Thromboinflammation in Myeloproliferative Neoplasms (MPN)—A Puzzle Still to Be Solved
Abstract
1. Introduction
2. Methodology of Literature Search
3. Inflammatory Cytokines
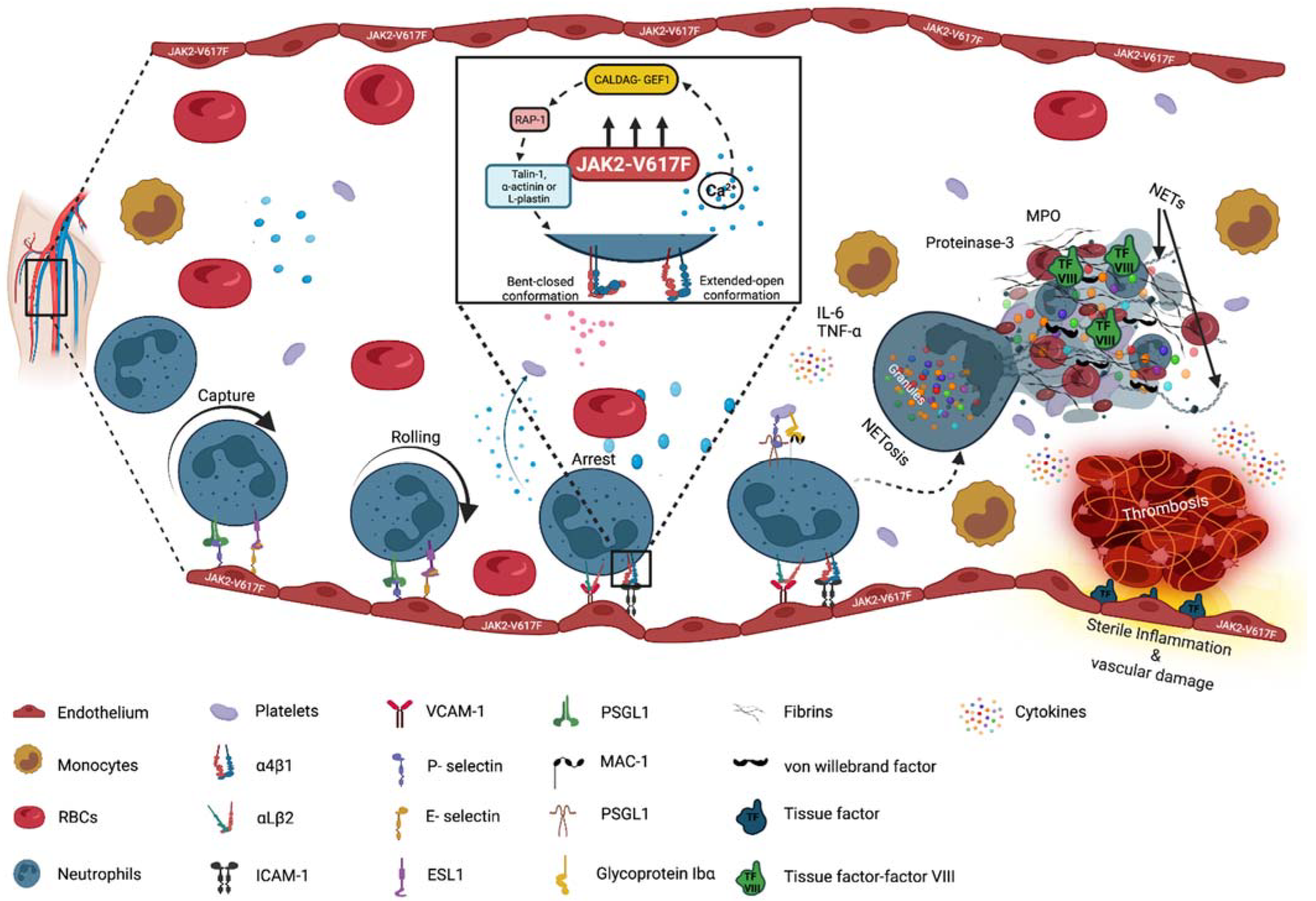
4. Thrombosis in MPN: The Roles of Integrin Activation and of NETosis
5. Signaling: JAKs and Leukocyte Integrins
6. Extracellular Vesicles (EVs) and Circulating Endothelial Cells (CEC) in Thrombogenesis
7. Current Therapeutic Strategies to Reduce the Prothrombotic Risk
8. Conclusions and Future Direction
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tefferi, A.; Thiele, J.; Orazi, A.; Kvasnicka, H.M.; Barbui, T.; Hanson, C.A.; Barosi, G.; Verstovsek, S.; Birgegard, G.; Mesa, R.; et al. Proposals and Rationale for Revision of the World Health Organization Diagnostic Criteria for Polycythemia Vera, Essential Thrombocythemia, and Primary Myelofibrosis: Recommendations from an Ad Hoc International Expert Panel. Blood 2007, 110, 1092–1097. [Google Scholar] [CrossRef]
- Vardiman, J.W.; Thiele, J.; Arber, D.A.; Brunning, R.D.; Borowitz, M.J.; Porwit, A.; Harris, N.L.; Le Beau, M.M.; Hellström-Lindberg, E.; Tefferi, A.; et al. The 2008 Revision of the World Health Organization (WHO) Classification of Myeloid Neoplasms and Acute Leukemia: Rationale and Important Changes. Blood 2009, 114, 937–951. [Google Scholar] [CrossRef]
- Spivak, J.L.; Silver, R.T. The Revised World Health Organization Diagnostic Criteria for Polycythemia Vera, Essential Thrombocytosis, and Primary Myelofibrosis: An Alternative Proposal. Blood 2008, 112, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Kralovics, R.; Passamonti, F.; Buser, A.S.; Teo, S.-S.; Tiedt, R.; Passweg, J.R.; Tichelli, A.; Cazzola, M.; Skoda, R.C. A Gain-of-Function Mutation of JAK2 in Myeloproliferative Disorders. N. Engl. J. Med. 2005, 352, 1779–1790. [Google Scholar] [CrossRef]
- Ugo, V.; James, C.; Vainchenker, W. A Unique Clonal JAK2 Mutation Leading to Constitutive Signalling Causes Polycythaemia Vera. Med. Sci. 2005, 434, 669–670. [Google Scholar] [CrossRef]
- Beer, P.A.; Campbell, P.J.; Scott, L.M.; Bench, A.J.; Erber, W.N.; Bareford, D.; Wilkins, B.S.; Reilly, J.T.; Hasselbalch, H.C.; Bowman, R.; et al. MPL Mutations in Myeloproliferative Disorders: Analysis of the PT-1 Cohort. Blood 2008, 112, 141–149. [Google Scholar] [CrossRef]
- Pardanani, A.D.; Levine, R.L.; Lasho, T.; Pikman, Y.; Mesa, R.A.; Wadleigh, M.; Steensma, D.P.; Elliott, M.A.; Wolanskyj, A.P.; Hogan, W.J.; et al. MPL515 Mutations in Myeloproliferative and Other Myeloid Disorders: A Study of 1182 Patients. Blood 2006, 108, 3472–3476. [Google Scholar] [CrossRef]
- Klampfl, T.; Gisslinger, H.; Harutyunyan, A.S.; Nivarthi, H.; Rumi, E.; Milosevic, J.D.; Them, N.C.; Berg, T.; Gisslinger, B.; Pietra, D.; et al. Somatic Mutations of Calreticulin in Myeloproliferative Neoplasms. N. Engl. J. Med. 2013, 369, 2379–2390. [Google Scholar] [CrossRef] [PubMed]
- Nangalia, J.; Massie, C.E.; Baxter, E.J.; Nice, F.L.; Gundem, G.; Wedge, D.C.; Avezov, E.; Li, J.; Kollmann, K.; Kent, D.G.; et al. Somatic CALR Mutations in Myeloproliferative Neoplasms with Nonmutated JAK2. N. Engl. J. Med. 2013, 369, 2391–2405. [Google Scholar] [CrossRef] [PubMed]
- McMullin, M.F.; Anderson, L.A. Aetiology of Myeloproliferative Neoplasms. Cancers 2020, 12, 1810. [Google Scholar] [CrossRef]
- Solans, M.; Sanvisens, A.; Ameijide, A.; Merino, S.; Rojas, D.; Alemán, A.; Banqueri, E.; Chico, M.; Marcos, A.I.; De Castro, V. Incidence of Myeloid Neoplasms in Spain (2002–2013): A Population-Based Study of the Spanish Network of Cancer Registries. Sci. Rep. 2022, 12, 323. [Google Scholar] [CrossRef]
- Ruggeri, M.; Tosetto, A.; Frezzato, M.; Rodeghiero, F. The Rate of Progression to Polycythemia Vera or Essential Thrombocythemia in Patients with Erythrocytosis or Thrombocytosis. Ann. Intern. Med. 2003, 139, 470–475. [Google Scholar] [CrossRef]
- Geyer, H.L.; Dueck, A.C.; Scherber, R.M.; Mesa, R.A. Impact of Inflammation on Myeloproliferative Neoplasm Symptom Development. Mediat. Inflamm. 2015, 2015, 284706. [Google Scholar] [CrossRef]
- Italiano, G.; Policitemia, S. Polycythemia Vera: The Natural History of 1213 Patients Followed for 20 Years. Gruppo Italiano Studio Policitemia. Ann. Intern. Med. 1995, 123, 656–664. [Google Scholar]
- Tefferi, A.; Rumi, E.; Finazzi, G.; Gisslinger, H.; Vannucchi, A.M.; Rodeghiero, F.; Randi, M.L.; Vaidya, R.; Cazzola, M.; Rambaldi, A.; et al. Survival and Prognosis among 1545 Patients with Contemporary Polycythemia Vera: An International Study. Leukemia 2013, 27, 1874–1881. [Google Scholar] [CrossRef]
- Kiladjian, J.J.; Chevret, S.; Dosquet, C.; Chomienne, C.; Rain, J.D. Treatment of Polycythemia Vera with Hydroxyurea and Pipobroman: Final Results of a Randomized Trial Initiated in 1980. J. Clin. Oncol. 2011, 29, 3907–3913. [Google Scholar] [CrossRef]
- Hultcrantz, M.; Kristinsson, S.Y.; Andersson, T.M.-L.; Landgren, O.; Eloranta, S.; Derolf, Å.R.; Dickman, P.W.; Hultcrantz, M.B.M.; Björkholm, M.; Institutet, K. Patterns of Survival Among Patients With Myeloproliferative Neoplasms Diagnosed in Sweden From 1973 to 2008: A Population-Based Study. J. Clin. Oncol. 2012, 30, 2995–3001. [Google Scholar] [CrossRef]
- Kroll, M.H.; Michaelis, L.C.; Verstovsek, S. Mechanisms of Thrombogenesis in Polycythemia Vera. Blood Rev. 2015, 29, 215–221. [Google Scholar] [CrossRef]
- Pei, Y.Q.; Wu, Y.; Wang, F.; Cui, W. Prognostic Value of CALR vs. JAK2V617F Mutations on Splenomegaly, Leukemic Transformation, Thrombosis, and Overall Survival in Patients with Primary Fibrosis: A Meta-Analysis. Ann. Hematol. 2016, 95, 1391–1398. [Google Scholar] [CrossRef]
- Masselli, E.; Pozzi, G.; Gobbi, G.; Merighi, S.; Gessi, S.; Vitale, M.; Carubbi, C. Cytokine Profiling in Myeloproliferative Neoplasms: Overview on Phenotype Correlation, Outcome Prediction, and Role of Genetic Variants. Cells 2020, 9, 2136. [Google Scholar] [CrossRef]
- Tefferi, A.; Vaidya, R.; Caramazza, D.; Finke, C.; Lasho, T.; Pardanani, A. Circulating Interleukin (IL)-8, IL-2R, IL-12, and IL-15 Levels Are Independently Prognostic in Primary Myelofibrosis: A Comprehensive Cytokine Profiling Study. J. Clin. Oncol. 2011, 29, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Edelmann, B.; Gupta, N.; Schnoeder, T.M.; Oelschlegel, A.M.; Shahzad, K.; Goldschmidt, J.; Philipsen, L.; Weinert, S.; Ghosh, A.; Saalfeld, F.C.; et al. JAK2-V617F Promotes Venous Thrombosis through Β1/Β2 Integrin Activation. J. Clin. Investig. 2018, 128, 4359–4371. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Rondina, M.T. The Era of Thromboinflammation: Platelets Are Dynamic Sensors and Effector Cells During Infectious Diseases. Front. Immunol. 2019, 10, 2204. [Google Scholar] [CrossRef] [PubMed]
