Opioidergic Signaling—A Neglected, Yet Potentially Important Player in Atopic Dermatitis
Abstract
1. Introduction
1.1. Key Aspects of the Pathogenesis of AD
1.2. Overview of Opioidergic Signaling

2. Role of the Opioidergic Signaling in AD
2.1. Expression of the Opioidergic System in the Skin
2.1.1. MOR, DOR, KOR, as Well as Their Major Endogenous Ligands, Are Present in the Skin
2.1.2. NOP Receptor, as Well as ζ (OGF) Receptor, Are Expressed in the Skin
2.1.3. Contradictory Data
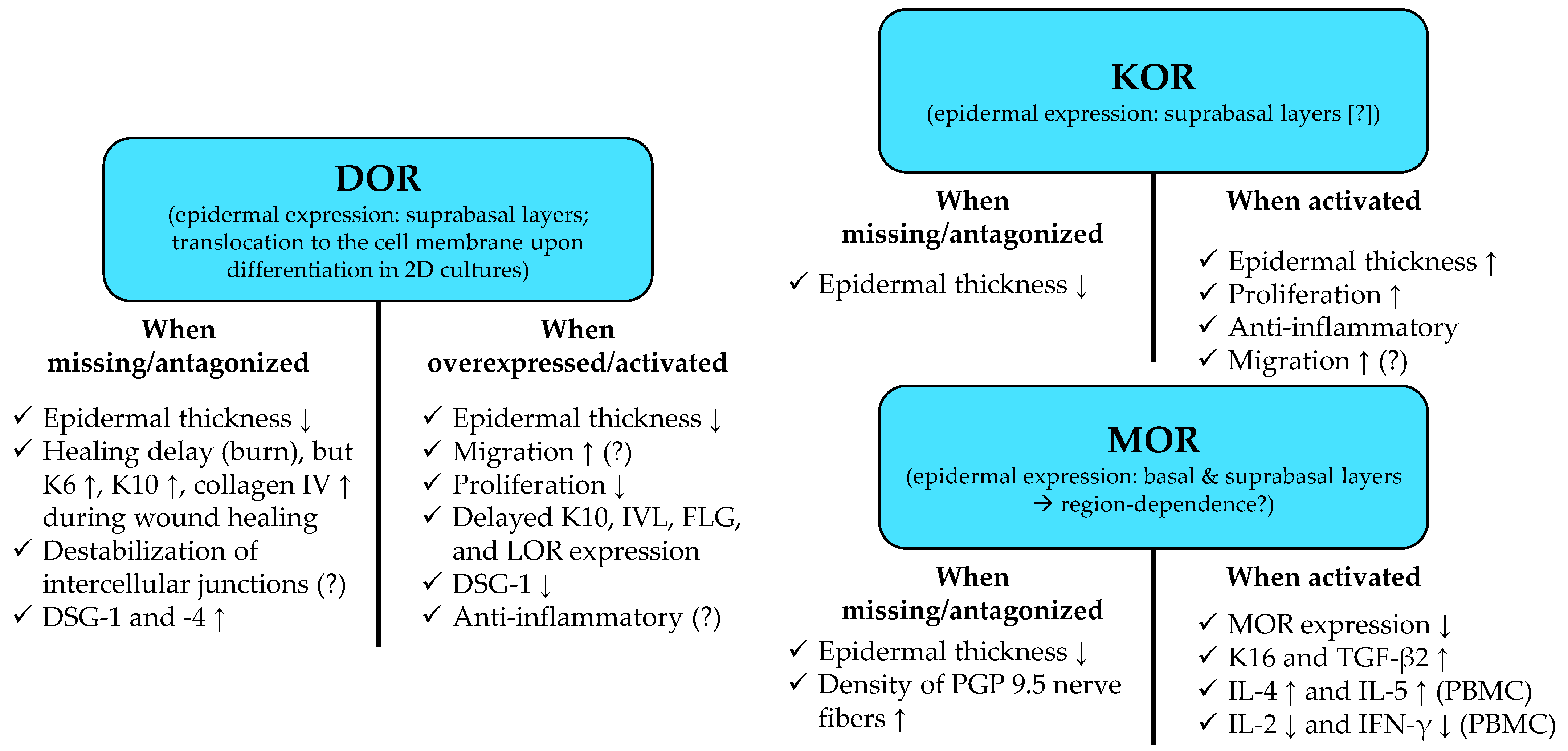
2.2. Opioidergic Signaling and the Proliferation/Differentiation Balance of Epidermal Keratinocytes—Putative Role in the Development of the Physicochemical Barrier
2.3. Opioidergic Signaling and the Cutaneous Immune System—Potentially AD-Relevant Aspects
2.3.1. Cutaneous Opioidergic Signaling Is Involved in Mediating Local Immune Responses
2.3.2. As Compared to MOR, KOR and DOR Are More Likely to Mediate Local Anti-Inflammatory Actions in the Skin
2.3.3. Effects of the Opioidergic Signaling on Mast Cells (MCs)
2.3.4. Opioidergic Signaling May Be Involved in Regulating the Balance between Th1- and Th2-Type Immune Response
2.4. Opioids in the Skin—Microbiota Communication
2.5. Opioidergic Signaling and Itch
2.5.1. Effects of MOR Antagonism and/or KOR Agonism on Pruritus—Animal Studies
2.5.2. A Deeper Insight into the Role of MOR and KOR in Controlling Pruritus—Itchy Circuits
2.5.3. Effects of MOR Antagonism and/or KOR Agonism on Pruritus—Human Studies
2.5.4. Cooperation between the Opioidergic Signaling and Certain “Itch Pathways”
2.5.5. Contradictory Data Regarding the Role of Opioidergic Signaling in Itch
2.6. Opioidergic Signaling and Stress
2.7. Outside the Box: Relationship with Cannabinoid Signaling and Electroacupuncture
2.8. Direct Evidence: Findings in AD
3. Discussion: Challenges, Open Questions, Promising Research Directions
3.1. Promising Possibilities: How Could Modulation of the Opioidergic Signaling Alleviate AD?
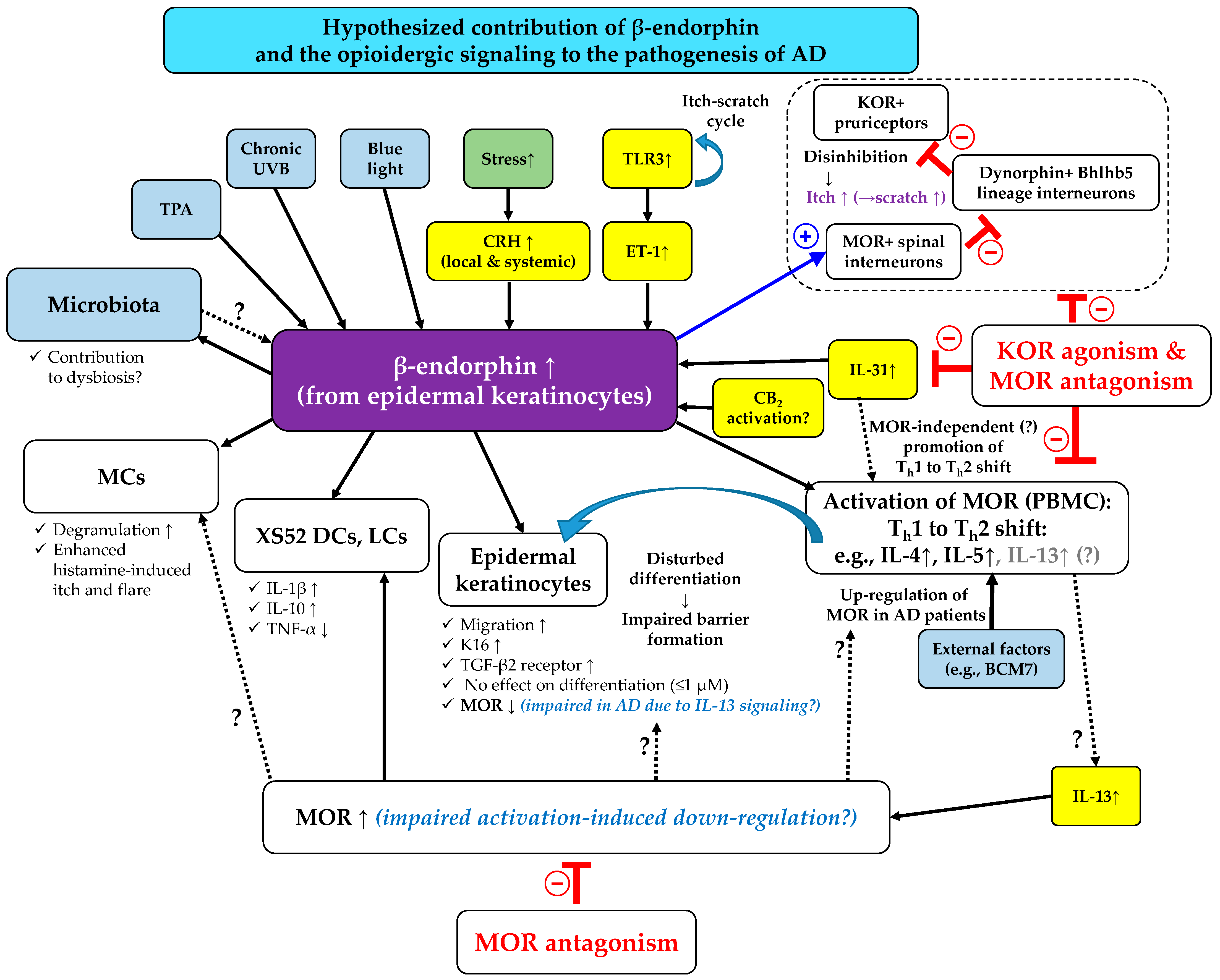
3.2. β-Endorphin: A Key Molecule That Connects Several AD-Relevant Signaling Pathways
3.3. Unexplored Territories, Contradictions, Upcoming Challenges
- Lack of KO-validated, subtype-specific antibodies
- Insufficient knowledge on the expression pattern and putative differential role, as well as ligand selectivity of the splice variants of the major receptors
- Signaling bias as well as receptor heteromerization
- Lack of receptor-subtype-selective agonists and antagonists
- Lack of sufficient knowledge on the biological characteristics of the splice variants of the endogenous ligands
- Unexplored subcellular distribution (surface vs. unavailable intracellular receptor pools)
- Regional and sex differences in the expression pattern of the receptors and ligands
4. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 5′GNTI | 5′-guanidinonaltrindole |
| ACTH | adrenocorticotropic hormone |
| AD | atopic dermatitis |
| AITC | allyl isothiocyanate |
| AP-1 | activator protein-1 |
| BCM7 | β-casomorphin-7 |
| CADESI | Canine Atopic Dermatitis Extent and Severity Index |
| cAMP | cyclic adenosine monophosphate |
| CB2 | cannabinoid receptor 2 |
| CCK | cholecystokinin |
| CCL2 | C-C motif chemokine ligand 2, also known as monocyte chemoattractant protein 1 |
| CPE | carboxypeptidase E |
| CREB | cAMP response element-binding protein |
| CRH | corticotropin-releasing hormone |
| CXCL1 | chemokine (C-X-C motif) ligand 1, also known as keratinocyte-derived cytokine |
| DMSO | dimethyl sulfoxide |
| DNFB | dinitrofluorobenzene |
| DOR | δ-opioid receptor |
| DRG | dorsal root ganglia |
| DSG | desmoglein |
| EGR-1 | early growth response 1 |
| ERK | extracellular signal-regulated protein kinase |
| ET | endothelin |
| EV | extracellular vesicle |
| FLG | filaggrin |
| G-CSF | granulocyte colony-stimulating factor |
| GIRK | G-protein-coupled inwardly rectifying potassium, also known as Kir-3 |
| GRP | gastrin releasing peptide |
| GRPR | gastrin releasing peptide receptor |
| HPA | hypothalamic—pituitary—adrenal cortex |
| i.d. | intradermal |
| IFN | interferon |
| IL | interleukin |
| IL-31RA | interleukin-31 receptor A |
| JNK | c-Jun N-terminal kinase |
| K | keratin |
| KC | keratinocyte-derived cytokine, also known as chemokine (C-X-C motif) ligand 1 |
| KLH | keyhole limpet hemocyanin |
| KOR | κ-opioid receptor |
| LC | Langerhans cell |
| LPS | lipopolysaccharide |
| MAPK | mitogen-activated protein kinase |
| MC | mast cell |
| MCP-1 | monocyte chemoattractant protein 1, also known as C-C motif chemokine ligand 2 |
| Mrgpr | Mas-related G-protein-coupled receptor |
| MIC | minimal inhibitory concentration |
| MIF | melanocyte inhibiting factor |
| MMP | matrix metalloproteinase |
| MOR | μ-opioid receptor |
| MRGPRX2 | MAS-related GPR family member X2 |
| MSH | melanocyte-stimulating hormone |
| mTOR | mammalian target of rapamycin |
| NK cell | natural killer cell |
| NOP receptor | nociceptin/orphanin FQ receptor |
| nor-BNI | nor-binaltorphimine |
| NPFF | neuropeptide FF |
| NPPB | natriuretic polypeptide b |
| OGF | opioid growth factor (Met-enkephalin) |
| p.o. | per os |
| p16INK4a | cyclin-dependent kinase inhibitor 2A |
| p21WAF1/CIP1 | cyclin-dependent kinase inhibitor 1 |
| PAR | protease-activate receptor |
| PBMC | peripheral blood monomorphonuclear cell |
| PC | prohormone convertase |
| PDYN | preprodynorphin |
| PENK | proenkephalin-A |
| PKA | protein kinase A |
| POMC | proopiomelanocortin |
| pP90RSK | 90-kDa Ribosomal S6 Kinase |
| PUVA | psoralen-ultraviolet A |
| R7bp | regulator of G protein signaling 7 binding protein |
| RTX | resiniferatoxin |
| s.c. | subcutaneous |
| SPF | specific pathogen-free |
| STAT | signal transducer and activator of transcription |
| TGase | transglutaminase |
| TGF | transforming growth factor |
| Th | helper T cell |
| TLR | toll-like receptor |
| TNF-α | tumor necrosis factor-α |
| TPA | 12-O-tetradecanoylphorbol-13-acetate |
| TRPA | transient receptor potential ankyrin |
| TRPM | transient receptor potential melastatin |
| TRPV | transient receptor potential vanilloid |
| VNTR | variable nucleotide repeat |
References
- Bieber, T. Atopic Dermatitis. N. Engl. J. Med. 2008, 358, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.E.M.; van de Kerkhof, P.; Czarnecka-Operacz, M. Psoriasis and Atopic Dermatitis. Dermatol. Ther. 2017, 7, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Langan, S.M.; Irvine, A.D.; Weidinger, S. Atopic Dermatitis. Lancet 2020, 396, 345–360. [Google Scholar] [CrossRef]
- Bocheva, G.S.; Slominski, R.M.; Slominski, A.T. Immunological Aspects of Skin Aging in Atopic Dermatitis. Int. J. Mol. Sci. 2021, 22, 5729. [Google Scholar] [CrossRef] [PubMed]
- Tóth, K.F.; Ádám, D.; Bíró, T.; Oláh, A. Cannabinoid Signaling in the Skin: Therapeutic Potential of the “C(Ut)Annabinoid” System. Molecules 2019, 24, 918. [Google Scholar] [CrossRef] [PubMed]
- Edwards, T.; Patel, N.U.; Blake, A.; Prabakaran, S.; Reimer, D.; Feldman, S.R.; Strowd, L.C. Insights into Future Therapeutics for Atopic Dermatitis. Expert Opin. Pharmacother. 2018, 19, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Vakharia, P.P.; Silverberg, J.I. New Therapies for Atopic Dermatitis: Additional Treatment Classes. J. Am. Acad. Dermatol. 2018, 78, S76–S83. [Google Scholar] [CrossRef]
- Erickson, S.; Heul, A.V.; Kim, B.S. New and Emerging Treatments for Inflammatory Itch. Ann. Allergy Asthma Immunol. 2021, 126, 13–20. [Google Scholar] [CrossRef]
- Soeberdt, M.; Kilic, A.; Abels, C. Small Molecule Drugs for the Treatment of Pruritus in Patients with Atopic Dermatitis. Eur. J. Pharmacol. 2020, 881, 173242. [Google Scholar] [CrossRef]
- Yang, N.; Chen, Z.; Zhang, X.; Shi, Y. Novel Targeted Biological Agents for the Treatment of Atopic Dermatitis. BioDrugs 2021, 35, 401–415. [Google Scholar] [CrossRef]
