Preparation and Characterization of Plasma-Derived Fibrin Hydrogels Modified by Alginate di-Aldehyde
Abstract
1. Introduction
2. Results and Discussion
2.1. Alginate-di-Aldehyde Preparation and Characterization
2.2. Gelation Time and Kinetics of Fibrin/ADA Gels
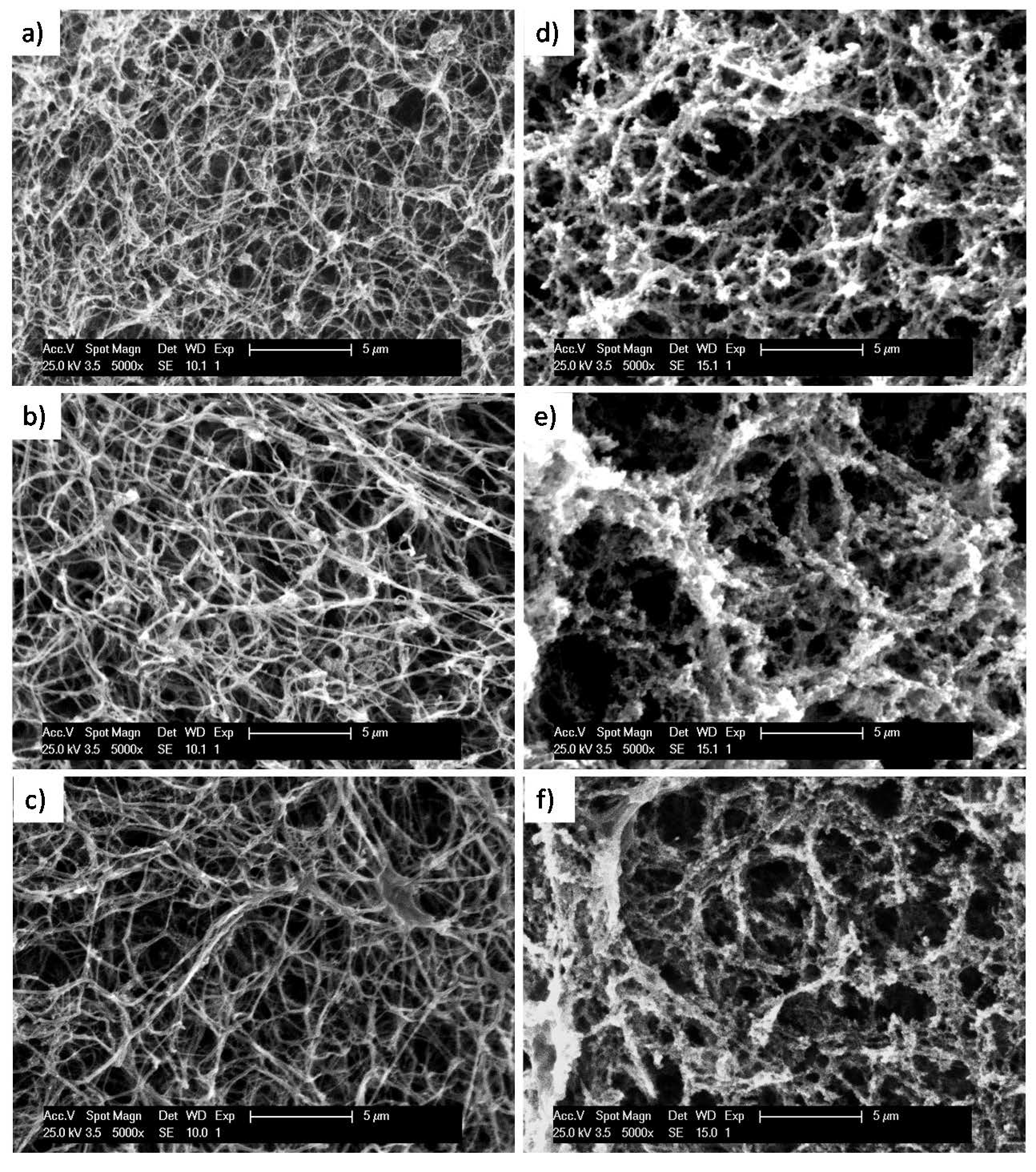
2.3. Microstructure of Fibrin/ADA Hydrogels
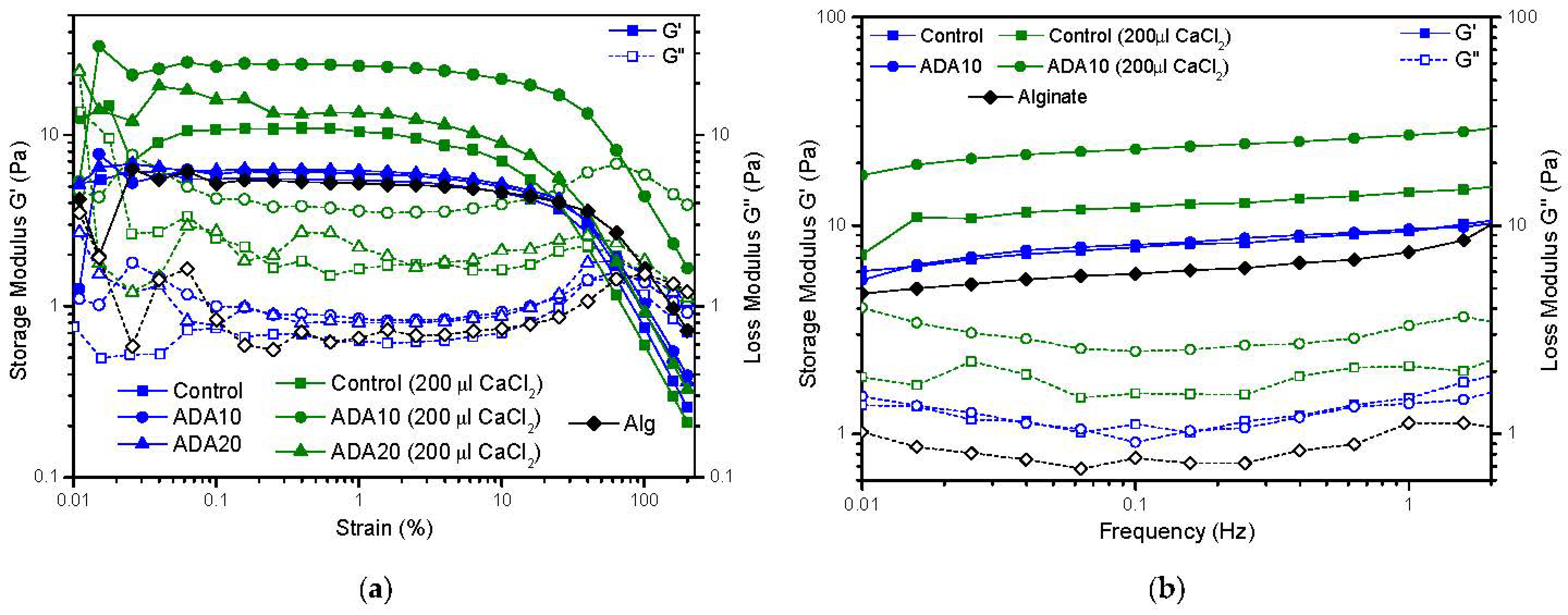
2.4. Rheological Characterization
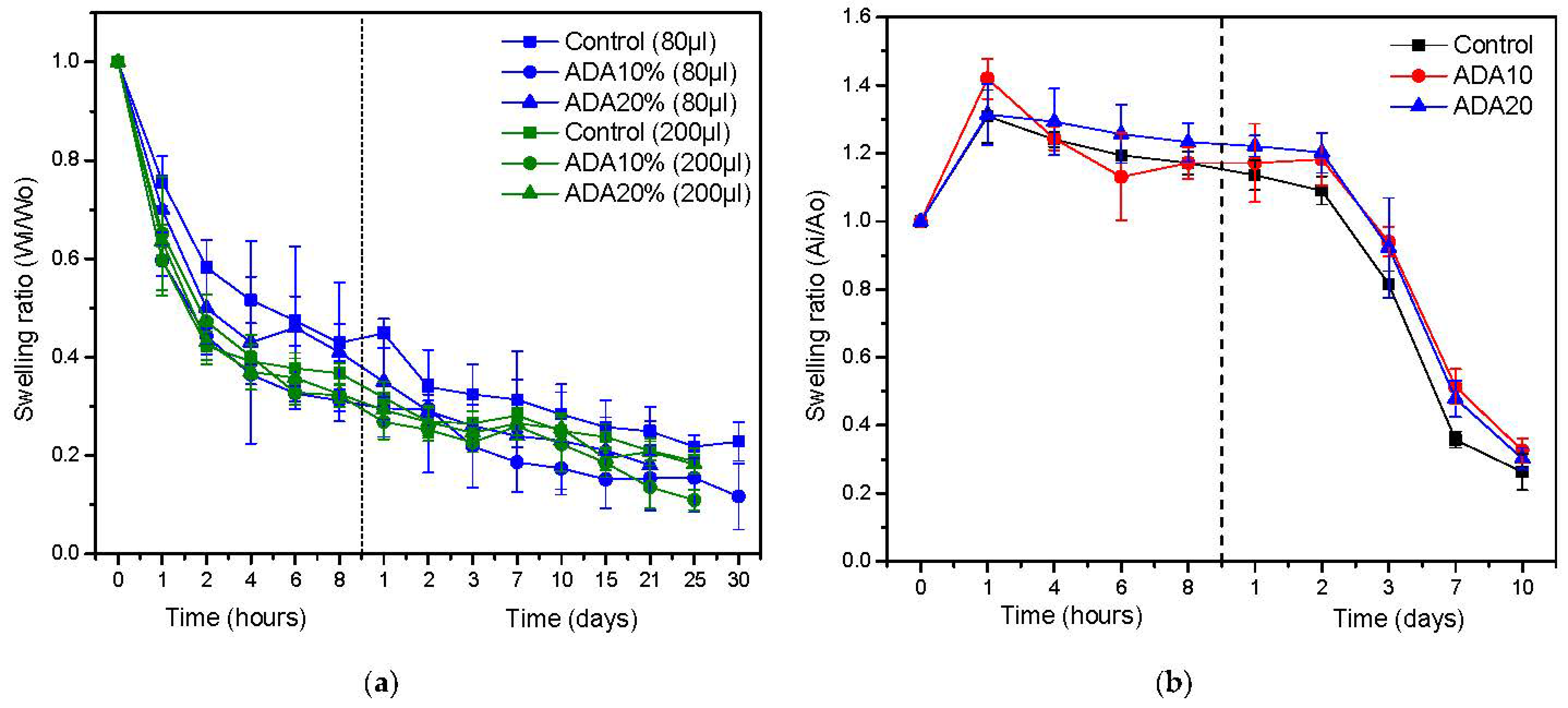
2.5. Fibrin/ADA Hydrogel Contraction
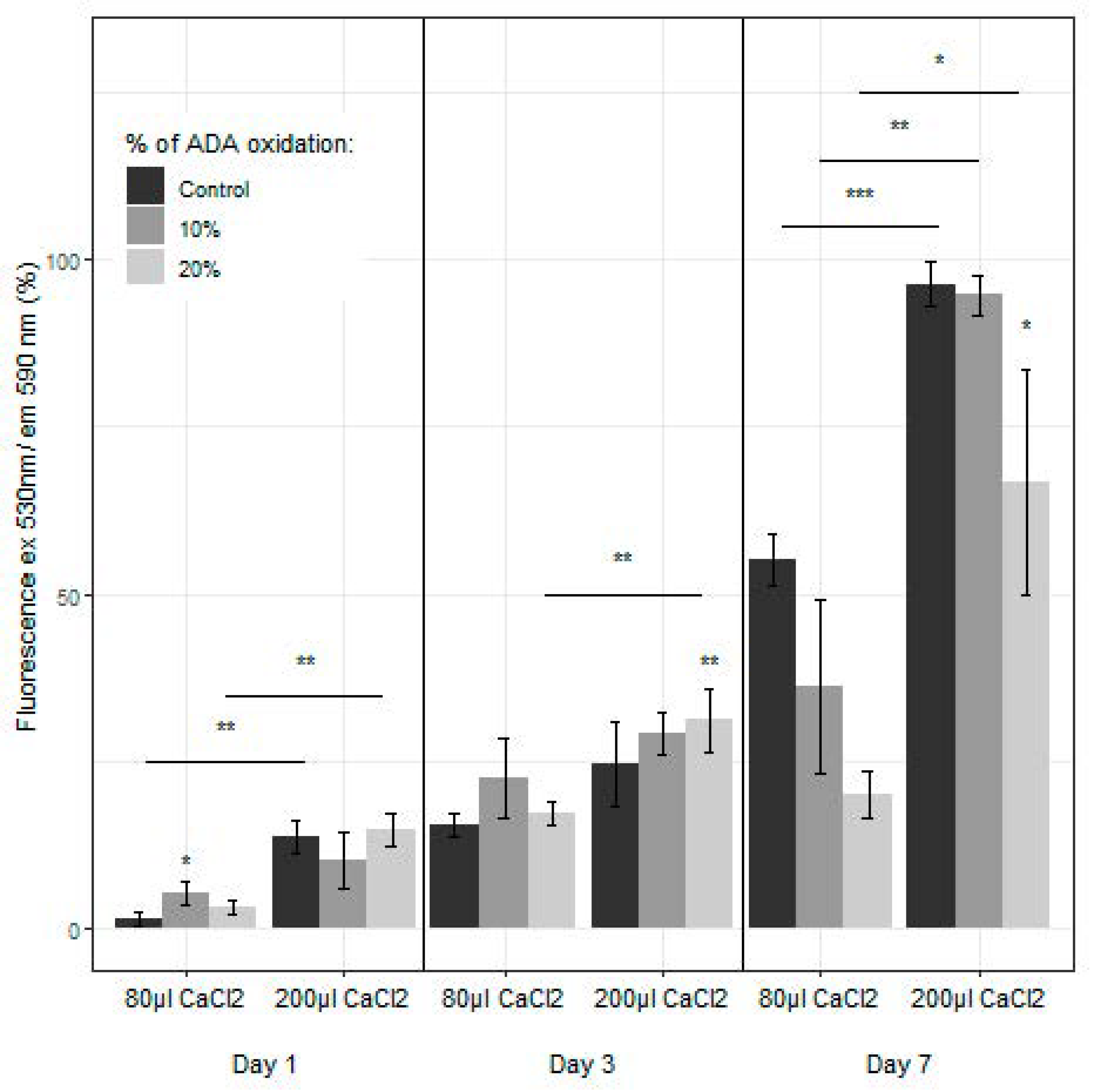
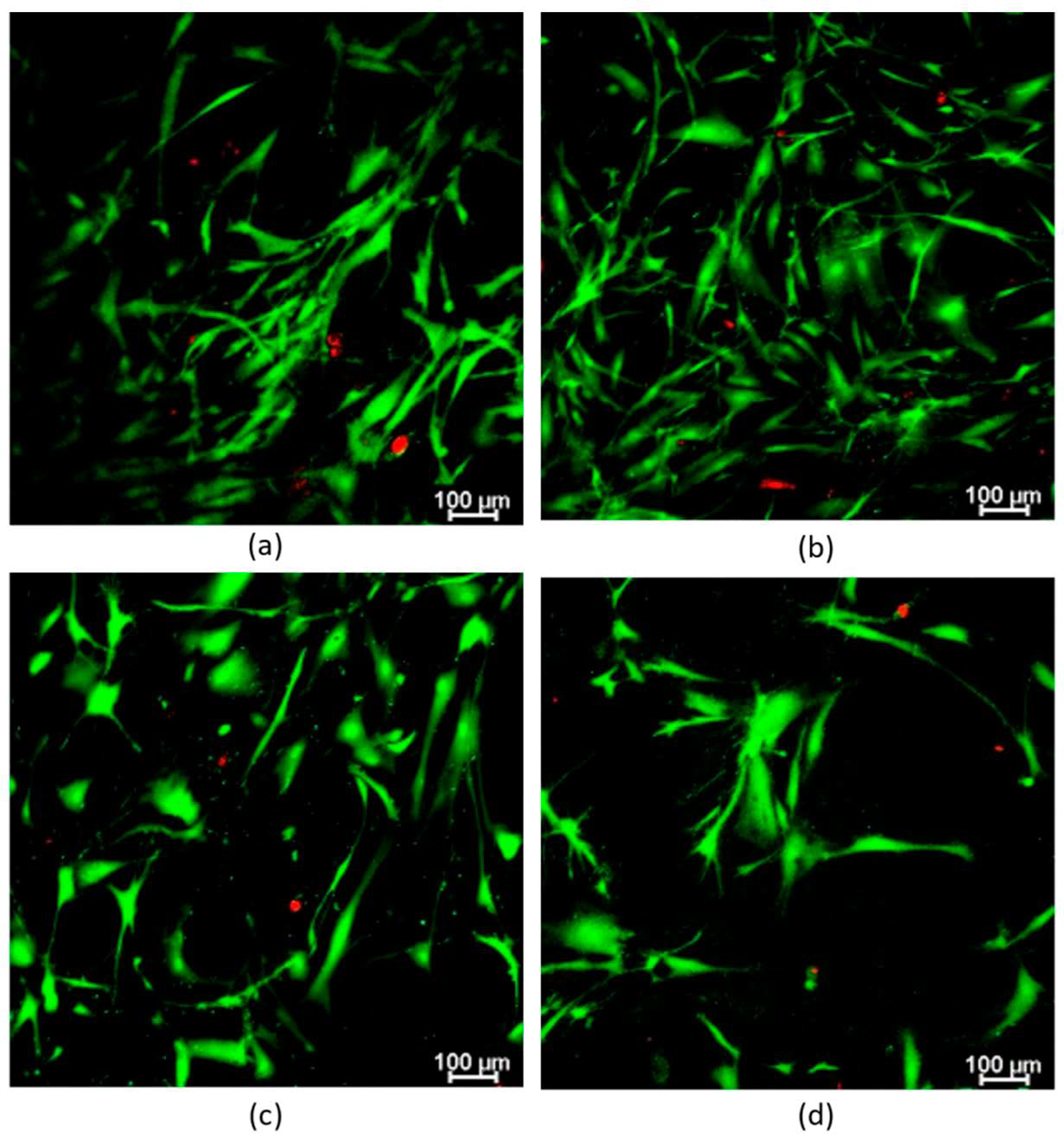
2.6. Proliferation and Live/Dead Assays: Encapsulated hFBs
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Alginate di-Aldehyde
3.3. Aldehyde Characterization
3.4. Proton Nuclear Magnetic Resonance
3.5. Characterization by FTIR-ATR (Fourier Transform Infrared Spectroscopy-Attenuated Total Reflection)
3.6. Thermogravimetric Analysis (TGA)
3.7. Gel Permeation Chromatography (GPC)
3.8. hFBs Culture
3.9. Fibrin/Alginate di-Aldehyde Hydrogel Preparation
3.10. Gelation Time and Kinetics
3.11. Microstructure Characterization of Fibrin/Alginate di-Aldehyde Hydrogels
3.12. Rheological Characterization
3.13. Contraction of FAD Hydrogels
3.14. hFB Proliferation Assay
3.15. Viability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vacanti, J.P.; Langer, R. Tissue engineering: The design and fabrication of living replacement devices for surgical reconstruction and transplantation. Lancet 1999, 354, 32–34. [Google Scholar] [CrossRef]
- Griffith, L.G.; Swartz, M.A. Capturing complex 3D tissue physiology in vitro. Nat. Rev. Mol. Cell Biol. 2006, 7, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface 2006, 3, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Marchant, R.E. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev. Med. Devices 2011, 8, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Holland, T.A.; Mikos, A.G. Review: Biodegradable Polymeric Scaffolds. Improvements in Bone Tissue Engineeringthrough Controlled Drug Delivery. In Tissue Engineering I; Lee, K., Kaplan, D., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 161–185. ISBN 978-3-540-31948-1. [Google Scholar]
- Gunatillake, P.A.; Adhikari, R.; Gadegaard, N. Biodegradable synthetic polymers for tissue engineering. Eur. Cells Mater. 2003, 5, 1–16. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed]
