Identification of Entry Inhibitors against Delta and Omicron Variants of SARS-CoV-2
Abstract
:1. Introduction
2. Results
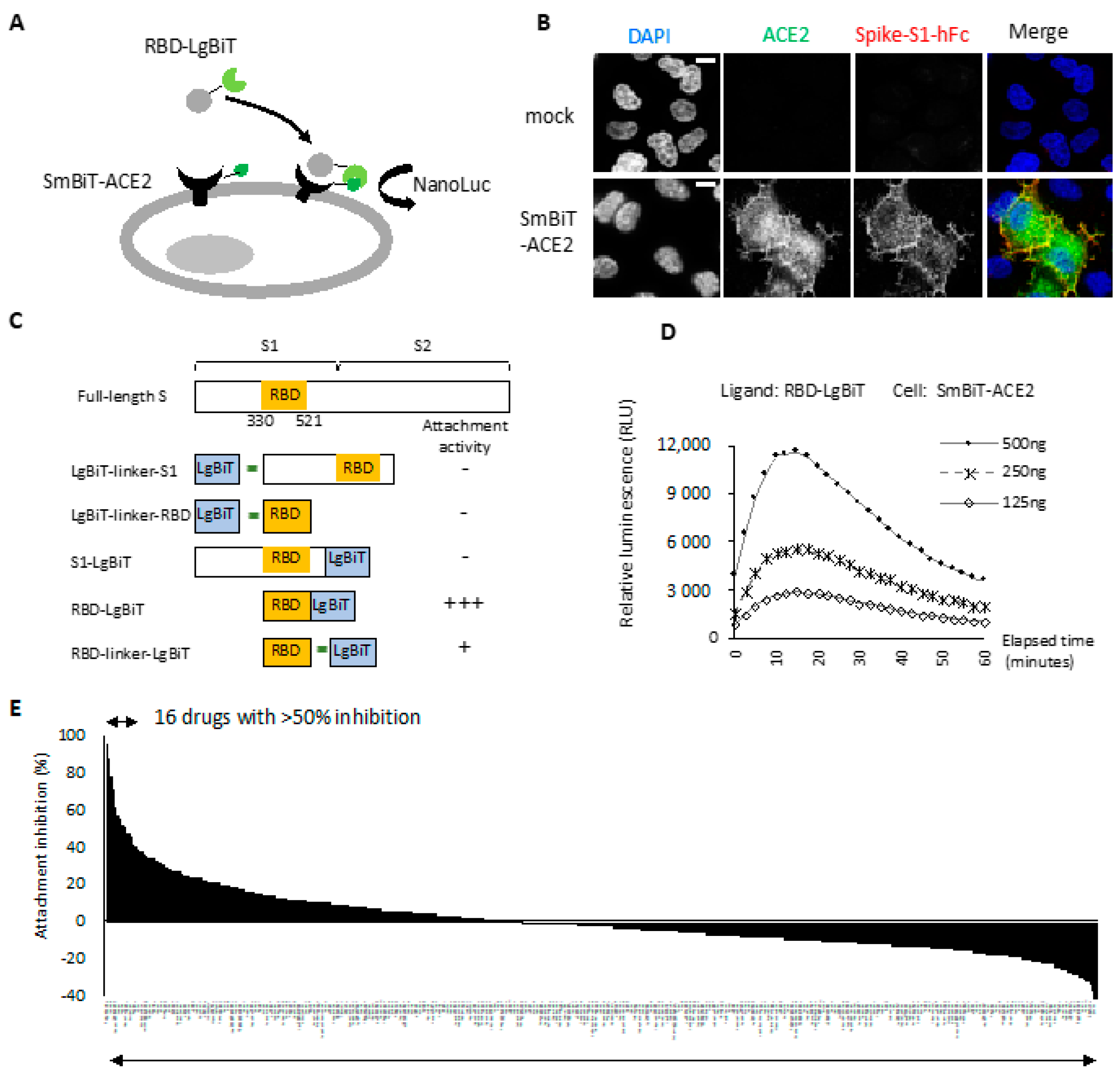
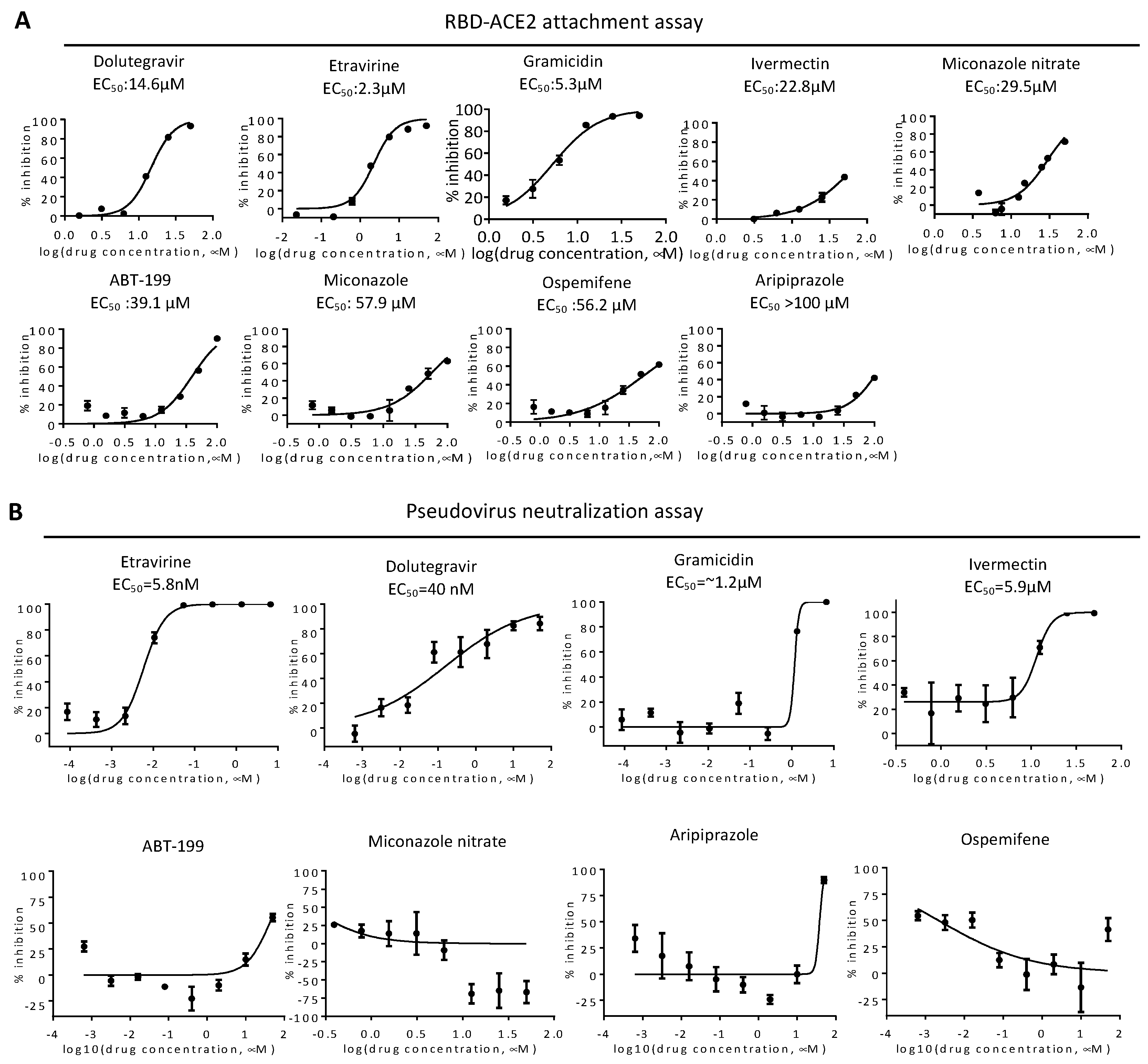
2.1. RBD-ACE2 Attachment Assay
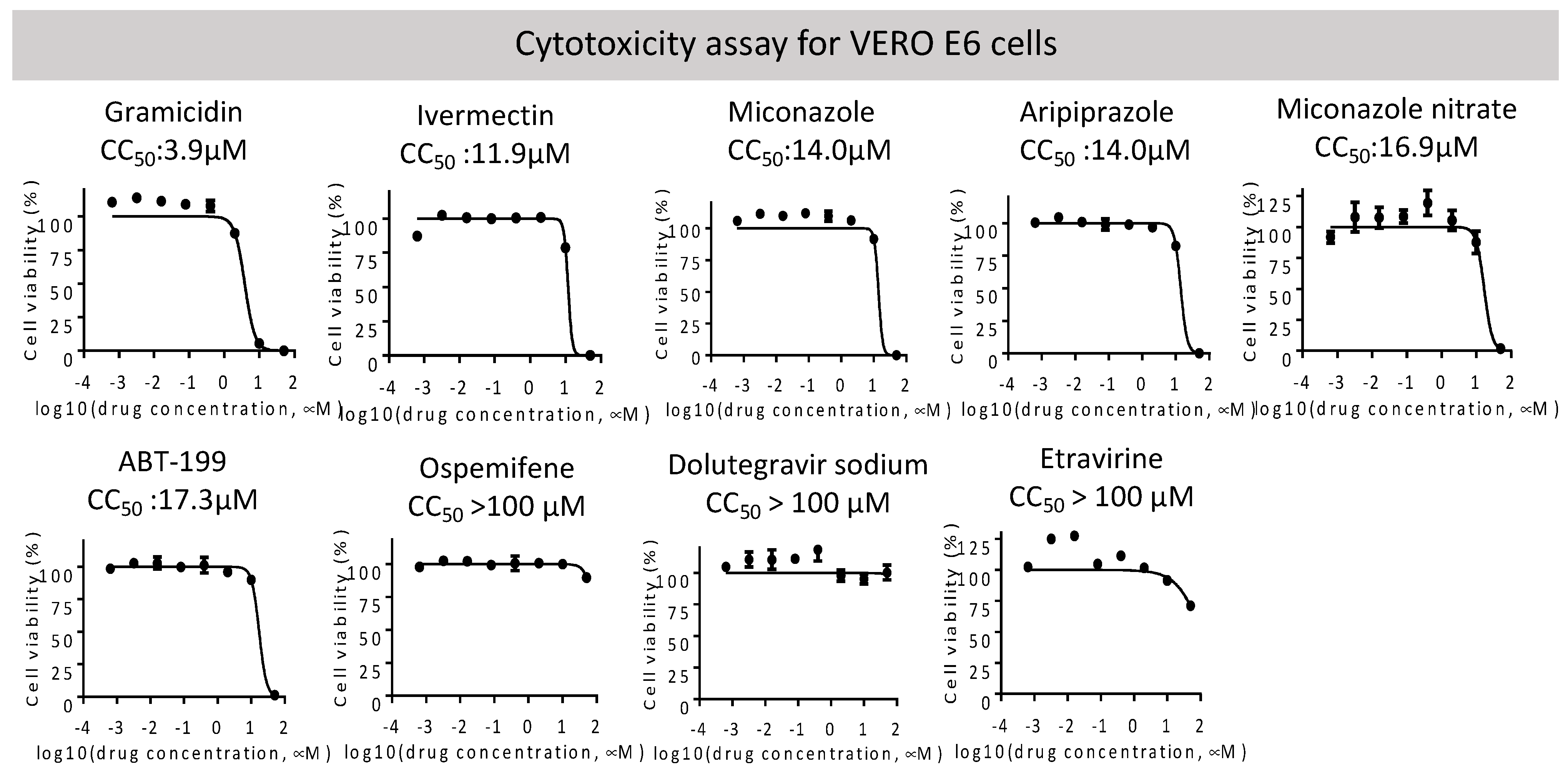
2.2. Entry Inhibitor Screening from an FDA-Approved Drug Library
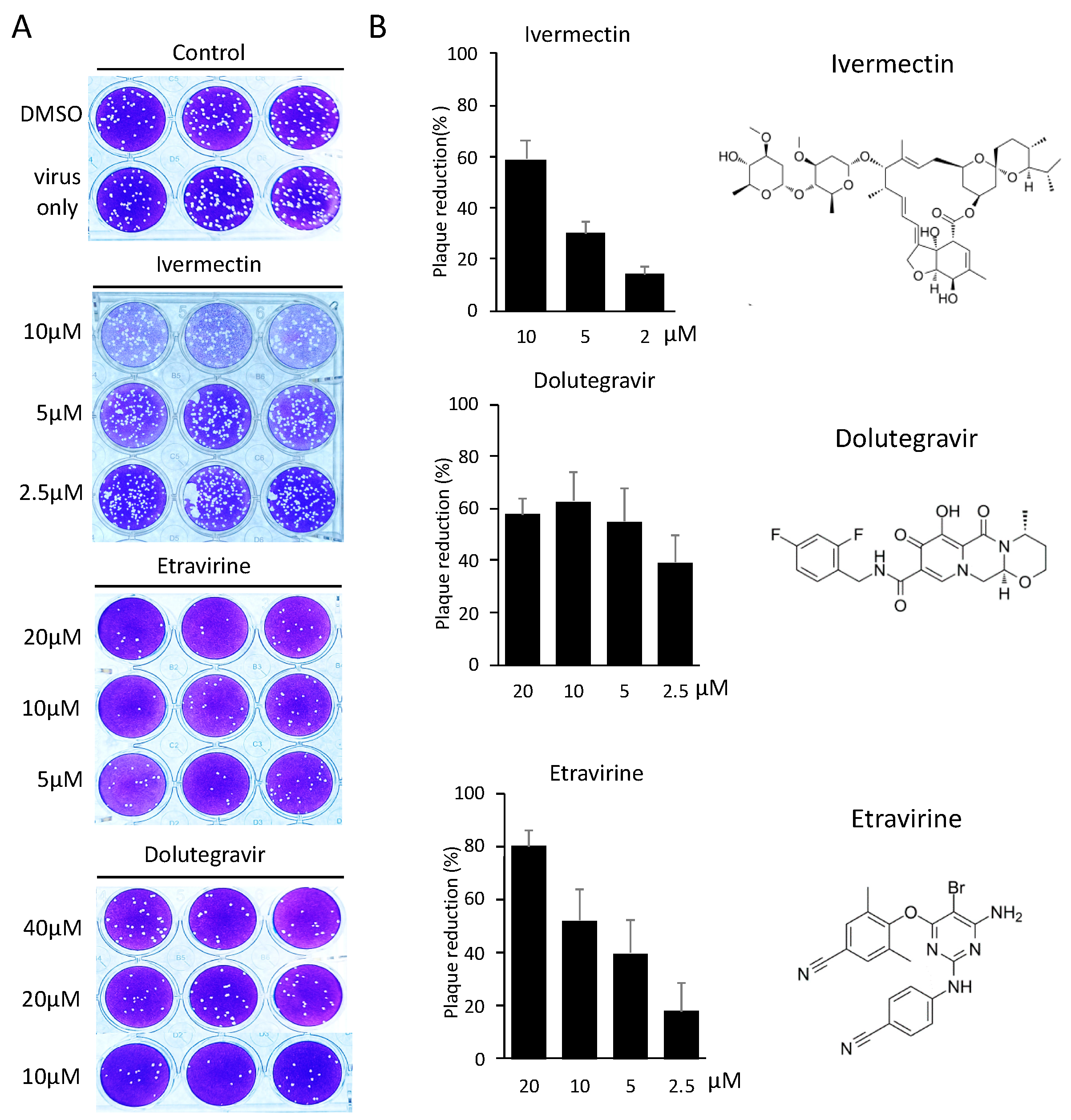
2.3. Neutralization of Spike Pseudovirus and SARS-CoV-2
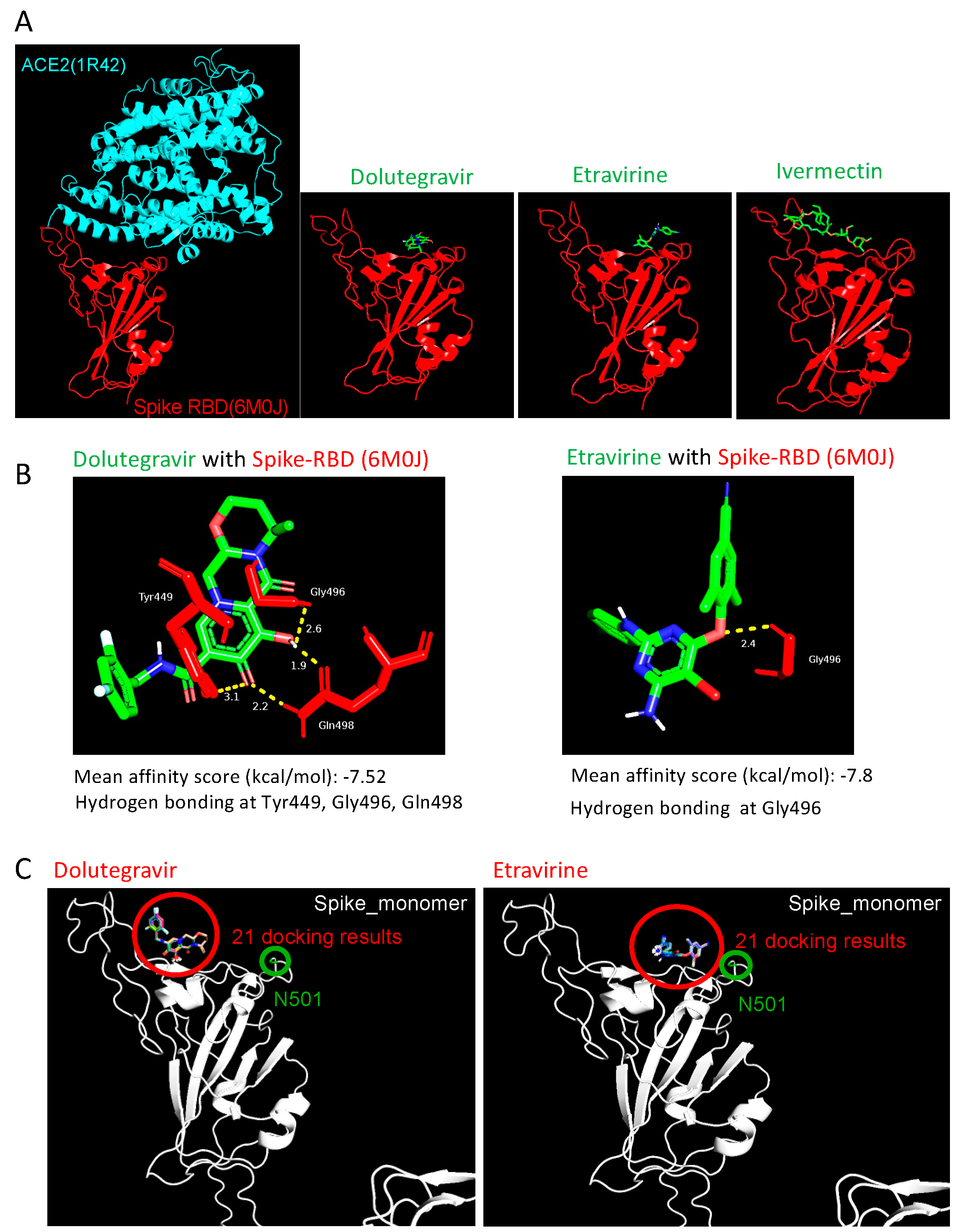
2.4. Etravirine and Dolutegravir Preferentially Interact with Spike RBD
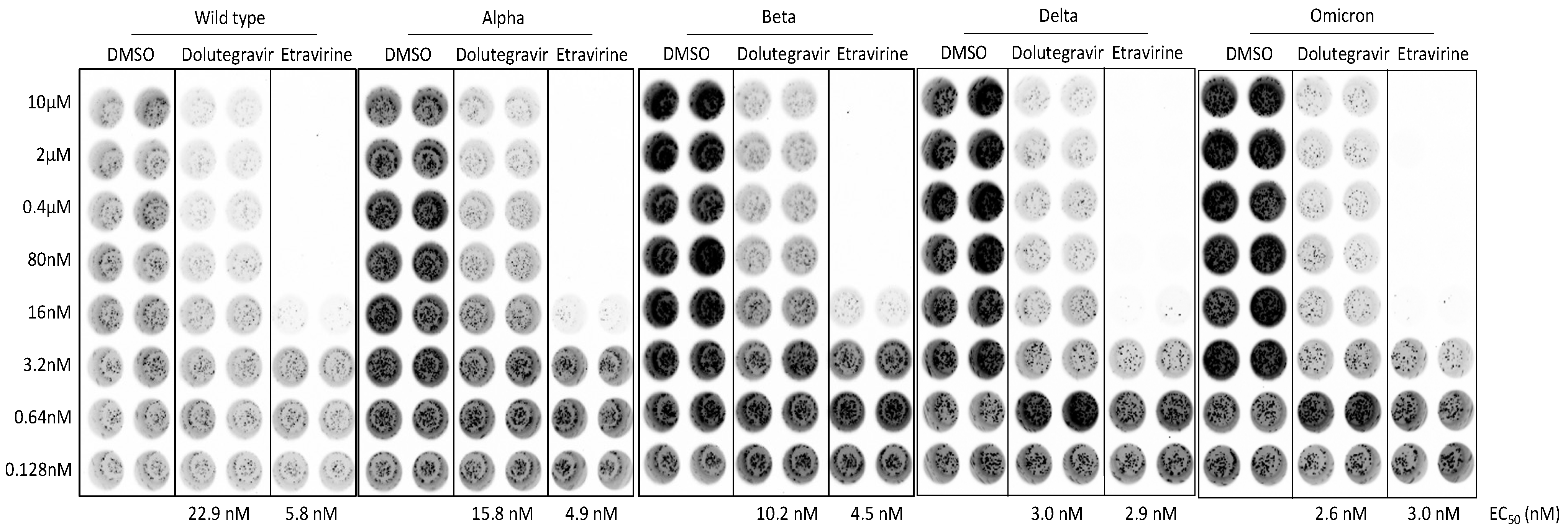
2.5. Dolutegravir and Etravirine as Pan-Entry Inhibitors of Predominant Viral Variants
3. Discussion
4. Materials and Methods
4.1. Molecular Cloning and Cell Culture
4.2. Indirect Immunofluorescence Staining
4.3. Recombinant RBD Fusion Protein
4.4. RBD-ACE2 Attachment Assay
4.5. Pseudovirus Neutralization Assay
4.6. SARS-CoV-2 Plaque Reduction Neutralization Test
4.7. Molecular Docking
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jackson, C.B.; Farzan, M.; Chen, B.; Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 2021, 23, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Wan, Y.; Luo, C.; Ye, G.; Geng, Q.; Auerbach, A.; Li, F. Cell entry mechanisms of SARS-CoV-2. Proc. Natl. Acad. Sci. USA 2020, 117, 11727–11734. [Google Scholar] [CrossRef]
- Shang, J.; Ye, G.; Shi, K.; Wan, Y.; Luo, C.; Aihara, H.; Geng, Q.; Auerbach, A.; Li, F. Structural basis of receptor recognition by SARS-CoV-2. Nature 2020, 581, 221–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lei, C.; Qian, K.; Li, T.; Zhang, S.; Fu, W.; Ding, M.; Hu, S. Neutralization of SARS-CoV-2 spike pseudotyped virus by recombinant ACE2-Ig. Nat. Commun. 2020, 11, 2070. [Google Scholar] [CrossRef] [Green Version]