- Kader, H.A.; Azeem, M.; Jwayed, S.A.; Al-Shehhi, A.; Tabassum, A.; Ayoub, M.A.; Hetta, H.F.; Waheed, Y.; Iratni, R.; Al-Dhaheri, A.; et al. Current Insights into Immunology and Novel Therapeutics of Atopic Dermatitis. Cells 2021, 10, 1392. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, J.I.; Pinter, A.; Alavi, A.; Lynde, C.; Bouaziz, J.-D.; Wollenberg, A.; Murrell, D.F.; Alpizar, S.; Laquer, V.; Chaouche, K.; et al. Nemolizumab Is Associated with a Rapid Improvement in Atopic Dermatitis Signs and Symptoms: Subpopulation (EASI ≥ 16) Analysis of Randomized Phase 2B Study. J. Eur. Acad. Dermatol. Venereol. JEADV 2021, 35, 1562–1568. [Google Scholar] [CrossRef] [PubMed]
- Torres, T.; Paiva-Lopes, M.J.; Gonçalo, M.; Claro, C.; Oliveira, M.; Gomes, J.; Vieira, A.P.; Amoedo, P.; Alpalhão, M.; Nogueira, M.; et al. Dupilumab for Atopic Dermatitis: A Real-World Portuguese Multicenter Retrospective Study. J. Dermatol. Treat. 2022, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kovács, D.; Fazekas, F.; Oláh, A.; Törőcsik, D. Adipokines in the Skin and in Dermatological Diseases. Int. J. Mol. Sci. 2020, 21, 9048. [Google Scholar] [CrossRef]
- Yang, G.H.; Lee, Y.B.; Kang, D.; Choi, E.; Nam, Y.; Lee, K.H.; You, H.-J.; Kang, H.J.; An, S.H.; Jeon, H. Overcome the Barriers of the Skin: Exosome Therapy. Biomater. Res. 2021, 25, 22. [Google Scholar] [CrossRef]
- Czarnowicki, T.; He, H.; Krueger, J.G.; Guttman-Yassky, E. Atopic Dermatitis Endotypes and Implications for Targeted Therapeutics. J. Allergy Clin. Immunol. 2019, 143, 1–11. [Google Scholar] [CrossRef]
- Narla, S.; Silverberg, J.I. The Role of Environmental Exposures in Atopic Dermatitis. Curr. Allergy Asthma Rep. 2020, 20, 74. [Google Scholar] [CrossRef]
- Piquero-Casals, J.; Carrascosa, J.M.; Morgado-Carrasco, D.; Narda, M.; Trullas, C.; Granger, C.; Fabbrocini, G. The Role of Photoprotection in Optimizing the Treatment of Atopic Dermatitis. Dermatol. Ther. 2021, 11, 315–325. [Google Scholar] [CrossRef]
- Jurakić Tončić, R.; Marinović, B. The Role of Impaired Epidermal Barrier Function in Atopic Dermatitis. Acta Dermatovenerol. Croat. ADC 2016, 24, 95–109. [Google Scholar]
- Furue, M. Regulation of Filaggrin, Loricrin, and Involucrin by IL-4, IL-13, IL-17A, IL-22, AHR, and NRF2: Pathogenic Implications in Atopic Dermatitis. Int. J. Mol. Sci. 2020, 21, 5382. [Google Scholar] [CrossRef]
- Nakahara, T.; Kido-Nakahara, M.; Tsuji, G.; Furue, M. Basics and Recent Advances in the Pathophysiology of Atopic Dermatitis. J. Dermatol. 2020, 48, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Proksch, E.; Brandner, J.M.; Jensen, J.-M. The Skin: An Indispensable Barrier. Exp. Dermatol. 2008, 17, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.M.; Proksch, E. The Skin’s Barrier. G. Ital. Dermatol. Venereol. 2009, 144, 689–700. [Google Scholar] [PubMed]
- Nedoszytko, B.; Reszka, E.; Gutowska-Owsiak, D.; Trzeciak, M.; Lange, M.; Jarczak, J.; Niedoszytko, M.; Jablonska, E.; Romantowski, J.; Strapagiel, D.; et al. Genetic and Epigenetic Aspects of Atopic Dermatitis. Int. J. Mol. Sci. 2020, 21, 6484. [Google Scholar] [CrossRef]
- Pappas, A. Epidermal Surface Lipids. Derm.-Endocrinol. 2009, 1, 72–76. [Google Scholar] [CrossRef]
- Shi, V.Y.; Leo, M.; Hassoun, L.; Chahal, D.S.; Maibach, H.I.; Sivamani, R.K. Role of Sebaceous Glands in Inflammatory Dermatoses. J. Am. Acad. Dermatol. 2015, 73, 856–863. [Google Scholar] [CrossRef]
- Grosshans, E.; Woehl, M. Abnormal vasomotor, sudoral and sebaceous reactions in atopic dermatitis. Ann. Dermatol. Venereol. 1982, 109, 151–162. [Google Scholar]
- Surber, C.; Abels, C.; Maibach, H. PH of the Skin: Issues and Challenges; Karger Book: Basel, Switzerland, 2018. [Google Scholar]
- Paller, A.S.; Kong, H.H.; Seed, P.; Naik, S.; Scharschmidt, T.C.; Gallo, R.L.; Luger, T.; Irvine, A.D. The Microbiome in Patients with Atopic Dermatitis. J. Allergy Clin. Immunol. 2019, 143, 26–35. [Google Scholar] [CrossRef]
- Zhang, E.; Tanaka, T.; Tajima, M.; Tsuboi, R.; Nishikawa, A.; Sugita, T. Characterization of the Skin Fungal Microbiota in Patients with Atopic Dermatitis and in Healthy Subjects. Microbiol. Immunol. 2011, 55, 625–632. [Google Scholar] [CrossRef]
- Morita, E.; Hide, M.; Yoneya, Y.; Kannbe, M.; Tanaka, A.; Yamamoto, S. An Assessment of the Role of Candida Albicans Antigen in Atopic Dermatitis. J. Dermatol. 1999, 26, 282–287. [Google Scholar] [CrossRef]
- Savolainen, J.; Lammintausta, K.; Kalimo, K.; Viander, M. Candida Albicans and Atopic Dermatitis. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 1993, 23, 332–339. [Google Scholar] [CrossRef]
- Fiedorowicz, E.; Kaczmarski, M.; Cieślińska, A.; Sienkiewicz-Szłapka, E.; Jarmołowska, B.; Chwała, B.; Kostyra, E. β-Casomorphin-7 Alters μ-Opioid Receptor and Dipeptidyl Peptidase IV Genes Expression in Children with Atopic Dermatitis. Peptides 2014, 62, 144–149. [Google Scholar] [CrossRef]
- Szántó, M.; Dózsa, A.; Antal, D.; Szabó, K.; Kemény, L.; Bai, P. Targeting the Gut-Skin Axis-Probiotics as New Tools for Skin Disorder Management? Exp. Dermatol. 2019, 28, 1210–1218. [Google Scholar] [CrossRef] [PubMed]
- Park, D.H.; Kim, J.W.; Park, H.-J.; Hahm, D.-H. Comparative Analysis of the Microbiome across the Gut-Skin Axis in Atopic Dermatitis. Int. J. Mol. Sci. 2021, 22, 4228. [Google Scholar] [CrossRef]
- Chen, G.; Chen, Z.-M.; Fan, X.-Y.; Jin, Y.-L.; Li, X.; Wu, S.-R.; Ge, W.-W.; Lv, C.-H.; Wang, Y.-K.; Chen, J.-G. Gut-Brain-Skin Axis in Psoriasis: A Review. Dermatol. Ther. 2021, 11, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Y.; Wu, L.; Xiao, S.; Ji, Y.; Tan, Y.; Jiang, C.; Zhang, G. Dysregulation of the Gut-Brain-Skin Axis and Key Overlapping Inflammatory and Immune Mechanisms of Psoriasis and Depression. Biomed. Pharmacother. 2021, 137, 111065. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.S.; Edwards, C.; Marks, R. A Study of White Dermographism in Atopic Dermatitis. J. Dermatol. Sci. 1996, 11, 148–153. [Google Scholar] [CrossRef]
- Ebbecke, U. Die lokale vasomotorische Reaktion (L.V.R.) der Haut und der inneren Organe. Pflüg. Arch. Für Gesamte Physiol. Menschen Tiere 1917, 169, 1–81. [Google Scholar] [CrossRef][Green Version]
- Steinhoff, M.; Steinhoff, A.; Homey, B.; Luger, T.A.; Schneider, S.W. Role of Vasculature in Atopic Dermatitis. J. Allergy Clin. Immunol. 2006, 118, 190–197. [Google Scholar] [CrossRef]
- Choi, J.E.; Di Nardo, A. Skin Neurogenic Inflammation. Semin. Immunopathol. 2018, 40, 249–259. [Google Scholar] [CrossRef]
- Yosipovitch, G.; Berger, T.; Fassett, M.S. Neuroimmune Interactions in Chronic Itch of Atopic Dermatitis. J. Eur. Acad. Dermatol. Venereol. JEADV 2020, 34, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Abels, C.; Soeberdt, M. Can We Teach Old Drugs New Tricks?—Repurposing of Neuropharmacological Drugs for Inflammatory Skin Diseases. Exp. Dermatol. 2019, 28, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- De Benedetto, A.; Agnihothri, R.; McGirt, L.Y.; Bankova, L.G.; Beck, L.A. Atopic Dermatitis: A Disease Caused by Innate Immune Defects? J. Investig. Dermatol. 2009, 129, 14–30. [Google Scholar] [CrossRef] [PubMed]
- Siiskonen, H.; Harvima, I. Mast Cells and Sensory Nerves Contribute to Neurogenic Inflammation and Pruritus in Chronic Skin Inflammation. Front. Cell. Neurosci. 2019, 13, 422. [Google Scholar] [CrossRef]
- Furue, K.; Ulzii, D.; Tanaka, Y.; Ito, T.; Tsuji, G.; Kido-Nakahara, M.; Nakahara, T.; Furue, M. Pathogenic Implication of Epidermal Scratch Injury in Psoriasis and Atopic Dermatitis. J. Dermatol. 2020, 47, 979–988. [Google Scholar] [CrossRef]
- Wong, L.-S.; Yen, Y.-T.; Lee, C.-H. The Implications of Pruritogens in the Pathogenesis of Atopic Dermatitis. Int. J. Mol. Sci. 2021, 22, 7227. [Google Scholar] [CrossRef]
- Bäsler, K.; Brandner, J.M. Tight Junctions in Skin Inflammation. Pflug. Arch. 2017, 469, 3–14. [Google Scholar] [CrossRef]
