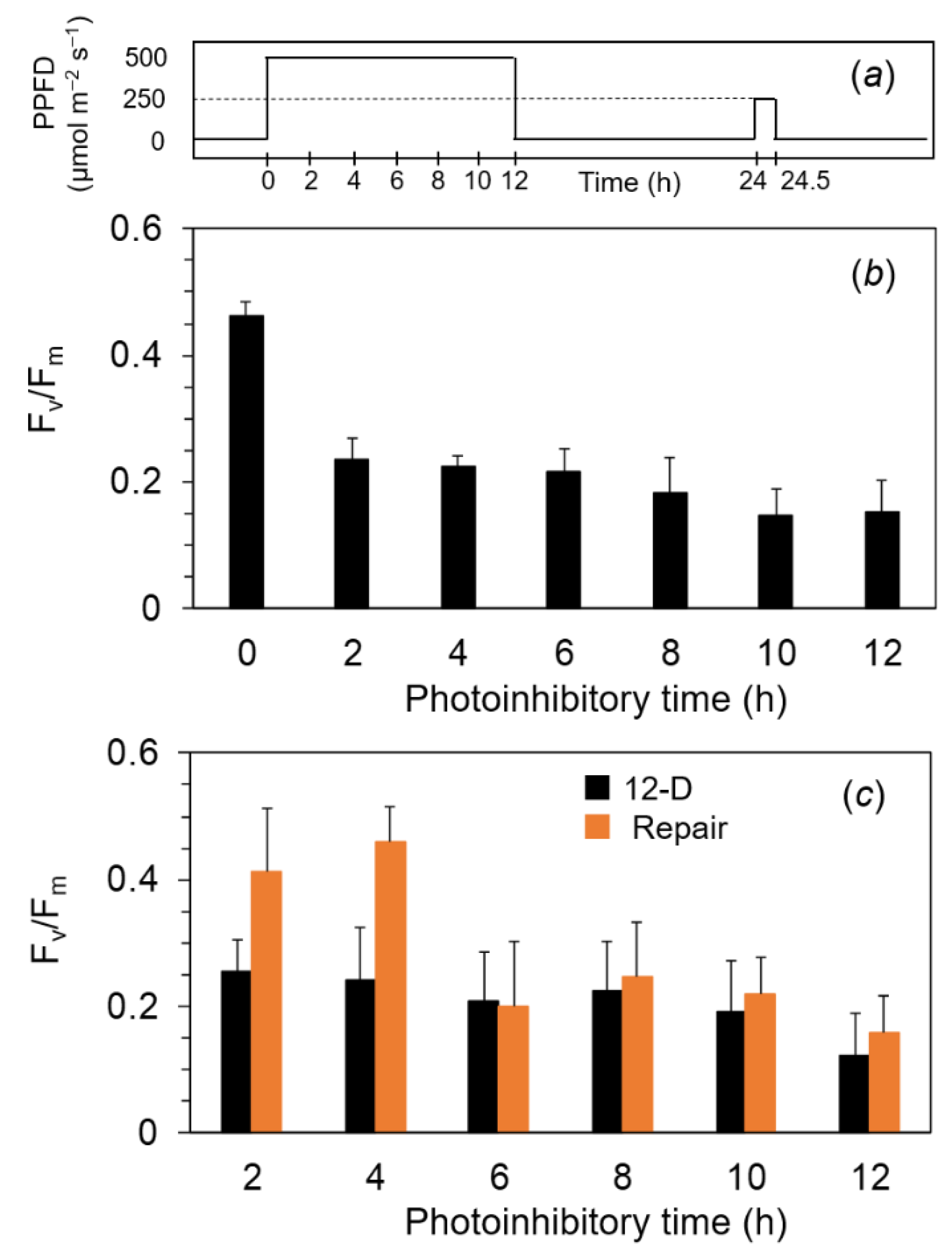
2.3. Quantum Yields of PSII and PSI of the PSII in the Photoinhibited Cells
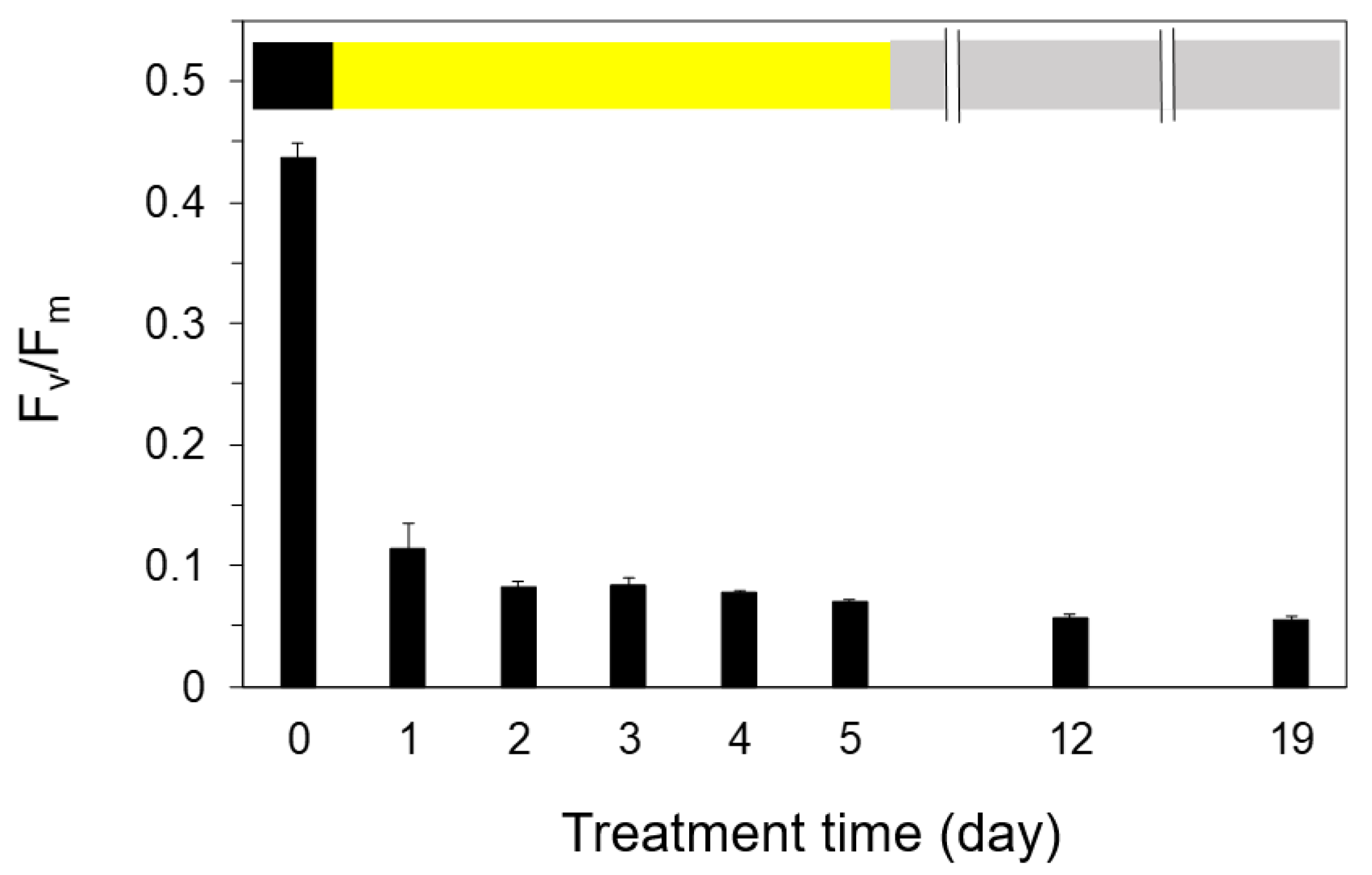
2.4. Photoinhibition under the Expected Natural Environment without the Ice in Winter
3. Discussion
4. Materials and Methods
4.1. Sites and Sample Collection
4.2. Vertical Profile of Water Temperature and Light Intensity under Ice and After Ice-off in the Lake
4.3. Photoinhibitory and Repair Treatments
4.4. Chlorophyll Fluorescence and 830 nm Absorbance Change Measurements
4.5. Neutral Red Uptake Assay
5. Conclusions and Future Remarks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FR | far-red |
| Fm | maximum fluorescence in the dark |
| Fm′ | maximum fluorescence in actinic light |
| Fo | minimum fluorescence in the dark |
| Fo′ | minimal fluorescence yield in actinic light |
| Fs′ | steady-state chlorophyll fluorescence level in actinic light |
| Fv | variable fluorescence (Fm–Fo) |
| ML | measuring light |
| NPQ | non-photochemical quenching of chlorophyll |
| PAM | pulse-amplitude modulation |
| P700 | PSI reaction center |
| PPFD | photosynthetic photon flux density |
| PSI | photosystem I |
| PSII | photosystem II |
| Pm | maximal P700 signal upon full oxidation |
| Pm′ | maximal P700 signal in the presence of actinic light |
| Po | complete reduction level of P700 signal |
| P | oxidized state P700 signal in the presence of actinic light |
| qL | fraction of open PSII centers |
| PQ | plastoquinone |
| SP | saturation pulse |
| Y(II) | quantum yield of the PSII photochemistry |
| Y(NPQ) | quantum yield of the light-induced non-photochemical energy quenching in the PSII antenna |
| Y(I) | quantum yield of the PSI photochemistry |
| Y(ND) | quantum yield of non-photochemical energy dissipation due to the donor-side limitation on PSI electron flow |
| Y(NA) | quantum yield of non-photochemical energy dissipation due to the acceptor-side limitation on PSI electron flow |
References
- Van den Hoek, C. Revision of the European Species of Cladophora; Brill Archive: Leiden, The Netherlands, 1963. [Google Scholar]
- John, D.M.; Whitton, B.A.; Brook, A.J.; York, P.V.; Johnson, L.R. The Freshwater Algal Flora of the British Isles: An Identification Guide to Freshwater and Terrestrial Algae; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Kurogi, M. Lake ball “marimo” in Lake Akan. Phycology 1980, 28, 168–169. [Google Scholar]
- Du, Z.-Y.; Lucker, B.F.; Zienkiewicz, K.; Miller, T.E.; Zienkiewicz, A.; Sears, B.B.; Kramer, D.M.; Benning, C. Galactoglycerolipid Lipase PGD1 Is Involved in Thylakoid Membrane Remodeling in Response to Adverse Environmental Conditions in Chlamydomonas. Plant Cell 2018, 30, 447–465. [Google Scholar] [CrossRef] [PubMed]
