Apoptosis-Inducing Factor 2 (AIF-2) Mediates a Caspase-Independent Apoptotic Pathway in the Tropical Sea Cucumber (Holothuria leucospilota)
Abstract
:1. Introduction
2. Results
2.1. Molecular Cloning and Sequence Analysis of HlAIF-2
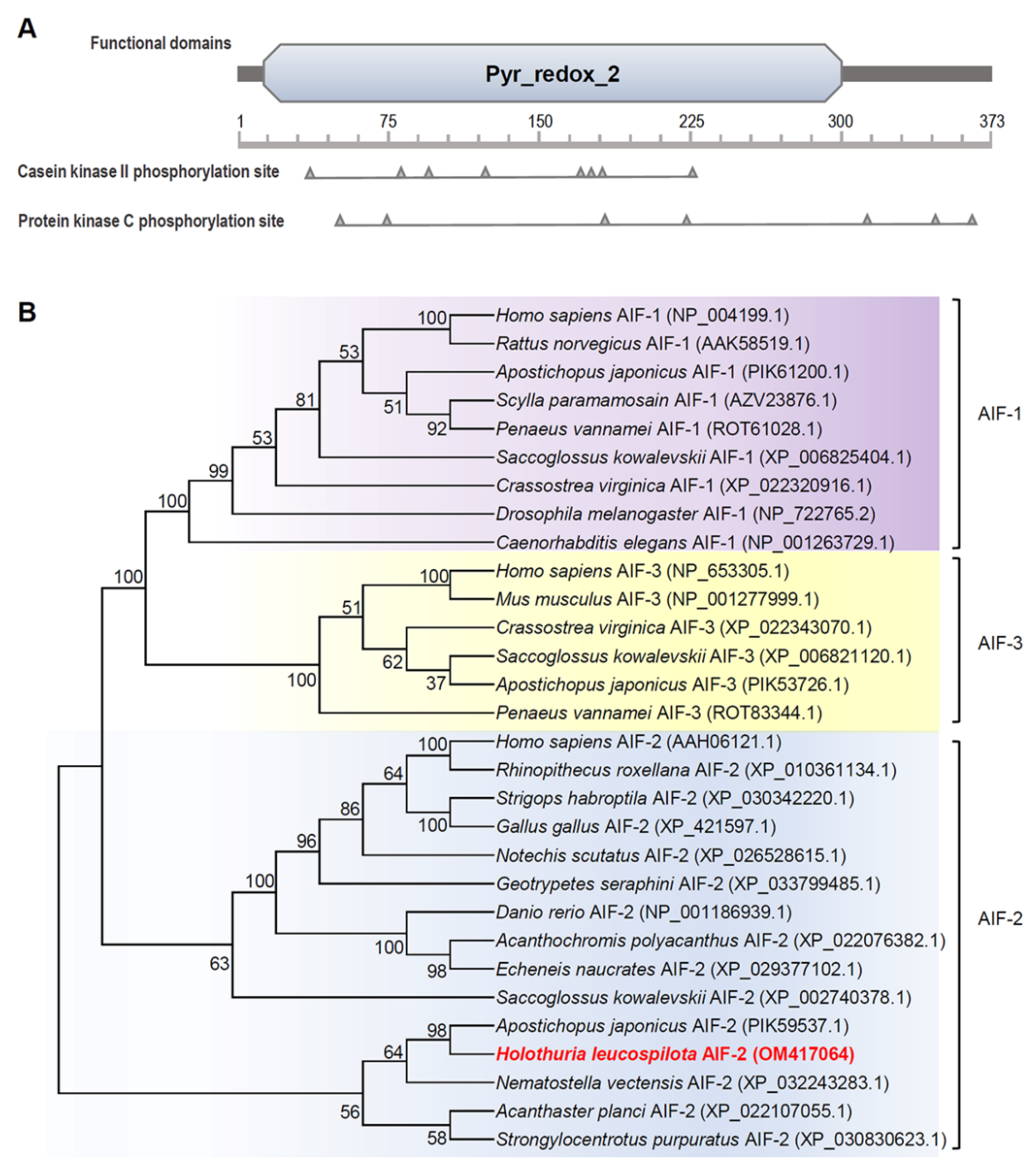
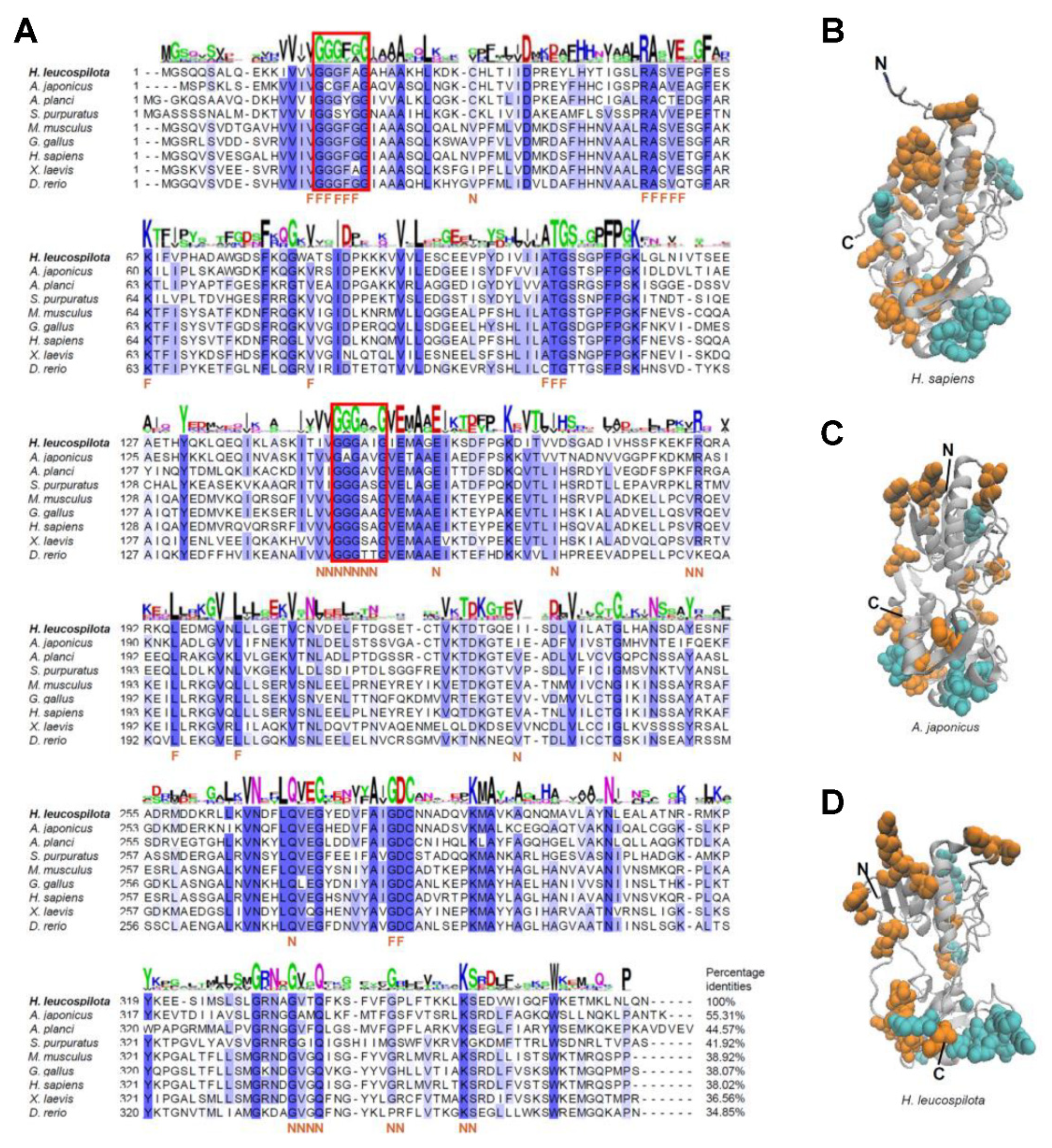
2.2. Phylogenetic, Homology and Structural Analysis
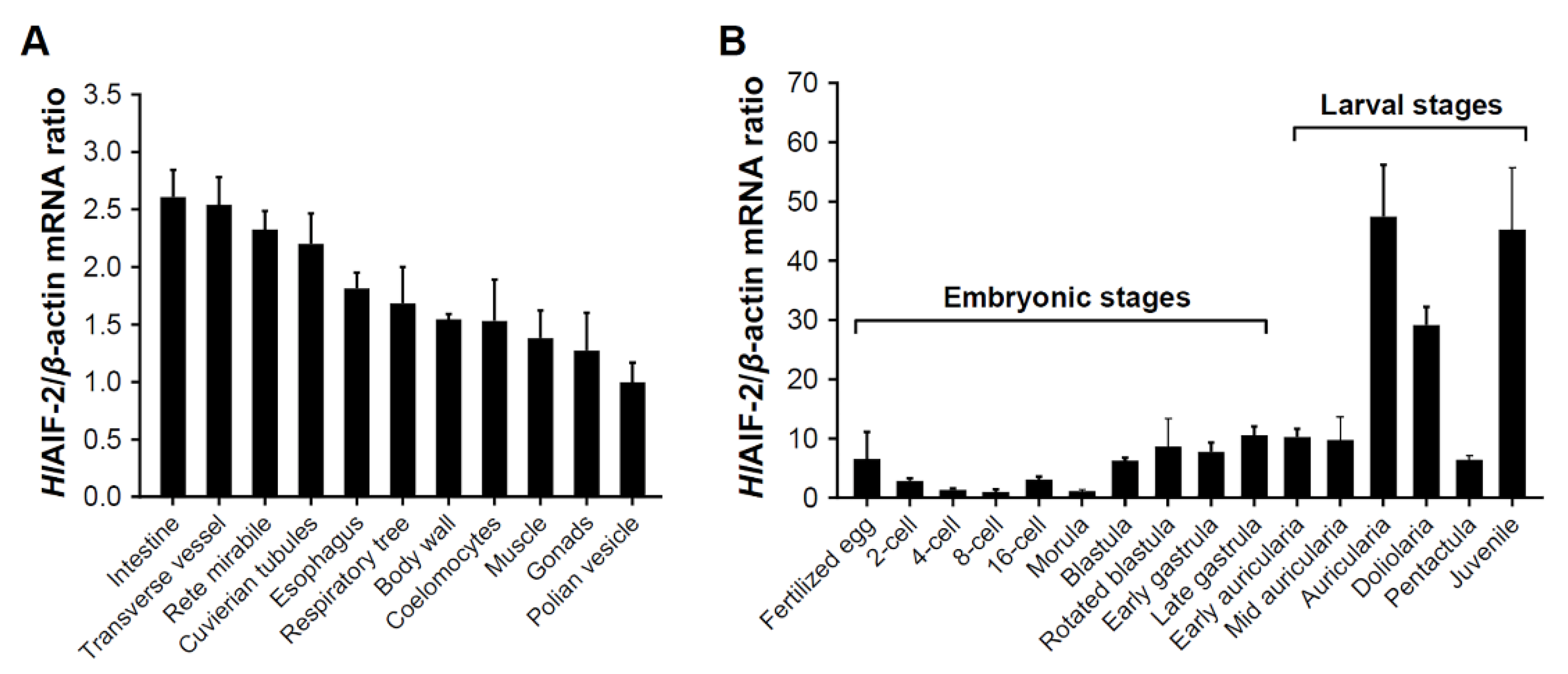
2.3. Expression Patterns of HlAIF-2 among Different Tissues
2.4. Expression Patterns of HlAIF-2 during Embryonic and Larval Development
2.5. HlAIF-2 Expression in Response to Challenges of CdCl2, LPS, and Poly (I:C)
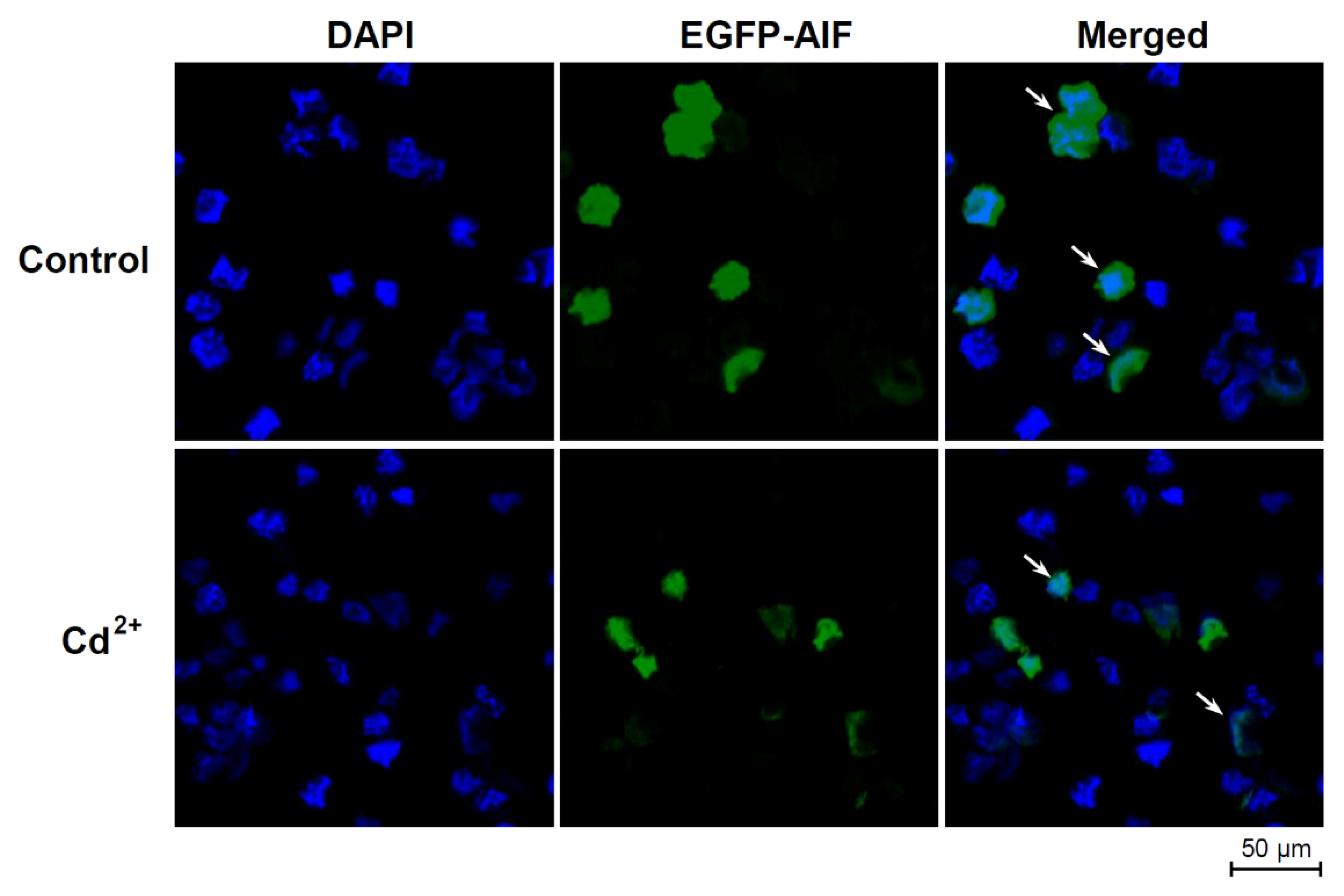
2.6. Subcellular Localization of HlAIF-2 in HEK293T Cells
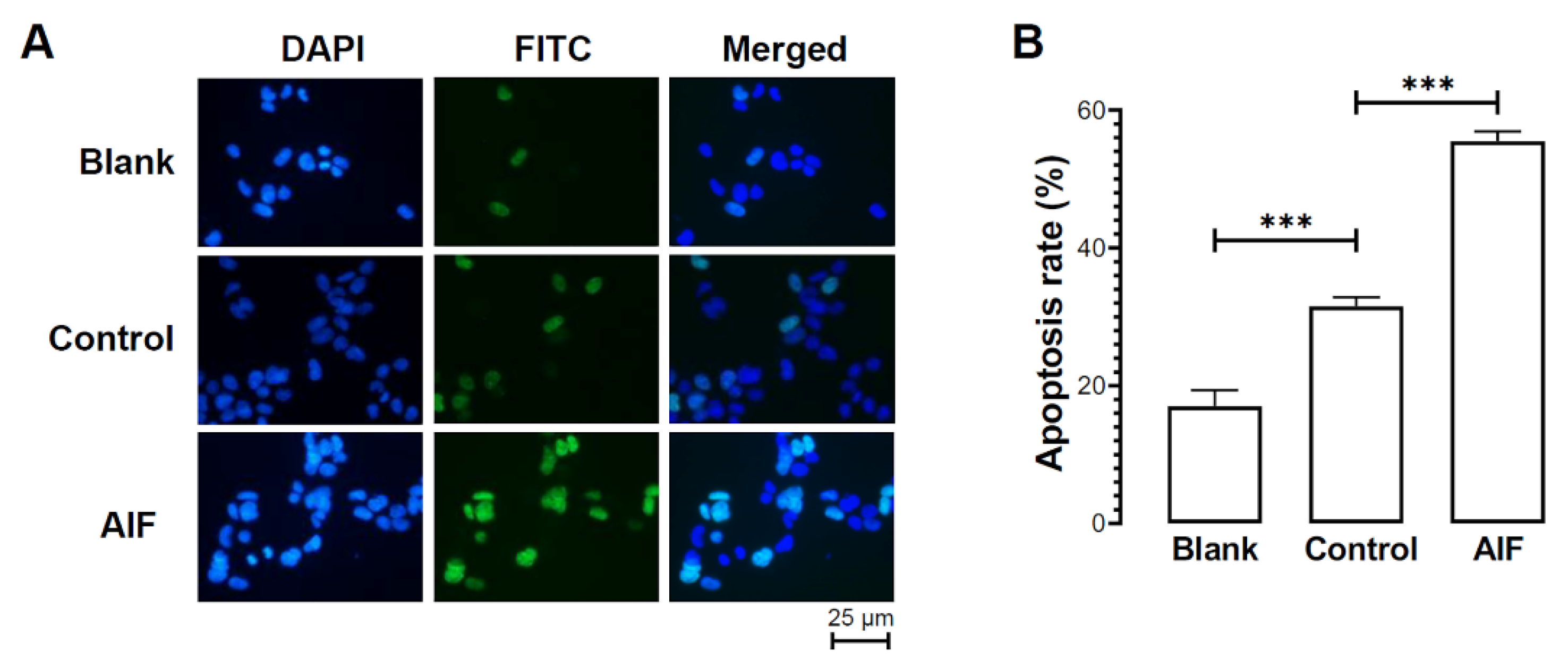
2.7. Effects of HlAIF-2 Overexpression on Cell Apoptosis
3. Discussion
4. Materials and Methods
4.1. Animals and Tissue Collection
4.2. Molecular Cloning of HlAIF-2 Full-Length cDNA
4.3. Bioinformatics Analysis
4.4. Tissue Distribution and Ontogeny of HlAIF-2 mRNA Expression
4.5. Primary Culture and Challenge of Coelomocytes
4.6. Detection of HlAIF-2 Transcript by Real-Time PCR
4.7. Plasmid Construction, Cell Line Culture and Transfection
4.8. Subcellular Localization and Translocation
4.9. Detection of Apoptosis
4.10. Data Transformation and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Danial, N.N.; Korsmeyer, S.J. Cell death: Critical control points. Cell 2004, 116, 205–219. [Google Scholar] [CrossRef] [Green Version]
- Sun, E.W.; Shi, Y.F. Apoptosis: The quiet death silences the immune system. Pharmacol. Ther. 2001, 92, 135–145. [Google Scholar] [CrossRef]