- Hoermann, G.; Greiner, G.; Valent, P. Cytokine Regulation of Microenvironmental Cells in Myeloproliferative Neoplasms. Mediat. Inflamm. 2015, 2015, 869242. [Google Scholar] [CrossRef] [PubMed]
- Longhitano, L.; Volti, G.L.; Giallongo, C.; Spampinato, M.; Barbagallo, I.; Di Rosa, M.; Romano, A.; Avola, R.; Tibullo, D.; Palumbo, G.A. The Role of Inflammation and Inflammasome in Myeloproliferative Disease. J. Clin. Med. 2020, 9, 2334. [Google Scholar] [CrossRef]
- Cacemiro, M.d.C.; Cominal, J.G.; Tognon, R.; Nunes, N.D.S.; Simões, B.P.; Pontes, L.L.D.F.; Catto, L.F.B.; Traina, F.; Souto, E.X.; Zambuzi, F.A.; et al. Philadelphia-Negative Myeloproliferative Neoplasms as Disorders Marked by Cytokine Modulation. Hematol. Transfus. Cell Ther. 2018, 40, 120–131. [Google Scholar] [CrossRef]
- Allain-Maillet, S.; Bosseboeuf, A.; Mennesson, N.; Bostoën, M.; Dufeu, L.; Choi, E.H.; Cleyrat, C.; Mansier, O.; Lippert, E.; Le Bris, Y.; et al. Anti-Glucosylsphingosine Autoimmunity, JAK2V617F-Dependent Interleukin-1β and JAK2V617F-Independent Cytokines in Myeloproliferative Neoplasms. Cancers 2020, 12, 2446. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, R.; Gangat, N.; Jimma, T.; Finke, C.M.; Lasho, T.L.; Pardanani, A.; Tefferi, A. Plasma Cytokines in Polycythemia Vera: Phenotypic Correlates, Prognostic Relevance, and Comparison with Myelofibrosis. Am. J. Hematol. 2012, 87, 1003–1005. [Google Scholar] [CrossRef] [PubMed]
- Hermouet, S.; Bigot-Corbel, E.; Gardie, B. Pathogenesis of Myeloproliferative Neoplasms: Role and Mechanisms of Chronic Inflammation. Mediat. Inflamm. 2015, 2015, 145293. [Google Scholar] [CrossRef] [PubMed]
- Forte, D.; Barone, M.; Morsiani, C.; Simonetti, G.; Fabbri, F.; Bruno, S.; Bandini, E.; Sollazzo, D.; Collura, S.; Deregibus, M.C.; et al. Distinct Profile of CD34+ Cells and Plasma-Derived Extracellular Vesicles from Triple-Negative Patients with Myelofibrosis Reveals Potential Markers of Aggressive Disease. J. Exp. Clin. Cancer Res. 2021, 40, 49. [Google Scholar] [CrossRef]
- Verstovsek, S.; Kantarjian, H.; Mesa, R.A.; Pardanani, A.D.; Cortes-Franco, J.; Thomas, D.A.; Estrov, Z.; Fridman, J.S.; Bradley, E.C.; Erickson-Viitanen, S.; et al. Safety and Efficacy of INCB018424, a JAK1 and JAK2 Inhibitor, in Myelofibrosis. N. Engl. J. Med. 2010, 363, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Skov, V.; Larsen, T.S.; Thomassen, M.; Riley, C.H.; Jensen, M.K.; Bjerrum, O.W.; Kruse, T.A.; Hasselbalch, H.C. Molecular Profiling of Peripheral Blood Cells from Patients with Polycythemia Vera and Related Neoplasms: Identification of Deregulated Genes of Significance for Inflammation and Immune Surveillance. Leuk. Res. 2012, 36, 1387–1392. [Google Scholar] [CrossRef] [PubMed]
- Pourcelot, E.; Trocme, C.; Mondet, J.; Bailly, S.; Toussaint, B.; Mossuz, P. Cytokine Profiles in Polycythemia Vera and Essential Thrombocythemia Patients: Clinical Implications. Exp. Hematol. 2014, 42, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Čokić, V.P.; Mitrović-Ajtić, O.; Beleslin-Čokić, B.B.; Marković, D.; Buač, M.; Diklić, M.; Kraguljac-Kurtović, N.; Damjanović, S.; Milenković, P.; Gotić, M.; et al. Proinflammatory Cytokine IL-6 and JAK-STAT Signaling Pathway in Myeloproliferative Neoplasms. Mediat. Inflamm. 2015, 2015, 453020. [Google Scholar] [CrossRef] [PubMed]
- Hermouet, S.; Godard, A.; Pineau, D.; Corre, I.; Raher, S.; Lippert, E.; Jacques, Y. Abnormal Production of Interleukin (IL)-11 and IL-8 in Polycythaemia Vera. Cytokine 2002, 20, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Øbro, N.F.; Grinfeld, J.; Belmonte, M.; Irvine, M.; Shepherd, M.S.; Rao, T.N.; Karow, A.; Riedel, L.M.; Harris, O.B.; Baxter, E.J.; et al. Longitudinal Cytokine Profiling Identifies GRO-α and EGF as Potential Biomarkers of Disease Progression in Essential Thrombocythemia. HemaSphere 2020, 4, e371. [Google Scholar] [CrossRef] [PubMed]
- Fleischman, A.G.; Aichberger, K.J.; Luty, S.B.; Bumm, T.G.; Petersen, C.L.; Doratotaj, S.; Vasudevan, K.B.; LaTocha, D.H.; Yang, F.; Press, R.D.; et al. TNFα Facilitates Clonal Expansion of JAK2V617F Positive Cells in Myeloproliferative Neoplasms. Blood 2011, 118, 6392–6398. [Google Scholar] [CrossRef] [PubMed]
- Campanelli, R.; Rosti, V.; Villani, L.; Castagno, M.; Moretti, E.; Bonetti, E.; Bergamaschi, G.; Balduini, A.; Barosi, G.; Massa, M. Evaluation of the Bioactive and Total Transforming Growth Factor Β1 Levels in Primary Myelofibrosis. Cytokine 2011, 53, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Panteli, K.; Bai, M.; Hatzimichael, E.; Zagorianakou, N.; Agnantis, N.J.; Bourantas, K. Serum Levels, and Bone Marrow Immunohistochemical Expression of, Vascular Endothelial Growth Factor in Patients with Chronic Myeloproliferative Diseases. Hematology 2007, 12, 481–486. [Google Scholar] [CrossRef]
- Ho, C.L.; Lasho, T.L.; Butterfield, J.H.; Tefferi, A. Global Cytokine Analysis in Myeloproliferative Disorders. Leuk. Res. 2007, 31, 1389–1392. [Google Scholar] [CrossRef] [PubMed]
- Najem, M.Y.; Couturaud, F.; Lemarié, C.A. Cytokine and Chemokine Regulation of Venous Thromboembolism. J. Thromb. Haemost. 2020, 18, 1009–1019. [Google Scholar] [CrossRef] [PubMed]
- Liberale, L.; Holy, E.W.; Akhmedov, A.; Bonetti, N.R.; Nietlispach, F.; Matter, C.M.; Mach, F.; Montecucco, F.; Beer, J.H.; Paneni, F.; et al. Interleukin-1β Mediates Arterial Thrombus Formation via NET-Associated Tissue Factor. J. Clin. Med. 2019, 8, 2072. [Google Scholar] [CrossRef] [PubMed]
- Bester, J.; Pretorius, E. Effects of IL-1β, IL-6 and IL-8 on Erythrocytes, Platelets and Clot Viscoelasticity. Sci. Rep. 2016, 6, 32188. [Google Scholar] [CrossRef] [PubMed]
- Newberry, K.J.; Naqvi, K.; Nguyen, K.T.; Cardenas-Turanzas, M.; Florencia Tanaka, M.; Pierce, S.; Verstovsek, S. Comorbidities Predict Worse Prognosis in Patients with Primary Myelofibrosis. Cancer 2014, 120, 2996–3002. [Google Scholar] [CrossRef] [PubMed]
- Schafer, A.I. Thrombotic, Vascular, and Bleeding Complications of the Myeloproliferative Neoplasms. Hematol. Oncol. Clin. N. Am. 2021, 35, 305–324. [Google Scholar] [CrossRef] [PubMed]
