1. Introduction
The polymeric biomaterials field includes a wide range of applications, as coatings in biomedical devices or as material in devices, such as catheters, heart valves, and dialysis membranes [
1]. The polymers that were initially developed for a variety of industrial applications were subsequently adapted to be used as biomaterials, based on their specific characteristics, namely the wettability, chemical composition, porosity, flexibility/stiffness, roughness—properties significant in relation to the existing physiological environment [
2,
3]. Therefore, the development of new polymeric materials with sustainability, multifunctionality, and advanced properties is a challenge of our days. This research helps us to better understand the interaction nature from the systems and the relation between the structure, composition, and surface properties of the polymeric materials.
Most biological reactions occur on the surfaces and at the interfaces. For this reason, the performance of a material in biological environment is initially dependent on its surface properties and also bulk properties of the basic polymers. Therefore, the material surface, through its distribution and functionality and hydrophilic/hydrophobic balance [
4], is the main factor that determines the biological response and, consequently, its biocompatibility [
5]. In this context, polysulfones (PSFs) are known as high performance polymers with an excellent film-forming potential, great thermal stability, and an excellent resistance to inorganic bases and acids [
6,
7,
8]. Besides these, other important and interesting properties of some types of PSFs, such as the transparency, hydrolytic stability, and resistance to heat, are relevant [
5]. Due to its high hydrophobicity which causes an increase in the hydraulic resistance to water flow, a decrease in the water permeability and restrains the practical inclination in water treatment, there are some /researches in progress concentrated on changing the polysulfones in a way to provide a proper balance between hydrophobicity and hydrophilicity with the aim to make these materials suitable for needed applications.
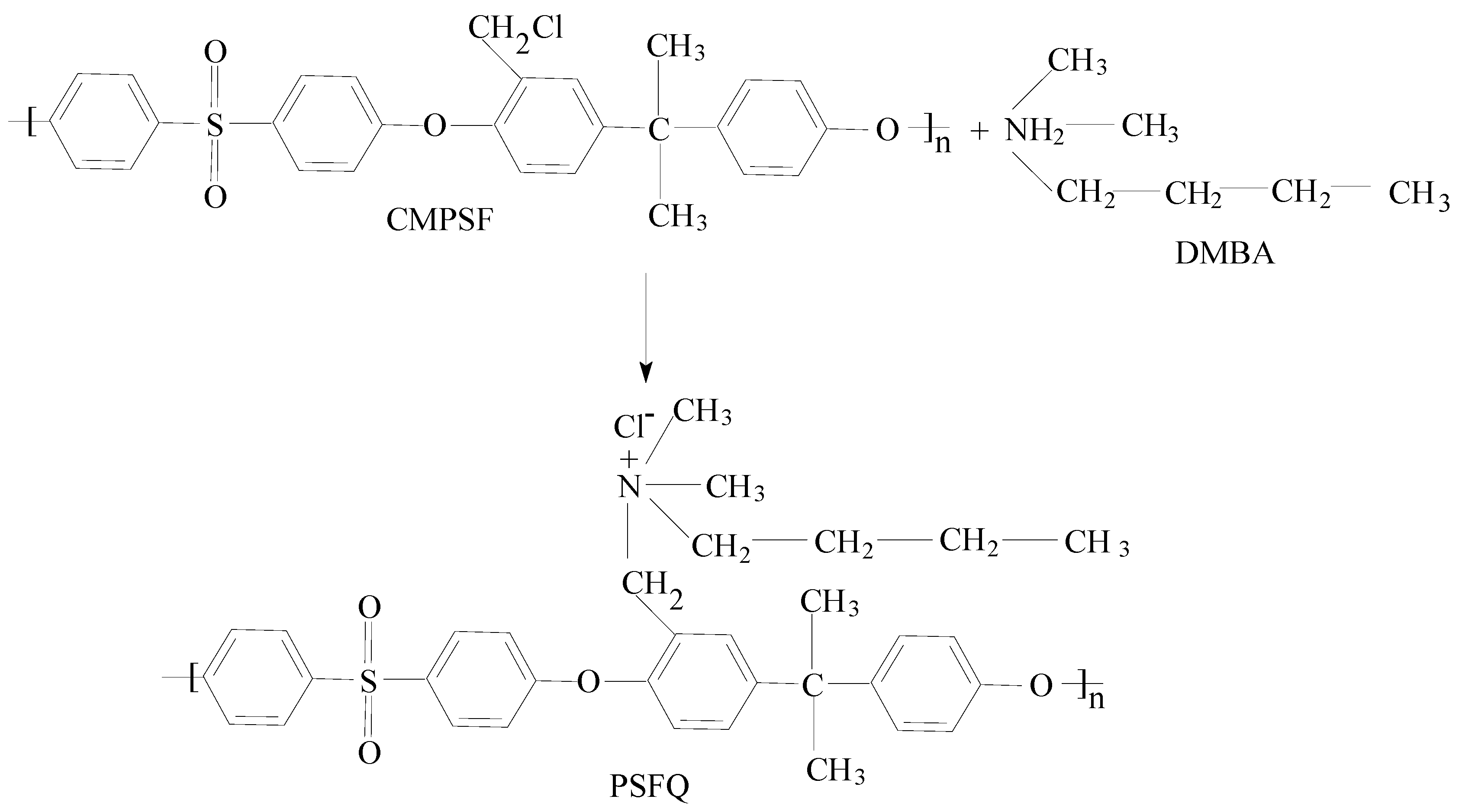
To meet these requirements, the structural modification of polymeric materials is essential. On the basis of the knowledge acquired from the structure–property relationships, a variety of polysulfones (PSFs) have been molecularly designed through the chloromethylation and quaternization reactions of PSFs [
9,
10,
11,
12]. Under these conditions, the functionalized PSFs membranes have not only advantages, e.g., antimicrobial, environmental, biocompatibility properties, but also disadvantages such as the fact that they behave as an obstacle in the separation process. This inconvenience can be surpassed by blending, thus improving in this way the water permeability and antifouling properties. Additionally, the added plasticizer improves the surface properties, the workability and membranes performance. Consequently, for an enhanced performance, design of blends/composites based on PSFs/functionalized PSFs using biologically inert and nontoxic compounds is preferred. The composite membranes consist of two materials with different characteristics: one of materials gives physico-chemical resistance, while the second one gives selectivity. In this context, the polysulfone-based composite membranes can be defined as an interphase limit-barrier which narrows the transport of various chemical species, in a selective way. From this perspective, in order to modify the inherent properties of PSFs/functionalized PSFs, conventional biodegradable polymers, such as cellulose and its derivatives (e.g., cellulose acetate phthalate (CAP)), polyvinyl alcohol (PVA), and polyesters have attracted attention and interest [
13,
14].
On the other hand, the surface modification has a relevant role in the biomedical applications due to the fact that the changes can help us to adjust the surface to the desired properties without making undesired changes in its bulk characteristics [
15,
16,
17]. The correlation of these modifications (chemical and/or physical) assure the biocompatibility of PSFs and, implicitly, the possibility of their action in a living organism or in contact with living system without negative consequences [
5].