- Rowley, J.A.; Madlambayan, G.; Mooney, D.J. Alginate hydrogels as synthetic extracellular matrix materials. Biomaterials 1999, 20, 45–53. [Google Scholar] [CrossRef]
- Reakasame, S.; Boccaccini, A.R. Oxidized Alginate-Based Hydrogels for Tissue Engineering Applications: A Review. Biomacromolecules 2018, 19, 3–21. [Google Scholar] [CrossRef]
- Baniasadi, H.; Mashayekhan, S.; Fadaoddini, S.; Haghirsharifzamini, Y. Design, fabrication and characterization of oxidized alginate-gelatin hydrogels for muscle tissue engineering applications. J. Biomater. Appl. 2016, 31, 152–161. [Google Scholar] [CrossRef]
- Park, C.H.; Woo, K.M. Fibrin-Based Biomaterial Applications in Tissue Engineering and Regenerative Medicine. In Biomimetic Medical Materials: From Nanotechnology to 3D Bioprinting; Noh, I., Ed.; Springer: Singapore, 2018; pp. 253–261. ISBN 978-981-13-0445-3. [Google Scholar]
- Geer, D.J.; Swartz, D.D.; Andreadis, S.T. Fibrin promotes migration in a three-dimensional in vitro model of wound regeneration. Tissue Eng. 2002, 8, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Sproul, E.; Nandi, S.; Brown, A. Fibrin biomaterials for tissue regeneration and repair. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Woodhead Publishing: Swaston, UK, 2017; pp. 127–150. ISBN 9780081008522. [Google Scholar]
- Ahmed, T.A.E.; Dare, E.V.; Hincke, M. Fibrin: A versatile scaffold for tissue engineering applications. Tissue Eng.-Part B Rev. 2008, 14, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Noori, A.; Ashrafi, S.J.; Vaez-Ghaemi, R.; Hatamian-Zaremi, A.; Webster, T.J. A review of fibrin and fibrin composites for bone tissue engineering. Int. J. Nanomed. 2017, 12, 4937–4961. [Google Scholar] [CrossRef] [PubMed]
- Mol, A.; Van Lieshout, M.I.; Dam-De Veen, C.G.; Neuenschwander, S.; Hoerstrup, S.P.; Baaijens, F.P.T.; Bouten, C.V.C. Fibrin as a cell carrier in cardiovascular tissue engineering applications. Biomaterials 2005, 26, 3113–3121. [Google Scholar] [CrossRef] [PubMed]
- De la Puente, P.; Ludeña, D. Cell culture in autologous fibrin scaffolds for applications in tissue engineering. Exp. Cell Res. 2014, 322, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Oseni, A.O.; Butler, P.E.; Seifalian, A.M. Rapid Production of Autologous Fibrin Hydrogels for Cellular Encapsulation in Organ Regeneration. In Organ Regeneration: Methods and Protocols; Basu, J., Ludlow, J.W., Eds.; Humana Press: Totowa, NJ, USA, 2013; pp. 145–152. ISBN 978-1-62703-363-3. [Google Scholar]