- Cerquozzi, S.; Tefferi, A. Blast Transformation and Fibrotic Progression in Polycythemia Vera and Essential Thrombocythemia: A Literature Review of Incidence and Risk Factors. Blood Cancer J. 2015, 5, 366. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-Related Inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, H.; Farkas, D.K.; Christiansen, C.F.; Hasselbalch, H.C.; Sørensen, H.T. Chronic Myeloproliferative Neoplasms and Subsequent Cancer Risk: A Danish Population-Based Cohort Study. Blood 2011, 118, 6515–6520. [Google Scholar] [CrossRef] [PubMed]
- Pettersson, H.; Knutsen, H.; Holmberg, E.; Andréasson, B. Increased Incidence of Another Cancer in Myeloproliferative Neoplasms Patients at the Time of Diagnosis. Eur. J. Haematol. 2015, 94, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Lussana, F.; Rambaldi, A. Inflammation and Myeloproliferative Neoplasms. J. Autoimmun. 2017, 85, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Koschmieder, S.; Mughal, T.I.; Hasselbalch, H.C.; Barosi, G.; Valent, P.; Kiladjian, J.-J.; Jeryczynski, G.; Gisslinger, H.; Jutzi, J.S.; Pahl, H.L.; et al. Myeloproliferative Neoplasms and Inflammation: Whether to Target the Malignant Clone or the Inflammatory Process or Both Exclusion of Nonmalignant Causes of Inflammation in Patients with Suspected Mpns. Leukemia 2016, 30, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Castejon, G.; Brough, D. Understanding the Mechanism of IL-1β Secretion. Cytokine Growth Factor Rev. 2011, 22, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Rai, S.; Hansen, N.; Hao-Shen, H.; Dirnhofer, S.; Tata, N.R.; Skoda, R.C. IL-1β Secreted from Mutant Cells Carrying JAK2-V617Ffavors Early Clonal Expansion and Promotes MPN Disease Initiation and Progression. Blood 2019, 134, 307. [Google Scholar] [CrossRef]
- Orjalo, A.V.; Bhaumik, D.; Gengler, B.K.; Scott, G.K.; Campisi, J. Cell Surface-Bound IL-1α Is an Upstream Regulator of the Senescence-Associated IL-6/IL-8 Cytokine Network. Proc. Natl. Acad. Sci. USA 2009, 106, 17031–17036. [Google Scholar] [CrossRef] [PubMed]
- Arranz, L.; del Mar Arriero, M.; Villatoro, A. Interleukin-1β as Emerging Therapeutic Target in Hematological Malignancies and Potentially in Their Complications. Blood Rev. 2017, 31, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zuo, X. Cytokines Frequently Implicated in Myeloproliferative Neoplasms. Cytokine X 2019, 1, 100005. [Google Scholar] [CrossRef] [PubMed]
- Barabanshikova, M.V.; Dubina, I.A.; Lapin, S.V.; Morozova, E.V.; Vlasova, J.J.; Ivanova, M.O.; Moiseev, I.S.; Afanasyev, B.V. Clinical Correlates and Prognostic Significance of IL-8, SIL-2R, and Immunoglobulin-Free Light Chain Levels in Patients with Myelofibrosis. Oncol. Res. Treat. 2017, 40, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, M.Z.; Bujko, K.; Cymer, M.; Thapa, A.; Adamiak, M.; Ratajczak, J.; Abdel-Latif, A.K.; Kucia, M. The Nlrp3 Inflammasome as a “Rising Star” in Studies of Normal and Malignant Hematopoiesis. Leukemia 2020, 34, 1512–1523. [Google Scholar] [CrossRef] [PubMed]
- Di Battista, V.; Bochicchio, M.T.; Giordano, G.; Napolitano, M.; Lucchesi, A. Genetics and Pathogenetic Role of Inflammasomes in Philadelphia Negative Chronic Myeloproliferative Neoplasms: A Narrative Review. Int. J. Mol. Sci. 2021, 22, 561. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, M.Z.; Adamiak, M.; Thapa, A.; Bujko, K.; Brzezniakiewicz-Janus, K.; Lenkiewicz, A.M. NLRP3 Inflammasome Couples Purinergic Signaling with Activation of the Complement Cascade for the Optimal Release of Cells from Bone Marrow. Leukemia 2019, 33, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yan, S.; Liu, N.; He, N.; Zhang, A.; Meng, S.; Ji, C.; Ma, D.; Ye, J. Genetic Polymorphisms and Expression of NLRP3 Inflammasome-Related Genes Are Associated with Philadelphia Chromosome-Negative Myeloproliferative Neoplasms. Hum. Immunol. 2020, 81, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Basiorka, A.A.; McGraw, K.L.; Eksioglu, E.A.; Chen, X.; Johnson, J.; Zhang, L.; Zhang, Q.; Irvine, B.A.; Cluzeau, T.; Sallman, D.A.; et al. The NLRP3 Inflammasome Functions as a Driver of the Myelodysplastic Syndrome Phenotype. Blood 2016, 128, 2960–2975. [Google Scholar] [CrossRef] [PubMed]
- Sallman, D.A.; List, A. The Central Role of Inflammatory Signaling in the Pathogenesis of Myelodysplastic Syndromes. Blood 2019, 133, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Xia, L.; Liu, Y.C.; Hochman, T.; Bizzari, L.; Aruch, D.; Lew, J.; Weinberg, R.; Goldberg, J.D.; Hoffman, R. Lipocalin Produced by Myelofibrosis Cells Affects the Fate of Both Hematopoietic and Marrow Microenvironmental Cells. Blood 2015, 126, 972–982. [Google Scholar] [CrossRef]
- Allegra, A.; Alonci, A.; Bellomo, G.; Campo, S.; Cannavò, A.; Penna, G.; Russo, S.; Centorrino, R.; Gerace, D.; Petrungaro, A.; et al. Increased Serum Levels of Neutrophil Gelatinase-Associated Lipocalin in Patients with Essential Thrombocythemia and Polycythemia Vera. Leuk. Lymphoma 2011, 52, 101–107. [Google Scholar] [CrossRef]
- Bauvois, B.; Susin, S.A. Revisiting Neutrophil Gelatinase-Associated Lipocalin (Ngal) in Cancer: Saint or Sinner? Cancers 2018, 10, 336. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Prakash, S.; Lu, M.; Tripodi, J.; Ye, F.; Najfeld, V.; Li, Y.; Schwartz, M.; Weinberg, R.; Roda, P.; et al. Spleens of Myelofibrosis Patients Contain Malignant Hematopoietic Stem Cells. J. Clin. Investig. 2012, 122, 3888–3899. [Google Scholar] [CrossRef]
- Kagoya, Y.; Yoshimi, A.; Tsuruta-Kishino, T.; Arai, S.; Satoh, T.; Akira, S.; Kurokawa, M. Regular Article MYELOID NEOPLASIA JAK2V617F 1 Myeloproliferative Neoplasm Clones Evoke Paracrine DNA Damage to Adjacent Normal Cells through Secretion of Lipocalin-2. Blood J. Am. Soc. Hematol. 2014, 124, 2996–3006. [Google Scholar] [CrossRef]
- Cacemiro, C.; Tognon, R.; Souza, N.; De Pinto, B.; Figueiredo-pontes, L.L.; De Fernando, L.; Catto, B.; Traina, F.; Xisto, E.; Albani, F.; et al. Hematology, Transfusion and Cell Therapy Original Article Philadelphia-Negative Myeloproliferative Neoplasms as Disorders Marked by Cytokine Modulation. Hematol. Transfus. Cell Ther. 2018, 40, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.J.; Baltay, M.; Getz, A.; Fuhrman, K.; Aster, J.C.; Hasserjian, R.P.; Pozdnyakova, O. Gene Expression Profiling Distinguishes Prefibrotic from Overtly Fibrotic Myeloproliferative Neoplasms and Identifies Disease Subsets with Distinct Inflammatory Signatures. PLoS ONE 2019, 14, e0216810. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.Y.; Brooks, S.A.; Craver, B.M.; Morse, S.J.; Nguyen, T.K.; Haghighi, N.; Garbati, M.R.; Fleischman, A.G. Defective Negative Regulation of Toll-like Receptor Signaling Leads to Excessive TNF-a in Myeloproliferative Neoplasm. Blood Adv. 2019, 3, 122–131. [Google Scholar] [CrossRef]