- Rogers, T.F.; Zhao, F.; Huang, D.; Beutler, N.; Burns, A.; He, W.T.; Limbo, O.; Smith, C.; Song, G.; Woehl, J.; et al. Isolation of potent SARS-CoV-2 neutralizing antibodies and protection from disease in a small animal model. Science 2020, 369, 956–963. [Google Scholar] [CrossRef] [PubMed]
- Cannalire, R.; Stefanelli, I.; Cerchia, C.; Beccari, A.R.; Pelliccia, S.; Summa, V. SARS-CoV-2 entry inhibitors: Small molecules and peptides targeting virus or host cells. Int. J. Mol. Sci. 2020, 21, 5707. [Google Scholar] [CrossRef]
- Xiu, S.; Dick, A.; Ju, H.; Mirzaie, S.; Abdi, F.; Cocklin, S.; Zhan, P.; Liu, X. Inhibitors of SARS-CoV-2 entry: Current and future opportunities. J. Med. Chem. 2020, 63, 12256–12274. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.R.; Fleming, T.R.; Longini, I.M.; Peto, R.; Briand, S.; Heymann, D.L.; Beral, V.; Snape, M.D.; Rees, H.; Ropero, A.M.; et al. SARS-CoV-2 variants and vaccines. N. Engl. J. Med. 2021, 385, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Volz, E.; Mishra, S.; Chand, M.; Barrett, J.C.; Johnson, R.; Geidelberg, L.; Hinsley, W.R.; Laydon, D.J.; Dabrera, G.; O’Toole, A.; et al. Assessing transmissibility of SARS-CoV-2 lineage B.1.1.7 in England. Nature 2021, 593, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.G.; Abbott, S.; Barnard, R.C.; Jarvis, C.I.; Kucharski, A.J.; Munday, J.D.; Pearson, C.A.B.; Russell, T.W.; Tully, D.C.; Washburne, A.D.; et al. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science 2021, 372, eabg3055. [Google Scholar] [CrossRef] [PubMed]
