Heat Shock Proteins Alterations in Rheumatoid Arthritis
Abstract
1. Introduction
2. Pathogenesis of Rheumatoid Arthritis
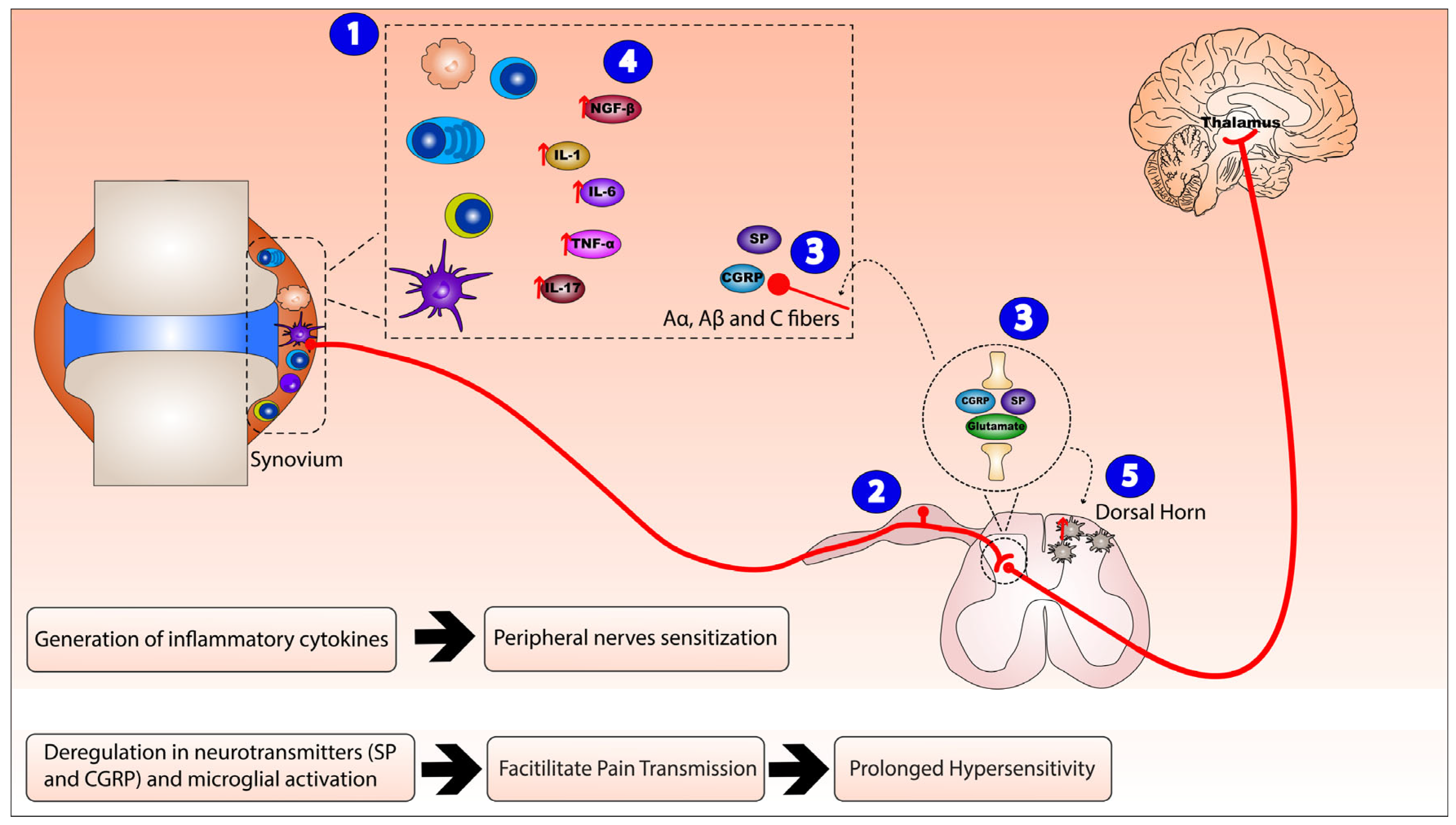
3. Neurogenic Inflammation and Rheumatoid Arthritis
4. Rheumatoid Arthritis and a Specific Pool of Immunogenic Proteins
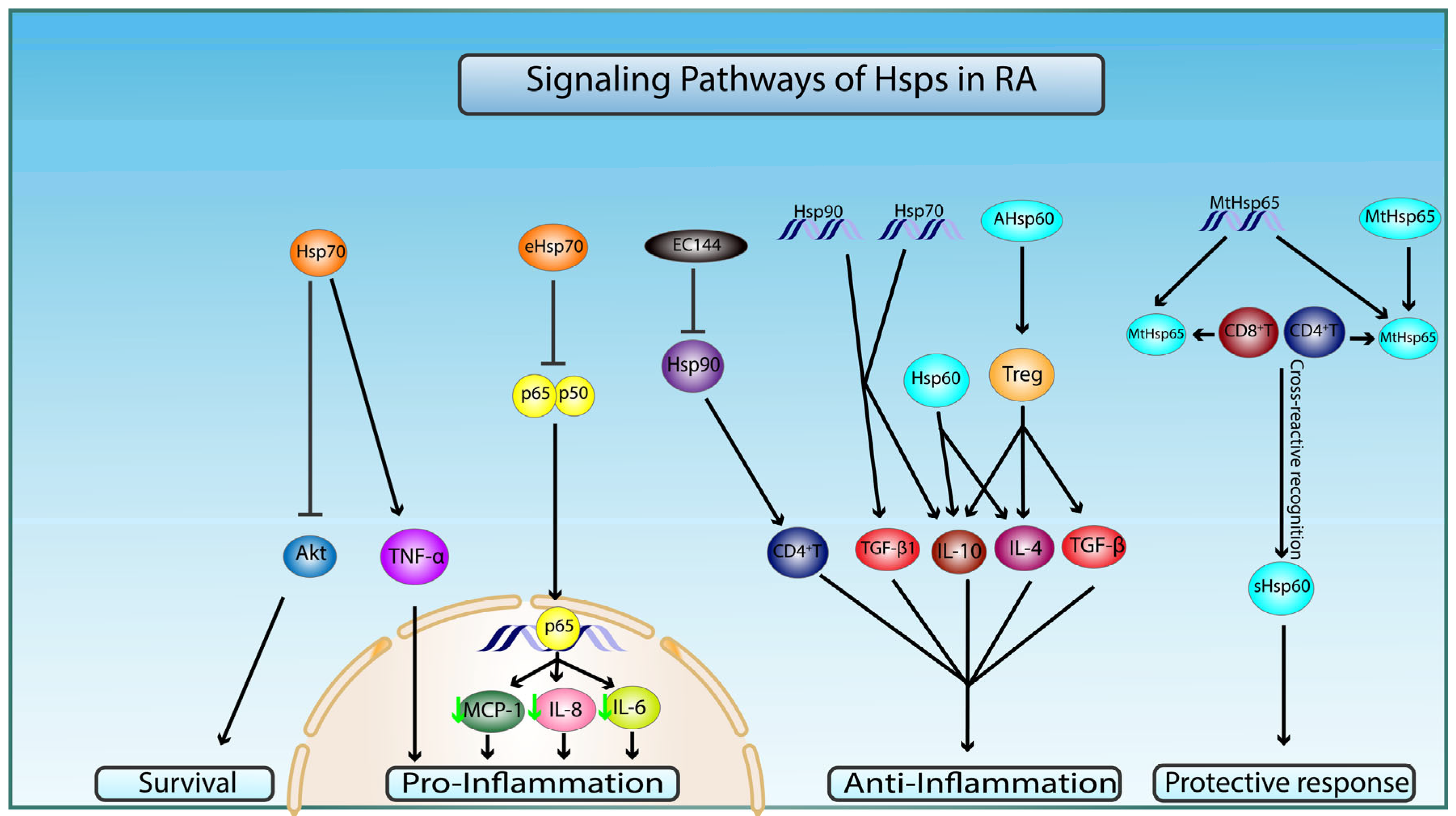
5. Rheumatoid Arthritis and Hsp27
6. Rheumatoid Arthritis and Hsp60
7. Rheumatoid Arthritis and Hsp70
8. Rheumatoid Arthritis and Hsp90
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Smith, H.R. What is the global prevalence of rheumatoid arthritis (RA) among different age groups and ethnicities? Medscape 2020, 1, 136. [Google Scholar]
- Lin, Y.J.; Anzaghe, M.; Schülke, S. Update on the Pathomechanism, Diagnosis, and Treatment Options for Rheumatoid Arthritis. Cells 2020, 9, 880. [Google Scholar] [CrossRef]
- Muravyev, Y.V. Extra-articular manifestations of rheumatoid arthritis. Nauchno-Prakticheskaya Revmatol. 2018, 56, 356–362. [Google Scholar] [CrossRef]
- Otero, M.; Goldring, M.B. Cells of the synovium in rheumatoid arthritis. Chondrocytes. Arthritis Res. Ther. 2007, 9, 220. [Google Scholar] [CrossRef] [PubMed]
- Choy, E. Understanding the dynamics: Pathways involved in the pathogenesis of rheumatoid arthritis. Rheumatology 2012, 51 (Suppl. 5), 113. [Google Scholar] [CrossRef]
- Yap, H.Y.; Tee, S.Z.Y.; Wong, M.M.T.; Chow, S.K.; Peh, S.C.; Teow, S.Y. Pathogenic Role of Immune Cells in Rheumatoid Arthritis: Implications in Clinical Treatment and Biomarker Development. Cells 2018, 7, 161. [Google Scholar] [CrossRef]
- Van der Kleij, H.P.M.; Forsythe, P.; Bienenstock, J. Autonomic Neuroimmunology. In Encyclopedia of Neuroscience; Elsevier Ltd.: Hamilton, ON, Canada, 2009; pp. 1003–1008. ISBN 9780080450469. [Google Scholar]
- Louis, S.M.; Jamieson, A.; Russell, N.J.W.; Dockray, G.J. The role of substance P and calcitonin gene-related peptide in neurogenic plasma extravasation and vasodilatation in the rat. Neuroscience 1989, 32, 581–586. [Google Scholar] [CrossRef]
- Ren, K.; Dubner, R. Interactions between the immune and nervous systems in pain. Nat. Med. 2010, 16, 1267–1276. [Google Scholar] [CrossRef]
- Bolon, B. Cellular and Molecular Mechanisms of Autoimmune Disease. Toxicol. Pathol. 2012, 40, 216–229. [Google Scholar] [CrossRef]