In above-mentioned context, the literature indicates various studies concerning the synthesis and some properties of a series of quaternary ammonium-functionalized polysulfones [
18,
19,
20,
21]. Starting from the charged polysulfones with different ionic chlorine contents, their morphology and antimicrobial activity were evaluated [
5]. The study object regarding their applications in membrane technology reveals that surface properties are influenced by the history of the processing protocols/techniques and by the charge density of the quaternized polysulfones. In addition, the adhesion of the microorganisms to the modified polysulfones solutions is determined in connection with the hydrophobic/hydrophilic characteristics of the polymers and tested microorganisms [
22,
23]. On the other hand, the systems based on functionalized polysulfones with tertiary amine have been obtained by mixing the solutions of functionalized polysulfone with a well-controlled content of different polymers [
18]. These studies have led to development of an innovative way of designing the membrane materials with a long-term stable hydrophilicity, increased porosity, improved workability, and a very good biocompatibility. The obtained results represent the basis for future tests regarding their functionality as ionic-exchange membranes for hemodialysis [
24].
In addition to combining the polymers properties by mixing, to induce a surface pattern with enhanced features, the processing protocols/techniques are very important. Thus, the nanomaterials obtained using the electrospinning technique possess a high surface area to volume ratio and large number of inter/intra-fibrous pores. The most relevant applications are drug delivery, tissue engineering, biosensors, filtration, wound dressing, and enzyme immobilization. The nanofibrous frames made by this method mimic the nanoscale properties of native extracellular components and can be adjusted according to their use.
For this reason, unlike the previous studies, due to the bactericidal properties of functionalized polysulfone (PSFQ), the present study offers a development path of a new generation of composites, namely PSFQ/CAP and PSFQ/PVA blends/composites, projected as films and fibers to improve their performance, becoming biologically inert and nontoxic compounds, with stable mechanical, morphological and adhesion properties during their use. In this sense, due to their common characteristics, namely high hydrophilicity, flexibility, and good performances in film forming, which represent the important tools for improving and controlling the hydrophilicity and porosity of membranes, CAP and PVA were selected to be used in this study. Moreover, depending on their characteristics, it will be possible to choose the most efficient mixtures for uses in the biomedical field. Through this study, we follow the contribution/effect brought by each of the polymers added to the polysulfone matrix to improve the properties of the obtained active surfaces (membranes/films/fibers) designed to achieve multi-performance in bioinspired integrated system.
2. Results and Discussion
2.1. Surface Chemistry Analysis
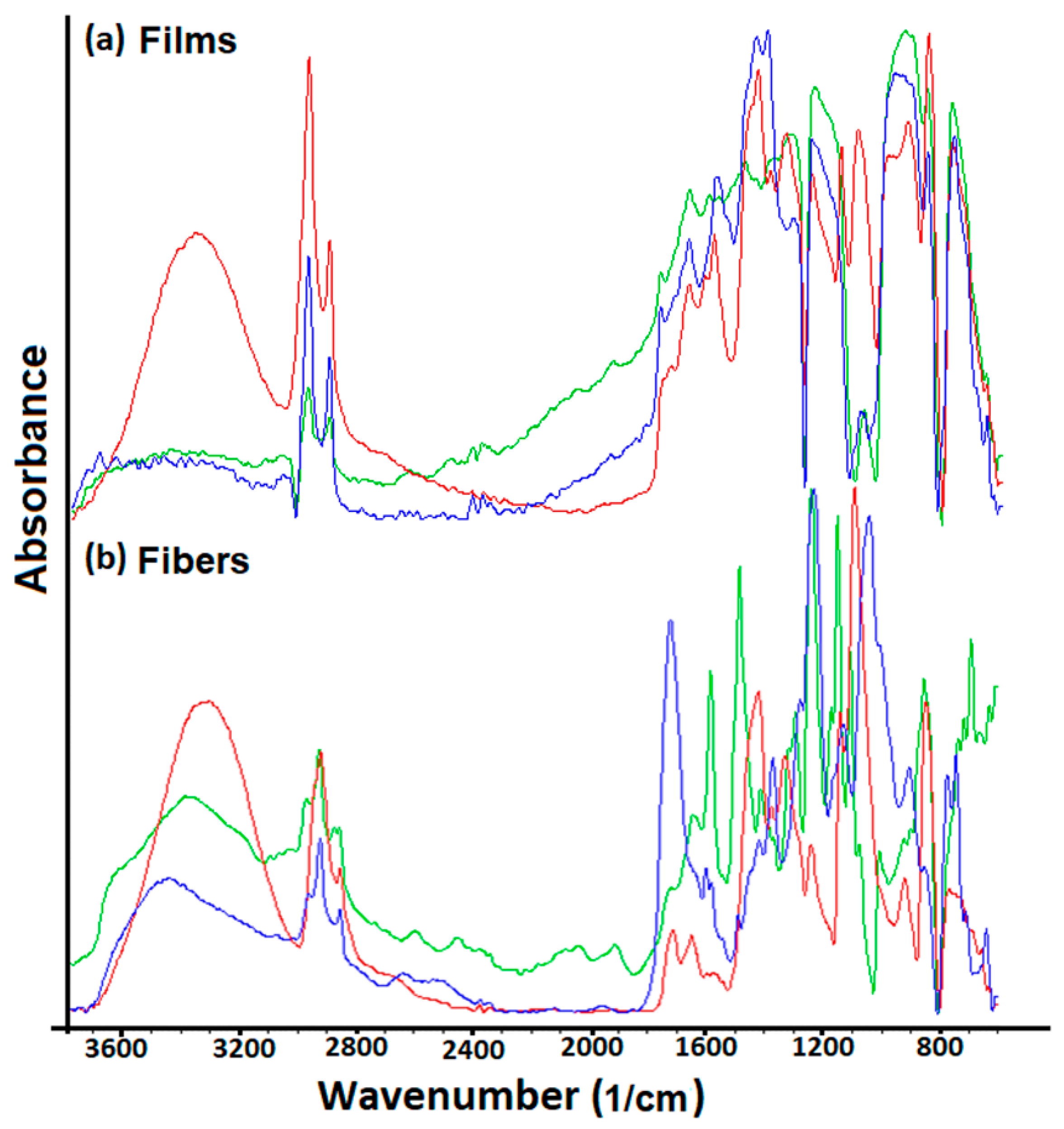
The spectroscopic methods are key tools in determining the presence of the functional groups on the polymeric surfaces (
Figure 1). Moreover, the inter- and intramolecular interactions due to the hydrogen bonding can be also determined through Fourier transform infrared spectra (FTIR) because these interactions affect the local electron density and a frequency shift could be observed [
25].