- Aurora, A.; Wrice, N.; Walters, T.J.; Christy, R.J.; Natesan, S. A PEGylated platelet free plasma hydrogel based composite scaffold enables stable vascularization and targeted cell delivery for volumetric muscle loss. Acta Biomater. 2018, 65, 150–162. [Google Scholar] [CrossRef]
- Anitua, E.; Nurden, P.; Prado, R.; Nurden, A.T.; Padilla, S. Autologous fibrin scaffolds: When platelet- and plasma-derived biomolecules meet fibrin. Biomaterials 2019, 192, 440–460. [Google Scholar] [CrossRef]
- Mazlyzam, A.L.; Aminuddin, B.S.; Fuzina, N.H.; Norhayati, M.M.; Fauziah, O.; Isa, M.R.; Saim, L.; Ruszymah, B.H.I. Reconstruction of living bilayer human skin equivalent utilizing human fibrin as a scaffold. Burns 2007, 33, 355–363. [Google Scholar] [CrossRef]
- Chellini, F.; Tani, A.; Zecchi-Orlandini, S.; Sassoli, C. Influence of platelet-rich and platelet-poor plasma on endogenous mechanisms of skeletal muscle repair/regeneration. Int. J. Mol. Sci. 2019, 20, 683. [Google Scholar] [CrossRef]
- Negri, S.; Federici, G.; Farinato, S.; Fila, C. Human plasma as a dermal scaffold for the generation of a completely autologous bioengineered skin. J. Clin. Rehabil. Tissue Eng. Res. 2009, 13, 9211–9216. [Google Scholar] [CrossRef]
- Growney, E.A.; Linder, H.R.; Garg, K.; Bledsoe, J.G.; Sell, S.A. Bio-conjugation of platelet-rich plasma and alginate through carbodiimide chemistry for injectable hydrogel therapies. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 1972–1984. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Ma, J.; Gan, S.; Chu, S.; Maldonado, M.; Zhou, J.; Ma, L.; Tang, S. Platelet poor plasma gel combined with amnion improves the therapeutic effects of human umbilical cord-derived mesenchymal stem cells on wound healing in rats. Mol. Med. Rep. 2017, 16, 3494–3502. [Google Scholar] [CrossRef] [PubMed]
- Kasemkijwattana, C.; Rungsinaporn, V.; Siripisitsak, T.; Kongmalai, P.; Boonprasert, R.; Charoenthamruksa, C.; Hongeng, S.; Wongkajornsilp, A.; Muangsomboon, S.; Chaipinyo, K.; et al. Autologous Fibrin-Base Scaffold for Chondrocytes and Bone Marrow Mesenchymal Stem Cells Implantation: The Development and Comparison to Conventional Fibrin Glue. J. Med. Assoc. Thai. 2016, 99, S99–S104. [Google Scholar] [PubMed]