- Kopeina, G.S.; Prokhorova, E.A.; Lavrik, I.N.; Boris, Z. Alterations in the nucleocytoplasmic transport in apoptosis: Caspases lead the way. Cell Prolif. 2018, 51, e12467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Candé, C.; Cohen, I.; Daugas, E.; Ravagnan, L.; Larochette, N.; Zamzami, N.; Kroemer, G. Apoptosis-inducing factor (AIF): A novel caspase-independent death effector released from mitochondria. Biochimie 2002, 84, 215–222. [Google Scholar] [CrossRef]
- Polster, B.M. AIF, reactive oxygen species, and neurodegeneration: A “complex” problem. Neurochem. Int. 2013, 62, 695–702. [Google Scholar] [CrossRef] [Green Version]
- Hangen, E.; Blomgren, K.; Bénit, P.; Kroemer, G.; Modjtahedi, N. Life with or without AIF. Trends Biochem. Sci. 2010, 35, 278–287. [Google Scholar] [CrossRef]
- Klim, J.; Gładki, A.; Kucharczyk, R.; Zielenkiewicz, U.; Kaczanowski, S. Ancestral State Reconstruction of the Apoptosis Machinery in the Common Ancestor of Eukaryotes. G3 Bethesda 2018, 8, 2121–2134. [Google Scholar] [CrossRef] [Green Version]
- Candé, C.; Cecconi, F.; Dessen, P.; Kroemer, G. Apoptosis-inducing factor (AIF): Key to the conserved caspase-independent pathways of cell death? J. Cell Sci. 2002, 115, 4727–4734. [Google Scholar] [CrossRef] [Green Version]
- Mande, S.S.; Sarfaty, S.; Allen, M.D.; Perham, R.N.; Hol, W.G. Protein-protein interactions in the pyruvate dehydrogenase multienzyme complex: Dihydrolipoamide dehydrogenase complexed with the binding domain of dihydrolipoamide acetyltransferase. Structure 1996, 4, 277–286. [Google Scholar] [CrossRef] [Green Version]
- Wu, M.; Xu, L.G.; Li, X.; Zhai, Z.; Shu, H.B. AMID, an apoptosis-inducing factor-homologous mitochondrion-associated protein, induces caspase-independent apoptosis. J. Biol. Chem. 2002, 277, 25617–25623. [Google Scholar] [CrossRef]