- Planas, D.; Veyer, D.; Baidaliuk, A.; Staropoli, I.; Guivel-Benhassine, F.; Rajah, M.M.; Planchais, C.; Porrot, F.; Robillard, N.; Puech, J.; et al. Reduced sensitivity of SARS-CoV-2 variant Delta to antibody neutralization. Nature 2021, 596, 276–280. [Google Scholar] [CrossRef] [PubMed]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of COVID-19 vaccines against the B.1.617.2 (Delta) variant. N. Engl. J. Med. 2021, 385, 585–594. [Google Scholar] [CrossRef]
- Tiecco, G.; Storti, S.; Degli Antoni, M.; Foca, E.; Castelli, F.; Quiros-Roldan, E. Omicron genetic and clinical peculiarities that may overturn SARS-CoV-2 pandemic: A literature review. Int. J. Mol. Sci. 2022, 23, 1987. [Google Scholar] [CrossRef] [PubMed]
- Dixon, A.S.; Schwinn, M.K.; Hall, M.P.; Zimmerman, K.; Otto, P.; Lubben, T.H.; Butler, B.L.; Binkowski, B.F.; Machleidt, T.; Kirkland, T.A.; et al. NanoLuc complementation reporter optimized for accurate measurement of protein interactions in cells. ACS Chem. Biol. 2016, 11, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Low, K.; Tan, S.Y.S.; Izgorodina, E.I. An ab initio study of the structure and energetics of hydrogen bonding in ionic liquids. Front. Chem. 2019, 7, 208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahbar, M.; Morsali, A.; Bozorgmehr, M.R.; Beyramabadi, S.A. Quantum chemical studies of chitosan nanoparticles as effective drug delivery systems for 5-fluorouracil anticancer drug. J. Mol. Liq. 2020, 302, 112495. [Google Scholar] [CrossRef]
