Half-Life Extension and Biodistribution Modulation of Biotherapeutics via Red Blood Cell Hitch-Hiking with Novel Anti-Band 3 Single-Domain Antibodies
Abstract
1. Introduction
2. Results
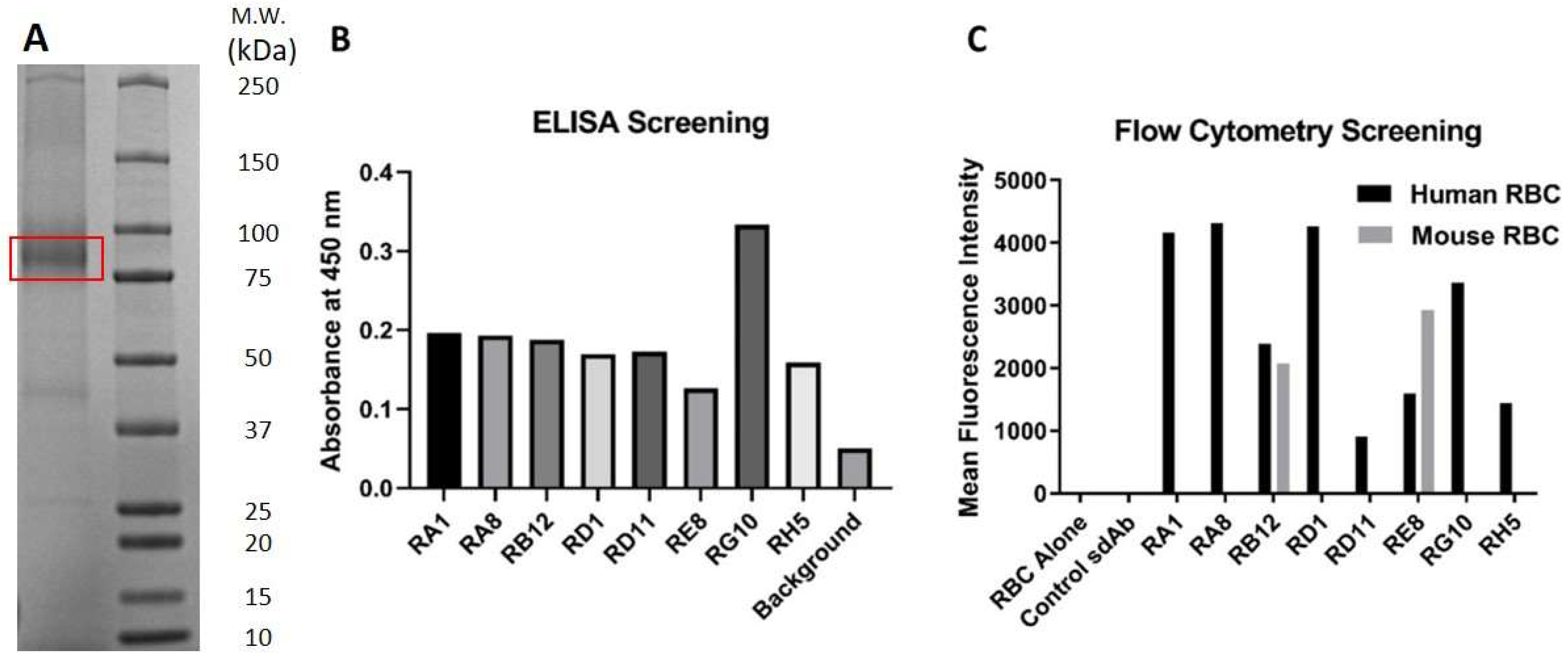
2.1. RBC Immunization, Panning, and Screening
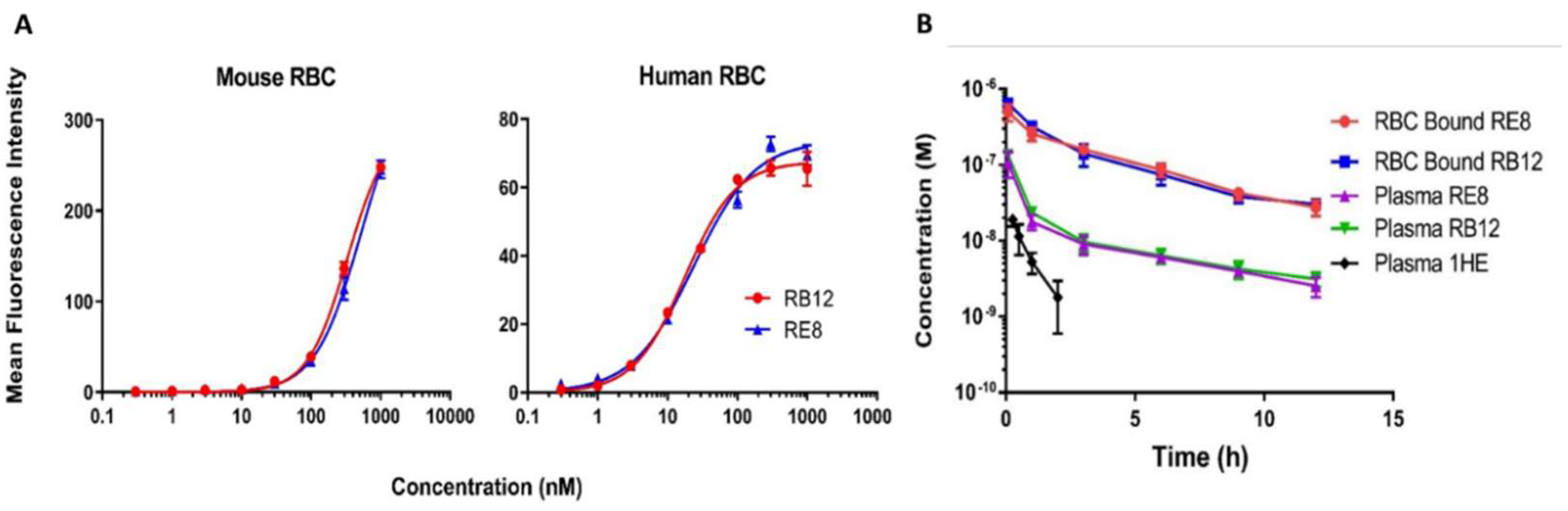
2.2. Characterizations of Binding Affinity and In Vivo Pharmacokinetics of RB12 and RE8
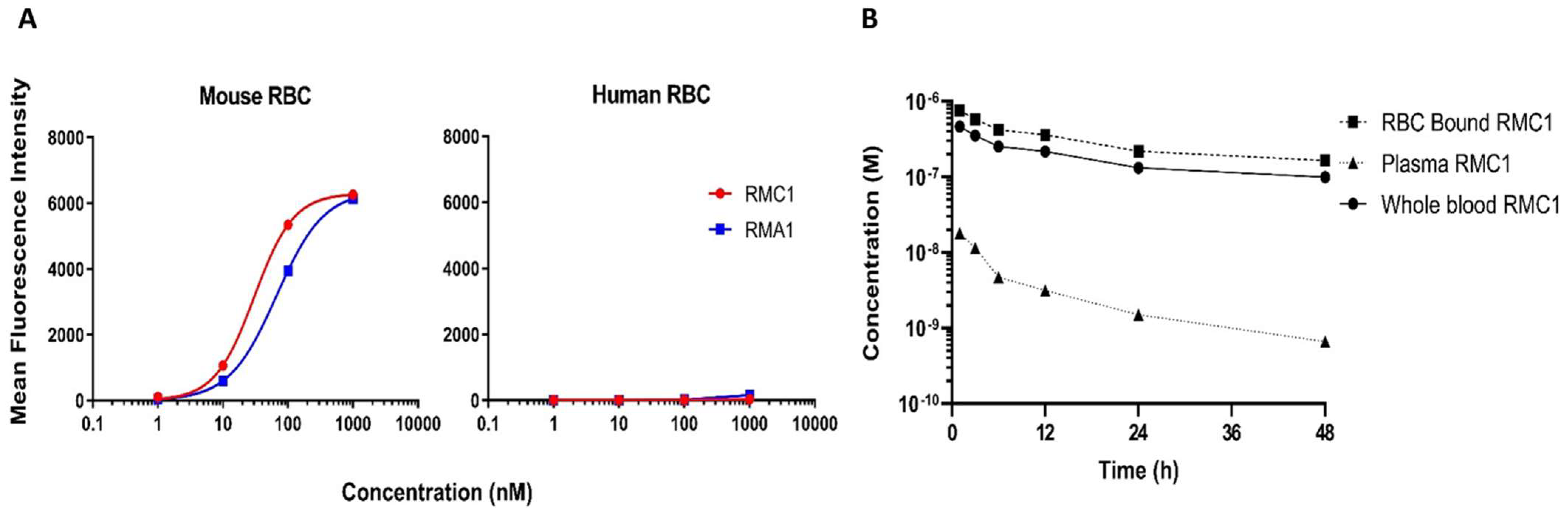
2.3. Affinity Maturation of RB12 and RE8
2.4. Development and Assessment of Anti-RBC-aTNF Fusion Bispecific Antibody
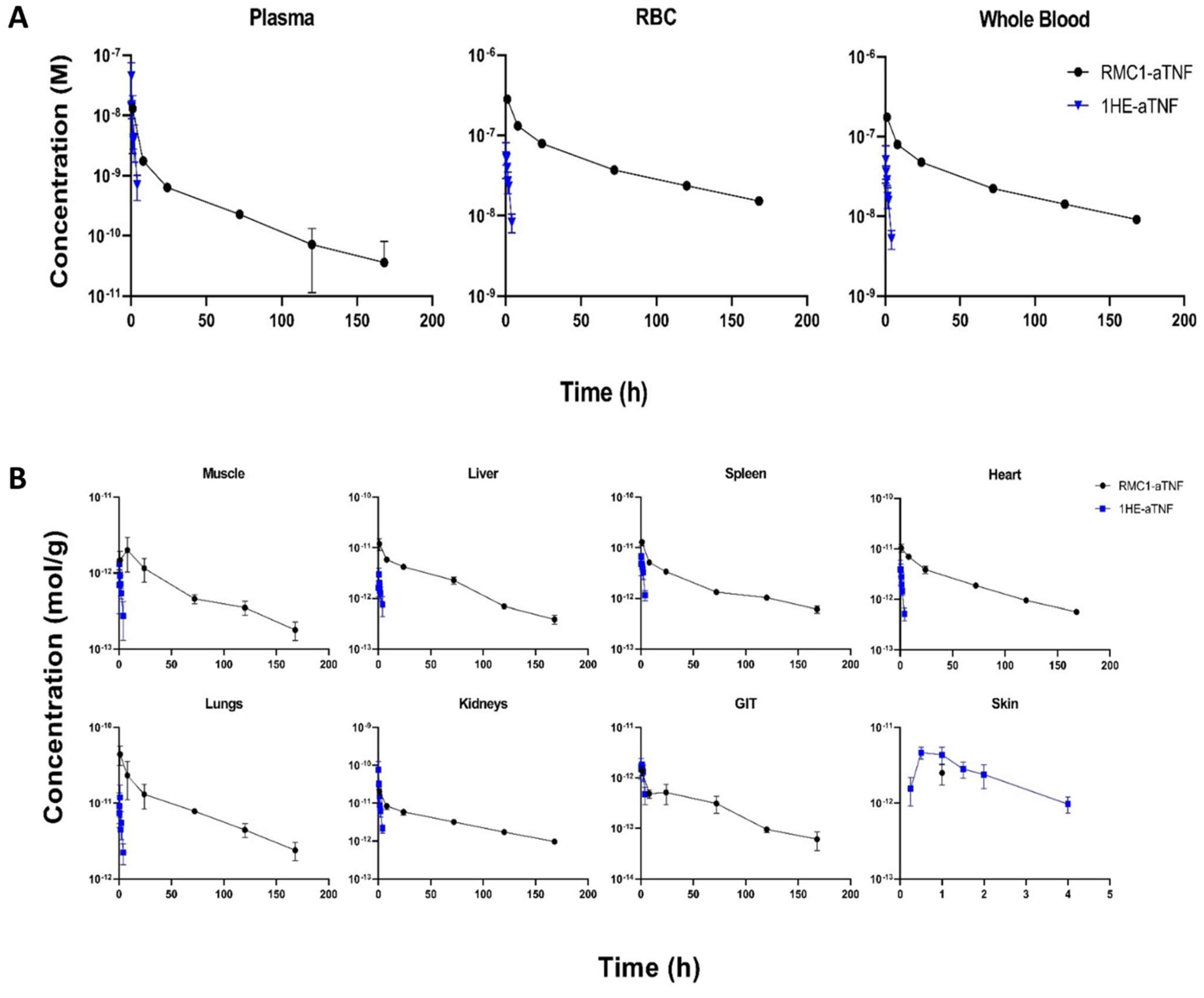
2.5. Pharmacokinetics and Biodistribution of the Anti-RBC-TNF-α Fusion Antibody
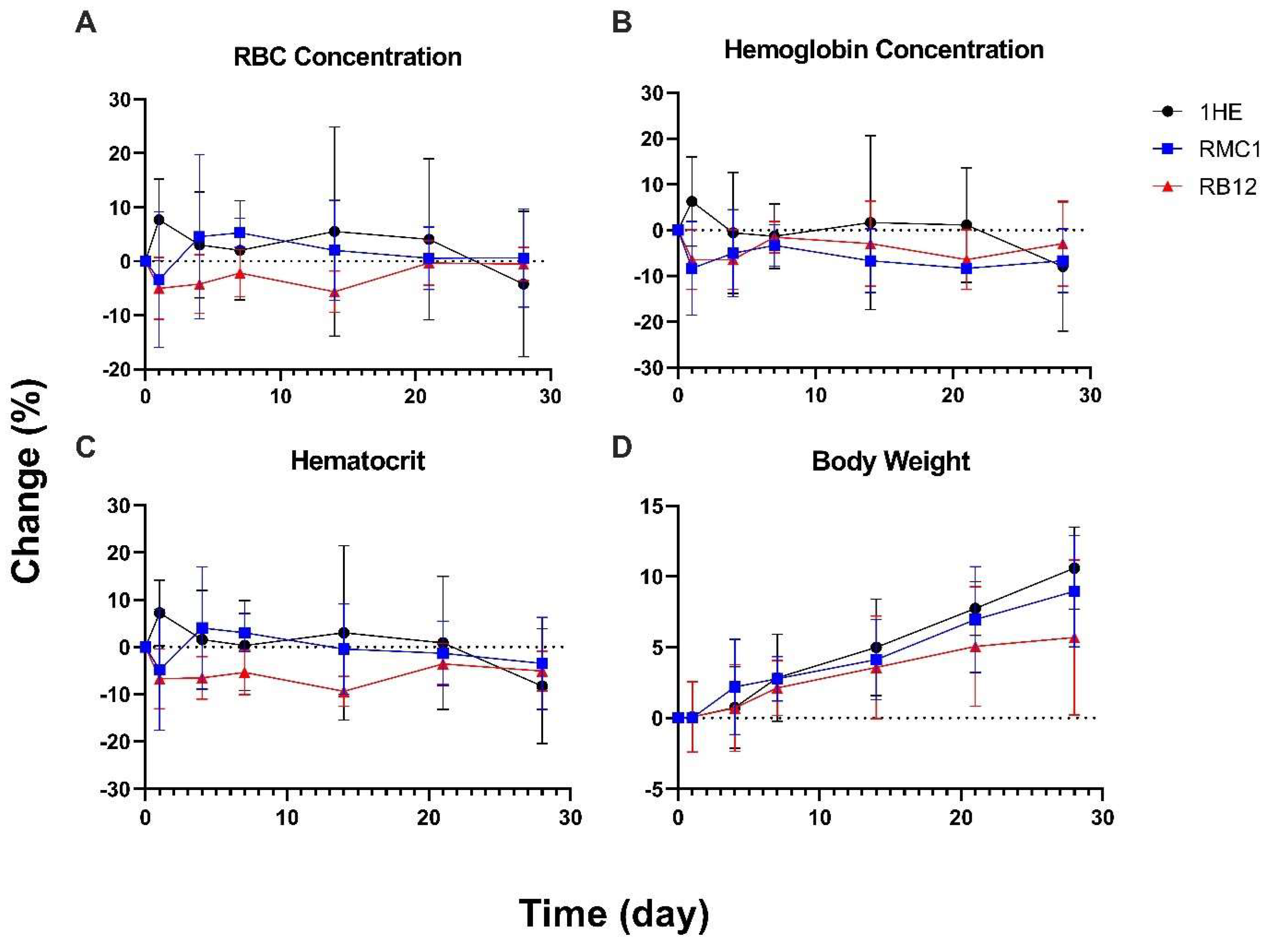
2.6. Hematological Analysis of RMC1-aTNF in Mice
3. Discussion
4. Materials and Methods
4.1. Extraction of Red Blood Cell B3p
4.2. Red Blood Cell Antigen Immunization and Phage-Display Library Construction
4.3. Phage Panning to Enrich sdAb Binders
4.4. Screening of sdAb Binders
4.5. Protein Expression and Purification
4.6. Flow Cytometry Analysis
4.7. Radiolabeling of Antibodies
4.8. Pharmacokinetics of Anti-B3p SdAbs
4.9. Hematology Study
4.10. Plasma and Tissue Biodistribution of Bispecific Fusion Constructs
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Walsh, G. Biopharmaceutical benchmarks 2018. Nat. Biotechnol. 2018, 36, 1136–1145. [Google Scholar] [CrossRef] [PubMed]
- Lobo, E.D.; Hansen, R.J.; Balthasar, J.P. Antibody pharmacokinetics and pharmacodynamics. J. Pharm. Sci. 2004, 93, 2645–2668. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, E.Q.; Balthasar, J.P. Monoclonal antibody pharmacokinetics and pharmacodynamics. Clin. Pharm. Ther. 2008, 84, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Thomas, V.A.; Balthasar, J.P. Understanding Inter-Individual Variability in Monoclonal Antibody Disposition. Antibodies 2019, 8, 56. [Google Scholar] [CrossRef] [PubMed]