- Doll, S.; Freitas, F.P.; Shah, R.; Aldrovandi, M.; da Silva, M.C.; Ingold, I.; Goya Grocin, A.; Xavier da Silva, T.N.; Panzilius, E.; Scheel, C.H.; et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature 2019, 575, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Hay, S.; Marshall, K.R.; Munro, A.W.; Scrutton, N.S. DNA binding suppresses human AIF-M2 activity and provides a connection between redox chemistry, reactive oxygen species, and apoptosis. J. Biol. Chem. 2007, 282, 30331–30340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, S.; Zhao, X.; Xu, H.; Chen, F.; Xu, Y.; Li, Z.; Sanchis, D.; Jin, L.; Zhang, Y.; Ye, J. AKT2 Blocks Nucleus Translocation of Apoptosis-Inducing Factor (AIF) and Endonuclease G (EndoG) While Promoting Caspase Activation during Cardiac Ischemia. Int. J. Mol. Sci. 2017, 18, 565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cregan, S.P.; Dawson, V.L.; Slack, R.S. Role of AIF in caspase-dependent and caspase-independent cell death. Oncogene 2004, 23, 2785–2796. [Google Scholar] [CrossRef] [Green Version]
- Liu, G.; Zou, H.; Luo, T.; Long, M.; Bian, J.; Liu, X.; Gu, J.; Yuan, Y.; Song, R.; Wang, Y.; et al. Caspase-Dependent and Caspase-Independent Pathways Are Involved in Cadmium-Induced Apoptosis in Primary Rat Proximal Tubular Cell Culture. PLoS ONE 2016, 11, e0166823. [Google Scholar] [CrossRef] [Green Version]
- Chang, Z.; Yang, M.; Ji, H. Molecular characterization and functional analysis of apoptosis-inducing factor (AIF) in palmitic acid-induced apoptosis in Ctenopharyngodon idellus kidney (CIK) cells. Fish Physiol. Biochem. 2021, 47, 213–224. [Google Scholar] [CrossRef]
- Gong, Y.; Kong, T.; Ren, X.; Chen, J.; Lin, S.; Zhang, Y.; Li, S. Exosome-mediated apoptosis pathway during WSSV infection in crustacean mud crab. PLoS Pathog. 2020, 16, e1008366. [Google Scholar] [CrossRef]
- Hu, W.Y.; Yao, C.L. Molecular and immune response characterizations of a novel AIF and cytochrome c in Litopenaeus vannamei defending against WSSV infection. Fish Shellfish Immunol. 2016, 56, 84–95. [Google Scholar] [CrossRef]