- Bugatti, S.; Vitolo, B.; Caporali, R.; Montecucco, C.; Manzo, A. B cells in rheumatoid arthritis: From pathogenic players to disease biomarkers. Biomed Res. Int. 2014, 2014, 1678. [Google Scholar] [CrossRef]
- Van Lent, P.L.E.M.; Van den Berg, W.B. Macrophages in rheumatoid arthritis. In Contemporary Targeted Therapies in Rheumatology; CRC Press: Boca Raton, FL, USA, 2007; pp. 45–61. ISBN 9780203694145. [Google Scholar]
- Lee, D.S.W.; Rojas, O.L.; Gommerman, J.L. B cell depletion therapies in autoimmune disease: Advances and mechanistic insights. Nat. Rev. Drug Discov. 2021, 20, 179–199. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.C.; Ssu, C.T.; Tsao, Y.P.; Liou, T.L.; Tsai, C.Y.; Chou, C.T.; Chen, M.H.; Leu, C.M. Cytotoxic T lymphocyte-associated antigen-4-Ig (CTLA-4-Ig) suppresses Staphylococcus aureus-induced CD80, CD86, and pro-inflammatory cytokine expression in human B cells. Arthritis Res. Ther. 2020, 22, 64. [Google Scholar] [CrossRef]
- Santha, M.; Durham, H.D.; Vigh, L.; Prodromou, C. Editorial: The Role of Heat Shock Proteins in Neuroprotection. Front. Pharmacol. 2020, 11, 1227. [Google Scholar] [CrossRef] [PubMed]
- Dukay, B.; Csoboz, B.; Tóth, M.E. Heat-shock proteins in neuroinflammation. Front. Pharmacol. 2019, 10, 920. [Google Scholar] [CrossRef] [PubMed]
- Scieglinska, D.; Krawczyk, Z.; Sojka, D.R.; Gogler-Pigłowska, A. Heat shock proteins in the physiology and pathophysiology of epidermal keratinocytes. Cell Stress Chaperones 2019, 24, 1027–1044. [Google Scholar] [CrossRef]
- Basset, C.A.; Rappa, F.; Lentini, V.L.; Barone, R.; Pitruzzella, A.; Unti, E.; Cappello, F.; Conway de Macario, E.; Macario, A.J.L.; Leone, A. Hsp27 and Hsp60 in human submandibular salivary gland: Quantitative patterns in healthy and cancerous tissues with potential implications for differential diagnosis and carcinogenesis. Acta Histochem. 2021, 123, 151771. [Google Scholar] [CrossRef]
- Li Volti, G. The Chaperonopathies-Diseases with Defective Molecular Chaperones. Eur. J. Histochem. 2013, 57, 15. [Google Scholar] [CrossRef]
- Kampinga, H.H.; Hageman, J.; Vos, M.J.; Kubota, H.; Tanguay, R.M.; Bruford, E.A.; Cheetham, M.E.; Chen, B.; Hightower, L.E. Guidelines for the nomenclature of the human heat shock proteins. Cell Stress Chaperones 2009, 14, 105–111. [Google Scholar] [CrossRef]