- Heaton, W.L.; Senina, A.V.; Pomicter, A.D.; Salama, M.E.; Clair, P.M.; Yan, N.; Bell, R.N.; Gililland, J.M.; Prchal, J.T.; O’Hare, T.; et al. Chronic Myeloproliferative Neoplasms Autocrine Tnf Signaling Favors Malignant Cells in Myelofibrosis in a Tnfr2-Dependent Fashion. Leukemia 2018, 32, 2399–2411. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Baldauf, C.K.; Haage, T.R.; Waldleben, A.M.; Richter, F.; Pfizenmaier, K.; Fischer, T. Anti-Inflammatory Treatment in MPN: Targeting TNFR1 and TNFR2 in JAK2-V617F–Induced Disease. Blood Adv. 2021, 5, 5349–5359. [Google Scholar] [CrossRef] [PubMed]
- White, T.A.; Johnson, T.; Zarzhevsky, N.; Tom, C.; Delacroix, S.; Holroyd, E.W.; Maroney, S.A.; Singh, R.; Pan, S.; Fay, W.P.; et al. Endothelial-Derived Tissue Factor Pathway Inhibitor Regulates Arterial Thrombosis but Is Not Required for Development or Hemostasis. Blood 2010, 116, 1787–1794. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guy, A.; Gourdou-Latyszenok, V.; Le Lay, N.; Peghaire, C.; Kilani, B.; Dias, J.V.; Duplaa, C.; Renault, M.A.; Denis, C.; Villeval, J.L.; et al. Vascular Endothelial Cell Expression of JAK2 V617F Is Sufficient to Promote a Pro-Thrombotic State Due to Increased P-Selectin Expression. Haematologica 2019, 104, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Guadall, A.; Lesteven, E.; Letort, G.; Toor, S.A.; Delord, M.; Pognant, D.; Brusson, M.; Verger, E.; Maslah, N.; Giraudier, S.; et al. Endothelial Cells Harbouring the JAK2 V617F Mutation Display Pro-Adherent and Pro-Thrombotic Features. Thromb. Haemost. 2018, 118, 1586–1599. [Google Scholar] [CrossRef]
- Yoshida, H.; Granger, D.N. Inflammatory Bowel Disease: A Paradigm for the Link between Coagulation and Inflammation. Inflamm. Bowel Dis. 2009, 15, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Bounes, F.V.; Mémier, V.; Marcaud, M.; Jacquemin, A.; Hamzeh-Cognasse, H.; Garcia, C.; Series, J.; Sié, P.; Minville, V.; Gratacap, M.P.; et al. Platelet Activation and Prothrombotic Properties in a Mouse Model of Peritoneal Sepsis. Sci. Rep. 2018, 8, 13536. [Google Scholar] [CrossRef] [PubMed]
- Bjørn, M.E.; Hasselbalch, H.C. The Role of Reactive Oxygen Species in Myelofibrosis and Related Neoplasms. Mediat. Inflamm. 2015, 2015, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Moisa, C.; Gaman, M.A.; Diaconu, C.C.; Gaman, A.M. Oxidative Stress Levels, JAK2V617F Mutational Status and Thrombotic Complications in Patients with Essential Thrombocythemia. Rev. Chim. 2019, 70, 2822–2825. [Google Scholar] [CrossRef]
- Gaman, A.M.; Moisa, C.; Diaconu, C.C.; Gaman, M.A. Crosstalk between Oxidative Stress, Chronic Inflammation and Disease Progression in Essential Thrombocythemia. Rev. Chim. 2019, 70, 3486–3489. [Google Scholar] [CrossRef]
- Multhoff, G.; Molls, M.; Radons, J. Chronic Inflammation in Cancer Development. Front. Immunol. 2012, 2, 98. [Google Scholar] [CrossRef] [PubMed]
- Hasselbalch, H.C.; Bjørn, M.E. MPNs as Inflammatory Diseases: The Evidence, Consequences, and Perspectives. Mediat. Inflamm. 2015, 2015, 1–16. [Google Scholar] [CrossRef]
- Zhao, H.; Wu, L.; Yan, G.; Chen, Y.; Zhou, M.; Wu, Y.; Li, Y. Inflammation and Tumor Progression: Signaling Pathways and Targeted Intervention. Signal Transduct. Target. Ther. 2021, 6, 263. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.A.; Pfeiffer, R.M.; Landgren, O.; Gadalla, S.; Berndt, S.I.; Engels, E.A. Risks of Myeloid Malignancies in Patients with Autoimmune Conditions. Br. J. Cancer 2009, 100, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Kristinsson, S.Y.; Björkholm, M.; Hultcrantz, M.; Derolf, Å.R.; Landgren, O.; Goldin Sigurdur, Y.; Kristinsson, L.R.; Björkholm, M.; Goldin, L.R.; Kristinsson, S.Y. Chronic Immune Stimulation Might Act As a Trigger for the Development of Acute Myeloid Leukemia or Myelodysplastic Syndromes. J. Clin. Oncol. 2011, 29, 2897–2903. [Google Scholar] [CrossRef] [PubMed]
- Kristinsson, S.Y.; Landgren, O.; Samuelsson, J.; Björkholm, M.; Goldin, L.R. Autoimmunity and the Risk of Myeloproliferative Neoplasms. Haematologica 2010, 95, 1216–1220. [Google Scholar] [CrossRef] [PubMed]
- Hermouet, S.; Vilaine, M. The JAK2 46/1 Haplotype: A Marker of Inappropriate Myelomonocytic Response to Cytokine Stimulation, Leading to Increased Risk of Inflammation, Myeloid Neoplasm, and Impaired Defense against Infection? Haematologica 2011, 96, 1575–1579. [Google Scholar] [CrossRef] [PubMed]
- Olcaydu, D.; Harutyunyan, A.; Jäger, R.; Berg, T.; Gisslinger, B.; Pabinger, I.; Gisslinger, H.; Kralovics, R. A Common JAK2 Haplotype Confers Susceptibility to Myeloproliferative Neoplasms. Nat. Genet. 2009, 41, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, S.; Fontanillas, P.; Flannick, J.; Manning, A.; Grauman, P.V.; Mar, B.G.; Lindsley, R.C.; Mermel, C.H.; Burtt, N.; Chavez, A.; et al. Age-Related Clonal Hematopoiesis Associated with Adverse Outcomes. N. Engl. J. Med. 2014, 371, 2488–2498. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.; Lee, J.; Mitchell, E.; Moore, L.; Baxter, E.J.; Hewinson, J.; Dawson, K.J.; Menzies, A.; Godfrey, A.L.; Green, A.R.; et al. Life Histories of Myeloproliferative Neoplasms Inferred from Phylogenies. Nature 2022, 602, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.; Lee, J.; Moore, L.; Baxter, J.E.; Hewinson, J.; Dawson, K.J.; Menzies, A.; Godfrey, A.L.; Green, A.R.; Campbell, P.J.; et al. Driver Mutation Acquisition in Utero and Childhood Followed by Lifelong Clonal Evolution Underlie Myeloproliferative Neoplasms. Blood 2020, 136, LBA-1. [Google Scholar] [CrossRef]
- Van Egeren, D.; Escabi, J.; Nguyen, M.; Liu, S.; Reilly, C.R.; Patel, S.; Kamaz, B.; Kalyva, M.; DeAngelo, D.J.; Galinsky, I.; et al. Reconstructing the Lineage Histories and Differentiation Trajectories of Individual Cancer Cells in Myeloproliferative Neoplasms. Cell Stem Cell 2021, 28, 514–523.e9. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.; Lee, J.; Moore, L.; Baxter, E.J.; Hewinson, J.; Dawson, K.J.; Menzies, A.; Godfrey, A.L.; Green, A.R.; Campbell, P.J.; et al. Phylogenetic Reconstruction of Myeloproliferative Neoplasm Reveals Very Early Origins and Lifelong Evolution. bioRxiv 2020. [Google Scholar] [CrossRef]