- Talagas, M.; Lebonvallet, N.; Berthod, F.; Misery, L. Lifting the Veil on the Keratinocyte Contribution to Cutaneous Nociception. Protein Cell 2020, 11, 239–250. [Google Scholar] [CrossRef]
- Talagas, M.; Lebonvallet, N.; Berthod, F.; Misery, L. Cutaneous Nociception: Role of Keratinocytes. Exp. Dermatol. 2019, 28, 1466–1469. [Google Scholar] [CrossRef]
- Klicznik, M.M.; Szenes-Nagy, A.B.; Campbell, D.J.; Gratz, I.K. Taking the Lead—How Keratinocytes Orchestrate Skin T Cell Immunity. Immunol. Lett. 2018, 200, 43–51. [Google Scholar] [CrossRef]
- Bitschar, K.; Wolz, C.; Krismer, B.; Peschel, A.; Schittek, B. Keratinocytes as Sensors and Central Players in the Immune Defense against Staphylococcus aureus in the Skin. J. Dermatol. Sci. 2017, 87, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Denda, M.; Nakanishi, S. Do Epidermal Keratinocytes Have Sensory and Information Processing Systems? Exp. Dermatol. 2021, 31, 459–474. [Google Scholar] [CrossRef]
- Furue, M.; Chiba, T.; Tsuji, G.; Ulzii, D.; Kido-Nakahara, M.; Nakahara, T.; Kadono, T. Atopic Dermatitis: Immune Deviation, Barrier Dysfunction, IgE Autoreactivity and New Therapies. Allergol. Int. 2017, 66, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Kuo, I.-H.; Yoshida, T.; De Benedetto, A.; Beck, L.A. The Cutaneous Innate Immune Response in Patients with Atopic Dermatitis. J. Allergy Clin. Immunol. 2013, 131, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Ono, S.; Kabashima, K. Novel Insights into the Role of Immune Cells in Skin and Inducible Skin-Associated Lymphoid Tissue (ISALT). Allergo J. Int. 2015, 24, 170–179. [Google Scholar] [CrossRef]
- Tsakok, T.; Woolf, R.; Smith, C.H.; Weidinger, S.; Flohr, C. Atopic Dermatitis: The Skin Barrier and Beyond. Br. J. Dermatol. 2019, 180, 464–474. [Google Scholar] [CrossRef]
- Wilson, S.R.; Thé, L.; Batia, L.M.; Beattie, K.; Katibah, G.E.; McClain, S.P.; Pellegrino, M.; Estandian, D.M.; Bautista, D.M. The Epithelial Cell-Derived Atopic Dermatitis Cytokine TSLP Activates Neurons to Induce Itch. Cell 2013, 155, 285–295. [Google Scholar] [CrossRef]
- Herman, A.; Herman, A.P. Antimicrobial Peptides Activity in the Skin. Skin Res. Technol. 2019, 25, 111–117. [Google Scholar] [CrossRef]
- Talagas, M.; Lebonvallet, N.; Leschiera, R.; Elies, P.; Marcorelles, P.; Misery, L. Intra-Epidermal Nerve Endings Progress within Keratinocyte Cytoplasmic Tunnels in Normal Human Skin. Exp. Dermatol. 2020, 29, 387–392. [Google Scholar] [CrossRef]
- Nasiri, G.; Azarpira, N.; Alizadeh, A.; Goshtasbi, S.; Tayebi, L. Shedding Light on the Role of Keratinocyte-Derived Extracellular Vesicles on Skin-Homing Cells. Stem Cell Res. Ther. 2020, 11, 421. [Google Scholar] [CrossRef]
- Than, U.T.T.; Leavesley, D.I.; Parker, T.J. Characteristics and Roles of Extracellular Vesicles Released by Epidermal Keratinocytes. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 2264–2272. [Google Scholar] [CrossRef] [PubMed]
- Dagnelie, M.-A.; Corvec, S.; Khammari, A.; Dréno, B. Bacterial Extracellular Vesicles: A New Way to Decipher Host-Microbiota Communications in Inflammatory Dermatoses. Exp. Dermatol. 2020, 29, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Spampinato, S.M. (Ed.) Opioid Receptors: Methods and Protocols, 2nd ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2021; ISBN 978-1-07-160883-8. [Google Scholar]
- Borsodi, A.; Bruchas, M.; Caló, G.; Chavkin, C.; Christie, M.J.; Civelli, O.; Connor, M.; Cox, B.M.; Devi, L.A.; Evans, C.; et al. Opioid Receptors (Version 2019.4) in the IUPHAR/BPS Guide to Pharmacology Database. IUPHAR/BPS Guide Pharmacol. CITE 2019, 2019, 4. [Google Scholar] [CrossRef]
- Sobczak, M.; Sałaga, M.; Storr, M.A.; Fichna, J. Physiology, Signaling, and Pharmacology of Opioid Receptors and Their Ligands in the Gastrointestinal Tract: Current Concepts and Future Perspectives. J. Gastroenterol. 2014, 49, 24–45. [Google Scholar] [CrossRef]
- Boom, M.; Niesters, M.; Sarton, E.; Aarts, L.; Smith, T.W.; Dahan, A. Non-Analgesic Effects of Opioids: Opioid-Induced Respiratory Depression. Curr. Pharm. Des. 2012, 18, 5994–6004. [Google Scholar] [CrossRef]
- Eisenstein, T.K. The Role of Opioid Receptors in Immune System Function. Front. Immunol. 2019, 10, 2904. [Google Scholar] [CrossRef]
- De Neve, J.; Barlow, T.M.A.; Tourwé, D.; Bihel, F.; Simonin, F.; Ballet, S. Comprehensive Overview of Biased Pharmacology at the Opioid Receptors: Biased Ligands and Bias Factors. RSC Med. Chem. 2021, 12, 828–870. [Google Scholar] [CrossRef]
- Che, T.; Dwivedi-Agnihotri, H.; Shukla, A.K.; Roth, B.L. Biased Ligands at Opioid Receptors: Current Status and Future Directions. Sci. Signal. 2021, 14, eaav0320. [Google Scholar] [CrossRef]
- Kudla, L.; Przewlocki, R. Influence of G Protein-Biased Agonists of μ-Opioid Receptor on Addiction-Related Behaviors. Pharmacol. Rep. 2021, 73, 1033–1051. [Google Scholar] [CrossRef]
- Kelemen, B.; Pinto, S.; Kim, N.; Lisztes, E.; Hanyicska, M.; Vladár, A.; Oláh, A.; Pénzes, Z.; Shu, B.; Vriens, J.; et al. The TRPM3 Ion Channel Mediates Nociception but Not Itch Evoked by Endogenous Pruritogenic Mediators. Biochem. Pharmacol. 2021, 183, 114310. [Google Scholar] [CrossRef]
- Vriens, J.; Owsianik, G.; Hofmann, T.; Philipp, S.E.; Stab, J.; Chen, X.; Benoit, M.; Xue, F.; Janssens, A.; Kerselaers, S.; et al. TRPM3 Is a Nociceptor Channel Involved in the Detection of Noxious Heat. Neuron 2011, 70, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Behrendt, M.; Gruss, F.; Enzeroth, R.; Dembla, S.; Zhao, S.; Crassous, P.-A.; Mohr, F.; Nys, M.; Louros, N.; Gallardo, R.; et al. The Structural Basis for an On-off Switch Controlling Gβγ-Mediated Inhibition of TRPM3 Channels. Proc. Natl. Acad. Sci. USA 2020, 117, 29090–29100. [Google Scholar] [CrossRef] [PubMed]
- Dembla, S.; Behrendt, M.; Mohr, F.; Goecke, C.; Sondermann, J.; Schneider, F.M.; Schmidt, M.; Stab, J.; Enzeroth, R.; Leitner, M.G.; et al. Anti-Nociceptive Action of Peripheral Mu-Opioid Receptors by G-Beta-Gamma Protein-Mediated Inhibition of TRPM3 Channels. eLife 2017, 6, e26280. [Google Scholar] [CrossRef] [PubMed]
- Quallo, T.; Alkhatib, O.; Gentry, C.; Andersson, D.A.; Bevan, S. G Protein Βγ Subunits Inhibit TRPM3 Ion Channels in Sensory Neurons. eLife 2017, 6, e26138. [Google Scholar] [CrossRef] [PubMed]
- Vangeel, L.; Benoit, M.; Miron, Y.; Miller, P.E.; De Clercq, K.; Chaltin, P.; Verfaillie, C.; Vriens, J.; Voets, T. Functional Expression and Pharmacological Modulation of TRPM3 in Human Sensory Neurons. Br. J. Pharmacol. 2020, 177, 2683–2695. [Google Scholar] [CrossRef]
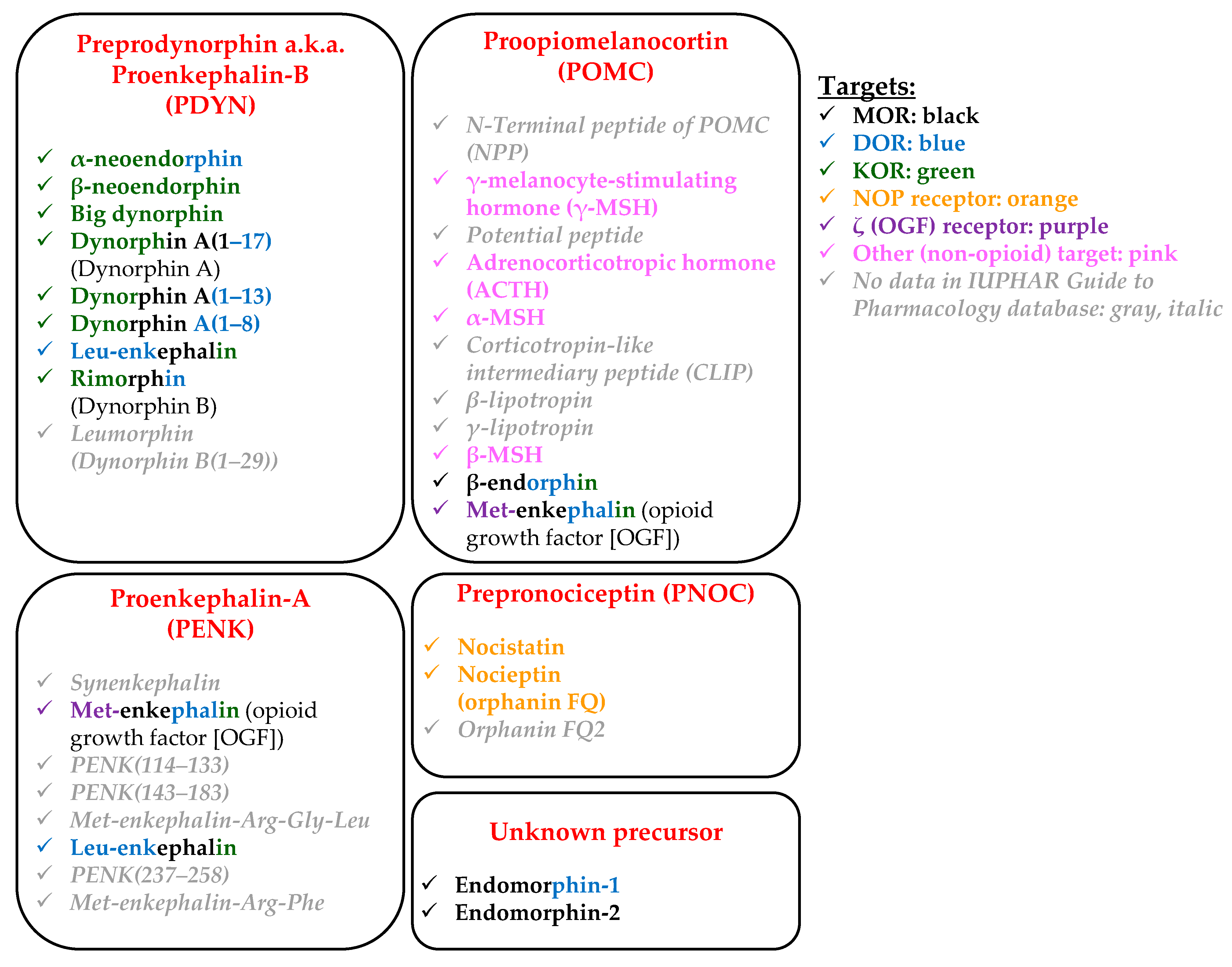
- Abrimian, A.; Kraft, T.; Pan, Y.-X. Endogenous Opioid Peptides and Alternatively Spliced Mu Opioid Receptor Seven Transmembrane Carboxyl-Terminal Variants. Int. J. Mol. Sci. 2021, 22, 3779. [Google Scholar] [CrossRef]
- Zagon, I.S.; Verderame, M.F.; McLaughlin, P.J. The Biology of the Opioid Growth Factor Receptor (OGFr). Brain Res. Brain Res. Rev. 2002, 38, 351–376. [Google Scholar] [CrossRef]
- Kren, N.P.; Zagon, I.S.; McLaughlin, P.J. Mutations in the Opioid Growth Factor Receptor in Human Cancers Alter Receptor Function. Int. J. Mol. Med. 2015, 36, 289–293. [Google Scholar] [CrossRef]
- Fricker, L.D.; Margolis, E.B.; Gomes, I.; Devi, L.A. Five Decades of Research on Opioid Peptides: Current Knowledge and Unanswered Questions. Mol. Pharmacol. 2020, 98, 96–108. [Google Scholar] [CrossRef]
- Kleczkowska, P.; Bojnik, E.; Leśniak, A.; Kosson, P.; Van den Eynde, I.; Ballet, S.; Benyhe, S.; Tourwé, D.; Lipkowski, A.W. Identification of Dmt-D-Lys-Phe-Phe-OH as a Highly Antinociceptive Tetrapeptide Metabolite of the Opioid-Neurotensin Hybrid Peptide PK20. Pharmacol. Rep. 2013, 65, 836–846. [Google Scholar] [CrossRef]
- Zagon, I.S.; Smith, J.P.; Conter, R.; McLaughlin, P.J. Identification and Characterization of Opioid Growth Factor Receptor in Human Pancreatic Adenocarcinoma. Int. J. Mol. Med. 2000, 5, 77–84. [Google Scholar] [PubMed]