Based on previous works [
22,
26], the FTIR spectra indicated the characteristic bands for the polymers studied. Thus, the FTIR spectrum for the PSFQ has presented the absorption bands, corresponding to -SO
2 from the PSF backbone, around 1330 and 1300 cm
−1 as asymmetric stretching, while the strong absorption band around 1150 cm
−1 corresponds to the symmetric stretching. The aromatic structure was confirmed by the appearance of the absorption bands at approximately 1590–1410 cm
−1 and those from 2990–2950 cm
−1 and 2894–2850 cm
−1 domains; bands attributed to the vibrations of aliphatic units (–CH
3 and –CH
2, respectively) were presented. The quaternary ammonium group was confirmed by the appearances of stretching vibration peak at approximately 1635 cm
−1. On the other hand, in CAP spectrum, the following peaks were observed: around 2980 and 2883 cm
−1, absorption bands are attributed to the asymmetric and symmetric stretching vibrations of methyl –C-H groups; at about 1750, 1725, and 1701 cm
−1 the adsorption peaks are associated with the –C=O carbonyl group vibrations, while the –C=C– linkage of the aromatic ring showed a peak at about 1599 cm
−1. At 1492 and 1284 cm
−1 were found the characteristic bands of stretching vibrations of –C-H bond from methylene groups and –C-O-C– bond of the ester groups, respectively. Peaks from region 1140 to 1071 cm
−1 were related to the –C-O– stretching vibrations of cyclic ether structure. Analysis of FTIR spectrum for PVA showed that the large band between 3500 and 3200 cm
−1 was attributed to the O-H stretching vibrations and intermolecular hydrogen bonding vibration [
14]. Two bands with maximum at 2940 and 2910 cm
−1 are attributed to the asymmetric and symmetric alkyl C-H stretching vibrations. The absorption peaks at 1650 and 1570 cm
−1 correspond to C=O stretching and C-O stretching of acetate group, and the peak from 834 cm
−1 is related to the C–C stretching.
Generally, in the 3100–3500 cm
−1 range, there is a wide absorption band attributed to free hydroxyl stretching vibrations (not hydrogen bonded) and to those that are self-associated (hydrogen bonded) with the hydroxyl groups [
14,
26]. The latter is more intense than that obtained for the free ones; however, the hydroxyl stretching may be affected by the presence of eventual hydrogen bonding interactions. Additionally, the FTIR carbonyl-hydroxyl stretching range or flexion mode hydroxyl–hydroxyl interactions are sensitive to the formation of new interactions between the polymer’s blends. In these conditions, when one of the polymers from the blend predominates, the appearance of a new band located at higher/lower wavelengths is identified, as a consequence of the inter- and intramolecular hydrogen bonding interactions between different species [
14,
26]. Therefore, the FTIR spectra shown in
Figure 1 recorded over the 4000–600 cm
−1 range include/highlight all these statements, identifying the different structuration types generated, on the one hand, by the orientation of the functional groups of the polymers during the processing processes and, on the other hand, by the occurrence of some interactions between different functional groups of the involved polymers in the formation of blends as a function of the organization form, film and/or fibers. All changes on the characteristic peaks revealed a good miscibility and interaction between components in the matrix due to the formation of the inter- and intramolecular hydrogen bonding.
Additionally, the evaluation of the intensity of characteristic vibration peak of functional groups recorded in ATR mode and their displacements to lower or higher values can demonstrate how the functional groups are distributed during the processing, casting solutions and/or electrospinning, and also how they are subsequently stabilized in the films/fibers structure. Moreover, in the case of a mixture of two polymers, the diffusion of a flexible polymer chain (e.g., CAP and/or PVA) into host matrix (PSFQ) is followed by the destruction of the weak bonds, changing the strength of some bonds among the atoms of the host polymer. Thus, other new bonds are created, and finally, the structural rearrangement of the entire macromolecular system takes place.
In this context, the intensities of the peaks characteristic to the functional groups, as detected in ATR mode at wavenumber around of 1635 cm−1 (quaternary ammonium group for PSFQ), 3465 cm−1 and 1650–1750 cm−1, respectively (the hydroxyl groups and carbonyl stretching region, respectively, for CAP), and also the -OH absorption band for PVA at approximatively 3400 cm−1, are more pronounced in the case of polymers organized as fibers compared to those obtained for films, as a result of the functional groups orientation during electrospinning experiments. This tendency of changing in the films/fibers structure as a result of orientation/rearrangement of functional groups onto or inside of the surfaces is highlighted through the variation of peaks intensity.
Consequently, the evaluation and comparison of FTIR spectra recorded in ATR mode (ATR-FTIR) of the PSFQ, CAP, and PVA polymers in fiber and film form and their blends are needed to offer the right views on the behavior of the materials and performances of the membranes and fibers, respectively, for future applications in biomedical domain.
2.2. Rheological and Wetting Parameters
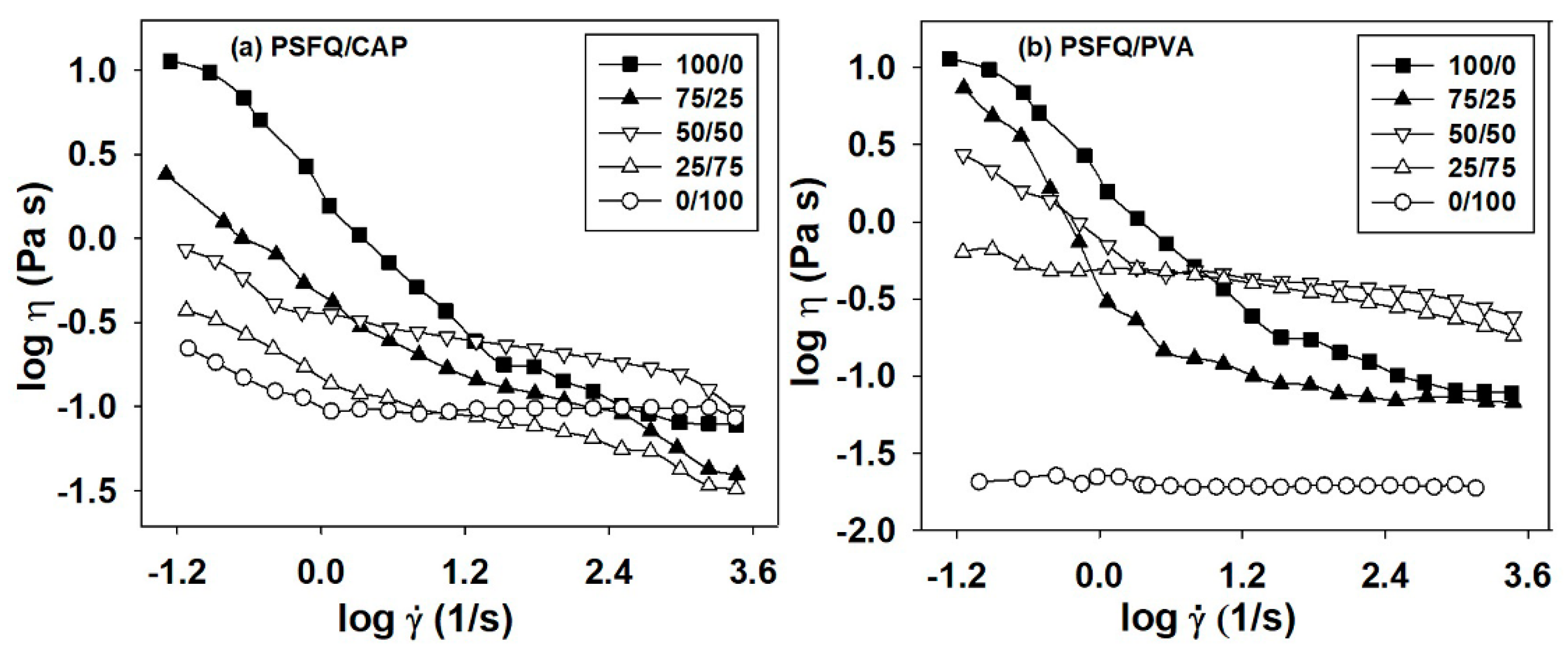
Rheological behavior of the polymer solutions plays an important role in defining the properties of the final products with applicability in certain areas, influencing the obtaining process of the films/fibers and, consequently, their surface properties. In this context, the rheological investigations of pure components (PSFQ, PVA, and CAP solutions in N-methyl-2-pyrrolidone, NMP) and of their blends, [
18,
26] in terms of the dynamic viscosity–shear rate dependence, where complex behavior appears as result of the polymers properties and blends composition that were previous analyzed (
Figure 2). Thus, the investigation of flow curves evidenced a shear thinning behavior at low shear rate, followed by a linear domain where the viscosity is constant (Newtonian plateau), when shear rate increases.