- Landolfi, R.; Marchioli, R.; Kutti, J.; Gisslinger, H.; Tognoni, G.; Patrono, C.; Barbui, T. Efficacy and Safety of Low-Dose Aspirin in Polycythemia Vera. N. Engl. J. Med. 2004, 350, 114–124. [Google Scholar] [CrossRef]
- Tefferi, A.; Elliott, M. Thrombosis in Myeloproliferative Disorders: Prevalence, Prognostic Factors, and the Role of Leukocytes and JAK2V617F. Semin. Thromb. Hemost. 2007, 33, 313–320. [Google Scholar] [CrossRef]
- Vannucchi, A.M.; Antonioli, E.; Guglielmelli, P.; Longo, G.; Pancrazzi, A.; Ponziani, V.; Bogani, C.; Ferrini, P.R.; Rambaldi, A.; Guerini, V.; et al. Prospective Identification of High-Risk Polycythemia Vera Patients Based on JAK2 V617F Allele Burden. Leukemia 2007, 21, 1952–1959. [Google Scholar] [CrossRef]
- Kaifie, A.; Kirschner, M.; Wolf, D.; Maintz, C.; Hänel, M.; Gattermann, N.; Gökkurt, E.; Platzbecker, U.; Hollburg, W.; Göthert, J.R.; et al. Bleeding, Thrombosis, and Anticoagulation in Myeloproliferative Neoplasms (MPN): Analysis from the German SAL-MPN-Registry. J. Hematol. Oncol. 2016, 9, 18. [Google Scholar] [CrossRef]
- Gecht, J.; Tsoukakis, I.; Kricheldorf, K.; Stegelmann, F.; Klausmann, M.; Griesshammer, M.; Schulz, H.; Hollburg, W.; Göthert, J.R.; Sockel, K.; et al. Kidney Dysfunction Is Associated with Thrombosis and Disease Severity in Myeloproliferative Neoplasms: Implications from the German Study Group for MPN Bioregistry. Cancers 2021, 13, 4086. [Google Scholar] [CrossRef]
- Swystun, L.L.; Liaw, P.C. The Role of Leukocytes in Thrombosis. Blood 2016, 128, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Yago, T.; Liu, Z.; Ahamed, J.; McEver, R.P. Cooperative PSGL-1 and CXCR2 Signaling in Neutrophils Promotes Deep Vein Thrombosis in Mice. Blood 2018, 132, 1426–1437. [Google Scholar] [CrossRef]
- Alvarez-Larrán, A.; Arellano-Rodrigo, E.; Reverter, J.C.; Domingo, A.; Villamor, N.; Colomer, D.; Cervantes, F. Increased Platelet, Leukocyte, and Coagulation Activation in Primary Myelofibrosis. Ann. Hematol. 2008, 87, 269–276. [Google Scholar] [CrossRef]
- Dhanesha, N.; Jain, M.; Tripathi, A.K.; Doddapattar, P.; Chorawala, M.; Bathla, G.; Nayak, M.K.; Ghatge, M.; Lentz, S.R.; Kon, S.; et al. Targeting Myeloid-Specific Integrin A9β1 Improves Short-and Long-Term Stroke Outcomes in Murine Models with Preexisting Comorbidities by Limiting Thrombosis and Inflammation. Circ. Res. 2020, 126, 1779–1794. [Google Scholar] [CrossRef]
- Awasthi, D.; Nagarkoti, S.; Kumar, A.; Dubey, M.; Singh, A.K.; Pathak, P.; Chandra, T.; Barthwal, M.K.; Dikshit, M. Oxidized LDL Induced Extracellular Trap Formation in Human Neutrophils via TLR-PKC-IRAK-MAPK and NADPH-Oxidase Activation. Free Radic. Biol. Med. 2016, 93, 190–203. [Google Scholar] [CrossRef]
- Delgado-Rizo, V.; Martínez-Guzmán, M.A.; Iñiguez-Gutierrez, L.; García-Orozco, A.; Alvarado-Navarro, A.; Fafutis-Morris, M. Neutrophil Extracellular Traps and Its Implications in Inflammation: An Overview. Front. Immunol. 2017, 8, 81. [Google Scholar] [CrossRef] [PubMed]
- Elaskalani, O.; Abdol Razak, N.B.; Metharom, P. Neutrophil Extracellular Traps Induce Aggregation of Washed Human Platelets Independently of Extracellular DNA and Histones. Cell Commun. Signal. 2018, 16, 1–15. [Google Scholar] [CrossRef]
- Wolach, O.; Sellar, R.S.; Martinod, K.; Cherpokova, D.; McConkey, M.; Chappell, R.J.; Silver, A.J.; Adams, D.; Castellano, C.A.; Schneider, R.K.; et al. Increased Neutrophil Extracellular Trap Formation Promotes Thrombosis in Myeloproliferative Neoplasms. Sci. Transl. Med. 2018, 10, eaan8292. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.F.; Hunter, R.W.; Harper, M.T.; Savage, J.S.; Siddiq, S.; Westbury, S.K.; Poole, A.W.; Mumford, A.D.; Hers, I. Dysfunction of the PI3 Kinase/Rap1/Integrin AIIbb3 Pathway Underlies Ex Vivo Platelet Hypoactivity in Essential Thrombocythemia. Blood 2013, 121, 1209–1219. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, C.M.; Manning, H.; Bennett, C.; Vasquez, L.; Severin, S.; Brain, L.; Mazharian, A.; Guerrero, J.A.; Li, J.; Soranzo, N.; et al. JAK2V617F Leads to Intrinsic Changes in Platelet Formation and Reactivity in a Knock-in Mouse Model of Essential Thrombocythemia. Blood 2013, 122, 3787–3797. [Google Scholar] [CrossRef] [PubMed]
- Lamrani, L.; Lacout, C.; Ollivier, V.; Denis, C.V.; Gardiner, E.; Noe, B.H.T.; Vainchenker, W.; Villeval, J.L.; Jandrot-Perrus, M. Hemostatic Disorders in a JAK2V617F-Driven Mouse Model of Myeloproliferative Neoplasm. Blood 2014, 124, 1136–1145. [Google Scholar] [CrossRef] [PubMed]
- Hauschner, H.; Horev, M.B.; Misgav, M.; Nagar, M.; Seligsohn, U.; Rosenberg, N.; Koren-Michowitz, M. Platelets from Calreticulin Mutated Essential Thrombocythemia Patients Are Less Reactive than JAK2 V617F Mutated Platelets. Am. J. Hematol. 2020, 95, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Etheridge, S.L.; Roh, M.E.; Cosgrove, M.E.; Sangkhae, V.; Fox, N.E.; Chen, J.; López, J.A.; Kaushansky, K.; Hitchcock, I.S. JAK2V617F-Positive Endothelial Cells Contribute to Clotting Abnormalities in Myeloproliferative Neoplasms. Proc. Natl. Acad. Sci. USA 2014, 111, 2295–2300. [Google Scholar] [CrossRef]
- De Grandis, M.; Cambot, M.; Wautier, M.-P.; Cassinat, B.; Chomienne, C.; Colin, Y.; Wautier, J.-L.; Le Van Kim, C.; El Nemer, W. JAK2V617F Activates Lu/BCAM-Mediated Red Cell Adhesion in Polycythemia Vera through an EpoR-Independent Rap1/Akt Pathway. Blood 2013, 121, 658–665. [Google Scholar] [CrossRef]
- Pearson, T.C.; Wetherley-Mein, G. Vascular Occlusive Episodes and Venous Hæmatocrit in Primary Proliferative Polycythæmlx. Lancet 1978, 312, 1219–1222. [Google Scholar] [CrossRef]
- McMullin, M.F.; Harrison, C.N.; Kiladjian, J.J.; Leone, G.; Prchal, J.T.; Gordeuk, V.R.; Marchioli, R.; Vannucchi, A.M.; Barbui, T.; Spivak, J.L. Treatment Target in Polycythemia Vera. N. Engl. J. Med. 2013, 368, 1554–1557. [Google Scholar] [CrossRef] [PubMed]
- Walton, B.L.; Lehmann, M.; Skorczewski, T.; Holle, L.A.; Beckman, J.D.; Cribb, J.A.; Mooberry, M.J.; Wufsus, A.R.; Cooley, B.C.; Homeister, J.W.; et al. Elevated Hematocrit Enhances Platelet Accumulation Following Vascular Injury. Blood 2017, 129, 2537–2546. [Google Scholar] [CrossRef] [PubMed]
- Vainchenker, W.; Kralovics, R. Genetic Basis and Molecular Pathophysiology of Classical Myeloproliferative Neoplasms. Blood 2017, 129, 667–679. [Google Scholar] [CrossRef] [PubMed]