- Wang, S.; Li, X.; Chen, M.; Storey, K.B.; Wang, T. A potential antiapoptotic regulation: The interaction of heat shock protein 70 and apoptosis-inducing factor mitochondrial 1 during heat stress and aestivation in sea cucumber. J. Exp. Zool A Ecol. Integr. Physiol. 2018, 329, 103–111. [Google Scholar] [CrossRef]
- Collard, M.; Eeckhaut, I.; Dehairs, F.; Dubois, P. Acid-base physiology response to ocean acidification of two ecologically and economically important holothuroids from contrasting habitats, Holothuria scabra and Holothuria parva. Environ. Sci. Pollut. Res. Int. 2014, 21, 13602–13614. [Google Scholar] [CrossRef]
- Huang, W.; Huo, D.; Yu, Z.; Ren, C.; Jiang, X.; Luo, P.; Chen, T.; Hu, C. Spawning, larval development and juvenile growth of the tropical sea cucumber Holothuria leucospilota. Aquaculture 2018, 488, 22–29. [Google Scholar] [CrossRef]
- Li, H.; Chen, T.; Sun, H.; Wu, X.; Jiang, X.; Ren, C. The first cloned echinoderm tumor necrosis factor receptor from Holothuria leucospilota: Molecular characterization and functional analysis. Fish Shellfish Immunol. 2019, 93, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.; Ren, C.; Chen, T.; Jiang, X.; Sun, H.; Huo, D.; Hu, C.; Wen, J. The first tropical sea cucumber caspase-8 from Holothuria leucospilota: Molecular characterization, involvement of apoptosis and inducible expression by immune challenge. Fish Shellfish Immunol. 2018, 72, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.; Ren, C.; Chen, T.; Huo, D.; Jiang, X.; Sun, H.; Hu, C. A novel caspase-6 from sea cucumber Holothuria leucospilota: Molecular characterization, expression analysis and apoptosis detection. Fish Shellfish Immunol. 2018, 80, 232–240. [Google Scholar] [CrossRef]