- Remigante, A.; Morabito, R.; Marino, A. Band 3 protein function and oxidative stress in erythrocytes. J. Cell Physiol. 2021, 236, 6225–6234. [Google Scholar] [CrossRef]
- Badior, K.E.; Casey, J.R. Molecular mechanism for the red blood cell senescence clock. IUBMB Life 2018, 70, 32–40. [Google Scholar] [CrossRef]
- Hamers-Casterman, C.; Atarhouch, T.; Muyldermans, S.; Robinson, G.; Hammers, C.; Songa, E.B.; Bendahman, N.; Hammers, R. Naturally occurring antibodies devoid of light chains. Nature 1993, 363, 446–448. [Google Scholar] [CrossRef]
- Greenberg, A.S.; Avila, D.; Hughes, M.; Hughes, A.; McKinney, E.C.; Flajnik, M.F. A new antigen receptor gene family that undergoes rearrangement and extensive somatic diversification in sharks. Nature 1995, 374, 168–173. [Google Scholar] [CrossRef]
- Hentrich, C.; Ylera, F.; Frisch, C.; Ten Haaf, A.; Knappik, A. Chapter 3—Monoclonal Antibody Generation by Phage Display: History, State-of-the-Art, and Future. In Handbook of Immunoassay Technologies; Vashist, S.K., Luong, J.H.T., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 47–80. [Google Scholar]
- Iezzi, M.E.; Policastro, L.; Werbajh, S.; Podhajcer, O.; Canziani, G.A. Single-Domain Antibodies and the Promise of Modular Targeting in Cancer Imaging and Treatment. Front. Immunol. 2018, 9, 273. [Google Scholar] [CrossRef]
- Arbabi-Ghahroudi, M. Camelid Single-Domain Antibodies: Historical Perspective and Future Outlook. Front. Immunol. 2017, 8, 1589. [Google Scholar] [CrossRef]
- Gonzalez-Sapienza, G.; Rossotti, M.A.; Tabares-da Rosa, S. Single-Domain Antibodies as Versatile Affinity Reagents for Analytical and Diagnostic Applications. Front. Immunol. 2017, 8, 977. [Google Scholar] [CrossRef] [PubMed]