- Benjannet, S.; Rondeau, N.; Day, R.; Chrétien, M.; Seidah, N.G. PC1 and PC2 Are Proprotein Convertases Capable of Cleaving Proopiomelanocortin at Distinct Pairs of Basic Residues. Proc. Natl. Acad. Sci. USA 1991, 88, 3564–3568. [Google Scholar] [CrossRef] [PubMed]
- Hook, V.; Funkelstein, L.; Toneff, T.; Mosier, C.; Hwang, S.-R. Human Pituitary Contains Dual Cathepsin L and Prohormone Convertase Processing Pathway Components Involved in Converting POMC into the Peptide Hormones ACTH, Alpha-MSH, and Beta-Endorphin. Endocrine 2009, 35, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Mousa, S.A.; Shakibaei, M.; Sitte, N.; Schäfer, M.; Stein, C. Subcellular Pathways of β-Endorphin Synthesis, Processing, and Release from Immunocytes in Inflammatory Pain. Endocrinology 2004, 145, 1331–1341. [Google Scholar] [CrossRef]
- Cesselin, F. Opioid and Anti-Opioid Peptides. Fundam. Clin. Pharmacol. 1995, 9, 409–433. [Google Scholar] [CrossRef]
- Gibula-Tarlowska, E.; Kotlinska, J.H. Crosstalk between Opioid and Anti-Opioid Systems: An Overview and Its Possible Therapeutic Significance. Biomolecules 2020, 10, 1376. [Google Scholar] [CrossRef]
- Leis, K.; Mazur, E.; Jabłońska, M.J.; Kolan, M.; Gałązka, P. Endocrine Systems of the Skin. Postepy Dermatol. Alergol. 2019, 36, 519–523. [Google Scholar] [CrossRef]
- Ramot, Y.; Böhm, M.; Paus, R. Translational Neuroendocrinology of Human Skin: Concepts and Perspectives. Trends Mol. Med. 2020, 27, 60–74. [Google Scholar] [CrossRef]
- Soeberdt, M.; Kilic, A.; Abels, C. Current and Emerging Treatments Targeting the Neuroendocrine System for Disorders of the Skin and Its Appendages. Exp. Dermatol. 2020, 29, 801–813. [Google Scholar] [CrossRef]
- Schmelz, M.; Paus, R. Opioids and the Skin: “Itchy” Perspectives beyond Analgesia and Abuse. J. Investig. Dermatol. 2007, 127, 1287–1289. [Google Scholar] [CrossRef]
- Bigliardi, P.L.; Dancik, Y.; Neumann, C.; Bigliardi-Qi, M. Opioids and Skin Homeostasis, Regeneration and Ageing—What’s the Evidence? Exp. Dermatol. 2016, 25, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T. On the Role of the Endogenous Opioid System in Regulating Epidermal Homeostasis. J. Investig. Dermatol. 2015, 135, 333–334. [Google Scholar] [CrossRef] [PubMed]
- Bigliardi, P.L.; Tobin, D.J.; Gaveriaux-Ruff, C.; Bigliardi-Qi, M. Opioids and the Skin—Where Do We Stand? Exp. Dermatol. 2009, 18, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Bigliardi-Qi, M.; Bigliardi, P. The Roles of Opioid Receptors in Cutaneous Wound Healing. Handb. Exp. Pharmacol. 2018, 247, 335–345. [Google Scholar] [CrossRef]
- Wang, Y.; Gupta, M.; Poonawala, T.; Farooqui, M.; Li, Y.; Peng, F.; Rao, S.; Ansonoff, M.; Pintar, J.E.; Gupta, K. Opioids and Opioid Receptors Orchestrate Wound Repair. Transl. Res. J. Lab. Clin. Med. 2017, 185, 13–23. [Google Scholar] [CrossRef]
- Zanello, S.B.; Jackson, D.M.; Holick, M.F. An Immunocytochemical Approach to the Study of Beta-Endorphin Production in Human Keratinocytes Using Confocal Microscopy. Ann. N. Y. Acad. Sci. 1999, 885, 85–99. [Google Scholar] [CrossRef]
- Bigliardi, P.L.; Bigliardi-Qi, M.; Buechner, S.; Rufli, T. Expression of Mu-Opiate Receptor in Human Epidermis and Keratinocytes. J. Investig. Dermatol. 1998, 111, 297–301. [Google Scholar] [CrossRef]
- Leong, C.; Neumann, C.; Ramasamy, S.; Rout, B.; Yi Wee, L.; Bigliardi-Qi, M.; Bigliardi, P.L. Investigating Endogenous Μ-Opioid Receptors in Human Keratinocytes as Pharmacological Targets Using Novel Fluorescent Ligand. PLoS ONE 2017, 12, e0188607. [Google Scholar] [CrossRef]
- Bigliardi-Qi, M.; Sumanovski, L.T.; Büchner, S.; Rufli, T.; Bigliardi, P.L. Mu-Opiate Receptor and Beta-Endorphin Expression in Nerve Endings and Keratinocytes in Human Skin. Dermatology 2004, 209, 183–189. [Google Scholar] [CrossRef]
- Boukamp, P.; Petrussevska, R.T.; Breitkreutz, D.; Hornung, J.; Markham, A.; Fusenig, N.E. Normal Keratinization in a Spontaneously Immortalized Aneuploid Human Keratinocyte Cell Line. J. Cell Biol. 1988, 106, 761–771. [Google Scholar] [CrossRef]
- Tominaga, M.; Ogawa, H.; Takamori, K. Possible Roles of Epidermal Opioid Systems in Pruritus of Atopic Dermatitis. J. Investig. Dermatol. 2007, 127, 2228–2235. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Zmijewski, M.A.; Zbytek, B.; Brozyna, A.A.; Granese, J.; Pisarchik, A.; Szczesniewski, A.; Tobin, D.J. Regulated Proenkephalin Expression in Human Skin and Cultured Skin Cells. J. Investig. Dermatol. 2011, 131, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Neumann, C.; Bigliardi-Qi, M.; Widmann, C.; Bigliardi, P.L. The δ-Opioid Receptor Affects Epidermal Homeostasis via ERK-Dependent Inhibition of Transcription Factor POU2F3. J. Investig. Dermatol. 2015, 135, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.; Schröder, W.; Jostock, R.; Schwartz, M.; Rosson, G.; Polydefkis, M. Nociceptin/Orphanin FQ Opioid Peptide-Receptor Expression in Pachyonychia Congenita. J. Peripher. Nerv. Syst. 2018, 23, 241–248. [Google Scholar] [CrossRef]
- Cheng, F.; McLaughlin, P.J.; Verderame, M.F.; Zagon, I.S. The OGF-OGFr Axis Utilizes the P16INK4a and P21WAF1/CIP1 Pathways to Restrict Normal Cell Proliferation. Mol. Biol. Cell 2009, 20, 319–327. [Google Scholar] [CrossRef]
- Wilson, R.P.; McLaughlin, P.J.; Lang, C.M.; Zagon, I.S. The Opioid Growth Factor, [Met5]-Enkephalin, Inhibits DNA Synthesis during Recornification of Mouse Tail Skin. Cell Prolif. 2000, 33, 63–73. [Google Scholar] [CrossRef]
- Salemi, S.; Aeschlimann, A.; Reisch, N.; Jüngel, A.; Gay, R.E.; Heppner, F.L.; Michel, B.A.; Gay, S.; Sprott, H. Detection of Kappa and Delta Opioid Receptors in Skin—Outside the Nervous System. Biochem. Biophys. Res. Commun. 2005, 338, 1012–1017. [Google Scholar] [CrossRef]
- Takahashi, N.; Tominaga, M.; Kosaka, R.; Kamata, Y.; Umehara, Y.; Matsuda, H.; Sakaguchi, A.; Ogawa, H.; Takamori, K. Involvement of Μ-Opioid Receptors and κ-Opioid Receptors in Itch-Related Scratching Behaviour of Imiquimod-Induced Psoriasis-like Dermatitis in Mice. Acta Derm. Venereol. 2017, 97, 928–933. [Google Scholar] [CrossRef]
- Wintzen, M.; de Winter, S.; Out-Luiting, J.J.; van Duinen, S.G.; Vermeer, B.J. Presence of Immunoreactive Beta-Endorphin in Human Skin. Exp. Dermatol. 2001, 10, 305–311. [Google Scholar] [CrossRef]
- Shah, P.K.; Borchardt, R.T. A Comparison of Peptidase Activities and Peptide Metabolism in Cultured Mouse Keratinocytes and Neonatal Mouse Epidermis. Pharm. Res. 1991, 8, 70–75. [Google Scholar] [CrossRef]
- Nissen, J.B.; Lund, M.; Stengaard-Pedersen, K.; Kragballe, K. Enkephalin-like Immunoreactivity in Human Skin Is Found Selectively in a Fraction of CD68-Positive Dermal Cells: Increase in Enkephalin-Positive Cells in Lesional Psoriasis. Arch. Dermatol. Res. 1997, 289, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Bigliardi-Qi, M.; Gaveriaux-Ruff, C.; Zhou, H.; Hell, C.; Bady, P.; Rufli, T.; Kieffer, B.; Bigliardi, P. Deletion of Delta-Opioid Receptor in Mice Alters Skin Differentiation and Delays Wound Healing. Differ. Res. Biol. Divers. 2006, 74, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Deo, P.N.; Deshmukh, R. Pathophysiology of Keratinization. J. Oral Maxillofac. Pathol. 2018, 22, 86–91. [Google Scholar] [CrossRef]
- Yang, L.; Fan, X.; Cui, T.; Dang, E.; Wang, G. Nrf2 Promotes Keratinocyte Proliferation in Psoriasis through Up-Regulation of Keratin 6, Keratin 16, and Keratin 17. J. Investig. Dermatol. 2017, 137, 2168–2176. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yin, M.; Zhang, L.-J. Keratin 6, 16 and 17-Critical Barrier Alarmin Molecules in Skin Wounds and Psoriasis. Cells 2019, 8, 807. [Google Scholar] [CrossRef] [PubMed]
- POU2F3 POU Class 2 Homeobox 3 [Homo Sapiens (Human)]—Gene—NCBI. Available online: https://www.ncbi.nlm.nih.gov/gene/25833 (accessed on 14 December 2021).