- Grinfeld, J.; Nangalia, J.; Green, A.R. Molecular Determinants of Pathogenesis and Clinical Phenotype in Myeloproliferative Neoplasms. Haematologica 2017, 102, 7–17. [Google Scholar] [CrossRef]
- Migliaccio, A.R.; Malara, A.; Aliabadi, H.M.; Bousoik, E. “Do We Know Jack” About JAK? A Closer Look at JAK/STAT Signaling Pathway. Front. Oncol. 2018, 1, 287. [Google Scholar] [CrossRef]
- Bharadwaj, U.; Kasembeli, M.M.; Robinson, P.; Tweardy, D.J. Targeting Janus Kinases and Signal Transducer and Activator of Transcription 3 to Treat Inflammation, Fibrosis, and Cancer: Rationale, Progress, and Caution. Pharmacol. Rev. 2020, 72, 486–526. [Google Scholar] [CrossRef]
- Wingelhofer, B.; Neubauer, H.A.; Valent, P.; Han, X.; Constantinescu, S.N.; Gunning, P.T.; Müller, M.; Moriggl, R. Implications of STAT3 and STAT5 Signaling on Gene Regulation and Chromatin Remodeling in Hematopoietic Cancer. Leukemia 2018, 32, 1713–1726. [Google Scholar] [CrossRef]
- Tiacci, E.; Ladewig, E.; Schiavoni, G.; Penson, A.; Fortini, E.; Pettirossi, V.; Wang, Y.; Rosseto, A.; Venanzi, A.; Vlasevska, S.; et al. Pervasive Mutations of JAK-STAT Pathway Genes in Classical Hodgkin Lymphoma. Blood 2018, 131, 2454–2465. [Google Scholar] [CrossRef]
- Liu, Y.; Easton, J.; Shao, Y.; Maciaszek, J.; Wang, Z.; Wilkinson, M.R.; Mccastlain, K.; Edmonson, M.; Pounds, S.B.; Shi, L.; et al. The Genomic Landscape of Pediatric and Young Adult T-Lineage Acute Lymphoblastic Leukemia. Nat. Genet. 2017, 49, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, S.; Bar-Natan, M.; Mascarenhas, J.O. JAKs to STATs: A Tantalizing Therapeutic Target in Acute Myeloid Leukemia. Blood Rev. 2020, 40, 100634. [Google Scholar] [CrossRef] [PubMed]
- Bartalucci, N.; Calabresi, L.; Balliu, M.; Martinelli, S.; Rossi, M.C.; Villeval, J.L.; Annunziato, F.; Guglielmelli, P.; Vannucchi, A.M. Inhibitors of the PI3K/MTOR Pathway Prevent STAT5 Phosphorylation in JAK2V617F Mutated Cells through PP2A/CIP2A Axis. Oncotarget 2017, 8, 96710–96724. [Google Scholar] [CrossRef]
- Guglielmelli, P.; Barosi, G.; Rambaldi, A.; Marchioli, R.; Masciulli, A.; Tozzi, L.; Biamonte, F.; Bartalucci, N.; Gattoni, E.; Lupo, M.L.; et al. Safety and Efficacy of Everolimus, a MTOR Inhibitor, as Single Agent in a Phase 1/2 Study in Patients with Myelofibrosis. Blood 2011, 118, 2069–2076. [Google Scholar] [CrossRef]
- Steelman, L.S.; Pohnert, S.C.; Shelton, J.G.; Franklin, R.A.; Bertrand, F.E.; McCubrey, J.A. JAK/STAT, Raf/MEK/ERK, PI3K/Akt and BCR-ABL in Cell Cycle Progression and Leukemogenesis. Leukemia 2004, 18, 189–218. [Google Scholar] [CrossRef]
- Guijarro-hernández, A.; Vizmanos, J.L. A Broad Overview of Signaling in Ph-negative Classic Myeloproliferative Neoplasms. Cancers 2021, 13, 984. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shiratori, I.; Uehori, J.; Ikawa, M.; Arase, H. Neutrophil Infiltration during Inflammation Is Regulated by PILRα via Modulation of Integrin Activation. Nat. Immunol. 2013, 14, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Hogg, N.; Patzak, I.; Willenbrock, F. Immunological Synapses The Insider’s Guide to Leukocyte Integrin Signalling and Function. Nat. Rev. Immunol. 2011, 11, 416–426. [Google Scholar] [CrossRef]
- Kinashi, T. Intracellular Signalling Controlling Integrin Activation in Lymphocytes. Nat. Rev. Immunol. 2005, 5, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Ley, K.; Laudanna, C.; Cybulsky, M.I.; Nourshargh, S. Getting to the Site of Inflammation: The Leukocyte Adhesion Cascade Updated. Nat. Rev. Immunol. 2007, 7, 678–689. [Google Scholar] [CrossRef]
- Montresor, A.; Bolomini-Vittori, M.; Toffali, L.; Rossi, B.; Constantin, G.; Laudanna, C. JAK Tyrosine Kinases Promote Hierarchical Activation of Rho and Rap Modules of Integrin Activation. J. Cell Biol. 2013, 203, 1003–1019. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, K.; Kinashi, T. Rap1 and Integrin Inside-out Signaling. Methods Mol. Biol. 2011, 757, 279–296. [Google Scholar] [CrossRef]
- Bromberger, T.; Klapproth, S.; Rohwedder, I.; Zhu, L.; Mittmann, L.; Reichel, C.A.; Sperandio, M.; Qin, J.; Moser, M. Direct Rap1/Talin1 Interaction Regulates Platelet and Neutrophil Integrin Activity in Mice. Blood J. Am. Soc. Hematol. 2018, 132, 2754–2762. [Google Scholar] [CrossRef] [PubMed]
- Montresor, A.; Toffali, L.; Constantin, G.; Laudanna, C. Chemokines and the Signaling Modules Regulating Integrin Affinity. Front. Immunol. 2012, 3, 127. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Edelmann, B.; Schnoeder, T.M.; Saalfeld, F.C.; Wolleschak, D.; Kliche, S.; Schraven, B.; Heidel, F.H.; Fischer, T. JAK2-V617F Activates Β1-Integrin-Mediated Adhesion of Granulocytes to Vascular Cell Adhesion Molecule 1. Leukemia 2017, 31, 1223–1226. [Google Scholar] [CrossRef]
- Bergmeier, W.; Goerge, T.; Wang, H.W.; Crittenden, J.R.; Baldwin, A.C.W.; Cifuni, S.M.; Housman, D.E.; Graybiel, A.M.; Wagner, D.D. Mice Lacking the Signaling Molecule CalDAG-GEFI Represent a Model for Leukocyte Adhesion Deficiency Type III. J. Clin. Investig. 2007, 117, 1699–1707. [Google Scholar] [CrossRef]
- Stadtmann, A.; Brinkhaus, L.; Mueller, H.; Rossaint, J.; Bolomini-Vittori, M.; Bergmeier, W.; Van Aken, H.; Wagner, D.D.; Laudanna, C.; Ley, K.; et al. Rap1a Activation by CalDAG-GEFI and P38 MAPK Is Involved in E-Selectin-Dependent Slow Leukocyte Rolling. Eur. J. Immunol. 2011, 41, 2074–2085. [Google Scholar] [CrossRef]
- Carbo, C.; Duerschmied, D.; Goerge, T.; Hattori, H.; Sakai, J.; Cifuni, S.M.; White, G.C.; Chrzanowska-Wodnicka, M.; Luo, H.R.; Wagner, D.D. Integrin-Independent Role of CalDAG-GEFI in Neutrophil Chemotaxis. J. Leukoc. Biol. 2010, 88, 313–319. [Google Scholar] [CrossRef]
- Ziegler, S.; Gartner, K.; Scheuermann, U.; Zoeller, T.; Hantzschmann, J.; Over, B.; Foermer, S.; Heeg, K.; Bekeredjian-Ding, I. Ca2+-Related Signaling Events Influence TLR9-Induced IL-10 Secretion in Human B Cells. Eur. J. Immunol. 2014, 44, 1285–1298. [Google Scholar] [CrossRef]
- Tang, S.; Chen, T.; Yang, M.; Wang, L.; Yu, Z.; Xie, B.; Qian, C.; Xu, S.; Li, N.; Cao, X.; et al. Extracellular Calcium Elicits Feedforward Regulation of the Toll-like Receptor-Triggered Innate Immune Response. Cell. Mol. Immunol. 2017, 14, 180–191. [Google Scholar] [CrossRef]
- Kosuru, R.; Chrzanowska, M. Molecular Sciences Integration of Rap1 and Calcium Signaling. Int. J. Mol. Sci. 2020, 21, 1616. [Google Scholar] [CrossRef]
- Mele, S.; Devereux, S.; Pepper, A.G.; Infante, E.; Ridley, A.J. Calcium-RasGRP2-Rap1 Signaling Mediates CD38-Induced Migration of Chronic Lymphocytic Leukemia Cells. Blood Adv. 2018, 2, 1551–1561. [Google Scholar] [CrossRef]