- Ruelland, E.; Vaultier, M.-N.; Zachowski, A.; Hurry, V. Chapter 2 Cold Signalling and Cold Acclimation in Plants. In Advances in Botanical Research; Elsevier: Amsterdam, The Netherlands, 2009; pp. 35–150. [Google Scholar]
- Mattila, H.; Mishra, K.B.; Kuusisto, I.; Mishra, A.; Novotná, K.; Šebela, D.; Tyystjärvi, E. Effects of low temperature on photoinhibition and singlet oxygen production in four natural accessions of Arabidopsis. Planta 2020, 252, 19. [Google Scholar] [CrossRef] [PubMed]
- Ermilova, E. Cold Stress Response: An Overview in Chlamydomonas. Front. Plant Sci. 2020, 11, 569437. [Google Scholar] [CrossRef]
- Kaur, M.; Saini, K.C.; Ojah, H.; Sahoo, R.; Gupta, K.; Kumar, A.; Bast, F. Abiotic stress in algae: Response, signaling and transgenic approaches. J. Appl. Phycol. 2022, 34, 1843–1869. [Google Scholar] [CrossRef]
- Bailey, S.; Thompson, E.; Nixon, P.J.; Horton, P.; Mullineaux, C.W.; Robinson, C.; Mann, N.H. A Critical Role for the Var2 FtsH Homologue of Arabidopsis thaliana in the Photosystem II Repair Cycle in vivo. J. Biol. Chem. 2002, 277, 2006–2011. [Google Scholar] [CrossRef]
- Kato, Y.; Sun, X.; Zhang, L.; Sakamoto, W. Cooperative D1 Degradation in the Photosystem II Repair Mediated by Chloroplastic Proteases in Arabidopsis. Plant Physiol. 2012, 159, 1428–1439. [Google Scholar] [CrossRef]
- Zhang, L.; Paakkarinen, V.; van Wijk, K.J.; Aro, E.-M. Co-translational Assembly of the D1 Protein into Photosystem II. J. Biol. Chem. 1999, 274, 16062–16067. [Google Scholar] [CrossRef]
- Kato, Y.; Sakamoto, W. Protein Quality Control in Chloroplasts: A Current Model of D1 Protein Degradation in the Photosystem II Repair Cycle. J. Biochem. 2009, 146, 463–469. [Google Scholar] [CrossRef]
- Aro, E.-M.; Virgin, I.; Andersson, B. Photoinhibition of Photosystem II. Inactivation, protein damage and turnover. Biochim. Et Biophys. Acta (BBA)-Bioenerg. 1993, 1143, 113–134. [Google Scholar] [CrossRef]
- Kornyeyev, D.; Holaday, S.; Logan, B. Predicting the Extent of Photosystem II Photoinactivation Using Chlorophyll a Fluorescence Parameters Measured during Illumination. Plant Cell Physiol. 2003, 44, 1064–1070. [Google Scholar] [CrossRef] [PubMed]