- Cubo, N.; Garcia, M.; Del Cañizo, J.F.; Velasco, D.; Jorcano, J.L. 3D bioprinting of functional human skin: Production and in vivo analysis. Biofabrication 2017, 9, 015006. [Google Scholar] [CrossRef]
- Park, Y.G.; Lee, I.H.; Park, E.S.; Kim, J.Y. Hydrogel and platelet-rich plasma combined treatment to accelerate wound healing in a nude mouse model. Arch. Plast. Surg. 2017, 44, 194–201. [Google Scholar] [CrossRef]
- Jalowiec, J.M.; D’Este, M.; Bara, J.J.; Denom, J.; Menzel, U.; Alini, M.; Verrier, S.; Herrmann, M. An in Vitro Investigation of Platelet-Rich Plasma-Gel as a Cell and Growth Factor Delivery Vehicle for Tissue Engineering. Tissue Eng.-Part C Methods 2016, 22, 49–58. [Google Scholar] [CrossRef]
- Zahn, J.; Loibl, M.; Sprecher, C.; Nerlich, M.; Alini, M.; Verrier, S.; Herrmann, M. Platelet-Rich Plasma as an Autologous and Proangiogenic Cell Delivery System. Mediat. Inflamm. 2017, 2017, 1075975. [Google Scholar] [CrossRef]
- Kim, Y.H.; Furuya, H.; Tabata, Y. Enhancement of bone regeneration by dual release of a macrophage recruitment agent and platelet-rich plasma from gelatin hydrogels. Biomaterials 2014, 35, 214–224. [Google Scholar] [CrossRef]
- Jain, E.; Chinzei, N.; Blanco, A.; Case, N.; Sandell, L.J.; Sell, S.; Rai, M.F.; Zustiak, S.P. Platelet-Rich Plasma Released From Polyethylene Glycol Hydrogels Exerts Beneficial Effects on Human Chondrocytes. J. Orthop. Res. 2019, 37, 2401–2410. [Google Scholar] [CrossRef]
- Gao, X.; Gao, L.; Groth, T.; Liu, T.; He, D.; Wang, M.; Gong, F.; Chu, J.; Zhao, M. Fabrication and properties of an injectable sodium alginate/PRP composite hydrogel as a potential cell carrier for cartilage repair. J. Biomed. Mater. Res. Part A 2019, 107, 2076–2087. [Google Scholar] [CrossRef]
- Nimal, T.R.; Baranwal, G.; Bavya, M.C.; Biswas, R.; Jayakumar, R. Anti-staphylococcal Activity of Injectable Nano Tigecycline/Chitosan-PRP Composite Hydrogel Using Drosophila melanogaster Model for Infectious Wounds. ACS Appl. Mater. Interfaces 2016, 8, 22074–22083. [Google Scholar] [CrossRef] [PubMed]
- Samberg, M.; Stone, R.; Natesan, S.; Kowalczewski, A.; Becerra, S.; Wrice, N.; Cap, A.; Christy, R. Platelet rich plasma hydrogels promote in vitro and in vivo angiogenic potential of adipose-derived stem cells. Acta Biomater. 2019, 87, 76–87. [Google Scholar] [CrossRef]