- Beirnaert, E.; Desmyter, A.; Spinelli, S.; Lauwereys, M.; Aarden, L.; Dreier, T.; Loris, R.; Silence, K.; Pollet, C.; Cambillau, C.; et al. Bivalent Llama Single-Domain Antibody Fragments against Tumor Necrosis Factor Have Picomolar Potencies due to Intramolecular Interactions. Front. Immunol. 2017, 8, 867. [Google Scholar] [CrossRef] [PubMed]
- Brenner, J.S.; Mitragotri, S.; Muzykantov, V.R. Red Blood Cell Hitchhiking: A Novel Approach for Vascular Delivery of Nanocarriers. Annu. Rev. Biomed. Eng. 2021, 23, 225–248. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; Fraternale, A.; Bianchi, M.; Magnani, M. Red Blood Cell Membrane Processing for Biomedical Applications. Front. Physiol. 2019, 10, 1070. [Google Scholar] [CrossRef]
- Hess, C.; Schifferli, J.A. Immune adherence revisited: Novel players in an old game. News Physiol. Sci. 2003, 18, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Murciano, J.-C.; Medinilla, S.; Eslin, D.; Atochina, E.; Cines, D.B.; Muzykantov, V.R. Prophylactic fibrinolysis through selective dissolution of nascent clots by tPA-carrying erythrocytes. Nat. Biotechnol. 2003, 21, 891–896. [Google Scholar] [CrossRef]
- Lindorfer, M.A.; Nardin, A.; Foley, P.L.; Solga, M.D.; Bankovich, A.J.; Martin, E.N.; Henderson, A.L.; Price, C.W.; Gyimesi, E.; Wozencraft, C.P.; et al. Targeting of Pseudomonas aeruginosa in the bloodstream with bispecific monoclonal antibodies. J. Immunol. 2001, 167, 2240–2249. [Google Scholar] [CrossRef]
- Asher, D.R.; Cerny, A.M.; Finberg, R.W. The erythrocyte viral trap: Transgenic expression of viral receptor on erythrocytes attenuates coxsackievirus B infection. Proc. Natl. Acad. Sci. USA 2005, 102, 12897–12902. [Google Scholar] [CrossRef]