- Bigliardi, P.L.; Neumann, C.; Teo, Y.L.; Pant, A.; Bigliardi-Qi, M. Activation of the δ-Opioid Receptor Promotes Cutaneous Wound Healing by Affecting Keratinocyte Intercellular Adhesion and Migration. Br. J. Pharmacol. 2015, 172, 501–514. [Google Scholar] [CrossRef] [PubMed]
- Bigliardi-Qi, M.; Gaveriaux-Ruff, C.; Pfaltz, K.; Bady, P.; Baumann, T.; Rufli, T.; Kieffer, B.L.; Bigliardi, P.L. Deletion of Mu- and Kappa-Opioid Receptors in Mice Changes Epidermal Hypertrophy, Density of Peripheral Nerve Endings, and Itch Behavior. J. Investig. Dermatol. 2007, 127, 1479–1488. [Google Scholar] [CrossRef] [PubMed]
- Soeberdt, M.; Molenveld, P.; Storcken, R.P.M.; Bouzanne des Mazery, R.; Sterk, G.J.; Autar, R.; Bolster, M.G.; Wagner, C.; Aerts, S.N.H.; van Holst, F.R.; et al. Design and Synthesis of Enantiomerically Pure Decahydroquinoxalines as Potent and Selective κ-Opioid Receptor Agonists with Anti-Inflammatory Activity in Vivo. J. Med. Chem. 2017, 60, 2526–2551. [Google Scholar] [CrossRef]
- Chéret, J.; Gherardini, J.; Soeberdt, M.; Hundt, J.E.; Abels, C.; Bertolini, M.; Paus, R. Non-Neuronal Kappa-Opioid Receptor Activation Enhances Epidermal Keratinocyte Proliferation, and Modulates Mast Cell Functions in Human Skin Ex Vivo. J. Dermatol. 2020, 47, 917–921. [Google Scholar] [CrossRef]
- Bigliardi, P.L.; Büchner, S.; Rufli, T.; Bigliardi-Qi, M. Specific Stimulation of Migration of Human Keratinocytes by Mu-Opiate Receptor Agonists. J. Recept. Signal Transduct. Res. 2002, 22, 191–199. [Google Scholar] [CrossRef]
- Yang, D.J.; Lee, K.S.; Ko, C.M.; Moh, S.H.; Song, J.; Hur, L.C.; Cheon, Y.W.; Yang, S.H.; Choi, Y.-H.; Kim, K.W. Leucine-Enkephalin Promotes Wound Repair through the Regulation of Hemidesmosome Dynamics and Matrix Metalloprotease. Peptides 2016, 76, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.J.; Moh, S.H.; Choi, Y.-H.; Kim, K.W. β-Neoendorphin Enhances Wound Healing by Promoting Cell Migration in Keratinocyte. Molecules 2020, 25, 4640. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.; Liu, H.; Fu, X.; Sheng, Z.; Qiu, J.; Cao, R. Expression of beta-endorphin and micro-opioid receptor during wound healing process in rat with deep partial-thickness scald. Zhonghua Shao Shang Za Zhi 2007, 23, 36–39. [Google Scholar] [PubMed]
- Cheng, B.; Liu, H.-W.; Fu, X.-B.; Sheng, Z.-Y.; Li, J.-F. Coexistence and Upregulation of Three Types of Opioid Receptors, Mu, Delta and Kappa, in Human Hypertrophic Scars. Br. J. Dermatol. 2008, 158, 713–720. [Google Scholar] [CrossRef]
- Bigliardi-Qi, M.; Bigliardi, P.L.; Eberle, A.N.; Büchner, S.; Rufli, T. Beta-Endorphin Stimulates Cytokeratin 16 Expression and Downregulates Mu-Opiate Receptor Expression in Human Epidermis. J. Investig. Dermatol. 2000, 114, 527–532. [Google Scholar] [CrossRef]
- Bigliardi, P.L.; Sumanovski, L.T.; Büchner, S.; Rufli, T.; Bigliardi-Qi, M. Different Expression of Mu-Opiate Receptor in Chronic and Acute Wounds and the Effect of Beta-Endorphin on Transforming Growth Factor Beta Type II Receptor and Cytokeratin 16 Expression. J. Investig. Dermatol. 2003, 120, 145–152. [Google Scholar] [CrossRef]
- Nissen, J.B.; Kragballe, K. Enkephalins Modulate Differentiation of Normal Human Keratinocytes in Vitro. Exp. Dermatol. 1997, 6, 222–229. [Google Scholar] [CrossRef]
- Bai, X.; Cao, X.; Qu, N.; Huang, H.; Handley, M.; Zhang, S.; Shan, F. Methionine Enkephalin Activates Autophagy and Stimulates Tumour Cell Immunogenicity in Human Cutaneous Squamous Cell Carcinoma. Int. Immunopharmacol. 2021, 96, 107733. [Google Scholar] [CrossRef]
- Shime, H.; Odanaka, M.; Tsuiji, M.; Matoba, T.; Imai, M.; Yasumizu, Y.; Uraki, R.; Minohara, K.; Watanabe, M.; Bonito, A.J.; et al. Proenkephalin + Regulatory T Cells Expanded by Ultraviolet B Exposure Maintain Skin Homeostasis with a Healing Function. Proc. Natl. Acad. Sci. USA 2020, 117, 20696–20705. [Google Scholar] [CrossRef]
- Nissen, J.B.; Egekvist, H.; Bjerring, P.; Kragballe, K. Effect of Intradermal Injection of Methionine-Enkephalin on Human Skin. Acta Derm. Venereol. 1999, 79, 23–26. [Google Scholar] [CrossRef]
- Nagui, N.A.; Ezzat, M.A.; Abdel Raheem, H.M.; Rashed, L.A.; Abozaid, N.A. Possible Role of Proenkephalin in Psoriasis. Clin. Exp. Dermatol. 2016, 41, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Nissen, J.B.; Avrach, W.W.; Hansen, E.S.; Stengaard-Pedersen, K.; Kragballe, K. Decrease in Enkephalin Levels in Psoriatic Lesions after Calcipotriol and Mometasone Furoate Treatment. Dermatology 1999, 198, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Al-Hashimi, M.; Scott, S.W.M.; Thompson, J.P.; Lambert, D.G. Opioids and Immune Modulation: More Questions than Answers. Br. J. Anaesth. 2013, 111, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Fell, G.L.; Robinson, K.C.; Mao, J.; Woolf, C.J.; Fisher, D.E. Skin β-Endorphin Mediates Addiction to UV Light. Cell 2014, 157, 1527–1534. [Google Scholar] [CrossRef]
- Albers, I.; Zernickel, E.; Stern, M.; Broja, M.; Busch, H.L.; Heiss, C.; Grotheer, V.; Windolf, J.; Suschek, C.V. Blue Light (Λ = 453 nm) Nitric Oxide Dependently Induces β-Endorphin Production of Human Skin Keratinocytes in-Vitro and Increases Systemic β-Endorphin Levels in Humans in-Vivo. Free Radic. Biol. Med. 2019, 145, 78–86. [Google Scholar] [CrossRef]
- Glinski, W.; Brodecka, H.; Glinska-Ferenz, M.; Kowalski, D. Increased Concentration of Beta-Endorphin in the Sera of Patients with Severe Atopic Dermatitis. Acta Derm. Venereol. 1995, 75, 9–11. [Google Scholar] [CrossRef]
- Lee, C.-H.; Chuang, H.-Y.; Shih, C.-C.; Jong, S.-B.; Chang, C.-H.; Yu, H.-S. Transepidermal Water Loss, Serum IgE and Beta-Endorphin as Important and Independent Biological Markers for Development of Itch Intensity in Atopic Dermatitis. Br. J. Dermatol. 2006, 154, 1100–1107. [Google Scholar] [CrossRef]
- Rupprecht, M.; Hornstein, O.P.; Schlüter, D.; Schäfers, H.J.; Koch, H.U.; Beck, G.; Rupprecht, R. Cortisol, Corticotropin, and Beta-Endorphin Responses to Corticotropin-Releasing Hormone in Patients with Atopic Eczema. Psychoneuroendocrinology 1995, 20, 543–551. [Google Scholar] [CrossRef]
- Lee, C.-H.; Hong, C.-H.; Yu, W.-T.; Chuang, H.-Y.; Huang, S.-K.; Chen, G.-S.; Yoshioka, T.; Sakata, M.; Liao, W.-T.; Ko, Y.-C.; et al. Mechanistic Correlations between Two Itch Biomarkers, Cytokine Interleukin-31 and Neuropeptide β-Endorphin, via STAT3/Calcium Axis in Atopic Dermatitis. Br. J. Dermatol. 2012, 167, 794–803. [Google Scholar] [CrossRef]
- Pilozzi, A.; Carro, C.; Huang, X. Roles of β-Endorphin in Stress, Behavior, Neuroinflammation, and Brain Energy Metabolism. Int. J. Mol. Sci. 2020, 22, 338. [Google Scholar] [CrossRef]
- Hosoi, J.; Ozawa, H.; Granstein, R.D. Beta-Endorphin Binding and Regulation of Cytokine Expression in Langerhans Cells. Ann. N. Y. Acad. Sci. 1999, 885, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T. Interleukin-13: Targeting an Underestimated Cytokine in Atopic Dermatitis. Allergy 2020, 75, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Ratnarajah, K.; Le, M.; Muntyanu, A.; Mathieu, S.; Nigen, S.; Litvinov, I.V.; Jack, C.S.; Netchiporouk, E. Inhibition of IL-13: A New Pathway for Atopic Dermatitis. J. Cutan. Med. Surg. 2021, 25, 315–328. [Google Scholar] [CrossRef]
- Bénard, A.; Cavaillès, P.; Boué, J.; Chapey, E.; Bayry, J.; Blanpied, C.; Meyer, N.; Lamant, L.; Kaveri, S.V.; Brousset, P.; et al. Mu-Opioid Receptor Is Induced by IL-13 within Lymph Nodes from Patients with Sézary Syndrome. J. Investig. Dermatol. 2010, 130, 1337–1344. [Google Scholar] [CrossRef]
- Frink, M.; Hsieh, Y.-C.; Hsieh, C.-H.; Pape, H.-C.; Choudhry, M.A.; Schwacha, M.G.; Chaudry, I.H. Keratinocyte-Derived Chemokine Plays a Critical Role in the Induction of Systemic Inflammation and Tissue Damage after Trauma-Hemorrhage. Shock 2007, 28, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Kaczyńska, K.; Kogut, E.; Zając, D.; Jampolska, M.; Andrzejewski, K.; Sulejczak, D.; Lipkowski, A.W.; Kleczkowska, P. Neurotensin-Based Hybrid Peptide’s Anti-Inflammatory Activity in Murine Model of a Contact Sensitivity Response. Eur. J. Pharm. Sci. 2016, 93, 84–89. [Google Scholar] [CrossRef]
- Clark, J.D.; Shi, X.; Li, X.; Qiao, Y.; Liang, D.; Angst, M.S.; Yeomans, D.C. Morphine Reduces Local Cytokine Expression and Neutrophil Infiltration after Incision. Mol. Pain 2007, 3, 28. [Google Scholar] [CrossRef] [PubMed]
- Earl, J.R.; Grootveld, M.C.; Blake, D.R.; Morris, C.J. Effect of Mu, Delta and Kappa Opioid Receptor Agonists on a Reactive Oxygen Species Mediated Model of Skin Inflammation. Skin Pharmacol. 1996, 9, 250–258. [Google Scholar] [CrossRef]
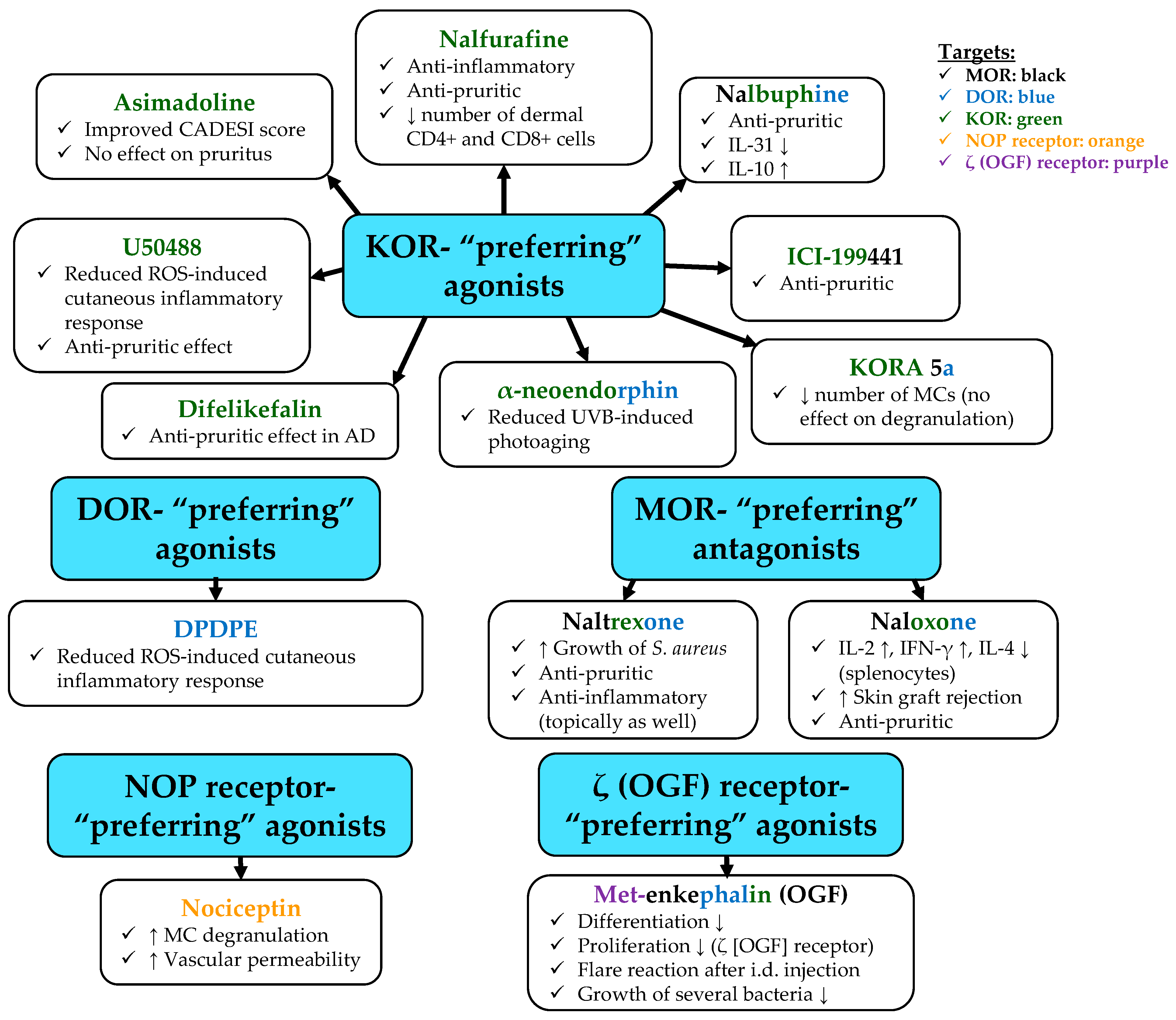
- Inan, S.; Torres-Huerta, A.; Jensen, L.E.; Dun, N.J.; Cowan, A. Nalbuphine, a Kappa Opioid Receptor Agonist and Mu Opioid Receptor Antagonist Attenuates Pruritus, Decreases IL-31, and Increases IL-10 in Mice with Contact Dermatitis. Eur. J. Pharmacol. 2019, 864, 172702. [Google Scholar] [CrossRef]
- Elliott, G.; Vanwersch, R.; Soeberdt, M.; Metze, D.; Lotts, T.; Ständer, S.; Abels, C. Topical Nalfurafine Exhibits Anti-Inflammatory and Anti-Pruritic Effects in a Murine Model of AD. J. Dermatol. Sci. 2016, 84, 351–354. [Google Scholar] [CrossRef]
- Marsella, R.; Ahrens, K.; Wilkes, R.; Soeberdt, M.; Abels, C. Topical κ-Opioid Receptor Agonist Asimadoline Improves Dermatitis in a Canine Model of Atopic Dermatitis. Exp. Dermatol. 2021, 31, 628–632. [Google Scholar] [CrossRef] [PubMed]
- Ádám, D.; Tóth, K.F.; Sárkány, F.; Soeberdt, M.; Abels, C.; Oláh, A.; Bíró, T. Activation of Κ-opioid Receptor (KOR) Suppresses Pro-inflammatory Response of Human Epidermal Keratinocytes. Exp. Dermatol. 2018, 27, 38. [Google Scholar] [CrossRef]
- Sucker, N.; Lüdiger, A.; Soeberdt, M.; Abels, C.; Luger, T.A.; Loser, K. Activation of Kappa-Opioid Receptor Signaling Ameliorates Ongoinginflammation in the Skin and the Gut. Exp. Dermatol. 2014, 23, e27. [Google Scholar] [CrossRef]
- Kilic, A.; Soeberdt, M.; Abels, C. Pruritus in Inflammatory Skin Diseases—A Brain Issue? Exp. Dermatol. 2019, 28, e85. [Google Scholar] [CrossRef]
- Lim, G.-E.; Park, J.E.; Cho, Y.H.; Lim, D.S.; Kim, A.-J.; Moh, S.H.; Lee, J.H.; Lee, J.S. Alpha-Neoendorphin Can Reduce UVB-Induced Skin Photoaging by Activating Cellular Autophagy. Arch. Biochem. Biophys. 2020, 689, 108437. [Google Scholar] [CrossRef]