- Jain, E.; Sheth, S.; Dunn, A.; Zustiak, S.P.; Sell, S.A. Sustained release of multicomponent platelet-rich plasma proteins from hydrolytically degradable PEG hydrogels. J. Biomed. Mater. Res. Part A 2017, 105, 3304–3314. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, M.S.; Patel, S.; Bansal, T. Improvising PRP for use in osteoarthritis knee- upcoming trends and futuristic view. J. Clin. Orthop. Trauma 2019, 10, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, M.S.; Patel, S.; John, R. PRP in OA knee—Update, current confusions and future options. SICOT-J 2017, 3, 27. [Google Scholar] [CrossRef] [PubMed]
- Natesan, S.; Stone, R.; Coronado, R.E.; Wrice, N.L.; Kowalczewski, A.C.; Zamora, D.O.; Christy, R.J. PEGylated Platelet-Free Blood Plasma-Based Hydrogels for Full-Thickness Wound Regeneration. Adv. Wound Care 2019, 8, 323–340. [Google Scholar] [CrossRef]
- Stone, R.; Wall, J.T.; Natesan, S.; Christy, R.J. PEG-plasma hydrogels increase epithelialization using a human ex vivo skin model. Int. J. Mol. Sci. 2018, 19, 3156. [Google Scholar] [CrossRef][Green Version]
- Burmeister, D.M.; Roy, D.C.; Becerra, S.C.; Natesan, S.; Christy, R.J. In situ delivery of fibrin-based hydrogels prevents contraction and reduces inflammation. J. Burn Care Res. 2018, 39, 40–53. [Google Scholar] [CrossRef]
- Montero, A.; Acosta, S.; Hernández, R.; Elvira, C.; Jorcano, J.L.; Velasco, D. Contraction of fibrin-derived matrices and its implications for in vitro human skin bioengineering. J. Biomed. Mater. Res. Part A 2021, 109, 500–514. [Google Scholar] [CrossRef]
- Tuan, T.L.; Song, A.; Chang, S.; Younai, S.; Nimni, M.E. In vitro fibroplasia: Matrix contraction, cell growth, and collagen production of fibroblasts cultured in fibrin gels. Exp. Cell Res. 1996, 223, 127–134. [Google Scholar] [CrossRef]
- Oss-Ronen, L.; Seliktar, D. Polymer-conjugated albumin and fibrinogen composite hydrogels as cell scaffolds designed for affinity-based drug delivery. Acta Biomater. 2011, 7, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Bidault, L.; Deneufchatel, M.; Hindié, M.; Vancaeyzeele, C.; Fichet, O.; Larreta-Garde, V. Fibrin-based interpenetrating polymer network biomaterials with tunable biodegradability. Polymer 2015, 62, 19–27. [Google Scholar] [CrossRef]