- Hahn, C.S.; French, O.G.; Foley, P.; Martin, E.N.; Taylor, R.P. Bispecific Monoclonal Antibodies Mediate Binding of Dengue Virus to Erythrocytes in a Monkey Model of Passive Viremia. J. Immunol. 2001, 166, 1057. [Google Scholar] [CrossRef]
- Taylor, R.P.; Sutherland, W.M.; Martin, E.N.; Ferguson, P.J.; Reinagel, M.L.; Gilbert, E.; Lopez, K.; Incardona, N.L.; Ochs, H.D. Bispecific monoclonal antibody complexes bound to primate erythrocyte complement receptor 1 facilitate virus clearance in a monkey model. J. Immunol. 1997, 158, 842–850. [Google Scholar]
- Sharma, R.; Zhao, H.; Al-Saleem, F.H.; Ubaid, A.S.; Puligedda, R.D.; Segan, A.T.; Lindorfer, M.A.; Bermudez, R.; Elias, M.; Adekar, S.P.; et al. Mechanisms of enhanced neutralization of botulinum neurotoxin by monoclonal antibodies conjugated to antibodies specific for the erythrocyte complement receptor. Mol. Immunol. 2014, 57, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Pretini, V.; Koenen, M.H.; Kaestner, L.; Fens, M.H.A.M.; Schiffelers, R.M.; Bartels, M.; Van Wijk, R. Red Blood Cells: Chasing Interactions. Front. Physiol. 2019, 10, 945. [Google Scholar] [CrossRef] [PubMed]
- Greineder, C.F.; Howard, M.D.; Carnemolla, R.; Cines, D.B.; Muzykantov, V.R. Advanced drug delivery systems for antithrombotic agents. Blood 2013, 122, 1565–1575. [Google Scholar] [CrossRef] [PubMed]
- Zaitsev, S.; Spitzer, D.; Murciano, J.C.; Ding, B.S.; Tliba, S.; Kowalska, M.A.; Marcos-Contreras, O.A.; Kuo, A.; Stepanova, V.; Atkinson, J.P.; et al. Sustained thromboprophylaxis mediated by an RBC-targeted pro-urokinase zymogen activated at the site of clot formation. Blood 2010, 115, 5241–5248. [Google Scholar] [CrossRef] [PubMed]