- Zhao, L.; Ren, C.; Chen, T.; Sun, H.; Wu, X.; Jiang, X.; Huang, W. The first cloned sea cucumber FADD from Holothuria leucospilota: Molecular characterization, inducible expression and involvement of apoptosis. Fish Shellfish Immunol. 2019, 89, 548–554. [Google Scholar] [CrossRef]
- Zhao, L.; Jiang, X.; Chen, T.; Sun, H.; Ren, C. Molecular characterization and functional analysis of MyD88 from the tropical sea cucumber, Holothuria leucospilota. Fish Shellfish Immunol. 2018, 83, 1–7. [Google Scholar] [CrossRef]
- Li, H.; Wu, X.; Chen, T.; Jiang, X.; Ren, C. Molecular characterization, inducible expression and functional analysis of an IKKbeta from the tropical sea cucumber Holothuria leucospilota. Fish Shellfish Immunol. 2020, 104, 622–632. [Google Scholar] [CrossRef]
- Li, H.; Chen, T.; Sun, H.; Wu, X.; Jiang, X.; Ren, C. Functional characterisation of Holothuria leucospilota Fas-associated death domain in the innate immune-related signalling pathways. Innate Immun. 2020, 26, 138–145. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen Recognition and Innate Immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef] [Green Version]
- Okereafor, U.; Makhatha, M.; Mekuto, L.; Uche-Okereafor, N.; Sebola, T.; Mavumengwana, V. Toxic metal implications on agricultural soils, plants, animals, aquatic life and human Health. Int. J. Environ. Res. Public Health 2020, 17, 2204. [Google Scholar] [CrossRef] [Green Version]
- Qiao, X.; Hou, L.; Wang, J.; Jin, Y.; Kong, N.; Li, J.; Wang, S.; Wang, L.; Song, L. Identification and characterization of an apoptosis-inducing factor 1 involved in apoptosis and immune defense of oyster, Crassostrea gigas. Fish Shellfish Immunol. 