These results indicate significant changes in the PSFQ/CAP and PSFQ/PVA systems as result of the specific molecular rearrangement of the polymeric chains in blend, determined by the plasticizer effect of CAP and PVA in the PSFQ solution. Moreover, according to the literature studies on the mechanism of shear thinning [
27,
28,
29], the shear flow could reduce the probability of interchain association at the expense of intrachain association, thus leading to a lower viscosity (
Figure 2a). This behavior is due to the higher orientation of the polymer chains of the CAP compared to those of PVA in the PSFQ solution. Therefore, we can conclude that these blends possess properties of pseudoplastic materials, characterized by reduced entanglement density and enhanced number of the oriented segments, as result of shear rates increasing.
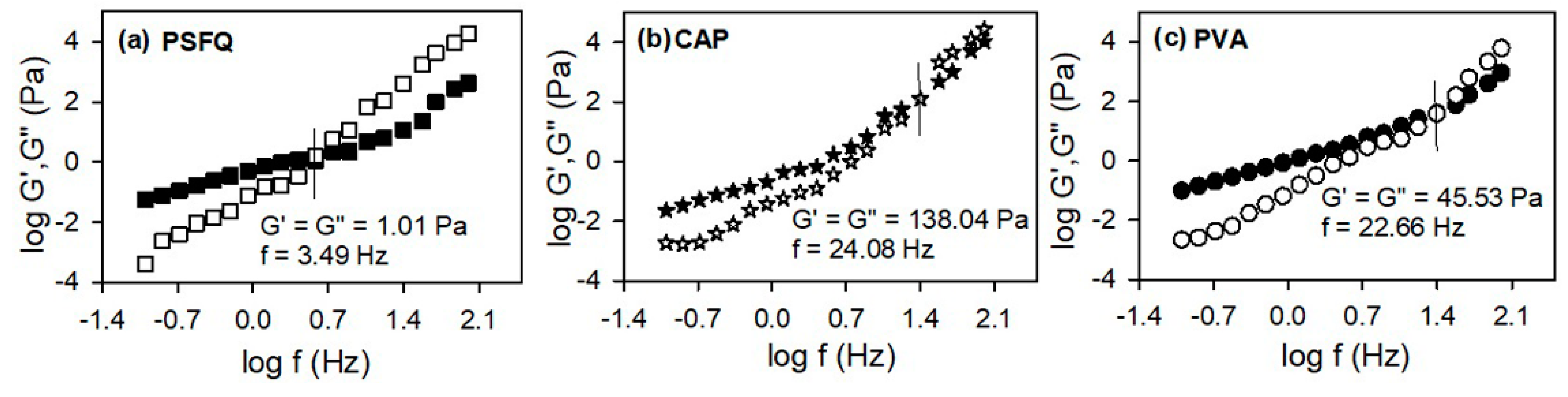
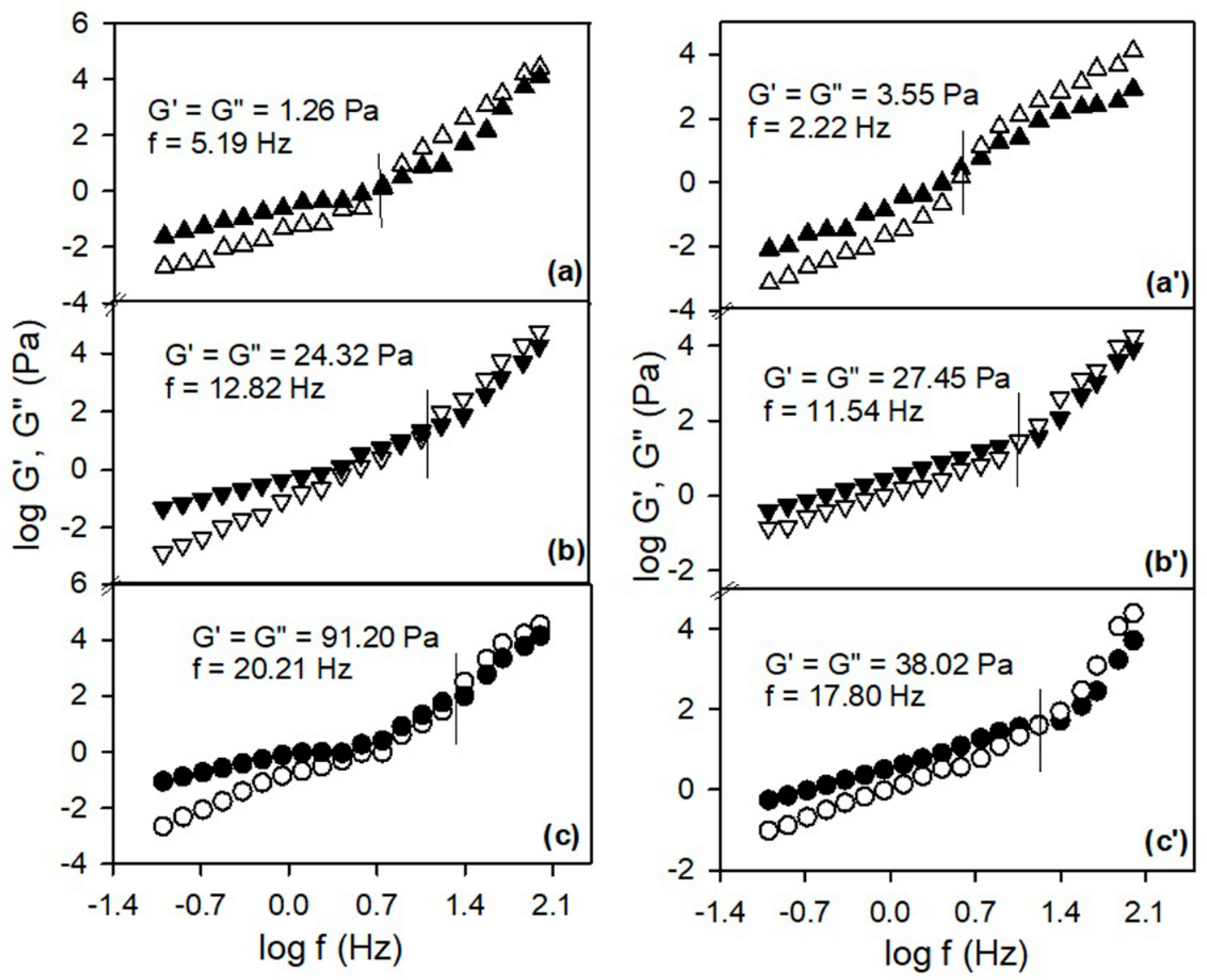
The above-mentioned findings concerning the influence of the structural characteristics of PSFQ, PVA, CAP and variation of the composition of the polymer blends on the chains mobility in the shear field was also confirmed by evaluation/analysis of the storage,
, and loss,
, moduli (
Figure 3 and
Figure 4). Thus, the rheological response for PSFQ, CAP, PVA pure polymers and their blends, at different compositions, over the low oscillatory frequency range exhibited a characteristic behavior typical for the viscous liquid, where
.