- Bhattacharjee, M.J.; Lin, J.J.; Chang, C.Y.; Chiou, Y.T.; Li, T.N.; Tai, C.W.; Shiu, T.F.; Chen, C.A.; Chou, C.Y.; Chakraborty, P.; et al. Identifying primate ACE2 variants that confer resistance to SARS-CoV-2. Mol. Biol. Evol. 2021, 38, 2715–2731. [Google Scholar] [CrossRef] [PubMed]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [Green Version]
- Schwinn, M.K.; Machleidt, T.; Zimmerman, K.; Eggers, C.T.; Dixon, A.S.; Hurst, R.; Hall, M.P.; Encell, L.P.; Binkowski, B.F.; Wood, K.V. CRISPR-mediated tagging of endogenous proteins with a luminescent peptide. ACS Chem. Biol. 2018, 13, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Caly, L.; Druce, J.D.; Catton, M.G.; Jans, D.A.; Wagstaff, K.M. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antivir. Res. 2020, 178, 104787. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, E.J.; Manguiat, K.; Wood, H.; Drebot, M. Two detailed plaque assay protocols for the quantification of infectious SARS-CoV-2. Curr. Protoc. Microbiol. 2020, 57, ecpmc105. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leung, K.; Shum, M.H.; Leung, G.M.; Lam, T.T.; Wu, J.T. Early transmissibility assessment of the N501Y mutant strains of SARS-CoV-2 in the United Kingdom, October to November 2020. Euro Surveill. 2021, 26, 2002106. [Google Scholar] [CrossRef] [PubMed]
- Lazzarin, A.; Campbell, T.; Clotet, B.; Johnson, M.; Katlama, C.; Moll, A.; Towner, W.; Trottier, B.; Peeters, M.; Vingerhoets, J.; et al. Efficacy and safety of TMC125 (etravirine) in treatment-experienced HIV-1-infected patients in DUET-2: 24-week results from a randomised, double-blind, placebo-controlled trial. Lancet 2007, 370, 39–48. [Google Scholar] [CrossRef]