- Akpalo, E.; Bidault, L.; Boissière, M.; Vancaeyzeele, C.; Fichet, O.; Garde, V.L. Fibrin-polyethylene oxide interpenetrating polymer networks: New self-supported biomaterials combining the properties of both protein gel and synthetic polymer. Acta Biomater. 2011, 7, 2418–2427. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C.; Barker, T.H. Fibrin-based biomaterials: Modulation of macroscopic properties through rational design at the molecular level. Acta Biomater. 2014, 10, 1502–1514. [Google Scholar] [CrossRef] [PubMed]
- Shikanov, A.; Xu, M.; Woodruff, T.K.; Shea, L.D. Interpenetrating fibrin-alginate matrices for in vitro ovarian follicle development. Biomaterials 2009, 30, 5476–5485. [Google Scholar] [CrossRef] [PubMed]
- Devi, M.P.; Sekar, M.; Chamundeswari, M.; Moorthy, A.; Krithiga, G.; Murugan, N.S.; Sastry, T.P. A novel wound dressing material fibrin chitosan sodium alginate. Indian Acad. Sci. 2012, 35, 1157–1163. [Google Scholar]
- Zhou, H.; Xu, H.H.K. The fast release of stem cells from alginate-fibrin microbeads in injectable scaffolds for bone tissue engineering. Biomaterials 2011, 32, 7503–7513. [Google Scholar] [CrossRef]
- Garcia-Orue, I.; Santos-Vizcaino, E.; Sanchez, P.; Gutierrez, F.B.; Aguirre, J.J.; Hernandez, R.M.; Igartua, M. Bioactive and degradable hydrogel based on human platelet-rich plasma fibrin matrix combined with oxidized alginate in a diabetic mice wound healing model. Mater. Sci. Eng. C 2022, 112695. [Google Scholar] [CrossRef]
- Zhao, H.; Heindel, N.D. Determination of Degree of Substitution of Formyl Groups in Polyaldehyde Dextran by the Hydroxylamine Hydrochloride Method. Pharm. Res. An Off. J. Am. Assoc. Pharm. Sci. 1991, 8, 400–402. [Google Scholar] [CrossRef]
- Gomez, C.G.; Rinaudo, M.; Villar, M.A. Oxidation of sodium alginate and characterization of the oxidized derivatives. Carbohydr. Polym. 2007, 67, 296–304. [Google Scholar] [CrossRef]
- Wang, L.; Hou, Y.; Zhong, X.; Hu, J.; Shi, F.; Mi, H. Preparation and catalytic performance of alginate-based Schiff Base. Carbohydr. Polym. 2019, 208, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Jejurikar, A.; Seow, X.T.; Lawrie, G.; Martin, D.; Jayakrishnan, A.; Grøndahl, L. Degradable alginate hydrogels crosslinked by the macromolecular crosslinker alginate dialdehyde. J. Mater. Chem. 2012, 22, 9751–9758. [Google Scholar] [CrossRef]