- Zaitsev, S.; Kowalska, M.A.; Neyman, M.; Carnemolla, R.; Tliba, S.; Ding, B.S.; Stonestrom, A.; Spitzer, D.; Atkinson, J.P.; Poncz, M.; et al. Targeting recombinant thrombomodulin fusion protein to red blood cells provides multifaceted thromboprophylaxis. Blood 2012, 119, 4779–4785. [Google Scholar] [CrossRef]
- Villa, C.H.; Pan, D.C.; Johnston, I.H.; Greineder, C.F.; Walsh, L.R.; Hood, E.D.; Cines, D.B.; Poncz, M.; Siegel, D.L.; Muzykantov, V.R. Biocompatible coupling of therapeutic fusion proteins to human erythrocytes. Blood Adv. 2018, 2, 165–176. [Google Scholar] [CrossRef]
- Khoory, J.; Estanislau, J.; Elkhal, A.; Lazaar, A.; Melhorn, M.I.; Brodsky, A.; Illigens, B.; Hamachi, I.; Kurishita, Y.; Ivanov, A.R.; et al. Ligation of Glycophorin A Generates Reactive Oxygen Species Leading to Decreased Red Blood Cell Function. PLoS ONE 2016, 11, e0141206. [Google Scholar] [CrossRef]
- Hsu, K. Exploring the Potential Roles of Band 3 and Aquaporin-1 in Blood CO2 Transport–Inspired by Comparative Studies of Glycophorin B-A-B Hybrid Protein GP. Mur. Front. Physiol. 2018, 9, 733. [Google Scholar] [CrossRef]
- Villa, C.H.; Pan, D.C.; Zaitsev, S.; Cines, D.B.; Siegel, D.L.; Muzykantov, V.R. Delivery of drugs bound to erythrocytes: New avenues for an old intravascular carrier. Ther. Deliv. 2015, 6, 795–826. [Google Scholar] [CrossRef]
- Paulitschke, M.; Nash, G.B.; Anstee, D.J.; Tanner, M.J.A.; Gratzer, W.B. Perturbation of Red Blood Cell Membrane Rigidity by Extracellular Ligands. Blood 1995, 86, 342–348. [Google Scholar] [CrossRef]
- Harmsen, M.M.; De Haard, H.J. Properties, production, and applications of camelid single-domain antibody fragments. Appl. Microbiol. Biotechnol. 2007, 77, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Zarschler, K.; Witecy, S.; Kapplusch, F.; Foerster, C.; Stephan, H. High-yield production of functional soluble single-domain antibodies in the cytoplasm of Escherichia coli. Microb. Cell Fact 2013, 12, 97. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, S.W.; Lin, S.C.; Stoll, B.R.; Baxter, L.T.; Munn, L.L.; Jain, R.K. Antibody-directed effector cell therapy of tumors: Analysis and optimization using a physiologically based pharmacokinetic model. Neoplasia 2002, 4, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.W.; Haber, E.; Yeatman, L.; Butler, V., Jr. Reversal of advanced digoxin intoxication with Fab fragments of digoxin-specific antibodies. N. Engl. J. Med. 1976, 294, 797–800. [Google Scholar] [CrossRef]
- Baud, F.J.; Sabouraud, A.; Vicaut, E.; Taboulet, P.; Lang, J.; Bismuth, C.; Rouzioux, J.M.; Scherrmann, J.M. Brief report: Treatment of severe colchicine overdose with colchicine-specific Fab fragments. N. Engl. J. Med. 1995, 332, 642–645. [Google Scholar] [CrossRef]
- Bordeau, B.M.; Abuqayyas, L.; Nguyen, T.D.; Chen, P.; Balthasar, J.P. Development and Evaluation of Competitive Inhibitors of Trastuzumab-HER2 Binding to Bypass the Binding-Site Barrier. Front. Pharm. 2022, 13, 837744. [Google Scholar] [CrossRef]
- Bordeau, B.M.; Yang, Y.; Balthasar, J.P. Transient Competitive Inhibition Bypasses the Binding Site Barrier to Improve Tumor Penetration of Trastuzumab and Enhance T-DM1 Efficacy. Cancer Res. 2021, 81, 4145–4154. [Google Scholar] [CrossRef]
- Chen, P.; Bordeau, B.M.; Zhang, Y.; Balthasar, J.P. Transient Inhibition of Trastuzumab-Tumor Binding to Overcome the "Binding-Site Barrier" and Improve the Efficacy of a Trastuzumab-Gelonin Immunotoxin. Mol. Cancer Ther. 2022, 21, 1573–1582. [Google Scholar] [CrossRef]