- Tsonev, T.D.; Hikosaka, K. Contribution of Photosynthetic Electron Transport, Heat Dissipation, and Recovery of Photoinactivated Photosystem II to Photoprotection at Different Temperatures in Chenopodium album Leaves. Plant Cell Physiol. 2003, 44, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Greer, D.; Berry, J.; Björkman, O. Photoinhibition of photosynthesis in intact bean leaves: Role of light and temperature, and requirement for chloroplast-protein synthesis during recovery. Planta 1986, 168, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Gombos, Z.; Wada, H.; Murata, N. The recovery of photosynthesis from low-temperature photoinhibition is accelerated by the unsaturation of membrane lipids: A mechanism of chilling tolerance. Proc. Natl. Acad. Sci. USA 1994, 91, 8787–8791. [Google Scholar] [CrossRef] [PubMed]
- Grennan, A.K.; Ort, D.R. Cool temperatures interfere with D1 synthesis in tomato by causing ribosomal pausing. Photosynth. Res. 2007, 94, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Terashima, I.; Funayama, S.; Sonoike, K. The site of photoinhibition in leaves of Cucumis sativus L. at low temperatures is photosystem I, not photosystem II. Planta 1994, 193, 300–306. [Google Scholar] [CrossRef]
- Sonoike, K. Photoinhibition of photosystem I. Physiol. Plant. 2011, 142, 56–64. [Google Scholar] [CrossRef]
- Suorsa, M.; Järvi, S.; Grieco, M.; Nurmi, M.; Pietrzykowska, M.; Rantala, M.; Kangasjärvi, S.; Paakkarinen, V.; Tikkanen, M.; Jansson, S.; et al. PROTON GRADIENT REGULATION5 Is Essential for Proper Acclimation of Arabidopsis Photosystem I to Naturally and Artificially Fluctuating Light Conditions. Plant Cell 2012, 24, 2934–2948. [Google Scholar] [CrossRef]
- Kono, M.; Noguchi, K.; Terashima, I. Roles of the Cyclic Electron Flow Around PSI (CEF-PSI) and O2-Dependent Alternative Pathways in Regulation of the Photosynthetic Electron Flow in Short-Term Fluctuating Light in Arabidopsis thaliana. Plant Cell Physiol. 2014, 55, 990–1004. [Google Scholar] [CrossRef]
- Zivcak, M.; Brestic, M.; Kunderlikova, K.; Sytar, O.; Allakhverdiev, S.I. Repetitive light pulse-induced photoinhibition of photosystem I severely affects CO2 assimilation and photoprotection in wheat leaves. Photosynth. Res. 2015, 126, 449–463. [Google Scholar] [CrossRef]
- Kudoh, H.; Sonoike, K. Irreversible damage to photosystem I by chilling in the light: Cause of the degradation of chlorophyll after returning to normal growth temperature. Planta 2002, 215, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Tikhonov, A.N. pH-Dependent regulation of electron transport and ATP synthesis in chloroplasts. Photosynth. Res. 2013, 116, 511–534. [Google Scholar] [CrossRef] [PubMed]
- Ruban, A.V. Nonphotochemical Chlorophyll Fluorescence Quenching: Mechanism and Effectiveness in Protecting Plants from Photodamage. Plant Physiol. 2016, 170, 1903–1916. [Google Scholar] [CrossRef] [PubMed]