- Basset, C.A.; Cappello, F.; Rappa, F.; Lentini, V.L.; Jurjus, A.R.; Conway de Macario, E.; Macario, A.J.L.; Leone, A. Molecular chaperones in tumors of salivary glands. J. Mol. Histol. 2020, 51, 109–115. [Google Scholar] [CrossRef]
- Basset, C.A.; Cappello, F.; Rappa, F.; Jurjus, A.R.; Conway de Macario, E.; Macario, A.J.L.; Leone, A. Chaperonin Hsp60 and Cancer Therapies. In Heat Shock Proteins in Human Diseases; Springer: Cham, Switzerland, 2020; pp. 31–52. [Google Scholar]
- Spierings, J.; van Eden, W. Heat shock proteins and their immunomodulatory role in inflammatory arthritis. Rheumatol. 2017, 56, 198–208. [Google Scholar] [CrossRef]
- Vidyasagar, A.; Wilson, N.A.; Djamali, A. Heat shock protein 27 (HSP27): Biomarker of disease and therapeutic target. Fibrogenes. Tissue Repair 2012, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sheth, K.; De, A.; Nolan, B.; Friel, J.; Duffy, A.; Ricciardi, R.; Miller-Graziano, C.; Bankey, P. Heat shock protein 27 inhibits apoptosis in human neutrophils. J. Surg. Res. 2001, 99, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Bruinsma, I.B.; de Jager, M.; Carrano, A.; Versleijen, A.A.M.; Veerhuis, R.; Boelens, W.; Rozemuller, A.J.M.; de Waal, R.M.W.; Verbeek, M.M. Small heat shock proteins induce a cerebral inflammatory reaction. J. Neurosci. 2011, 31, 11992–12000. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Alford, K.A.; Glennie, S.; Turrell, B.R.; Rawlinson, L.; Saklatvala, J.; Dean, J.L.E. Heat shock protein 27 functions in inflammatory gene expression and transforming growth factor-β-activated kinase-1 (TAK1)-mediated signaling. J. Biol. Chem. 2007, 282, 6232–6241. [Google Scholar] [CrossRef]
- Sedlackova, L.; Sosna, A.; Vavrincova, P.; Frýdl, J.; Guerriero, V.; Raynes, D.A.; Hromadnikova, I. Heat shock protein gene expression profile may differentiate between rheumatoid arthritis, osteoarthritis, and healthy controls. Scand. J. Rheumatol. 2011, 40, 354–357. [Google Scholar] [CrossRef]
- Batulan, Z.; Pulakazhi Venu, V.K.; Li, Y.; Koumbadinga, G.; Alvarez-Olmedo, D.G.; Shi, C.; O’Brien, E.R. Extracellular release and signaling by heat shock protein 27: Role in modifying vascular inflammation. Front. Immunol. 2016, 7, 285. [Google Scholar] [CrossRef]