- Lansdon, E.B.; Brendza, K.M.; Hung, M.; Wang, R.; Mukund, S.; Jin, D.; Birkus, G.; Kutty, N.; Liu, X. Crystal structures of HIV-1 reverse transcriptase with etravirine (TMC125) and rilpivirine (TMC278): Implications for drug design. J. Med. Chem. 2010, 53, 4295–4299. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.J.; Li, W.; Berta, D.; Badaoui, M.; Ballandras-Colas, A.; Nans, A.; Kotecha, A.; Rosta, E.; Engelman, A.N.; Cherepanov, P. Structural basis of second-generation HIV integrase inhibitor action and viral resistance. Science 2020, 367, 806–810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walmsley, S.L.; Antela, A.; Clumeck, N.; Duiculescu, D.; Eberhard, A.; Gutierrez, F.; Hocqueloux, L.; Maggiolo, F.; Sandkovsky, U.; Granier, C.; et al. Dolutegravir plus abacavir-lamivudine for the treatment of HIV-1 infection. N. Engl. J. Med. 2013, 369, 1807–1818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Indu, P.; Rameshkumar, M.R.; Arunagirinathan, N.; Al-Dhabi, N.A.; Valan Arasu, M.; Ignacimuthu, S. Raltegravir, Indinavir, Tipranavir, Dolutegravir, and Etravirine against main protease and RNA-dependent RNA polymerase of SARS-CoV-2: A molecular docking and drug repurposing approach. J. Infect. Public Health 2020, 13, 1856–1861. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.J.; Jha, R.K.; Amera, G.M.; Jain, M.; Singh, E.; Pathak, A.; Singh, R.P.; Muthukumaran, J.; Singh, A.K. Targeting SARS-CoV-2: A systematic drug repurposing approach to identify promising inhibitors against 3C-like proteinase and 2′-O-ribose methyltransferase. J. Biomol. Struct. Dyn. 2020, 39, 2679–2692. [Google Scholar] [CrossRef] [Green Version]