- Kono, M.; Yamori, W.; Suzuki, Y.; Terashima, I. Photoprotection of PSI by Far-Red Light Against the Fluctuating Light-Induced Photoinhibition in Arabidopsis thaliana and Field-Grown Plants. Plant Cell Physiol. 2017, 58, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Miyake, C. Molecular Mechanism of Oxidation of P700 and Suppression of ROS Production in Photosystem I in Response to Electron-Sink Limitations in C3 Plants. Antioxidants 2020, 9, 230. [Google Scholar] [CrossRef]
- Yamori, W.; Shikanai, T. Physiological Functions of Cyclic Electron Transport Around Photosystem I in Sustaining Photosynthesis and Plant Growth. Annu. Rev. Plant Biol. 2016, 67, 81–106. [Google Scholar] [CrossRef]
- Burlacot, A.; Dao, O.; Auroy, P.; Cuiné, S.; Li-Beisson, Y.; Peltier, G. Alternative photosynthesis pathways drive the algal CO2-concentrating mechanism. Nature 2022, 605, 366–371. [Google Scholar] [CrossRef]
- Takizawa, K.; Cruz, J.A.; Kanazawa, A.; Kramer, D.M. The thylakoid proton motive force in vivo. Quantitative, non-invasive probes, energetics, and regulatory consequences of light-induced pmf. Biochim. Et Biophys. Acta (BBA)-Bioenerg. 2007, 1767, 1233–1244. [Google Scholar] [CrossRef]
- Tikhonov, A.N. The cytochrome b6f complex at the crossroad of photosynthetic electron transport pathways. Plant Physiol. Biochem. 2014, 81, 163–183. [Google Scholar] [CrossRef]
- Kono, M.; Terashima, I. Elucidation of Photoprotective Mechanisms of PSI Against Fluctuating Light photoinhibition. Plant Cell Physiol. 2016, 57, 1405–1414. [Google Scholar] [CrossRef]
- Tikkanen, M.; Mekala, N.R.; Aro, E.-M. Photosystem II photoinhibition-repair cycle protects Photosystem I from irreversible damage. Biochim. Et Biophys. Acta (BBA)-Bioenerg. 2014, 1837, 210–215. [Google Scholar] [CrossRef]
- Cano-Ramirez, D.L.; Saskia de Fraine, T.; Griffiths, O.G.; Dodd, A.N. Photosynthesis and circadian rhythms regulate the buoyancy of marimo lake balls. Curr. Biol. 2018, 28, R869–R870. [Google Scholar] [CrossRef]
- Yoshida, T. Marimo Research. Kushiro Board Educ. 1994, 3, 1–58. [Google Scholar]
- Sonoike, K.; Terashima, I. Mechanism of photosystem-I photoinhibition in leaves of Cucumis sativus L. Planta 1994, 194, 287–293. [Google Scholar] [CrossRef]
- Ramalho, J.C.; Campos, P.S.; Quartin, V.L.; Silva, M.J.; Nunes, M.A. High Irradiance Impairments on Photosynthetic Electron Transport, Ribulose-1,5-bisphosphate Carboxylase/oxygenase and N Assimilation as a Function of N Availability in Coffea arabica L. Plants. J. Plant Physiol. 1999, 154, 319–326. [Google Scholar] [CrossRef]