In addition, the overlap frequencies that delimit the viscous flow from the elastic one, occur at high frequencies, reflecting the influence of polymer chain flexibility and arranging the polymers in following order: PSFQ < PVA < CAP (
Figure 3). Therefore, the rheological behavior of PSFQ/PVA and PSFQ/CAP blends is influenced by specific interactions, like hydrogen bonding and association phenomena from the complex systems. In conclusion, as a result of the shear rates and overlap frequencies increasing, the rheological data were reflected in morphological changes induced by the PVA or CAP presence in PSFQ solution.
Successful application of a polymeric material in a biological environment is determined by its bulk and surface properties and is also is mediated by the combination of the solution and surface specific characteristics that are required for specific application [
4,
30]. These combined studies contribute to a better knowledge of the system interactions nature, and of the relation between the shear deformation and texture. Therefore, the surface of the biomaterial represents a key factor, determining the biological response and, therefore, the biocompatibility [
31]. Thus, properties such as the flexibility/stiffness, wettability, porosity, roughness of the biomaterial’s surface, are highly significant with respect to the existing physiological environment [
2].
It is known that the flexibility of polymers is closely related with their hydrophilicity, so besides the rheological studies, in order to design blends based on quaternized polysulfones as biomaterials, the interpretation of their surface characteristics and control of physical or/and chemical modificationsmust be taken into account. Generally, their biocompatibility is mainly affected by the surface characteristics, including hydrophilic/hydrophobic balance of the surface, distribution and functionality, and surface smoothness [
32]. Hence, the evaluation of the hydrophobic/hydrophilic balance is essential for studying the compounds used as biomaterials with different applications in biomedicine and biotechnology. In particular, as is shown in the rheological studies, the modification of surface functional groups—as a result of change in the polymer blends composition and polymers nature from the casting solutions (PSFQ/CAP and PSFQ/PVA)—leads to an increase in flexibility and workability of the films. Therefore, this modification plays an important role in biomedical applications because the changes can be used to adapt the surface to the desired characteristics without compromising its bulk properties. Thus, the effect of the different functional groups on the surface properties was evaluated from water contact angle measurements (
) by surface free energy,
, (Equation (1) using the total surface tension of water,
= 72.80 mN/m [
33])) which expresses the balance between the surface hydrophobicity and hydrophilicity.
As is known, the water contact angle () is an indicator of the wettability of the film surface [
5,
33], and the approach of the polymer surface energy allows us to correlate the surface tension parameters (
) with their chemical nature. Thus, the hydrophobic surfaces, with high contact angles, were converted into hydrophilic surfaces, with low contact angles, through the CAP or/and PVA addition in PSFQ matrix (
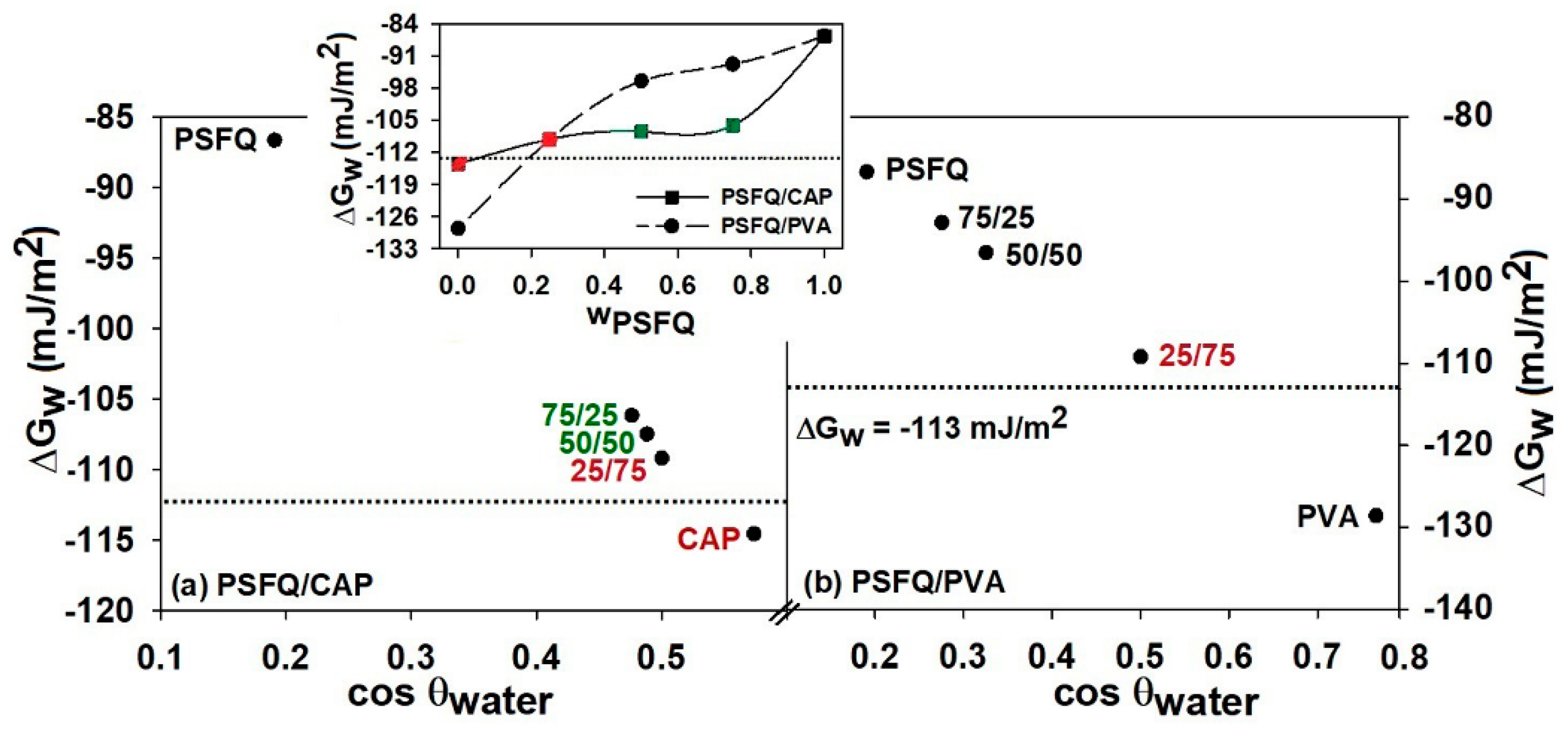
Figure 5). Thus, this versatility, in function of the final application requirements, is mainly attributed to the presence of a great number of functional groups (e.g., hydroxyl, carbonyl) which confer to PSFQ chain a high flexibility and hydrophilicity.