- Verma, V.; Verma, P.; Ray, P.; Ray, A.R. 2, 3-Dihydrazone cellulose: Prospective material for tissue engineering scaffolds. Mater. Sci. Eng. C 2008, 28, 1441–1447. [Google Scholar] [CrossRef]
- Huamani-Palomino, R.G.; Jacinto, C.R.; Alarcón, H.; Mejía, I.M.; López, R.C.; Silva, D.d.O.; Cavalheiro, E.T.G.; Venâncio, T.; Dávalos, J.Z.; Valderrama, A.C. Chemical modification of alginate with cysteine and its application for the removal of Pb(II) from aqueous solutions. Int. J. Biol. Macromol. 2019, 129, 1056–1068. [Google Scholar] [CrossRef] [PubMed]
- Whyte, C.S.; Chernysh, I.N.; Domingues, M.M.; Connell, S.; Weisel, J.W.; Ariens, R.A.S.; Mutch, N.J. Polyphosphate delays fibrin polymerisation and alters the mechanical properties of the fibrin network. Thromb. Haemost. 2016, 116, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Ryan, E.A.; Mockros, L.F.; Weisel, J.W.; Lorand, L. Structural origins of fibrin clot rheology. Biophys. J. 1999, 77, 2813–2826. [Google Scholar] [CrossRef]
- Vorwald, C.E.; Gonzalez-Fernandez, T.; Joshee, S.; Sikorski, P.; Leach, J.K. Tunable fibrin-alginate interpenetrating network hydrogels to support cell spreading and network formation. Acta Biomater. 2020, 108, 142–152. [Google Scholar] [CrossRef]
- Sun, Y.; Giraudier, O.; Garde, V.L. Rheological characterization and dissolution kinetics of fibrin gels crosslinked by a microbial transglutaminase. Biopolymers 2005, 77, 257–263. [Google Scholar] [CrossRef]
- Distler, T.; Ruther, F.; Boccaccini, A.R.; Detsch, R. Development of 3D Biofabricated Cell Laden Hydrogel Vessels and a Low-Cost Desktop Printed Perfusion Chamber for In Vitro Vessel Maturation. Macromol. Biosci. 2019, 19, 1900245. [Google Scholar] [CrossRef]
- Somasekharan, L.T.; Kasoju, N.; Raju, R.; Bhatt, A. Formulation and characterization of alginate dialdehyde, gelatin, and platelet-rich plasma-based bioink for bioprinting applications. Bioengineering 2020, 7, 108. [Google Scholar] [CrossRef]
- Sarker, B.; Singh, R.; Silva, R.; Roether, J.A.; Kaschta, J.; Detsch, R.; Schubert, D.W.; Cicha, I.; Boccaccini, A.R. Evaluation of fibroblasts adhesion and proliferation on alginate-gelatin crosslinked hydrogel. PLoS ONE 2014, 9, 107952. [Google Scholar] [CrossRef] [PubMed]