- Nakano, R.; Ishida, H.; Kobayashi, M.; Makino, A.; Mae, T. Biochemical changes associated with in vivo RbcL fragmentation by reactive oxygen species under chilling-light conditions. Plant Biol. 2010, 12, 35–45. [Google Scholar] [CrossRef]
- Zhang, S.; Scheller, H.V. Photoinhibition of Photosystem I at Chilling Temperature and Subsequent Recovery in Arabidopsis thaliana. Plant Cell Physiol. 2004, 45, 1595–1602. [Google Scholar] [CrossRef]
- Tsuyama, M.; Kobayashi, Y. Reduction of the primary donor P700 of photosystem I during steady-state photosynthesis under low light in Arabidopsis. Photosynth. Res. 2009, 99, 37–47. [Google Scholar] [CrossRef]
- Yamori, W.; Makino, A.; Shikanai, T. A physiological role of cyclic electron transport around photosystem I in sustaining photosynthesis under fluctuating light in rice. Sci. Rep. 2016, 6, 20147. [Google Scholar] [CrossRef]
- Sejima, T.; Takagi, D.; Fukayama, H.; Makino, A.; Miyake, C. Repetitive Short-Pulse Light Mainly Inactivates Photosystem I in Sunflower Leaves. Plant Cell Physiol. 2014, 55, 1184–1193. [Google Scholar] [CrossRef]
- Allahverdiyeva, Y.; Mustila, H.; Ermakova, M.; Bersanini, L.; Richaud, P.; Ajlani, G.; Battchikova, N.; Cournac, L.; Aro, E.-M. Flavodiiron proteins Flv1 and Flv3 enable cyanobacterial growth and photosynthesis under fluctuating light. Proc. Natl. Acad. Sci.USA 2013, 110, 4111–4116. [Google Scholar] [CrossRef]
- Bulychev, A.A.; Cherkashin, A.A.; Muronets, E.M.; Elanskaya, I.V. Photoinduction of electron transport on the acceptor side of PSI in Synechocystis PCC 6803 mutant deficient in flavodiiron proteins Flv1 and Flv3. Biochim. Et Biophys. Acta (BBA)-Bioenerg. 2018, 1859, 1086–1095. [Google Scholar] [CrossRef]
- Chaux, F.; Johnson, X.; Auroy, P.; Beyly-Adriano, A.; Te, I.; Cuiné, S.; Peltier, G. PGRL1 and LHCSR3 Compensate for Each Other in Controlling Photosynthesis and Avoiding Photosystem I Photoinhibition during High Light Acclimation of Chlamydomonas Cells. Mol. Plant 2017, 10, 216–218. [Google Scholar] [CrossRef]
- Chaux, F.; Burlacot, A.; Mekhalfi, M.; Auroy, P.; Blangy, S.; Richaud, P.; Peltier, G. Flavodiiron Proteins Promote Fast and Transient O2 Photoreduction in Chlamydomonas. Plant Physiol. 2017, 174, 1825–1836. [Google Scholar] [CrossRef]
- Valledor, L.; Furuhashi, T.; Hanak, A.-M.; Weckwerth, W. Systemic Cold Stress Adaptation of Chlamydomonas reinhardtii. Mol. Cell. Proteom. 2013, 12, 2032–2047. [Google Scholar] [CrossRef]