2021, 119, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Lin, T.; Zhang, Y.; Zheng, J.; Bonanno, J.A. Molecular cloning and characterization of a human AIF-like gene with ability to induce apoptosis. J. Biol. Chem. 2005, 280, 19673–19681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srivaths, A.; Ramanathan, S.; Sakthivel, S.; Habeeb, S. Insights from the Molecular Modelling and Docking Analysis of AIF-NLS complex to infer Nuclear Translocation of the Protein. Bioinformation 2018, 14, 132–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.; Soh, J. Cadmium-induced apoptosis is mediated by the translocation of AIF to the nucleus in rat testes. Toxicol. Lett. 2009, 188, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Miramar, M.D.; Costantini, P.; Ravagnan, L.; Saraiva, L.M.; Haouzi, D.; Brothers, G.; Penninger, J.M.; Peleato, M.L.; Kroemer, G.; Susin, S.A. NADH Oxidase Activity of Mitochondrial Apoptosis-inducing Factor. J. Biol. Chem. 2001, 276, 16391–16398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daugas, E.; Nochy, D.; Ravagnan, L.; Loeffler, M.; Susin, S.A.; Zamzami, N.; Kroemer, G. Apoptosis-inducing factor (AIF): A ubiquitous mitochondrial oxidoreductase involved in apoptosis. FEBS Lett. 2000, 476, 118–123. [Google Scholar] [CrossRef]
- Hangen, E.; De Zio, D.; Bordi, M.; Zhu, C.; Dessen, P.; Caffin, F.; Lachkar, S.; Perfettini, J.L.; Lazar, V.; Benard, J.; et al. A brain-specific isoform of mitochondrial apoptosis-inducing factor: AIF2. Cell Death Differ. 2010, 17, 1155–1166. [Google Scholar] [CrossRef]
- Sanders, E.J.; Parker, E. Expression of apoptosis-inducing factor during early neural differentiation in the chick embryo. Histochem. J. 2002, 34, 161–166. [Google Scholar] [CrossRef]
- Liao, Y. Fauna Sincia: Phylum Echinodermata Class. Holothuroidea; Science Press: Beijing, China, 1997. [Google Scholar]