- Bech, E.M.; Pedersen, S.L.; Jensen, K.J. Chemical Strategies for Half-Life Extension of Biopharmaceuticals: Lipidation and Its Alternatives. ACS Med. Chem. Lett. 2018, 9, 577–580. [Google Scholar] [CrossRef]
- Khodabakhsh, F.; Salimian, M.; Hedayati, M.H.; Ahangari Cohan, R.; Norouzian, D. Challenges and advancements in the pharmacokinetic enhancement of therapeutic proteins. Prep. Biochem. Biotechnol. 2021, 51, 519–529. [Google Scholar] [CrossRef]
- Merten, H.; Brandl, F.; Zimmermann, M.; Schaefer, J.V.; Irpinio, L.; Sand, K.M.K.; Nilsen, J.; Andersen, J.T.; Zangemeister-Wittke, U.; Plückthun, A. Half-life extension of efficiently produced DARPin serum albumin fusions as a function of FcRn affinity and recycling. Eur. J. Pharm. Biopharm. 2021, 167, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Su, W.; Zhang, W.; Wang, P.; Sattler, M.; Zou, P. Recent Advances in Half-life Extension Strategies for Therapeutic Peptides and Proteins. Curr. Pharm. Des. 2019, 24, 4932–4946. [Google Scholar] [CrossRef]
- Zaman, R.; Islam, R.A.; Ibnat, N.; Othman, I.; Zaini, A.; Lee, C.Y.; Chowdhury, E.H. Current strategies in extending half-lives of therapeutic proteins. J. Control. Release 2019, 301, 176–189. [Google Scholar] [CrossRef] [PubMed]
- Sivilotti, M.L. Oxidant stress and haemolysis of the human erythrocyte. Toxicol. Rev. 2004, 23, 169–188. [Google Scholar] [CrossRef] [PubMed]
- Casey, J.R.; Lieberman, D.M.; Reithmeier, R.A. Purification and characterization of band 3 protein. Methods Enzym. 1989, 173, 494–512. [Google Scholar] [CrossRef]
- Pimplikar, S.W.; Reithmeier, R.A. Affinity chromatography of Band 3, the anion transport protein of erythrocyte membranes. J. Biol. Chem. 1986, 261, 9770–9778. [Google Scholar] [CrossRef]