- Ishida, S.; Morita, K.; Kishine, M.; Takabayashi, A.; Murakami, R.; Takeda, S.; Shimamoto, K.; Sato, F.; Endo, T. Allocation of Absorbed Light Energy in PSII to Thermal Dissipations in the Presence or Absence of PsbS Subunits of Rice. Plant Cell Physiol. 2011, 52, 1822–1831. [Google Scholar] [CrossRef]
- Ishida, S.; Uebayashi, N.; Tazoe, Y.; Ikeuchi, M.; Homma, K.; Sato, F.; Endo, T. Diurnal and Developmental Changes in Energy Allocation of Absorbed Light at PSII in Field-Grown Rice. Plant Cell Physiol. 2014, 55, 171–182. [Google Scholar] [CrossRef]
- Ono, T.-A.; Inoue, Y. Photoactivation of the Water-Oxidation System in Isolated Intact Chloroplasts Prepared from Wheat Leaves Grown under Intermittent Flash Illumination. Plant Physiol. 1982, 69, 1418–1422. [Google Scholar] [CrossRef]
- He, J.; Chow, W.S. The rate coefficient of repair of photosystem II after photoinactivation. Physiol. Plant. 2003, 118, 297–304. [Google Scholar] [CrossRef]
- Tóth, S.Z.; Schansker, G.; Kissimon, J.; Kovács, L.; Garab, G.; Strasser, R. Biophysical studies of photosystem II-related recovery processes after a heat pulse in barley seedlings (Hordeum vulgare L.). J. Plant Physiol. 2005, 162, 181–194. [Google Scholar] [CrossRef]
- Takahashi, S.; Badger, M.R. Photoprotection in plants: A new light on photosystem II damage. Trends Plant Sci. 2011, 16, 53–60. [Google Scholar] [CrossRef]
- DeEll, J.R.; Toivonen, P.M. Practical Applications of Chlorophyll Fluorescence in Plant Biology; Springer Science & Business Media: Berlin, Germany, 2003. [Google Scholar]
- Falkowski, P.G.; Koblfzek, M.; Gorbunov, M.; Kolber, Z. Development and application of variable chlorophyll fluorescence techniques in marine ecosystems. In Chlorophyll a Fluorescence; Springer: Berlin/Heidelberg, Germany, 2004; pp. 757–778. [Google Scholar]
- Raven, J.A.; Maberly, S.C. Plant productivity of inland waters. In Chlorophyll a Fluorescence; Springer: Berlin/Heidelberg, Germany, 2004; pp. 779–793. [Google Scholar]
- Papageorgiou, G.C.; Tsimilli-Michael, M.; Stamatakis, K. The fast and slow kinetics of chlorophyll a fluorescence induction in plants, algae and cyanobacteria: A viewpoint. Photosynth. Res. 2007, 94, 275–290. [Google Scholar] [CrossRef]
- Butler, W.L. Energy Distribution in the Photochemical Apparatus of Photosynthesis. Annu. Rev. Plant Physiol. 1978, 29, 345–378. [Google Scholar] [CrossRef]
- Genty, B.; Briantais, J.-M.; Baker, N.R. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta (BBA)-Gen. Subj. 1989, 990, 87–92. [Google Scholar] [CrossRef]
- Hendrickson, L.; Furbank, R.T.; Chow, W.S. A Simple Alternative Approach to Assessing the Fate of Absorbed Light Energy Using Chlorophyll Fluorescence. Photosynth. Res. 2004, 82, 73–81. [Google Scholar] [CrossRef]