- Nakano, H.; Murabe, N.; Amemiya, S.; Nakajima, Y. Nervous system development of the sea cucumber Stichopus japonicus. Dev. Biol. 2006, 292, 205–212. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Ruan, Y.; Chen, T.; Yu, Z.; Huo, D.; Li, X.; Wu, F.; Jiang, X.; Ren, C. First echinoderm alpha-amylase from a tropical sea cucumber (Holothuria leucospilota): Molecular cloning, tissue distribution, cellular localization and functional production in a heterogenous E.coli system with codon optimization. PLoS ONE 2020, 15, e0239044. [Google Scholar] [CrossRef]
- Huo, D.; Jiang, X.; Wu, X.; Ren, C.; Yu, Z.; Liu, J.; Li, H.; Ruan, Y.; Wen, J.; Chen, T.; et al. First echinoderm trehalase from a tropical sea cucumber (Holothuria leucospilota): Molecular cloning and mRNA expression in different tissues, embryonic and larval stages, and under a starvation challenge. Gene 2018, 665, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Wu, F.; Ren, C.; Jiang, X.; Zhang, X.; Li, X.; Luo, P.; Hu, C.; Chen, T. Aquaculture of the tropical sea cucumber, Stichopus monotuberculatus: Induced spawning, detailed records of gonadal and embryonic development, and improvements in larval breeding by digestive enzyme supply in diet. Aquaculture 2021, 540, 736690. [Google Scholar] [CrossRef]
- Kannan, K.; Jain, S.K. Oxidative stress and apoptosis. Pathophysiology 2000, 7, 153–163. [Google Scholar] [CrossRef]
- Candé, C.; Vahsen, N.; Garrido, C.; Kroemer, G. Apoptosis-inducing factor (AIF): Caspase-independent after all. Cell Death Differ. 2004, 11, 591–595. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, L.; Qu, T.; Tang, X.; Li, L.; Zhang, G. Conservation and divergence of mitochondrial apoptosis pathway in the Pacific oyster, Crassostrea gigas. Cell Death Dis. 2017, 8, e2915. [Google Scholar] [CrossRef] [Green Version]
- Vařecha, M.; Amrichová, J.; Zimmermann, M.; Ulman, V.; Lukášová, E.; Kozubek, M. Bioinformatic and image analyses of the cellular localization of the apoptotic proteins endonuclease G, AIF, and AMID during apoptosis in human cells. Apoptosis 2007, 12, 1155–1171. [Google Scholar] [CrossRef]