- Genç, H.; Hazur, J.; Karakaya, E.; Dietel, B.; Bider, F.; Groll, J.; Alexiou, C.; Boccaccini, A.R.; Detsch, R.; Cicha, I. Differential responses to bioink-induced oxidative stress in endothelial cells and fibroblasts. Int. J. Mol. Sci. 2021, 22, 2358. [Google Scholar] [CrossRef] [PubMed]
- Emami, Z.; Ehsani, M.; Zandi, M.; Foudazi, R. Controlling alginate oxidation conditions for making alginate-gelatin hydrogels. Carbohydr. Polym. 2018, 198, 509–517. [Google Scholar] [CrossRef] [PubMed]
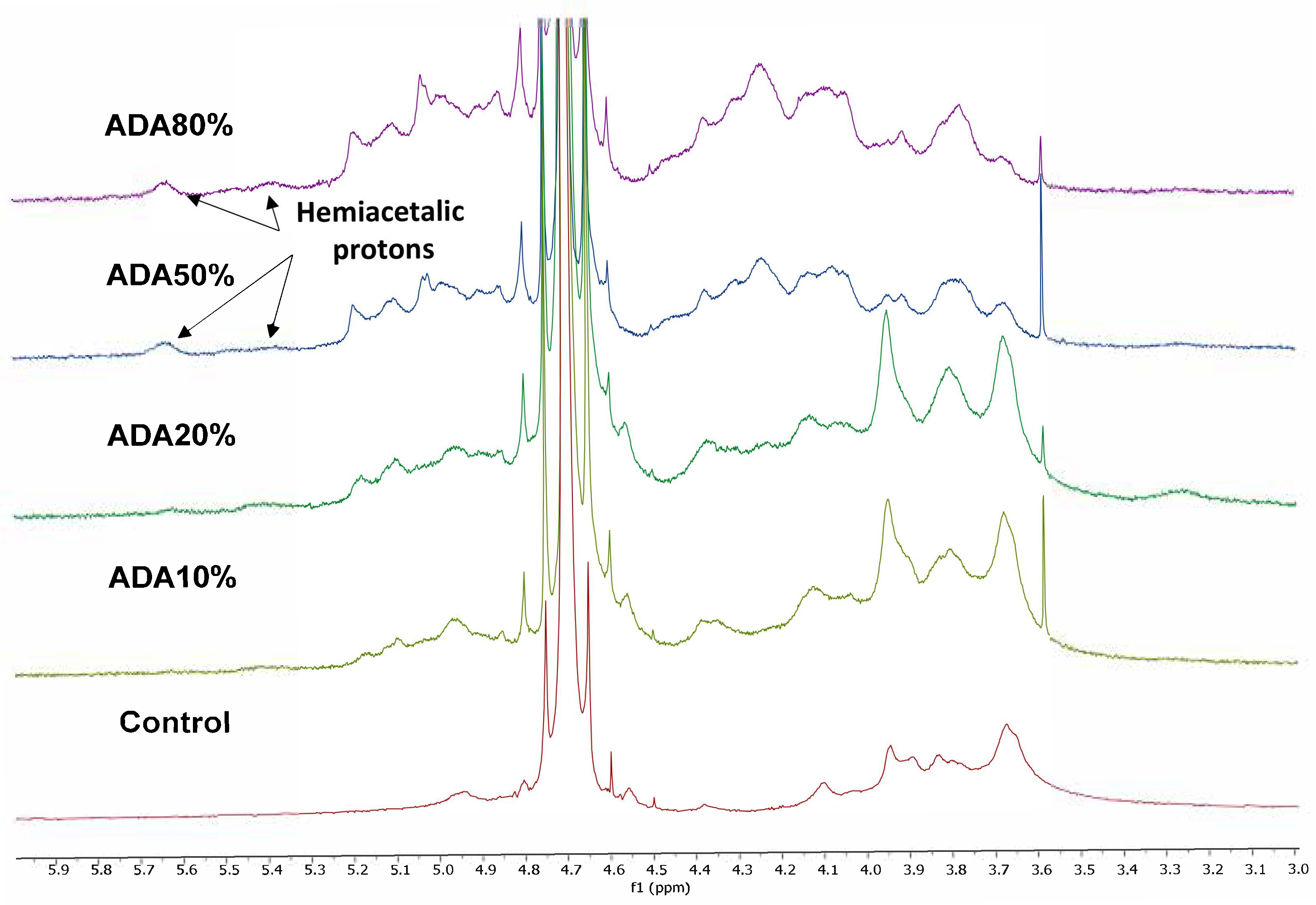
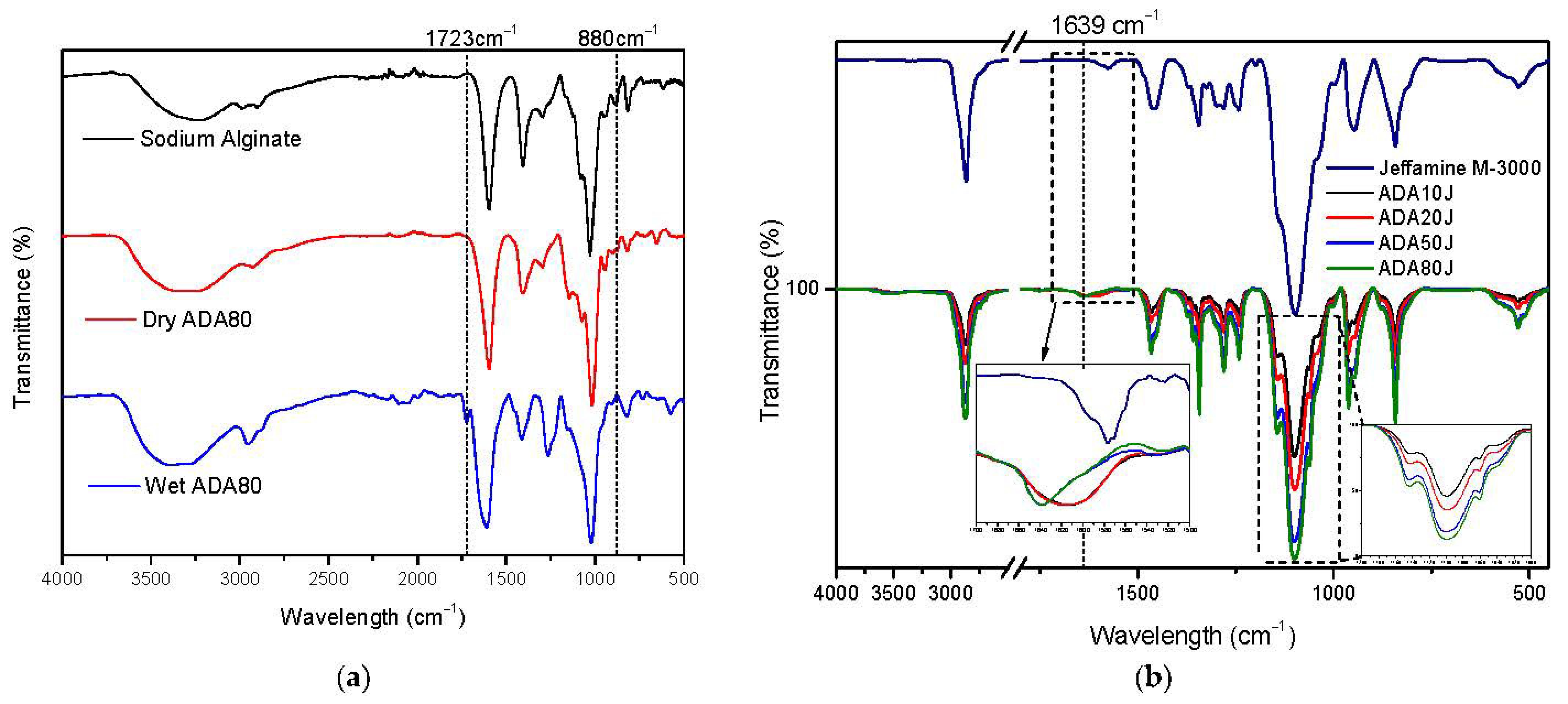

| Theoretical Oxidation (%) | Oxidation (%) 1 | MW (GPC) | PI | Maximum Concentration in Final Hydrogel (mg/mL) |
|---|---|---|---|---|
| 0 | 0 | 467,000 | 2.3 | - |
| 10 | 7.7 ± 0.5 | 178,000 | 1.9 | 3 |
| 20 | 17 ± 2.6 | 69,000 | 1.6 | 3 |
| 50 | 46.4 ± 2.5 | 32,000 | 2.9 | 0 |
| 80 | 74.1 ± 3.1 | 33,000 | 2.5 | 0 |
| Theoretical Oxidation (%) | NaIO4 (mmol) | Sample Name |
|---|---|---|
| 0 | 0 | Sodium Alginate |
| 10 | 0.5 | ADA10 |
| 20 | 1 | ADA20 |
| 50 | 2.5 | ADA50 |
| 80 | 4 | ADA80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanz-Horta, R.; Matesanz, A.; Jorcano, J.L.; Velasco, D.; Acedo, P.; Gallardo, A.; Reinecke, H.; Elvira, C. Preparation and Characterization of Plasma-Derived Fibrin Hydrogels Modified by Alginate di-Aldehyde. Int. J. Mol. Sci. 2022, 23, 4296. https://doi.org/10.3390/ijms23084296
Sanz-Horta R, Matesanz A, Jorcano JL, Velasco D, Acedo P, Gallardo A, Reinecke H, Elvira C. Preparation and Characterization of Plasma-Derived Fibrin Hydrogels Modified by Alginate di-Aldehyde. International Journal of Molecular Sciences. 2022; 23(8):4296. https://doi.org/10.3390/ijms23084296
Chicago/Turabian StyleSanz-Horta, Raúl, Ana Matesanz, José Luis Jorcano, Diego Velasco, Pablo Acedo, Alberto Gallardo, Helmut Reinecke, and Carlos Elvira. 2022. "Preparation and Characterization of Plasma-Derived Fibrin Hydrogels Modified by Alginate di-Aldehyde" International Journal of Molecular Sciences 23, no. 8: 4296. https://doi.org/10.3390/ijms23084296
APA StyleSanz-Horta, R., Matesanz, A., Jorcano, J. L., Velasco, D., Acedo, P., Gallardo, A., Reinecke, H., & Elvira, C. (2022). Preparation and Characterization of Plasma-Derived Fibrin Hydrogels Modified by Alginate di-Aldehyde. International Journal of Molecular Sciences, 23(8), 4296. https://doi.org/10.3390/ijms23084296