- Kramer, D.M.; Johnson, G.; Kiirats, O.; Edwards, G.E. New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth. Res. 2004, 79, 209–218. [Google Scholar] [CrossRef]
- Klughammer, C.; Schreiber, U. Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the Saturation Pulse method. PAM Appl. Notes 2008, 1, 201–247. [Google Scholar]
- Oxborough, K.; Baker, N.R.B. Resolving chlorophyll a fluorescence images of photosynthetic efficiency into photochemical and non-photochemical components—Calculation of qP and Fv-/Fm-; without measuring Fo. Photosynth. Res. 1997, 54, 135–142. [Google Scholar] [CrossRef]
- Klughammer, C.; Schreiber, U. An improved method, using saturating light pulses, for the determination of photosystem I quantum yield via P700+-absorbance changes at 830 nm. Planta 1994, 192, 261–268. [Google Scholar] [CrossRef]
- Repetto, G.; del Peso, A.; Zurita, J.L. Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat. Protoc. 2008, 3, 1125–1131. [Google Scholar] [CrossRef]
- Harden, E.A.; Falshaw, R.; Carnachan, S.M.; Kern, E.R.; Prichard, M.N. Virucidal activity of polysaccharide extracts from four algal species against herpes simplex virus. Antivir. Res. 2009, 83, 282–289. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obara, A.; Ogawa, M.; Oyama, Y.; Suzuki, Y.; Kono, M. Effects of High Irradiance and Low Water Temperature on Photoinhibition and Repair of Photosystems in Marimo (Aegagropila linnaei) in Lake Akan, Japan. Int. J. Mol. Sci. 2023, 24, 60. https://doi.org/10.3390/ijms24010060
Obara A, Ogawa M, Oyama Y, Suzuki Y, Kono M. Effects of High Irradiance and Low Water Temperature on Photoinhibition and Repair of Photosystems in Marimo (Aegagropila linnaei) in Lake Akan, Japan. International Journal of Molecular Sciences. 2023; 24(1):60. https://doi.org/10.3390/ijms24010060
Chicago/Turabian StyleObara, Akina, Mari Ogawa, Yoichi Oyama, Yoshihiro Suzuki, and Masaru Kono. 2023. "Effects of High Irradiance and Low Water Temperature on Photoinhibition and Repair of Photosystems in Marimo (Aegagropila linnaei) in Lake Akan, Japan" International Journal of Molecular Sciences 24, no. 1: 60. https://doi.org/10.3390/ijms24010060
APA StyleObara, A., Ogawa, M., Oyama, Y., Suzuki, Y., & Kono, M. (2023). Effects of High Irradiance and Low Water Temperature on Photoinhibition and Repair of Photosystems in Marimo (Aegagropila linnaei) in Lake Akan, Japan. International Journal of Molecular Sciences, 24(1), 60. https://doi.org/10.3390/ijms24010060