- Catani, L.; Cavo, M.; Palandri, F. The Power of Extracellular Vesicles in Myeloproliferative Neoplasms: “Crafting” a Microenvironment That Matters. Cells 2021, 10, 2316. [Google Scholar] [CrossRef]
- Găman, M.A.; Cozma, M.A.; Dobrică, E.C.; Crețoiu, S.M.; Găman, A.M.; Diaconu, C.C. Liquid Biopsy and Potential Liquid Biopsy-Based Biomarkers in Philadelphia-Negative Classical Myeloproliferative Neoplasms: A Systematic Review. Life 2021, 11, 677. [Google Scholar] [CrossRef]
- Fel, A.; Lewandowska, A.E.; Petrides, P.E.; Wiśniewski, J.R. Comparison of Proteome Composition of Serum Enriched in Extracellular Vesicles Isolated from Polycythemia Vera Patients and Healthy Controls. Proteomes 2019, 7, 20. [Google Scholar] [CrossRef]
- Charpentier, A.; Lebreton, A.; Rauch, A.; Bauters, A.; Trillot, N.; Nibourel, O.; Tintillier, V.; Wemeau, M.; Demory, J.-L.; Preudhomme, C.; et al. Microparticle Phenotypes Are Associated with Driver Mutations and Distinct Thrombotic Risks in Essential Thrombocythemia. Haematologica 2016, 101, e365–e368. [Google Scholar] [CrossRef]
- Woywodt, A.; Scheer, J.; Hambach, L.; Buchholz, S.; Ganser, A.; Haller, H.; Hertenstein, B.; Haubitz, M. Circulating Endothelial Cells as a Marker of Endothelial Damage in Allogeneic Hematopoietic Stem Cell Transplantation. Blood 2004, 103, 3603–3605. [Google Scholar] [CrossRef]
- Torres, C.; Fonseca, A.M.; Leander, M.; Matos, R.; Morais, S.; Campos, M.; Lima, M. Circulating Endothelial Cells in Patients with Venous Thromboembolism and Myeloproliferative Neoplasms. PLoS ONE 2013, 8, e81574. [Google Scholar] [CrossRef]
- Treliński, J.; Wierzbowska, A.; Krawczyńska, A.; Sakowicz, A.; Pietrucha, T.; Smolewski, P.; Robak, T.; Chojnowski, K. Circulating Endothelial Cells in Essential Thrombocythemia and Polycythemia Vera: Correlation with JAK2-V617F Mutational Status, Angiogenic Factors and Coagulation Activation Markers. Int. J. Hematol. 2010, 91, 792–798. [Google Scholar] [CrossRef]
- Barbui, T.; Barosi, G.; Birgegard, G.; Cervantes, F.; Finazzi, G.; Griesshammer, M.; Harrison, C.; Hasselbalch, H.C.; Hehlmann, R.; Hoffman, R.; et al. Philadelphia-Negative Classical Myeloproliferative Neoplasms: Critical Concepts and Management Recommendations From European LeukemiaNet. J. Clin. Oncol. 2011, 29, 761–770. [Google Scholar] [CrossRef]
- Tefferi, A.; Barbui, T. Polycythemia Vera and Essential Thrombocythemia: 2019 Update on Diagnosis, Risk-Stratification and Management. Am. J. Hematol. 2019, 94, 133–143. [Google Scholar] [CrossRef]
- Tefferi, A.; Barbui, T. Polycythemia Vera and Essential Thrombocythemia: 2021 Update on Diagnosis, Risk-Stratification and Management. Am. J. Hematol. 2020, 95, 1599–1613. [Google Scholar] [CrossRef]
- Dillinger, J.G.; Sollier, C.B.D.; Sideris, G.; Ronez, E.; Henry, P.; Drouet, L. Twice Daily Aspirin to Improve Biological Aspirin Efficacy in Patients with Essential Thrombocytemia. Thromb. Res. 2012, 129, 91–94. [Google Scholar] [CrossRef]
- Pascale, S.; Petrucci, G.; Dragani, A.; Habib, A.; Zaccardi, F.; Pagliaccia, F.; Pocaterra, D.; Ragazzoni, E.; Rolandi, G.; Rocca, B.; et al. Aspirin-Insensitive Thromboxane Biosynthesis in Essential Thrombocythemia Is Explained by Accelerated Renewal of the Drug Target. Blood 2012, 119, 3595–3603. [Google Scholar] [CrossRef]
- Tremblay, D.; Kosiorek, H.E.; Dueck, A.C.; Hoffman, R. Evaluation of Therapeutic Strategies to Reduce the Number of Thrombotic Events in Patients With Polycythemia Vera and Essential Thrombocythemia. Front. Oncol. 2021, 10, 636675. [Google Scholar] [CrossRef]
- Kreher, S.; Ochsenreither, S.; Trappe, R.U.; Pabinger, I.; Bergmann, F.; Petrides, P.E.; Koschmieder, S.; Matzdorff, A.; Tiede, A.; Griesshammer, M.; et al. Prophylaxis and Management of Venous Thromboembolism in Patients with Myeloproliferative Neoplasms: Consensus Statement of the Haemostasis Working Party of the German Society of Hematology and Oncology (DGHO), the Austrian Society of Hematology and Oncolo. Ann. Hematol. 2014, 93, 1953–1963. [Google Scholar] [CrossRef]
- Alvarez-Larrán, A.; Martínez-Avilés, L.; Hernández-Boluda, J.C.; Ferrer-Marín, F.; Antelo, M.L.; Burgaleta, C.; Mata, M.I.; Xicoy, B.; Martínez-Trillos, A.; Gómez-Casares, M.T.; et al. Busulfan in Patients with Polycythemia Vera or Essential Thrombocythemia Refractory or Intolerant to Hydroxyurea. Ann. Hematol. 2014, 93, 2037–2043. [Google Scholar] [CrossRef]
- Harrison, C.N.; Campbell, P.J.; Buck, G.; Wheatley, K.; East, C.L.; Bareford, D.; Wilkins, B.S.; van der Walt, J.D.; Reilly, J.T.; Grigg, A.P.; et al. Hydroxyurea Compared with Anagrelide in High-Risk Essential Thrombocythemia. N. Engl. J. Med. 2005, 353, 33–45. [Google Scholar] [CrossRef]
- Vannucchi, A.M.; Kiladjian, J.J.; Griesshammer, M.; Masszi, T.; Durrant, S.; Passamonti, F.; Harrison, C.N.; Pane, F.; Zachee, P.; Mesa, R.; et al. Ruxolitinib versus Standard Therapy for the Treatment of Polycythemia Vera. N. Engl. J. Med. 2015, 372, 426–435. [Google Scholar] [CrossRef]
- Passamonti, F.; Griesshammer, M.; Palandri, F.; Egyed, M.; Benevolo, G.; Devos, T.; Callum, J.; Vannucchi, A.M.; Sivgin, S.; Bensasson, C.; et al. Ruxolitinib for the Treatment of Inadequately Controlled Polycythaemia Vera without Splenomegaly (RESPONSE-2): A Randomised, Open-Label, Phase 3b Study. Lancet Oncol. 2017, 18, 88–99. [Google Scholar] [CrossRef]
- Signorelli, H. A Double-Blind, Placebo-Controlled Trial of Ruxolitinib for Myelofibrosis. Yearb. Pathol. Lab. Med. 2013, 2013, 368–369. [Google Scholar] [CrossRef]
- Duncan, P.W.; Sullivan, K.J.; Behrman, A.L.; Azen, S.P.; Wu, S.S.; Nadeau, S.E.; Dobkin, B.H.; Rose, D.K.; Tilson, J.K.; Cen, S.; et al. Body-Weight-Supported Treadmill Rehabilitation after Stroke. N. Engl. J. Med. 2011, 364, 2026–2036. [Google Scholar] [CrossRef]
- Masciulli, A.; Ferrari, A.; Carobbio, A.; Ghirardi, A.; Barbui, T. Ruxolitinib for the Prevention of Thrombosis in Polycythemia Vera: A Systematic Review and Meta-Analysis. Blood Adv. 2020, 4, 380–386. [Google Scholar] [CrossRef]
- Samuelson, B.T.; Vesely, S.K.; Chai-Adisaksopha, C.; Scott, B.L.; Crowther, M.; Garcia, D. The Impact of Ruxolitinib on Thrombosis in Patients with Polycythemia Vera and Myelofibrosis: A Meta-Analysis. Blood Coagul. Fibrinolysis 2016, 27, 648–652. [Google Scholar] [CrossRef]