- Ekelem, C.; Juhasz, M.; Khera, P.; Mesinkovska, N.A. Utility of Naltrexone Treatment for Chronic Inflammatory Dermatologic Conditions: A Systematic Review. JAMA Dermatol. 2019, 155, 229–236. [Google Scholar] [CrossRef]
- Nakashima, C.; Ishida, Y.; Kitoh, A.; Otsuka, A.; Kabashima, K. Interaction of Peripheral Nerves and Mast Cells, Eosinophils, and Basophils in the Development of Pruritus. Exp. Dermatol. 2019, 28, 1405–1411. [Google Scholar] [CrossRef]
- Wang, F.; Yang, T.-L.B.; Kim, B.S. The Return of the Mast Cell: New Roles in Neuroimmune Itch Biology. J. Investig. Dermatol. 2020, 140, 945–951. [Google Scholar] [CrossRef]
- Casale, T.B.; Bowman, S.; Kaliner, M. Induction of Human Cutaneous Mast Cell Degranulation by Opiates and Endogenous Opioid Peptides: Evidence for Opiate and Nonopiate Receptor Participation. J. Allergy Clin. Immunol. 1984, 73, 775–781. [Google Scholar] [CrossRef]
- Levy, J.H.; Brister, N.W.; Shearin, A.; Ziegler, J.; Hug, C.C.; Adelson, D.M.; Walker, B.F. Wheal and Flare Responses to Opioids in Humans. Anesthesiology 1989, 70, 756–760. [Google Scholar] [CrossRef]
- Blunk, J.A.; Schmelz, M.; Zeck, S.; Skov, P.; Likar, R.; Koppert, W. Opioid-Induced Mast Cell Activation and Vascular Responses Is Not Mediated by Mu-Opioid Receptors: An in Vivo Microdialysis Study in Human Skin. Anesth. Analg. 2004, 98, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Babina, M.; Wang, Z.; Roy, S.; Guhl, S.; Franke, K.; Artuc, M.; Ali, H.; Zuberbier, T. MRGPRX2 Is the Codeine Receptor of Human Skin Mast Cells: Desensitization through β-Arrestin and Lack of Correlation with the FcεRI Pathway. J. Investig. Dermatol. 2020, 141, 1286–1296.e4. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Kitaichi, K.; Hiramatsu, K.; Yoshida, M.; Ito, Y.; Kume, H.; Yamaki, K.; Suzuki, R.; Takagi, K. Intradermal Application of Nociceptin Increases Vascular Permeability in Rats: The Possible Involvement of Histamine Release from Mast Cells. Eur. J. Pharmacol. 2000, 407, 327–332. [Google Scholar] [CrossRef]
- Jackson-Cowan, L.; Cole, E.F.; Arbiser, J.L.; Silverberg, J.I.; Lawley, L.P. TH2 Sensitization in the Skin-Gut-Brain Axis: How Early-Life Th2-Mediated Inflammation May Negatively Perpetuate Developmental and Psychologic Abnormalities. Pediatr. Dermatol. 2021, 38, 1032–1039. [Google Scholar] [CrossRef] [PubMed]
- Sacerdote, P.; Gaspani, L.; Panerai, A.E. The Opioid Antagonist Naloxone Induces a Shift from Type 2 to Type 1 Cytokine Pattern in Normal and Skin-Grafted Mice. Ann. N. Y. Acad. Sci. 2000, 917, 755–763. [Google Scholar] [CrossRef]
- Sacerdote, P.; Manfredi, B.; Gaspani, L.; Panerai, A.E. The Opioid Antagonist Naloxone Induces a Shift from Type 2 to Type 1 Cytokine Pattern in BALB/CJ Mice. Blood 2000, 95, 2031–2036. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Balasubramanian, S.; Sumandeep, S.; Charboneau, R.; Wang, J.; Melnyk, D.; Beilman, G.J.; Vatassery, R.; Barke, R.A. Morphine Directs T Cells toward T(H2) Differentiation. Surgery 2001, 130, 304–309. [Google Scholar] [CrossRef]
- Roy, S.; Wang, J.; Charboneau, R.; Loh, H.H.; Barke, R.A. Morphine Induces CD4+ T Cell IL-4 Expression through an Adenylyl Cyclase Mechanism Independent of the Protein Kinase A Pathway. J. Immunol. 2005, 175, 6361–6367. [Google Scholar] [CrossRef]
- Karaji, A.G.; Reiss, D.; Matifas, A.; Kieffer, B.L.; Gavériaux-Ruff, C. Influence of Endogenous Opioid Systems on T Lymphocytes as Assessed by the Knockout of Mu, Delta and Kappa Opioid Receptors. J. Neuroimmune Pharmacol. 2011, 6, 608–616. [Google Scholar] [CrossRef]
- Di Domenico, E.G.; Cavallo, I.; Capitanio, B.; Ascenzioni, F.; Pimpinelli, F.; Morrone, A.; Ensoli, F. Staphylococcus aureus and the Cutaneous Microbiota Biofilms in the Pathogenesis of Atopic Dermatitis. Microorganisms 2019, 7, 301. [Google Scholar] [CrossRef]
- Hong, S.-W.; Kim, M.-R.; Lee, E.-Y.; Kim, J.H.; Kim, Y.-S.; Jeon, S.G.; Yang, J.-M.; Lee, B.-J.; Pyun, B.-Y.; Gho, Y.S.; et al. Extracellular Vesicles Derived from Staphylococcus aureus Induce Atopic Dermatitis-like Skin Inflammation. Allergy 2011, 66, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Vlachos, C.; Gaitanis, G.; Alexopoulos, E.C.; Papadopoulou, C.; Bassukas, I.D. Phospholipase Activity after β-Endorphin Exposure Discriminates Malassezia Strains Isolated from Healthy and Seborrhoeic Dermatitis Skin. J. Eur. Acad. Dermatol. Venereol. 2013, 27, 1575–1578. [Google Scholar] [CrossRef] [PubMed]
- Zagon, I.S.; McLaughlin, P.J. An Opioid Growth Factor Regulates the Replication of Microorganisms. Life Sci. 1992, 50, 1179–1187. [Google Scholar] [CrossRef]
- Toll, L.; Berzetei-Gurske, I.P.; Polgar, W.E.; Brandt, S.R.; Adapa, I.D.; Rodriguez, L.; Schwartz, R.W.; Haggart, D.; O’Brien, A.; White, A.; et al. Standard Binding and Functional Assays Related to Medications Development Division Testing for Potential Cocaine and Opiate Narcotic Treatment Medications. NIDA Res. Monogr. 1998, 178, 440–466. [Google Scholar] [PubMed]
- Poulain, R.; Horvath, D.; Bonnet, B.; Eckhoff, C.; Chapelain, B.; Bodinier, M.C.; Déprez, B. From Hit to Lead. Combining Two Complementary Methods for Focused Library Design. Application to Mu Opiate Ligands. J. Med. Chem. 2001, 44, 3378–3390. [Google Scholar] [CrossRef]
- Rota, S.; Kaya, K.; Timliothlu, O.; Karaca, O.; Yzdep, S.; Ocal, E. Do the Opioids Have an Antibacterial Effect? Can. J. Anaesth. 1997, 44, 679–680. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, P.H.; Renkonen, O.V. Antimicrobial Activity of Bupivacaine and Morphine. Anesthesiology 1985, 62, 178–179. [Google Scholar] [CrossRef] [PubMed]
- Karas, J.A.; Wong, L.J.M.; Paulin, O.K.A.; Mazeh, A.C.; Hussein, M.H.; Li, J.; Velkov, T. The Antimicrobial Activity of Cannabinoids. Antibiotics 2020, 9, 406. [Google Scholar] [CrossRef]
- Flury, U.; Cahill, J.L.; Nixon, R.L. Occupational Contact Dermatitis Caused by Opioids: A Case Series. Contact Dermat. 2019, 81, 332–335. [Google Scholar] [CrossRef]
- Ständer, S.; Steinhoff, M.; Schmelz, M.; Weisshaar, E.; Metze, D.; Luger, T. Neurophysiology of Pruritus: Cutaneous Elicitation of Itch. Arch. Dermatol. 2003, 139, 1463–1470. [Google Scholar] [CrossRef]
- Meng, J.; Steinhoff, M. Molecular Mechanisms of Pruritus. Curr. Res. Transl. Med. 2016, 64, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Tóth, B.I.; Szallasi, A.; Bíró, T. Transient Receptor Potential Channels and Itch: How Deep Should We Scratch? Handb. Exp. Pharmacol. 2015, 226, 89–133. [Google Scholar] [CrossRef] [PubMed]
- Yosipovitch, G.; Rosen, J.D.; Hashimoto, T. Itch: From Mechanism to (Novel) Therapeutic Approaches. J. Allergy Clin. Immunol. 2018, 142, 1375–1390. [Google Scholar] [CrossRef] [PubMed]
- Yosipovitch, G.; Arendt-Nielsen, L.; Andersen, H. Itch and Pain: Similarities, Interactions, and Differences. Wolters Kluwer Health: Philadelphia, PA, USA, 2020; ISBN 978-1-975153-03-8. [Google Scholar]
- Frazier, W.; Bhardwaj, N. Atopic Dermatitis: Diagnosis and Treatment. Am. Fam. Physician 2020, 101, 590–598. [Google Scholar] [PubMed]
- Ishiuji, Y. Addiction and the Itch-Scratch Cycle. What Do They Have in Common? Exp. Dermatol. 2019, 28, 1448–1454. [Google Scholar] [CrossRef]
- Greaves, M.W. Pathogenesis and Treatment of Pruritus. Curr. Allergy Asthma Rep. 2010, 10, 236–242. [Google Scholar] [CrossRef]
- Sutaria, N.; Adawi, W.; Goldberg, R.; Roh, Y.S.; Choi, J.; Kwatra, S.G. Itch: Pathogenesis and Treatment. J. Am. Acad. Dermatol. 2021, 86, 17–34. [Google Scholar] [CrossRef]
- Umehara, Y.; Kiatsurayanon, C.; Trujillo-Paez, J.V.; Chieosilapatham, P.; Peng, G.; Yue, H.; Nguyen, H.L.T.; Song, P.; Okumura, K.; Ogawa, H.; et al. Intractable Itch in Atopic Dermatitis: Causes and Treatments. Biomedicines 2021, 9, 229. [Google Scholar] [CrossRef]
- Lipman, Z.M.; Yosipovitch, G. Substance Use Disorders and Chronic Itch. J. Am. Acad. Dermatol. 2021, 84, 148–155. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, K.; Inan, S.; Siebert, D.; Holzgrabe, U.; Lee, D.Y.W.; Huang, P.; Li, J.-G.; Cowan, A.; Liu-Chen, L.-Y. Comparison of Pharmacological Activities of Three Distinct Kappa Ligands (Salvinorin A, TRK-820 and 3FLB) on Kappa Opioid Receptors in Vitro and Their Antipruritic and Antinociceptive Activities in Vivo. J. Pharmacol. Exp. Ther. 2005, 312, 220–230. [Google Scholar] [CrossRef]
- Lee, B.; Elston, D.M. The Uses of Naltrexone in Dermatologic Conditions. J. Am. Acad. Dermatol. 2019, 80, 1746–1752. [Google Scholar] [CrossRef] [PubMed]
- Reich, A.; Szepietowski, J.C. Non-Analgesic Effects of Opioids: Peripheral Opioid Receptors as Promising Targets for Future Anti-Pruritic Therapies. Curr. Pharm. Des. 2012, 18, 6021–6024. [Google Scholar] [CrossRef]
- Cowan, A.; Kehner, G.B.; Inan, S. Targeting Itch with Ligands Selective for κ Opioid Receptors. Handb. Exp. Pharmacol. 2015, 226, 291–314. [Google Scholar] [CrossRef]
- Phan, N.Q.; Bernhard, J.D.; Luger, T.A.; Ständer, S. Antipruritic Treatment with Systemic μ-Opioid Receptor Antagonists: A Review. J. Am. Acad. Dermatol. 2010, 63, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Phan, N.Q.; Lotts, T.; Antal, A.; Bernhard, J.D.; Ständer, S. Systemic Kappa Opioid Receptor Agonists in the Treatment of Chronic Pruritus: A Literature Review. Acta Derm. Venereol. 2012, 92, 555–560. [Google Scholar] [CrossRef]
- Bigliardi, P.L.; Bigliardi-Qi, M. Peripheral Opioids. In Itch: Mechanisms and Treatment; Carstens, E., Akiyama, T., Eds.; Frontiers in Neuroscience; CRC Press: Boca Raton, FL, USA, 2014; ISBN 978-1-4665-0543-8. [Google Scholar]
- Farmer, W.S.; Marathe, K.S. Atopic Dermatitis: Managing the Itch. In Management of Atopic Dermatitis: Methods and Challenges; Fortson, E.A., Feldman, S.R., Strowd, L.C., Eds.; Advances in Experimental Medicine and Biology; Springer International Publishing: Cham, Switzerland, 2017; pp. 161–177. ISBN 978-3-319-64804-0. [Google Scholar]
- Inan, S.; Cowan, A. Antipruritic Effects of Kappa Opioid Receptor Agonists: Evidence from Rodents to Humans. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2022; Volume 271, pp. 275–292. [Google Scholar]
- Melo, H.; Basso, L.; Iftinca, M.; MacNaughton, W.K.; Hollenberg, M.D.; McKay, D.M.; Altier, C. Itch Induced by Peripheral Mu Opioid Receptors Is Dependent on TRPV1-Expressing Neurons and Alleviated by Channel Activation. Sci. Rep. 2018, 8, 15551. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Ko, M.-C. Distinct Functions of Opioid-Related Peptides and Gastrin-Releasing Peptide in Regulating Itch and Pain in the Spinal Cord of Primates. Sci. Rep. 2015, 5, 11676. [Google Scholar] [CrossRef]