- Laudanski, K.; De, A.; Miller-Graziano, C. Exogenous heat shock protein 27 uniquely blocks differentiation of monocytes to dendritic cells. Eur. J. Immunol. 2007, 37, 2812–2824. [Google Scholar] [CrossRef]
- Fouani, M.; Basset, C.A.; Jurjus, A.R.; Leone, L.G.; Tomasello, G.; Leone, A. Salivary gland proteins alterations in the diabetic milieu. J. Mol. Histol. 2021, 52, 893–904. [Google Scholar] [CrossRef]
- Grundtman, C.; Kreutmayer, S.B.; Almanzar, G.; Wick, M.C.; Wick, G. Heat shock protein 60 and immune inflammatory responses in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 960–968. [Google Scholar] [CrossRef]
- Coelho, V.; Faria, A.M.C. HSP60: Issues and insights on its therapeutic use as an immunoregulatory agent. Front. Immunol. 2012, 2, 97. [Google Scholar] [CrossRef][Green Version]
- Quintana, F.J.; Cohen, I.R. The HSP60 immune system network. Trends Immunol. 2011, 32, 89–95. [Google Scholar] [CrossRef]
- Zanin-Zhorov, A.; Cahalon, L.; Tal, G.; Margalit, R.; Lider, O.; Cohen, I.R. Heat shock protein 60 enhances CD4+ CD25+ regulatory T cell function via innate TLR2 signaling. J. Clin. Invest. 2006, 116, 2022–2032. [Google Scholar] [CrossRef] [PubMed]
- Swaroop, S.; Sengupta, N.; Suryawanshi, A.R.; Adlakha, Y.K.; Basu, A. HSP60 plays a regulatory role in IL-1β-induced microglial inflammation via TLR4-p38 MAPK axis. J. Neuroinflammation 2016, 13, 486. [Google Scholar] [CrossRef] [PubMed]
- Liyanagamage, D.S.N.K.; Martinus, R.D. Role of Mitochondrial Stress Protein HSP60 in Diabetes-Induced Neuroinflammation. Mediators Inflamm. 2020, 7, 3516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Sun, L.; Zhu, H.; Wang, L.; Wu, W.; Xie, J.; Gu, J. Microglial LOX-1 reacts with extracellular HSP60 to bridge neuroinflammation and neurotoxicity. Neurochem. Int. 2012, 61, 1021–1035. [Google Scholar] [CrossRef]
- Juwono, J.; Martinus, R.D. Does Hsp60 Provide a Link between Mitochondrial Stress and Inflammation in Diabetes Mellitus? J. Diabetes Res. 2016, 1, 7571. [Google Scholar] [CrossRef]
- Märker, T.; Sell, H.; Zilleßen, P.; Glöde, A.; Kriebel, J.; Margriet Ouwens, D.; Pattyn, P.; Ruige, J.; Famulla, S.; Roden, M.; et al. Heat shock protein 60 as a mediator of adipose tissue inflammation and insulin resistance. Diabetes 2012, 61, 615–625. [Google Scholar] [CrossRef]