- Rosa, S.T.; Rossotti, M.; Carleiza, C.; Carrión, F.; Pritsch, O.; Ahn, K.C.; Last, J.A.; Hammock, B.D.; González-Sapienza, G. Competitive Selection from Single Domain Antibody Libraries Allows Isolation of High-Affinity Antihapten Antibodies That Are Not Favored in the llama Immune Response. Anal. Chem. 2011, 83, 7213–7220. [Google Scholar] [CrossRef]
- Garg, A.; Balthasar, J.P. Physiologically-based pharmacokinetic (PBPK) model to predict IgG tissue kinetics in wild-type and FcRn-knockout mice. J. Pharmacokinet. Pharmacodyn. 2007, 34, 687–709. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.D.; Bordeau, B.M.; Zhang, Y.; Mattle, A.G.; Balthasar, J.P. Half-Life Extension and Biodistribution Modulation of Biotherapeutics via Red Blood Cell Hitch-Hiking with Novel Anti-Band 3 Single-Domain Antibodies. Int. J. Mol. Sci. 2023, 24, 475. https://doi.org/10.3390/ijms24010475
Nguyen TD, Bordeau BM, Zhang Y, Mattle AG, Balthasar JP. Half-Life Extension and Biodistribution Modulation of Biotherapeutics via Red Blood Cell Hitch-Hiking with Novel Anti-Band 3 Single-Domain Antibodies. International Journal of Molecular Sciences. 2023; 24(1):475. https://doi.org/10.3390/ijms24010475
Chicago/Turabian StyleNguyen, Toan D., Brandon M. Bordeau, Yu Zhang, Anna G. Mattle, and Joseph P. Balthasar. 2023. "Half-Life Extension and Biodistribution Modulation of Biotherapeutics via Red Blood Cell Hitch-Hiking with Novel Anti-Band 3 Single-Domain Antibodies" International Journal of Molecular Sciences 24, no. 1: 475. https://doi.org/10.3390/ijms24010475
APA StyleNguyen, T. D., Bordeau, B. M., Zhang, Y., Mattle, A. G., & Balthasar, J. P. (2023). Half-Life Extension and Biodistribution Modulation of Biotherapeutics via Red Blood Cell Hitch-Hiking with Novel Anti-Band 3 Single-Domain Antibodies. International Journal of Molecular Sciences, 24(1), 475. https://doi.org/10.3390/ijms24010475