- Heyer, G.; Groene, D.; Martus, P. Efficacy of Naltrexone on Acetylcholine-Induced Alloknesis in Atopic Eczema. Exp. Dermatol. 2002, 11, 448–455. [Google Scholar] [CrossRef]
- Feng, J.; Chen, Y.; Xiong, J.; Chen, X.; Liang, J.; Ji, W. The Kinin B1 Receptor Mediates Alloknesis in a Murine Model of Inflammation. Neurosci. Lett. 2014, 560, 31–35. [Google Scholar] [CrossRef]
- Nakasone, T.; Sato, T.; Matsushima, Y.; Inoue, T.; Kamei, C. Characteristics of Scratching Behavior in ADJM Mice (Atopic Dermatitis from Japanese Mice). Immunopharmacol. Immunotoxicol. 2015, 37, 202–206. [Google Scholar] [CrossRef]
- Pandey, M.; Zhang, J.-H.; Mishra, S.K.; Adikaram, P.R.; Harris, B.; Kahler, J.F.; Loshakov, A.; Sholevar, R.; Genis, A.; Kittock, C.; et al. A Central Role for R7bp in the Regulation of Itch Sensation. Pain 2017, 158, 931–944. [Google Scholar] [CrossRef] [PubMed]
- Martemyanov, K.A.; Yoo, P.J.; Skiba, N.P.; Arshavsky, V.Y. R7BP, a Novel Neuronal Protein Interacting with RGS Proteins of the R7 Family. J. Biol. Chem. 2005, 280, 5133–5136. [Google Scholar] [CrossRef] [PubMed]
- Inan, S.; Dun, N.J.; Cowan, A. Antipruritic Effect of Nalbuphine, a Kappa Opioid Receptor Agonist, in Mice: A Pan Antipruritic. Molecules 2021, 26, 5517. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, T.; Carstens, M.I.; Piecha, D.; Steppan, S.; Carstens, E. Nalfurafine Suppresses Pruritogen- and Touch-Evoked Scratching Behavior in Models of Acute and Chronic Itch in Mice. Acta Derm. Venereol. 2015, 95, 147–150. [Google Scholar] [CrossRef]
- Szöllősi, A.G.; Oláh, A.; Lisztes, E.; Griger, Z.; Tóth, B.I. Pruritus: A Sensory Symptom Generated in Cutaneous Immuno-Neuronal Crosstalk. Front. Pharmacol. 2022, 13, 745658. [Google Scholar] [CrossRef]
- Kupari, J.; Usoskin, D.; Parisien, M.; Lou, D.; Hu, Y.; Fatt, M.; Lönnerberg, P.; Spångberg, M.; Eriksson, B.; Barkas, N.; et al. Single Cell Transcriptomics of Primate Sensory Neurons Identifies Cell Types Associated with Chronic Pain. Nat. Commun. 2021, 12, 1510. [Google Scholar] [CrossRef]
- Nguyen, M.Q.; von Buchholtz, L.J.; Reker, A.N.; Ryba, N.J.; Davidson, S. Single-Nucleus Transcriptomic Analysis of Human Dorsal Root Ganglion Neurons. eLife 2021, 10, e71752. [Google Scholar] [CrossRef]
- Usoskin, D.; Furlan, A.; Islam, S.; Abdo, H.; Lönnerberg, P.; Lou, D.; Hjerling-Leffler, J.; Haeggström, J.; Kharchenko, O.; Kharchenko, P.V.; et al. Unbiased Classification of Sensory Neuron Types by Large-Scale Single-Cell RNA Sequencing. Nat. Neurosci. 2015, 18, 145–153. [Google Scholar] [CrossRef]
- LaMotte, R.H.; Dong, X.; Ringkamp, M. Sensory Neurons and Circuits Mediating Itch. Nat. Rev. Neurosci. 2014, 15, 19–31. [Google Scholar] [CrossRef]
- Dong, X.; Dong, X. Peripheral and Central Mechanisms of Itch. Neuron 2018, 98, 482–494. [Google Scholar] [CrossRef]
- Ikoma, A.; Steinhoff, M.; Ständer, S.; Yosipovitch, G.; Schmelz, M. The Neurobiology of Itch. Nat. Rev. Neurosci. 2006, 7, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Xu, Q.; Guo, C.; Guan, Y.; Liu, Q.; Dong, X. Leaky Gate Model: Intensity-Dependent Coding of Pain and Itch in the Spinal Cord. Neuron 2017, 93, 840–853.e5. [Google Scholar] [CrossRef]
- Sun, Y.-G.; Chen, Z.-F. A Gastrin-Releasing Peptide Receptor Mediates the Itch Sensation in the Spinal Cord. Nature 2007, 448, 700–703. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-J.; Sun, Y.-G. Central Circuit Mechanisms of Itch. Nat. Commun. 2020, 11, 3052. [Google Scholar] [CrossRef] [PubMed]
- Davidson, S.; Zhang, X.; Khasabov, S.G.; Simone, D.A.; Giesler, G.J. Relief of Itch by Scratching: State-Dependent Inhibition of Primate Spinothalamic Tract Neurons. Nat. Neurosci. 2009, 12, 544–546. [Google Scholar] [CrossRef]
- Ross, S.E.; Mardinly, A.R.; McCord, A.E.; Zurawski, J.; Cohen, S.; Jung, C.; Hu, L.; Mok, S.I.; Shah, A.; Savner, E.M.; et al. Loss of Inhibitory Interneurons in the Dorsal Spinal Cord and Elevated Itch in Bhlhb5 Mutant Mice. Neuron 2010, 65, 886–898. [Google Scholar] [CrossRef]
- Chiang, M.C.; Hachisuka, J.; Todd, A.J.; Ross, S.E. Insight into B5-I Spinal Interneurons and Their Role in the Inhibition of Itch and Pain. Pain 2016, 157, 544–545. [Google Scholar] [CrossRef]
- Kardon, A.P.; Polgár, E.; Hachisuka, J.; Snyder, L.M.; Cameron, D.; Savage, S.; Cai, X.; Karnup, S.; Fan, C.R.; Hemenway, G.M.; et al. Dynorphin Acts as a Neuromodulator to Inhibit Itch in the Dorsal Horn of the Spinal Cord. Neuron 2014, 82, 573–586. [Google Scholar] [CrossRef]
- Snyder, L.M.; Ross, S.E. Itch and Its Inhibition by Counter Stimuli. Handb. Exp. Pharmacol. 2015, 226, 191–206. [Google Scholar] [CrossRef]
- Snyder, L.M.; Chiang, M.C.; Loeza-Alcocer, E.; Omori, Y.; Hachisuka, J.; Sheahan, T.D.; Gale, J.R.; Adelman, P.C.; Sypek, E.I.; Fulton, S.A.; et al. Kappa Opioid Receptor Distribution and Function in Primary Afferents. Neuron 2018, 99, 1274–1288.e6. [Google Scholar] [CrossRef]
- Liu, X.-Y.; Liu, Z.-C.; Sun, Y.-G.; Ross, M.; Kim, S.; Tsai, F.-F.; Li, Q.-F.; Jeffry, J.; Kim, J.-Y.; Loh, H.H.; et al. Unidirectional Cross-Activation of GRPR by MOR1D Uncouples Itch and Analgesia Induced by Opioids. Cell 2011, 147, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.-Y.; Ginosar, Y.; Yazdi, J.; Hincker, A.; Chen, Z.-F. Cross-Talk between Human Spinal Cord μ-Opioid Receptor 1Y Isoform and Gastrin-Releasing Peptide Receptor Mediates Opioid-Induced Scratching Behavior. Anesthesiology 2019, 131, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, E.; Lim, G.; Ross, S.E. Evaluation of Therapies for Peripheral and Neuraxial Opioid-Induced Pruritus Based on Molecular and Cellular Discoveries. Anesthesiology 2021, 135, 350–365. [Google Scholar] [CrossRef] [PubMed]
- Sathyamurthy, A.; Johnson, K.R.; Matson, K.J.E.; Dobrott, C.I.; Li, L.; Ryba, A.R.; Bergman, T.B.; Kelly, M.C.; Kelley, M.W.; Levine, A.J. Massively Parallel Single Nucleus Transcriptional Profiling Defines Spinal Cord Neurons and Their Activity during Behavior. Cell Rep. 2018, 22, 2216–2225. [Google Scholar] [CrossRef]
- Häring, M.; Zeisel, A.; Hochgerner, H.; Rinwa, P.; Jakobsson, J.E.T.; Lönnerberg, P.; La Manno, G.; Sharma, N.; Borgius, L.; Kiehn, O.; et al. Neuronal Atlas of the Dorsal Horn Defines Its Architecture and Links Sensory Input to Transcriptional Cell Types. Nat. Neurosci. 2018, 21, 869–880. [Google Scholar] [CrossRef]
- Nguyen, E.; Lim, G.; Ding, H.; Hachisuka, J.; Ko, M.-C.; Ross, S.E. Morphine Acts on Spinal Dynorphin Neurons to Cause Itch through Disinhibition. Sci. Transl. Med. 2021, 13, eabc3774. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, C.; Yao, H.; Chen, O.; Rahman, S.; Gu, Y.; Zhao, J.; Huh, Y.; Ji, R.-R. Central Opioid Receptors Mediate Morphine-Induced Itch and Chronic Itch via Disinhibition. Brain J. Neurol. 2021, 144, 665–681. [Google Scholar] [CrossRef]
- Lansu, K.; Karpiak, J.; Liu, J.; Huang, X.-P.; McCorvy, J.D.; Kroeze, W.K.; Che, T.; Nagase, H.; Carroll, F.I.; Jin, J.; et al. In Silico Design of Novel Probes for the Atypical Opioid Receptor MRGPRX2. Nat. Chem. Biol. 2017, 13, 529–536. [Google Scholar] [CrossRef]
- Dickie, A.C.; Bell, A.M.; Iwagaki, N.; Polgár, E.; Gutierrez-Mecinas, M.; Kelly, R.; Lyon, H.; Turnbull, K.; West, S.J.; Etlin, A.; et al. Morphological and Functional Properties Distinguish the Substance P and Gastrin-Releasing Peptide Subsets of Excitatory Interneuron in the Spinal Cord Dorsal Horn. Pain 2019, 160, 442–462. [Google Scholar] [CrossRef]
- Albisetti, G.W.; Pagani, M.; Platonova, E.; Hösli, L.; Johannssen, H.C.; Fritschy, J.-M.; Wildner, H.; Zeilhofer, H.U. Dorsal Horn Gastrin-Releasing Peptide Expressing Neurons Transmit Spinal Itch But Not Pain Signals. J. Neurosci. 2019, 39, 2238–2250. [Google Scholar] [CrossRef]
- Fjellner, B.; Hägermark, O. Potentiation of Histamine-Induced Itch and Flare Responses in Human Skin by the Enkephalin Analogue FK-33-824, Beta-Endorphin and Morphine. Arch. Dermatol. Res. 1982, 274, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Heyer, G.R.; Hornstein, O.P. Recent Studies of Cutaneous Nociception in Atopic and Non-Atopic Subjects. J. Dermatol. 1999, 26, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Bigliardi, P.L.; Stammer, H.; Jost, G.; Rufli, T.; Büchner, S.; Bigliardi-Qi, M. Treatment of Pruritus with Topically Applied Opiate Receptor Antagonist. J. Am. Acad. Dermatol. 2007, 56, 979–988. [Google Scholar] [CrossRef]
- Sullivan, J.R.; Watson, A. Naltrexone: A Case Report of Pruritus from an Antipruritic. Australas. J. Dermatol. 1997, 38, 196–198. [Google Scholar] [CrossRef]
- Inui, S. Nalfurafine Hydrochloride to Treat Pruritus: A Review. Clin. Cosmet. Investig. Dermatol. 2015, 8, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Kremer, A.E.; Mettang, T. Pruritus in systemic diseases: Common and rare etiologies. Internist 2019, 60, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, H.; Ebata, T.; Takamori, K.; Miyasato, K.; Muramatsu, T.; Nakamoto, H.; Kurihara, M.; Yanagita, T.; Suzuki, H. Efficacy and Safety of a Novel ĸ-Agonist for Managing Intractable Pruritus in Dialysis Patients. Am. J. Nephrol. 2012, 36, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, A.; Krajewski, P.; Kozioł-Gałczyńska, M.; Szepietowski, J.C. Opioid Receptors Expression in the Skin of Haemodialysis Patients Suffering from Uraemic Pruritus. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 2368–2372. [Google Scholar] [CrossRef]
- Kupczyk, P.; Reich, A.; Hołysz, M.; Gajda, M.; Wysokińska, E.; Kobuszewska, A.; Nevozhay, D.; Nowakowska, B.; Strzadała, L.; Jagodziński, P.P.; et al. Opioid Receptors in Psoriatic Skin: Relationship with Itch. Acta Derm. Venereol. 2017, 97, 564–570. [Google Scholar] [CrossRef]
- Bigliardi-Qi, M.; Lipp, B.; Sumanovski, L.T.; Buechner, S.A.; Bigliardi, P.L. Changes of Epidermal Mu-Opiate Receptor Expression and Nerve Endings in Chronic Atopic Dermatitis. Dermatology 2005, 210, 91–99. [Google Scholar] [CrossRef]
- Schteingart, C.D.; Menzaghi, F.; Jiang, G.; Alexander, R.V.; Sueiras-Diaz, J.; Spencer, R.H.; Chalmers, D.T.; Luo, Z. Synthetic Peptide Amides. U.S. Patent US7402564 B1, 22 July 2008. [Google Scholar]
- Investor Relations|Cara Therapeutics. Available online: http://ir.caratherapeutics.com/ (accessed on 13 January 2022).