- Bersuker, K.; Hendricks, J.M.; Li, Z.; Magtanong, L.; Ford, B.; Tang, P.H.; Roberts, M.A.; Tong, B.; Maimone, T.J.; Zoncu, R.; et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 2019, 575, 688–692. [Google Scholar] [CrossRef]
- Wu, X.F.; Chen, T.; Huo, D.; Yu, Z.H.; Ruan, Y.; Cheng, C.H.; Jiang, X.; Ren, C.H. Transcriptomic analysis of sea cucumber (Holothuria leucospilota) coelomocytes revealed the echinoderm cytokine response during immune challenge. BMC Genom. 2020, 21, 306. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Chen, T.; Wu, X.; Jiang, X.; Luo, P.; E, Z.; Hu, C.; Ren, C. Apoptosis-Inducing Factor 2 (AIF-2) Mediates a Caspase-Independent Apoptotic Pathway in the Tropical Sea Cucumber (Holothuria leucospilota). Int. J. Mol. Sci. 2022, 23, 3008. https://doi.org/10.3390/ijms23063008
Li X, Chen T, Wu X, Jiang X, Luo P, E Z, Hu C, Ren C. Apoptosis-Inducing Factor 2 (AIF-2) Mediates a Caspase-Independent Apoptotic Pathway in the Tropical Sea Cucumber (Holothuria leucospilota). International Journal of Molecular Sciences. 2022; 23(6):3008. https://doi.org/10.3390/ijms23063008
Chicago/Turabian StyleLi, Xiaomin, Ting Chen, Xiaofen Wu, Xiao Jiang, Peng Luo, Zixuan E, Chaoqun Hu, and Chunhua Ren. 2022. "Apoptosis-Inducing Factor 2 (AIF-2) Mediates a Caspase-Independent Apoptotic Pathway in the Tropical Sea Cucumber (Holothuria leucospilota)" International Journal of Molecular Sciences 23, no. 6: 3008. https://doi.org/10.3390/ijms23063008
APA StyleLi, X., Chen, T., Wu, X., Jiang, X., Luo, P., E, Z., Hu, C., & Ren, C. (2022). Apoptosis-Inducing Factor 2 (AIF-2) Mediates a Caspase-Independent Apoptotic Pathway in the Tropical Sea Cucumber (Holothuria leucospilota). International Journal of Molecular Sciences, 23(6), 3008. https://doi.org/10.3390/ijms23063008