- Bhaskaran, K.; Rentsch, C.T.; MacKenna, B.; Schultze, A.; Mehrkar, A.; Bates, C.J.; Eggo, R.M.; Morton, C.E.; Bacon, S.C.J.; Inglesby, P.; et al. HIV infection and COVID-19 death: A population-based cohort analysis of UK primary care data and linked national death registrations within the OpenSAFELY platform. Lancet HIV 2021, 8, e24–e32. [Google Scholar] [CrossRef]
- O’Boyle, N.M.; Banck, M.; James, C.A.; Morley, C.; Vandermeersch, T.; Hutchison, G.R. Open babel: An open chemical toolbox. J. Cheminform. 2011, 3, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanner, M.F. Python: A programming language for software integration and development. J. Mol. Graph. Model 1999, 17, 57–61. [Google Scholar] [PubMed]
- Towler, P.; Staker, B.; Prasad, S.G.; Menon, S.; Tang, J.; Parsons, T.; Ryan, D.; Fisher, M.; Williams, D.; Dales, N.A.; et al. ACE2 X-ray structures reveal a large hinge-bending motion important for inhibitor binding and catalysis. J. Biol. Chem. 2004, 279, 17996–18007. [Google Scholar] [CrossRef] [Green Version]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| No. | Hits | Positive Screening RBD Attachment Assay | Negative Screening HiBiT Assay † | ||||
|---|---|---|---|---|---|---|---|
| % Inhibition (100 μM) | % Inhibition (20 μM) | * Dose Response | % Inhibition (100 μM) | % Inhibition (20 μM) | INHIBIT NanoBiT ‡ | ||
| 1 | Thonzonium Bromide | 99 | 93 | Yes | 52 | 36 | Yes |