- Khodorova, A.; Navarro, B.; Jouaville, L.S.; Murphy, J.-E.; Rice, F.L.; Mazurkiewicz, J.E.; Long-Woodward, D.; Stoffel, M.; Strichartz, G.R.; Yukhananov, R.; et al. Endothelin-B Receptor Activation Triggers an Endogenous Analgesic Cascade at Sites of Peripheral Injury. Nat. Med. 2003, 9, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Szöllősi, A.G.; McDonald, I.; Szabó, I.L.; Meng, J.; van den Bogaard, E.; Steinhoff, M. TLR3 in Chronic Human Itch: A Keratinocyte-Associated Mechanism of Peripheral Itch Sensitization. J. Investig. Dermatol. 2019, 139, 2393–2396.e6. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Moriyama, M.; Feld, M.; Buddenkotte, J.; Buhl, T.; Szöllösi, A.; Zhang, J.; Miller, P.; Ghetti, A.; Fischer, M.; et al. New Mechanism Underlying IL-31-Induced Atopic Dermatitis. J. Allergy Clin. Immunol. 2018, 141, 1677–1689.e8. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Luo, R.; Chen, Y.; Sun, C.; Wang, J.; Zhou, L.; Li, Y.; Deng, L. Interleukin-31 Promotes Helper T Cell Type-2 Inflammation in Children with Allergic Rhinitis. Pediatr. Res. 2015, 77, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Yeo, H.; Ahn, S.S.; Lee, Y.H.; Shin, S.Y. Regulation of Pro-Opiomelanocortin (POMC) Gene Transcription by Interleukin-31 via Early Growth Response 1 (EGR-1) in HaCaT Keratinocytes. Mol. Biol. Rep. 2020, 47, 5953–5962. [Google Scholar] [CrossRef]
- Tsuji, M.; Arai, I.; Miyagawa, K.; Miyagishi, H.; Saito, A.; Takeda, K.; Takeda, H.; Akiyama, N.; Saito, S. Involvement of Interleukin-31 Receptor A in Morphine-Induced Itching and Antinociception in Mice. Eur. J. Pain 2019, 23, 378–388. [Google Scholar] [CrossRef]
- Nordlind, K.; Azmitia, E.C.; Slominski, A. The Skin as a Mirror of the Soul: Exploring the Possible Roles of Serotonin. Exp. Dermatol. 2008, 17, 301–311. [Google Scholar] [CrossRef]
- Jafferany, M.; Ferreira, B.R.; Abdelmaksoud, A.; Mkhoyan, R. Management of Psychocutaneous Disorders: A Practical Approach for Dermatologists. Dermatol. Ther. 2020, 33, e13969. [Google Scholar] [CrossRef]
- Theoharides, T.C. Effect of Stress on Neuroimmune Processes. Clin. Ther. 2020, 42, 1007–1014. [Google Scholar] [CrossRef]
- Tóth, B.I.; Oláh, A.; Szöllosi, A.G.; Czifra, G.; Bíró, T. “Sebocytes’ Makeup”: Novel Mechanisms and Concepts in the Physiology of the Human Sebaceous Glands. Pflug. Arch. 2011, 461, 593–606. [Google Scholar] [CrossRef]
- Szöllősi, A.G.; Oláh, A.; Bíró, T.; Tóth, B.I. Recent Advances in the Endocrinology of the Sebaceous Gland. Derm.-Endocrinol. 2017, 9, e1361576. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C. The Impact of Psychological Stress on Mast Cells. Ann. Allergy Asthma Immunol. 2020, 125, 388–392. [Google Scholar] [CrossRef]
- Slominski, A.T.; Zmijewski, M.A.; Zbytek, B.; Tobin, D.J.; Theoharides, T.C.; Rivier, J. Key Role of CRF in the Skin Stress Response System. Endocr. Rev. 2013, 34, 827–884. [Google Scholar] [CrossRef] [PubMed]
- Vasiadi, M.; Therianou, A.; Sideri, K.; Smyrnioti, M.; Sismanopoulos, N.; Delivanis, D.A.; Asadi, S.; Katsarou-Katsari, A.; Petrakopoulou, T.; Theoharides, A.; et al. Increased Serum CRH Levels with Decreased Skin CRHR-1 Gene Expression in Psoriasis and Atopic Dermatitis. J. Allergy Clin. Immunol. 2012, 129, 1410–1413. [Google Scholar] [CrossRef] [PubMed]
- Orita, K.; Hiramoto, K.; Inoue, R.; Sato, E.F.; Kobayashi, H.; Ishii, M.; Inoue, M. Strong Exercise Stress Exacerbates Dermatitis in Atopic Model Mice, NC/Nga Mice, While Proper Exercise Reduces It. Exp. Dermatol. 2010, 19, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K.; Kobayashi, H.; Sekiyama, A.; Sato, E.F.; Tsuruta, D.; Ishii, M. Mild Exercise Suppresses Exacerbation of Dermatitis by Increasing Cleavage of the β-Endorphin from Proopiomelanocortin in NC/Nga Mice. J. Clin. Biochem. Nutr. 2013, 52, 58–63. [Google Scholar] [CrossRef]
- Gao, F.; Zhang, L.-H.; Su, T.-F.; Li, L.; Zhou, R.; Peng, M.; Wu, C.-H.; Yuan, X.-C.; Sun, N.; Meng, X.-F.; et al. Signaling Mechanism of Cannabinoid Receptor-2 Activation-Induced β-Endorphin Release. Mol. Neurobiol. 2016, 53, 3616–3625. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M.; Porreca, F.; Lai, J.; Albrecht, P.J.; Rice, F.L.; Khodorova, A.; Davar, G.; Makriyannis, A.; Vanderah, T.W.; Mata, H.P.; et al. CB2 Cannabinoid Receptor Activation Produces Antinociception by Stimulating Peripheral Release of Endogenous Opioids. Proc. Natl. Acad. Sci. USA 2005, 102, 3093–3098. [Google Scholar] [CrossRef] [PubMed]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.-Z.; Xie, X.-Q.; Altmann, K.-H.; Karsak, M.; Zimmer, A. Beta-Caryophyllene Is a Dietary Cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef]
- Katsuyama, S.; Mizoguchi, H.; Kuwahata, H.; Komatsu, T.; Nagaoka, K.; Nakamura, H.; Bagetta, G.; Sakurada, T.; Sakurada, S. Involvement of Peripheral Cannabinoid and Opioid Receptors in β-Caryophyllene-Induced Antinociception. Eur. J. Pain 2013, 17, 664–675. [Google Scholar] [CrossRef]
- Jung, D.-L.; Lee, S.-D.; Choi, I.-H.; Na, H.-S.; Hong, S.-U. Effects of Electroacupuncture on Capsaicin-Induced Model of Atopic Dermatitis in Rats. J. Dermatol. Sci. 2014, 74, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Georgala, S.; Schulpis, K.H.; Papaconstantinou, E.D.; Stratigos, J. Raised Beta-Endorphin Serum Levels in Children with Atopic Dermatitis and Pruritus. J. Dermatol. Sci. 1994, 8, 125–128. [Google Scholar] [CrossRef]
- Ständer, S.; Gunzer, M.; Metze, D.; Luger, T.; Steinhoff, M. Localization of Mu-Opioid Receptor 1A on Sensory Nerve Fibers in Human Skin. Regul. Pept. 2002, 110, 75–83. [Google Scholar] [CrossRef]
- Lotts, T.; Born, I.; Ständer, S. High Epidermal Kappa Opioid Receptor Expression Levels Dominate Atopicdermatitis While in Prurigo Nodularis Mu Opioid Receptors Are Abundantlyexpressed. Exp. Dermatol. 2014, 23, e41. [Google Scholar] [CrossRef]
- Schneeweiss, M.; Lüdiger, A.; Chartolani, N.; Soeberdt, M.; Knie, U.; Luger, T.A.; Abels, C.; Loser, K. Topical Treatment with a Novel Kappa-Opioid Receptor Agonist Ameliorates Atopic Dermatitis. Exp. Dermatol. 2018, 27, e47. [Google Scholar] [CrossRef]
- Soeberdt, M.; Masur, C.; Knie, U.; Metze, D.; Abels, C. Safety, Tolerability, Pharmacokinetics and Efficacy of Kappa-Opioid Receptor Agonist WOL071-007-Containing Topical Formulations in Atopic Dermatitis Patients. Exp. Dermatol. 2018, 27, e20. [Google Scholar] [CrossRef]
- Zimprich, A.; Kraus, J.; Wöltje, M.; Mayer, P.; Rauch, E.; Höllt, V. An Allelic Variation in the Human Prodynorphin Gene Promoter Alters Stimulus-Induced Expression. J. Neurochem. 2000, 74, 472–477. [Google Scholar] [CrossRef]
- Greisenegger, E.K.; Zimprich, A.; Zimprich, F.; Stingl, G.; Kopp, T. Analysis of the Prodynorphin Promoter Polymorphism in Atopic Dermatitis and Disease-Related Pruritus. Clin. Exp. Dermatol. 2009, 34, 728–730. [Google Scholar] [CrossRef]
- Totsuka, A.; Omori-Miyake, M.; Kawashima, M.; Yagi, J.; Tsunemi, Y. Expression of Keratin 1, Keratin 10, Desmoglein 1 and Desmocollin 1 in the Epidermis: Possible Downregulation by Interleukin-4 and Interleukin-13 in Atopic Dermatitis. Eur. J. Dermatol. 2017, 27, 247–253. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ádám, D.; Arany, J.; Tóth, K.F.; Tóth, B.I.; Szöllősi, A.G.; Oláh, A. Opioidergic Signaling—A Neglected, Yet Potentially Important Player in Atopic Dermatitis. Int. J. Mol. Sci. 2022, 23, 4140. https://doi.org/10.3390/ijms23084140
Ádám D, Arany J, Tóth KF, Tóth BI, Szöllősi AG, Oláh A. Opioidergic Signaling—A Neglected, Yet Potentially Important Player in Atopic Dermatitis. International Journal of Molecular Sciences. 2022; 23(8):4140. https://doi.org/10.3390/ijms23084140
Chicago/Turabian StyleÁdám, Dorottya, József Arany, Kinga Fanni Tóth, Balázs István Tóth, Attila Gábor Szöllősi, and Attila Oláh. 2022. "Opioidergic Signaling—A Neglected, Yet Potentially Important Player in Atopic Dermatitis" International Journal of Molecular Sciences 23, no. 8: 4140. https://doi.org/10.3390/ijms23084140
APA StyleÁdám, D., Arany, J., Tóth, K. F., Tóth, B. I., Szöllősi, A. G., & Oláh, A. (2022). Opioidergic Signaling—A Neglected, Yet Potentially Important Player in Atopic Dermatitis. International Journal of Molecular Sciences, 23(8), 4140. https://doi.org/10.3390/ijms23084140