- De Launay, D.; Van De Sande, M.G.H.; De Hair, M.J.H.; Grabiec, A.M.; Van De Sande, G.P.M.; Lehmann, K.A.; Wijbrandts, C.A.; Van Baarsen, L.G.M.; Gerlag, D.M.; Tak, P.P.; et al. Selective involvement of ERK and JNK mitogen-activated protein kinases in early rheumatoid arthritis (1987 ACR criteria compared to 2010 ACR/EULAR criteria): A prospective study aimed at identification of diagnostic and prognostic biomarkers as well as th. Ann. Rheum. Dis. 2012, 71, 415–423. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Kamalakannan, V.; Kirthika, S.; Haripriya, K.; Babu, S.; Narayanan, R.B. Wolbachia heat shock protein 60 induces pro-inflammatory cytokines and apoptosis in monocytes in vitro. Microbes Infect. 2012, 14, 610–618. [Google Scholar] [CrossRef]
- Anderton, S.M.; van der Zee, R.; Noordzij, A.; van Eden, W. Differential mycobacterial 65-kDa heat shock protein T cell epitope recognition after adjuvant arthritis-inducing or protective immunization protocols. J. Immunol. 1994, 152, 3656–3664. [Google Scholar] [PubMed]
- Ragno, S.; Colston, M.J.; Lowrie, D.B.; Winrow, V.R.; Blake, D.R.; Tascon, R. Protection of rats from adjuvant arthritis by immunization with naked DNA encoding for mycobacterial heat shock protein 65. Arthritis Rheum. 1997, 40, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Vercoulen, Y.; van Teijlingen, N.H.; de Kleer, I.M.; Kamphuis, S.; Albani, S.; Prakken, B.J. Heat shock protein 60 reactive T cells in juvenile idiopathic arthritis: What is new? Arthritis Res. Ther. 2009, 11, 231. [Google Scholar] [CrossRef] [PubMed]
- Prakken, B.J.; Roord, S.; Van Kooten, P.J.S.; Wagenaar, J.P.A.; Van Eden, W.; Albani, S.; Wauben, M.H.M. Inhibition of adjuvant-induced arthritis by interleukin-10-driven regulatory cells induced via nasal administration of a peptide analog of an arthritis-related heat-shock protein 60 T cell epitope. Arthritis Rheum. 2002, 46, 1937–1946. [Google Scholar] [CrossRef]
- Sharma, D.; Masison, D. Hsp70 Structure, Function, Regulation and Influence on Yeast Prions. Protein Pept. Lett. 2009, 16, 571–581. [Google Scholar] [CrossRef]
- Zylicz, M.; Wawrzynow, A. Insights into the function of Hsp70 chaperones. IUBMB Life 2001, 51, 283–287. [Google Scholar]
- Tukaj, S.; Mantej, J.; Sobala, M.; Potrykus, K.; Sitko, K. Autologous extracellular Hsp70 exerts a dual role in rheumatoid arthritis. Cell Stress Chaperones 2020, 25, 1105–1110. [Google Scholar] [CrossRef]