According to the literature, a polymer surface can be considered more hydrophilic for
mJ/m
2, whereas, when
mJ/m
2, it should be considered more hydrophobic. Taking into account these statements, the polymers’ nature and modification of the composition in the studied systems (PSFQ/CAP and PSFQ/PVA) determine a decline in the contact angles and an appropriate increase in their surface wettability compared with the PSFQ sample, caused by the addition of polar groups to the PSFQ matrix, which exhibit high hydrophilicity (see
Figure 5). The modification of the wetting characteristics of the studied surfaces can be caused by the polymer chains orientation and rearrangement of the polar groups on the film surface, after the blending process, due to the increase in the local polar moments. Moreover, analyzing the data obtained for the two studied systems, it can be stated that in the case of the PSFQ/CAP system due to the carbonyl and hydroxyl groups which increase the polarity, the surfaces have an improved hydrophilicity compared to PVA. However, we can conclude that the negative values of
reveal an increased wettability, implicitly a high hydrophilicity—a property useful for biomedical applications—of the films obtained from solutions based on PSFQ- blends.
2.3. Surface Morphology Analysis
As it was observed from the study in solution, the conformational modifications of PSFQ chains in polymers blends, generated by specific interactions are reflected in the topographic organization of the polysulfonic films induced by the nature and content of the added polymers. Therefore, supported by literature [
4,
22,
30], our results reported that the chain shape of a polymer in solution could affect the polymer morphology in bulk. Thus, in order to understand the effects induced by the presence of CAP or/and PVA in the polysulfonic matrix, a comparative analysis on the film surface aspects for PSFQ, PVA, CAP pure polymers and their blends is necessary. The addition CAP or PVA—which are flexible and hydrophilic polymers, namely to the polysulfonic matrix as plasticizers—improves the material properties by the interactions that it generates, such as intermolecular hydrogen bonds, and, consequently, changes the structure of the polymer both at molecular and morphological level.
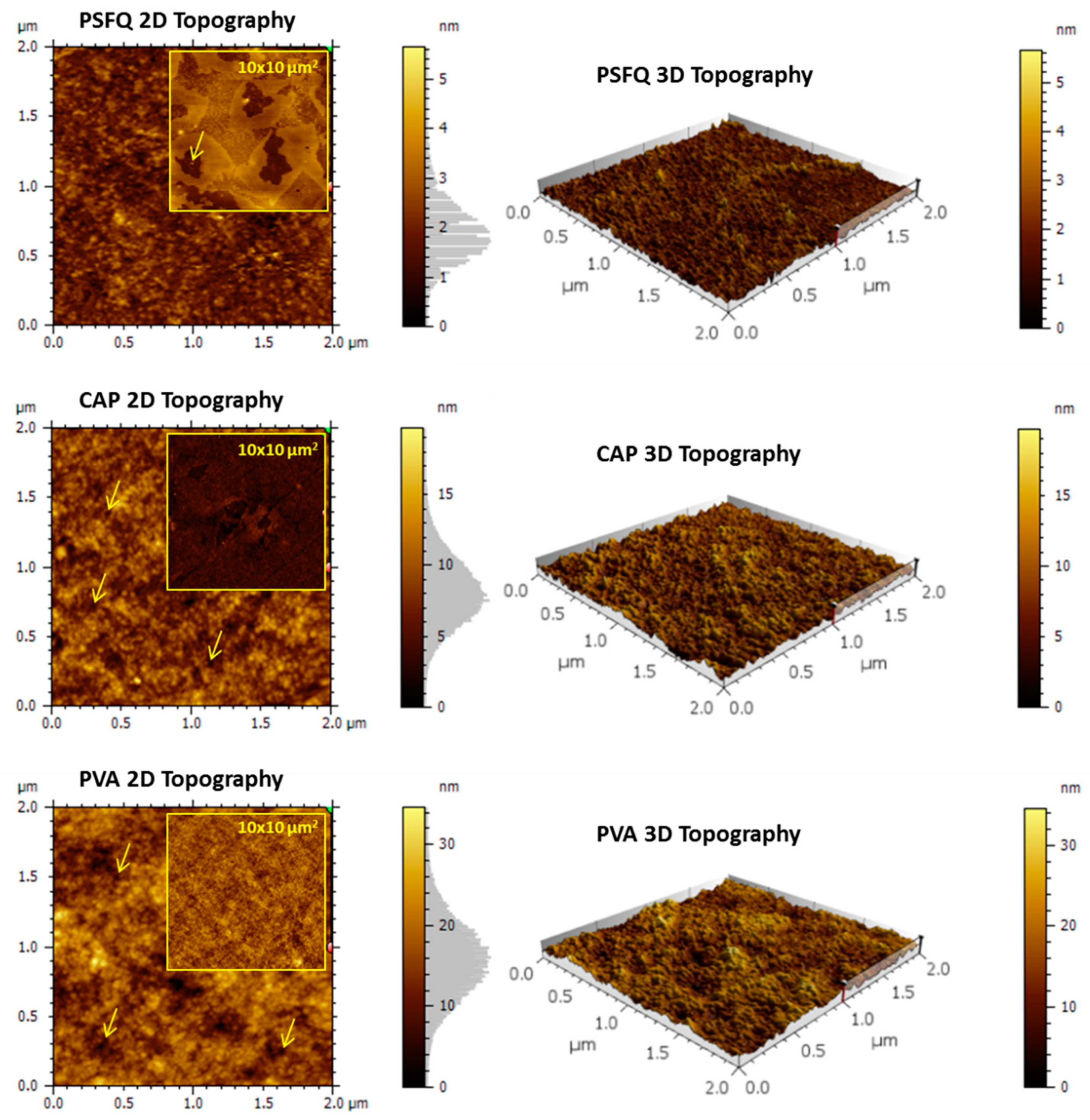
The morphology dictated by the roughness and the existence of porous formations are determining factors in order to obtain high-performance surfaces used in the biomedicine. In this context, atomic force microscopy (AFM) investigations of the films obtained from PSFQ- based blends can clarify the polymer influence, namely CAP or/and PVA, on the morphological properties, as well as its ability to improve the film-forming characteristics (
Figure 6 and
Figure 7). In this context, according to behavior evidenced by rheological measurements, the CAP or/and PVA added in different amounts acts as a plasticizer, generating an increase in the flexibility/workability of the films. Moreover, in accordance with data obtained through contact angle measurements was found that the addition of a large amount of CAP or/and PVA to PSFQ matrix (compositions containing less than 50 wt.% PSFQ in the blends) has conducted an increase in surface hydrophilicity. Therefore, the expected effect, to introduce modifications at the nanoscale in PSFQ matrix, is given by the structural and compositional characteristics of the other two polymers (CAP and PVA) used as hydrophilic additives and pore-forming agents. For this reason, the chosen films for further investigations were those containing high content of CAP or PVA, namely 25/75 wt./wt. compositions of the PSFQ/CAP or PSFQ/PVA blends, respectively (
Figure 7). On the other hand, considering the excellent characteristics of CAP (e.g., a high chemical reactivity, the flexible structure of the polymer chain, and high hydrophilicity due to hydroxyl, carbonyl groups, etc.), the blends based on PSFQ—PSFQ/CAP—would bring new benefits to those already known from the literature, leading to materials with improved properties suitable for targeted applications.