- Deininger, M.; Radich, J.; Burn, T.C.; Huber, R.; Paranagama, D.; Verstovsek, S. Brief Report The Effect of Long-Term Ruxolitinib Treatment on JAK2p.V617F Allele Burden in Patients with Myelofibrosis. Blood 2015, 126, 1551–1554. [Google Scholar] [CrossRef]
- Huang, B.-T.; Zeng, Q.-C.; Zhao, W.-H.; Li, B.-S.; Chen, R.-L. Interferon-2b Gains High Sustained Response Therapy for Advanced Essential Thrombocythemia and Polycythemia Vera with JAK2V617F Positive Mutation. Leuk. Res. 2014, 38, 1177–1183. [Google Scholar] [CrossRef]
- Masarova, L.; Patel, K.P.; Newberry, K.J.; Cortes, J.; Borthakur, G.; Konopleva, M.; Estrov, Z.; Kantarjian, H.; Verstovsek, S. Pegylated Interferon Alfa-2a in Patients with Essential Thrombocythaemia or Polycythaemia Vera: A Post-Hoc, Median 83 Month Follow-up of an Open-Label, Phase 2 Trial. Lancet Haematol. 2017, 4, e165–e175. [Google Scholar] [CrossRef]
- Kiladjian, J.J.; Mesa, R.A.; Hoffman, R. The Renaissance of Interferon Therapy for the Treatment of Myeloid Malignancies. Blood 2011, 117, 4706–4715. [Google Scholar] [CrossRef]
- Hasan, S.; Lacout, C.; Marty, C.; Cuingnet, M.; Solary, E.; Vainchenker, W.; Villeval, J.L. JAK2V617F Expression in Mice Amplifies Early Hematopoietic Cells and Gives Them a Competitive Advantage That Is Hampered by IFNa. Blood 2013, 122, 1464–1477. [Google Scholar] [CrossRef]
- Mullally, A.; Bruedigam, C.; Poveromo, L.; Heidel, F.H.; Purdon, A.; Vu, T.; Austin, R.; Heckl, D.; Breyfogle, L.J.; Kuhn, C.P.; et al. Depletion of Jak2V617F Myeloproliferative Neoplasm-Propagating Stem Cells by Interferon-α in a Murine Model of Polycythemia Vera. Blood 2013, 121, 3692–3702. [Google Scholar] [CrossRef]
- Kiladjian, J.J.; Chomienne, C.; Fenaux, P. Interferon-α Therapy in Bcr-Abl-Negative Myeloproliferative Neoplasms. Leukemia 2008, 22, 1990–1998. [Google Scholar] [CrossRef] [PubMed]
- Quesada, J.R.; Talpaz, M.; Rios, A.; Kurzrock, R.; Gutterman, J.U. Clinical Toxicity of Interferons in Cancer Patients: A Review. J. Clin. Oncol. 1986, 4, 234–243. [Google Scholar] [CrossRef]
- Yacoub, A.; Mascarenhas, J.; Kosiorek, H.; Prchal, J.T.; Berenzon, D.; Baer, M.R.; Ritchie, E.; Silver, R.T.; Kessler, C.; Winton, E.; et al. Pegylated Interferon Alfa-2a for Polycythemia Vera or Essential Thrombocythemia Resistant or Intolerant to Hydroxyurea. Blood 2019, 134, 1498–1509. [Google Scholar] [CrossRef]
- Kjær, L.; Cordua, S.; Holmström, M.O.; Thomassen, M.; Kruse, T.A.; Pallisgaard, N.; Larsen, T.S.; De Stricker, K.; Skov, V.; Hasselbalch, H.C. Differential Dynamics of CALR Mutant Allele Burden in Myeloproliferative Neoplasms during Interferon Alfa Treatment. PLoS ONE 2016, 11, e0165336. [Google Scholar] [CrossRef] [PubMed]
- Dove, A. CD18 Trials Disappoint Again. Nat. Biotechnol. 2000, 18, 817–818. [Google Scholar] [CrossRef]
- Slack, R.J.; Macdonald, S.J.F.; Roper, J.A.; Jenkins, R.G.; Hatley, R.J.D. Emerging Therapeutic Opportunities for Integrin Inhibitors. Nat. Rev. Drug Discov. 2022, 21, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Ley, K.; Rivera-Nieves, J.; Sandborn, W.J.; Shattil, S. Integrin-Based Therapeutics: Biological Basis, Clinical Use and New Drugs. Nat. Rev. Drug Discov. 2016, 15, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Gao, H.; Shi, C.; Erhardt, P.W.; Pavlovsky, A.; Soloviev, D.A.; Bledzka, K.; Ustinov, V.; Zhu, L.; Qin, J.; et al. Leukocyte Integrin Mac-1 Regulates Thrombosis via Interaction with Platelet GPIbα. Nat. Commun. 2017, 8, 15559. [Google Scholar] [CrossRef]
| References | ||||
|---|---|---|---|---|
| Cytokine | Main Function | PV | ET | MF |
| IL-1β * | Pro-inflammatory, acute phase, stimulating TH/B-cells, proliferation, apoptosis, and differentiation | [26,27] | [26,27] | [21,26,27,28,29,30] |
| IL-1Ra * | Blocking IL-1 | [27,28] | [27] | [21,27,28,31] |
| IL-5 | Growth factor of eosinophils, enhancing B-cell proliferation, and antibody production | [26,27,28,29] | [26,27,28] | [26,27,28,32] |
| IL-6 * | Pro- and anti-inflammatory, acute phase, differentiation, and cytokine production | [26,27,28,29,33,34] | [26,27,29,33,34] | [21,26,31,33,34] |
| IL-8 * | Chemotaxis, activating and degranulating neutrophils, and angiogenesis | [27,28,29,33,35] | [27,29,33] | [21,27,31] |
| IL-10 | Anti-inflammatory and inhibition of pro-inflammatory cytokines | [26,27,32,33] | [26,29,33] | [21,26,27,28,29] |
| TNF-α * | Pro-inflammatory, acute phase, cytokine production, proliferation, and apoptosis | [26,27,29,33,36] | [26,27,29,33,36,37] | [21,26,27,28,30,31,36,37] |
| IFN-α * | Anti-viral | [26,27] | [26,27] | [21,26,27,28,29] |
| IFN-γ * | Promoting TH1 and the cellular immune response and activating macrophages | [26,27,28,33] | [27,29,33] | [26,27,30,31] |
| TGF-β * | Inhibiting growth, activating leucocytes, inducing TReg, apoptotic, antiangiogenic, and healing wounds | [29,38] | [29,38] | [38] |
| VEGF | Vascular growth factor: vasculogenesis and angiogenesis | [27,28,29,33,39] | [27,29,33,39] | [21,27,28,31,32,39] |
| CCL2 (*) (MCP-1) | Recruiting monocytes, activating macrophages, histamine release of basophilic cells, stimulating TH2 | [28,33] | [27,29,33] | [21,27,29] |
| CCL3 (MIP1-a) | Stimulating TH1 and DC | [26,28] | [26] | [21,26,28,29,31] |
| CCL4 (MIP1-b) | Stimulating DC | [26,27,28] | [26,27] | [21,27,29,40] |
| CXCL9 (MIG) | Activation of the acquired immune system | [27,28,32] | [27,32] | [21,27,28,29,32] |
| CXCL10 | Pro-inflammatory, anti-angiogenetic, and stimulating TH1 | [26,27,28,32,36] | [21,26,27,28,29,36] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhuria, V.; Baldauf, C.K.; Schraven, B.; Fischer, T. Thromboinflammation in Myeloproliferative Neoplasms (MPN)—A Puzzle Still to Be Solved. Int. J. Mol. Sci. 2022, 23, 3206. https://doi.org/10.3390/ijms23063206
Bhuria V, Baldauf CK, Schraven B, Fischer T. Thromboinflammation in Myeloproliferative Neoplasms (MPN)—A Puzzle Still to Be Solved. International Journal of Molecular Sciences. 2022; 23(6):3206. https://doi.org/10.3390/ijms23063206
Chicago/Turabian StyleBhuria, Vikas, Conny K. Baldauf, Burkhart Schraven, and Thomas Fischer. 2022. "Thromboinflammation in Myeloproliferative Neoplasms (MPN)—A Puzzle Still to Be Solved" International Journal of Molecular Sciences 23, no. 6: 3206. https://doi.org/10.3390/ijms23063206
APA StyleBhuria, V., Baldauf, C. K., Schraven, B., & Fischer, T. (2022). Thromboinflammation in Myeloproliferative Neoplasms (MPN)—A Puzzle Still to Be Solved. International Journal of Molecular Sciences, 23(6), 3206. https://doi.org/10.3390/ijms23063206