- Kang, E.H.; Kim, D.J.; Lee, E.Y.; Lee, Y.J.; Lee, E.B.; Song, Y.W. Downregulation of heat shock protein 70 protects rheumatoid arthritis fibroblast-like synoviocytes from nitric oxide-induced apoptosis. Arthritis Res. Ther. 2009, 11, 2797. [Google Scholar] [CrossRef]
- Mantej, J.; Polasik, K.; Piotrowska, E.; Tukaj, S. Autoantibodies to heat shock proteins 60, 70, and 90 in patients with rheumatoid arthritis. Cell Stress Chaperones 2019, 24, 283–287. [Google Scholar] [CrossRef]
- Quintana, F.J.; Carmi, P.; Mor, F.; Cohen, I.R. Inhibition of adjuvant-induced arthritis by DNA vaccination with the 70-kd or the 90-kd human heat-shock protein: Immune cross-regulation with the 60-kd heat-shock protein. Arthritis Rheum. 2004, 50, 3712–3720. [Google Scholar] [CrossRef]
- Kakimura, J.-I.; Kitamura, Y.; Takata, K.; Umeki, M.; Suzuki, S.; Shibagaki, K.; Taniguchi, T.; Nomura, Y.; Gebicke-Haerter, P.J.; Smith, M.A.; et al. Microglial activation and amyloid-β clearance induced by exogenous heat-shock proteins. FASEB J. 2002, 16, 601–603. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Zuo, X.; Zhou, Y.; Zhang, B.; Shi, Y.; Liu, M.; Wang, K.; McMillian, D.R.; Xiao, X. Extracellular heat shock protein 70 inhibits tumour necrosis factor-α induced proinflammatory mediator production in fibroblast-like synoviocytes. Arthritis Res. Ther. 2008, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, B.; Chaabo, K.; Hall, C.; Garrood, T.; Mant, T.; Allen, E.; Vincent, A.; Vasconcelos, J.C.; Prevost, A.T.; Panayi, G.S.; et al. Safety and patient response as indicated by biomarker changes to binding immunoglobulin protein in the phaseI/IIA RAGULA clinical trial in rheumatoid arthritis. Rheumatology 2016, 55, 1993–2000. [Google Scholar] [CrossRef] [PubMed]
- Van Eden, W.; Van Der Zee, R.; Prakken, B. Heat-shock proteins induce T-cell regulation of chronic inflammation. Nat. Rev. Immunol. 2005, 5, 318–330. [Google Scholar] [CrossRef]
- Caldas-Lopes, E.; Cerchietti, L.; Ahn, J.H.; Clement, C.C.; Robles, A.I.; Rodina, A.; Moulick, K.; Taldone, T.; Gozrnan, A.; Guo, Y.; et al. Hsp90 inhibitor PU-H71, a multimodal inhibitor of malignancy, induces complete responses in triple-negative breast cancer models. Proc. Natl. Acad. Sci. USA 2009, 106, 8368–8373. [Google Scholar] [CrossRef]