Analysing the texture of the samples recorded by 3D and 2D AFM images, different morphologies are observed (
Figure 6 and
Figure 7) highlighting that the nature and composition of the polymers incorporated in the PSFQ matrix (CAP and/or PVA) are responsible for the formation of areas with pores and grains of different sizes and densities. In accordance with the above mentioned, the AFM image recorded for PSFQ (
Figure 6) indicates the appearance of irregular, porous formations, randomly disposed in some areas, (the average diameter of these pores being of 2.16 ± 0.28 μm). Instead, in the case of CAP and PVA samples (
Figure 6), the AFM images show a well-balanced surface relief with a nanoporous structure aspect (hardly observable on the 10 × 10 μm
2 areas—inserted images). This change is in accordance with the previous statements regarding the flexibility and hydrophilicity of the samples. The average diameter of the nanopores detected and calculated using the Pore Analysis Module was 56 ± 17 nm for the CAP sample and 78 ± 15 nm for the PVA sample. Some of them are evidenced using arrows in
Figure 6.
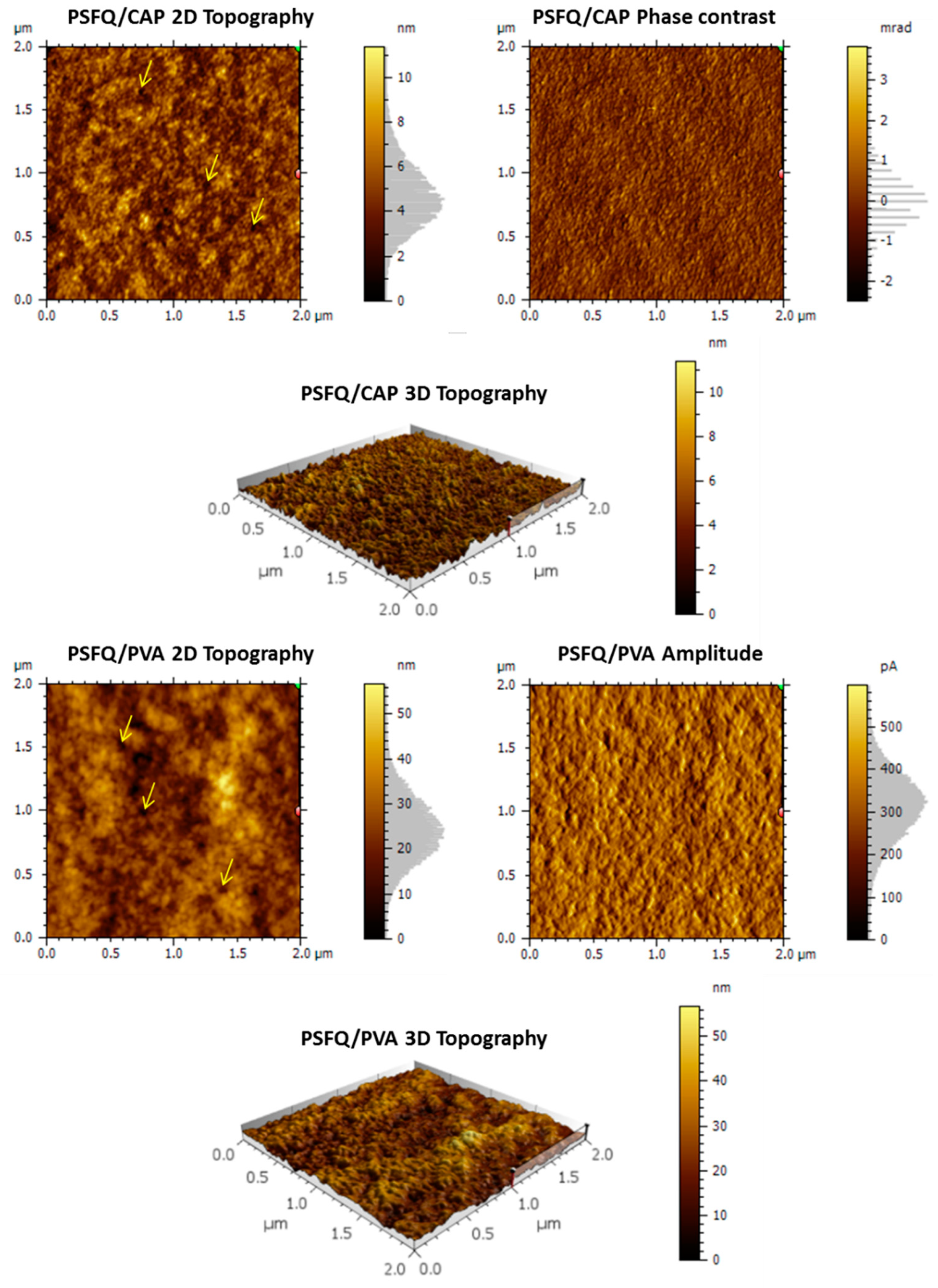
On the other hand, when the PSFQ matrix is modified with a certain amount of plasticizer (i.e., 0.75 weight fractions of CAP or PVA), the surface of films obtained from PSFQ/CAP or PSFQ/PVA blends, respectively, become more complex (see
Figure 7). Increasing the CAP or PVA content generates an increase in the number of nanopores but with different characteristics, namely the sizes being of 63 ± 12 nm for 25/75 wt./wt. composition of PSFQ/CAP blend compared to 101 ± 21 nm for 25/75 wt./wt. composition of PSFQ/PVA blend. This changing trend in film morphology coincides with the shear thinning behavior, evidenced from rheological data, as an effect of the conformation modification of polymeric chains in solution. The phase contrast and amplitude images presented in
Figure 7 indicate that there is no phase contrast (phase separation) is presented at this scale, the phase shift/amplitude shift being due only to the differences in the surface relief heights. Moreover, the microstructural organization of the studied films is related to the surfaces wettability property as result of the chains orientation effect and polar group’s rearrangement on the films surface.
The same changes in the texture and morphology of the polymer films can be seen also in the SEM images (
Figure 8a–c). Thus, the SEM images for the PSFQ sample (
Figure 8a) show a material with pores at the surface of the polymer, whose walls are more contoured. The SEM image for pure PVA film (
Figure 8c) displays a more compact structure, which can confirm the presence of a homogenous surface due to the polymer characteristics, with no clearly visible pores. Instead, SEM images for CAP film (
Figure 8b) show a homogenous surface with layered microparticles in the form of rectangles with variable sides between 0.5 and 4 microns, with well-defined roughness as wavy nano ripples (20–50 nm average roughness) developed on the surface of each microparticle.
On the other hand, the electrospinning of PSFQ, CAP, and PVA solutions gave continuous cylindrical and thin fibers that were defect-free and randomly-oriented with inter-related voids among the fibers (
Figure 8a’–c’). The examination of SEM micrographs revealed that PSFQ fiber (
Figure 8a’) present loose smooth fibers with silky ribbon-like shape, with the width around 1 micron and average length over 150 microns. The surface of the PSFQ fibers is homogeneous with no roughness. Meanwhile, the CAP fiber (
Figure 8b’) consists of loose fibers with overall tubular structure with the fiber radius of around 200 nm and average fiber length over 50 microns. The surface of the fibers shows roughness with wave-like ripples perpendicular to the fiber axis. As can be seen in the
Figure 8c’, the PVA fiber consists in close-layered fibers with the fiber radius of around 200 nm and average fiber length over 100 microns. The surface of the fibers shows homogeneous aspect with no roughness. The differences between the morphology and polymers form have a relevant contribution to their behavior as biomaterials.