| 2 | Dabrafenib (GSK2118436) | 96 | 85 | Yes | 97 | 88 | Yes |
| 3 | Dolutegravir sodium (GSK1349572) | 96 | 82 | Yes | 62 | 1 | No |
| 4 | Etravirine (TMC125) | 90 | 78 | Yes | 31 | 0 | No |
| 5 | Crystal Violet | 99 | 73 | Yes | 86 | 17 | Yes |
| 6 | Gramicidin | 95 | 70 | Yes | 35 | 0 | No |
| 7 | Clevidipine butyrate | 93 | 69 | Yes | 93 | 79 | Yes |
| 8 | Nitazoxanide | 93 | 69 | Yes | 95 | 77 | Yes |
| 9 | Nimodipine | 96 | 64 | Yes | 97 | 79 | Yes |
| 10 | Felodipine | 96 | 63 | Yes | 99 | 88 | Yes |
| 11 | Nicardipine hydrochloride | 86 | 58 | Yes | 88 | 64 | Yes |
| 12 | Aripiprazole | 71 | 56 | Yes | 21 | 13 | No |
| 13 | Phenazopyridine hydrochloride | 91 | 52 | Yes | 83 | 48 | Yes |
| 14 | Miconazole nitrate | 83 | 52 | Yes | 12 | 3 | No |
| 15 | ABT-199 | 96 | 51 | Yes | 43 | 0 | No |
| 16 | Miconazole | 79 | 50 | Yes | 15 | 6 | No |
| 17 | Dronedarone hydrochloride | 93 | 41 | Yes | 86 | 21 | Yes |
| 18 | Atovaquone | 95 | 39 | Yes | 96 | 54 | Yes |
| 19 | Ospemifene | 68 | 39 | Yes | 8 | 3 | No |
| 20 | Ivermectin | 68 | 29 | Yes | 4 | 0 | No |
| Name (CAS ID) | Major Target | Application | EC50 (μM) | |||
|---|---|---|---|---|---|---|
| VeroE6 CC50 (μM) | RBD Attachment | Pseudovirus Wild Type | SARS-CoV-2 PRNT | |||
| Dolutegravir sodium 1051375-19-9 | HIV integrase | HIV integrase inhibitor | >100 | 14.6 | 0.04 | 4.2 |
| Etravirine 269055-15-4 | HIV reverse transcriptase | Non-nucleoside reverse transcriptase inhibitor (NNRTI) for HIV | >100 | 2.3 | 0.006 | 7.7 |
| Miconazole nitrate 22832-87-7 | Unknown | Anti-fungal agent | 16.9 | 29.5 | n.a. | n.a |