- Van Eden, W.; Jansen, M.A.A.; Ludwig, I.; Van Kooten, P.; Van der Zee, R.; Broere, F. The Enigma of Heat Shock Proteins in Immune Tolerance. Front. Immunol 2017, 8, 1599. [Google Scholar] [CrossRef]
- Tukaj, S.; Węgrzyn, G. Anti-Hsp90 therapy in autoimmune and inflammatory diseases: A review of preclinical studies. Cell Stress Chaperones 2016, 21, 213–218. [Google Scholar] [CrossRef]
- Rice, J.W.; Veal, J.M.; Fadden, R.P.; Barabasz, A.F.; Partridge, J.M.; Barta, T.E.; Dubois, L.G.; Huang, K.H.; Mabbett, S.R.; Silinski, M.A.; et al. Small molecule inhibitors of Hsp90 potently affect inflammatory disease pathways and exhibit activity in models of rheumatoid arthritis. Arthritis Rheum. 2008, 58, 3765–3775. [Google Scholar] [CrossRef]
- Yun, T.; Harning, E.; Giza, K.; Rabah, D.; Li, P.; Arndt, J.; Luchetti, D.; Biamonte, M.; Shi, J.; Lundgren, K.; et al. Inflammation and Autoimmunity Immune Responses in Models of Protein 90, Blocks Innate and Adaptive EC144, a Synthetic Inhibitor of Heat Shock. J. Immunol. 2011, 186, 563–575. [Google Scholar] [CrossRef]
- Liu, Z.; Li, X.; Qiu, L.; Zhang, X.; Chen, L.; Cao, S.; Wang, F.; Meng, S. Treg suppress CTL responses upon immunization with HSP gp96. Eur. J. Immunol. 2009, 39, 3110–3120. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fouani, M.; Basset, C.A.; Mangano, G.D.; Leone, L.G.; Lawand, N.B.; Leone, A.; Barone, R. Heat Shock Proteins Alterations in Rheumatoid Arthritis. Int. J. Mol. Sci. 2022, 23, 2806. https://doi.org/10.3390/ijms23052806
Fouani M, Basset CA, Mangano GD, Leone LG, Lawand NB, Leone A, Barone R. Heat Shock Proteins Alterations in Rheumatoid Arthritis. International Journal of Molecular Sciences. 2022; 23(5):2806. https://doi.org/10.3390/ijms23052806
Chicago/Turabian StyleFouani, Malak, Charbel A. Basset, Giuseppe D. Mangano, Lavinia G. Leone, Nada B. Lawand, Angelo Leone, and Rosario Barone. 2022. "Heat Shock Proteins Alterations in Rheumatoid Arthritis" International Journal of Molecular Sciences 23, no. 5: 2806. https://doi.org/10.3390/ijms23052806
APA StyleFouani, M., Basset, C. A., Mangano, G. D., Leone, L. G., Lawand, N. B., Leone, A., & Barone, R. (2022). Heat Shock Proteins Alterations in Rheumatoid Arthritis. International Journal of Molecular Sciences, 23(5), 2806. https://doi.org/10.3390/ijms23052806