Following the studies performed, we can conclude that the components are stabilized in the structure of the obtained films and fibers and also considering the superior properties acquired, namely improved flexibility, hydrophilicity, and specific molecular microarchitecture—properties required by biomedical applications. Studied materials can be recommended as surfaces/porous scaffolds with expected future developments in the biomedical fields.
2.4. Water Uptake Ability and Water Vapour Sorption Behavior
Adsorption isotherms are relevant for modeling, simulating, and optimizing of the studied systems in practical applications. In this context, considering the shape of the water sorption curves presented in
Figure 9 and
Figure 10, these can be associated with isotherms of Type IV, according to the IUPAC classification. This type of isotherm with hysteresis can be interpreted as being specific to the porous surfaces, providing information about the samples surface. Therefore, the following occurs: a reduced water vapor sorption capacity at low values of relative humidity, RH (0–10%), moderate sorption at intermediate values of RH, and a high increase of water vapour sorption at RH values close to 100%. The values of the surface parameters determined from adsorption/desorption isotherms for all studied samples are summarized in
Table 1.
The samples required an increased equilibrium time (30/60 min) for correct registration. The equilibrium is becoming more difficult to reach, especially for higher humidities, due to the possible cleavage of the physical bonds in the sample. In this condition, the surface characteristic properties are given by the sigmoidal shape and the presence of the hysteresis. They are reflecting adsorption via mono-multilayer (mainly multilayer) and capillary condensation in pores. The hysteresis loops and water vapour uptake levels are strongly influenced by the hydrophobic/hydrophilic balance modification. Moreover, the swelling and water vapour sorption behavior depend on material structure (composition, water affinity related to the presence of polar groups), surface area, morphology (topography and porosity, pores sizes and shape). The pores sizes and shapes, as well as the presence of polar groups with water affinity, are important in moisture sorption behavior of the samples. Thus, there are not high differences between the porous, macroporous and dense 3D structures (
Figure 9 and
Figure 10).
To appreciate the specific surface area (
Table 1), Brunauer–Emmett–Teller kinetic model (BET, Equation (2)) was applied by modeling the sorption isotherms registered under dynamic conditions.
where the parameters involved are as follows: weight of sorbed water—
; weight of water forming a monolayer—
; sorption constant—C, relative humidity—
.
The BET model describes the sorption isotherms up to a relative humidity of 40%, depending on the type of sorption isotherm and on the type of material. BET model can characterize the isotherms of types II but also types I, III and IV. The nature of the functional groups of the polymers from the system influence the values of water vapour capacities and, as a consequence of this fact, influence also the specific surface area and the average pore size of the studied samples.
By applying the Barrett, Joyner and Halenda model (BJF, Equation (3)), based on calculation methods for cylindrical pores, the average pore size,
, (
Table 1) was determined using the desorption branch of the isotherm. The desorbed amount of vapor is due either to the evaporation of the liquid core or to desorption of a multilayer. Pore size distribution is outlined as the distribution of pore volume. The relationship between pore volume and moisture uptake can be defined knowing the density of the adsorbed phase. The first assumption of mesopore size analysis is that this phase is equivalent to the liquid phase of the adsorbate.
where W is absorption percentage,
is the adsorbed phase density, and A is specific surface area evaluated by BET method.
Investigations on studied surface, based on the water vapor sorption data, have demonstrated that on their surface exist nanosized pores whose number varies from sample to sample, as a function of the structural peculiarity of polymers and the number of polymers in the blend. It is notable the porogen effect of the CAP and PVA which generates structures with a high specific surface area and small pore size into the polysulfone matrix. That is why it is necessary to provide a high pore density and, consequently, a high permeability. On the other hand, analyzing the results, values of the water vapors’ sorption capacities and surface characteristics for studied fibers increase compared to the ones corresponding to the films. This tendency of changing as a result of orientation/rearrangement functional groups (highlighted by ATR-FTIR data) is consistent with the topographical reorganization of the films/fibers, revealing that the specific molecular rearrangements produced in the system through modification of the mixing ratio of polymers influence the surface properties. Consequently, it can be stated that the surface properties of the materials are those that dictate their functionality, performance and implicitly their biological ability, and not their specific surface.
2.5. Testing the Antibacterial Capacity
One of the important issues in the medical field is the obtaining of the porous scaffolds with specific physical and biological properties. In this context, materials used for applications in this domain must be designed to stimulate the specific cell response at the molecular level, inducing specific attachment, proliferation, and differentiation of cells. In addition to cell–material interaction, the antibacterial properties also play an important role in the medical field. From this perspective, the selection of the biomaterials constitutes the most important factor for the success of practical applications in health and medicine field. Thus, it is well known that some positively charged polymer materials present antibacterial properties because they kill the bacteria by disrupting the negatively charged cell bacteria membrane (containing phosphatidylethanolamine as the major component) as result a of their charge. In this regard, the functionalized polysulfones with quaternary ammonium groups (PSFQ) are the most explored kind of polymeric biocide that offers some advantages including good antibacterial activity, reactivity, films and fibers forming capacity, and favorable hydrophilicity [
26,
34].
On the other hand, although a single-component polymer material cannot satisfy all requirements, the design and preparation of the multicomponent polymer systems constitute a viable strategy, assuring the progress in the multifunctional biomaterials domain. Therefore, the modification procedures increase hydrophilicity and porosity of surface, having an important effect on the flux and fouling reduction, improving the antibacterial activity and blood compatibility. In this context, the use of polymeric materials with antimicrobial properties, namely PSFQ, CAP and PVA, gains an increasing interest from both academic and medical perspectives. In particular, the antibacterial activity of PSFQ, CAP and PVA, and their blends (70/30 wt./wt. composition of PSFQ/CAP and PSFQ/PVA blends) using
Escherichia coli (
E. coli) and
Staphylococcus aureus (
S. aureus) microorganisms in terms of the inhibition zone diameter, was evaluated (
Figure 11). The obtained results indicate the presence of well-defined inhibitory zones, the aspect which demonstrates that these materials have antibacterial activity against tested microorganisms.
Knowing from the literature the nature of the tested microorganisms as a result of the cell walls components, namely hydrophilic
E. coli and hydrophobic
S. aureus [
35], we found that
E. coli is more sensitive to the investigated materials than
S. aureus. On the other hand, it should be noted that the inhibition effect of the tested samples on the
S. aureus and
E. coli microorganisms are slightly different because all polymers present hydrophilic character. Thus, considering the hydrophilic character of the studied samples (hydrophilicity of tested samples varying in the order of PSFQ < PVA < CAP), the inhibition of
E. coli to PSFQ is slightly pronounced comparatively with the inhibition for PVA and CAP (values of the inhibition zone diameter being 23 mm for PSFQ > 22 mm for PVA > 20 mm for CAP). Moreover, the analysis of the inhibition zone diameters measured against to
E. coli and
S. aureus for studied blends (70/30 wt./wt. composition of PSFQ/CAP and PSFQ/PVA blends) indicates that the character of pure polymers gives the blends the same variation on antimicrobial activity.
Additionally, we mention that the obtained results are consistent with the experimental data previously presented [
26] for the PSFQ and CAP samples at concentrations varying between 0.5 and 2 g/dL, maintaining the same inhibition trend. Consequently, antibacterial efficacy of the studied materials, using
Staphylococcus aureus and
Escherichia coli microorganisms, was demonstrated, and the obtained data indicate the potential for their application in health and medicine fields.