| Gramicidin 1405-97-6 | MRP1 | Anti-bacterial | 3.9 | 5.3 | 1.2 | n.a. |
| Ivermectin 70288-86-7 | GluCl channel; P2X purinergic receptor | Broad-spectrum antiparasitic drug | 11.9 | 22.8 | 5.9 | n.a. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, R.K.-L.; Li, T.-N.; Chang, S.-Y.; Chao, T.-L.; Kuo, C.-H.; Pan, M.Y.-C.; Chiou, Y.-T.; Liao, K.-J.; Yang, Y.; Wu, Y.-H.; et al. Identification of Entry Inhibitors against Delta and Omicron Variants of SARS-CoV-2. Int. J. Mol. Sci. 2022, 23, 4050. https://doi.org/10.3390/ijms23074050
Lee RK-L, Li T-N, Chang S-Y, Chao T-L, Kuo C-H, Pan MY-C, Chiou Y-T, Liao K-J, Yang Y, Wu Y-H, et al. Identification of Entry Inhibitors against Delta and Omicron Variants of SARS-CoV-2. International Journal of Molecular Sciences. 2022; 23(7):4050. https://doi.org/10.3390/ijms23074050
Chicago/Turabian StyleLee, Richard Kuan-Lin, Tian-Neng Li, Sui-Yuan Chang, Tai-Ling Chao, Chun-Hsien Kuo, Max Yu-Chen Pan, Yu-Ting Chiou, Kuan-Ju Liao, Yi Yang, Yi-Hsuan Wu, and et al. 2022. "Identification of Entry Inhibitors against Delta and Omicron Variants of SARS-CoV-2" International Journal of Molecular Sciences 23, no. 7: 4050. https://doi.org/10.3390/ijms23074050
APA StyleLee, R. K.-L., Li, T.-N., Chang, S.-Y., Chao, T.-L., Kuo, C.-H., Pan, M. Y.-C., Chiou, Y.-T., Liao, K.-J., Yang, Y., Wu, Y.-H., Huang, C.-H., Juan, H.-F., Hsieh, H.-P., & Wang, L. H.-C. (2022). Identification of Entry Inhibitors against Delta and Omicron Variants of SARS-CoV-2. International Journal of Molecular Sciences, 23(7), 4050. https://doi.org/10.3390/ijms23074050






